Abstract
Interest in omega-3 fatty acids (colloquially known as fish oils) to lower residual cardiovascular risk in statin-treated patients has increased markedly in the wake of recent cardiovascular outcome trials. The triglyceride-lowering effects of omega-3 fatty acids are generally thought to occur by reduced hepatic VLDL production. In this issue of the JCI, Grevengoed et al. used mouse models and human plasma samples to reveal an additional mechanism whereby these polyunsaturated fatty acids can lower plasma triglycerides. Their findings indicate that omega-3 fatty acid–derived N-acyl taurines (NATs) greatly accumulate in bile and also in plasma following omega-3 supplementation. The authors further show that one of these NATs (C22:6 NAT) inhibited intestinal triglyceride hydrolysis and lipid absorption, which resulted in lower plasma triglycerides and protection against hepatic triacylglycerol accumulation in mice fed a high-fat diet. The findings open a potential avenue for triglyceride lowering by omega-3 fatty acids conjugated to taurine.
A promising strategy for reducing residual cardiovascular disease risk?
LDL cholesterol lowering by statins and nonstatin drugs has markedly reduced atherosclerotic cardiovascular disease (CVD) risk, but a residual risk remains in many patients (1). Elevated triglyceride-rich lipoproteins (TRLs; VLDL and chylomicrons) and their remnant lipoprotein particles (1, 2), systemic inflammation (3), lipoprotein(a) (1), as well as diabetes and associated dyslipidemia (4, 5) are thought to contribute to the residual risk. Recent clinical trials have begun to address the concept that targeting residual risk factors could provide cardioprotection above that conferred by LDL cholesterol lowering alone. For example, the CANTOS trial (NCT01327846) revealed that suppressing systemic inflammation (IL-1β blockade) provided a small protection in individuals at high risk of a second CVD event, approximately 90% of whom were on statin therapy (6).
Research on the omega-3 fatty acids eicosapentaenoic acid (C20:5; EPA) and docosahexaenoic acid (C22:6; DHA) has intensified after the REDUCE-IT (NCT01492361) cardiovascular outcome trial revealed that purified EPA ethyl ester (icosapent ethyl) had protective effects in statin-treated individuals (7). Study participants who had established CVD or diabetes and elevated plasma triglycerides and were treated with a high dose of icosapent ethyl had reduced plasma triglycerides and protection from major cardiovascular events, as compared with participants treated with placebo (mineral oil). These results generated much interest, because omega-3 fatty acids not only reduce plasma triglycerides, but also reduce various inflammatory processes and thrombosis, acting as precursors of proresolving/antiinflammatory lipid mediators (8–10). Omega-3 fatty acids, or at least a –––high dose of EPA (7, 11), appeared to be a promising strategy for reducing residual CVD risk (Figure 1).
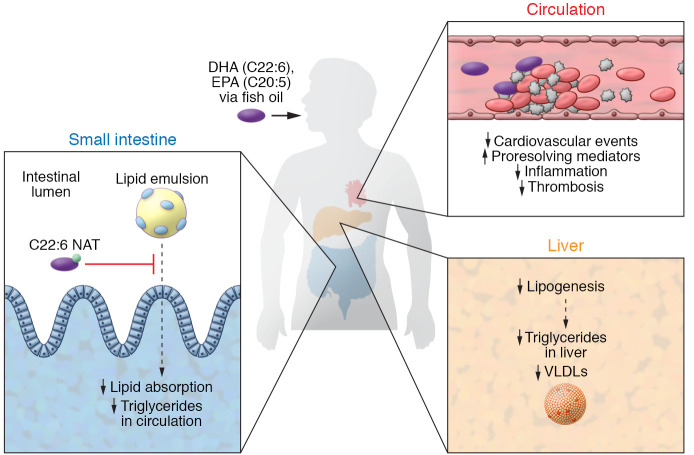
Figure 1. Model for omega-3 fatty acid cardioprotection.
