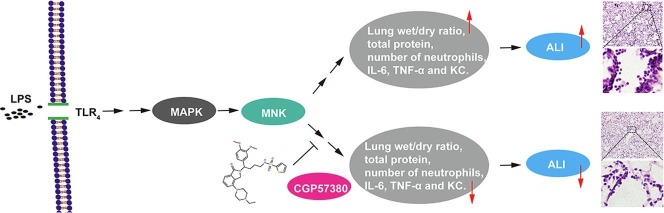
Graphical abstract
Keywords: Acute lung injury, MAPKs, MNK, CGP57380, Inflammatory cytokines
Abstract
Acute lung injury (ALI) or its more severe form, known as acute respiratory distress syndrome (ARDS), is characterized by an initial exudative phase, expression of proinflammatory mediators, activation of inflammatory leukocytes, and impairment of the lung endothelium and epithelium. Despite numerous, novel therapeutic strategies have been developed regarding the pathophysiology of ALI, current treatment is mainly supportive, as specific therapies have not been established in the past few decades. The MAP kinase-interacting kinases (MNK1 and MNK2) are serine threonine kinases which are activated by mitogen-activated protein kinases (MAPKs), regulate protein synthesis by phosphroylating eukaryotic translation initiation factor 4E (eIF4E). Although studies have shown that MAPKs pathway is involved in anti-inflammatory and preventing tissue injury processes, the role of MNKs in ALI has, until now, remained relatively unexplored. Here, we investigated whether partial inhibition of MAPKs pathway by targeting MNKs was effective in the prevention and treatment of ALI. C57BL6 mice were pretreated with MNK1 and MNK2 inhibitor (CGP57380, 30 mg/kg) for 30 min and then challenged with 5 mg/kg LPS for 6 h. The results showed that pretreatment with CGP57380 not only significantly attenuated LPS-induced lung wet/dry ratio, as well as protein content, total cells and neutrophils in bronchoalveolar lavage fluid (BALF), but also decreased the production of pro-inflammatory mediators such as interleukin-6 (IL-6), tumor necrosis factor alpha (TNF-α) and keratinocyte-derived chemoattractant (KC). In addition, CGP57380 was observed to significantly suppress LPS-stimulated phosphorylation of eIF4E and MAPKs in the mouse bone marrow-derived macrophages (BMDMs). The involvement of MNK2 in lung injury was further evident by MNK2 knockout mice. MNK2 deficiency resulted in the attenuated lung histopathological changes, as also reflected by reductions in neutrophil counts, and the less LPS-induced the production of IL-6, TNF-α and KC in mouse BALF. Taken together, these findings demonstrated for the first time that MNK inhibition could effectively reduce the LPS-induced ALI in mice, suggesting a novel and potential application for MNK-based therapy to treat this serious disease.
1. Introduction
Acute lung injury (ALI) and the more severe acute respiratory distress syndrome (ARDS) are the severe pulmonary inflammatory disease which occurs due to various environmental triggers, characterized by rapid alveolar injury, pulmonary infiltrates, severe hypoxemia, uncontrolled inflammatory response, and neutrophil accumulation, with persistent high morbidity and mortality in the critically ill patients [1], [2], [3]. It occurs mainly in young adults, previously fit people, and is responsible for thousands of pediatric and adult deaths worldwide each year. It is estimated that ALI accounts for approximately 79,000 deaths per year in the United States which would make it one of the top 8 causes of death in 2018. Both ALI and ARDS bear a large long-term illness and disability burden on the individual sufferer and on society [4]. Despite extensive research, profound understanding of ALI pathogenesis and recent advances in therapeutic strategies being trialed, mortality rates from ALI remain high at approximately 30% or 40% [5], [6]. This means the discovery and development of effective pharmacotherapies remain a major focus for research.
Pulmonary injury is a complex and multidimensional process, involving dysfunction of alveolar-capillary barrier, activation of immune and inflammatory system, infiltration of neutrophils and macrophages, and leaking of alveolar albumin [7], [8]. Research efforts in the field of ALI have primarily focused on the innate immune system and have typically conceptually viewed ALI as a syndrome of hyper-inflammation. Emerging evidence indicates that dysregulation of cytokines in acute inflammation is a crucial step in mediating, amplifying, and perpetuating ALI processes [1], [2], [3], [4], [9]. Lipopolysaccharide (LPS), a main component of bacterial cell walls, has been identified as a key factor in ALI development, which can trigger a rapid and robust inflammatory response and impair the normal function of immune cells, such as macrophages. Results from previous studies showed that continuous activation of macrophages was one of the key steps responsible for accelerating ALI progress [9], [10], [11], [12]. Macrophages perform multiple functions at different stages of ALI through the production of pro-inflammatory mediators including chemokines and cytokines such as interleukin-6 (IL-6), interleukin-12 (IL-12) and tumor necrosis factor alpha (TNF-α) [13], [14], [15]. Toll-like receptor 4 (TLR4) is a transmembrane protein encoded by the TLR4 gene, which is involved in the innate immune response [16], [17], [18]. A number of studies have shown that LPS is the ligand of TLR4 and stimulates the inflammatory response of the lungs by binding to TLR4 [19], [20], [21], [22]. It is now verified that TLR4-mediated mitogen-activated protein kinases (MAPKs) signaling pathways are responsible for the production of a variety of pro-inflammatory cytokines [23], [24], [25], [26]. Following that, expression levels of IL-6, TNF-α and other pro-inflammatory cytokines are considerably enhanced [24]. Thus, blocking the TLR4-mediated MAPKs signaling pathways can inhibit the development of ALI induced by LPS.
The MAP kinase-interacting kinases (MNK1 and MNK2) are serine threonine kinases which are activated by MAPKs, regulating protein synthesis by phosphroylating eukaryotic translation initiation factor 4E (eIF4E) [27], [28], [29]. Although numerous studies have shown that MAPK pathways are involved in the anti-inflammatory and tissue injury preventing processes [30], the role of MNKs in ALI has remained relatively unexplored until now. Here, we investigated whether partial inhibition of MAPK pathways by targeting MNKs was effective in the prevention and treatment of ALI, and potential mechanisms that might be involved.
2. Materials and methods
2.1. Materials and reagents
Most chemical reagents including LPS, CGP57380, pentobarbital sodium, DMSO, paraformaldehyde, and H&E staining solution were purchased from Sigma-Aldrich China Branch (Shanghai, China). The cell culture reagents including DMEM, penicillin, streptomycin, trypsin, and phosphate-buffered saline were purchased from Life Technologies of Thermo Fisher Scientific (China) (Shanghai, China). Fetal bovine serum (FBS) was purchased from EVERY GREEN by Zhejiang Tianhang Biotechnology (Hangzhou, Zhejiang, China). The antibodies against eIF4E, phospho-eIF4E, phospho-ERK, phospho-JNK and phospho-p38 MAPK used in Western blotting were purchased from Cell signaling Technology (Beverly, MA, USA). The FITC-labeled anti-mouse Ly-6G (Gr-1) antibody was obtained from eBioscience (Grand Island, NY). The GAPDH antibody was purchased from Proteintech (Wuhan, Hubei, China). The secondary antibody (horseradish peroxidase-conjugated goat anti-rabbit IgG antibody) was obtained from Mei5 Biotechnology (Beijing, China). The enhanced chemiluminescence (ECL) kit was purchased from Thermo Fisher Scientific (China) (Shanghai, China).
