Abstract
OBJECTIVES:
Lung cancer is the leading cause of cancer-related deaths worldwide. However, factors associated with the survival of patients with advanced non-small-cell lung cancer (NSCLC) who received only hospice care are largely unclear. In this study, we aimed to determine the prognostic factors correlated with survival in patients with advanced NSCLC who had undergone hospice care only.
METHODS:
A total of 102 patients with recurrent stage III/IV NSCLC after traditional treatment failure were investigated. Survival was measured from the date of enrollment to December 2019 or the time of death. Tumor tissues were collected, and DNA sequencing was performed to identify somatic mutations. Data on clinical factors of patients were collected and analyzed by univariate and multivariate analyses. Overall survival analysis was conducted using the Kaplan-Meier method.
RESULTS:
The 6-month, 1-year, and 2-year overall survival rates of the 102 patients with metastatic NSCLC were 17.65%, 3.92%, and 0.98%, respectively. The median overall survival of the 102 patients was 3.15 months. Tumor location in the peripheral lung, epidermal growth factor receptor (EGFR) inhibitor history, low tumor mutation load, adenocarcinoma, and poor performance status score were associated with prolonged survival compared with tumor location in the central lung, no EGFR inhibitor history, high tumor mutation load, squamous cell carcinoma, and good performance status score (p=0.045, p=0.003, p=0.045, p=0.021, and p=0.0003, respectively).
CONCLUSIONS:
EGFR inhibitor treatment history and tumor mutation load are risk factors for the overall survival of patients with stage III/IV NSCLC who have undergone only hospice care. These results provide a critical clinical basis for further study of nontraditional anti-tumor responses induced by EGFR inhibitors.
Keywords: Lung Cancer, Hospice Care, EGFR Inhibitor, Overall Survival, Tumor Mutation
INTRODUCTION
Lung cancer is the leading cause of cancer-related deaths worldwide (1). Non-small-cell lung cancer (NSCLC), which accounts for approximately 85% of all lung cancer cases, is often diagnosed at a late stage and has a poor prognosis (2). Traditional treatment strategies, including surgical resection and chemotherapy, are most commonly used in lung cancer treatment. However, the survival prognosis achieved with these conventional therapies is still unsatisfactory, with a 5-year survival rate of only approximately 15% (3-4). Recently, epidermal growth factor receptor-tyrosine kinase inhibitors (EGFR-TKIs) gefitinib, erlotinib, and AZD-92912 have been used to treat unresectable or recurrent lung cancer and have shown significantly improved clinical outcomes in patients with NSCLC with EGFR mutations (5-15). Nonetheless, tumors will inevitably develop drug resistance, and tumor recurrence has been observed (16-18).
Some patients who experienced disease progression, continued on conventional treatments following failures. It has been shown that continuing EGFR inhibitor treatment is beneficial in many patients even after they develop resistance to EGFR inhibitors on the basis of the hypothesis that a population of EGFR inhibitor-sensitive cells remains during disease progression, and resistant cells may be detected radiographically before widespread dissemination occurs (19). Other patients receive only hospice care. Patients who discontinue EGFR inhibitor treatment have a higher risk of symptomatic progression and increase in tumor size, which may lead to a much more rapid progression of the cancer (20-21). Despite the success of EGFR inhibitor treatment, questions on whether the benefits of continuing EGFR inhibitor treatment are temporary or long-term, how the overall survival is affected after EGFR inhibitor discontinuation, and what factors are correlated with overall survival in patients with metastatic stage III/IV NSCLC who receive hospice care remain unanswered. In this study, we aimed to determine the prognostic factors that are correlated with the survival of patients with advanced NSCLC who had received only hospice care.
SUBJECTS AND METHODS
Patient selection
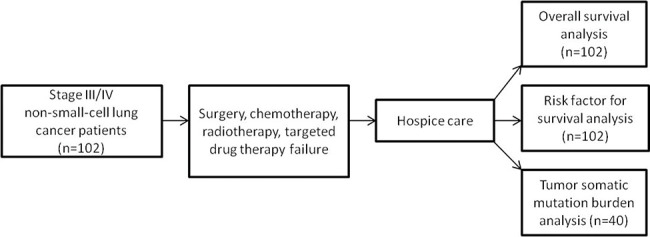
A total of 102 patients with stage III/IV advanced NSCLC between December 2015 and April 2019 were included in this study. The study protocol was in accordance with the ethical guidelines outlined in the Declaration of Helsinki, as revised in 2013, and was approved by the Tianjin Anti-Cancer Association and ethics committee of the Tianjin Beichen Hospital. Informed consent was obtained from all patients enrolled in this study. Patients were selected according to the following inclusion criteria: 1) stage III/IV NSCLC according to the National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology (version 3.2016), Non-Small-Cell Lung Cancer Stage Classification; 2) disease progression on standard treatments, including surgery, chemotherapy, radiotherapy, and targeted drug therapy, and the absence of an active treatment option; 3) current hospice care only; and 4) a diagnosis of NSCLC confirmed by biopsy. Patients were excluded from the present study if they 1) did not meet the abovementioned criteria; 2) continued to receive antineoplastic therapies intended at prolonging survival, such as traditional cytotoxic, targeted, or immune-based therapies, despite disease progression; or 2) had incomplete medical records. It is noteworthy that patients with pleural effusion were all confirmed as having malignant pleural effusion by thoracentesis. None of the enrolled patients received any other survival-prolonging treatments during the follow-up period. All patient data were retrospectively collected from detailed hospital medical records after clinical sample detection. The clinical and demographic characteristics of all patients are summarized in Table 1. A study flowchart is shown in Figure 1.
