Abstract
Background
Quality of care incentives and reimbursements for cardiovascular testing differ between insurance providers. We hypothesized that there are differences in the use of guideline‐concordant testing between Medicaid versus commercial insurance patients <65 years, and between Medicare Advantage versus Medicare fee‐for‐service patients ≥65 years.
Methods and Results
Using data from the Colorado All‐Payer Claims Database from 2015 to 2018, we identified patients eligible to receive a high‐value test recommended by guidelines: assessment of left ventricular function among patients hospitalized with acute myocardial infarction or incident heart failure, or a low‐value test that provides minimal patient benefit: stress testing prior to low‐risk surgery or routine stress testing within 2 years of percutaneous coronary intervention or coronary artery bypass graft surgery. Among 145 616 eligible patients, 37% had fee‐for‐service Medicare, 18% Medicare Advantage, 22% Medicaid, and 23% commercial insurance. Using multilevel logistic regression models adjusted for patient characteristics, Medicaid patients were less likely to receive high‐value testing for acute myocardial infarction (odds ratio [OR], 0.84 [0.73–0.98]; P=0.03) and heart failure (OR, 0.59 [0.51–0.70]; P<0.01) compared with commercially insured patients. Medicare Advantage patients were more likely to receive high‐value testing for acute myocardial infarction (OR, 1.35 [1.15–1.59]; P<0.01) and less likely to receive low‐value testing after percutaneous coronary intervention/ coronary artery bypass graft (OR, 0.63 [0.55–0.72]; P<0.01) compared with Medicare fee‐for‐service patients.
Conclusions
Guideline‐concordant testing was less likely to occur among patients with Medicaid compared with commercial insurance, and more likely to occur among patients with Medicare Advantage compared with fee‐for‐service Medicare. Insurance plan features may provide valuable targets to improve guideline‐concordant testing.
Keywords: health policy, imaging, quality of care
Subject Categories: Health Services, Quality and Outcomes
Clinical Perspective
What Is New?
A patient’s health insurance provider is an independent predictor of whether they will receive diagnostic cardiovascular testing in certain clinical scenarios described in guidelines.
Compared with commercially insured patients, Medicaid patients were less likely to receive high‐value testing to assess left ventricular function when hospitalized for acute myocardial infarction or incident heart failure.
Compared with Medicare fee‐for‐service patients, Medicare Advantage patients were more likely to receive high‐value testing for acute myocardial infarction and less likely to receive low‐value stress testing after percutaneous coronary intervention/coronary artery bypass graft.
What Are the Clinical Implications?
Overuse and underuse of diagnostic tests can worsen patient outcomes and lead to high healthcare costs.
Our results suggest that reimbursement and quality incentives may affect use of guideline‐concordant testing, and that features of insurance plans could be useful targets to optimize use of testing to improve patient outcomes and reduce healthcare costs.
There is a growing emphasis on providing the right medical care to the right individuals at the right time. Diagnostic cardiovascular tests, such as echocardiography and stress tests, have received particular scrutiny because of regional variation and rapid rises in rates of testing. 1 , 2 , 3 Clinical practice guidelines explicitly define situations where diagnostic tests are considered high value because they can guide therapy and improve patient outcomes, such as assessment of left ventricular ejection fraction (LVEF) among patients hospitalized with acute myocardial infarction (AMI). 4 , 5 Other practice guidelines, such as the American Board of Internal Medicine’s Choosing Wisely program, describe tests that are low value because they are unlikely to provide patient benefit, such as routine stress testing in individuals prior to low‐risk noncardiac surgery. 6
Prior studies have shown differences in use of high‐ and low‐value diagnostic testing based on patient characteristics such as age and sex, but little is known about the impact of health insurance providers. 7 , 8 , 9 , 10 , 11 , 12 There is evidence that differences in insurance plan features could lead to differences in the overall volume and quality of healthcare services provided. 13 , 14 , 15 In particular, differences in reimbursement for testing, such as the typically lower reimbursements for testing provided by Medicaid, or incentives to provide evidence‐based care, such as quality incentives provided by Medicare Advantage plans, could lead to significant differences in high‐ and low‐value testing. Therefore, understanding the associations between insurance provider and guideline‐concordant testing is important to identify previously unknown disparities in testing, as well as potential new opportunities to improve use of testing.
Accordingly, we used a large all‐payer administrative claims database from the state of Colorado to examine variations in use of diagnostic testing in 2 high‐value clinical scenarios: (1) assessment of LVEF after hospitalization for AMI and (2) assessment of LVEF after hospitalization for incident heart failure (HF); and 2 low‐value clinical scenarios: (1) preoperative stress testing prior to low‐risk surgery and (2) routine stress testing within 2 years after percutaneous coronary intervention (PCI) or coronary artery bypass graft surgery (CABG). Hereafter, we refer to the use of testing in these scenarios as high‐value and low‐value testing. Consistent with prior studies, we separately examined Medicaid versus commercial insurance patients <65 years, and fee‐for‐service Medicare versus Medicare Advantage patients ≥65 years. 16 , 17 We hypothesized that insurance provider would be independently associated with use of high‐ and low‐value testing after adjustment for patient characteristics.
Methods
Data Source and Study Population
We used data from the Colorado All‐Payer Claims Database, which is a comprehensive administrative data set that includes inpatient, outpatient, physician, and facility claims on nearly all patients who received care in the state. Reporting is required for all insurance companies and plans, with the exception of federal health facilities (e.g., Veterans Health Administration hospitals) and self‐insured group health plans. Thus, it is not a voluntary effort that could lead to reporting bias. The Colorado All‐Payer Claims Database also includes beneficiary demographics including age and sex, insurance carrier, and hospital identifiers, but does not include reliable data on race/ethnicity. These data are made available for purchase by researchers. Because of the sensitive nature of the data collected for this study, requests to access the data set from qualified researchers trained in human subject confidentiality protocols may be sent to the Center for Improving Value in Healthcare at info@civhc.org .