2.2. Animals model
All experimental procedures were approved by the Institutional Animal Care and Use Committee of Wuhan University, and all experiments were performed according to the Guide for the Care and Use of Laboratory Animals. Eight week-old wild-type (MNK2+/+, purchased from Beijing Vital River Laboratory Animal Technology Co., Ltd., Beijing, China) and MNK2 deficient (MNK2−/−, obtained from Center for Animal Experiment, Wuhan University, China) male C57BL6 mice (18–20 g) were housed in a conventional animal facility at constant 21 ℃ in a 12-h light–dark cycle with unrestricted access to food and water. To induce ALI, after anesthesia with intraperitoneally injection of 50 mg/kg pentobarbital sodium, LPS (5 mg/kg) was intratracheally instilled in wild-type and MNK2-knockout mice. Before LPS exposure, some wild-type mice were intraperitoneally injected with at 30 mg/kg of CGP57380 or DMSO for 30 min. The ALI and lung inflammation were determined in the mice by measuring alveolar exudate, neutrophil infiltration, and lung wet/dry weight ratio. Five mice were used in each group.
2.3. Leukocytes in the bronchoalveolar lavage fluid (BALF)
After the lungs had been washed with 0.5 ml of sterile saline three times, BALF samples were collected as previously described [31]. After lysis of red cells, the remaining cells were washed, then resuspended with PBS and counted. For neutrophil analysis in BALF, FITC-labeled anti-mouse Ly-6G (Gr-1) antibody was used for labeling, the labeled neutrophils were counted using a flow cytometer (FACScanto, BD Biosciences, San Jose, CA) and the data were analyzed with FlowJo7.6.
2.4. Lung wet/dry ratio
The lung edema was determined by the ratio of lung wet/dry weight. Fresh right lung was cleansed and weighed to obtain the wet weight and then was dried in an oven at 60 °C for at least 24 h to assess the dry weight. The left lung was collected for histopathological analysis.
2.5. Histopathological examination
Left lung tissues were fixed in 4% paraformaldehyde, embedded in paraffin, and cut into 5 μm thick sections. The sections were stained with H&E using standard protocols, as reported previously [32]. The sections were examined and photographed using a light microscope (RX51, Olympus Optical Co. Ltd., Tokyo, Japan) at × 200 magnification after staining.
2.6. Cytokine detection by ELISA
The amounts of IL-6, TNF-α and keratinocyte-derived chemoattractant (KC) in BALF were measured by commercially available ELISA kits (R&D System, Minneapolis, MN, USA) in accordance with the manufacturer’s instructions.
2.7. Cell culture
Bone marrow-derived macrophages (BMDMs) were isolated from the bones of male C57BL/6 mice. The isolated cells were cultured in DMEM supplemented with antibiotics (100 units/mL of penicillin and 100 μg/mL streptomycin), 10% FBS, and 10% culture supernatant of L929 cell-conditioned medium (as a source of Macrophage-Colony Stimulating Factor) for 7 d (37 °C, 5% CO2). Cells were treated with indicated concentrations of CGP57380; 0.1% DMSO was added into culture medium as the solvent control.
2.8. Western blot analysis
The BMDMs at 2 × 106 cells per well density in 6-well plates were pretreated with CGP57380 for 15 min, then stimulated with 100 ng/ml LPS for 1 h. The cells were homogenized in radioimmunoprecipitation assay lysis buffer (Wuhan Boster Biological Technology, Ltd., Wuhan, China) supplemented with phenylmethanesulfonyl fluoride (Beyotime, Jiangsu, China). A bicinchoninic acid extraction kit (Wuhan Boster Biological Technology, Ltd.) was used to determine the protein concentration. The 20 μg denatured protein samples were separated on SDS-PAGE followed by transfer to the nitrocellulose membrane. After blocking, the membranes were incubated with primary antibodies at 4 °C overnight. The primary antibodies were against eIF4E, phospho-eIF4E, phospho-ERK, phospho-JNK, phospho-p38 MAPK, and GAPDH. After washing, the membranes were incubated for 2 h with the secondary antibody at room temperature. The bands were visualized using enhanced chemiluminescence (ECL) stain kit and then auto-radiographed.
2.9. Statistical analysis
Unless otherwise stated, experiment results were performed at least three times independently. The ‘n’ of each group was also indicated in the figure legends. Data are presented as the means ± standard deviation (SDs). The GraphPad Prism program or one-way ANOVA followed by the Holm-Sidak’s method were used for the statistics analysis as appropriate. Differences were considered statistically significant if P < 0.05.
3. Results
3.1. MNK inhibitor CGP57380 reduces LPS induced ALI
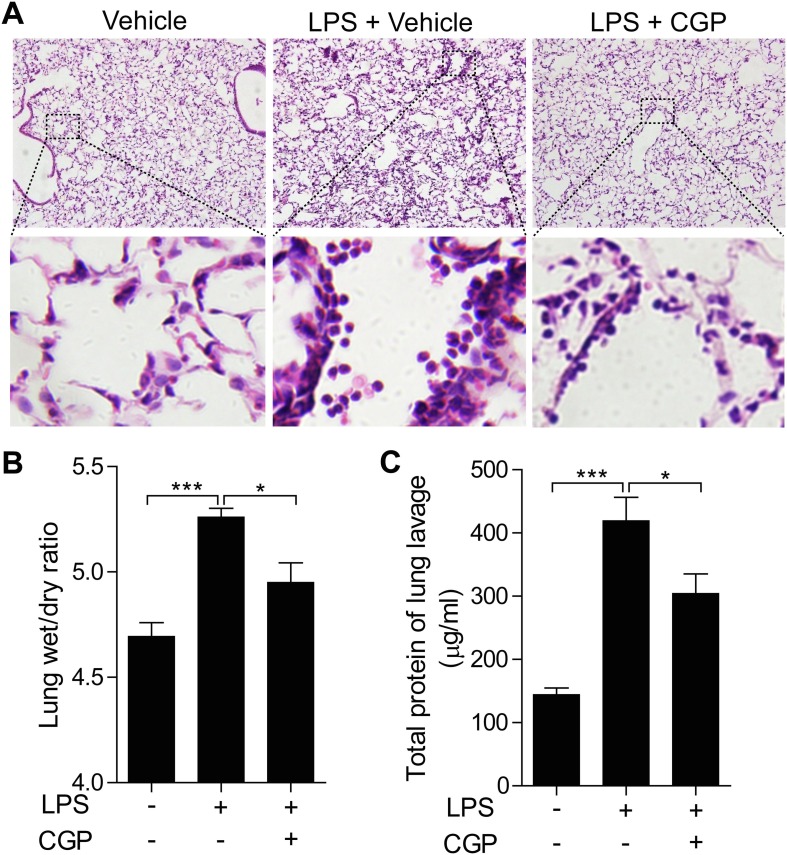
To determine the role of MNKs in the development of ALI, we first determined the effects of MNK inhibitor CGP57380 on the LPS induced mouse lung injury. CGP57380 is a potent inhibitor for both MNK1 and MNK2 with the similar IC50 value [36]. Intratracheally instillation with LPS in mice was used to introduce the ALI. The histological studies showed that LPS treatment induced the hemorrhage and infiltration of inflammatory cells in compared to the control group, and pretreatment with MNK inhibitor CGP57380 decreased both hemorrhage and inflammatory cells (Fig. 1 A). While the increase of lung wet/dry ratio caused by LPS was significantly reduced by the MNK inhibitor, the high protein contents in the BALF of LPS treated mice were also greatly decreased (Fig. 1B and 1C). Together, these data suggest that MNK inhibitor CGP57380 was able to decrease the LPS induced mouse lung injury.