Table 1. Patients’ clinical and demographic characteristics at baseline.
| Characteristic | Mean±SD or no. of cases (n=102) | Range |
|---|---|---|
| Sex (male-female) | 57-45 | - |
| Age (year) | 65.64±11.20 | 28-91 |
| Weight (kg) | 62.50±9.45 | 40.5-83 |
| Height (cm) | 167.0±7.43 | 150-186 |
| Smoking history (yes-no) | 71-31 | - |
| Pleural effusion (yes-no) | 55-47 | - |
| Tumor size (cm) | 4.34±1.64 | 1.4-10.1 |
| Tumor site (central-peripheral) | 42-60 | - |
| CEA (ng/mL) | 71.40±88.73 | 0.91-1005 |
| CA125 (U/mL) | 110.03±257.67 | 11.47-2482.1 |
| CA153 (U/mL) | 50.10±103.37 | 4.5-1016.2 |
| EGFR mutation (yes-no) | 17-31** | - |
| Previous treatments | ||
| Surgery (yes-no) | 7-95 | - |
| Local radiotherapy (yes-no) | 89-13 | - |
| Chemotherapy (yes-no) | 73-29 | - |
| EGFR inhibitor (yes-no) | 17-85 | - |
| Lymphocyte (%) | 18.01±8.68 | 3.3-44.5 |
| Brain metastases (yes-no) | 11-91 | - |
| Bone metastases (yes-no) | 35-67 | - |
| Tumor histology (SQ-AD) | 48-54 | - |
| ECOG PS (0/1/2-3) | 31-71 | - |
48 of 102 performed EGFR mutation analysis.
Figure 1. Flowchart of the study.
Hospice care
Hospice care is defined as supportive care at the end of life when life prolongation is not the primary treatment goal and disease-modifying therapies are no longer provided (22). Hospice care is particularly intended at improving the quality of life of patients through pain relief; treatment of fever and cough caused by lung tumor; and physical, psychosocial, and spiritual care. In this study, hospice care included the use of analgesics, antipyretic and cough medications, and antibiotics, with attention to psychological and spiritual aspects of care. None of the enrolled patients received antineoplastic treatments intended at improving survival, including conventional chemotherapy, radiotherapy, targeted drug therapy, or immunotherapy, during the follow-up period.
Assessment of somatic tumor cell mutation by next-generation sequencing
We followed a genotyping panel designed to detect 508 cancer-associated genes on the basis of the biobanking system and in conjunction with the clinic and pathology laboratory (23). DNA was extracted from tumor samples of 40 patients immediately after biopsy using the TIANamp Genomic DNA Kit (TIANGEN) according to the manufacturer’s instructions. Then, exome libraries were constructed from the isolated DNA. Barcoded next-generation sequencing libraries were constructed (Hengjia Biotech), and exome captures were sequenced on the HiSeq X Ten System (Illumina, USA).
Serological test for carcinoembryonic antigen (CEA), carcinoma antigen 125 (CA125), and carcinoma antigen 153 (CA153) and lymphocyte percentage analysis
Serum CEA, CA125, and CA153 levels were determined by a luminescence-based method using detection kits (Access CEA, Cat. #33200; Access OV Monitor, Cat. #386357; Access BR Monitor, Cat. #387620, Beckman Coulter Inc.) and subsequently measured using the Automatic Luminescence Immunoassay Analyzer (UniCel DxI 800, Beckman Coulter). Lymphocyte percentage was tested by fluorescence-activated cell sorting using the auto hematology analyzer (BC-6800, Mindray).
Follow-up assessments
For survival analysis, all patients were followed up from the date of initial enrollment until December 2019 or until the time of death. Subsequent follow-up examinations included assessment of tumor biomarkers CEA, CA125, and CA153; peripheral blood T-cell counts; and tumor DNA sequencing of 40 patients from whom tissue samples were available.
Statistical analysis
Univariate and multivariate analyses were performed to evaluate the possible factors that were correlated with patient prognosis and survival outcomes. Survival time was defined as the duration from the date of enrollment to the date of death or December 2019. The enrollment date for each patient in the study was the date of start of hospice care only. Survival curves were plotted, and overall survival rates were estimated using the Kaplan-Meier method and compared using the log-rank test or Cox’s proportional hazard model. Variables with p<0.6 on univariate analysis were entered into the multivariate model (Supplemental Table 1). The Cox regression method was used for multivariate analyses. Variables with p<0.05 were considered statistically significant. All statistical analyses were performed using the Statistical Package for Social Sciences (SPSS 16.0 for Windows; SPSS Inc., Chicago, IL) and GraphPad Prism 5.0 (USA).
RESULTS
Overall survival analysis of all patients with advanced stage III/IV NSCLC who had undergone hospice care only
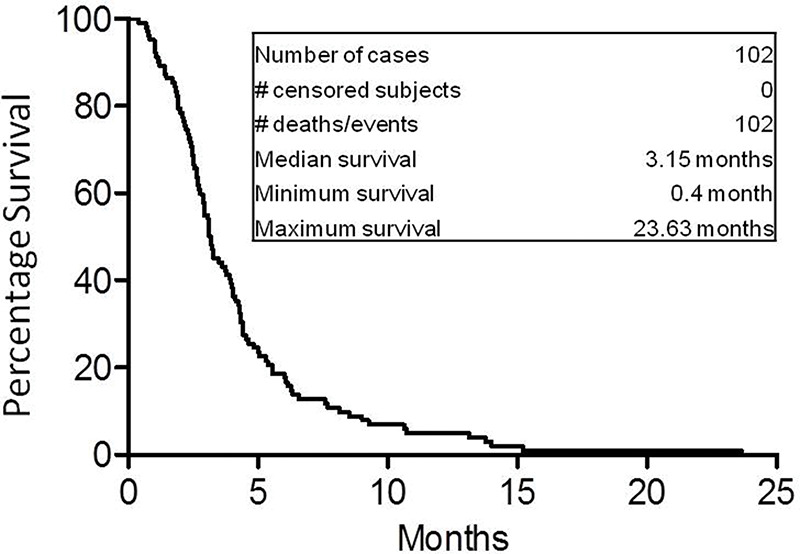
The 6-month, 1-year, and 2-year overall survival rates of the 102 patients with metastatic NSCLC were 17.65%, 3.92%, and 0.98%, respectively. The median overall survival of the 102 patients was 3.15 months (Figure 2), which was consistent with that in previous studies, which reported a median overall survival of 3-5 months (24-26). These results provide greatly important basic survival data to researchers evaluating new treatment strategies for patients with recurrent stage III/IV NSCLC who show progression on multiple conventional treatments.
