Graphical abstract
Keywords: Dipeptidyl peptidase, DPP4 inhibitor, Sitagliptin, Immunity, Innate immunity, Neutrophil, Macrophage
Abstract
Dipeptidyl peptidase (DPP)-4 inhibitors are a class of orally available, small molecule inhibitors that prolong the insulinotropic activity of the incretin hormone glucagon-like peptide-1 (GLP-1) and are highly effective for the treatment of Type-2 diabetes. DPP4 can also cleave several immunoregulatory peptides including chemokines. Emerging evidence continues to implicate DPP4 inhibitors as immunomodulators, with recent findings suggesting DPP4 inhibitors modify specific aspects of innate immunity. This review summarises recent insights into how DPP4 inhibitors could be implicated in endothelial, neutrophil and monocyte/macrophage mediated immunity. Additionally, this review highlights additional avenues of research with DPP4 inhibitors in the context of the COVID-19 pandemic.
1. Introduction
Dipeptidyl peptidase (DPP)-4 is a transmembrane serine protease that has emerged as a potent pharmacological target for the treatment of Type-2 diabetes. DPP4 is part of the S9B sub-family of serine proteases that also includes the structurally homologous: fibroblast activation protein (FAP), DPP8 and DPP9. These proteases all have unique enzyme activity, cleaving N-terminal dipeptides from regulatory peptides with a proline or alanine residue in the penultimate position (i.e. X-Pro or X-Ala) [1], [2]. DPP4 protein expression and enzyme activity has been localised to the apical surface of endothelial, epithelial cells and activated lymphocytes, with abundant expression in the lungs, kidney, liver, and small intestine [3]. An enzymatically active, soluble form of DPP4 has also been identified and is thought to result from the enzymatic shedding of membrane bound DPP4 from various cell types, including adipocytes, smooth muscle cells, and specialised T-lymphocytes [4], [5].
Sitagliptin (Januvia, Merck) was the first, highly selective dipeptidyl peptidase (DPP)-4 inhibitor to gain FDA approval as an orally available, small molecule inhibitor with clinical indication for type-2 diabetes [6]. This class of inhibitors acts by blocking DPP4 mediated cleavage of the incretin hormone, glucagon-like peptide-1 (GLP-1) and GIP, prolonging its insulinotropic activity [2], [7]. Since then, many DPP4 inhibitors, broadly classed as the ‘gliptins’ and each with discrete structural differences and binding mechanisms, have entered the pharmaceutical market (Table 1 ). Whilst the clinical efficacy of DPP4 inhibitors have been reported in several large scale clinical studies, and meta-analyses [8], [9], emerging evidence continues to suggest complex immuno-regulatory roles for DPPs with potential implications for long-term use (i.e. daily ingestion for more than 3 months) of DPP4 inhibitors [10].
Table 1.
Selective DPP4 inhibitors approved for the clinical management of type-2 diabetes.
| Inhibitor | Company | Approving body and Year | Chemical Structures |
IC50 (nmol/L) |
Reference | ||
|---|---|---|---|---|---|---|---|
| DPP4 | DPP8 | DPP9 | |||||
| Sitagliptin (Januvia) | Merck & Company | FDA/US 2006 |  |
18 | 4.8x104 | >1.x105 | [20] |
| Vildagliptin (Galvus) | Novartis | EU 2007 |  |
100 | 9x103 | – | [21] |
| Saxagliptin (Onglyza) | Bristol-Myers Squibb & Astra Zeneca | FDA/US 2009 |  |
4 | – | – | [22] |
| Linagliptin (Tradjenta) | Eli Lilly & Company and Boehringer Ingelheim | FDA/US 2011 |  |
1 | 4x104 | >1x104 | [23] |
| Gemigliptin | LG Life Sciences | Korea 2012 |  |
10 | 2.7x105 | 2.3x104 | [24] |
| Anagliptin (Suiny) | Sanwa Kagaku | Japan 2012 |  |
3.8–5.3 | 68 | 60 | [25] |
| Teneligliptin (Tenelia) | Mitsubishi Tanabe Pharma | Japan 2012 |  |
0.37 | 260 | 540 | [26] |
| Alogliptin (Nesina) | Takeda | FDA/US 2013 |  |
7 | >1x105 | >1x105 | [27] |
| Imigliptin | Xuanzhu Pharma Co Ltd | China 2014 |  |
9 | >1x105 | >1x105 | [28] |
| Trelagliptin | Japan 2015 |  |
1.3 | – | – | [29] | |
| Omarigliptin (MK-3102) | Merck & Co | Japan 2015 |  |
1.6 | >6.7x104 | >6.7x104 | [30] |
| Evogliptin(Suganon) | Korea 2015 |  |
0.9 | – | – | [31] | |
Chemical structures adapted from National Centre for Biotechnology Information (NCBI). PubChem Compound Database
Innate and adaptive immunity are no longer considered distinct entities, with fluidity between the two responses maintaining a state of immune homeostasis [11]. The role of DPP4 in adaptive immune responses has been previously described [12]; however, an understanding of the importance of DPP4 in innate immunity is emerging. Ohnuma et al has described links between DPP4, the innate and adaptive immune system, reporting that interactions between DPP4 on T-cells and caveolin-1 on antigen presenting cells lead to costimulatory signals that result in the proliferation of antigen specific T cells [13]. Recent, large cohort studies have also suggested that DPP4 inhibitor use could be associated with an increased risk for pancreatitis in a small sub-group of patients [14], [15].
Some of the earliest published studies used DPP4 inhibitors to investigate whether DPP4 enzyme activity modulated T-cell activity and function [16], [17]. Inhibition of DPP4 in mitogen stimulated T-cells from human blood suppressed IL-2 and IFN-γ production, while also reducing T-cell proliferation [17], with similar findings also reported in antigen stimulated T-cell lines [16]. These initial findings suggested that the DPP4 inhibitors were blocking the enzymatic activity of DPP4 against immunoregulatory substrates such as chemokines [18]. However, early iterations of DPP4 inhibitors were non-selective, and off-target effects via binding of DPP8 or DPP9 could not be excluded [19].
We have previously reported on DPP4 inhibitors as potential immunomodulators in inflammatory disease [2]. The global proliferation of DPP4 inhibitors, and recent insights into their possible effects on immune activity, necessitates a comprehensive update on the role DPP4 inhibitors may have in regulating innate immunity, and how they could modify disease risk. This review summarises recent insights into how highly selective DPP4 inhibitors could modulate the cellular response in innate immunity, including epithelial and endothelial cell, neutrophil and macrophage driven immunity.
2. DPP4 inhibitors: Clinical safety
The safety and efficacy of the DPP4 inhibitors has been reported in several meta-analyses. A systematic review of 29 studies published between 2004 and 2007 by Amori et al found that Sitagliptin and Vildagliptin were highly effective for blood sugar management and were more likely to reduce glycosylated haemoglobin compared to placebo [8]. The risk of adverse reactions, including infections and gastrointestinal complications such as nausea, diarrhoea and vomiting, was relatively low overall in patients receiving DPP4 inhibitor therapy [8]. Urinary tract infections were the most common adverse event for all DPP4 inhibitors compared with non-incretin based hypoglycaemic agents [8]. Subsequent studies by Williams-Herman et al and Karagiannis et al have reported no significant differences in the rates of infections, including upper respiratory tract and urinary tract infections, between patients treated with DPP4 inhibitors compared to placebo [9], [32] or conventional diabetic therapies [9].
Although rare, acute pancreatitis is the most reported serious adverse event associated with DPP4 inhibitor use. A comparison of adverse event reports lodged to the FDA over a 9 year period found that Sitagliptin use increased the odds ratio for pancreatitis 6-fold compared to other therapies [33]. In contrast, a large retrospective study that analysed pharmaceutical claims data from 786,656 patients found that the incidence of pancreatitis was unchanged in patients receiving Sitagliptin compared to other type-2 diabetes medications [34]. Recent meta-analyses of the DPP4 inhibitors Sitagliptin, Alogliptin, Linagliptin, Saxagliptin and Vildagliptin found a marginally higher risk of developing acute pancreatitis with DPP4 inhibitor use [14], [15]. The Cardiovascular and Renal Microvascular Outcome Study With Linagliptin in Patients With Type 2 Diabetes Mellitus (CARMELINA) trial also reported a small but increased risk of pancreatitis in patients taking Linagliptin [35]. However, the Cardiovascular Outcome Study of Linagliptin vs Glimepiride in Type 2 Diabetes (CAROLINA) study found no increased risk of pancreatitis in the Linagliptin group [36]. Acute pancreatitis is characterised by a TNF-α, IL-1, IL-6 and IL-8 pro-inflammatory immune response [37], with neutrophils and macrophages driving this response [38]. Whilst the exact mechanisms associated with DPP4 inhibitor use and pancreatitis are still unknown, their effects on innate immunity could reveal novel insights into their pharmacological actions.
