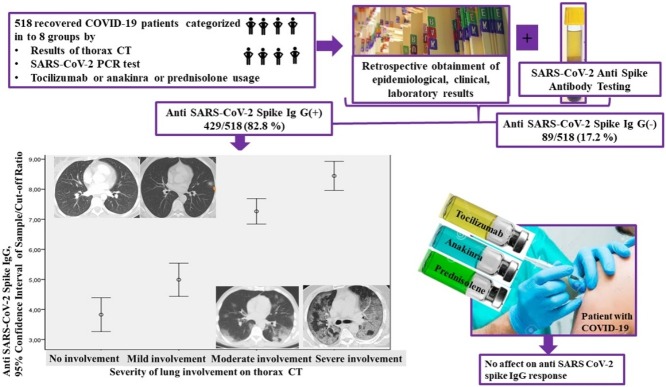
Graphical abstract
Keywords: Tocilizumab, Anakinra, Prednisolone, SARS-CoV-2, Antibody
Abstract
Objectives
Disease severity, previous medications and immunosuppressive agents could affect the antibody response against SARS-CoV-2. This study aimed to analyze variables affecting the humoral response to SARS-CoV-2.
Methods
This prospective cohort study included adult patients who recovered from COVID-19 and were admitted to a COVID-19 follow-up unit. Eight patient groups were defined in accordance with the results of thoracic computed tomography (CT), SARS-CoV-2 PCR test, and tocilizumab or anakinra use during active disease. Anti-S IgG antibodies were determined by ELISA in serum samples. Anti-S positive and negative cases were compared.
Results
A total of 518 patients were included in the study. SARS-CoV-2 IgG antibodies were positive in 82.8% of patients. SARS-CoV-2 PCR positivity, extent of lung involvement on CT, and time to antibody testing were independently associated with antibody positivity. Tocilizumab, anakinra or prednisolone use was not a factor affecting the antibody response. The rate of antibody response and sample/CO values among antibody-positive patients showed a linear relationship with the extent of lung involvement on CT.
Conclusions
The use of tocilizumab, anakinra and prednisolone for COVID-19 did not affect the antibody response against SARS-CoV-2. The main driver of antibody response among patients with COVID-19 was the extent of pulmonary involvement on CT.
Introduction
A protective immune response against SARS-CoV-2 requires synchronized function of viral protein-specific CD4+ and CD8+ T cells as well as B cells. COVID-19 patients with severe infection are more likely to have asynchronous immune responses. Whether having a synchronized or asynchronized immune response, the vast majority of patients with PCR-confirmed SARS-CoV-2 infection produce antibodies against SARS-CoV-2 and those with the most severe disease have higher titers of anti-SARS-CoV-2 antibodies compared with asymptomatic or mild cases (Rydyznski Moderbacher et al., 2020, Wajnberg et al., 2020a, Wajnberg et al., 2020b). However, in addition to the severity of infection, many other factors – including comorbidities, previous medication and current treatments – could also affect the antibody response to SARS-CoV-2 in COVID-19. After the publication of some observational studies showing beneficial effects of anti-cytokine treatments for COVID-19, biologic agents blocking the activity of IL-6 and IL-1, such as tocilizumab and anakinra, respectively, have been widely used to treat COVID-19 patients who developed findings of cytokine storm syndrome (Toniati et al., 2020). In addition, glucocorticoid treatment has become a standard of care for severe COVID-19 patients after demonstration of the beneficial effect of dexamethasone on mortality (RECOVERY Collaborative Group et al., 2020). All of these immunosuppressive treatments could affect the antibody response against SARS-CoV-2 (Maeda et al., 2010, Roll et al., 2011, Dinarello, 2009).
This study analyzed all potential factors that may affect the antibody response to SARS- CoV-2 in a cohort of patients with different levels of disease severity and SARS-CoV-2 PCR results. It specifically aimed to investigate whether or not this response was affected by tocilizumab, anakinra or prednisolone treatment.