Figure 2. Overall survival (OS) curve of all 102 patients with advanced stage III/IV NSCLC. The 6-month, 1-year, and 2-year OS rates were 17.65%, 3.92%, and 0.98%, respectively. The median OS of all 102 studied patients was 3.15 months.
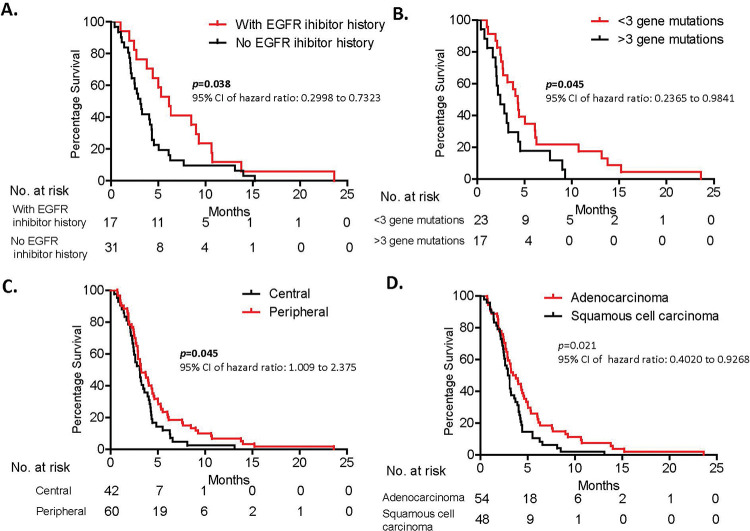
Correlation of EGFR inhibitor history with prolonged survival
Forty-eight of the 102 patients had undergone EGFR mutation evaluation, among whom 17 patients were found to have an EGFR mutation and EGFR inhibitor treatment history and 31 had no EGFR mutation or EGFR inhibitor treatment history. Detailed gene mutation signatures of each patient are shown in Supplemental Tables 2 and 3. The median overall survival time of patients with and without an EGFR inhibitor history were 6.13 months and 3.10 months, respectively (=0.038; 95% confidence interval [CI], 0.2998-0.9667) (Figure 3A, Table 2). This result suggests that EGFR inhibitor history was associated with a longer survival time than was no EGFR inhibitor history in patients. This leads to an extremely interesting question: Do EGFR inhibitors affect patient tumor progression and regression fate? Meanwhile, this result was based on a relatively small population of patients, and larger studies are needed to further confirm this finding.
Figure 3. A. Patients with an EGFR inhibitor history showed better survival than did patients without an EGFR inhibitor history. B. Patients with a higher tumor mutation load survived for shorter periods than did patients with a lower tumor mutation burden. C. Patients with a tumor in the peripheral lung lived for significantly longer than did patients with a tumor in the central lung. D. There is statistical difference in overall survival between patients with squamous cell carcinoma and those with adenocarcinoma. Survival analysis was conducted using the Kaplan-Meier method.
Table 2. Factors associated with overall survival in 102 patients with advanced NSCLC according to univariate analysis.
| Characteristic | Median or no. of cases | Median survival time (months)<median vs. ≥median(or left vs. right) | p-value | Chi-square | 95% CI of hazard ratio |
|---|---|---|---|---|---|
| Sex (male-female) | 57-45 | 3.07 vs. 4.23 | 0.0305 | 4.683 | 1.042 to 2.309 |
| Age (year) | 67 | 3.10 vs. 3.27 | 0.5676 | 0.3266 | 0.5887 to 1.337 |
| Weight (kg) | 162 | 3.27 vs. 3.10 | 0.6156 | 0.2520 | 0.6066 to 1.345 |
| Height (cm) | 170 | 3.97 vs. 2.92 | 0.1085 | 2.577 | 0.4759 to 1.077 |
| Smoke history (yes-no) | 71-31 | 3.2 0 vs. 3.10 | 0.7461 | 0.1048 | 0.6018 to 1.439 |
| Pleural effusion (yes-no) | 55-47 | 3.07 vs. 3.27 | 0.6862 | 0.1633 | 0.7309 to 1.610 |
| Tumor size (cm) | 4.35 | 2.93 vs. 3.23 | 0.5509 | 0.3557 | 0.5904 to 1.325 |
| Tumor site (central-peripheral) | 42-60 | 3.08 vs. 3.25 | 0.0451 | 4.013 | 1.009 to 2.375 |
| CEA (ng/mL) | 43.79 | 3.20 vs. 3.09 | 0.2187 | 1.513 | 0.5124 to 1.165 |
| CA125 (u/mL) | 65.99 | 2.85 vs. 3.95 | 0.8087 | 0.0586 | 0.7034 to 1.570 |
| CA153 (u/mL) | 34.39 | 3.47 vs. 3.02 | 0.4449 | 0.5835 | 0.5745 to 1.276 |
| EGFR mutation (yes-no) | 17-31* | 6.13 vs. 3.10 | 0.0381 | 4.298 | 0.2998 to 0.9667 |
| Number of tumor somatic mutations | 3** | 4.27 vs. 2.43 | 0.0451 | 4.017 | 0.2365 to 0.9841 |
| Previous treatments | |||||
| Surgery (yes-no) | 7-95 | 3.10 vs. 3.20 | 0.4324 | 0.6165 | 0.5879 to 3.459 |
| Local radiotherapy (yes-no) | 89-13 | 3.20 vs. 3.10 | 0.7088 | 0.1394 | 0.4807 to 1.646 |
| Chemotherapy (yes-no) | 73-29 | 3.20 vs. 2.93 | 0.5201 | 0.4137 | 0.7518 to 1.758 |
| EGFR inhibitor (yes-no) | 17-31* | 6.13 vs. 3.10 | 0.0381 | 4.298 | 0.2998 to 0.9667 |
| Chemotherapy cycles | 3 | 3.60 vs. 3.10 | 0.3435 | 0.8973 | 0.5440 to 1.236 |
| Lymphocyte (%) | 19.6 | 2.87 vs. 4.01 | 0.0894 | 2.885 | 0.9484 to 2.097 |
| Brain metastases (yes-no) | 11-91 | 2.87 vs. 3.20 | 0.9912 | 0.0001 | 0.5281 to 1.880 |
| Bone metastases (yes-no) | 35-67 | 3.10 vs. 3.20 | 0.8146 | 0.0550 | 0.6287 to 1.440 |
| Metastases (visceral-bone) | 17-35 | 2.93 vs. 3.10 | 0.6422 | 0.3714 | 0.6540 to 2.243 |
| Disease stage (III-IV) | 38-64 | 3.20 vs. 3.10 | 0.6973 | 0.2791 | 0.7367 to 1.701 |
| Tumor histology (SQ-AD)* | 48-54 | 3.00 vs. 3.62 | 0.0205 | 5.367 | 0.4020 to 0.9268 |
| ECOG PS (0/1/2-3) | 31-71 | 4.58 vs. 2.77 | 0.0003 | 13.05 | 0.3125 to 0.7082 |
48 of 102 underwent EGFR mutation analysis;
40 of 102 patients underwent 508 gene panel analysis (23). SQ, squamous cell carcinoma; AD, adenocarcinoma
Higher somatic mutation load is associated with worse overall survival
We were able to collect tumor tissues from 40 of 102 patients and performed the 508 gene panel next-generation DNA sequencing analysis. A range of 0 to 9 mutations was detected in 40 patients (Supplemental Table 2). Univariate analysis of these 40 patients showed that >3 somatic mutations was associated with a shorter overall survival than with <3 somatic mutations (4.27 months vs. 2.43 months, p=0.045; 95% CI, 0.2365-0.9841) (Figure 3B, Table 2). This result implies that tumor cell mutation load impacts tumor cell growth, migration, and progression through possibly multiple unknown signaling pathways. Further mechanistic studies are necessary to better understand lung cancer progression.