The omega-3 fatty acids EPA (C20:5) and DHA (C22:6) affect processes that are likely involved in protection against CVD, although the cardioprotective effects of EPA and DHA are intensely debated, given the results of recent cardiovascular outcome trials. Individual omega-3 fatty acids may have specific effects; their protective effects are possibly related to reduced plasma triglycerides through reduced hepatic lipogenesis and VLDL production, increased TRL (chylomicron) lipolysis, increased antiinflammatory and proresolving lipid mediators, and reduced thrombosis. Grevengoed et al. (19) showed that biliary C22:6 NAT prevents intestinal triglyceride absorption, providing an additional mechanism for how DHA lowers plasma and liver triglycerides.
However, the cardioprotective effects of omega-3 fatty acids and their mechanisms of action are far from clear. Furthermore, EPA and DHA had distinct effects in some studies (8, 12). Following the REDUCE-IT trial, the recent STRENGTH cardiovascular outcome trial (NCT02104817) investigated the effect of a high-dose carboxylic acid formulation of EPA and DHA in statin-treated patients. This trial was terminated early because of a predicted lack of effect on cardiovascular outcome over that of the corn oil placebo (13). The reasons for these discrepant results are debated, as the triglyceride-lowering effect achieved by the omega-3 fatty acids in REDUCE-IT and STRENGTH was similar. Different placebos in the two trials and the formulations and mixture of EPA and DHA in STRENGTH versus the EPA ester in REDUCE-IT have been brought forward as possible explanations (14, 15). Definite answers to the questions of whether omega-3 fatty acids prevent residual cardiovascular risk and whether the mechanism involves triglyceride lowering are, therefore, lacking.
Mechanisms for omega-3 fatty acid triglyceride lowering
It is thought that omega-3 fatty acids lower plasma triglycerides by increasing fatty acid oxidation, which suppresses hepatic lipogenesis (16, 17), and subsequent VLDL production (Figure 1). Because hepatic triacylglycerol (also known as triglyceride) accumulation is associated with hepatic steatosis, which can lead to steatohepatitis and reduced liver function, an EPA derivative is currently in clinical trial for the prevention of nonalcoholic steatohepatitis (NCT04052516). Other studies have shown that omega-3 fatty acids can increase the clearance of chylomicrons, reducing the half-life of TRLs in circulation (16–18). The exact mechanism for how omega-3 fatty acids reduce plasma triglycerides and liver triglycerides is unclear.
The study by Grevengoed and colleagues, published in this issue of the JCI, provides an additional mechanism for how omega-3 fatty acids can lower plasma and liver triglycerides, through the generation of N-acyl taurines (19). Taurine, an organic compound derived from cysteine, when conjugated to bile acids, aids in the emulsification and absorption of fats. Moreover, N-acyl conjugates of taurine have various biological activities. In a previous study, the authors had cleverly generated a mouse model to study the biological effects of N-acyl taurines (NATs). NATs are degraded (hydrolyzed) by the enzyme fatty acid amide hydrolase (FAAH), but FAAH also degrades N-acylethanolamines (20). The research group, therefore, generated a mouse model harboring an amino acid substitution in FAAH (S268D) that selectively disrupts endogenous NAT hydrolytic activity (21). Characterizing this mouse model, the researchers observed increased plasma levels of DHA-containing NAT (C22:6 NAT), and EPA-containing NAT (C20:5 NAT), as well as other NATs. The most abundant NAT was oleic acid–containing NAT (C18:1 NAT), which they demonstrated improves glucose homeostasis (21).