Methods
Study design and population
This prospective cohort study included all consecutive adult patients who: were diagnosed with probable or confirmed COVID-19 at the current hospital, had recovered from the disease, were admitted to a COVID-19 follow-up unit between June and September 2020, and fulfilled the below specific group criteria. The definitions of the World Health Organization for a COVID-19 case were used: A patient who meets the appropriate clinical criteria: acute onset of fever and cough, or acute onset of any three or more of the following signs or symptoms: fever, cough, general weakness/fatigue, headache, myalgia, sore throat, coryza, dyspnea, anorexia/nausea/vomiting, diarrhea, and altered mental status; resides or has traveled to an area with community transmission anytime within the 14 days prior to symptom onset; and either with chest imaging showing findings suggestive of COVID-19 disease and/or with a positive SARS-CoV-2 PCR test result (World Health Organization (WHO, 2020). To include all patients with different disease severity and microbiologic result status, eight patient groups were defined in accordance with the results of thoracic computed tomography (CT), SARS-CoV-2 PCR test and use of tocilizumab or/and anakinra during the active phase of COVID-19 disease before enrollment into the study: Group 1: Patients treated with at least one dose of tocilizumab and/or anakinra for COVID-19; Group 2: Patients with a positive SARS-CoV-2 PCR test and severe involvement on thoracic CT; Group 3: Patients with a positive SARS-CoV-2 PCR test and moderate involvement on thoracic CT; Group 4: Patients with a negative SARS-CoV-2 PCR test and severe involvement on thoracic CT; Group 5: Patients with a negative SARS-CoV-2 PCR test and moderate involvement on thoracic CT; Group 6: Patients with a positive SARS-CoV-2 PCR test and mild involvement on thoracic CT; Group 7: Patients with a negative SARS-CoV-2 PCR test and mild involvement on thoracic CT; Group 8: Patients with a positive SARS-CoV-2 PCR test and no involvement on thoracic CT.
The extent of lung involvement on thoracic CT was graded as absent, minimal (<25%), moderate (25–50%), and severe (>50%) (Jalaber et al., 2020). Bilateral and multifocal ground-glass opacities predominating in the peripheral sections of the lungs were defined as specific CT features of COVID-19 pneumonia. Tocilizumab, anakinra and prednisolone were used in cases of hyperinflammatory response as follows: single or two 400 mg doses of tocilizumab, anakinra with a daily dose ranging between 2 × 100 mg SC to 3 × 200 mg IV, and at least 80 mg/day prednisolone for 5 days.
Laboratory examination
The laboratory diagnosis of COVID-19 was based on RT-qPCR and anti-SARS-CoV-2 antibodies. Viral RNA extraction from respiratory specimens was performed using a manual kit (Bioeksen the Co Ltd. R & D Technologies, Turkey). RT-qPCR process was performed on a Rotor-Gene Q 5 Plex Real Time PCR device (Qiagen, Germany) using a national kit (Bioeksen the Co Ltd. R & D Technologies, Turkey). The Anti-SARS-CoV-2 ELISA (IgG) kit (Euroimmun, Germany) was used to detect IgG antibodies developed against the S1 domain of the SARS-CoV-2 spike protein in serum samples.
Data collection and outcomes
Physical examination was performed and blood samples were drawn from patients after consent was obtained. The serum samples were stored at −20 °C until antibody testing. The epidemiological, clinical features and admission laboratory tests and thoracic CT results were retrospectively obtained from the online hospital database.
Statistical analysis
Statistical analysis was performed using the Statistical Package for the Social Sciences (SPSS) 21.0 (SPSS Inc., Chicago, IL, USA). Univariate analysis was performed using Chi-square, Fisher’s exact, Student’s t-, and Mann–Whitney U tests, where appropriate. For the multivariate logistic regression analysis, significant (with p < 0.05) factors identified with univariate analysis were included to the model to determine the independent predictors of SARS-CoV-2 spike IgG antibody seroconversion. Colinear variables were excluded, collinearity between predictors was tested by correlation matrix. All factors included into the multivariate logistic regression analysis are shown in Table 1 . Hosmer-Lemeshow goodness-of-fit statistics were used to assess the model fit. Kruskal-Wallis tests were conducted to compare the non-normally distributed parameters and ordinal variables between the groups, and the level of pulmonary involvement on thoracic CT.
Table 1.
Comparison of patients with and without a positive SARS-CoV-2 antibodies.