Potential correlation of demographic characteristics with patient prognoses
In addition to EGFR inhibitor history and tumor somatic mutation burden, we also found that tumor location within the lung and patient performance status score were associated with survival outcome. Patients with tumors in the central lung had extended survival compared with patients with tumors in the peripheral lung (3.25 months vs. 3.08 months, p=0.045; 95% CI, 1.009-2.375) (Figure 3C, Table 2). A patient performance status score-ECOG performance status of 0-2 was correlated with better survival than with an ECOG PS of 3 (4.58 months vs. 2.77 months, p=0.0003; 95% CI, 0.3125-0.7082 ) (Table 2). Furthermore, patients with adenocarcinoma and EGFR mutations showed a longer overall survival than did patients with squamous cell carcinoma and without EGFR mutation (p=0.021 and p=0.038, respectively). Further multivariate analysis showed that tumor pathology, ECOG performance status, and tumor site were independent factors associated with the overall survival of these patients (p=0.012, p=0.019, p=0.035, respectively). Curiously, clinical factors including pleural effusion; tumor size; tumor biomarkers CEA, CA125, and CA153; chemotherapy cycles; and brain metastases did not significantly affect survival in these patients and were therefore not considered risk factors (p=0.686, p=0.5509, p=0.219, p=0.809, p=0.445, p=0.344, and p=0.991, respectively; Figure 3D, Table 2). Our findings suggest that these clinical factors may not be correlated with survival in this terminal disease stage. However, these results are based on a small population of cases and particular background of patients and need to be further confirmed with extended studies of a larger size.
DISCUSSION
Recently, much progress has been made toward advancing lung cancer treatments, such as EGFR mutation-based targeted therapies (27). However, the prognoses of late-stage lung cancer remain poor, as almost all patients eventually develop drug resistance and disease progression within one or two years (28-29). This is particularly important in Asia, as approximately 40% of patients with NSCLC carry EGFR mutations (23). What causes the difference between EGFR mutant and non-mutant tumors and how the tumor biology changes after EGFR inhibitor treatment failure remain largely unknown. Our group, along with many others, has previously shown that patients with EGFR-positive NSCLC have a worse overall survival compared with patients with EGFR-negative NSCLC (23,30-32). This result suggests that EGFR mutant tumor cells may be activated in multiple tumor proliferation and migration pathways that are associated with EGFR signaling. Therefore, blocking EGFR signaling has the potential to reverse or slow tumor progression.
Here, we report that patients with advanced stage III/IV NSCLC and an EGFR inhibitor treatment history have a significantly longer overall survival than do patients without an EGFR inhibitor treatment history (p=0.038). We have shown for the first time that EGFR inhibitor treatment may benefit even patients with NSCLC undergoing hospice care only, with a better overall survival. Another interpretation from our findings could be that patients with different EGFR statuses have distinct tumor biology. EGFR acts as a cytoplasmic kinase and mediates many/several important growth factors, signaling both extracellularly and intracellularly. We hypothesize that EGFR inhibitor treatment eliminates EGFR mutant cells while sparing non-mutant cells. Compared with EGFR mutant cells, these non-mutant cells proliferate at a much slower rate and, as a consequence, contribute to longer survival in patients previously treated with EGFR inhibitors. However, this result was based on a small sample size, and large-scale studies are needed to confirm our findings.
The correlation of survival and somatic mutation load has been reported in several different cancer types (33-35). High somatic mutation is correlated with decreased progression-free survival in multiple myeloma and tumor progression in melanoma (33-34). Interestingly, tumor mutation load is significantly correlated with the clinical outcome of anti-CTLA4 antibody and adoptive T-cell treatment in melanoma. The clinical benefit of anti-PD-1 treatment in lung cancer is also found to be strongly associated with tumor mutation load status (35). These results suggest that tumor cells with high mutation load are more aggressive in nature but are simultaneously more sensitive to immunotherapy. Consistent with these findings, we showed that patients with metastatic stage III/IV NSCLC and a higher tumor mutation load survived for shorter periods than did patients with metastatic stage III/IV NSCLC with a lower tumor mutation load (p=0.045). It is noteworthy that these patients were not under any antineoplastic treatment, and the result reflects the actual effect of tumor mutation load on overall survival.
Additionally, we analyzed other clinical factors that may be associated with the survival of patients with advanced stage III/IV NSCLC who were under hospice care only. Tumor site and performance status score were significantly correlated with patient survival, whereas tumor size; pleural effusion; tumor biomarkers CEA, CA125, and CA153; chemotherapy cycles; and brain metastases were not. Patients with tumors that initiated in or were localized to the peripheral lung lived longer than did those with tumors in the central lung. The reason for this is that tumors developing in the central lung are more likely to block the main airway and consequently lead to death. We also measured the overall survival of all patients, showing that the median overall survival was 3.15 months. The 6-month, 1-year, and 2-year overall survival rates were 17.65%, 3.92%, and 0.98%, respectively. These data establish a primary survival baseline for developing new treatment strategies in future studies.