3. Dipeptidyl peptidases and innate immunity
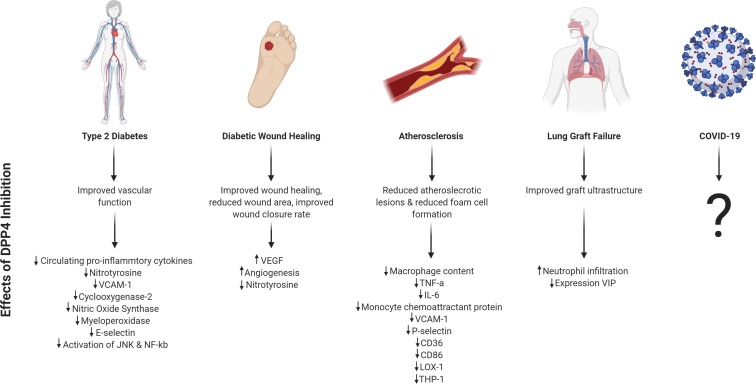
Emerging evidence suggests that DPP4 inhibitors could modulate innate immune responses, with potential clinical implications. Whilst the role of DPP4 in adaptive immunity has been previously described, it is increasingly apparent that DPP4 inhibitors can modify specific components of innate immunity, including vascular endothelial cell, neutrophil and monocyte/macrophage mediated responses (Fig. 1 ). The diverse range of DPP4 substrates, with overlapping roles in innate immunity, suggests multiple pathways that could be impacted by DPP4 inhibition (Table 2 ).
Fig. 1.
Summary of main health conditions discussed in this review and the described physiological and molecular effects of DPP4 inhibition. ↓ indicates decreased expression and ↑ indicates increased expression. Image created with BioRender.com.
Table 2.
Candidate DPP4 substrates with identified roles in innate immune responses.
| Substrate | N-terminal sequence | Reported Roles in Innate Immunity | Biological Effect of DPP4 cleavage |
|---|---|---|---|
| Eotaxin (CCL11) | Gly-Pro-Ala | Eosinophil and basophil migration | Reduced chemotactic activity |
| Erythropoietin | Ala-Pro-Pro | Inhibits production of macrophage derived inflammatory cytokines; enhances macrophage phagocytic activity | Inactivation |
| Gastrin-releasing peptide (GRP) | Val-Pro-Leu | Induces neutrophil migration | Inactivation |
| Glucagon like peptide 1 (GLP-1) | His-Ala-Glu | Induces macrophage polarization | Inactivation |
| Granulocyte-Colony Stimulating Factor(G-CSF) | Glu-Ala-Thr | Stimulates neutrophil proliferation & differentiation | Inactivation |
| Granulocyte Macrophage-Colony-Stimulating Factor (GM-CSF) | Ala-Pro-Ala | Recruitment of neutrophils & monocytes; stimulates macrophage activity | Inactivation |
| HMGB1 | Gly-Lys-Gly | Stimulates expression of cell adhesion molecules; upregulates pro-inflammatory cytokine production | Inactivation |
| IP10 (CXCL10) | Val-Pro-Leu | Chemoattractant for monocytes/macrophages & NK cells; stimulates NK cell degranulation | Reduced chemotactic potential |
| ITAC (CXCL11) | Phe-Pro-Ala | Chemoattractant for monocytes/macrophages & NK cells | Reduced chemotactic potential |
| IL-3 | Ala-Pro-Met | Monocyte activation & differentiation | Inactivation |
| Macrophage-derived chemokine (MDC, CCL22) | Gly-Pro-Tyr | Recruitment of dendritic cells, NK & Th2 cells | Reduced chemotactic potential |
| Monokine induced by IFN-γ (Mig, CXCL9) | Tyr-Pro-Val | Recruitment of T-cells, | Reduced chemotactic potential |
| Neuropeptide-Y (NPY) | Tyr-Pro-Ser | Stimulates secretion of pro-inflammatory cytokines by macrophages; stimulates phagocytic activity | Altered receptor specificity |
| RANTES (CCL5) | Ser-Pro-Tyr | Recruitment of macrophages and NK cells | Altered receptor specificity |
| Stromal-cell derived factor (SDF-1, CXCL12) | Lys-Pro-Val | Monocyte chemoattractant | Reduced chemotactic potential |
| Pituitary adenylate-cyclase-activating polypeptide (PACAP) | His-Ser-Asp | Inhibit macrophage chemotaxis, phagocytosis & ROS production; inhibits pro-inflammatory cytokine production by macrophages | Inactivation |
| Substance P | Arg-Pro-Lys | Stimulate neutrophil & macrophage phagocytic activity; stimulates pro-inflammatory cytokine production in mast cells; enhances NK cell cytotoxicity | Inactivation |
| Vasoactive Intestinal Peptide (VIP) | His-Ser-Asp | Inhibit macrophage chemotaxis, phagocytosis & ROS production; inhibits pro-inflammatory cytokine production by macrophages | Inactivation |
3.1. Vascular endothelium
Endothelial cells lining the vascular system are vital for maintaining blood flow and vessel tonicity and have important roles in regulating the innate immune response [39]. When activated, endothelial cells secrete an array of cytokines, chemokines, and the cellular adhesion molecules, intracellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1) [40], required for the recruitment, activation, binding and tethering of polymorphonuclear leukocytes, including monocytes and neutrophils. Endothelial dysfunction is defined as a chronic and continual endothelial activation [41] and has been implicated in the pathogenesis of cardiovascular disease and other inflammatory disorders, such as inflammatory bowel disease and rheumatoid arthritis [41].
DPP4 inhibitors have been associated with significant improvements to vascular endothelial function in type-2 diabetes, independent of their anti-hyperglycaemic action. In patients with type-2 diabetes and existing coronary artery disease, endothelial function was significantly improved after six months of treatment with Sitagliptin (50 mg/day) compared to conventional therapy [42]. Rizzo et al found a reduction in circulating pro-inflammatory cytokines and the oxidative stress marker, nitrotyrosine in a prospective, randomized open label study of Type-2 diabetics taking either Sitagliptin or Vildagliptin [43]. The authors reported superior effects with Vildagliptin, suggesting glycaemic control and inhibitor kinetics were responsible for the immunomodulatory effects. However, the authors did not exclude the possibility of a non-glycaemic effect of DPP4 inhibitors in regulating endothelial cell mediated innate immunity [43].
Glucose independent effects of DPP4 inhibitors have been described in pre-clinical studies as modifying endothelial mediated immunity. Kroller-Schon et al found that vascular function was maintained in linagliptin treated rats with lipopolysaccharide (LPS)-induced septic shock compared to control groups [44]. The authors also reported that Linagliptin treatment attenuated vascular oxidative stress markers. Linagliptin also reduced expression of the inflammatory markers vascular cell adhesion molecule (VCAM)-1, cyclooxygenase-2, nitric oxide synthase and myeloperoxidase [44]. Using the DPP4 inhibitor, Gemigliptin, Hwang et al also demonstrated decreased expression of VCAM-1 and the neutrophil binding protein, E-selectin, in LPS stimulated human umbilical vein endothelial cells (HUVEC) cells in vitro [45]. Gemigliptin also blocked LPS-mediated activation of pro-inflammatory transcription factors JNK and NF-κβvia an Akt/AMP-activated protein kinase dependent pathway in vitro [45]. Monocyte (THP-1 cell) adhesion to LPS stimulated HUVECS was also reduced, as was pro-inflammatory cytokine expression by HUVECs in vitro. It is likely that the effects of DPP4 inhibitors in modifying endothelial cell mediated innate immunity are via a combination of glucose dependent and independent mechanisms. Additional studies are required to determine not only the molecular pathways involved, but also how different DPP4 inhibitors exhibit different modes of action.
3.2. Neutrophils
Neutrophils are highly specialized anti-microbial immune cells and are the first responders to invading pathogens [46]. Neutrophils employ an array of cellular and non-cellular mechanisms to destroy invading pathogens that include phagocytosis and the secretion of potent antimicrobials, proteases, and oxidative species [47], [48]. Additionally, neutrophils have a key role in driving the secondary immune response, recruiting and activating macrophages, dendritic cells and lymphocytes, to the site of inflammation via secretion of various cytokines and peroxidases [49]. Pre-clinical and in vitro studies have suggested that DPP4 inhibitors can modulate specific aspects of neutrophil recruitment and activity.