| Characteristics | Probable + confirmed COVID-19 cases (n = 518) |
p-Value | Confirmed COVID-19 cases (n = 311) |
p-Value | ||
|---|---|---|---|---|---|---|
| Patients with positive anti-SARS CoV-2 IgG (n = 429) | Patients with negative anti-SARS CoV-2 IgG (n = 89) | Patients with positive anti-SARS CoV-2 IgG (n = 280) | Patients with negative anti-SARS CoV-2 IgG (n = 31) | |||
| Age, mean ± SD | 48.99 ± 13.69 | 49.75 ± 16.33 | 0.647a | 48.15 ± 14.44 | 43.32 ± 12.21 | 0.047a |
| Gender, female, n (%) | 184 (42.9) | 43 (48.3) | 0.348a | 123 (43.9) | 18 (58.1) | 0.134a |
| Hypertension, n (%) | 98 (22.8) | 25 (28.1) | 0.290 | 58 (20.7) | 3 (9.7) | 0.230 |
| Diabetes mellitus, n (%) | 73 (17.0) | 16 (17.9) | 0.827 | 44 (15.7) | 2 (6.4) | 0.282 |
| Hypothyroidism, n (%) | 23 (5.3) | 6(6.7) | 0.610 | 18 (6.4) | 3 (9.7) | 0.453 |
| Rheumatological diseases, n (%) | 21 (4.9) | 8 (9.0) | 0.282 | 13 (4.6) | 2 (6.4) | 0.814 |
| Asthma, n (%) | 19 (4.4) | 6 (6.7) | 0.354 | 14 (5.0) | 2 (6.4) | 0.666 |
| Cancer, n (%) | 15 (3.5) | 3 (3.4) | 1.000 | 9 (3.2) | 1 (3.2) | 1.000 |
| Ischemic heart disease, n (%) | 13 (3.0) | 5 (5.6) | 0.213 | 7 (2.5) | 0 (0) | 1.000 |
| Chronic obstructive pulmonary disease, n (%) | 9 (2.1) | 3 (3.4) | 0.442 | 5 (1.8) | 0 (0) | 1.000 |
| Previous tuberculosis, n (%) | 7 (1.6) | 4 (4.5) | 0.102 | 6 (2.1) | 1 (3.2) | 0.524 |
| Solid organ transplantation, n (%) | 4 (0.9) | 6 (6.7) | 0.003a | 2 (0.7) | 0 (0) | 1.000 |
| Congestive heart failure, n (%) | 3 (0.7) | 3 (3.4) | 0.066 | 3 (1.1) | 0 (0) | 1.000 |
| Chronic renal failure, n (%) | 4 (0.9) | 1 (1.1) | 1.000 | 1 (0.4) | 0 (0) | 1.000 |
| Previous medications | ||||||
| Immunosuppressivesb | 14 (3.3) | 8 (8.9) | 0.015a | 9 (3.2) | 1 (3.2) | 0.656 |
| Clinical and laboratory findings on admission for COVID-19 | ||||||
| Body temperature, mean ± SD | 37.4 ± 1.0 | 37.0 ± 0.89 | 0.001a | 37.4 ± 0.7 | 36.9 ± 07 | 0.014a |
| Peripheral arterial oxygen saturation (SpO2), mean ± SD | 95.6 ± 3.9 | 96.1 ± 4.5 | 0.007 | 95.9 ± 4.0 | 98.2 ± 1.1 | <0.001 |
| Blood leucocyte count, /mm3, mean ± SD | 6211 ± 5827 | 8904 ± 4004 | <0.001 | 5821 ± 2280 | 8405 ± 5179 | 0.001 |
| Blood neutrophil count /mm3, mean ± SD | 3941 ± 2105 | 6305 ± 3978 | <0.001a | 3780 ± 2011 | 5771 ± 5575 | 0.032a |
| Blood lymphocyte count, /mm3, mean ± SD | 1396 ± 651 | 1780 ± 985 | 0.001a | 1415 ± 644 | 1742 ± 815 | 0.073 |
| Serum CRP level, mg/L, mean ± SD | 48.46 ± 61.94 | 46.17 ± 68.54 | 0.038a | 45.29 ± 65.53 | 13.56 ± 28.92 | <0.001a |
| Serum ferritin level, mg/dL, mean ± SD | 453.4 ± 715.5 | 256.1 ± 441.7 | <0.001a | 420.9 ± 732.0 | 84.5 ± 105.2 | <0.001a |
| Serum D-dimer level, mg/dL, mean ± SD | 1012 ± 1817 | 1060 ± 1174 | 0.771 | 942 ± 1635 | 768 ± 964 | 0.064 |
| Hospitalized for COVID-19, n (%) | 282 (65.7) | 46 (51.7) | 0.012a | 169 (60.4) | 6 (19.4) | <0.001a |
| Presence of pneumonia, n (%) | 371 (86.5) | 71 (79.8) | 0.104 | 220 (78.6) | 13 (41.9) | <0.001a |
| Need for nasal O2 support, n (%) | 144 (33.6) | 16 (18.0) | 0.010a | 82 (29.3) | 0 (0) | 0.002a |
| Need for high-flow nasal oxygen, n (%) | 25 (5.8) | 5 (5.6) | 0.728 | 15 (5.4) | 0 (0) | 0.347 |
| Need for mechanical ventilation, n (%) | 13 (3.0) | 2 (2.2) | 0.672 | 6 (2.1) | 0 (0) | 0.599 |
| Need for intensive care unit support, n (%) | 23 (5.4) | 7 (7.9) | 0.485 | 13 (4.6) | 0 (0) | 0.393 |
| SARS-CoV-2 PCR positivity, n (%) | 280 (65.3) | 31 (34.8) | <0.001a | |||
| Extent of lung involvement on thoracic CT, n (%) | <0.001a | <0.001a | ||||