Taken together, this study reveals that EGFR inhibitor history and tumor mutation load may influence the prognosis of patients even at the terminal disease stage. While the changes in tumor biology after EGFR inhibitor treatment and how immunogenic tumors continue to progress after treatment remain unknown, ongoing research is crucial to better understand the role of the EGFR signaling pathway and tumor mutation load in lung tumor cell fate determination and, in turn, help determine appropriate treatment modulation in patients with advanced lung cancer.
AUTHOR CONTRIBUTIONS
Lan Y was responsible for the study design. Zhou S was responsible for the data collection and analysis. Feng W was responsible for the data collection. Qiao Y was responsible for the study design and project supervision. Du X was responsible for the study design and data analysis. Li F was responsible for the manuscript writing and data analysis.
ACKNOWLEDGMENTS
This work was supported by the Tianjin Beichen Hospital.
APPENDIX.
Supplemental Table 1. Multivariate analysis showed independent factors associated with the overall survival rates of 102 patients with NSCLC.
| Variables | SE | p-value | Exp (β) | 95% CI for Exp (β) |
|---|---|---|---|---|
| Age:>67 years vs. ≤67 years | 0.570 | 0.710 | 0.809 | 0.265 to 2.472 |
| Sex: female vs. male | 0.865 | 0.446 | 1.933 | 0.355 to 10.528 |
| Height: >170 cm vs. ≤170 cm | 0.705 | 0.778 | 0.820 | 0.206 to 3.261 |
| Tumor size: >4.35 cm vs. ≤5.35 cm | 0.558 | 0.312 | 0.569 | 0.191 to 1.699 |
| CEA: >43.79 ng/mL vs. ≤43.79 ng/mL | 0.566 | 0.030 | 3.424 | 1.130 to 10.380 |
| CA153: >34.39 u/mL vs. ≤34.39 u/mL | 0.496 | 0.591 | 1.305 | 0.494 to 3.447 |
| EGFR inhibitor history: yes vs. no | 0.933 | 0.794 | 0.783 | 0.126 to 4.878 |
| EGFR mutation: yes vs. no | 0.546 | 0.051 | 2.897 | 0.994 to 8.441 |
| Lymphocyte: >19.6% vs. ≤19.6% | 0.495 | 0.908 | 1.059 | 0.401 to 2.796 |
| Tumor pathology: SQ vs. AD* | 0.793 | 0.012 | 7.275 | 1.536 to 34.445 |
| ECOG PS: 0/1/2 vs. 3 | 0.652 | 0.019 | 4.626 | 1.289 to 16.602 |
| Tumor site: central vs. peripheral | 0.907 | 0.035 | 6.796 | 1.148 to 40.223 |
| Chemotherapy cycles: ≥3 vs. <3 | 0.694 | 0.176 | 0.391 | 0.101 to 1.524 |
| Chemotherapy: yes vs. no | 0.797 | 0.066 | 4.327 | 0.908 to 20.628 |
| Number of tumor somatic mutations: ≥3 vs. <3 | 0.724 | 0.077 | 3.591 | 0.870 to 14.829 |
SQ, squamous cell carcinoma; AD, adenocarcinoma.
Bold values indicate significant difference.
Supplemental Table 2. Gene mutation results of 40 patients according to 508 gene panel analysis.
| Patient ID | No. of gene mutation | Gene | Base mutation | Amino acids mutation |
|---|---|---|---|---|
| Pt. 12 | 1 | TSC2 | c.4519T>C | p.S1507P |
| Pt. 15 | 3 | EGFR | c.2573T>G | p.L858R |
| TP53 | c.859G>T | p.E287* | ||
| CDK4 | c.71G>T | p.R24L | ||
| EGFR | / | 1.7copy number again | ||
| Pt. 19 | 9 | TP53 | c.337T>G | p.F113V |
| RB1 | c.1450_1451del | p.M484Vfs*8 | ||
| CD22 | c.A139A>G | p.R47G | ||
| PIK3C2A | c.1201C>T | p.R401C | ||
| EPHA5 | c.2767C>A | p.P923T | ||
| NEK11 | c.25A>G | p.K9E | ||
| STAG2 | c.3446A>T | p.H1149L | ||
| EPHA5 | c.688T>G | p.Y230D | ||
| ATM | c.4966A>G | p.K1656E | ||
| Pt. 21 | 1 | ERBB2 | c.338G>A | p.R113Q |
| Pt. 22 | 1 | EGFR | c.2573T>G | p.L858R |
| Pt. 23 | 6 | RAD52 | c.1037C>A | p.S346X |
| CREBBP | c.1651C>A | p.L551I | ||
| TEK | c.2029C>A | p.Q677K | ||
| PIK3C3 | c.718G>A | p.G240S | ||
| MPL | c.1120A>G | p.T374A | ||
| PIK3C2G | c.3299_3306dup | p.Y1103Vfs*15 | ||
| Pt. 25 | 2 | TP53 | c.469G>T | p.V157F |
| CDKN2A | c.250G>A | p.D84N | ||
| Pt. 26 | 7 | NOTCH2 | c.2548G>A | p.E850K |
| APC | c.4666dup | p.T1556Nfs*3 | ||
| TP53 | c.532del | p.H178Tfs*69 | ||
| MS4A1 | c.161C>A | p.A54D | ||
| KEAP1 | c.1337A>T | p.E446V | ||
| IGF1R | c.758A>G | p.H253R | ||
| MLH3 | c.2032A>G | p.N678D | ||
| Pt. 28 | 1 | EGFR | c.2235_2249del | p.745_750del |
| Pt. 31 | 9 | PTCH1 | c.C3853>T | p.Q1285X |