Impaired intestinal absorption
In the present study (19), C57BL/6 mice were first fed a fish oil diet, which resulted in large increases of polyunsaturated NATs in bile and markedly increased C22:6 NAT in plasma. The increases in plasma C22:6 NAT and C20:5 NAT in response to DHA/EPA supplementation were nicely confirmed in humans. The fish oil diet slowed intestinal lipid absorption but not hepatic VLDL production. Previous studies have shown that omega-3 fatty acids prevent hepatic VLDL production (16), which suggests that the conditions in the Grevengoed et al. study (19) failed to support sufficient VLDL production and that effects of the omega-3 fatty acids might have gone undetected. The researchers were able to mimic the suppressed intestinal lipid absorption achieved by omega-3 fatty acid feeding by administrating C22:6 NAT by oral gavage. Similarly, FAAH-S268D mice exhibited increased polyunsaturated NATs in bile. Because C22:6 NAT prevented the uptake of triolein into intestinal enterocytes, and because it did not directly inhibit pancreatic lipase activity in vitro, but prevented triglyceride hydrolysis in a lipid emulsion, the authors hypothesized that C22:6 NAT impairs triglyceride hydrolysis in the intestine through a mechanism independent of direct pancreatic lipase inactivation (Figure 1). C22:6 NAT reduced the micelle size in bovine bile (in vitro), which the authors suggested might account for a reduced ability of pancreatic lipase to hydrolyze the micelles. In vivo, intestinal triglycerides and micelles are surrounded by phospholipids, cholesterol, and bile salts (22), and C22:6 NAT might act quite differently. It is, for example, possible that C22:6 NAT alters the pancreatic lipase-colipase complex, other lipases, the inhibition of enzymatic activity by bile salts, the structure of micelles, bile acid reabsorption, and subsequent activation of hepatic farnesoid X receptors, which reduces triglycerides (23), or interactions with microbiota. In future studies, it will be interesting to more thoroughly elucidate the mechanism whereby C22:6 NAT reduces triglycerides.
To further support the conclusion that polyunsaturated NATs impair intestinal lipid absorption, FAAH-S268D mice were fed a fish oil diet to show that levels of both plasma triglycerides and liver triacylglycerol were (rather modestly) reduced, without a difference in VLDL production. Moreover, subcutaneous injection of C22:6 NAT for two weeks lowered plasma triglycerides and liver triglycerides, as well as glucose levels, without decreasing weight gain. It is possible that subcutaneous injection of C22:6 NAT could exert systemic effects beyond those caused by biliary C22:6 NAT. Although concentrations of plasma omega-3 NATs are lower than those in bile, it would be interesting to investigate whether they alter lipoprotein particle size, metabolism, and lipolysis, as observed in the bile. Of note, Grevengoed et al. used C57BL/6 mice that carry most of their cholesterol in HDL rather than LDL, which therefore limits the interpretation of the TRL metabolism data. Furthermore, C22:6 NAT was shown to indirectly increase plasma GLP-1 levels after a high-fat meal (19). To what extent the known triglyceride-lowering effects of GLP-1 (24) might have contributed to the effects of C22:6 NAT needs further consideration.
Finally, as an elegant proof-of-concept experiment to verify that the effect of C22:6 NAT on liver triglycerides was not explained by reduced hepatic de novo lipogenesis, FAAH-S268D mice were fed a high-sucrose diet to drive de novo lipogenesis from a carbohydrate source. These mice maintained constant hepatic triglyceride levels when injected with C22:6 NAT, supporting the proposal that C22:6 NAT prevents hepatic lipid accumulation by inhibiting intestinal lipid absorption.
Future research on NATs and fish oils
Together, these results convincingly demonstrate that elevated C22:6 NAT can prevent lipid absorption. However, the role of physiologically elevated C22:6 NAT levels cannot be assessed as of yet, because the enzyme or enzymes responsible for its synthesis remain unidentified. Thus, preventing C22:6 NAT degradation, or providing C22:6 NAT through gavage or injection, does not necessarily mimic the effects of physiologically regulated NAT levels.