| None, n (%) | 58 (13.5) | 18 (20.2) | 58 (20.7) | 18 (58.1) | ||
| Mild, n (%) | 112 (26.1) | 50 (56.2) | 73 (26.1) | 12 (38.7) | ||
| Moderate, n (%) | 138 (32.2) | 15 (16.8) | 79 (28.2) | 1 (3.2) | ||
| Severe, n (%) | 121 (28.2) | 6 (6.7) | 70 (25.0) | 0 (0) | ||
| Patient groups | <0.001 | <0.001 | ||||
| Group 1, n (%) | 77 (17.9) | 3 (3.4) | 45 (16.1) | 0 (0) | ||
| Group 2, n (%) | 38 (8.9) | 0 (0) | 38 (13.6) | 0 (0) | ||
| Group 3, n (%) | 66 (15.4) | 1 (1.1) | 66 (23.5) | 1 (3.2) | ||
| Group 4, n (%) | 26 (6.1) | 5 (5.6) | – | – | ||
| Group 5, n (%) | 52 (12.1) | 12 (13.5) | – | – | ||
| Group 6, n (%) | 73 (17.0) | 12 (13.5) | 73 (26.1) | 12 (38.7) | ||
| Group 7, n (%) | 39 (9.1) | 38 (42.7) | – | – | ||
| Group 8, n (%) | 58 (13.5) | 18 (20.2) | 58 (20.7) | 18 (58.1) | ||
| Severe disease (either needing supplemental oxygen or severe involvement on thoracic CT), n (%) | 176 (41.0) | 16 (17.9) | <0.001 | 99 (35.4) | 0 (0) | <0.001 |
| Anti-SARS-CoV-2 treatments | ||||||
| Favipiravir ± others, n (%) | 138 (32.2) | 20 (22.5) | 0.070 | 94 (33.6) | 2 (6.5) | 0.001a |
| Hydroxychloroquine ± others, n (%) | 414 (96.5) | 85 (95.5) | 0.656 | 269 (96.1) | 31 (100) | 1.000 |
| Tocilizumab or anakinra for COVID-19 | 0.006 | 0.120 | ||||
| Tocilizumab, n (%) | 55 (12.8) | 1 (1.1) | 32 (11.4) | 0 (0) | ||
| Anakinra, n (%) | 10 (2.3) | 1 (1.1) | 7 (2.5) | 0 (0) | ||
| Tocilizumab = anakinra, n (%) | 12 (2.8) | 1 (1.1) | 6 (2.1) | 0 (0) | ||
| Tocilizumab and/or anakinra for COVID-19, n (%) | 77 (17.9) | 3 (3.4) | <0.001a | 45 (16.1) | 0 (0) | 0.012a |
| Prednisolone for COVID-19, n (%) | 21 (4.9) | 0 (0) | 0.034a | 11 (3.9) | 0 (0) | 0.610 |
| Convalescent plasma for COVID-19, n (%) | 5 (1.2) | 0 (0) | 0.594 | 3 (1.1) | 0 (0) | 1.000 |
| IVIG for COVID-19, n (%) | 8 (0.7) | 1 (1.1) | 1.000 | 5 (1.8) | 0 (0) | 1.000 |
| Time from hospital admission to antibody testing, /mm3, mean ± SD | 86.90 ± 43.44 | 62.15 ± 37.20 | <0.001a | 80.84 ± 42.41 | 77.64 ± 42.29 | 0.712 |
| Blood lymphocyte count at the antibody testing, /mm3, mean ± SD | 2144 ± 750 | 2053 ± 588 | 0.484 | 2118 ± 695 | 2154 ± 533 | 0.601 |
Abbreviations: SARS CoV-2, severe acute respiratory syndrome coronavirus-2; IgG, immunoglobulin G; CT, computed tomography; PCR, polymerase chain reaction; COVID-19, Coronavirus disease 2019; IVIG, intravenous immunoglobulin.
Entered into multivariate analysis.
Including prednisolones and/or monoclonal immunosuppressive antibodies and/or other immunosuppressives.
Results
A total of 518 patients (63.3% hospitalized, 36.7% outpatients) were included in the study. The mean age of the patients was 49.2 years, and 56.2% were male. While 442 of patients had pneumonia (85%), 76 had noncomplicated disease without pneumonia (15%); 192 of 518 patients (37%) were categorized as having severe disease who either required oxygen support or had severe involvement on thoracic CT. Nasopharyngeal SARS-CoV-2 PCR test was positive in 60% of the patients. The mean time from hospital admission to antibody testing was 82 days, and 25, 50 and 75 percentiles of testing time were 42, 76 and 125 days, respectively. SARS-CoV-2 IgG antibodies were positive in 82.8% of patients, the rate was 90% and 72% among PCR positive and negative cases, respectively; and 94% of PCR-positive patients with no antibody response had either no (52%) or minor (42%) involvement on thoracic CT. The demographic, and clinical features of patients with and without antibody response are shown in Table 1.