| KMT2D | c.10636C >T | p.Q3546X | ||
| CARD11 | c.3179G>A | p.C1060Y | ||
| EXT1 | c.1813C>T | p.R605W | ||
| TRAF7 | c.694C>T | p.R232W | ||
| DNMT3A | c.1574C>T | p.A525V | ||
| CCND3 | c.391G>A | p.A131T | ||
| ERBB2 | c.1830G>T | p.K610N | ||
| TAF1 | c.3650G>A | p.R1217H | ||
| Pt. 35 | 2 | CHUK | c.464T>C | p.V155A |
| PTCH1 | c.2225G>A | p.R742H | ||
| Pt. 39 | 1 | TP53 | c.527G>T | p.C176F |
| Pt. 42 | 2 | KRAS | c.34G>A | p.G12S |
| TP53 | c.775G>T | p.D259Y | ||
| Pt. 44 | 9 | ABL2 | c.2948C>T | p.A983V |
| BRCA2 | c.8187G>T | p.K2729N | ||
| DOCK2 | c.4768A>G | p.R1590G | ||
| ERBB2 | c.380G>A | p.R127Q | ||
| Pt. 44 | 7 | EGFR | c.2573T>G | p.L858R |
| TP53 | c.584T>C | p.I195T | ||
| CDKN2A | c.315C>A | p.D105E | ||
| NTRK1 | c.2026C>T | p.R676C | ||
| ALK | c.3311C>T | p.S1104F | ||
| FGFR2 | c.160C>T | p.P54S | ||
| EGFR | / | 1.7copy number again | ||
| Pt. 47 | 2 | EGFR | c.2237_2254del | p.E746_S752delinsA |
| EGFR | c.2255C>T | p.S752F | ||
| BRCA1 | c.718C>T | p.Q240X | ||
| Pt. 50 | 5 | AKT1 | c.607C>T | p.Q203X |
| BRAF | c.1568C>T | p.P523L | ||
| FGFR1 | c.748C>T | p.R250W | ||
| NF1 | c.4998G>C | p.R1666S | ||
| GOPC | c.1151G>A | p.R384H | ||
| Pt. 53 | 7 | EGFR | c.2573T>G | p.L858R |
| KMT2C | c.817delG | p.V273Wfs*82 | ||
| NFE2L3 | c.1826_1829del | p.C611Tfs*22 | ||
| SMC1A | c.3103C>T | p.R1035X | ||
| FAT3 | c.12637C>T | p.R4213C | ||
| SMAD4 | c.608C>T | p.P203L | ||
| PRKCG | c.614G>A | p.R205Q | ||
| Pt. 63 | 3 | EGFR | c.2573T>G | p.L858R |
| KRAS | c.35G>A | p.G12D | ||
| TSC2 | c.4594C>T | p.Q1532X | ||
| Pt. 64 | 2 | STK11 | c.1062C>G | p.F354L |
| TP53 | c.430C>T | p.Q144X | ||
| Pt. 65 | 6 | NTRK1 | c.865C>T | p.Q289X |
| PMS2 | c.80G>T | p.C27F | ||
| ATM | c.7358G>C | p.R2453P | ||
| SMAD4 | c.608C>T | p.P203L | ||
| TSC1 | c.2696C>G | p.T899S | ||
| PTEN | c.235G>A | p.A79T | ||
| Pt. 66 | 1 | TP53 | c.524G>A | p.R175H |
| Pt. 67 | 2 | EGFR | c.2573T>G | p.L858R |
| TP53 | c.430C>T | p.Q144X | ||
| Pt. 68 | 4 | DDR2 | c.1267C>T | p.Q423X |
| MET | c.1039G>A | p.A347T | ||
| PTCH1 | c.4144C>T | p.H1382Y | ||
| HER2 | c.2579C>T | p.A860V | ||
| Pt. 69 | 5 | EGFR | c.2232_2233ins | p.I744_K745insKIP VAI |
| MSH3 | c.1180C>T | p.Q394X | ||
| AR | c.1369_1377del | p.G457_G459del | ||
| PTCH1 | c.1628G>A | p.R543H | ||
| TSC2 | c.2023G>A | p.A675T | ||
| Pt. 70 | 2 | PIK3CA | c.1633G>A | p.E545K |
| TP53 | c.524G>A | p.R175H | ||
| TP53 | c.1006G>T | p.E336X | ||
| Pt. 71 | 2 | EGFR | c.2156G>C | p.G719A |
| TP53 | c.661G>T | p.E221X | ||
| TP53 | c.745A>T | p.R249W | ||
| Pt. 72 | 1 | TP53 | c.524G>A | p.R175H |
| Pt. 73 | 1 | EGFR | c.2237_2251delinsT AG | p.E746_T751delins VA |
| Pt. 74 | 5 | SMARCA4 | c.1189C>T | p.R397X |
| DPYD | c.1850C>T | p.T617M | ||
| XPO1 | c.740T>C | p.I247T | ||
| PRKAA1 | c.1382G>A | p.R461Q | ||
| SMC1A | c.1990C>T | p.R664W | ||
| Pt. 75 | 2 | EGFR | c.2235_2249del | p.745_750del |
| TP53 | c.724T>A | p.C242S | ||
| Pt. 76 | 5 | U2AF1 | c.101C>T | p.S34F |
| CD22 | c.2182C>T | p.Q728X | ||
| SETD2 | c.1814A>C | p.K605T | ||
| TNFAIP3 | c.1344G>C | p.W448C | ||
| CARD11 | c.157G>A | p.D53N | ||
| Pt. 77 | 3 | MLH1 | c.1192C>T | p.Q398X |
| AR | c.2257C>T | p.R753X | ||
| TSC2 | c.5051_5068del | p.1684_1690del | ||
| Pt. 78 | 2 | EGFR | c.2575G>A | p.A859T |
| TP53 | c.524G>A | p.R175H | ||
| Pt. 79 | 2 | NF1 | c.2023G>T | p.G675X |
| TP53 | c.532delC | p.H178fs | ||
| Pt. 80 | 2 | EGFR | c.2573T>G | p.L858R |
| EGFR | c.2240T>C | p.L747S | ||
| TP53 | c.832C>T | p.P278S | ||
| Pt. 81 | 3 | ERBB2 | Copy number gain | |
| TP53 | c.245_246insCACC | p.P82fs | ||
| MET | c.3520G>T | p.V1174F | ||
| Pt. 82 | 0 | None | ||
| Pt. 83 | 2 | TP53 | c.536A>G | p.H179R |
| TSC2 | c.2023G>A | p.A675T | ||
| Pt. 84 | 2 | U2AF1 | c.101C>T | p.S34F |
| TP53 | c.994-1G>T |
Supplemental Table 3. EGFR mutation results of eight patients who had undergone single EGFR gene analysis.