For this study, we included patients aged 18 to 95 years between 2015 and 2018 in the Colorado All‐Payer Claims Database. We excluded patients without at least 1 year of enrollment in insurance prior to an index testing event. We excluded critical access hospitals and hospitals with fewer than 40 hospitalizations for AMI or HF during the study period. International Classification of Diseases, Ninth Revision (ICD‐9) and Tenth Revision (ICD‐10) codes and Current Procedural Technology codes used are provided in Table S1: PCI, CABG surgery, and diagnostic cardiovascular tests; Table S2: low‐risk surgeries; and Table S3: hospitalizations for AMI and HF. Comorbid conditions were identified using algorithms provided by the Centers for Medicare and Medicaid Services Chronic Conditions Warehouse, 18 and were assessed based on claims filed in the year prior to eligibility for high‐ or low‐value testing. The study was approved by the University of Colorado Institutional Review Board. Because this was a retrospective study using administrative claims, it was deemed low risk and thus exempt from the need for informed consent. Dr Kini had full access to all of the data in the study and takes responsibility for their integrity and the data analysis.
Identification of High‐ and Low‐Value Cardiovascular Tests
Overview
We identified use of 2 high‐value tests, assessment of LVEF after hospitalization for AMI or incident HF, and 2 low‐value tests, preoperative stress testing prior to low‐risk surgery and routine stress testing within 2 years after coronary revascularization with PCI or CABG surgery, using the methods described below.
High‐Value Assessment of LVEF Among Patients Hospitalized With AMI
Using methods previously validated by Miller et al., 11 we identified all patients discharged with a primary diagnosis of AMI, and excluded those in whom LVEF assessment may not have been indicated according to clinical practice guidelines 19 , 20 : (1) patients who died during hospitalization or were discharged to hospice, and (2) patients readmitted with myocardial infarction within 90 days of a prior hospitalization, because readmission may have been related to the initial event and would not necessarily require another assessment (Figure 1A). If the same patient had another AMI hospitalization beyond 90 days following a prior AMI hospitalization, they were considered eligible for LVEF assessment again. To define the completion of testing, we included any test of LVEF (i.e., echocardiography, nuclear single photon emission computed tomography, left ventriculography, cardiac positron emission tomography, cardiac computed tomography angiography, or cardiac magnetic resonance imaging) billed between the day of admission for the index hospitalization for AMI and the 60 days following discharge.
Figure 1. Flow diagrams for creation of high‐value testing cohorts.
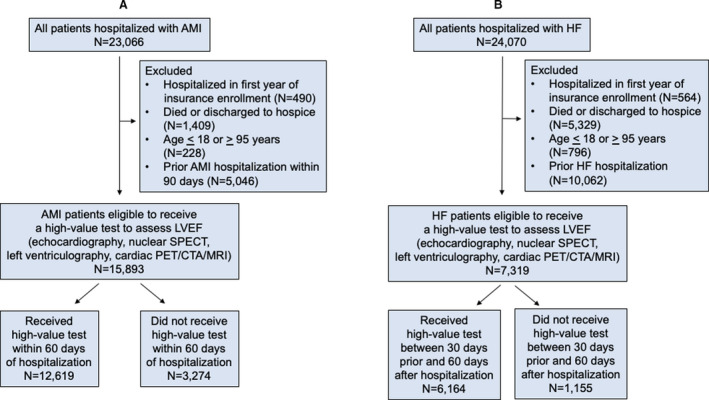
Patients hospitalized with acute myocardial infarction (AMI) (A) and heart failure (HF) (B). CTA indicates computed tomography angiography; LVEF, left ventricular ejection fraction; MRI, magnetic resonance imaging; PET, positron emission tomography; and SPECT, single photon emission computed tomography).
High‐Value Assessment of LVEF Among Patients Hospitalized With HF
Using methods similar to those previously validated by Curtis et al. 12 and Farmer et al., 10 we identified all patients discharged with a new primary diagnosis of HF (i.e., incident HF, patients without a primary diagnosis of HF on an inpatient claim or 2 outpatient claims in the 2 years prior to hospitalization). We excluded patients who died during their hospitalization or were discharged to hospice (Figure 1B). Patients were unique (i.e., could only be included in the cohort once because only patients with incident HF were considered eligible for testing). To define the completion of testing, we included any test to assess LVEF (i.e., echocardiography, nuclear single photon emission computed tomography, left heart catheterization with left ventriculography, or cardiac positron emission tomography/cardiac computed tomography angiography/magnetic resonance imaging) between 30 days prior to the day of admission of the index hospitalization for HF to capture tests that may have prompted hospitalization and 60 days after the day of discharge.
Among both the AMI and HF cohorts, we performed the following sensitivity analyses: (1) increasing the time interval for testing from 60 to 90 days following discharge, and (2) excluding 2 safety‐net hospitals that provide care for the largest proportion of Medicaid patients in the state.
Low‐Value Preoperative Stress Testing Among Patients Undergoing Low‐Risk Surgery
Among all patients undergoing low‐risk surgery (e.g., knee or shoulder arthroscopy, cataract surgery, laparoscopic cholecystectomy, inguinal hernia repair, mastectomy, hysteroscopy, or transurethral prostatectomy), we identified patients for whom stress testing was likely to be low value based on Appropriate Use Criteria and Choosing Wisely (Figure 2A). 5 , 6 We used methods previously validated by Kerr et al. 21 and Schwartz et al. 22 We treated as low value any stress test performed in the 60 days prior to the low‐risk surgery. We excluded surgeries that occurred within 1 year after a prior surgery. If the same patient had another low‐risk surgery at least 1 year after a prior surgery, they were assessed for low‐value stress testing again. We excluded tests that were billed during an acute care visit to an emergency department or during another hospitalization, as these may have been performed for appropriate clinical indications.
Figure 2. Flow diagrams for creation of low‐value testing cohorts.