On univariate analysis the following were found to be related to antibody positivity: solid organ transplantation (p = 0.003); use of immunosuppressive drugs before COVID-19 diagnosis (p = 0.01); presence of fever; low WBC, neutrophil and lymphocyte counts; high CRP and ferritin levels on admission; need for oxygen support; hospitalization; severity of involvement on thoracic CT; nasopharyngeal SARS-CoV-2 PCR positivity; time from hospital admission to antibody testing; tocilizumab, anakinra or prednisolone use for COVID-19 (Table 1). On multivariate analysis, SARS-CoV-2 PCR positivity and extent of involvement on thoracic CT and time from hospital admission to antibody testing were found to be independently associated with antibody positivity (Table 2 ).
Table 2.
Multivariate analysis of factors affecting SARS-CoV-2 spike antibody response.
| Characteristics | Probable + confirmed COVID-19 cases (n = 518) |
Confirmed COVID-19 cases (n = 311) |
||||
|---|---|---|---|---|---|---|
| p-Value | OR | 95% CI | p-Value | OR | 95% CI | |
| SARS-CoV-2 PCR test positivity | <0.001 | 5.639 | 2.806–11.331 | NA | NA | NA |
| Moderate/severe lung involvement on thoracic CT | <0.001 | 8.193 | 3.824–17.552 | 0.034 | 10.95 | 1.20–99. 81 |
| Time from hospital admission to antibody testing, /mm3, mean ± SD | <0.001 | 1.015 | 1.007–1.023 | – | – | – |
| Previous immunosuppressive drug usage | 0.027 | 0.213 | 0.054–0.835 | – | – | – |
Abbreviations; SARS CoV-2, severe acute respiratory syndrome coronavirus-2; PCR, polymerase chain reaction; CT, computed tomography.
The analysis of only the confirmed COVID-19 cases (with a positive result of SARS-CoV-2 PCR test) showed that the following were found as the factors associated with antibody response: older age (p = 0.047); hospitalization (p < 0.001); presence of fever (p = 0.014) and pneumonia (p = 0.001); higher serum CRP (p < 0.001) and ferritin levels (p < 0.001); lower blood WBC (p = 0.001) and PNL counts (p = 0.032); moderate/severe lung involvement on thoracic CT (p = 0.001); need for oxygen support (p = 0.002); use of favipiravir (p = 0.001) and tocilizumab and/or anakinra (p = 0.012). Although serum D-dimer levels were found to be higher, and blood lymphocyte counts were found to be lower among patients with a positive SARS-CoV-2 spike IgG, the differences did not reach statistical significance (p = 0.064 and p = 0.07, respectively) (Table 1). Moderate/severe lung involvement on thoracic CT was also found to be an independent factor of SARS-CoV-2 spike antibody positivity among confirmed cases (OR 10.95, 95% CI 1.20–99.81, p = 0.034) (Table 2).
Both the rate of antibody response and sample/cut-off (S/CO) values among antibody-positive patients showed a linear relationship between the extent of lung involvement on thoracic CT (Figure 1 ) and both were significantly different between the groups (p < 0.05). The S/CO levels were found to be 2.21, 1.69 and 1.16 times higher in patients with severe, mild and moderate involvement compared with no lung involvement, respectively. Groups including severe COVID-19 cases (Groups 1–3) showed significantly higher titer responses compared with the groups including milder cases (Groups 5–8) (p < 0.001). The antibody concentrations among patients with positive antibody results (429 patients) were significantly higher in patients who were treated with either tocilizumab, anakinra or prednisolone (p < 0.05). However, the S/CO levels of those patients were found to be similar with the S/CO levels of patients with severe disease (p > 0.05) (Table 3 ).
Figure 1.
Distribution of sample/cut-off ratios of patients according to lung involvement on thoracic CT.
Table 3.
Comparison of antibody levels among patients with a positive antibody result and severe disease.