| Patient ID | EGFR status |
|---|---|
| Pt. 6 | No EGFR mutation |
| Pt. 11 | No EGFR mutation |
| Pt. 24 | EGFR Exon21 p.L858R |
| Pt. 27 | EGFR Exon 19 c.2235_2249del p.746_750del 7.17% |
| Pt. 41 | No EGFR mutation |
| Pt. 48 | No EGFR mutation |
| Pt. 60 | EGFR Exon21 p.L858R |
| Pt. 34 | No EGFR mutation |
Footnotes
No potential conflict of interest was reported.
REFERENCES
- 1.Torre LA, Siegel RL, Jemal A. Lung Cancer Statistics. Adv Exp Med Biol. 2016;893:1–19. doi: 10.1007/978-3-319-24223-1. [DOI] [PubMed] [Google Scholar]
- 2.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2017. CA Cancer J Clin. 2017;67(1):7–30. doi: 10.3322/caac.21387. [DOI] [PubMed] [Google Scholar]
- 3.Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D. Global cancer statistics. CA Cancer J Clin. 2011;61(2):69–90. doi: 10.3322/caac.20107. [DOI] [PubMed] [Google Scholar]
- 4.Reddy C, Chilla D, Boltax J. Lung cancer screening: a review of available data and current guidelines. Hosp Pract (1995) 2011;39(4):107–12. doi: 10.3810/hp.2011.10.929. [DOI] [PubMed] [Google Scholar]
- 5.Rosell R, Carcereny E, Gervais R, Vergnenegre A, Massuti B, Felip E, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol. 2012;13(3):239–46. doi: 10.1016/S1470-2045(11)70393-X. [DOI] [PubMed] [Google Scholar]
- 6.Maemondo M, Inoue A, Kobayashi K, Sugawara S, Oizumi S, Isobe H, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med. 2010;362(25):2380–8. doi: 10.1056/NEJMoa0909530. [DOI] [PubMed] [Google Scholar]
- 7.Sequist LV, Yang JC, Yamamoto N, O'Byrne K, Hirsh V, Mok T, et al. Phase III study of afatinib or cisplatin plus pemetrexed in patients with metastatic lung adenocarcinoma with EGFR mutations. J Clin Oncol. 2013;31(27):3327–34. doi: 10.1200/JCO.2012.44.2806. [DOI] [PubMed] [Google Scholar]
- 8.Paz-Ares L, Tan EH, O'Byrne K, Zhang L, Hirsh V, Boyer M, et al. Afatinib versus gefitinib in patients with EGFR mutation-positive advanced non-small-cell lung cancer: overall survival data from the phase IIb LUX-Lung 7 trial. Ann Oncol. 2017;28(2):270–7. doi: 10.1093/annonc/mdw611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wu YL, Cheng Y, Zhou X, Lee KH, Nakagawa K, Niho S, et al. Dacomitinib versus gefitinib as first-line treatment for patients with EGFR-mutation-positive non-small-cell lung cancer (ARCHER 1050): a randomised, open-label, phase 3 trial. Lancet Oncol. 2017;18(11):1454–66. doi: 10.1016/S1470-2045(17)30608-3. [DOI] [PubMed] [Google Scholar]
- 10.Solomon BJ, Mok T, Kim DW, Wu YL, Nakagawa K, Mekhail T, et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N Engl J Med. 2014;371(23):2167–77. doi: 10.1056/NEJMoa1408440. [DOI] [PubMed] [Google Scholar]
- 11.Peters S, Camidge DR, Shaw AT, Gadgeel S, Ahn JS, Kim DW, et al. Alectinib versus Crizotinib in Untreated ALK-Positive Non-Small-Cell Lung Cancer. N Engl J Med. 2017;377(9):829–38. doi: 10.1056/NEJMoa1704795. [DOI] [PubMed] [Google Scholar]
- 12.Shaw AT, Kim TM, Crinò L, Gridelli C, Kiura K, Liu G, et al. Ceritinib versus chemotherapy in patients with ALK-rearranged non-small-cell lung cancer previously given chemotherapy and crizotinib (ASCEND-5): a randomised, controlled, open-label, phase 3 trial. Lancet Oncol. 2017;18(7):874–86. doi: 10.1016/S1470-2045(17)30339-X. [DOI] [PubMed] [Google Scholar]
- 13.Kim DW, Tiseo M, Ahn MJ, Reckamp KL, Hansen KH, Kim SW, et al. Brigatinib in Patients With Crizotinib-Refractory Anaplastic Lymphoma Kinase-Positive Non-Small-Cell Lung Cancer: A Randomized, Multicenter Phase II Trial. J Clin Oncol. 2017;35(22):2490–8. doi: 10.1200/JCO.2016.71.5904. [DOI] [PubMed] [Google Scholar]
- 14.Shaw AT, Bauer TM, Felip E, Besse B, James LP, Clancy JS, et al. Clinical activity and safety of PF-06463922 from a dose escalation study in patients with advanced ALK+ or ROS1+NSCLC. J Clin Oncol. 2015;33(15 Suppl):8018. [Google Scholar]
- 15.Shaw AT, Ou SH, Bang YJ, Camidge DR, Solomon BJ, Salgia R, et al. Crizotinib in ROS1-rearranged non-small-cell lung cancer. N Engl J Med. 2014;371(21):1963–71. doi: 10.1056/NEJMoa1406766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Santarpia M, Gil N, Rosell R. Strategies to overcome resistance to tyrosine kinase inhibitors in non-small-cell lung cancer. Expert Rev Clin Pharmacol. 2015;8(4):461–77. doi: 10.1586/17512433.2015.1055252. [DOI] [PubMed] [Google Scholar]
- 17.Mok TS, Wu YL, Ahn MJ, Garassino MC, Kim HR, Ramalingam SS, et al. Osimertinib or Platinum-Pemetrexed in EGFR T790M-Positive Lung Cancer. N Engl J Med. 2017;376(7):629–40. doi: 10.1056/NEJMoa1612674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ou Q, Wu X, Bao H, Tong X, Wang X, Zhang X, et al. Investigating novel resistance mechanisms to third generation EGFR TKI osimertinib in non-small cell lung cancer patients using next generation sequencing. J Clin Oncol. 2017;35(15 Suppl):2572. doi: 10.1200/JCO.2017.35.15_suppl.2572. [DOI] [Google Scholar]