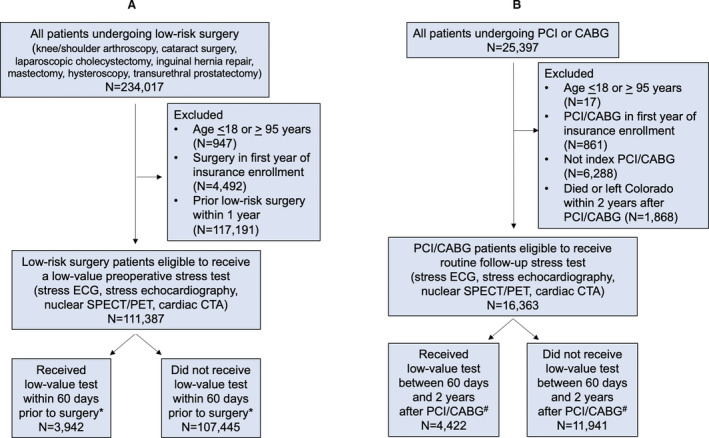
Patients undergoing low‐risk surgery (A) and coronary revascularization (B). CABG indicates coronary artery bypass graft surgery; CTA, computed tomography angiography; ECG, electrocardiography; PCI, percutaneous coronary intervention; PET, positron emission tomography; and SPECT, single photon emission computed tomography. *Tests related to acute care (emergency department visits or hospital admissions) were not considered low value. #Tests related to acute care (emergency department visits or hospital admissions) or that were followed by repeat coronary revascularization were not considered low value.
Low‐Value Routine Stress Testing Within 2 Years After PCI or CABG
For patients who underwent PCI or CABG, we identified patients for whom stress testing was likely to be low value based on Appropriate Use Criteria and Choosing Wisely (Figure 2B). 6 , 7 We used methods similar to those used by Shah et al. 23 and Bradley et al. 24 We identified all patients who underwent an initial index PCI or CABG. We excluded subsequent PCI and CABG procedures performed on the same patient (i.e., patients were assessed for low‐value stress testing only once). We identified whether patients received a cardiac stress test between 60 days 2 years after the index procedure. We excluded stress tests within the first 60 days after the index procedure because these tests may have been performed to guide staged revascularization (i.e., PCI of intermediate severity stenosis identified during the index PCI procedure). We excluded tests that were billed during an acute care visit to an emergency department or during another hospitalization, as these may have been performed for appropriate clinical indications. We did not exclude stress tests that were followed by repeat PCI or CABG, because the designation of low‐value stress testing in this scenario is not dependent on the outcome of testing. 5
Outcome Measures, Covariates, and Statistical Analysis
We calculated the rate of each high‐ and low‐value test overall and stratified by insurance type. The denominator was the number of patients eligible for each clinical scenario; the numerator was the number of patients who received the test. In the AMI and low‐risk surgery cohorts, patients could be eligible to receive testing more than once (i.e., 90 days after prior AMI hospitalization or 1 year after prior low‐risk surgery). We performed sensitivity analyses counting only the index AMI hospitalization or low‐risk surgery. We allowed for overlap between time periods of patients eligible to receive high‐ and low‐value testing in different scenarios (e.g., a patient undergoing low‐risk surgery and then hospitalized for AMI could have been counted in both low‐ and high‐value testing cohorts).
We used multivariable logistic regression models with 2‐sided P values to assess associations between high‐ and low‐value testing and insurance payer, the key independent variable. Models were multilevel to account for clustering of patients by hospital, and adjusted for a variety of patient‐level covariates. Covariates were selected for inclusion in the model based on prior studies of high‐ and low‐value care, prior studies examining differences in healthcare utilization between insurance plan types, and clinical reasoning. Patient‐level covariates included in the final models were age, sex, and the presence or absence of diabetes mellitus, hypertension, hyperlipidemia, stroke, chronic kidney disease, chronic pulmonary disease, atrial fibrillation, cancer, ischemic heart disease, and HF, except in the HF cohort where patients with a prior history of HF were excluded. We assessed for interaction between insurance type and age, sex, and selected comorbid conditions for each high‐ and low‐value test.
We separately assessed (1) Medicaid versus commercial insurance patients <65 years, and (2) Medicare fee‐for‐service versus Medicare Advantage patients ≥65 years. This approach is consistent with prior studies and was used to minimize sampling bias, because Medicaid and commercially insured patients are typically ages <65 years, whereas Medicare fee‐for‐service and Medicare Advantage patients are typically aged ≥65 years. 16 , 17 SAS (version 9.4) and Stata (version 13.1) software were used for data cleaning and statistical analysis.
Results
High‐Value Testing
Cohort Characteristics
We identified 15 893 patients eligible for high‐value testing for AMI and 7319 patients eligible for high‐value testing for HF (Table 1). For the AMI cohort, the mean age of patients was 67 years [SD, 13.2 years], and 30% were women. The proportion of patients enrolled in fee‐for‐service Medicare was 44%, in Medicare Advantage was 12%, in Medicaid was 20%, and in commercial insurance was 20%. For the HF cohort, the mean age of patients was 70 years [SD, 15.4 years], and 41% were women. The proportion of patients enrolled in fee‐for‐service Medicare was 44%, in Medicare Advantage was 16%, in Medicaid was 21%, and in commercial insurance was 14%. Comorbid conditions including chronic pulmonary disease, hypertension, and diabetes mellitus were common among both the AMI and HF cohorts.
Table 1.