Fig. 1.
CGP57380 depressed LPS-induced mouse lung injury. (A) C57BL/6 mice were pretreated with CGP57380 for 30 min and then were challenged with 5 mg/kg LPS for 6 h. H&E staining revealed typical ALI manifestation, and CGP57380 attenuated LPS-induced histopathological changes. Higher-power images listed below showed the changes of inflammatory cell infiltration and alveolar septal thickening. (B-C) Lung wet/dry ratio and total BALF protein were significantly lower in the CGP57380-treated mice than that challenged with LPS (P < 0.05). Five mice were used in each group (n = 5). Data are expressed as mean ± SD. *** P < 0.01; * P < 0.05.
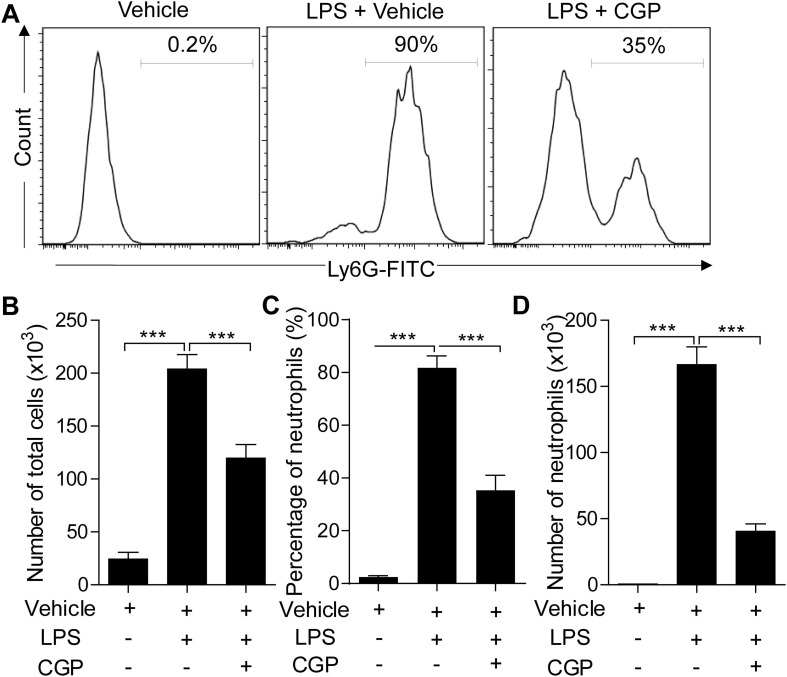
To further confirm the effects of CGP57380 on the lung injury, we next examined the infiltration of neutrophils in the BALF. The neutrophils were analyzed with flow cytometry analysis. LPS treatment induced a high number of leukocytes collected in the BALF. About 80% of the cells were the neutrophils. CGP57380 pretreatment reduced the total leukocytes and neutrophils in BALF (Fig. 2 ), indicating that the CGP57380 could prevent the infiltration of neutrophils in the alveoli after LPS induction.
Fig. 2.
CGP57380 decreased inflammatory cell accumulation in BALF from ALI mice. (A) The total BALF cells and neutrophils were measured and counted based on flow cytometry analysis. (B-C) The total cells and neutrophils were remarkably reduced in the CGP57380-treated mice than that challenged with LPS (P < 0.01). Five mice were used in each group (n = 5). Data are expressed as mean ± SD. *** P < 0.01.
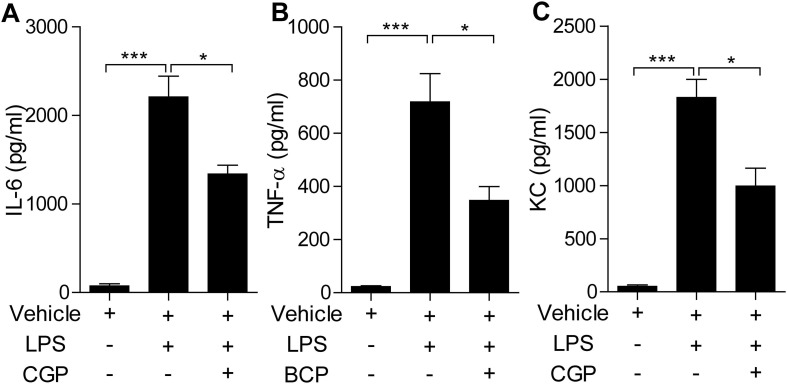
We then examined the contents of pro-inflammatory cytokines and chemokines in the BALF by ELISA. The data showed that LPS treatment induced the increase of IL-6, TNF-α, and KC in the BALF. And pretreatment of CGP57380 diminished their contents in the BALF in about half (Fig. 3 ). Together, LPS intratracheally instillation induced lung injury could be attenuated by MNK inhibitor CGP57380 pretreatment.
Fig. 3.
Cytokines and chemokine in BALF were detected by ELISA. (A) IL-6 in BALF were notably increased by LPS administration compared with vehicle one (P<0.01), whereas their levels were significantly reduced in the LPS/ CGP57380 group (P<0.05). (B) TNF-α in BALF were remarkably increased by LPS administration compared with vehicle one (P<0.01), whereas their levels were notably reduced in the LPS/ CGP57380 group (P<0.05). (C) KC in BALF were outstandingly increased by LPS administration compared with vehicle one (P<0.01), whereas their levels were considerably reduced in the LPS/ CGP57380 group (P<0.05). Five mice were used in each group (n = 5). Data are expressed as mean ± SD. *** P < 0.01; * P < 0.05.