| Feature | (n) | S/CO level (median ± SD) | p-Value |
|---|---|---|---|
| Patients with a positive SARS-CoV-2 antibody result (n = 429) | |||
| Need for oxygen support | No (285) | 5.55 ± 2.82 | <0.001 |
| Yes (144) | 8.40 ± 2.77 | ||
| SARS-CoV-2 PCR test positivity | No (149) | 6.71 ± 3.04 | 0.388 |
| Yes (280) | 6.44 ± 3.15 | ||
| Level of lung involvement on thoracic CT | None (58) | 3.82 ± 2.14 | <0.001 |
| Mild (112) | 4.99 ± 2.94 | ||
| Moderate (138) | 7.26 ± 2.50 | ||
| Severe (121) | 8.44 ± 2.67 | ||
| Patient treated with either tocilizumab or anakinra | No (352) | 6.15 ± 3.12 | <0.001 |
| Yes (77) | 8.30 ± 2.39 | ||
| Patient treated with tocilizumab + anakinra | No (417) | 6.57 ± 3.11 | 0.171 |
| Yes (12) | 7.76 ± 3.12 | ||
| Patient treated with tocilizumab | No (374) | 6.24 ± 3.11 | <0.001 |
| Yes (55) | 8.55 ± 2.26 | ||
| Patient treated with anakinra | No (419) | 6.51 ± 3.73 | 0.145 |
| Yes (10) | 7.71 ± 1.63 | ||
| Patient treated with corticosteroid for COVID-19 | No (405) | 6.39 ± 3.08 | 0.001 |
| Patient treated with corticosteroid | Yes (21) | 8.73 ± 2.77 | |
| before COVID-19 diagnosis | No (416) | 6.52 ± 3.09 | 0.553 |
| Yes (10) | 5.97 ± 3.66 | ||
| Patients with a positive SARS-CoV-2 antibody result and severe disease (n = 176) | |||
| Patient treated with either tocilizumab or anakinra | No (104) | 8.22 ± 2.88 | 0.644 |
| Yes (72) | 8.44 ± 2.33 | ||
| Patient treated with tocilizumab | No (123) | 8.18 ± 2.82 | 0.473 |
| Yes (53) | 8.60 ± 2.28 | ||
| Patient treated with anakinra | No (168) | 8.33 ± 2.71 | 0.733 |
| Yes (8) | 7.99 ± 1.53 | ||
| Patient treated with tocilizumab + anakinra | No (165) | 8.33 ± 2.65 | 0.898 |
| Yes (11) | 7.99 ± 3.05 | ||
| Patient treated with corticosteroid for COVID-19 | No (156) | 8.18 ± 2.66 | 0.223 |
| Yes (19) | 9.19 ± 2.49 | ||
| Patient treated with corticosteroid before COVID-19 diagnosis | No (170) | 8.38 ± 2.59 | 0.032 |
| Yes (5) | 5.04 ± 3.12 | ||
Abbreviations: SARS CoV-2, severe acute respiratory syndrome coronavirus-2; PCR, polymerase chain reaction; CT, computed tomography; COVID-19, Coronavirus disease 2019.
Discussion
This study evaluated the factors affecting antibody responses against SARS-CoV-2 in a cohort of COVID-19 patients with different levels of severity and PCR test results, and with and without tocilizumab and/or anakinra and/or prednisolone treatment. It found that antibody response was independently associated with the level of lung involvement on thoracic CT. A total of 95%, 90% and 69% of patients with severe, moderate and mild lung involvement, respectively, were positive for SARS-CoV-2 antibody. The S/CO level was found to be 2.21, 1.69 and 1.16 times higher in patients with severe, mild and moderate CT involvement than in those with no involvement, respectively. When the analysis was limited to only confirmed cases, moderate/severe lung involvement on thoracic CT was found to be an independent factor affecting the antibody seroconversion. The extent of involvement on thoracic CT was shown to be directly correlated with disease severity in COVID-19 in previous studies (Feng et al., 2020). There are also studies showing the correlations between the severity of infection and either involvement of lung on thoracic CT or antibody responses to SARS-CoV-2 (Chen et al., 2020). The decrease in the number of CD4+ T cells and the increase in IL-6 levels were reported to be correlated with the volume of lung lesions in critically ill COVID-19 patients in one study (He et al., 2020). However, we found no other study showing a direct relationship between the extent of lung involvement on thoracic CT and the rate and level of antibody response. Therefore, based on the findings of the current study, antibody responses and levels could be predicted from the level of lung involvement on thoracic CT, even in patients without a positive PCR test.
It was also found that the antibody response rate was higher in patients who were treated with either tocilizumab, anakinra or prednisolone compared with patients who were not administered these drugs on univariate analysis. However, the use of those anticytokines and prednisolone for the treatment of COVID-19 were not found to be independent factors affecting the antibody response rate. There could be so many reasons why this study failed to obtain less SARS-CoV-2 antibody seroconversion in COVID-19 patients who were treated with drugs affecting the immune system. First of all, it is well known that the potential adverse effects of immunosuppressive or immunomodulatory agents are related to both the average dose and the cumulative duration of use (Slade and Hepburn, 1983). In the case of COVID-19, those drugs were used for no more than 1 week or a maximum of 2 weeks; this could be one of the reasons why no adverse effect of those drugs on antibody response to SARS-CoV-2 was found. The finding of lower antibody response among COVID-19 patients who were on long-term immunosuppressive drugs also supports that idea.