Characteristics of Patients Eligible for High‐Value Testing
| Acute Myocardial Infarction, N=15 893 | Heart Failure, N=7319 | |||
|---|---|---|---|---|
| Tested, N=12 619 | Not Tested, N=3274 | Tested, N=6164 | Not Tested, N=1155 | |
| Age, y, mean (SD) | 66.7 (13.2) | 67.1 (13.1) | 70.4 (15.4) | 71.2 (16.0) |
| Women, n (%) | 3883 (31) | 933 (27) | 2576 (42) | 421 (36) |
| Insurance, n (%) | ||||
| Medicare fee‐for‐service | 5458 (43) | 1579 (48) | 2818 (46) | 560 (48) |
| Medicare Advantage | 1542 (12) | 326 (10) | 1009 (16) | 138 (12) |
| Medicaid | 2412 (19) | 692 (21) | 1223 (20) | 300 (26) |
| Commercial | 2577 (20) | 541 (17) | 910 (15) | 125 (11) |
| Unknown | 630 (5) | 136 (4) | 204 (3) | 32 (3) |
| Comorbid conditions, n (%) | ||||
| Atrial fibrillation | 754 (6) | 181 (5) | 989 (16) | 171 (15) |
| Chronic pulmonary disease | 1855 (15) | 387 (11) | 1329 (22) | 193 (17) |
| Hyperlipidemia | 1210 (10) | 289 (8) | 529 (9) | 135 (12) |
| Hypertension | 6326 (51) | 1230 (35) | 3547 (58) | 522 (45) |
| Stroke | 556 (4) | 122 (4) | 263 (4) | 61 (5) |
| Cancer (breast, colorectal, prostate, or lung) | 506 (4) | 113 (3) | 363 (6) | 43 (4) |
| Chronic kidney disease | 3576 (29) | 698 (20) | 2330 (38) | 399 (35) |
| Diabetes mellitus | 3885 (31) | 759 (22) | 2109 (34) | 316 (27) |
| Heart failure | 1781 (14) | 419 (12) | n/a | n/a |
| Ischemic heart disease | 4490 (36) | 1033 (30) | 1860 (30) | 330 (29) |
n/a, not applicable.
Association of Testing With Insurance Provider
Overall, 79% of patients received high‐value testing for AMI, and 84% of patients received high‐value testing for HF. Rates of testing stratified by insurance provider ranged from 78% to 83% for AMI, and 81% to 90% for HF (Figure 3). Rates of testing increased slightly but similarly across insurance providers in a sensitivity analysis varying the time period for testing from 60 to 90 days following discharge (Table S4). After multivariable adjustment, patients with Medicaid were less likely than patients with commercial insurance to receive high‐value testing for both AMI (odds ratio [OR], 0.84 [0.73–0.98]; P=0.03) and HF (OR, 0.59 [0.51–0.70]; P<0.001; Table 3). Patients with Medicare Advantage were more likely to receive high‐value testing than patients with Medicare fee‐for‐service for AMI (OR, 1.35 [1.15–1.59]; P<0.001), but equally likely to receive testing for HF (OR, 1.03 [0.78–1.36]; P=0.84; Table 3). The odds of receiving testing were similar in sensitivity analyses (1) excluding safety‐net hospitals, and (2) excluding the 3.7% of patients with AMI who were eligible for testing more than once. There were no significant differences in use of testing by sex; for women compared with men the OR for AMI was 0.95 (0.87–1.05), and the OR for HF was 1.01 (0.86–1.20).
Figure 3. Rates of high‐value testing by health insurance provider.
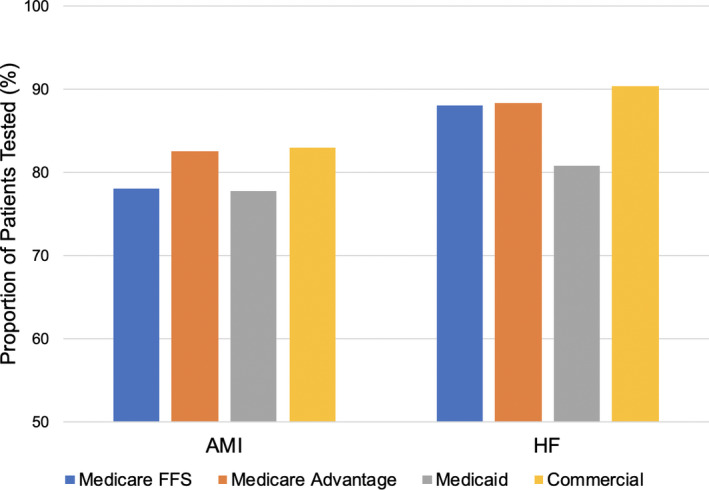
The proportion of patients receiving high‐value testing to assess left ventricular ejection fraction after hospitalization for acute myocardial infarction (AMI) or incident heart failure (HF) by insurance provider. FFS indicates fee‐for‐service.
Table 3.
Adjusted Associations of Health Insurance Provider With High‐ and Low‐Value Testing
| High‐Value Testing | Low‐Value Testing | |||||||
|---|---|---|---|---|---|---|---|---|
| AMI | HF | Low‐Risk Surgery | PCI/CABG | |||||
| OR | P Value | OR | P Value | OR | P Value | OR | P Value | |
| Medicare Advantage (compared with Medicare fee‐for‐service) | 1.35 (1.15–1.59) | <0.001 | 1.03 (0.78–1.36) | 0.84 | 1.06 (0.87–1.29) | 0.59 | 0.63 (0.55–0.72) | <0.001 |
| Medicaid (compared with commercial) | 0.84 (0.73–0.98) | 0.03 | 0.59 (0.51–0.70) | <0.001 | 1.10 (0.94–1.27) | 0.23 | 0.94 (0.81–1.10) | 0.44 |
All models were adjusted for patient characteristics including age, sex, and comorbid conditions. AMI indicates acute myocardial infarction; CABG, coronary artery bypass graft; HF, heart failure; and PCI, percutaneous coronary intervention.
