Abstract
Background
Cardiovascular involvement in coronavirus disease 2019 (COVID‐19) is common and leads to worsened mortality. Diagnostic cardiovascular studies may be helpful for resource appropriation and identifying patients at increased risk for death.
Methods and Results
We analyzed 887 patients (aged 64±17 years) admitted with COVID‐19 from March 1 to April 3, 2020 in New York City with 12 lead electrocardiography within 2 days of diagnosis. Demographics, comorbidities, and laboratory testing, including high sensitivity cardiac troponin T (hs‐cTnT), were abstracted. At 30 days follow‐up, 556 patients (63%) were living without requiring mechanical ventilation, 123 (14%) were living and required mechanical ventilation, and 203 (23%) had expired. Electrocardiography findings included atrial fibrillation or atrial flutter (AF/AFL) in 46 (5%) and ST‐T wave changes in 306 (38%). 27 (59%) patients with AF/AFL expired as compared to 181 (21%) of 841 with other non‐life‐threatening rhythms (P<0.001). Multivariable analysis incorporating age, comorbidities, AF/AFL, QRS abnormalities, and ST‐T wave changes, and initial hs‐cTnT ≥20 ng/L showed that increased age (HR 1.04/year), elevated hs‐cTnT (HR 4.57), AF/AFL (HR 2.07), and a history of coronary artery disease (HR 1.56) and active cancer (HR 1.87) were associated with increased mortality.
Conclusions
Myocardial injury with hs‐cTnT ≥20 ng/L, in addition to cardiac conduction perturbations, especially AF/AFL, upon hospital admission for COVID‐19 infection is associated with markedly increased risk for mortality than either diagnostic abnormality alone.
Keywords: arrhythmia, atrial fibrillation, COVID‐19, troponin
Subject Categories: Atrial Fibrillation
Nonstandard Abbreviations and Acronyms
- AFL
atrial flutter
- AF/AFL
atrial fibrillation or atrial flutter
- COVID‐19
coronavirus disease 2019
- HFpEF
heart failure with preserved ejection fraction
- HFrEF
heart failure with reduced ejection fraction
- hs‐cTnT
high‐sensitivity cardiac troponin T
- IQR
interquartile range
- SARS‐CoV‐2
severe acute respiratory syndrome coronavirus 2
Clinical Perspective
What Is New?
This retrospective study of 887 mostly hospitalized patients with coronavirus disease 2019 (COVID‐19) shows that cardiac conduction abnormalities on 12 lead ECG are common and are correlated with increased mortality.
The presence of atrial fibrillation or atrial flutter is a particularly adverse marker, with 59% of patients with atrial fibrillation or atrial flutter dying within 30 days, as compared to 21% of patients with other rhythms.
What Are the Clinical Implications?
Older age, a history of coronary artery disease, and active cancer and findings of an elevated cardiac troponin and atrial fibrillation on presentation are associated with increased mortality.
The first cases of coronavirus disease 2019 (COVID‐19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2), were diagnosed in Wuhan, China in December 2019. 1 Since that time, COVID‐19 has developed into a pandemic that has been diagnosed in millions of individuals globally with hundreds of thousands of deaths.
COVID‐19 has been shown to be particularly dangerous in older populations and in those with pre‐existing comorbidities, such as cardiovascular disease, diabetes mellitus, hypertension, obesity, and pulmonary disease, in populations in China, 2 Italy, 3 and in the United States. 4 , 5 The most prominent clinical manifestation of COVID‐19 is pulmonary involvement with the acute respiratory distress syndrome, but there has been increasing recognition that cardiac complications are important contributors to COVID‐19 mortality. 6 In particular, cardiac injury as indicated by elevated troponin levels has been identified as a marker for adverse events, and significant ECG abnormalities, such as ST elevations, heart block, and ventricular tachycardia, have been reported in patients with COVID‐19. 5 , 6 , 7 , 8 , 9 Implicated pathophysiologic pathways of cardiac involvement of COVID‐19 include myocardial ischemia from epicardial and microvascular coronary thrombosis caused by the hypercoagulable state associated with COVID‐19, demand‐supply mismatching from hypoxemia and shock, systemic inflammation, and direct viral myocardial infiltration leading to myocarditis. 10 , 11 , 12 , 13
Although ECG abnormalities have been reported in small case series, there has not yet been a systematic ECG analysis in a large population of patients with COVID‐19 or a determination that specific arrhythmias confer excess risk. In this study, we aimed to describe the ECG findings that can be used to identify those patients most likely to be have adverse outcomes.
Methods
Patients, Setting, and Data Collection
Patients older than 18 years of age who tested positive for SARS‐CoV‐2 using a reverse‐transcriptase–polymerase‐chain‐reaction (RT‐PCR) assay of a nasopharyngeal or oropharyngeal sample from March 1, 2020 through April 3, 2020 at Columbia University Irving Medical Center and New York‐Presbyterian Allen Hospital and who had a 12‐lead ECG within the first 2 days of diagnosis or presentation were included in the study. In the initial phases of the pandemic analyzed in this study, SARS‐CoV‐2 testing was mostly reserved for patients who were planned for admission. This study was conducted with approval from the Columbia University Irving Medical Center Institutional Review Board and the need for informed consent was waived. The data that support the findings of this study are available from the corresponding author on reasonable request. We abstracted the following data: demographics, comorbidities, laboratory findings, electrocardiography, and outcomes. Comorbidities were manually assessed by physicians and included the following: hypertension, diabetes mellitus, obesity (defined as body mass index ≥30 kg/m2), lung disease (including asthma, chronic obstructive pulmonary disease, interstitial lung disease, or any primary lung disease that required home oxygen therapy), chronic kidney disease stage 3 to 5 (CKD), heart failure with reduced ejection fraction defined as heart failure with left ventricular ejection fraction <50% (HFrEF), heart failure with preserved ejection fraction defined as heart failure with left ventricular ejection fraction ≥50% (HFpEF), coronary artery disease defined as a history of obstruction of the left main coronary artery ≥50% or other coronary artery ≥70% (CAD), active cancer (which was defined as metastatic cancer, cancer that required treatment within the last 6 months, or cancer undergoing active observation), or personal history of cancer that did not meet the active cancer definition. We excluded non‐metastatic nonmelanoma skin cancers from inclusion in the cancer criteria.