3.2. MNK inhibitor CGP57380 decreases inflammatory response in macrophages
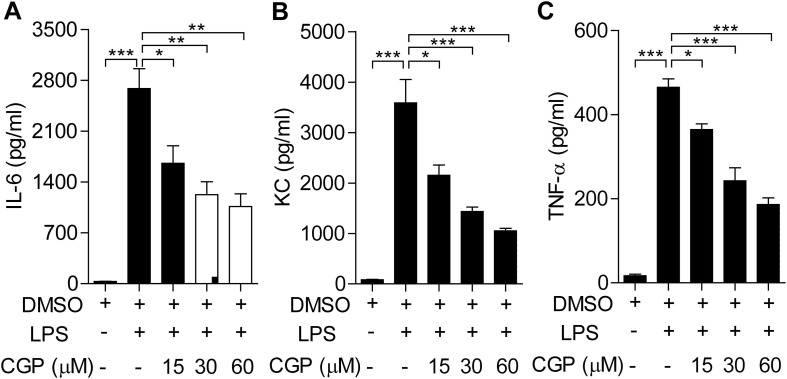
Macrophages, including alveoli residential macrophages play a pivotal role in the ALI [33], [34]. Next we determined the effects of MNK inhibitor CGP57380 on the macrophage inflammatory response after LPS stimulation. We measured the amounts of pro-inflammatory IL-6, TNF-a, and KC in BMDMs as the way to determine the macrophage inflammatory response. Challenging of BMDMs with LPS resulted in the profound increase of IL-6, KC and TNF-α, respectively. Moreover, pretreatment of the BMDMs with different concentrations of CGP57380 inhibited the increase in a concentration-dependent manner (Fig. 4 ), clearly suggesting that macrophage inflammatory response is at least partially dependent on MNKs.
Fig. 4.
CGP57380 inhibited cytokines and chemokine production in LPS-stimulated BMDMs. (A) Analysis of LPS-induced cytokines and chemokine expression. BMDMs were pretreated with 0.1% DMSO or CGP57380 as indicated for 15 min, then stimulated with 100 ng/ml LPS for 6 h. (B-D) LPS-induced expression of cytokines and chemokine was inhibited by CGP57380 in a concentration-dependent manner. Statistical analysis was performed using one-way ANOVA. n = 3. *P < 0.05, **P < 0.01, ***P < 0.001.
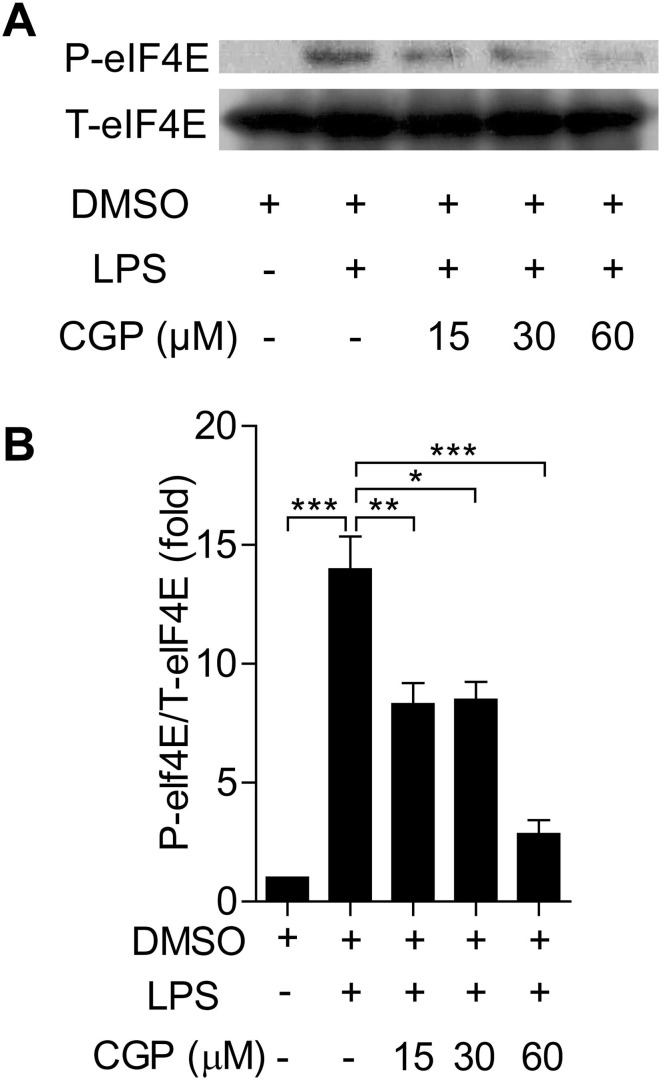
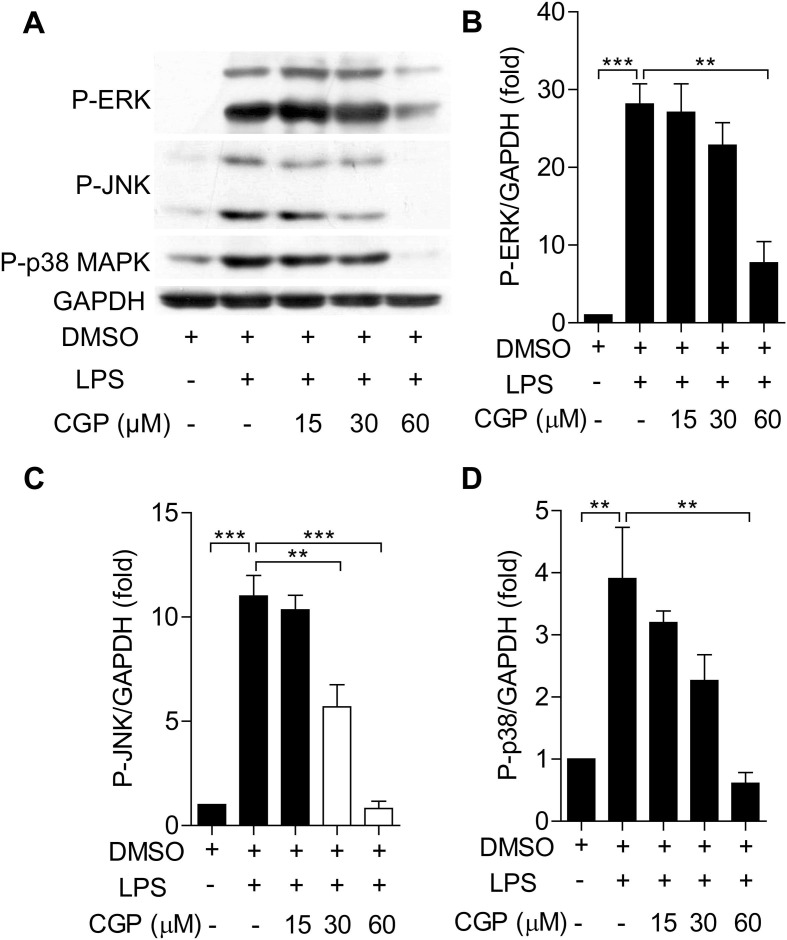
To understand the effects of MNK inhibition on the MNK upstream and downstream signal molecules, we used Western blot to determine the phosphorylation of these molecules. In Fig. 5 , challenging of BMDMs with LPS resulted in the increased levels of phosphorylated eIF4E, this increase was inhibited by the pretreatment of CGP57380 in a concentration-dependent manner. Interestingly, LPS challenging also resulted in the increased phosphorylation of MAPKs (ERK, p38, JNK), pretreatment with CGP57380 diminished the increased phosphorylation of these MAPKs at a higher concentration (Fig. 6 ). Taken together, it is likely that MNKs are involved in the inflammatory response macrophage and inhibition with CGP57380 interrupted the downstream signaling and reduced the inflammatory response.