Low‐Value Testing
Cohort Characteristics
We identified 111 387 patients eligible for low‐value testing prior to low‐risk surgery and 16 363 patients eligible for low‐value testing after PCI/CABG (Table 2). For the low‐risk surgery cohort, the mean age of patients was 64 years [SD, 16.1 years], and 56% were women. The proportion of patients enrolled in fee‐for‐service Medicare was 30%, in Medicare Advantage was 17%, in Medicaid was 24%, and in commercial insurance was 25%. For the PCI/CABG cohort, the mean age of patients was 70 years [SD, 15.7 years], and 26% were women. The proportion of patients enrolled in fee‐for‐service Medicare was 57%, in Medicare Advantage was 10%, in Medicaid was 16%, and in commercial insurance was 15%. Comorbid conditions including hypertension, chronic kidney disease, and diabetes mellitus were common among both the low‐risk surgery and PCI/CABG cohorts.
Table 2.
Characteristics of Patients Eligible for Low‐Value Testing
| Low‐Risk Surgery, N=111 387 | PCI/CABG, N=16 363 | |||
|---|---|---|---|---|
| Tested, N=3942 | Not Tested, N=107 445 | Tested, N=4422 | Not Tested, N=11 941 | |
| Age, y, mean (SD) | 64.0 (17.0) | 63.4 (16.8) | 70.3 (15.5) | 69.9 (15.7) |
| Women, n (%) | 2025 (51) | 60 199 (56) | 1206 (27) | 3016 (25) |
| Insurance, n (%) | ||||
| Medicare fee‐for‐service | 1427 (38) | 32 434 (30) | 2553 (58) | 6766 (57) |
| Medicare Advantage | 722 (18) | 18 130 (17) | 293 (7) | 1379 (12) |
| Medicaid | 803 (20) | 25 558 (24) | 807 (18) | 1779 (15) |
| Commercial | 668 (17) | 27 310 (25) | 646 (15) | 1746 (15) |
| Unknown | 143 (4) | 4192 (4) | 123 (3) | 271 (2) |
| Comorbid conditions, n (%) | ||||
| Atrial fibrillation | 567 (14) | 3375 (3) | 251 (6) | 859 (7) |
| Chronic pulmonary disease | 707 (18) | 8464 (8) | 618 (14) | 1755 (15) |
| Hyperlipidemia | 464 (12) | 3846 (4) | 516 (12) | 1446 (12) |
| Hypertension | 2405 (61) | 34 136 (32) | 2524 (57) | 6467 (54) |
| Stroke | 232 (6) | 1592 (1) | 170 (4) | 553 (5) |
| Cancer (breast, colorectal, prostate, or lung) | 568 (14) | 7891 (7) | 278 (6) | 740 (6) |
| Chronic kidney disease | 1272 (32) | 15 997 (15) | 938 (21) | 2742 (23) |
| Diabetes mellitus | 1171 (30) | 17 970 (17) | 1357 (31) | 3757 (31) |
| Heart failure | 817 (21) | 4135 (4) | 636 (14) | 2122 (18) |
| Ischemic heart disease | 1377 (35) | 11 189 (10) | 2349 (53) | 6139 (51) |
CABG indicates coronary artery bypass graft surgery; and PCI, percutaneous coronary intervention.
Association of Testing With Insurance Provider
Overall, 4% of patients received low‐value testing prior to low‐risk surgery, and 27% of patients received low‐value testing after PCI/CABG. Rates of testing stratified by insurance provider ranged from 2% to 5% for low‐risk surgery and 19% to 37% for PCI/CABG (Figure 4). After multivariable adjustment, there were no significant differences in the use of low‐value testing between patients with Medicaid compared with commercial insurance for either low‐risk surgery (OR, 1.10 [0.94–1.27]; P=0.23) or PCI/CABG (OR, 0.94 [0.81–1.10]; P=0.44; Table 3). Patients with Medicare Advantage were less likely to receive low‐value testing after PCI/CABG compared with Medicare fee‐for‐service (OR, 0.63 [0.55–0.72]; P<0.001), but equally likely to receive low‐value testing prior to low‐risk surgery (OR, 1.06 [0.87–1.29]; P=0.59; Table 3). The odds of receiving testing were similar in a sensitivity analysis excluding the 3.8% of low‐risk surgery patients who were eligible for testing more than once. There were no significant differences in use of testing by sex; for women compared with men, the OR for low‐risk surgery was 0.93 (0.84–1.03) and the OR for PCI/CABG was 0.92 (0.84–1.01).
Figure 4. Rates of low‐value testing by health insurance provider.
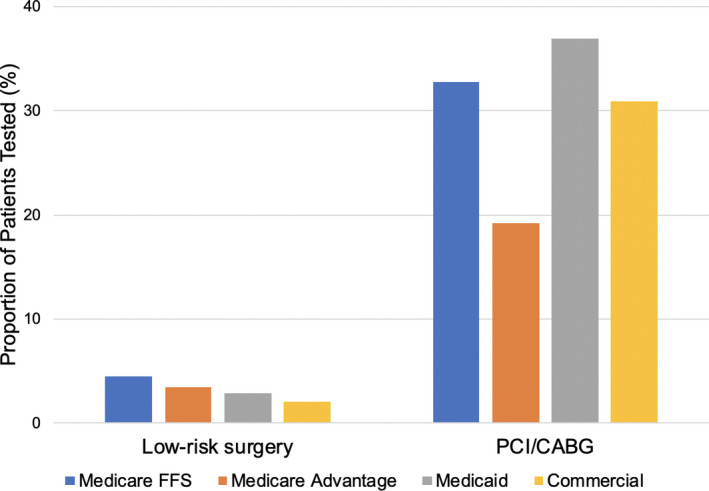
The proportion of patients receiving low‐value stress testing prior to low‐risk surgery and after percutaneous coronary intervention (PCI)/coronary artery bypass graft surgery (CABG) by health insurance provider. FFS indicates fee‐for‐service.