IL-6 works as a B cell differentiation factor, which induces activated B cells to produce immunoglobulin (Muraguchi et al., 1988). A T cell-dependent antibody response against virus infection was found to be impaired in IL-6-deficient mice (Kopf et al., 1994). The current study found that tocilizumab, an IL-6 receptor blocker, did not affect the antibody response in patients with COVID-19. Another study also reported that tocilizumab did not impair the viral-specific antibody responses in COVID-19 (Masiá et al., 2020). The antibody response to influenza vaccine in rheumatoid arthritis patients was not hampered by tocilizumab (Mori et al., 2012). IL-1 could affect the antibody response as a result of its effects on T cells and IL-6 production (Dinarello, 2009). However, the current study found that either anakinra alone or combined with tocilizumab did not affect antibody responses in patients with COVID-19. It found no other study evaluating the effect of anakinra on antibody response in COVID-19 patients. In one study, anakinra showed no significant difference in anti-pneumococcal antibody responses (Quartier et al., 2011). One explanation of the current results may be that hyperactivation of both innate and acquired immunity develops because of continuous and high-level pathogen and danger-associated molecular pattern signals in severe COVID-19, and targeting only IL-1 or IL-6 inhibitors could not be adequate enough to control or suppress dysregulated immune responses including humoral response. The failure of recent randomized clinical trials of tocilizumab for treating patients with severe COVID-19 (Stone et al., 2020) and the positive effect of personalized treatment decisions on early mortality in COVID-19 patients (Garcia-Vidal et al., 2020) also support that idea. This was also the case in previous sepsis trials: based on the assumption that sepsis mortality was driven by excessive inflammation, many trials have been performed with a variety of anti-inflammatory agents and none of them have shown benefit, strongly suggesting that inhibition of specific components of the inflammatory response does not improve survival in all-comers with sepsis and selected based on severity of disease. The key to success of either anti-inflammatory or immunostimulatory type of immunomodulation is to identify the patients who may benefit from a particular intervention. Considering the complexity of the host response during COVID-19 and the diversity of pathophysiologic mechanisms at play, it is unlikely that the current “one-target” and “one-size-fits-all” strategy will control all parts of the dysregulated immune system (Van der Poll and Wiersinga, 2020). Another explanation may be that tocilizumab and/or anakinra are not specifically targeting B cells; antibody production may not simply depend on IL-1 or IL-6. IL-6 was not required for influenza virus-specific antibody responses by tonsillar mononuclear cells in an in vitro study (Costelloe et al., 1993). Lastly, an inflammatory environment in severe patients with cytokine storm results in exhaustion of lymphocytes and lymphopenia, and improvement of inflammatory status may be associated with better immune functions against SARS-CoV-2, both in humoral and cellular immunity (Riva et al., 2020).
Although glucocorticoids cause no significant acute changes in B cells (Olnes et al., 2016), the number of circulating B lymphocytes and levels of IgG may be reduced with short-term glucocorticoid administration (Slade and Hepburn, 1983). In the current study, prednisolone used for COVID-19 treatment was not found to be associated with the antibody response. In another study, hospitalized COVID-19 patients received corticosteroids and/or tocilizumab; no detrimental effect was found on antibody responses. Although differences in antibody response favored patients receiving corticosteroids, such differences vanished after adjustment (Masiá et al., 2021). It was same in the current study, where patients treated with either tocilizumab, anakinra or prednisolone had higher antibody responses than those who did not take these medications, but none were found to be an independent factor for antibody response on multivariate analysis. As a result, it may be concluded that tocilizumab, anakinra and prednisolone, even in combination, do not negatively impact the humoral immune response against SARS-CoV-2.
The current study found that seroconversion in COVID-19 cases was related to the duration between hospital admission and antibody testing (OR 1.02; 95% CI 1.01–1.03; p < 0.001). It was shown that SARS-CoV-2 IgG antibodies develop over a period of 7–50 days from symptom onset, with a median of 24 days (Wajnberg et al., 2020a). Although the mean time from hospital admission to antibody testing was 82 days in the current study, antibody testing was performed before 42 days in 25% of the patients, and as a result some of them could not produce adequate antibodies in a given time. As 76% of the patients with a negative antibody response had mild disease, either with no involvement or mild involvement on thoracic CT, another explanation of time dependence of seroconversion could be the longer duration of antibody response among asymptomatic and mild cases. It was also shown that seroconversion in mild COVID-19 cases might take longer time to rise (Wajnberg et al., 2020a, Wajnberg et al., 2020b). Another explanation for this could be due to a mild and transient response; antibodies become negative in a short time among mild or asymptomatic cases (Poland et al., 2020). In the convalescent phase, IgG titres in symptomatic individuals were found to remain significantly higher than those in asymptomatic individuals (Adams et al., 2020). Notably, in one study, IgG titers declined during the convalescent phase in both symptomatic and asymptomatic individuals, with 13% and 40% of symptomatic and asymptomatic individuals becoming IgG seronegative within 2–3 months following infection (Long et al., 2020). Finally, some of the current patients might not have had COVID-19 at all, as the rate of antibody positivity was found to be 72% without PCR positivity. However, 31 of 89 antibody negative patients were confirmed COVID-19 cases with a positive PCR test. The current analysis of the only confirmed COVID-19 cases showed that the time from hospital admission to antibody testing was not found to be a factor affecting the antibody response. This could be related to the very early sampling and false negativity of PCR testing among PCR-negative cases, which could be the result of delayed antibody responses among those cases. As it was not planned to assess the kinetics of the antibody response, a final comment on this issue cannot be made.