Fig. 5.
CGP57380 decreased the phosphorylation of eIF4E in LPS-treated BMDMs. (A) Western blot analysis of LPS-induced eIF4E expression. BMDMs were pretreated with 0.1% DMSO or CGP57380 as indicated for 15 min, then stimulated with 100 ng/ml LPS for 6 h. LPS-induced expression of eIF4E was inhibited by CGP57380 in a concentration-dependent manner (A-B). Statistical analysis was performed using one-way ANOVA. n = 3. *P < 0.05, **P < 0.01, ***P < 0.001.
Fig. 6.
CGP57380 reduced the phosphorylation of MAPKs in LPS-treated BMDMs. (A) Western blot analysis of LPS-induced P-ERK, P-JNK and P-p38 expression. BMDMs were pretreated with 0.1% DMSO or CGP57380 as indicated for 15 min before stimulation of LPS (100 ng/ml). The expressions of P-ERK, P-JNK and P-p38 were detected as above. (B-D) Densitometric analysis of the ratios of P-p38/T-p38, P-ERK/T-ERK, and P-JNK/T-JNK was performed using Image software. LPS-induced expression of MAPKs was inhibited by CGP57380 in a concentration -dependent manner (A-D). Statistical analysis was performed using one-way ANOVA. n = 3. **P < 0.05, ***P < 0.01.
3.3. MNK2 knockout attenuates LPS-induced ALI
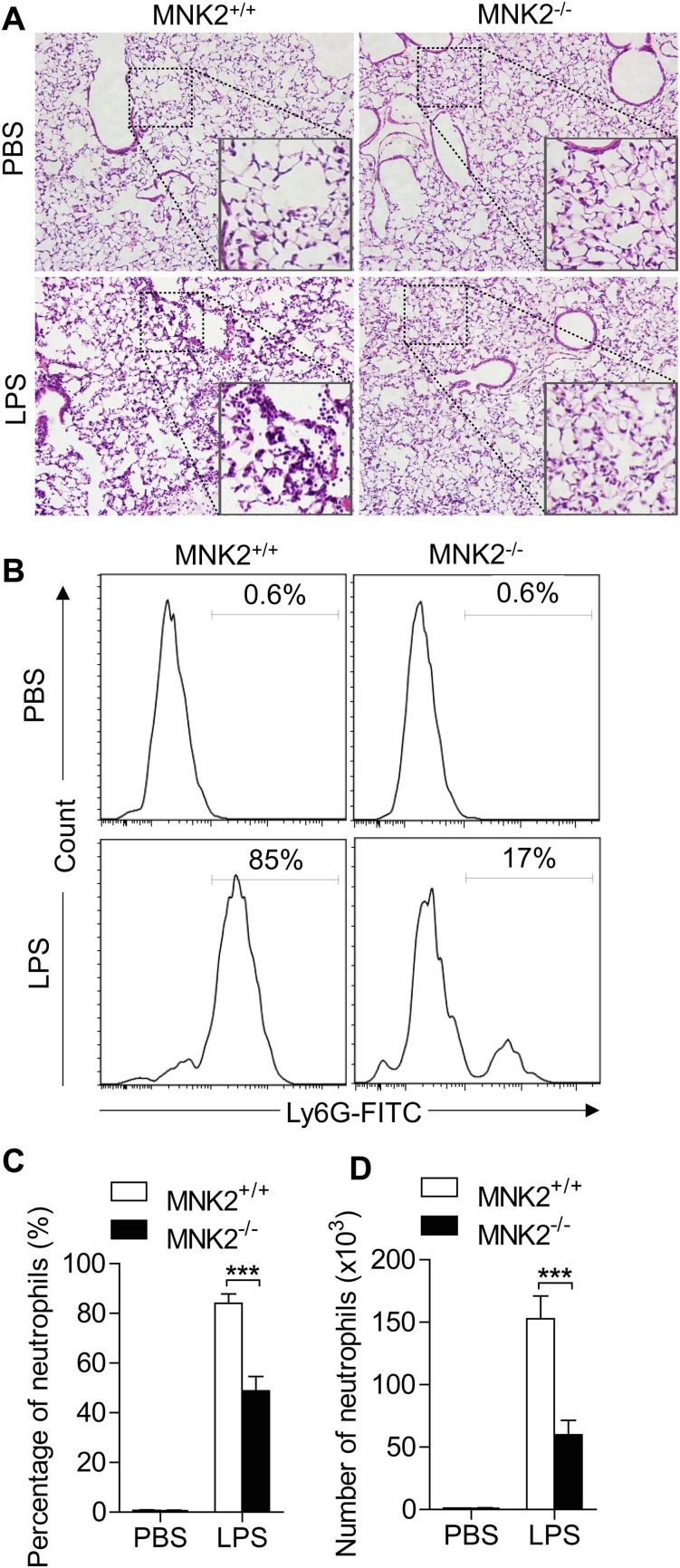
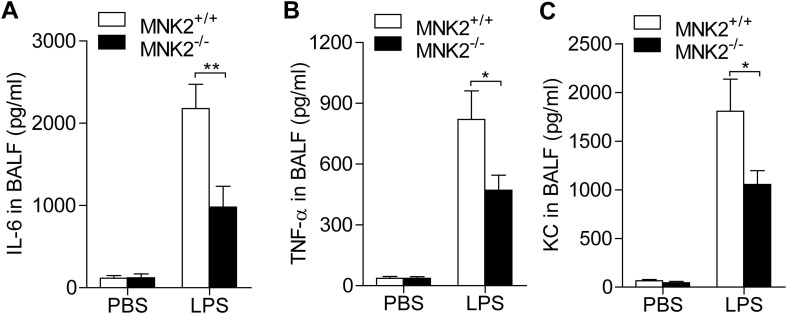
Since compound CGP57380 is able to inhibit both MNK1 and MNK2, to further determine the role of MNK2 in the lung injury, we took the advantage of MNK2 knockout mice. LPS induced lung injury was compared in the MNK2-/- and wild type mice. Histological studies showed that MNK2 deficient resulted in a much less lung injury as shown by less hemorrhage and alveoli infiltration of inflammatory cells (Fig. 7 A). Flow cytometry analysis of neutrophils in BALF showed that neutrophil alveoli infiltration was reduced at least in half (Fig. 7 B and 7C). These results confirmed the involvement of MNK2 in LPS induced lung injury. The proinflammatory cytokines in BALF were also determined. The LPS induced productions of IL-6, TNF-α, and KC were greatly decreased in the BALF of MNK2 deficient mice (Fig. 8 ).