Discussion
Summary of Findings and Extension of Prior Knowledge
Our study found that health insurance provider is independently associated with use of certain high‐ and low‐value diagnostic cardiovascular tests. Using multivariable models adjusted for a variety of patient characteristics, we found that (1) patients with Medicaid were less likely to receive high‐value testing when hospitalized with AMI or incident HF compared with patients with commercial insurance, and (2) patients with Medicare Advantage were more likely to receive high‐value testing for AMI and less likely to receive low‐value testing after PCI/CABG compared with patients with Medicare fee‐for‐service. Thus, across 4 high‐ and low‐value diagnostic testing scenarios, Medicaid patients were less likely to receive guideline‐concordant testing compared with commercially insured patients, whereas Medicare Advantage patients were more likely to receive guideline‐concordant testing compared with fee‐for‐service Medicare patients.
Our study has several strengths. First, although prior studies have assessed variation in the use of certain high‐ and low‐value diagnostic tests, they have been limited to single insurance providers such as Medicare fee‐for‐service, 12 , 21 or single health systems such as Kaiser Permanente 10 and the Veterans Health Administration. 24 Our study finds that a patient’s insurance provider is a significant correlate of high‐ and low‐value testing after adjusting for a variety of patient‐level factors. Second, we assessed the use of 4 common high‐ and low‐value tests spanning a variety of different cardiovascular conditions. This provided an opportunity to broadly understand the association of insurance providers and insurance plan features on guideline‐concordant testing. Our results suggest that differences in insurance plan features, such as reimbursement and quality of care incentives, may lead to differences in use of high‐ and low‐value testing. Insurance plan features may also provide valuable targets to improve use of testing.
Medicaid Versus Commercial Insurance
Prior studies have shown that patients with Medicaid may receive fewer high‐value tests and treatments compared with patients with commercial insurance. 13 , 14 For example, a recent study found that Medicaid patients hospitalized with AMI received fewer revascularization procedures compared with matched patients with commercial insurance. 25 These disparities may occur for several reasons. First, Medicaid patients often receive care at safety‐net hospitals and clinics that have fewer resources to provide care overall and to invest in quality‐improvement programs in particular. 26 Second, there may be differences in the characteristics of clinicians who predominantly serve patients with a particular insurance status or who attend on hospital teams serving these patients. 27 Third, Medicaid typically provides lower reimbursement rates for services compared with other insurance providers, which can lead to delays or reduced access to care. 28 Our sensitivity analysis showed that the proportion of patients who were receiving delayed testing, within 90 days rather than 60 days, was similar across insurance categories, which suggests that overall access to care, rather than delayed access to care, may have contributed to our observation that Medicaid patients were less likely to receive high‐value testing. Finally, although we adjusted for a variety of patient characteristics, unmeasured differences between Medicaid and commercially insured patients, such as socioeconomic factors, could have contributed to the observed differences in use of testing.
Medicare Advantage Versus Traditional Fee‐for‐Service Medicare
Our study also suggests that insurance plans that measure and incentivize quality metrics may lead to more guideline‐concordant care. Patients enrolled in Medicare Advantage were not only more likely to receive high‐value testing for AMI, but also less likely to receive low‐value testing after PCI/CABG. Other studies have also found that Medicare Advantage patients may receive care that is more concordant with guidelines compared with patients with Medicare fee‐for‐service. 15 , 29 Managed care plans such as Medicare Advantage often directly incentivize the delivery of evidence‐based care that has been shown to improve patient outcomes. Many of these quality metrics form the basis on which Medicare Advantage plans receive star ratings and performance bonuses from the Centers for Medicare and Medicaid Services. One quality metric specifically addresses the chronic management of coronary artery disease, which may have contributed to the differences in testing among patients with AMI and patients with PCI/CABG observed in our study. Our results suggest that efforts to measure and report certain quality metrics may be effective in improving guideline‐concordant use of diagnostic testing.
Potential Clinical Consequences
Underuse of high‐value testing and overuse of low‐value testing can both worsen patient outcomes. Beneficial therapies that reduce mortality are often initiated based on results of high‐value tests, such as initiation and dose titration of β‐blockers in patients with incident HF and reduced systolic function. 19 Similarly, performance of low‐value testing can prompt unnecessary subsequent invasive procedures, such as coronary angiography, that carry risks of bleeding and stroke. Low‐value cardiovascular testing also contributes to medical overspending and unnecessary downstream consequences. Therefore, the observed differences in use of testing in our study represent an opportunity to optimize testing to improve patient outcomes and reduce healthcare costs.
Limitations
Although we accounted for a wide variety of patient‐level variables in our multilevel regression models, unmeasured confounders, such as tobacco use or family history, could have contributed to observed differences in use of high‐ and low‐value testing. We studied the use of high‐ and low‐value tests in specific scenarios that could be identified using administrative claims data. Use of other high‐ and low‐value tests, as well as tests of uncertain appropriateness, may differ. Our data set did not have information on whether symptoms may have prompted stress testing after PCI/CABG. Some stress tests were likely clinically indicated, and it is difficult to ascertain whether the numbers of clinically indicated tests vary by insurance type. Similar to prior studies, we excluded tests performed in association with acute‐care visits that may have been prompted by changes in symptoms. Race/ethnicity was not available in our data set, so we were unable to adjust for these demographics in our models. Hospital‐level characteristics were not available in our data set. Although we tested for interaction between comorbid conditions and insurance type and adjusted for comorbidities in our regression models, residual confounding is still a possibility given the imbalance of comorbid conditions between the groups that did and did not receive testing. We did not have sufficient power to assess for differences in clinical outcomes between patients who received or did not receive high‐ or low‐value testing. Our data set did not include information on prior authorizations or disapprovals of such requests. We used all‐payer data from the state of Colorado, which provided important information on insurance plan enrollment but may have limited the generalizability of our findings.
Conclusions
Health insurance provider is independently associated with use of certain high‐ and low‐value diagnostic cardiovascular tests. Medicaid patients were less likely to receive guideline‐concordant testing compared with commercially insured patients, whereas Medicare Advantage patients were more likely to receive guideline‐concordant testing compared with fee‐for‐service Medicare patients. Our results suggest that reimbursement and quality incentives may affect use of guideline‐concordant testing, and that features of insurance plans could be useful targets to improve use of testing.