- 19.Becker K, Xu Y. Management of tyrosine kinase inhibitor resistance in lung cancer with EGFR mutation. World J Clin Oncol. 2014;5(4):560–7. doi: 10.5306/wjco.v5.i4.560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Riely GJ, Kris MG, Zhao B, Akhurst T, Milton DT, Moore E, et al. Prospective assessment of discontinuation and reinitiation of erlotinib or gefitinib in patients with acquired resistance to erlotinib or gefitinib followed by the addition of everolimus. Clin Cancer Res. 2007;13(17):5150–5. doi: 10.1158/1078-0432.CCR-07-0560. [DOI] [PubMed] [Google Scholar]
- 21.Chaft JE, Oxnard GR, Sima CS, Kris MG, Miller VA, Riely GJ. Disease flare after tyrosine kinase inhibitor discontinuation in patients with EGFR-mutant lung cancer and acquired resistance to erlotinib or gefitinib: implications for clinical trial design. Clin Cancer Res. 2011;17(19):6298–303. doi: 10.1158/1078-0432.CCR-11-1468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bhattacharya P, Dessain SK, Evans TL. Palliative Care in Lung Cancer: When to Start. Curr Oncol Rep. 2018;20(11):90. doi: 10.1007/s11912-018-0731-9. [DOI] [PubMed] [Google Scholar]
- 23.Li F, Du X, Zhang H, Ju T, Chen C, Qu Q, et al. Next-generation sequencing of Chinese stage IV lung cancer patients reveals an association between EGFR mutation status and survival outcome. Clin Genet. 2017;91(3):488–93. doi: 10.1111/cge.12809. [DOI] [PubMed] [Google Scholar]
- 24.Vergnenàgre A, Hominal S, Tchalla AE, Bérard H, Monnet I, Fraboulet G, et al. Assessment of palliative care for advanced non-small-cell lung cancer in France: a prospective observational multicenter study (GFPC 0804 study) Lung Cancer. 2013;82(2):353–7. doi: 10.1016/j.lungcan.2013.07.014. [DOI] [PubMed] [Google Scholar]
- 25.Duggan KT, Hildebrand Duffus S, D'Agostino RB, Jr, Petty WJ, Streer NP, Stephenson RC. The Impact of Hospice Services in the Care of Patients with Advanced Stage Nonsmall Cell Lung Cancer. J Palliat Med. 2017;20(1):29–34. doi: 10.1089/jpm.2016.0064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Duggan KJ, Wiltshire J, Strutt R, Boxer MM, Berthelsen A, Descallar J, et al. Palliative care and psychosocial care in metastatic non-small cell lung cancer: factors affecting utilisation of services and impact on patient survival. Support Care Cancer. 2019;27(3):911–9. doi: 10.1007/s00520-018-4379-7. [DOI] [PubMed] [Google Scholar]
- 27.Forde PM, Ettinger DS. Targeted therapy for non-small-cell lung cancer: past, present and future. Expert Rev Anticancer Ther. 2013;13(6):745–58. doi: 10.1586/era.13.47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Jackman D, Pao W, Riely GJ, Engelman JA, Kris MG, Jänne PA, et al. Clinical definition of acquired resistance to epidermal growth factor receptor tyrosine kinase inhibitors in non-small-cell lung cancer. J Clin Oncol. 2010;28(2):357–60. doi: 10.1200/JCO.2009.24.7049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Camidge DR, Pao W, Sequist LV. Acquired resistance to TKIs in solid tumours: learning from lung cancer. Nat Rev Clin Oncol. 2014;11(8):473–81. doi: 10.1038/nrclinonc.2014.104. [DOI] [PubMed] [Google Scholar]
- 30.Scagliotti GV, Selvaggi G, Novello S, Hirsch FR. The biology of epidermal growth factor receptor in lung cancer. Clin Cancer Res. 2004;10(12 Pt 2):4227s–4232s. doi: 10.1158/1078-0432.CCR-040007. [DOI] [PubMed] [Google Scholar]
- 31.Veale D, Kerr N, Gibson GJ, Kelly PJ, Harris AL. The relationship of quantitative epidermal growth factor receptor expression in non-small cell lung cancer to long term survival. Br J Cancer. 1993;68(1):162–5. doi: 10.1038/bjc.1993.306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Volm M, Rittgen W, Drings P. Prognostic value of ERBB-1, VEGF, cyclin A, FOS, JUN and MYC in patients with squamous cell lung carcinomas. Br J Cancer. 1998;77(4):663–9. doi: 10.1038/bjc.1998.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Miller A, Asmann Y, Cattaneo L, Braggio E, Keats J, Auclair D, et al. High somatic mutation and neoantigen burden are correlated with decreased progression-free survival in multiple myeloma. Blood Cancer J. 2017;7(9):e612. doi: 10.1038/bcj.2017.94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tiffen J, Wilson S, Gallagher SJ, Hersey P, Filipp FV. Somatic Copy Number Amplification and Hyperactivating Somatic Mutations of EZH2 Correlate With DNA Methylation and Drive Epigenetic Silencing of Genes Involved in Tumor Suppression and Immune Responses in Melanoma. Neoplasia. 2016;18(2):121–32. doi: 10.1016/j.neo.2016.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Roszik J, Haydu LE, Hess KR, Oba J, Joon AY, Siroy AE, et al. Novel algorithmic approach predicts tumor mutation load and correlates with immunotherapy clinical outcomes using a defined gene mutation set. BMC Med. 2016;14(1):168. doi: 10.1186/s12916-016-0705-4. [DOI] [PMC free article] [PubMed] [Google Scholar]