Laboratory Data
Laboratory data were abstracted from the electronic health record. This included high‐sensitivity cardiac troponin T (hs‐cTnT), complete blood counts, and inflammatory markers including C‐reactive protein, ferritin, interleukin‐6, procalcitonin, D‐dimer, and lactate dehydrogenase. The first lab test performed during the index presentation was defined as the initial lab test. In addition, the maximum value for each lab throughout the index presentation was also recorded.
Electrocardiogram
An initial 12‐lead ECG performed within 2 days of admission or diagnosis of COVID‐19 was abstracted and analyzed in all 887 patients using the MUSE Cardiology Information System (GE Healthcare, Chicago, IL, United States). All initial ECGs were analyzed by a board‐certified electrophysiologist (E.W., J.D., J.M., H.Y., F.E., and D.S.) with a standardized reading protocol including the following: rate, rhythm, QRS axis, ECG intervals, QRS wave amplitude and morphology, and ST and T wave morphology. The ST segment was measured at 80 ms after the J‐point, and pathologic ST depressions or elevations were defined as ≥1 mm in 2 contiguous leads. Patients with QRS complexes ≥120 ms in length were excluded from ST and T wave analysis. The QT was corrected using the Bazett formula (QTc), and the corrected QT using the Fridericia formula was also recorded. ECG’s were broadly categorized into one of the following 3 categories:
Normal: ECG with normal sinus rhythm with no significant abnormalities in rate, rhythm, axis, or wave morphology.
Borderline: ECG that did not meet abnormal criteria but did have any of the following: PR >240 ms, QTc (Bazett) 470–499 ms, a single ventricular or atrial ectopic beat in a 10 second ECG strip, atrial abnormalities, an incomplete right bundle‐branch block, or a ventricular rate >100.
Abnormal: ECG that met any of the following criteria: ventricular rate <50, widened QRS with duration ≥120 ms, abnormal QRS axis, left bundle‐branch block, right‐bundle branch block, nonspecific interventricular conduction delay, left anterior fascicular block, left posterior fascicular block, corrected QT (Bazett) ≥500 ms, any ST or T wave abnormality, signs of right ventricular strain by S1Q3T3 or right ventricular hypertrophy, significant atrial arrhythmia, second or third degree atrioventricular block, atrial or ventricular pacing, more than 1 atrial or ventricular ectopic beat in a 10 second ECG strip, Q wave infarct, QRS voltage abnormality with left ventricular hypertrophy or low voltage, or ventricular pre‐excitation.
In order to determine which classes of ECG variables were most impactful on prognosis, the ECG abnormalities were stratified into the 3 sub‐categories, which included the following: (1) a rhythm diagnosis of AF/AFL versus any another rhythm, including sinus rhythms, ectopic atrial rhythms, or atrially paced rhythm, (2) abnormal QRS morphology, which included any of left bundle‐branch block, right bundle‐branch block, nonspecific interventricular conduction delay, left anterior fascicular block, left posterior fascicular block, left ventricular hypertrophy, low QRS voltage, Q wave infarct, or ventricular pacing, and (3) any ST or T wave abnormality.
Clinical Outcomes
The dates of admission, COVID‐19 diagnosis, discharge, intubation, extubation, and death were abstracted, if applicable. For outcomes, patients were classified into one of 3 mutually exclusive groups: (1) living, managed in the emergency department or admitted to the hospital, and did not require mechanical ventilation, (2) living and required mechanical ventilation at any time, and (3) expired. Clinical outcomes were assessed at 2 times. First, the receipt of and timing of mechanical ventilation was manually abstracted on April 16 to 17 at a minimum of 14 days after admission or diagnosis of COVID‐19. Second, mortality was again assessed on May 2 and censored at 30 days after admission or diagnosis of COVID‐19. Association with adverse clinical outcomes was defined using both overall mortality and a composite of death or the need for mechanical ventilation (“composite adverse outcome”) as end points.
Statistical Analysis
Demographics, comorbidities, laboratory findings, ECG parameters, and clinical outcomes were described using means, medians, and frequencies, as appropriate. The Shapiro‐Wilk test was used to assess normality of key variables. If assumptions of normality were met, mean, standard deviation, and 95% CIs were described. Non‐parametric results were described with median and interquartile ranges. Categorical variables were analyzed using chi‐square analysis and with the Fisher’s exact test for counts <5. Continuous variable means were compared using t tests. The Kaplan‐Meier method was used to estimate cumulative 30‐day risk of death when patients were stratified by overall ECG categorization and initial hs‐cTnT level. In order to assess for the impact of the ECG findings and hs‐cTnT on outcomes in the context of demographics and comorbidities, a multivariable Cox proportional‐hazards regression model that included age, sex, comorbidities, initial hs‐cTnT, and the ECG findings of atrial fibrillation/flutter, QRS abnormalities, and ST‐T wave abnormalities was used to perform an adjusted analysis of survival. The variable effect size of ECG abnormalities on 30‐day mortality, based on whether initial high sensitivity cardiac troponin T (hs‐cTnT) was >20 ng/L, was further explored in this model. Three separate interaction terms (between elevated hs‐cTnT + each individual ECG abnormality [AF/AFL, QRS abnormalities, and ST‐T wave abnormalities]) were tested. These 3 interaction terms were added to the model alone (without the other 2 interaction terms) or together (all 3 interaction terms added to the model). None of these interaction terms were significant. These interaction terms were, therefore, omitted from the final model. All reported P values are two‐sided and P values <0.05 were determined to be significant. Statistical tests were performed in Python 3.4 (Wilmington, DE) and SPSS v26 (Chicago, IL).
Results
Patient Characteristics, Comorbidities, and Outcomes
A total of 1258 patients had positive SARS‐CoV‐2 PCR tests during the study period. Of these 1258 patients, a total of 887 had ECGs performed within 2 days of presentation or diagnosis of COVID‐19 and were included in the final study cohort. Of these 887 patients, 556 (63%) were living and never required mechanical ventilation, 123 (14%) patients were living and required mechanical ventilation, and 208 (23%) had expired at the time of data abstraction (Figure 1). Clinical characteristics, outcomes, and comorbidities are summarized in Table 1.