Sources of Funding
This research was supported by National Institutes of Health/ National Heart, Lung, and Blood Institute grant K23HL143208 (PI: Kini). Dr Raghavan is supported by the Veterans Health Administration Career Development Award IK2‐CX001907‐01. Dr Khazanie is supported by National Institutes of Health/National Heart, Lung, and Blood Institute grant K23HL145122. Dr Ho is supported by grants from the National Heart, Lung, and Blood Institute, Veterans Health Administration Health Services Research and Development, and University of Colorado School of Medicine.
Disclosures
Dr Ho has a research agreement with Bristol‐Myers Squibb through the University of Colorado. He serves as the Deputy Editor for Circulation: Cardiovascular Quality and Outcomes. Dr Masoudi has a contract with the American College of Cardiology for his role as Chief Science Officer, National Cardiovascular Data Registries. He serves as a consultant to TurningPoint Healthcare Solutions. The remaining authors have no disclosures to report.
Supporting information
Tables S1–S4
(J Am Heart Assoc. 2021;10:e018877. DOI: 10.1161/JAHA.120.018877.)
Supplementary Material for this article is available at https://www.ahajournals.org/doi/suppl/10.1161/JAHA.120.018877
For Sources of Funding and Disclosures, see page 9.
References
- 1. Andrus BW, Welch HG. Medicare services provided by cardiologists in the United States: 1999–2008. Circ Cardiovasc Qual Outcomes. 2012;5:31–36. DOI: 10.1161/CIRCOUTCOMES.111.961813 [DOI] [PubMed] [Google Scholar]
- 2. Mark DB, Anderson JL, Brinker JA, Brophy JA, Casey DE, Cross RR, Edmundowicz D, Hachamovitch R, Hlatky MA, Jacobs JE, et al. ACC/AHA/ASE/ASNC/HRS/IAC/Mended Hearts/NASCI/RSNA/SAIP/SCAI/SCCT/SCMR/SNMMI 2014 health policy statement on use of noninvasive cardiovascular imaging: a report of the American College of Cardiology Clinical Quality Committee. J Am Coll Cardiol. 2014;63:698–721. DOI: 10.1016/j.jacc.2013.02.002. [DOI] [PubMed] [Google Scholar]
- 3. Safavi KC, Li SX, Dharmarajan K, Venkatesh AK, Strait KM, Lin H, Lowe TJ, Fazel R, Nallamothu BK, Krumholz HM. Hospital variation in the use of noninvasive cardiac imaging and its association with downstream testing, interventions, and outcomes. JAMA Intern Med. 2014;174:546–553. DOI: 10.1001/jamainternmed.2013.14407 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Douglas PS, Garcia MJ, Haines DE, Lai WW, Manning WJ, Patel AR, Picard MH, Polk DM, Ragosta M, Ward RP, et al. ACCF/ASE/AHA/ASNC/HFSA/HRS/SCAI/SCCM/SCCT/SCMR 2011 appropriate use criteria for echocardiography. J Am Coll Cardiol. 2011;57:1126–1166. DOI: 10.1016/j.jacc.2010.11.002 [DOI] [PubMed] [Google Scholar]
- 5. Wolk MJ, Bailey SR, Doherty JU, Douglas PS, Hendel RC, Kramer CM, Min JK, Patel MR, Rosenbaum L, Shaw LJ, et al. ACCF/AHA/ASE/ASNC/HFSA/HRS/SCAI/SCCT/SCMR/STS 2013 multimodality appropriate use criteria for the detection and risk assessment of stable ischemic heart disease. J Am Coll Cardiol. 2014;63:380–406. DOI: 10.1016/j.jacc.2013.11.009 [DOI] [PubMed] [Google Scholar]
- 6. Cassel CK, Guest JA. Choosing wisely: helping physicians and patients make smart decisions about their care. JAMA. 2012;307:1801–1802. DOI: 10.1001/jama.2012.476. [DOI] [PubMed] [Google Scholar]
- 7. Fonseca R, Negishi K, Otahal P, Marwick TH. Temporal changes in appropriateness of cardiac imaging. J Am Coll Cardiol. 2015;65:763–773. DOI: 10.1016/j.jacc.2014.11.057 [DOI] [PubMed] [Google Scholar]
- 8. Ladapo JA, Blecker S, O'Donnell M, Jumkhawala SA, Douglas PS. Appropriate use of cardiac stress testing with imaging: a systematic review and meta‐analysis. PLoS One. 2016;11:e0161153. DOI: 10.1371/journal.pone.0161153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kini V, Viragh T, Magid D, Masoudi FA, Moghtaderi A, Black B. Trends in high‐and low‐value cardiovascular diagnostic testing in fee‐for‐service Medicare, 2000–2016. JAMA Netw Open. 2019;2:e1913070. DOI: 10.1001/jamanetworkopen.2019.13070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Farmer SA, Lenzo J, Magid DJ, Gurwitz JH, Smith DH, Hsu G, Sung SH, Go AS. Hospital‐level variation in use of cardiovascular testing for adults with incident heart failure: findings from the cardiovascular research network heart failure study. JACC Cardiovasc Imaging. 2014;7:690–700. DOI: 10.1016/j.jcmg.2014.02.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Miller AL, Dib C, Li L, Chen AY, Amsterdam E, Funk M, Saucedo JF, Wang TY. Left ventricular ejection fraction assessment among patients with acute myocardial infarction and its association with hospital quality of care and evidence‐based therapy use. Circ Cardiovasc Qual Outcomes. 2012;5:662–671. DOI: 10.1161/CIRCOUTCOMES.112.965012 [DOI] [PubMed] [Google Scholar]