Grevengoed and colleagues raise several other interesting questions. Omega-3 fatty acids exert antiinflammatory effects as precursors of the proresolving lipid mediators resolvins, maresins, and protectins (9). The protective effects of DHA on hepatic lipid accumulation have been described to be mediated by maresin 1 (25). It would be interesting to determine whether omega-3 NATs affect the proresolving lipid mediators. Furthermore, EPA may have physical and biological properties distinct from those of DHA (8). Different NATs also have different biological effects (19, 21). Given the positive result of REDUCE-IT, an exploration of the biological effects of C20:5 NAT is worthwhile. Mouse models can also be used to investigate whether C20:5 NAT or C22:6 NAT protects against atherosclerosis. Finally, and most important, understanding the biological effects of omega-3 NATs in humans is a crucial area of future investigation.
In summary, the findings of Grevengoed et al. (19) further reveal the emerging picture of how omega-3 fatty acids might exert protective effects in dyslipidemia, hepatic steatosis, and CVD.
Acknowledgments
Work in KEB’s laboratory is supported by grants from the NIH (P01HL092969, P01HL151328, R01HL149685, and R35HL150754).
Version 1. 03/15/2021
Electronic publication
Footnotes
Conflict of interest: The author has declared that no conflict of interest exists.
Copyright: © 2021, American Society for Clinical Investigation.
Reference information: J Clin Invest. 2021;131(6):e147558. https://doi.org/10.1172/JCI147558.
References
- 1.Hoogeveen RC, Ballantyne CM. Residual cardiovascular risk at low LDL: remnants, lipoprotein(a), and inflammation. Clin Chem. 2021;67(1):143–153. doi: 10.1093/clinchem/hvaa252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Lawler PR, et al. Residual risk of atherosclerotic cardiovascular events in relation to reductions in very-low-density lipoproteins. J Am Heart Assoc. 2017;6(12):e007402. doi: 10.1161/JAHA.117.007402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ridker PM, et al. Comparison of interleukin-6, C-reactive protein, and low-density lipoprotein cholesterol as biomarkers of residual risk in contemporary practice: secondary analyses from the Cardiovascular Inflammation Reduction Trial. Eur Heart J. 2020;41(31):2952–2961. doi: 10.1093/eurheartj/ehaa160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stitziel NO, et al. Emerging targets for cardiovascular disease prevention in diabetes. Trends Mol Med. 2020;26(8):744–757. doi: 10.1016/j.molmed.2020.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Xiao C, et al. Pharmacological targeting of the atherogenic dyslipidemia complex: the next frontier in CVD prevention beyond lowering LDL cholesterol. Diabetes. 2016;65(7):1767–1778. doi: 10.2337/db16-0046. [DOI] [PubMed] [Google Scholar]
- 6.Ridker PM, et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N Engl J Med. 2017;377(12):1119–1131. doi: 10.1056/NEJMoa1707914. [DOI] [PubMed] [Google Scholar]
- 7.Bhatt DL, et al. Cardiovascular risk reduction with icosapent ethyl for hypertriglyceridemia. N Engl J Med. 2019;380(1):11–22. doi: 10.1056/NEJMoa1812792. [DOI] [PubMed] [Google Scholar]
- 8.Mason RP, et al. Emerging mechanisms of cardiovascular protection for the omega-3 fatty acid eicosapentaenoic acid. Arterioscler Thromb Vasc Biol. 2020;40(5):1135–1147. doi: 10.1161/ATVBAHA.119.313286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Serhan CN, Levy BD. Resolvins in inflammation: emergence of the pro-resolving superfamily of mediators. J Clin Invest. 2018;128(7):2657–2669. doi: 10.1172/JCI97943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Conte MS, et al. Pro-resolving lipid mediators in vascular disease. J Clin Invest. 2018;128(9):3727–3735. doi: 10.1172/JCI97947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yokoyama M, et al. Effects of eicosapentaenoic acid on major coronary events in hypercholesterolaemic patients (JELIS): a randomised open-label, blinded endpoint analysis. Lancet. 2007;369(9567):1090–1098. doi: 10.1016/S0140-6736(07)60527-3. [DOI] [PubMed] [Google Scholar]