Normal wound healing is characterised by an integrated cascade of events, with neutrophil recruitment and activation a key component to prevent infection by pathogenic organisms [50]. Patients with diabetes have impaired leucocyte function, which can lead to impaired neutrophil migration to the wound [51]. However, in chronic, non-healing wounds, such as those seen in diabetics, persistence of neutrophils at the wound site can exacerbate tissue damage via release of reactive oxygen species, anti-microbial peptides and proteases [52]. In a prospective, randomised open label trial, Marfella et al investigated the effects of Vildagliptin on wound healing in diabetic chronic ulcers. Treatment with Vildagliptin for three months significantly reduced overall wound area, wound related complications and improved wound closure rates, compared to patients on conventional therapies [53]. The authors reported increased expression of vascular endothelial growth factor and increased angiogenesis in the Vildagliptin group [53]. Although leukocyte infiltration was not specifically quantified, a decrease in nitrotyrosine, which is associated with activated neutrophils, was reported in Vildagliptin treated patients. In a murine model of diabetic wound healing, Schurmann et al reported reduced numbers of infiltrating neutrophils to the wound in mice treated with Linagliptin [54], with the authors suggesting an indirect effect of Linagliptin on neutrophil recruitment via COX-2 and MIP-2 [54]. This pre-clinical data supports the observations seen in humans, and suggests DPP4 inhibition may be protective via a possible COX-2 and MIP-2 mediated mechanism.
The effects of a panel of five DPP4 inhibitors on neutrophil functions has been investigated in human neutrophils in vitro. Linagliptin dose-dependently (1 μM to 100 μM) reduced ROS production in LPS-stimulated neutrophils; however, the DPP4 inhibitors, Sitagliptin, Vildagliptin, Alogliptin and Saxagliptin, had little effect on in vitro ROS production from human neutrophils [44]. Linagliptin also reduced the adhesion of LPS-activated human neutrophils to an endothelial monolayer in vitro [44]. Unlike other DPP4 inhibitors, Linagliptin is a xanthine analogue and Kroller-Schon and colleagues have suggested potential off-target effects as a result of interactions with xanthine oxidase, an enzyme involved in the production of ROS and nitrogen species [55]; however, activity from xanthine oxidase was not specifically measured.
DPP4 inhibition has also been found to modify aspects of neutrophil activity in a mouse model of ischemia–reperfusion injury induced lung graft failure [56]. Treatment of lung-allografts with the irreversible DPP4 inhibitor, AB192 (bis(4-acetamidophenyl) 1-(s)-prolylpyrrolidine-2(r,s)-phosphonate), prior to surgical transplant improved graft ultrastructure and reduced graft neutrophil infiltration, as determined by markers Ly-6-G and myeloperoxidase activity, compared to untreated controls [56]. The authors also reported that DPP4 inhibitor treatment was associated with increased expression of vasoactive intestinal peptide (VIP) in lung grafts. VIP has been previously been shown to suppress neutrophil infiltration, suggesting a possible indirect mechanism of DPP4 inhibition on neutrophil activity. Our group has previously reported that the DPP4 inhibitors, Ile-Pyrr-(2-CN)*TFA and Ile-Thia were associated with lowered colonic myeloperoxidase activity in mice with dextran sulfate sodium induced colitis [57]. Whilst the current evidence suggests that DPP4 inhibitors can modify neutrophil responses in experimental models of inflammation, the specific mechanisms and clinical relevance to humans are yet to be elucidated.
Enzymatically active DPP4 may also act as a neutrophil chemo-repellent. Using an in vitro chemotaxis model, unstimulated and TNF-α stimulated human derived peripheral blood neutrophils were found to migrate away from recombinant DPP4 in a dose dependant manner [58]. When the non-selective DPP4 inhibitors Diprotin A and DPPI 1c hydrochloride were added in combination with the recombinant DPP4 gradient, the chemo-repulsive activity of DPP4 was significantly attenuated suggesting that the ability of DPP4 to act as a chemo-repellent is dependent on its enzymatic activity [58]. The authors also demonstrated that treating mice with aerosolized recombinant DPP4 reduced neutrophil infiltration to the lungs induced by bleomycin, although no changes to other inflammatory cell types were observed [58]. In a separate study of collagen-induced-arthritis, injection of recombinant DPP4 to the joint reduced neutrophil and macrophage infiltration, improving joint inflammation and disease severity [59]; however, DPP4 inhibitors were not used in this study. Whilst these findings could have implications for the long-term use of DPP4 inhibitors on neutrophil recruitment, the absence of data using more highly selective, clinically relevant DPP4 inhibitors mean this effect cannot be clearly delineated.
The collective evidence suggests that DPP4 inhibitors may modify neutrophil recruitment and activity; however, the specific mechanisms involved are still unclear. It is likely that DPP4 inhibitors have both direct and indirect effects on neutrophil functions during specific inflammatory conditions. Additional studies should seek to investigate whether long-term DPP4 inhibitor therapy affects innate immune responses in patients that have become septic or are exposed to infectious pathogens. Furthermore, the precise mechanisms of DPP4 inhibitors in modifying neutrophil functions should be deciphered to determine any possible therapeutic application.
3.3. Monocytes and macrophages
DPP4 activity and expression has been characterised in monocytes, macrophages [60] and in subpopulations of dendritic cells [61], [62], [63]. Atherosclerosis is a complex, inflammatory disorder in which macrophages are directly involved in disease pathogenesis [64]. In diabetic apolipoprotein E deficient (ApoE-/-) mouse models of atherosclerosis, treatment with either Sitagliptin, Anagliptin or a Vildagliptin analogue has been associated with fewer atherosclerotic lesions compared with respective controls [65], [66], [67]. Macrophage content and infiltration into the plaques was also reduced in animals treated with a DPP4 inhibitor [67], [68]. Reduced expression of the pro-inflammatory cytokines TNF-α [65], IL-6 and monocyte chemoattractant protein 1, as well as serum adhesion molecules, VCAM-1 and P-selectin [66], have been identified in the vascular tissue, suggesting that DPP4 inhibitors may cause changes to circulating and locally expressed inflammatory mediators that could modify monocyte and macrophage recruitment and infiltration. Dai et al reported that Sitagliptin in vitro prevented the formation of foam cells, a key component of atherosclerotic plaques, when exposed to oxidized low-density lipoprotein [69]. Sitagliptin also reduced the expression of scavenger receptors including CD36 and LOX-1 in THP-1 cells and CD68 in human monocytes, which are involved in the internalization of oxidized low-density lipoprotein [69].
Shah et al determined the effects of Alogliptin on adipose tissue inflammation in mice deficient for the low-density lipoprotein receptor (LDLR-/-) and fed a high fat diet to induce obesity [70]. Alogliptin treatment for 12 weeks led to reduced number of macrophages within adipose tissue, as detected by immunohistochemistry [70]. Alogliptin treatment was associated with reduced numbers of pro-inflammatory M1 macrophages (CD11b+Ly6Chigh) in adipose tissue, and increased gene expression of M2 macrophage markers, CD163 and Ym-1 [70]. The authors also reported that Alogliptin treatment was associated with reduced gene expression of monocyte derived pro-inflammatory cytokines. Using the monocytic cell line, THP-1, monocyte migration was also found to be reduced in vitro following treatment with either Alogliptin or Sitagliptin. Follow-up studies by Shah et al in an ApoE-/- mouse model of atherosclerosis found monocyte migration to the aortic plaque was reduced in vivo by Sitagliptin [70].
A role for soluble DPP4 protein in stimulating inflammatory responses in macrophages has also been proposed. Ikeda et al found that the addition of enzymatically active, soluble DPP4, dose dependently increased the gene and protein expression of TNF-α in lipopolysaccharide stimulated THP-1 cells and freshly isolated monocytes [71]. The authors also reported that soluble DPP4 mediated increased production of signal transduction proteins that include NF-κB, ERK and c-FOS [71]. Interestingly, treatment with Sitagliptin (1 μM) ameliorated these effects, suggesting that the observed effects were dependent on DPP4 enzyme activity [71]. Lee et al also reported that addition of exogenous recombinant DPP4 to a lipopolysaccharide stimulated macrophage cell line, RAW264.7, in vitro enhanced the expression of TLR4, TLR2, iNOS and cytokines, IL-6, IL-1β and TNF-α [72]. The authors found that Vildagliptin (0.01 μM–10 μM) treatment reduced iNOS expression, JNK phosphorylation and NF- κB activation, as well as suppressing the increase in production of the cytokines TNF-α, IL-6, and IL-12 p40 [72].
DPP4 inhibitor selectivity has been an important consideration in gliptin drug design to mitigate off-target effects from other DPP family members. Matheeussen et al reported high DPP8/9 immunostaining of DPP8 and DPP9 in the macrophage rich regions of human atherosclerotic plaques [60]. DPP8/9 enzyme activity was also high in the cell lysate of the human monocyte cell line, U937 and in monocyte differentiated macrophages [60]. The authors also reported increased DPP8/9 activity in vitro in PMA stimulated U937 cells and monocyte differentiated macrophages. Treatment of lipopolysaccharide or IFN-γ stimulated M1 macrophages with the DPP8/9 inhibitor, 1G244, reduced production of pro-inflammatory cytokines, IL-6, IL-1β and TNF-α [60]. DPP9 silencing using siRNA suggested that the immuno-regulatory effects could be attributable to DPP9 [60]. Macrophage activation was also investigated by Waumans et al in murine bone marrow derived macrophages. Treatment with a DPP8/9 inhibitor reduced TNF-α and IL-6 secretion from macrophages, indicating reduced cell activation [73]. This suggests that at least some effects of DPP inhibition could be due to DPP9.