Fig. 7.
MNK2 deficiency attenuated LPS-induced ALI in mice. (A) WT and MNK2 deficient mice were treated with 5 mg/kg LPS for 6 h. H&E staining revealed typical murine ALI manifestation in WT mice, and MNK2 deficiency attenuated LPS-induced histopathological changes. Higher-power images listed below showed the changes of inflammatory cell infiltration and alveolar septal thickening. (B) The BALF neutrophils were measured and counted based on flow cytometry analysis. (B-C) The neutrophils were remarkably reduced in MNK2 deficient mice than that in WT mice (P < 0.01). Five mice were used in each group (n = 5). Data are expressed as mean ± SD. *** P < 0.01.
Fig. 8.
MNK2 deficiency diminished lung inflammatory responses in LPS-induced mice. WT and MNK2 knockout mice were challenged with 5 mg/kg LPS for 6 h, and then cytokines and chemokine in BALF were detected by ELISA. (A) IL-6 level in MNK2 deficient mice were significantly decreased by LPS administration compared with WT ones (P<0.01). (B) TNF-α production in MNK2 deficient mice were notably reduced by LPS treatment compared with WT ones (P<0.05). (C) KC content in MNK2 deficient mice were remarkably declined by LPS management compared with WT ones (P<0.05). Five mice were used in each group (n = 5). Data are expressed as mean ± SD. * P < 0.05, ** P < 0.01.
4. Discussion
ALI is a complex and multidimensional process including inflammatory response of several types cells in lungs. MAPKs signaling pathway mediated the production of many pro-inflammatory cytokines and chemokines in these cells. As MNK kinases are effectors of MAPK pathways, these observations suggest that they may play important roles in mediating cytokine production at the translational level [35]. MNKs are also the MAPKs downstream molecules, controlling the translation efficiency by phosphorylating eIF4E and playing a pivotal role in the production of pro-inflammatory cytokines such as TNF-α, IL-6. Both kinases are not dispensable in the development of T cells [35]. However, in spite of their roles in inflammation, the individual role of MNK1 and MNK2 in ALI has not been well studied, not mention the potential as pharmaceutical target for ALI or ARDS. In this report, we used both pharmacological reagent and knockout animals to explore the potential of MNK2 as the therapeutic targeting molecules for ALI.
Because of their important roles in inflammation, several compounds were developed as inhibitors for MNKs. Among them, CGP57380 has been used as a MNK inhibitor both in intro and in vivo. Although some thought CGP57380 is a potent for MNK1, however, detail pharmacological studies indicate it is an inhibitor for both MNK1 and MNK2 with the even low IC50 value for MNK2 when ATP was used as substrate [36]. Another interesting compound is the cercosporamide. It has been suggested to more selective to MNK2. However, it also inhibits other kinases, for example it has IC50 value about 0.031 uM for Jak3. In addition, cell biological studies indicated treatment with cercosporamide also induced apoptosis, therefore it has only demonstrated in anti-cancer studies [37]. In contrary, CGP57380 has been studies in non-cancer inflammatory disease models, such asthma, rheumatoid arthritis and bowel disease [38], [39]. Therefore, it is more reasonable for us to use CGP57380 in our ALI model.
In our studies, we first determined the effects of CGP57380 on the LPS induced mouse ALI. Our results from histological studies, protein contents and neutrophil numbers in BALF, and the production of pro-inflammatory cytokines and chemokines demonstrated that CGP57380 is able to reduce the LPS induced ALI in mice, clearly suggesting the involvement of MNK1 and/or MNK2 in the development of lung injury. In the isolated BMDMs, we also demonstrated the inhibitory effects of CGP57380 on the inflammatory response in macrophages. Our data showed that CGP57380 is able to decrease the amounts of IL-6, TNF-α and KC. Furthermore, CGP57380 is able to inhibit the phosphorylation of eIF4E even at low concentration. These results suggest that CGP57380 is targeting to the MNK mediating signaling pathway to regulate the macrophage inflammatory response and eventually reduce the LPS-induced ALI. CGP57380 is not a specific MNK1 or MNK2 inhibitor, and also inhibit other kinases [40], [41], [42]. The interpretation our observation with CGP57380 may need caution. Otherwise, our study of MNK2 deficient mice may complement the limitations presented by CGP57380. Our data from MNK2 deficient mice clearly indicate the involvement of MNK2 in the development of lung inflammation and lung injury. Taken together, our studies suggest that targeting MNK2 may provide new strategy for ALI therapy.
In summary, our results demonstrated for the first time that MNKs, especially MNK2, play a pivotal role in the development of infection induced lung inflammation and injury. With the development and availability of new compounds as MNK specific inhibitor, pharmaceutical targeting MNKs, or at least MNK2, may provide new therapeutic strategy for ALI.
CRediT authorship contribution statement
Jianfeng Gao: Conceptualization, Investigation, Formal analysis, Validation, Data curation, Writing - original draft. Li Teng: Investigation, Methodology, Formal analysis, Validation, Data curation. Sijun Yang: Formal analysis, Methodology, Validation. Shuguang Huang: Investigation, Data curation, Methodology. Linrui Li: Methodology, Software, Investigation. Li Zhou: Investigation, Data curation. Guoquan Liu: Investigation, Data curation, Writing - original draft. Hongbin Tang: Resources, Supervision, Funding acquisition, Validation.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by the grants from the National Natural Science Foundation of China (Grant No 81670069), the National Key Research and Development Program of China (Grant No 2018YFC1200302), the Natural Science Foundation of Hubei (Grant No 2019CFB679), the Health Commission of Hubei Province Scientific Research Project (Grant No WJ2021Q031) and the Teaching Research Project of Medical Department of Wuhan University (Grant No 2020078). We thank Prof. Hongliang Li (Wuhan University) for kindly providing the MNK2 deficient mice.