- 12.Suzuki-Kemuriyama N, et al. Different effects of eicosapentaenoic and docosahexaenoic acids on atherogenic high-fat diet-induced non-alcoholic fatty liver disease in mice. PLoS One. 2016;11(6):0157580. doi: 10.1371/journal.pone.0157580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nicholls SJ, et al. Effect of high-dose omega-3 fatty acids vs corn oil on major adverse cardiovascular events in patients at high cardiovascular risk: the STRENGTH randomized clinical trial. JAMA. 2020;324(22):2268–2280. doi: 10.1001/jama.2020.22258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sharma G, et al. Effects of omega-3 fatty acids on major adverse cardiovascular events: what matters most: the drug, the dose, or the placebo? JAMA. 2020;324(22):2262–2264. doi: 10.1001/jama.2020.22387. [DOI] [PubMed] [Google Scholar]
- 15.Curfman G. Do omega-3 fatty acids benefit health? JAMA. 2020;324(22):2280–2281. doi: 10.1001/jama.2020.22898. [DOI] [PubMed] [Google Scholar]
- 16.Shearer GC, et al. Fish oil -- how does it reduce plasma triglycerides? Biochim Biophys Acta. 2012;1821(5):843–851. doi: 10.1016/j.bbalip.2011.10.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Oscarsson J, Hurt-Camejo E. Omega-3 fatty acids eicosapentaenoic acid and docosahexaenoic acid and their mechanisms of action on apolipoprotein B-containing lipoproteins in humans: a review. Lipids Health Dis. 2017;16(1):149. doi: 10.1186/s12944-017-0541-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Park Y, Harris WS. Omega-3 fatty acid supplementation accelerates chylomicron triglyceride clearance. J Lipid Res. 2003;44(3):455–463. doi: 10.1194/jlr.M200282-JLR200. [DOI] [PubMed] [Google Scholar]
- 19.Grevengoed TJ, et al. An abundant biliary fatty acid metabolite derived from dietary omega-3 polyunsaturated fatty acids regulates triglycerides. J Clin Invest. 2021;131(7):e143861. doi: 10.1172/JCI143861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Long JZ, et al. An anatomical and temporal portrait of physiological substrates for fatty acid amide hydrolase. J Lipid Res. 2011;52(2):337–344. doi: 10.1194/jlr.M012153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Grevengoed TJ, et al. N-acyl taurines are endogenous lipid messengers that improve glucose homeostasis. Proc Natl Acad Sci U S A. 2019;116(49):24770–24778. doi: 10.1073/pnas.1916288116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Pignol D, et al. Critical role of micelles in pancreatic lipase activation revealed by small angle neutron scattering. J Biol Chem. 2000;275(6):4220–4224. doi: 10.1074/jbc.275.6.4220. [DOI] [PubMed] [Google Scholar]
- 23.Lefebvre P, et al. Role of bile acids and bile acid receptors in metabolic regulation. Physiol Rev. 2009;89(1):147–191. doi: 10.1152/physrev.00010.2008. [DOI] [PubMed] [Google Scholar]
- 24.Mulvihill EE. Regulation of intestinal lipid and lipoprotein metabolism by the proglucagon-derived peptides glucagon like peptide 1 and glucagon like peptide 2. Curr Opin Lipidol. 2018;29(2):95–103. doi: 10.1097/MOL.0000000000000495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Han YH, et al. A maresin 1/RORα/12-lipoxygenase autoregulatory circuit prevents inflammation and progression of nonalcoholic steatohepatitis. J Clin Invest. 2019;129(4):1684–1698. doi: 10.1172/JCI124219. [DOI] [PMC free article] [PubMed] [Google Scholar]