Figure 1. Patient outcomes.
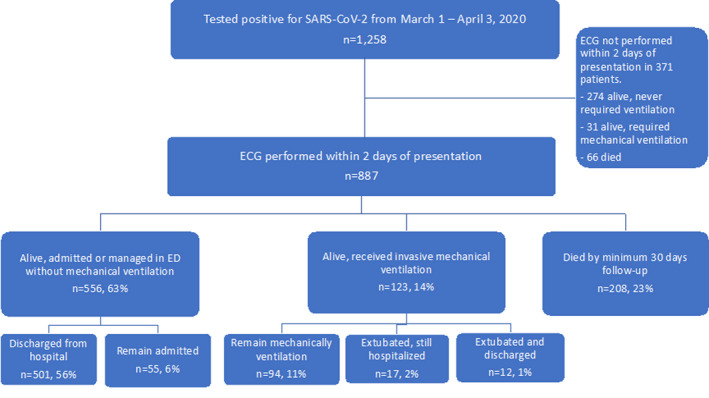
This diagram demonstrates patient outcomes. One thousand two hundred and fifty‐eight patients were diagnosed with COVID‐19 from March 1 to April 3, 2020 in the study with a minimum of 30 days of follow‐up. Of those patients, 887 had an ECG performed within 2 days of presentation, after the exclusion of 371 patients who did not have an ECG within 2 days. Of those 887 patients, 556 were living without requiring mechanical ventilation (of whom 501 had been discharged and 55 remained admitted). 123 patients were living and had required mechanical ventilation, of whom 94 remained intubated, 17 had been extubated but remained hospitalized, and 12 has been extubated and discharged from the hospital. A total of 208 patients had expired by 30 days of follow‐up. COVID‐19 indicates coronavirus disease 2019; and ED, emergency department.
Table 1.
Patient Characteristics
| Total | % | Living, No Mechanical Ventilation | % | Living, Required Mechanical Ventilation | % | Expired | % | P value (death or ventilated vs living) | |
|---|---|---|---|---|---|---|---|---|---|
| Total (N) | 887 | 556 | 63% | 123 | 14% | 208 | 23% | ||
| Demographics | |||||||||
| Age (y), mean | 64.1 | 61.0 | 59.2 | 75.2 | <0.001 | ||||
| Age <60 | 319 | 36% | 239 | 43% | 51 | 41% | 29 | 14% | |
| Age ≥60 | 568 | 64% | 317 | 57% | 72 | 59% | 179 | 86% | |
| Female sex | 374 | 42% | 250 | 45% | 41 | 33% | 83 | 40% | 0.041 |
| Male sex | 513 | 58% | 306 | 55% | 82 | 67% | 125 | 60% | |
| Comorbidities | |||||||||
| No comorbidities | 127 | 14% | 100 | 18% | 20 | 16% | 7 | 3% | <0.001 |
| Hypertension | 541 | 61% | 308 | 55% | 69 | 56% | 164 | 79% | <0.001 |
| Diabetes mellitus | 350 | 39% | 196 | 35% | 43 | 35% | 111 | 53% | 0.001 |
| Obesity | 309 | 35% | 196 | 35% | 51 | 41% | 62 | 30% | 0.722 |
| Pulmonary disease | 142 | 16% | 88 | 16% | 17 | 14% | 37 | 18% | 0.857 |
| CKD | 147 | 17% | 76 | 14% | 15 | 12% | 56 | 27% | 0.003 |
| HFrEF | 63 | 7% | 35 | 6% | 3 | 2% | 25 | 12% | 0.228 |
| HFpEF | 41 | 5% | 22 | 4% | 3 | 2% | 16 | 8% | 0.223 |
| CAD | 104 | 12% | 56 | 10% | 8 | 7% | 40 | 19% | 0.048 |
| Cancer, active | 37 | 4% | 19 | 3% | 6 | 5% | 12 | 6% | 0.147 |
| Cancer, history | 55 | 6% | 31 | 6% | 7 | 6% | 17 | 8% | 0.320 |
| Two or more comorbidities | 542 | 61% | 317 | 57% | 67 | 54% | 158 | 76% | 0.001 |
This table displays baseline characteristics in the 887‐patient cohort. Data is displayed for the total cohort, as well as for the following 3 groups: (1) patients who were living and did not require mechanical ventilation, (2) patients who were living and required mechanical ventilation, and (3) patients who expired. Comparisons were performed between patients with a favorable outcome (living and not requiring mechanical ventilation) versus a composite of those with who expired or required mechanical ventilation. CKD indicates stage 3 or greater chronic kidney disease; CAD indicates obstructive coronary artery disease; HFrEF indicates heart failure with reduced ejection fraction which was defined as a clinical diagnosis of systolic heart failure or a baseline echocardiogram with left ventricular ejection fraction <50%; and HFpEF indicates heart failure with preserved ejection fraction.
The mean age was 64.1 years (standard deviation 17.2, range 18–100) and 374 (42%) patients were female. Common comorbidities included hypertension in 541 patients (61%), diabetes mellitus (n=350, 39%), obesity (n=309, 35%), lung disease (n=142, 16%), and CKD (n=147, 17%). Comorbid cardiovascular disease included coronary artery disease (n=104, 12%), HFrEF (n=63, 7%), and HFpEF (n=41, 5%). On univariate analysis, patients who had worse outcomes, as defined by death or the requirement for mechanical ventilation, were more commonly male (P=0.041), older in age (P<0.001), and had a history of hypertension (P<0.001), diabetes mellitus (P=0.001), CKD (P=0.003), and CAD (P=0.048).
