More than 2 million people have died from COVID-19, caused by SARS-CoV-2.1 In an unprecedented effort to develop vaccines to control the COVID-19 pandemic, mRNA, protein subunit, and viral vector-based vaccines have been developed within an extraordinarily swift timeframe. However, the efficacy of these vaccines (ie, their ability to reduce the incidence of severe disease and death from COVID-19) can vary considerably. For example, among 43 448 adults, the efficacy of the mRNA-based COVID-19 vaccine produced by Pfizer and BioNTech ranged between 29·5% and 68·4% against symptomatic COVID-19 after the first dose, and between 90·3 and 97·6% after the second dose.2 By comparison, in an interim analysis of ongoing clinical trials (involving 23 484 participants), the corresponding efficacy of two standard doses of the ChAdOx1 nCoV-19 adenovirus vector vaccine produced by AstraZeneca ranged between 41·0% and 75·2%.3
Although data from phase 3 trials indicate that factors such as age and biological sex might not be as prominent in modulating the efficacy of certain COVID-19 vaccines (eg, in case of the mRNA-based COVID-19 vaccine produced by Pfizer and BioNTech),2 the role of sleep in this context is unclear. As suggested by previous studies, sleep duration at the time of vaccination against viral infections can affect the immune response (figure ). For instance, 10 days after vaccination against the seasonal influenza virus (1996–97), IgG antibody titres in individuals who were immunised after four consecutive nights of sleep restricted to 4 h were less than half of those measured in individuals without such sleep deficits.4 Similarly, shorter actigraphy-based sleep duration was associated with a lower secondary antibody response to hepatitis B vaccination.5 Sleep might also boost aspects of virus-specific adaptive cellular immunity. Compared to wakefulness, sleep in the night following vaccination against hepatitis A doubled the relative proportion of virus-specific T helper cells, which are known to play a prominent role in host-protective immune responses.6 Interestingly, in individuals who slept the night after the first vaccination, the increase in the fraction of interferon-γ (IFN-γ)-positive immune cells at weeks 0–8 was significantly more pronounced than in those who had stayed awake on that night.6 IFN-γ directly inhibits viral replication and activates immune responses to eliminate viruses, thus protecting the host against virus-induced pathogenesis and lethality.7 Further emphasising the importance of sleep in the fight against viral pandemics, lack of sleep in the night after vaccination against the 2009 H1N1 influenza virus was found to reduce the early-phase production of H1N1-specific antibodies in men but not women.8 Finally, nocturnal sleep has been shown to promote a cytokine milieu supporting adaptive cellular immune responses, such as decreased activity of the anti-inflammatory cytokine interleukin-10 and increased activity of the pro-inflammatory cytokine interleukin-12.9 Although these data suggest that extending sleep duration at the time of vaccination can boost host immune responses, there is no evidence indicating that sleep quality and moderate-to-severe obstructive sleep apnoea are related to antibody responses to vaccination against viruses.5, 10, 11
Figure.
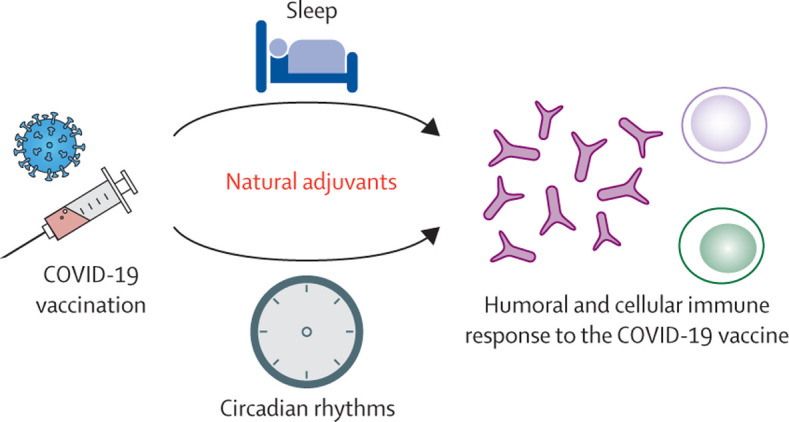
Post-vaccination sleep and morning timing of vaccination as possible immune adjuvants for COVID-19 vaccination
Whether reduced antibody production due to sleep loss can impact vaccine efficacy remains largely undetermined. In one study investigating the impact of acute sleep loss in the night following vaccination against hepatitis A in healthy young adults, a small subsample of individuals failed to reach the clinically significant antibody level at week 20—the threshold for an additional vaccination.6 For most healthy people, sleep loss in the night after vaccination might be of minor concern with respect to the vaccine's efficacy. However, among those whose immune systems' ability to fight infectious diseases is compromised or absent (eg, immunosuppressed individuals), extending sleep duration during the night after the vaccination might help ensure an adequate response to vaccines and potentially contribute to reducing the incidence of severe disease. Furthermore, emerging variants of SARS-CoV-2 might modulate vaccine efficacy against COVID-19. In particular, mutations found in the variant B.1.351 might reduce vaccine-derived neutralisation of SARS-CoV-2 by the mRNA vaccines by about threefold to sixfold.12 In this context, the difference in antibody levels, due to differences in sleep duration in the night after vaccination, might become clinically more significant. Encouragingly, for some individuals, sleep duration might even have increased during the COVID-19 pandemic, possibly as a result of greater work flexibility that enables improved daily activities with individual sleep–wake preferences.13
Since the immune system exhibits marked circadian rhythmicity,14 the timing of vaccination might also affect the immune response to COVID-19 vaccines. For instance, one study found that administering hepatitis A and influenza vaccines in the morning instead of the afternoon results in an almost twofold higher antibody titre 4 weeks later, an effect only seen in men.15 Thus, it is possible that administering COVID-19 vaccines in the morning might result in higher antibody titres. However, several uncertainties remain, such as how to determine the appropriate time of vaccination for night-shift workers. This group often has chronic circadian disruption and exhibits a markedly greater risk of COVID-19 diagnosis.16
Given the urgency of achieving effective global COVID-19 vaccination, we strongly advocate gathering information about individuals' sleep patterns preceding and following vaccination, as well as information about vaccination timing. Combined with data such as baseline serostatus, possible re-infections, work schedules, and comorbidities, monitoring of sleep and the timing of vaccination could provide more conclusive information for public health agencies, health-care providers, patients, and vaccine developers about the importance of these factors for optimising vaccine efficacy.