Although not yet clearly defined, DPP4 and DPP8/9 appear to have specific roles in regulating monocyte and macrophage function and activity. Further studies are necessary to better understand the specific mechanisms of DPPs and whether their roles are confined to specific sub-populations of monocytes and macrophages. Understanding the clinical relevance of DPP4 inhibition in monocyte-macrophage functions may also support the repurposing of FDA approved DPP4 inhibitors as pharmacological targets for the management of chronic inflammatory disorders like atherosclerosis.
4. DPP4 inhibitors and COVID-19
It is prudent to discuss the findings of this review in the context of current global coronavirus disease-19 (COVID-19) pandemic. DPP4/CD26 is the receptor for the Middle East Respiratory Syndrome-Corona Virus (MERS-CoV) [74], binding the amino terminal receptor-binding spike domain S1. The emergence of the novel SARS-CoV-2 virus in Wuhan, China in late 2019 initiated global discussions about whether DPP4/CD26 represented a therapeutic target for COVID-19 as a potential receptor for the SARS-CoV-2 spike protein [75], [76]. However, the identification of angiotensin converting enzyme-2 (ACE-2) as the main receptor for virus entry [77], [78] and the inability for DPP4 to bind SARS-CoV-2 [79] suggest that DPP4 inhibitors would have little effect in preventing virus pathogenicity. Whilst the role of the innate immune system in COVID-19 is still being unravelled, it is becoming clear that elements of innate immunity are dysregulated during COVID-19 and may play a role in the development of the cytokine storm [80]. The immunomodulatory effects of DPP4 inhibitors discussed in this review suggest a therapeutic benefit that could be investigated in COVID-19 patients. Type-2 diabetes has been associated with poor prognosis in patients with COVID-19 [81]. To date, only one study has reported data on DPP4 inhibitor use in COVID-19 patients, with no advantage reported in patients on a DPP4 inhibitor [82] (primary endpoint: tracheal intubation and/or death within 7 days of admission). With millions of type-2 diabetics already prescribed DPP4 inhibitors globally, it will be interesting to determine outcomes in this patient cohort to glean any clues of therapeutic benefit or otherwise for DPP4 inhibitors.
5. Summary
DPP4 inhibitors have emerged as safe and effective therapeutic options for the management of hyperglycaemia in type-2 diabetes. Whilst early work with non-selective DPP4 inhibitors suggested broad roles for DPPs in immunity, the emergence of highly selective DPP inhibitors has revealed roles for this enzyme family in specific immune responses (Table 3 ).
Table 3.
Summary of immunomodulatory effects of DPP4 inhibitors.
| Inhibitor | Immunomodulatory Effects | Cell Type | Functional Effects | Reference |
|---|---|---|---|---|
| Sitagliptin | ↓IL-6, ↓IL-1b, ↓TNF-α, ↓MCP-1, ↓VCAM-1, ↓ICAM-1 | Endothelial cells | Improved endothelial function, ↓atherosclerotic lesions ↓macrophage infiltration | [42] |
| ↓IL-6, ↓MCP-1, ↓VCAM-1, ↓P-selectin | Macrophages & Endothelial cells | ↓Formation of atherosclerotic lesions | [66] | |
| ↓IL-6, ↓LOX-1, ↓CD36, ↓CD68 | Macrophages | ↓Foam cell formation | [69] | |
| ↓TNF- α , ↓NF- κB | Monocytes | Not reported | [71] | |
| Vildagliptin | ↓IL-6, ↓IL-18, ↓nitrotyrosine | NA | ↓Oxidative stress & ↓markers of systemic inflammation in type-2 diabetics | [43] |
| ↓nitrotyrosine, | Neutrophils | ↓wound area, ↑wound healing, ↑angiogenesis, ↓oxidative stress | [53] | |
| Not reported | Macrophages | ↓Macrophage infiltration, ↓atherosclerotic lesions | [67], [83] | |
| ↓iNOS, ↓NF- κB , ↓TNF- α , ↓IL-6, ↓IL-12 p40, ↓IL-1 β | Macrophages | Not reported | [72] | |
| Linagliptin | ↓VCAM-1, ↓Cox-2, ↓NOS-2, ↓oxidative burst, ↓RONS | Endothelial cells & neutrophils | Improved vascular function, ↓oxidative stress | [44] |
| ↓Cox-2 ↓MIP-2 | Neutrophils | Accelerated wound healing, ↓neutrophil infiltration to wound site | [54] | |
| Gemigliptin | ↓VCAM-1, ↓E-selectin, ↓JNK, ↓NF- κB , ↓IL-6, ↓IL-1β, ↓TNF- α , ↓MCP-1 | Endothelial cells | ↓Monocyte cell adhesion, inhibited foam cell formation | [45] |
| Anagliptin | ↓TNF- α , ↓NF-κB | Monocytes & macrophages | ↓Monocyte adhesion, ↓macrophage infiltration,↓atherosclerotic lesions | [65] |
| Alogliptin | ↓TNF-a, ↓IL-6, ↓MCP-1 | Monocytes | ↓Monocyte migration, ↓atherosclerotic lesions | [70] |
| AB192 | ↑VIP, ↓MPO activity | Neutrophils | ↓neutrophil infiltration in lung allografts | [56] |
Cox, cyclooxygenase; IL, interleukin; iNOS, inducible isoform nitric oxide synthase; JNK, c-Jun N-terminal kinase; LOX-1, oxidized low-density lipoprotein receptor 1; MPO, myeloperoxidase; MCP, monocyte chemoattractant protein; MIP, macrophage inflammatory protein; NF, nuclear factor; RONS, reactive oxygen & nitrogen species; TNF, tumour necrosis factor; VCAM, vascular cell adhesion molecule; VIP, vasoactive intestinal polypeptide.
Although a clear mechanism for the immunomodulatory actions of DPP4 inhibitors in innate immunity has not yet been clearly described, the broad repertoire of candidate DPP4 substrates, including chemokines, cytokines and neuropeptides indicates possible avenues for investigation (Fig. 2 ). DPP4 has been referred to as a ‘moonlighting protein’, with cell type dependent physiological actions via either its enzymatic activity or its co-stimulatory activity through via its binding partners [10], [84]. The findings that different DPP4 inhibitors can have differential effects on immune cell activation and function suggest effects that could be independent of DPP4 enzyme activity. Studies that have reported on the binding properties of different DPP4 inhibitors found evidence of conformational change in the DPP4 protein [85], [86], [87]. Whether this could explain the disparate effects of the DPP4 inhibitors remains to be seen; however, additional studies are required to dissect this phenomenon.
Fig. 2.
Proposed model for role of DPP4 inhibitors in modulating innate immune responses. DPP4 cleaves several immunoregulatory peptides with demonstrated roles in stimulating release of chemotactic factors, promoting tethering and migration of innate immune cells and stimulate innate immune cell activity, including secretion of reactive oxygen species and phagocytic activity. DPP4 inhibitors could block the proteolytic cleavage of these regulatory factors, leading to downstream anti-inflammatory effects.
The accumulating in vitro and in vivo evidence points to a direct effect of DPP4 inhibitors on innate immunity. However, these effects appear to be multi-factorial, and the glycaemic actions of DPP4 inhibition via GLP-1 could also in part explain the anti-inflammatory actions of DPP4 inhibitors. The chemorepellent properties of the DPP4 protein, as well as the capacity for soluble DPP4 to stimulate inflammatory pathways implies a complex regulatory role for DPP4 in immune cell activation. Observations that these effects could be reversed by DPP4 inhibitors lends further support to the hypothesis that conformational changes induced by DPP4 inhibitors could affect biological activity.
The anti-inflammatory actions of DPP4 inhibitors reported in clinical and pre-clinical studies of endothelial dysfunction, atherosclerosis and diabetic ulcers suggest an additional utility for this drug class. The COVID-19 pandemic has seen a global effort to identify novel pharmaceuticals that could combat this disease. The reported effects of DPP4 inhibitors on suppressing innate immune responses suggests possible applications for COVID-19 and other respiratory infections that could be further investigated. To conclude, the proliferation of DPP4 inhibitors for type-2 diabetes is indicative of their therapeutic benefit and general safety profile. The immunomodulatory effects of DPP4 inhibitors on innate immunity suggest they have further potential clinical applications; however, the precise mechanisms of these inhibitors must be determined to ensure optimum and targeted effects.