Laboratory Findings
Median laboratory values are described in Table 2. Abnormal cardiac biomarkers were common in the cohort. A hs‐cTnT was checked in 830 (93.6%) patients, and the median value of the initial test was 16 ng/L (IQR 8–41, institutional normal ranges <15 ng/L in women and <23 ng/L in men). In 361 of 830 patients (43%), the first value was ≥20 ng/L. Initial hs‐cTnT was associated with composite adverse outcomes, with a median hs‐cTnT of 11 ng/L (IQR <6 to 24) in patients who were living and did not require mechanical ventilation versus 31 ng/L (IQR 14–75) in patients who expired or required mechanical ventilation. Peak hs‐cTnT was also associated with composite adverse outcomes, with a median of 11 ng/L (IQR 7–27) in patients who were living and did not require mechanical ventilation and 76 ng/L (IQR 32–177) in patients who expired or required mechanical ventilation. In addition to hs‐cTnT, elevated NT‐proBNP (N‐terminal pro‐B‐type natriuretic peptide), elevated inflammatory markers, renal failure, and elevated D‐dimer were frequently present and associated with adverse outcomes in univariate analysis.
Table 2.
Laboratory Data
| Total | Living, No Mechanical Ventilation | Living, Required Mechanical Ventilation | Expired | P value (Death or Ventilated vs Favorable Outcome) | ||
|---|---|---|---|---|---|---|
| Cardiac | ||||||
| hs‐cTnT (n=830) | Initial | 16 (8–41) | 11 (6–24) | 16 (8–37) | 41 (22–96) | <0.001 |
| Peak | 23 (9–75.5) | 11 (7–27) | 64 (28–160) | 83 (36–195) | <0.001 | |
| NT‐proBNP (n=252) | Initial | 352 (73.4–1717) | 291 (46.7–1129) | 144 (48.5–435) | 1359 (349–4814) | <0.001 |
| Peak | 361 (73.4–1867) | 291 (46.7–1129) | 144 (61.6–481) | 1423 (359.2–4814) | <0.001 | |
| Inflammatory | ||||||
| WBC (n=879) | Initial | 7.07 (5.3–9.7) | 6.5 (5.1–8.6) | 9.68 (6.5–13.6) | 8.14 (5.4–11) | <0.001 |
| Peak | 10.11 (7.2–15.9) | 8.07 (6.3–10.7) | 20.85 (16–27) | 14.13 (9.8–19.8) | <0.001 | |
| C‐reactive protein (n=815) | Initial | 106.44 (52.4–189) | 88.03 (36–154.1) | 152.7 (66.4–234.2) | 148.14 (94–223.4) | <0.001 |
| Peak | 167.92 (86.1–286.2) | 113.55 (52.8–187.5) | 300 (266.2–300) | 238.23 (146.5–300) | <0.001 | |
| Sedimentation rate (n=794) | Initial | 68 (46–94.8) | 66 (44–90) | 70 (52–94) | 74 (52–101) | 0.005 |
| Peak | 90 (62–117.8) | 78 (50.5–105.5) | 117.5 (95.3–130) | 100 (74–125) | 0.001 | |
| Procalcitonin (n=823) | Initial | 0.22 (0.1–0.6) | 0.15 (0.1–0.3) | 0.37 (0.2–1) | 0.51 (0.2–1.8) | <0.001 |
| Peak | 0.37 (0.1–2.2) | 0.18 (0.1–0.4) | 3.22 (1.4–9.5) | 2.01 (0.5–8) | <0.001 | |
| Ferritin (n=809) | Initial | 715.1 (352.2–1307) | 609.25 (286.2–1110.3) | 1082.5 (625.8–1934) | 820 (396.1–1595) | <0.001 |
| Peak | 940 (441.6–1969) | 712.3 (342.1–1322.8) | 1948 (1082.5–3289.8) | 1351 (642.8–3075) | <0.001 | |
| Interleukin‐6 (n=691) | Initial | 86.3 (38.3–240.6) | 48.55 (20.7–91.1) | 222.8 (72.9–315) | 146.9 (85.4–315) | <0.001 |
| Peak | 38.4 (9–169.8) | 15 (5–50.6) | 315 (138.1–315) | 121.1 (35.5–315) | <0.001 | |
| Lactate dehydrogenase (n=802) | Initial | 393.5 (293–515) | 349 (268–440) | 486 (374.5–629.5) | 496.5 (350.8–660.3) | <0.001 |
| Peak | 456.5 (336–665.8) | 376 (293.3–481.3) | 708.5 (566.5–911) | 662 (475.3–935.3) | <0.001 | |
| D‐dimer (n=755) | Initial | 1.3 (0.8–2.7) | 1.1 (0.6–1.8) | 2.1 (1–4.8) | 2.1 (1.2–4.4) | <0.001 |
| Peak | 2.1 (1–7.6) | 1.2 (0.7–2.4) | 20 (6.2–20) | 4.4 (2.1–17.1) | <0.001 | |
| Kidney function | ||||||
| Creatinine (n=876) | Initial | 1.1 (0.8–1.6) | 1.0 (0.8–1.4) | 1.1 (0.9–1.5) | 1.4 (1–2.4) | <0.001 |
| Peak | 1.3 (0.9–3) | 1.1 (0.8–1.5) | 3.4 (1.4–6.8) | 2.9 (1.4–5.4) | <0.001 | |
| Blood urea nitrogen (n=876) | Initial | 18 (302–302) | 15 (99–99) | 17 (80–80) | 27 (123–123) | <0.001 |
| Peak | 29 (376–376) | 19 (139–139) | 73 (88–88) | 60.5 (149–149) | <0.001 | |
This table displays median laboratory findings with interquartile ranges in 887 patients with both initial and peak laboratory findings. The median values for the total cohort are displayed, as well as those for the following 3 groups: (1) patients who were living and did not require mechanical ventilation, (2) patients who were living and required mechanical ventilation, and (3) patients who expired. Comparisons are performed between patients with a favorable outcome (living and not requiring mechanical ventilation) versus a composite of those who expired or required mechanical ventilation. Elevated hs‐cTnT, elevated NT‐proBNP, elevated inflammatory markers, and abnormal kidney function were associated with composite adverse outcomes on univariate analysis. hs‐cTnT indicates high sensitivity cardiac troponin T; NT‐proBNP, N‐terminal pro‐B‐type natriuretic peptide; and WBC, white blood cell count.