Acknowledgments
We declare no competing interests.
References
- 1.Wu A, Peng Y, Huang B, et al. Genome composition and divergence of the novel coronavirus (2019-nCoV) originating in China. Cell Host Microbe. 2020;27:325–328. doi: 10.1016/j.chom.2020.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Polack FP, Thomas SJ, Kitchin N, et al. Safety and efficacy of the BNT162b2 mRNA Covid-19 vaccine. N Engl J Med. 2020;383:2603–2615. doi: 10.1056/NEJMoa2034577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Voysey M, Clemens SAC, Madhi SA, et al. Safety and efficacy of the ChAdOx1 nCoV-19 vaccine (AZD1222) against SARS-CoV-2: an interim analysis of four randomised controlled trials in Brazil, South Africa, and the UK. Lancet. 2021;397:99–111. doi: 10.1016/S0140-6736(20)32661-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Spiegel K, Sheridan JF, Van Cauter E. Effect of sleep deprivation on response to immunization. JAMA. 2002;288:1471–1472. doi: 10.1001/jama.288.12.1471-a. [DOI] [PubMed] [Google Scholar]
- 5.Prather AA, Hall M, Fury JM, et al. Sleep and antibody response to hepatitis B vaccination. Sleep. 2012;35:1063–1069. doi: 10.5665/sleep.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lange T, Dimitrov S, Bollinger T, Diekelmann S, Born J. Sleep after vaccination boosts immunological memory. J Immunol. 2011;187:283–290. doi: 10.4049/jimmunol.1100015. [DOI] [PubMed] [Google Scholar]
- 7.Presti RM, Pollock JL, Dal Canto AJ, O'Guin AK, Virgin HW., 4th Interferon gamma regulates acute and latent murine cytomegalovirus infection and chronic disease of the great vessels. J Exp Med. 1998;188:577–588. doi: 10.1084/jem.188.3.577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Benedict C, Brytting M, Markström A, Broman JE, Schiöth HB. Acute sleep deprivation has no lasting effects on the human antibody titer response following a novel influenza A H1N1 virus vaccination. BMC Immunol. 2012;13:1. doi: 10.1186/1471-2172-13-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lange T, Dimitrov S, Fehm HL, Westermann J, Born J. Shift of monocyte function toward cellular immunity during sleep. Arch Intern Med. 2006;166:1695–1700. doi: 10.1001/archinte.166.16.1695. [DOI] [PubMed] [Google Scholar]
- 10.Dopp JM, Wiegert NA, Moran JJ, Muller D, Weber S, Hayney MS. Humoral immune responses to influenza vaccination in patients with obstructive sleep apnea. Pharmacotherapy. 2007;27:1483–1489. doi: 10.1592/phco.27.11.1483. [DOI] [PubMed] [Google Scholar]
- 11.Prather AA, Pressman SD, Miller GE, Cohen S. Temporal links between self-reported sleep and antibody responses to the influenza vaccine. Int J Behav Med. 2021;28:151–158. doi: 10.1007/s12529-020-09879-4. [DOI] [PubMed] [Google Scholar]
- 12.Liu Y, Liu J, Xia H, et al. Neutralizing activity of BNT162b2-elicited serum—preliminary report. N Engl J Med. 2021 doi: 10.1056/nejmc2102017. published online Feb 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Blume C, Schmidt MH, Cajochen C. Effects of the COVID-19 lockdown on human sleep and rest-activity rhythms. Curr Biol. 2020;30:R795–R797. doi: 10.1016/j.cub.2020.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Druzd D, Matveeva O, Ince L, et al. Lymphocyte circadian clocks control lymph node trafficking and adaptive immune responses. Immunity. 2017;46:120–132. doi: 10.1016/j.immuni.2016.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Phillips AC, Gallagher S, Carroll D, Drayson M. Preliminary evidence that morning vaccination is associated with an enhanced antibody response in men. Psychophysiology. 2008;45:663–666. doi: 10.1111/j.1469-8986.2008.00662.x. [DOI] [PubMed] [Google Scholar]
- 16.Rizza S, Coppeta L, Grelli S, et al. High body mass index and night shift work are associated with COVID-19 in health care workers. J Endocrinol Invest. 2020 doi: 10.1007/s40618-020-01397-0. published online Aug 27. [DOI] [PMC free article] [PubMed] [Google Scholar]