CRediT authorship contribution statement
R. Yazbeck: Conceptualization, Investigation, Resources, Writing - original draft, Writing - review & editing, Visualization, Supervision. S.E. Jaenisch: Investigation, Writing - original draft, Writing - review & editing. C.A. Abbott: Conceptualization, Writing - review & editing, Supervision.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Sulda M.L., Abbott C.A., Macardle P.J., Hall R.K., Kuss B.J. Expression and prognostic assessment of dipeptidyl peptidase IV and related enzymes in B-cell chronic lymphocytic leukemia. Cancer Biol. Ther. 2010;10(2):180–189. doi: 10.4161/cbt.10.2.12168. [DOI] [PubMed] [Google Scholar]
- 2.Yazbeck R., Howarth G.S., Abbott C.A. Dipeptidyl peptidase inhibitors, an emerging drug class for inflammatory disease? Trends Pharmacol. Sci. 2009;30(11):600–607. doi: 10.1016/j.tips.2009.08.003. [DOI] [PubMed] [Google Scholar]
- 3.Yazbeck R., Howarth G.S., Butler R.N., Geier M.S., Abbott C.A. Biochemical and histological changes in the small intestine of mice with dextran sulfate sodium colitis. J. Cell. Physiol. 2011;226(12):3219–3224. doi: 10.1002/jcp.22682. [DOI] [PubMed] [Google Scholar]
- 4.Casrouge A., Sauer A.V., Barreira da Silva R., Tejera-Alhambra M., Sánchez-Ramón S., ICAReB, Cancrini C., Ingersoll M.A., Aiuti A., Albert M.L. Lymphocytes are a major source of circulating soluble dipeptidyl peptidase 4. Clin. Exp. Immunol. 2018;194(2):166–179. doi: 10.1111/cei.13163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lettau M., Dietz M., Vollmers S., Armbrust F., Peters C., Dang T.M., Chitadze G., Kabelitz D., Janssen O. Degranulation of human cytotoxic lymphocytes is a major source of proteolytically active soluble CD26/DPP4. Cell Mol. Life Sci. 2020;77(4):751–764. doi: 10.1007/s00018-019-03207-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Herman G.A., Stevens C., Van Dyck K., Bergman A., Yi B., De Smet M., Snyder K., Hilliard D., Tanen M., Tanaka W., Wang A.Q., Zeng W., Musson D., Winchell G., Davies M.J., Ramael S., Gottesdiener K.M., Wagner J.A. Pharmacokinetics and pharmacodynamics of sitagliptin, an inhibitor of dipeptidyl peptidase IV, in healthy subjects: results from two randomized, double-blind, placebo-controlled studies with single oral doses. Clin. Pharmacol. Ther. 2005;78(6):675–688. doi: 10.1016/j.clpt.2005.09.002. [DOI] [PubMed] [Google Scholar]
- 7.Connolly B.A., Sanford D.G., Chiluwal A.K., Healey S.E., Peters D.E., Dimare M.T., Wu W., Liu Y., Maw H., Zhou Y., Li Y., Jin Z., Sudmeier J.L., Lai J.H., Bachovchin W.W. Dipeptide boronic acid inhibitors of dipeptidyl peptidase IV: determinants of potency and in vivo efficacy and safety. J. Med. Chem. 2008;51(19):6005–6013. doi: 10.1021/jm800390n. [DOI] [PubMed] [Google Scholar]
- 8.Amori R.E., Lau J., Pittas A.G. Efficacy and safety of incretin therapy in type 2 diabetes: systematic review and meta-analysis. JAMA. 2007;298(2):194–206. doi: 10.1001/jama.298.2.194. [DOI] [PubMed] [Google Scholar]
- 9.Karagiannis T., Paschos P., Paletas K., Matthews D.R., Tsapas A. Dipeptidyl peptidase-4 inhibitors for treatment of type 2 diabetes mellitus in the clinical setting: systematic review and meta-analysis. BMJ. 2012;344 doi: 10.1136/bmj.e1369. [DOI] [PubMed] [Google Scholar]
- 10.Klemann C., Wagner L., Stephan M., von Horsten S. Cut to the chase: a review of CD26/dipeptidyl peptidase-4's (DPP4) entanglement in the immune system. Clin. Exp. Immunol. 2016;185(1):1–21. doi: 10.1111/cei.12781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Iwasaki A., Medzhitov R. Control of adaptive immunity by the innate immune system. Nat. Immunol. 2015;16(4):343–353. doi: 10.1038/ni.3123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ohnuma K., Dang N.H., Morimoto C. Revisiting an old acquaintance: CD26 and its molecular mechanisms in T cell function. Trends Immunol. 2008;29(6):295–301. doi: 10.1016/j.it.2008.02.010. [DOI] [PubMed] [Google Scholar]
- 13.Ohnuma K., Takahashi N., Yamochi T., Hosono O., Dang N.H., Morimoto C. Role of CD26/dipeptidyl peptidase IV in human T cell activation and function. Front. Biosci. 2008;13:2299–2310. doi: 10.2741/2844. [DOI] [PubMed] [Google Scholar]
- 14.S. Chen, E. Zhao, W. Li, J. Wang, Association between dipeptidyl peptidase-4 inhibitor drugs and risk of acute pancreatitis: A meta-analysis, Medicine (Baltimore) 96(48) (2017) e8952-e8952. [DOI] [PMC free article] [PubMed]
- 15.Pinto L.C., Rados D.V., Barkan S.S., Leitão C.B., Gross J.L. Dipeptidyl peptidase-4 inhibitors, pancreatic cancer and acute pancreatitis: A meta-analysis with trial sequential analysis. Sci. Rep. 2018;8(1):782. doi: 10.1038/s41598-017-19055-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Flentke G.R., Munoz E., Huber B.T., Plaut A.G., Kettner C.A., Bachovchin W.W. Inhibition of dipeptidyl aminopeptidase IV (DP-IV) by Xaa-boroPro dipeptides and use of these inhibitors to examine the role of DP-IV in T-cell function. Proc. Natl. Acad. Sci. U S A. 1991;88(4):1556–1559. doi: 10.1073/pnas.88.4.1556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Schon E., Demuth H.U., Eichmann E., Horst H.J., Korner I.J., Kopp J., Mattern T., Neubert K., Noll F., Ulmer A.J., et al. Dipeptidyl peptidase IV in human T lymphocytes. Impaired induction of interleukin 2 and gamma interferon due to specific inhibition of dipeptidyl peptidase IV. Scand. J. Immunol. 1989;29(2):127–132. doi: 10.1111/j.1365-3083.1989.tb01108.x. [DOI] [PubMed] [Google Scholar]
- 18.De Meester I., Korom S., Van Damme J., Scharpe S. CD26, let it cut or cut it down. Immunol. Today. 1999;20(8):367–375. doi: 10.1016/s0167-5699(99)01486-3. [DOI] [PubMed] [Google Scholar]
- 19.Lankas G.R., Leiting B., Roy R.S., Eiermann G.J., Beconi M.G., Biftu T., Chan C.C., Edmondson S., Feeney W.P., He H., Ippolito D.E., Kim D., Lyons K.A., Ok H.O., Patel R.A., Petrov A.N., Pryor K.A., Qian X., Reigle L., Woods A., Wu J.K., Zaller D., Zhang X., Zhu L., Weber A.E., Thornberry N.A. Dipeptidyl peptidase IV inhibition for the treatment of type 2 diabetes: potential importance of selectivity over dipeptidyl peptidases 8 and 9. Diabetes. 2005;54(10):2988–2994. doi: 10.2337/diabetes.54.10.2988. [DOI] [PubMed] [Google Scholar]
- 20.Biftu T., Feng D., Qian X., Liang G.B., Kieczykowski G., Eiermann G., He H., Leiting B., Lyons K., Petrov A., Sinha-Roy R., Zhang B., Scapin G., Patel S., Gao Y.D., Singh S., Wu J., Zhang X., Thornberry N.A., Weber A.E. (3R)-4-[(3R)-3-Amino-4-(2,4,5-trifluorophenyl)butanoyl]-3-(2,2,2-trifluoroethyl)-1,4-diazepan-2-one, a selective dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. Bioorg. Med. Chem. Lett. 2007;17(1):49–52. doi: 10.1016/j.bmcl.2006.09.099. [DOI] [PubMed] [Google Scholar]
- 21.Brandt I., Joossens J., Chen X., Maes M.B., Scharpe S., De Meester I., Lambeir A.M. Inhibition of dipeptidyl-peptidase IV catalyzed peptide truncation by Vildagliptin ((2S)-{[(3-hydroxyadamantan-1-yl)amino]acetyl}-pyrrolidine-2-carbonitrile) Biochem. Pharmacol. 2005;70(1):134–143. doi: 10.1016/j.bcp.2005.04.009. [DOI] [PubMed] [Google Scholar]