Electrocardiography and Cardiovascular Biomarkers
Rate and Rhythm
The ECG findings in this cohort are summarized in Table 3. The median ventricular rate was 90 beats per minute. Atrial rhythms included sinus rhythm (n=582, 66%), sinus tachycardia (n=226, 25%), atrial fibrillation (n=40, 5%), and atrial flutter (n=6, 1%). Ectopic atrial and ventricular beats were present in 67 (8%) and 48 patients (5%), respectively. Patients with AF/AFL as compared to patients with sinus rhythm, ectopic atrial rhythm, or atrially paced rhythm (P<0.001) were at higher risk for adverse outcomes. Of the 46 patients with AF/AFL on the presenting ECG, 27 (59%) expired in comparison to 181 (21%) of 841 patients with sinus rhythm, ectopic atrial rhythm, or atrially paced rhythm (P<0.001) (Figure 2). Of the 46 patients with AF/AFL on admission, 25 (54%) had pre‐existing AF/AFL and, in 21 (46%), it was a new diagnosis. Mortality was similar between pre‐existing AF/AFL (14 patients, 56%) and newly diagnosed AF/AFL (13 patients, 62%).
Table 3.
Electrocardiography Findings in Patients With COVID‐19
| Total | % | Living, No Mechanical Ventilation | % | Living, Required Mechanical Ventilation | % | Expired | % | P Value (Expired or Ventilated vs. Favorable Outcome) | |
|---|---|---|---|---|---|---|---|---|---|
| Total | 887 | 556 | 63 | 123 | 14 | 208 | 23 | ||
| Normal sinus rhythm | 583 | 66 | 389 | 70 | 80 | 65 | 114 | 55 | 0.002 |
| Sinus bradycardia | 15 | 2 | 13 | 2 | 0 | 0 | 2 | 1 | 0.043 |
| Sinus tachycardia | 225 | 25 | 131 | 24 | 39 | 32 | 55 | 26 | 0.067 |
| AF/AFL | 46 | 5 | 15 | 3 | 4 | 3 | 27 | 13 | <0.001 |
| Paced rhythm | 12 | 1 | 6 | 1 | 0 | 0 | 6 | 3 | 0.434 |
| Other | 6 | 1 | 2 | 0 | 0 | 0 | 4 | 2 | 0.269 |
| Intervals | |||||||||
| PR interval, ms | 150 | 150 | 148 | 150 | 0.238 | ||||
| QRS interval, ms | 86 | 86 | 86 | 88 | <0.001 | ||||
| QT, ms | 364 | 370 | 358 | 361 | 0.529 | ||||
| QTc (Bazett), ms | 445 | 442 | 441 | 452 | 0.019 | ||||
| QTc (Fridericia), ms | 416 | 415 | 408 | 422 | 0.365 | ||||
| Other ECG findings | |||||||||
| Ectopic atrial beat | 67 | 8 | 33 | 6 | 3 | 2 | 31 | 15 | 0.032 |
| Ectopic ventricular beat | 48 | 5 | 23 | 4 | 2 | 2 | 23 | 11 | 0.180 |
| QTc < 460 ms | 603 | 74 | 395 | 71 | 91 | 74 | 117 | 56 | 0.004 |
| QTc ≥ 460 ms | 209 | 26 | 130 | 23 | 23 | 19 | 56 | 27 | 0.86 |
| QTc < 500 ms | 44 | 59 | 19 | 3 | 7 | 6 | 18 | 9 | 0.006 |
| QTc ≥ 500 ms | 31 | 41 | 12 | 2 | 2 | 2 | 17 | 8 | 0.005 |
| LVH | 99 | 11 | 51 | 9 | 12 | 10 | 36 | 17 | 0.015 |
| Low voltage | 30 | 3 | 19 | 3 | 2 | 2 | 9 | 4 | 1.0 |
| RBBB | 25 | 3 | 15 | 3 | 4 | 3 | 6 | 3 | 1.0 |
| RBBB + LAFB or LPFB | 17 | 2 | 4 | 1 | 2 | 2 | 11 | 5 | <0.001 |
| LBBB | 13 | 1 | 3 | 1 | 1 | 1 | 9 | 4 | 0.003 |
| IVCD | 10 | 1 | 5 | 1 | 2 | 2 | 3 | 1 | 0.71 |
| Paced rhythm | 10 | 1 | 4 | 1 | 0 | 0 | 6 | 3 | 0.13 |
| ST‐T wave analysis (N) | 812 | 525 | 114 | 173 | |||||
| Normal ST and T wave | 506 | 62 | 346 | 66 | 68 | 60 | 92 | 53 | 0.003 |
| Abnormal ST or T wave | 306 | 38 | 179 | 34 | 46 | 40 | 81 | 47 | 0.003 |
| Significant ST depression | 31 | 4 | 15 | 3 | 5 | 4 | 11 | 6 | 0.083 |
| Significant ST elevation | 11 | 1 | 5 | 1 | 3 | 3 | 3 | 2 | 0.890 |
| Significant ST depression or elevation | 42 | 5 | 20 | 4 | 8 | 7 | 14 | 8 | 0.015 |
This table describes the distribution of various ECG abnormalities in 887 patients with univariate analysis comparing patients who were admitted and did not require mechanical ventilation versus a composite of those who expired or were living but required mechanical ventilation. Analysis of ST and T wave abnormalities was restricted to the 812 patients with QRS duration <120 ms, and significant ST depressions or elevations were defined as ≥1 mm deviation 80 ms after the J point in two contiguous leads. AF/AFL indicates atrial fibrillation or atrial flutter; IVCD indicates nonspecific interventricular conduction delay; LAFB, left anterior fascicular block; LVH, left ventricular hypertrophy, LBBB, left bundle branch block; LPFB, left posterior fascicular block; and RBBB, right bundle branch block.
Figure 2. Kaplan‐Meier estimates of survival by presence of absence of atrial fibrillation or atrial flutter (AF/AFL) on presenting ECG.