- 12. Curtis LH, Greiner MA, Shea AM, Whellan DJ, Hammill BG, Schulman KA, Douglas PS. Assessment of left ventricular function in older Medicare beneficiaries with newly diagnosed heart failure. Circ Cardiovasc Qual Outcomes. 2011;4:85–91. DOI: 10.1161/CIRCOUTCOMES.110.958587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Hasan O, Orav EJ, Hicks LS. Insurance status and hospital care for myocardial infarction, stroke, and pneumonia. J Hosp Med. 2010;5:452–459. DOI: 10.1002/jhm.687 [DOI] [PubMed] [Google Scholar]
- 14. Epstein AJ, Ketcham JD, Rathore SS, Groeneveld PW. Variations in the use of an innovative technology by payer: the case of drug‐eluting stents. Med Care. 2012;50:1. DOI: 10.1097/MLR.0b013e31822d5de9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Figueroa JF, Blumenthal DM, Feyman Y, Frakt AB, Turchin A, Doros G, Gao Q, Song Y, Maddox KE. Differences in management of coronary artery disease in patients with Medicare Advantage vs traditional fee‐for‐service Medicare among cardiology practices. JAMA Cardiol. 2019;4:265–271. DOI: 10.1001/jamacardio.2019.0007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Unger JM, Blanke CD, LeBlanc M, Barlow WE, Vaidya R, Ramsey SD, Hershman DL. Association of patient demographic characteristics and insurance status with survival in cancer randomized clinical trials with positive findings. JAMA Netw Open. 2020;3:e203842. DOI: 10.1001/jamanetworkopen.2020.3842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Slatore CG, Au DH, Gould MK. An official American Thoracic Society systematic review: insurance status and disparities in lung cancer practices and outcomes. Am J Respir Crit Care Med. 2010;182:1195–1205. DOI: 10.1164/rccm.2009-038ST [DOI] [PubMed] [Google Scholar]
- 18. Chronic Conditions Data Warehouse. Centers for Medicare and Medicaid Services. Available at: https://www2.ccwdata.org/web/guest/condition‐categories. Accessed March 20, 2020. [Google Scholar]
- 19. O’Gara PT, Kushner FG, Ascheim DD, Casey DE, Chung MK, de Lemos JA, Ettinger SM, Fang JC, Fesmire FM, Franklin BA, et al. 2013 ACCF/AHA guideline for the management of ST‐elevation myocardial infarction: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;127:529–555. DOI: 10.1161/CIR.0b013e3182742c84 [DOI] [PubMed] [Google Scholar]
- 20. Amsterdam EA, Wenger NK, Brindis RG, Casey DE Jr, Ganiats TG, Holmes DR Jr, Jaffe AS, Jneid H, Kelly RF, Kontos MC, et al. 2014 AHA/ACC guideline for the management of patients with non‐ST‐elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation. 2014;130:2354–2394. DOI: 10.1161/CIR.0000000000000133 [DOI] [PubMed] [Google Scholar]
- 21. Kerr EA, Chen J, Sussman JB, Klamerus ML, Nallamothu BK. Stress testing before low‐risk surgery: so many recommendations, so little overuse. JAMA Intern Med. 2015;175:645–647. DOI: 10.1001/jamainternmed.2014.7877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Schwartz AL, Landon BE, Elshaug AG, Chernew ME, McWilliams JM. Measuring low‐value care in Medicare. JAMA Intern Med. 2014;174:1067–1076. DOI: 10.1001/jamainternmed.2014.1541 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Shah BR, Cowper PA, O'Brien SM, Jensen N, Drawz M, Patel MR, Douglas PS, Peterson ED. Patterns of cardiac stress testing after revascularization in community practice. J Am Coll Cardiol. 2010;56:1328–1334. DOI: 10.1016/j.jacc.2010.03.093 [DOI] [PubMed] [Google Scholar]
- 24. Bradley SM, Hess E, Winchester DE, Sussman JB, Aggarwal V, Maddox TM, Barón AE, Rumsfeld JS, Ho PM. Stress testing after percutaneous coronary intervention in the veterans affairs healthcare system: insights from the Veterans Affairs Clinical Assessment, Reporting, and Tracking Program. Circ Cardiovasc Qual Outcomes. 2015;8:486–492. DOI: 10.1161/CIRCOUTCOMES.114.001561 [DOI] [PubMed] [Google Scholar]
- 25. Patel N, Gupta A, Doshi R, Kalra R, Bajaj NS, Arora G, Arora P. In‐hospital management and outcomes after ST‐segment–elevation myocardial infarction in Medicaid beneficiaries compared with privately insured individuals. Circ Cardiovasc Qual Outcomes. 2019;12:e004971. DOI: 10.1161/CIRCOUTCOMES.118.004971 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Hasnain‐Wynia R, Baker DW, Nerenz D, Feinglass J, Beal AC, Landrum MB, Behal R, Weissman JS. Disparities in health care are driven by where minority patients seek care: examination of the hospital quality alliance measures. Arch Intern Med. 2007;167:1233–1239. DOI: 10.1001/archinte.167.12.1233 [DOI] [PubMed] [Google Scholar]
- 27. Spencer CS, Gaskin DJ, Roberts ET. The quality of care delivered to patients within the same hospital varies by insurance type. Health Aff (Millwood). 2013;32:1731–1739. DOI: 10.1377/hlthaff.2012.1400 [DOI] [PubMed] [Google Scholar]
- 28. Medicaid Access Study Group . Access of Medicaid recipients to outpatient care. N Engl J Med. 1994;330:1426–1430. DOI: 10.1056/NEJM199405193302007 [DOI] [PubMed] [Google Scholar]
- 29. Huckfeldt PJ, Escarce JJ, Rabideau B, Karaca‐Mandic P, Sood N. Less intense postacute care, better outcomes for enrollees in Medicare Advantage than those in fee‐for‐service. Health Aff (Millwood). 2017;36:91–100. DOI: 10.1377/hlthaff.2016.1027 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Tables S1–S4