References
- 1.Mowery N.T., Terzian W.T.H., Nelson A.C. Acute lung injury. Curr. Probl. Surg. 2020;57(5):100777. doi: 10.1016/j.cpsurg.2020.100777. [DOI] [PubMed] [Google Scholar]
- 2.Butt Y., Kurdowska A., Allen T.C. Acute Lung Injury: A Clinical and Molecular Review. Arch. Pathol. Lab. Med. 2016;140(4):345–350. doi: 10.5858/arpa.2015-0519-RA. [DOI] [PubMed] [Google Scholar]
- 3.Mokra D., Kosutova P. Biomarkers in acute lung injury. Respir. Physiol. Neurobiol. 2015;209:52–58. doi: 10.1016/j.resp.2014.10.006. [DOI] [PubMed] [Google Scholar]
- 4.Saguil A., Fargo M.V. Acute respiratory distress syndrome: diagnosis and management. Am. Fam. Physician. 2020;101(12):730–738. [PubMed] [Google Scholar]
- 5.Wu K., Huang S.S., Kuo Y.H., Ho Y.L., Yang C.S., Chang Y.S., Huang G.J. Ugonin M, a Helminthostachys zeylanica constituent, prevents LPS-induced acute lung injury through TLR4-mediated MAPK and NF-κB signaling pathways. Molecules. 2017;22(4):573. doi: 10.3390/molecules22040573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.N. Costa, G.R. Júnior, A.A. Alemany, L. Belotti, D.H. Zati, M. Cavalcante, M. Veras, S. Ribeiro, E.G. Kallás, P.H. Saldiva, M. Dolhnikoff, L.F. Silva, Early and late pulmonary effects of nebulized LPS in mice: An acute lung injury model, PLoS One 12(9) (2017) e0185474. [DOI] [PMC free article] [PubMed]
- 7.Hughes K.T., Beasley M.B. Pulmonary manifestations of acute lung injury: more than just diffuse alveolar damage. Arch. Pathol. Lab. Med. 2017;141(7):916–922. doi: 10.5858/arpa.2016-0342-RA. [DOI] [PubMed] [Google Scholar]
- 8.Malaviya R., Sunil V.R., Venosa A., Vayas K.N., Businaro R., Heck D.E., Laskin J.D., Laskin D.L. Macrophages and inflammatory mediators in pulmonary injury induced by mustard vesicants. Ann. N. Y. Acad. Sci. 2016;1374(1):168–175. doi: 10.1111/nyas.13123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li X., Ye C., Mulati M., Sun L., Qian F. Ellipticine blocks synergistic effects of IL-17A and TNF-α in epithelial cells and alleviates severe acute pancreatitis-associated acute lung injury. Biochem. Pharmacol. 2020;177 doi: 10.1016/j.bcp.2020.113992. [DOI] [PubMed] [Google Scholar]
- 10.Liang Y., Yang N., Pan G., Jin B., Wang S., Ji W. Elevated IL-33 promotes expression of MMP2 and MMP9 via activating STAT3 in alveolar macrophages during LPS-induced acute lung injury. Cell. Mol. Biol. Lett. 2018;23:52. doi: 10.1186/s11658-018-0117-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bao X., Zhang Q., Liu N., Zhuang S., Li Z., Meng Q., Sun H., Bai J., Zhou X., Tang L. Characteristics of circular RNA expression of pulmonary macrophages in mice with sepsis-induced acute lung injury. J. Cell Mol. Med. 2019;23(10):7111–7115. doi: 10.1111/jcmm.14577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li D., Ren W., Jiang Z., Zhu L. Regulation of the NLRP3 inflammasome and macrophage pyroptosis by the p38 MAPK signaling pathway in a mouse model of acute lung injury. Mol. Med. Rep. 2018;18(5):4399–4409. doi: 10.3892/mmr.2018.9427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tian X., Xie G., Xiao H., Ding F., Bao W., Zhang M. CXCR4 knockdown prevents inflammatory cytokine expression in macrophages by suppressing activation of MAPK and NF-κB signaling pathways. Cell Biosci. 2019;9:55. doi: 10.1186/s13578-019-0315-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang Y., Xu Y., Zhang P., Ruan W., Zhang L., Yuan S., Pang T., Jia A.Q. Smiglaside A ameliorates LPS-induced acute lung injury by modulating macrophage polarization via AMPK-PPARγ pathway. Biochem. Pharmacol. 2018;156:385–395. doi: 10.1016/j.bcp.2018.09.002. [DOI] [PubMed] [Google Scholar]
- 15.Artyomov M.N., Sergushichev A., Schilling J.D. Integrating immunometabolism and macrophage diversity. Semin. Immunol. 2016;28(5):417–424. doi: 10.1016/j.smim.2016.10.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Assadiasl S., Mousavi M.J., Amirzargar A. Toll-like receptor 4 in renal transplant. Exp. Clin. Transplant. 2018;16(3):245–252. [PubMed] [Google Scholar]
- 17.García Bueno B., Caso J.R., Madrigal J.L., Leza J.C. Innate immune receptor Toll-like receptor 4 signalling in neuropsychiatric diseases. Neurosci. Biobehav. Rev. 2016;64:134–147. doi: 10.1016/j.neubiorev.2016.02.013. [DOI] [PubMed] [Google Scholar]
- 18.Olejnik J., Hume A.J., Mühlberger E., Dutch R.E. Toll-like receptor 4 in acute viral infection: Too much of a good thing. PLoS Pathog. 2018;14(12):e1007390. doi: 10.1371/journal.ppat.1007390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Swanson L., Katkar G.D., Tam J., Pranadinata R.F., Chareddy Y., Coates J., Anandachar M.S., Castillo V., Olson J., Nizet V., Kufareva I., Das S., Ghosh P. TLR4 signaling and macrophage inflammatory responses are dampened by GIV/Girdin. Proc Natl Acad Sci USA. 2020;117(43):26895–26906. doi: 10.1073/pnas.2011667117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Ciesielska A., Matyjek M., Kwiatkowska K. TLR4 and CD14 trafficking and its influence on LPS-induced pro-inflammatory signaling. Cell. Mol. Life Sci. 2021 doi: 10.1007/s00018-020-03656-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Robertson S.A., Wahid H.H., Chin P.Y., Hutchinson M.R., Moldenhauer L.M., Keelan J.A. Toll-like receptor-4: a new target for preterm labour pharmacotherapies? Curr. Pharm. Des. 2018;24(9):960–973. doi: 10.2174/1381612824666180130122450. [DOI] [PubMed] [Google Scholar]