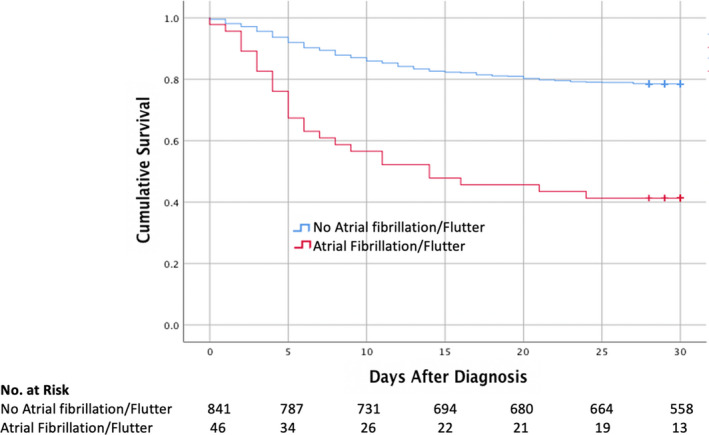
This figure demonstrates Kaplan‐Meier survival estimates at 30 days for patients stratified by presence (n=46) or absence (n=841) of AF/AFL on the presenting ECG. A total of 27 patients (59%) with AF/AFL on their presenting ECG expired, as compared to 181 (21%) of 841 patients without AF/AFL (P<0.001).
QRS Complex, ST Segment, and T Wave Analysis
Specific QRS abnormalities included pathologic Q waves (n=71, 8%), left ventricular hypertrophy (LVH) (n=99, 11%), low QRS voltage (n=30, 3%), right bundle branch block (RBBB) (n=25, 3%), RBBB + either left anterior fascicular block or left posterior fascicular block (n=17, 2%), left bundle branch block (LBBB) (n=13, 1%), nonspecific interventricular conduction delay (IVCD) (n=10, 1%), and ventricular pacing (n=10, 1%). ST segment and T wave abnormalities were assessed in a total of 812 patients with a QRS duration <120 ms. Nonspecific ST changes were seen in 82 patients (10%) and T wave abnormalities were observed in 235 patients (29%). The ST segment was significantly abnormal in 42 patients (5%), with 31 (3%) having ST depressions and 11 (1%) having ST elevations ≥1 mm in 2 contiguous leads. Patients with ST‐T wave abnormalities were at higher risk of composite adverse outcomes than patients with normal ST and T waves (P<0.001). Of the 11 patients with ST elevations, the ECG showed diffuse ST elevations in 7 patients and regional elevations in 4 patients. Of those patients with ST elevations who had further cardiovascular work‐up, 5 had an echocardiogram with 2 showing reduced and 3 having preserved left ventricular systolic function. None of the 11 patients underwent coronary angiography.
A prolonged QT was present in 240 patients (27%) as defined by a QTc >460 ms in patients with narrow QRS complexes and >500 ms in patients with QRS duration ≥120 ms.
ECG, hs‐cTnT, and Outcomes
The median initial hs‐cTnT was 13 ng/L (IQR 7–31) in patients with normal ST and T waves, 22 ng/L (IQR 9–50.5) in patients with T wave abnormalities or nonspecific ST changes, 30 ng/L (IQR 17–75) in patients with ST depressions, and 76 ng/L (IQR 14.5–125.5) in patients with ST elevations. Similarly, there was a signal towards higher hs‐cTnT in patients with other severe ECG abnormalities, such as QRS widening (Table 4). In an analysis of 830 patients who underwent both an ECG within 2 days of presentation and hs‐cTnT testing, the combination of an abnormal ECG and hs‐cTnT ≥20 was associated with increased 30‐day mortality, with death occurring in 132 of 267 patients (49%) with both abnormal features versus 15 of 264 (6%) patients with a normal ECG and hs‐cTnT <20 ng/L (P<0.001) (Figure S1).
Table 4.
ECG Findings and High Sensitivity Cardiac Troponin T
| ECG finding | n | hs‐cTnT, median (IQR) |
|---|---|---|
| QRS ≥120 ms | 71 | 52 (23–90) |
| QRS <120 ms | 756 | 14 (7–35) |
| Normal sinus rhythm | 545 | 15 (8–37) |
| Non‐normal sinus rhythm | 282 | 17 (7–47) |
| AF/AFL | 45 | 44 (20–87) |
| Atrial ectopic beats, any | 60 | 30 (16–69) |
| Ventricular ectopic beats, any | 43 | 30 (16–64) |
| Q wave infarct | 67 | 28 (13–59) |
| Left ventricular hypertrophy | 92 | 28 (13–60) |
| Normal ST‐T wave* | 471 | 13 (7–31) |
| T wave abnormality or nonspecific ST change* | 245 | 22 (9–51) |
| ST depression ≥1 mm* | 29 | 30 (17–75) |
| ST elevation ≥1 mm* | 11 | 76 (15–126) |
This table displays ECG findings, the total number of patients with each ECG finding that had a high sensitivity cardiac troponin T (hs‐cTnT) measured during their hospitalization, and the median initial hs‐cTnT in ng/L in patients with each initial ECG findings with interquartile ranges. *Analysis of ST and T wave changes was restricted to patients with QRS duration <120 milliseconds. AF/AFL indicates atrial fibrillation or atrial flutter.
Using a Cox regression model that included age, sex, all comorbidities, abnormal hs‐cTnT ≥20 ng/L, ECG abnormalities subcategorized by a rhythm diagnosis of AF/AFL, abnormal QRS morphology, and ST and T wave abnormalities, the findings of older age, an elevated hs‐cTnT, a history of coronary artery disease, active cancer, and a rhythm diagnosis of AF/AFL were significantly associated with mortality (Table 5).
Table 5.