- 22.Wang A., Dorso C., Kopcho L., Locke G., Langish R., Harstad E., Shipkova P., Marcinkeviciene J., Hamann L., Kirby M.S. Potency, selectivity and prolonged binding of saxagliptin to DPP4: maintenance of DPP4 inhibition by saxagliptin in vitro and ex vivo when compared to a rapidly-dissociating DPP4 inhibitor. BMC Pharmacol. 2012;12:2. doi: 10.1186/1471-2210-12-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Thomas L., Eckhardt M., Langkopf E., Tadayyon M., Himmelsbach F., Mark M. (R)-8-(3-amino-piperidin-1-yl)-7-but-2-ynyl-3-methyl-1-(4-methyl-quinazolin-2-ylm ethyl)-3,7-dihydro-purine-2,6-dione (BI 1356), a novel xanthine-based dipeptidyl peptidase 4 inhibitor, has a superior potency and longer duration of action compared with other dipeptidyl peptidase-4 inhibitors. J. Pharmacol. Exp. Ther. 2008;325(1):175–182. doi: 10.1124/jpet.107.135723. [DOI] [PubMed] [Google Scholar]
- 24.Kim S.H., Jung E., Yoon M.K., Kwon O.H., Hwang D.M., Kim D.W., Kim J., Lee S.M., Yim H.J. Pharmacological profiles of gemigliptin (LC15-0444), a novel dipeptidyl peptidase-4 inhibitor, in vitro and in vivo. Eur. J. Pharmacol. 2016;788:54–64. doi: 10.1016/j.ejphar.2016.06.016. [DOI] [PubMed] [Google Scholar]
- 25.Kato N., Oka M., Murase T., Yoshida M., Sakairi M., Yamashita S., Yasuda Y., Yoshikawa A., Hayashi Y., Makino M., Takeda M., Mirensha Y., Kakigami T. Discovery and pharmacological characterization of N-[2-({2-[(2S)-2-cyanopyrrolidin-1-yl]-2-oxoethyl}amino)-2-methylpropyl]-2-methyl pyrazolo[1,5-a]pyrimidine-6-carboxamide hydrochloride (anagliptin hydrochloride salt) as a potent and selective DPP-IV inhibitor. Bioorg. Med. Chem. 2011;19(23):7221–7227. doi: 10.1016/j.bmc.2011.09.043. [DOI] [PubMed] [Google Scholar]
- 26.Yoshida T., Akahoshi F., Sakashita H., Kitajima H., Nakamura M., Sonda S., Takeuchi M., Tanaka Y., Ueda N., Sekiguchi S., Ishige T., Shima K., Nabeno M., Abe Y., Anabuki J., Soejima A., Yoshida K., Takashina Y., Ishii S., Kiuchi S., Fukuda S., Tsutsumiuchi R., Kosaka K., Murozono T., Nakamaru Y., Utsumi H., Masutomi N., Kishida H., Miyaguchi I., Hayashi Y. Discovery and preclinical profile of teneligliptin (3-[(2S,4S)-4-[4-(3-methyl-1-phenyl-1H-pyrazol-5-yl)piperazin-1-yl]pyrrolidin-2-y lcarbonyl]thiazolidine): a highly potent, selective, long-lasting and orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. Bioorg. Med. Chem. 2012;20(19):5705–5719. doi: 10.1016/j.bmc.2012.08.012. [DOI] [PubMed] [Google Scholar]
- 27.Feng J., Zhang Z., Wallace M.B., Stafford J.A., Kaldor S.W., Kassel D.B., Navre M., Shi L., Skene R.J., Asakawa T., Takeuchi K., Xu R., Webb D.R., Gwaltney S.L., 2nd Discovery of alogliptin: a potent, selective, bioavailable, and efficacious inhibitor of dipeptidyl peptidase IV. J. Med. Chem. 2007;50(10):2297–2300. doi: 10.1021/jm070104l. [DOI] [PubMed] [Google Scholar]
- 28.Shu C., Ge H., Song M., Chen J.H., Zhou H., Qi Q., Wang F., Ma X., Yang X., Zhang G., Ding Y., Zhou D., Peng P., Shih C.K., Xu J., Wu F. Discovery of Imigliptin, a Novel Selective DPP-4 Inhibitor for the Treatment of Type 2 Diabetes. ACS Med. Chem. Lett. 2014;5(8):921–926. doi: 10.1021/ml5001905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.McKeage K. Trelagliptin: First Global Approval. Drugs. 2015;75(10):1161–1164. doi: 10.1007/s40265-015-0431-9. [DOI] [PubMed] [Google Scholar]
- 30.Biftu T., Sinha-Roy R., Chen P., Qian X., Feng D., Kuethe J.T., Scapin G., Gao Y.D., Yan Y., Krueger D., Bak A., Eiermann G., He J., Cox J., Hicks J., Lyons K., He H., Salituro G., Tong S., Patel S., Doss G., Petrov A., Wu J., Xu S.S., Sewall C., Zhang X., Zhang B., Thornberry N.A., Weber A.E. Omarigliptin (MK-3102): a novel long-acting DPP-4 inhibitor for once-weekly treatment of type 2 diabetes. J. Med. Chem. 2014;57(8):3205–3212. doi: 10.1021/jm401992e. [DOI] [PubMed] [Google Scholar]
- 31.McCormack P.L. Evogliptin: First Global Approval. Drugs. 2015;75(17):2045–2049. doi: 10.1007/s40265-015-0496-5. [DOI] [PubMed] [Google Scholar]
- 32.Williams-Herman D., Round E., Swern A.S., Musser B., Davies M.J., Stein P.P., Kaufman K.D., Amatruda J.M. Safety and tolerability of sitagliptin in patients with type 2 diabetes: a pooled analysis. BMC Endocr. Disord. 2008;8:14. doi: 10.1186/1472-6823-8-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Elashoff M., Matveyenko A.V., Gier B., Elashoff R., Butler P.C. Pancreatitis, pancreatic, and thyroid cancer with glucagon-like peptide-1-based therapies. Gastroenterology. 2011;141(1):150–156. doi: 10.1053/j.gastro.2011.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Garg R., Chen W., Pendergrass M. Acute pancreatitis in type 2 diabetes treated with exenatide or sitagliptin: a retrospective observational pharmacy claims analysis. Diabetes Care. 2010;33(11):2349–2354. doi: 10.2337/dc10-0482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hanssen N.M., Jandeleit-Dahm K.A. Dipeptidyl peptidase-4 inhibitors and cardiovascular and renal disease in type 2 diabetes: What have we learned from the CARMELINA trial? Diab. Vasc. Dis. Res. 2019;16(4):303–309. doi: 10.1177/1479164119842339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rosenstock J., Kahn S.E., Johansen O.E., Zinman B., Espeland M.A., Woerle H.J., Pfarr E., Keller A., Mattheus M., Baanstra D., Meinicke T., George J.T., von Eynatten M., McGuire D.K., Marx N., f.t.C. Investigators Effect of Linagliptin vs Glimepiride on Major Adverse Cardiovascular Outcomes in Patients With Type 2 Diabetes: The CAROLINA Randomized Clinical Trial. JAMA. 2019;322(12):1155–1166. doi: 10.1001/jama.2019.13772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Norman J. The role of cytokines in the pathogenesis of acute pancreatitis. Am. J. Surg. 1998;175(1):76–83. doi: 10.1016/s0002-9610(97)00240-7. [DOI] [PubMed] [Google Scholar]
- 38.Habtezion A. Inflammation in acute and chronic pancreatitis. Curr. Opin. Gastroenterol. 2015;31(5):395–399. doi: 10.1097/MOG.0000000000000195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Rajendran P., Rengarajan T., Thangavel J., Nishigaki Y., Sakthisekaran D., Sethi G., Nishigaki I. The vascular endothelium and human diseases. Int. J. Biol. Sci. 2013;9(10):1057–1069. doi: 10.7150/ijbs.7502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Krishnaswamy G., Kelley J., Yerra L., Smith J.K., Chi D.S. Human endothelium as a source of multifunctional cytokines: molecular regulation and possible role in human disease. J. Interferon. Cytokine Res. 1999;19(2):91–104. doi: 10.1089/107999099314234. [DOI] [PubMed] [Google Scholar]
- 41.Steyers C.M., 3rd, Miller F.J., Jr. Endothelial dysfunction in chronic inflammatory diseases. Int. J. Mol. Sci. 2014;15(7):11324–11349. doi: 10.3390/ijms150711324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Matsubara J., Sugiyama S., Akiyama E., Iwashita S., Kurokawa H., Ohba K., Maeda H., Fujisue K., Yamamoto E., Kaikita K., Hokimoto S., Jinnouchi H., Ogawa H. Dipeptidyl Peptidase-4 Inhibitor, Sitagliptin, Improves Endothelial Dysfunction in Association With Its Anti-Inflammatory Effects in Patients With Coronary Artery Disease and Uncontrolled Diabetes. Circ. J. 2013;77(5):1337–1344. doi: 10.1253/circj.cj-12-1168. [DOI] [PubMed] [Google Scholar]