- 22.Molteni M., Gemma S., Rossetti C. The Role of Toll-Like Receptor 4 in Infectious and Noninfectious Inflammation. Mediators Inflamm. 2016;2016:6978936. doi: 10.1155/2016/6978936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.J.L. Lai, Y.H. Liu, C. Liu, M.P. Qi, R.N. Liu, X.F. Zhu, Q.G. Zhou, Y.Y. Chen, A.Z. Guo, CM. Hu, Indirubin Inhibits LPS-Induced Inflammation via TLR4 Abrogation Mediated by the NF-kB and MAPK Signaling Pathways, Inflammation 40(1) (2017) 1-12. [DOI] [PubMed]
- 24.Wang W., Weng J., Yu L., Huang Q., Jiang Y., Guo X. Role of TLR4-p38 MAPK-Hsp27 signal pathway in LPS-induced pulmonary epithelial hyperpermeability. BMC Pulm. Med. 2018;18(1):178. doi: 10.1186/s12890-018-0735-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Li R., Guo Y., Zhang Y., Zhang X., Zhu L., Yan T. Salidroside Ameliorates Renal Interstitial Fibrosis by Inhibiting the TLR4/NF-κB and MAPK Signaling Pathways. Int. J. Mol. Sci. 2019;20(5):1103. doi: 10.3390/ijms20051103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Leitner G.R., Wenzel T.J., Marshall N., Gates E.J., Klegeris A. Targeting toll-like receptor 4 to modulate neuroinflammation in central nervous system disorders. Expert Opin. Ther. Targets. 2019;23(10):865–882. doi: 10.1080/14728222.2019.1676416. [DOI] [PubMed] [Google Scholar]
- 27.Xie J., Merrett J.E., Jensen K.B., Proud C.G. The MAP kinase-interacting kinases (MNKs) as targets in oncology. Expert Opin. Ther. Targets. 2019;23(3):187–199. doi: 10.1080/14728222.2019.1571043. [DOI] [PubMed] [Google Scholar]
- 28.Diab S., Kumarasiri M., Yu M., Teo T., Proud C., Milne R., Wang S. MAP kinase-interacting kinases–emerging targets against cancer. Chem. Biol. 2014;21(4):441–452. doi: 10.1016/j.chembiol.2014.01.011. [DOI] [PubMed] [Google Scholar]
- 29.Buxade M., Parra-Palau J.L., Proud C.G. The Mnks: MAP kinase-interacting kinases (MAP kinase signal-integrating kinases) Front Biosci. 2008;13:5359–5373. doi: 10.2741/3086. [DOI] [PubMed] [Google Scholar]
- 30.Ren J., Su D., Li L., Cai H., Zhang M., Zhai J., Li M., Wu X., Hu K. Anti-inflammatory effects of Aureusidin in LPS-stimulated RAW264.7 macrophages via suppressing NF-κB and activating ROS- and MAPKs-dependent Nrf2/HO-1 signaling pathways. Toxicol. Appl. Pharmacol. 2020;387 doi: 10.1016/j.taap.2019.114846. [DOI] [PubMed] [Google Scholar]
- 31.Zhang D., Lee H., Wang X., Groot M., Sharma L., Dela Cruz C.S., Jin Y. A potential role of microvesicle-containing miR-223/142 in lung inflammation. Thorax. 2019;74(9):865–874. doi: 10.1136/thoraxjnl-2018-212994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Matute-Bello G., Downey G., Moore B.B., Groshong S.D., Matthay M.A., Slutsky A.S., Kuebler W.M. Acute Lung Injury in Animals Study Group. An official American Thoracic Society workshop report: features and measurements of experimental acute lung injury in animals. Am. J. Respir. Cell Mol. Biol. 2011;44(5):725–738. doi: 10.1165/rcmb.2009-0210ST. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hu G., Christman J.W. Editorial: Alveolar Macrophages in Lung Inflammation and Resolution. Front. Immunol. 2019;10:2275. doi: 10.3389/fimmu.2019.02275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Laskin D.L., Malaviya R., Laskin J.D. Role of macrophages in acute lung injury and chronic fibrosis induced by pulmonary toxicants. Toxicol. Sci. 2019;168(2):287–301. doi: 10.1093/toxsci/kfy309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Joshi S., Platanias L.C. Mnk kinase pathway: Cellular functions and biological outcomes. World J. Biol. Chem. 2014;5(3):321–333. doi: 10.4331/wjbc.v5.i3.321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Basnet S.K., Diab S., Schmid R., Yu M., Yang Y., Gillam T.A., Teo T., Li P., Peat T., Albrecht H., Wang S. Identification of a highly conserved allosteric binding site on Mnk1 and Mnk2. Mol. Pharmacol. 2015;88(5):935–948. doi: 10.1124/mol.115.100131. [DOI] [PubMed] [Google Scholar]
- 37.Konicek B.W., Stephens J.R., McNulty A.M., Robichaud N., Peery R.B., Dumstorf C.A., Dowless M.S., Iversen P.W., Parsons S., Ellis K.E., McCann D.J., Pelletier J., Furic L., Yingling J.M., Stancato L.F., Sonenberg N., Graff J.R. Therapeutic inhibition of MAP kinase interacting kinase blocks eukaryotic initiation factor 4E phosphorylation and suppresses outgrowth of experimental lung metastases. Cancer Res. 2011;71(5):1849–1857. doi: 10.1158/0008-5472.CAN-10-3298. [DOI] [PubMed] [Google Scholar]
- 38.Seidel P., Sun Q., Costa L., Lardinois D., Tamm M., Roth M. The MNK-1/eIF4E pathway as a new therapeutic pathway to target inflammation and remodelling in asthma. Cell. Signal. 2016;28(10):1555–1562. doi: 10.1016/j.cellsig.2016.07.004. [DOI] [PubMed] [Google Scholar]
- 39.S. Joshi, L.C. Platanias, Mnk Kinases in Cytokine Signaling and Regulation of Cytokine Responses, Biomol Concepts, 3(2) (2012) 127-139. [DOI] [PMC free article] [PubMed]
- 40.Dreas A., Mikulski M., Milik M., Fabritius C.H., Brzózka K., Rzymski T. Mitogen-activated protein kinase (MAPK) interacting kinases 1 and 2 (MNK1 and MNK2) as targets for cancer therapy: recent progress in the development of MNK inhibitors. Curr. Med. Chem. 2017;24(28):3025–3053. doi: 10.2174/0929867324666170203123427. [DOI] [PubMed] [Google Scholar]
- 41.M. Grzmil, R.M. Huber, D. Hess, S. Frank, D. Hynx, G. Moncayo, D. Klein, A. Merlo, B.A. Hemmings, MNK1 pathway activity maintains protein synthesis in rapalog-treated gliomas, J. Clin. Invest. 124(2) (2014) 742-754. [DOI] [PMC free article] [PubMed]
- 42.B.K. Gorentla, S. Krishna, J. Shin, M. Inoue, M.L. Shinohara, J.M. Grayson, R. Fukunaga, X.P. Zhong, Mnk1 and 2 are dispensable for T cell development and activation but important for the pathogenesis of experimental autoimmune encephalomyelitis, J. Immunol. 190(3) (2013) 1026-1037. [DOI] [PMC free article] [PubMed]