Cox Regression Model
| Variable | Hazard Ratio (95% CI) | Beta Coefficient | Standard Error | Significance | |
|---|---|---|---|---|---|
| AF/AFL | 2.07 | (1.33–3.23) | 0.73 | 0.23 | 0.001 |
| QRS abnormality | 1.09 | (0.75–1.59) | 0.08 | 0.19 | 0.662 |
| ST‐T wave abnormality | 1.06 | (0.79–1.43) | 0.06 | 0.15 | 0.692 |
| Initial hs‐cTnT ≥20 ng/L | 4.57 | (3.02–6.92) | 1.52 | 0.21 | <0.001 |
| Age per year | 1.04 | (1.03–1.05) | 0.04 | 0.01 | <0.001 |
| Male sex | 1.25 | (0.91–1.72) | 0.22 | 0.16 | 0.169 |
| Hypertension | 1.03 | (0.69–1.52) | 0.03 | 0.20 | 0.892 |
| Diabetes mellitus | 1.32 | (0.96–1.80) | 0.28 | 0.16 | 0.084 |
| CKD | 1.06 | (0.75–1.52) | 0.06 | 0.18 | 0.732 |
| Primary lung disease | 1.27 | (0.86–1.87) | 0.24 | 0.20 | 0.232 |
| Coronary artery disease | 1.56 | (1.04–2.33) | 0.44 | 0.21 | 0.032 |
| Obesity | 1.16 | (0.83–1.62) | 0.14 | 0.17 | 0.401 |
| HFrEF | 1.23 | (0.73–2.07) | 0.21 | 0.27 | 0.442 |
| HFpEF | 1.10 | (0.62–1.96) | 0.10 | 0.30 | 0.748 |
| Active cancer | 1.87 | (1.00–3.48) | 0.63 | 0.32 | 0.049 |
| History of cancer | 1.10 | (0.64–1.89) | 0.09 | 0.28 | 0.742 |
In this Cox regression model, initial high sensitivity troponin T (hs‐cTnT) ≥20, older age, a rhythm diagnosis of AF/AFL versus sinus rhythms, ectopic atrial rhythms, and atrially paced rhythms, a history of CAD, and active cancer were associated with increased morality. The variable effect size of ECG abnormalities on 30‐day mortality based on whether initial high sensitivity cardiac troponin T (hs‐cTnT) was >20 ng/L was further explored in this model. Three separate interaction terms (between elevated hs‐cTnT + each individual ECG abnormality [AF/AFL, QRS abnormalities, and ST‐T wave abnormalities] were tested. These 3 interaction terms were added to the model alone (without the other 2 interaction terms) or together (all 3 interaction terms added to the model). Neither of these interaction terms were significant. These interaction terms were therefore omitted from the model. AF/AFL indicates atrial fibrillation or atrial flutter, CKD indicates stage 3 or greater chronic kidney disease, HFrEF indicates heart failure with reduced ejection fraction which was defined as a clinical diagnosis of systolic heart failure or a baseline echocardiogram with left ventricular ejection fraction <50%, HFpEF indicates heart failure with preserved ejection fraction.
Discussion
The heart is a unique organ, composed of myofibers, with its own conduction system. COVID‐19 can damage myocardium either through thrombosis leading to ischemia and infarction, or myocarditis through inflammation, but evidently it may also cause cardiac arrhythmias. As found in prior studies, 6 , 14 we found a high rate of cardiac injury in hospitalized patients with COVID‐19 as defined by elevated hs‐cTnT. Additionally, we identified a wide range of abnormal ECG findings, including Q wave infarcts, atrial arrhythmias, ectopic beats, and ST and T wave changes, all of which were more commonly present in patients who experienced adverse outcomes. This study suggests that when COVID‐19 adversely affects both myocardium and cardiac conduction together, it is indicative of more profound cardiovascular involvement and is predictive of increased risk for death. Namely, the combination of elevated cardiac troponin and AF/AFL on ECG multivariable modeling was most significantly associated with increased mortality.
The associations between elevated troponin and AF/AFL with adverse outcomes have several possible explanations. First, direct COVID‐19 myocarditis may occur, and case studies have been published of patients with clinically diagnosed COVID‐19‐related myocarditis 15 or myopericarditis. 16 Secondly, the systemic coagulopathy that has been documented in severe COVID‐19 infection could lead to microvascular or epicardial coronary thrombosis. 17 Thirdly, COVID‐19 may result in a stress cardiomyopathy and cardiac injury from systemic inflammation, either as an isolated entity or accompanied by multi‐system organ failure with hypoxemia and shock. Finally, systemic inflammation can also lead to increased sympathetic activation, which may precipitate serious cardiac arrhythmias, such as AF/AFL. As some of these pathways may be targeted with specific therapies, such as anticoagulation or anti‐inflammatory therapy, determining underlying mechanisms remains critical. The pattern of ECG abnormalities may provide important clues to the underlying pathophysiology, such as diffuse ST elevations seen in myocarditis or focal changes that could indicate coronary thrombosis. During the initial phases of the pandemic, overwhelming patient volumes, scare resources, and concern for staff safety has limited the use of advanced cardiovascular imaging in patients with COVID‐19. As we move into the next phase of the pandemic, increased pathologic study by autopsy or endomyocardial biopsy could be combined with more widespread use of cardiovascular imaging with echocardiography, cardiac magnetic resonance imaging, and angiography to more fully delineate the pathophysiologic mechanisms responsible for cardiac injury and guide further study and treatment.
Finally, it is possible that the troponin and AF/AFL seen in this study may be surrogates of more advanced age and pre‐existing cardiovascular comorbidities. We therefore attempted to control for several important comorbidities and age, in addition to ECG categorization and hs‐cTnT, using Cox proportional hazards models. After multivariable adjustment, we found that elevated hs‐cTnT and AF/AFL remained significantly associated with adverse outcomes. As a result, these tests provide important prognostic information, even after controlling for baseline comorbidities, and clearly identify a high‐risk group of patients with COVID‐19 who warrant aggressive monitoring and treatment. It is possible that inclusion of additional laboratory findings, such as other inflammatory markers, renal function, and markers of coagulopathy, into risk models would provide further prognostic accuracy.
Study Limitations
As a retrospective study of an ongoing pandemic, this study has significant limitations. (1) Some patients remain hospitalized and their final outcomes are unknown. The inclusion of 30‐day mortality data helps to reduce the impact of this limitation, but prolonged hospitalizations with severe COVID‐19 are common and some late adverse outcomes may be missed. (2) Patients who are discharged home are assumed to have good outcomes if re‐admission did not occur at our medical center, and it is probable that re‐admissions and adverse outcomes (eg, death) that occur at home or other centers were incompletely captured. (3) Data were abstracted from the medical record, and comorbidities are likely incompletely characterized. (4) This analysis begins at the time of presentation to the hospital, and the timing of the onset of symptoms and the relationship to the time of presentation is not available. (5) This study does not systematically include cardiac imaging findings, which would provide additional clues as to the pathophysiology of cardiac involvement. (6) The analyzed initial ECGs were obtained within 2 days of admission. It is possible that treatment with medications with possible cardiac toxicity and QT prolongation, such as azithromycin and hydroxychloroquine, would have been administered by this time and could affect the ECG results. (7) We excluded patients from the present analysis who did not have ECGs performed within 2 days of admission. It is possible that this may reduce generalizability to all admitted patients with COVID‐19.