- 43.Rizzo M.R., Barbieri M., Marfella R., Paolisso G. Reduction of oxidative stress and inflammation by blunting daily acute glucose fluctuations in patients with type 2 diabetes: role of dipeptidyl peptidase-IV inhibition. Diabetes Care. 2012;35(10):2076–2082. doi: 10.2337/dc12-0199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kroller-Schon S., Knorr M., Hausding M., Oelze M., Schuff A., Schell R., Sudowe S., Scholz A., Daub S., Karbach S., Kossmann S., Gori T., Wenzel P., Schulz E., Grabbe S., Klein T., Munzel T., Daiber A. Glucose-independent improvement of vascular dysfunction in experimental sepsis by dipeptidyl-peptidase 4 inhibition. Cardiovasc. Res. 2012;96(1):140–149. doi: 10.1093/cvr/cvs246. [DOI] [PubMed] [Google Scholar]
- 45.Hwang H.J., Chung H.S., Jung T.W., Ryu J.Y., Hong H.C., Seo J.A., Kim S.G., Kim N.H., Choi K.M., Choi D.S., Baik S.H., Yoo H.J. The dipeptidyl peptidase-IV inhibitor inhibits the expression of vascular adhesion molecules and inflammatory cytokines in HUVECs via Akt- and AMPK-dependent mechanisms. Mol. Cell Endocrinol. 2015;405:25–34. doi: 10.1016/j.mce.2015.01.025. [DOI] [PubMed] [Google Scholar]
- 46.Witko-Sarsat V., Rieu P., Descamps-Latscha B., Lesavre P., Halbwachs-Mecarelli L. Neutrophils: molecules, functions and pathophysiological aspects. Lab. Invest. 2000;80(5):617–653. doi: 10.1038/labinvest.3780067. [DOI] [PubMed] [Google Scholar]
- 47.Falloon J., Gallin J.I. Neutrophil granules in health and disease. J. Allergy Clin. Immunol. 1986;77(5):653–662. doi: 10.1016/0091-6749(86)90404-5. [DOI] [PubMed] [Google Scholar]
- 48.Kolaczkowska E., Kubes P. Neutrophil recruitment and function in health and inflammation. Nat. Rev. Immunol. 2013;13(3):159–175. doi: 10.1038/nri3399. [DOI] [PubMed] [Google Scholar]
- 49.Kumar V., Sharma A. Neutrophils: Cinderella of innate immune system. Int. Immunopharmacol. 2010;10(11):1325–1334. doi: 10.1016/j.intimp.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 50.Blakytny R., Jude E. The molecular biology of chronic wounds and delayed healing in diabetes. Diabet. Med. 2006;23(6):594–608. doi: 10.1111/j.1464-5491.2006.01773.x. [DOI] [PubMed] [Google Scholar]
- 51.A. Alavi, R.G. Sibbald, D. Mayer, L. Goodman, M. Botros, D.G. Armstrong, K. Woo, T. Boeni, E.A. Ayello, R.S. Kirsner, Diabetic foot ulcers: Part I. Pathophysiology and prevention, J Am Acad Dermatol 70(1) (2014) 1 e1-18; quiz 19-20. [DOI] [PubMed]
- 52.Pradhan L., Nabzdyk C., Andersen N.D., LoGerfo F.W., Veves A. Inflammation and neuropeptides: the connection in diabetic wound healing. Expert. Rev. Mol. Med. 2009;11 doi: 10.1017/S1462399409000945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Marfella R., Sasso F.C., Rizzo M.R., Paolisso P., Barbieri M., Padovano V., Carbonara O., Gualdiero P., Petronella P., Ferraraccio F., Petrella A., Canonico R., Campitiello F., Della Corte A., Paolisso G., Canonico S. Dipeptidyl peptidase 4 inhibition may facilitate healing of chronic foot ulcers in patients with type 2 diabetes. Exp. Diabetes Res. 2012 doi: 10.1155/2012/892706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Schurmann C., Linke A., Engelmann-Pilger K., Steinmetz C., Mark M., Pfeilschifter J., Klein T., Frank S. The dipeptidyl peptidase-4 inhibitor linagliptin attenuates inflammation and accelerates epithelialization in wounds of diabetic ob/ob mice. J. Pharmacol. Exp. Ther. 2012;342(1):71–80. doi: 10.1124/jpet.111.191098. [DOI] [PubMed] [Google Scholar]
- 55.Grisham M.B., Granger D.N. Neutrophil-mediated mucosal injury. Role of reactive oxygen metabolites. Digestive Diseases Sci. 1988;33(3 Suppl):6S–15S. doi: 10.1007/BF01538126. [DOI] [PubMed] [Google Scholar]
- 56.Jungraithmayr W., De Meester I., Matheeussen V., Inci I., Augustyns K., Scharpe S., Weder W., Korom S. Inhibition of CD26/DPP IV attenuates ischemia/reperfusion injury in orthotopic mouse lung transplants: the pivotal role of vasoactive intestinal peptide. Peptides. 2010;31(4):585–591. doi: 10.1016/j.peptides.2009.12.012. [DOI] [PubMed] [Google Scholar]
- 57.Yazbeck R., Sulda M.L., Howarth G.S., Bleich A., Raber K., von Horsten S., Holst J.J., Abbott C.A. Dipeptidyl peptidase expression during experimental colitis in mice. Inflamm. Bowel Dis. 2010;16(8):1340–1351. doi: 10.1002/ibd.21241. [DOI] [PubMed] [Google Scholar]
- 58.Herlihy S.E., Pilling D., Maharjan A.S., Gomer R.H. Dipeptidyl peptidase IV is a human and murine neutrophil chemorepellent. J. Immunol. 2013;190(12):6468–6477. doi: 10.4049/jimmunol.1202583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Herlihy S.E., Brown M.L., Pilling D., Weeks B.R., Myers L.K., Gomer R.H. Role of the neutrophil chemorepellent soluble dipeptidyl peptidase IV in decreasing inflammation in a murine model of arthritis. Arthritis Rheumatol. 2015;67(10):2634–2638. doi: 10.1002/art.39250. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Matheeussen V., Waumans Y., Martinet W., Van Goethem S., Van der Veken P., Scharpe S., Augustyns K., De Meyer G.R., De Meester I. Dipeptidyl peptidases in atherosclerosis: expression and role in macrophage differentiation, activation and apoptosis. Basic Res. Cardiol. 2013;108(3):350. doi: 10.1007/s00395-013-0350-4. [DOI] [PubMed] [Google Scholar]
- 61.Gliddon D.R., Howard C.J. CD26 is expressed on a restricted subpopulation of dendritic cells in vivo. Eur. J. Immunol. 2002;32(5):1472–1481. doi: 10.1002/1521-4141(200205)32:5<1472::AID-IMMU1472>3.0.CO;2-Q. [DOI] [PubMed] [Google Scholar]
- 62.Nakano H., Moran T.P., Nakano K., Gerrish K.E., Bortner C.D., Cook D.N. Complement receptor C5aR1/CD88 and dipeptidyl peptidase-4/CD26 define distinct hematopoietic lineages of dendritic cells. J. Immunol. 2015;194(8):3808–3819. doi: 10.4049/jimmunol.1402195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhong J., Rao X., Deiuliis J., Braunstein Z., Narula V., Hazey J., Mikami D., Needleman B., Satoskar A.R., Rajagopalan S. A potential role for dendritic cell/macrophage-expressing DPP4 in obesity-induced visceral inflammation. Diabetes. 2013;62(1):149–157. doi: 10.2337/db12-0230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Moore K.J., Sheedy F.J., Fisher E.A. Macrophages in atherosclerosis: a dynamic balance. Nat. Rev. Immunol. 2013;13(10):709–721. doi: 10.1038/nri3520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ervinna N., Mita T., Yasunari E., Azuma K., Tanaka R., Fujimura S., Sukmawati D., Nomiyama T., Kanazawa A., Kawamori R., Fujitani Y., Watada H. Anagliptin, a DPP-4 inhibitor, suppresses proliferation of vascular smooth muscles and monocyte inflammatory reaction and attenuates atherosclerosis in male apo E-deficient mice. Endocrinology. 2013;154(3):1260–1270. doi: 10.1210/en.2012-1855. [DOI] [PubMed] [Google Scholar]
- 66.Zeng Y., Li C., Guan M., Zheng Z., Li J., Xu W., Wang L., He F., Xue Y. The DPP-4 inhibitor sitagliptin attenuates the progress of atherosclerosis in apolipoprotein-E-knockout mice via AMPK- and MAPK-dependent mechanisms. Cardiovasc Diabetol. 2014;13:32. doi: 10.1186/1475-2840-13-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Terasaki M., Nagashima M., Watanabe T., Nohtomi K., Mori Y., Miyazaki A., Hirano T. Effects of PKF275-055, a dipeptidyl peptidase-4 inhibitor, on the development of atherosclerotic lesions in apolipoprotein E-null mice. Metabolism. 2012;61(7):974–977. doi: 10.1016/j.metabol.2011.11.011. [DOI] [PubMed] [Google Scholar]