Conclusions
Cardiac injury in the setting of COVID‐19 can lead to myocardial injury and rhythm disturbances, and the findings of both an elevated cardiac troponin and atrial arrhythmias on admission portends an ominous prognosis. Further study is needed to clarify the mechanisms of cardiovascular involvement in COVID‐19.
Sources of Funding
Dr. Madhavan has received support from an institutional grant by the National Institutes of Health/National Heart, Lung, and Blood Institute to Columbia University Irving Medical Center (T32 HL007854). KJC is supported by NIH/NHLBI Grant K23 HL148528. EYW was supported by NIH R01 HL152236, the Esther Aboodi Endowed Professorship at Columbia University, and the Wu Family Research fund.
Disclosures
TJP owns stock in Abbott Laboratories, AbbVie, Inc, Baxter International, and Edwards Lifesciences, has received research funding from the Amyloidosis Foundation David Seldin Memorial Junior Research Grant, and has research grant funding pending from EIDOS Therapeutic, which is unrelated to this research.
Supporting information
Figures S1
Acknowledgments
We would like to thank the NewYork‐Presbyterian Hospital’s nurses, nurse practitioners, physician assistants, and residents for their heroic efforts during the pandemic.
(J Am Heart Assoc. 2021;10:e018476. DOI: 10.1161/JAHA.120.018476.)
Supplementary Material for this article is available at https://www.ahajournals.org/doi/suppl/10.1161/JAHA.120.018476
For Sources of Funding and Disclosures, see page 11.
References
- 1. Huang C, Wang Y, Li X, Ren L, Zhao J, Hu YI, Zhang LI, Fan G, Xu J, Gu X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, Xiang J, Wang Y, Song B, Gu X, et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Grasselli G, Zangrillo A, Zanella A, Antonelli M, Cabrini L, Castelli A, Cereda D, Coluccello A, Foti G, Fumagalli R, et al. Baseline characteristics and outcomes of 1591 patients infected with SARS‐CoV‐2 admitted to ICUs of the lombardy region, Italy. JAMA. 2020;323:1574–1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Team CC‐R . Preliminary estimates of the prevalence of selected underlying health conditions among patients with coronavirus disease. United States, February 12‐March 28, 2020. MMWR Morb Mortal Wkly Rep. 2020;69(13):382–386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Goyal P, Choi JJ, Pinheiro LC, Schenck EJ, Chen R, Jabri A, Satlin MJ, Campion TR, Nahid M, Ringel JB, et al. Clinical characteristics of Covid‐19 in New York City. N Engl J Med. 2020;382:2372–2374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Guo T, Fan Y, Chen M, Wu X, Zhang L, He T, Wang H, Wan J, Wang X, Lu Z. Cardiovascular implications of fatal outcomes of patients with coronavirus disease 2019 (COVID‐19). JAMA Cardiol. 2020;5:811. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lippi G, Plebani M. Laboratory abnormalities in patients with COVID‐2019 infection. Clin Chem Lab Med. 2020;58:1131–1134. [DOI] [PubMed] [Google Scholar]
- 8. Chen T, Wu DI, Chen H, Yan W, Yang D, Chen G, Ma KE, Xu D, Yu H, Wang H, et al. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368:m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Kochav SM, Coromilas E, Nalbandian A, Ranard LS, Gupta A, Chung MK, Gopinathannair R, Biviano AB, Garan H, Wan EY. Cardiac arrhythmias in COVID‐19 infection. Circ Arrhythm Electrophysiol. 2020;13:e008719. DOI: 10.1161/CIRCEP.120.008719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Driggin E, Madhavan MV, Bikdeli B, Chuich T, Laracy J, Biondi‐Zoccai G, Brown TS, Der Nigoghossian C, Zidar DA, Haythe J, et al. Cardiovascular considerations for patients, health care workers, and health systems during the COVID‐19 pandemic. J Am Coll Cardiol. 2020;75:2352–2371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Clerkin KJ, Fried JA, Raikhelkar J, Sayer G, Griffin JM, Masoumi A, Jain SS, Burkhoff D, Kumaraiah D, Rabbani LeRoy, et al. COVID‐19 and cardiovascular disease. Circulation. 2020;141:1648–1655. [DOI] [PubMed] [Google Scholar]
- 12. Madjid M, Safavi‐Naeini P, Solomon SD, Vardeny O. Potential effects of coronaviruses on the cardiovascular system: a review. JAMA Cardiol. 2020;5:831–840. [DOI] [PubMed] [Google Scholar]
- 13. Tavazzi G, Pellegrini C, Maurelli M, Belliato M, Sciutti F, Bottazzi A, Sepe PA, Resasco T, Camporotondo R, Bruno R, et al. Myocardial localization of coronavirus in COVID‐19 cardiogenic shock. Eur J Heart Fail. 2020;22:911–915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Shi S, Qin MU, Shen BO, Cai Y, Liu T, Yang F, Gong W, Liu XU, Liang J, Zhao Q, et al. Association of cardiac injury with mortality in hospitalized patients with COVID‐19 in Wuhan, China. JAMA Cardiol. 2020;5(7):802. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Inciardi RM, Lupi L, Zaccone G, Italia L, Raffo M, Tomasoni D, Cani DS, Cerini M, Farina D, Gavazzi E, et al. Cardiac involvement in a patient with coronavirus disease 2019 (COVID‐19). JAMA Cardiol. 2020;5:819. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Hua A, O'Gallagher K, Sado D, Byrne J. Life‐threatening cardiac tamponade complicating myo‐pericarditis in COVID‐19. Eur Heart J. 2020;41:2130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bikdeli B, Madhavan MV, Jimenez D, Chuich T, Dreyfus I, Driggin E, Nigoghossian CD, Ageno W, Madjid M, Guo Y, et al. COVID‐19 and thrombotic or thromboembolic disease: Implications for prevention, antithrombotic therapy, and follow‐up: JACC state‐of‐the‐art review. J Am Coll Cardiol. 2020;75:2950–2973. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figures S1