- 68.Vittone F., Liberman A., Vasic D., Ostertag R., Esser M., Walcher D., Ludwig A., Marx N., Burgmaier M. Sitagliptin reduces plaque macrophage content and stabilises arteriosclerotic lesions in Apoe (-/-) mice. Diabetologia. 2012;55(8):2267–2275. doi: 10.1007/s00125-012-2582-5. [DOI] [PubMed] [Google Scholar]
- 69.Dai Y., Wang X., Ding Z., Dai D., Mehta J.L. DPP-4 inhibitors repress foam cell formation by inhibiting scavenger receptors through protein kinase C pathway. Acta Diabetol. 2014;51(3):471–478. doi: 10.1007/s00592-013-0541-3. [DOI] [PubMed] [Google Scholar]
- 70.Shah Z., Kampfrath T., Deiuliis J.A., Zhong J., Pineda C., Ying Z., Xu X., Lu B., Moffatt-Bruce S., Durairaj R., Sun Q., Mihai G., Maiseyeu A., Rajagopalan S. Long-term dipeptidyl-peptidase 4 inhibition reduces atherosclerosis and inflammation via effects on monocyte recruitment and chemotaxis. Circulation. 2011;124(21):2338–2349. doi: 10.1161/CIRCULATIONAHA.111.041418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ikeda T., Kumagai E., Iwata S., Yamakawa A. Soluble CD26/Dipeptidyl Peptidase IV Enhances the Transcription of IL-6 and TNF-alpha in THP-1 Cells and Monocytes. PLoS ONE. 2013;8(6) doi: 10.1371/journal.pone.0066520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lee D.S., Lee E.S., Alam M.M., Jang J.H., Lee H.S., Oh H., Kim Y.C., Manzoor Z., Koh Y.S., Kang D.G., Lee D.H. Soluble DPP-4 up-regulates toll-like receptors and augments inflammatory reactions, which are ameliorated by vildagliptin or mannose-6-phosphate. Metabolism. 2016;65(2):89–101. doi: 10.1016/j.metabol.2015.10.002. [DOI] [PubMed] [Google Scholar]
- 73.Waumans Y., Vliegen G., Maes L., Rombouts M., Declerck K., Van Der Veken P., Vanden Berghe W., De Meyer G.R., Schrijvers D., De Meester I. The Dipeptidyl Peptidases 4, 8, and 9 in Mouse Monocytes and Macrophages: DPP8/9 Inhibition Attenuates M1 Macrophage Activation in Mice. Inflammation. 2016;39(1):413–424. doi: 10.1007/s10753-015-0263-5. [DOI] [PubMed] [Google Scholar]
- 74.Raj V.S., Mou H., Smits S.L., Dekkers D.H.W., Müller M.A., Dijkman R., Muth D., Demmers J.A.A., Zaki A., Fouchier R.A.M., Thiel V., Drosten C., Rottier P.J.M., Osterhaus A.D.M.E., Bosch B.J., Haagmans B.L. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature. 2013;495(7440):251–254. doi: 10.1038/nature12005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.R. Dalan, Is DPP4 inhibition a comrade or adversary in COVID-19 infection, Diabetes Res. Clin. Pract. 164 (2020) 108216-108216. [DOI] [PMC free article] [PubMed]
- 76.Strollo R., Pozzilli P. DPP4 inhibition: Preventing SARS-CoV-2 infection and/or progression of COVID-19? Diabetes Metab. Res. Rev. 2020 doi: 10.1002/dmrr.3330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wang Q., Zhang Y., Wu L., Niu S., Song C., Zhang Z., Lu G., Qiao C., Hu Y., Yuen K.Y., Wang Q., Zhou H., Yan J., Qi J. Structural and Functional Basis of SARS-CoV-2 Entry by Using Human ACE2. Cell. 2020;181(4):894–904 e9. doi: 10.1016/j.cell.2020.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Hoffmann M., Kleine-Weber H., Schroeder S., Kruger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A., Muller M.A., Drosten C., Pohlmann S. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181(2):271–280 e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Xi C.R., Di Fazio A., Nadvi N.A., Patel K., Xiang M.S.W., Zhang H.E., Deshpande C., Low J.K.K., Wang X.T., Chen Y., McMillan C.L.D., Isaacs A., Osborne B., Vieira de Ribeiro A.J., McCaughan G.W., Mackay J.P., Church W.B., Gorrell M.D. A Novel Purification Procedure for Active Recombinant Human DPP4 and the Inability of DPP4 to Bind SARS-CoV-2. Molecules. 2020;25(22) doi: 10.3390/molecules25225392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Birra D., Benucci M., Landolfi L., Merchionda A., Loi G., Amato P., Licata G., Quartuccio L., Triggiani M., Moscato P. COVID 19: a clue from innate immunity. Immunol. Res. 2020;68(3):161–168. doi: 10.1007/s12026-020-09137-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Apicella M., Campopiano M.C., Mantuano M., Mazoni L., Coppelli A., Del Prato S. COVID-19 in people with diabetes: understanding the reasons for worse outcomes. Lancet Diabetes Endocrinol. 2020 doi: 10.1016/S2213-8587(20)30238-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cariou B., Hadjadj S., Wargny M., Pichelin M., Al-Salameh A., Allix I., Amadou C., Arnault G., Baudoux F., Bauduceau B., Borot S., Bourgeon-Ghittori M., Bourron O., Boutoille D., Cazenave-Roblot F., Chaumeil C., Cosson E., Coudol S., Darmon P., Disse E., Ducet-Boiffard A., Gaborit B., Joubert M., Kerlan V., Laviolle B., Marchand L., Meyer L., Potier L., Prevost G., Riveline J.P., Robert R., Saulnier P.J., Sultan A., Thebaut J.F., Thivolet C., Tramunt B., Vatier C., Roussel R., Gautier J.F., Gourdy P., C. investigators Phenotypic characteristics and prognosis of inpatients with COVID-19 and diabetes: the CORONADO study. Diabetologia. 2020;63(8):1500–1515. doi: 10.1007/s00125-020-05180-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Terasaki M., Nagashima M., Nohtomi K., Kohashi K., Tomoyasu M., Sinmura K., Nogi Y., Katayama Y., Sato K., Itoh F., Watanabe T., Hirano T. Preventive effect of dipeptidyl peptidase-4 inhibitor on atherosclerosis is mainly attributable to incretin's actions in nondiabetic and diabetic apolipoprotein E-null mice. PLoS ONE. 2013;8(8) doi: 10.1371/journal.pone.0070933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Boonacker E., Van Noorden C.J. The multifunctional or moonlighting protein CD26/DPPIV. Eur. J. Cell Biol. 2003;82(2):53–73. doi: 10.1078/0171-9335-00302. [DOI] [PubMed] [Google Scholar]
- 85.Arulmozhiraja S., Matsuo N., Ishitsubo E., Okazaki S., Shimano H., Tokiwa H. Comparative Binding Analysis of Dipeptidyl Peptidase IV (DPP-4) with Antidiabetic Drugs - An Ab Initio Fragment Molecular Orbital Study. PLoS ONE. 2016;11(11) doi: 10.1371/journal.pone.0166275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sheehan S.M., Mest H.J., Watson B.M., Klimkowski V.J., Timm D.E., Cauvin A., Parsons S.H., Shi Q., Canada E.J., Wiley M.R., Ruehter G., Evers B., Petersen S., Blaszczak L.C., Pulley S.R., Margolis B.J., Wishart G.N., Renson B., Hankotius D., Mohr M., Zechel J.C., Michael Kalbfleisch J., Dingess-Hammond E.A., Boelke A., Weichert A.G. Discovery of non-covalent dipeptidyl peptidase IV inhibitors which induce a conformational change in the active site. Bioorg. Med. Chem. Lett. 2007;17(6):1765–1768. doi: 10.1016/j.bmcl.2006.12.074. [DOI] [PubMed] [Google Scholar]
- 87.Ansorge S., Nordhoff K., Bank U., Heimburg A., Julius H., Breyer D., Thielitz A., Reinhold D., Tager M. Novel aspects of cellular action of dipeptidyl peptidase IV/CD26. Biol. Chem. 2011;392(3):153–168. doi: 10.1515/BC.2011.008. [DOI] [PubMed] [Google Scholar]