This study had some limitations. Although it prospectively included patients during the convalescent phase, the clinical and laboratory data of active COVID-19 were retrospectively obtained from the hospital database; as a result, it could not get some of the data, including the duration of symptoms, total duration of prednisolone use, and the time from the symptoms to thoracic CT evaluation. As SARS-CoV-2 RNA PCR was negative in 40% of the patients, some of them might not have had COVID-19 at all. The thoracic CT results were included for analysis, and 72% of patients with a negative PCR test had a positive antibody test result, which showed that selection of patients was appropriate.
This study had some strengths. It analyzed factors affecting antibody response in a large number of patients with different disease severity and PCR result status. For the first time, it showed a direct association between antibody response rate and S/CO ratio and extent of lung involvement on CT. It also showed that anakinra treatment does not affect the antibody response in patients with severe COVID-19.
Conclusion
Although tocilizumab, anakinra and prednisolones have some effects on T cells, B cells and antibody response, their use during the course of COVID-19 was not found to be associated with a negative effect on antibody response. The main driver of the antibody response to SARS-CoV-2 and level of it among patients with COVID-19 was found to be the extent of lung involvement on thoracic CT, which correlated well with the severity of infection. As a result, the antibody response to SARS-CoV-2 and levels of it could be predicted by thoracic CT.
Authors’ contributions
SB, SŞY and SM conceived and designed the study. SB and SŞY analyzed the results, and wrote the manuscript. All authors reviewed and approved the final version for submission.
Funding
This work was supported by the Scientific Research Projects Coordination Unit of Istanbul University (Project number: 36884).
Conflict of interest
No conflict of interest was declared by the authors.
Acknowledgement
The authors thank Yunus Çatma for his help in data collection.
References
- Adams E.R., Ainsworth M., Anand R., Andersson M.I., Auckland K., Baillie J.K. National COVID Testing Scientific Advisory Panel. Antibody testing for COVID-19: a report from the National COVID Scientific Advisory Panel. Wellcome Open Res. 2020;5:139. doi: 10.12688/wellcomeopenres.15927.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen Y., Zuiani A., Fischinger S., Mullur J., Atyeo C., Travers M. Quick COVID-19 healers sustain anti-SARS-CoV-2 antibody production. Cell. 2020;183(6):1496–1507.e16. doi: 10.1016/j.cell.2020.10.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costelloe K.E., Smith S.H., Callard R.E. Interleukin 6 is not required for antigen-specific antibody responses by human B cells. Eur J Immunol. 1993;23(4):984–987. doi: 10.1002/eji.1830230436. [DOI] [PubMed] [Google Scholar]
- Dinarello C.A. Immunological and inflammatory functions of the Interleukin-1 family. Annu Rev Immunol. 2009;27:519–550. doi: 10.1146/annurev.immunol.021908.132612. [DOI] [PubMed] [Google Scholar]
- Feng Z., Yu Q., Yao S., Luo L., Zhou W., Mao X. Early prediction of disease progression in COVID-19 pneumonia patients with chest CT and clinical characteristics. Nat Commun. 2020;11(1) doi: 10.1038/s41467-020-18786-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Vidal C., Moreno-García E., Hernández-Meneses M., Puerta-Alcalde P., Chumbita M., Garcia-Pouton N. Personalized therapy approach for hospitalized patients with COVID-19. Clin Infect Dis. 2020 doi: 10.1093/cid/ciaa964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- He S., Zhou C., Lu D., Yang H., Xu H., Wu G. Relationship between chest CT manifestations and immune response in COVID-19 patients. Int J Infect Dis. 2020;98:125–129. doi: 10.1016/j.ijid.2020.06.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jalaber C., Lapotre T., Morcet-Delattre T., Ribet F., Jouneau S., Lederlin M. Chest CT in COVID-19 pneumonia: a review of current knowledge. Diagn Interv Imaging. 2020;101(7–8):431–437. doi: 10.1016/j.diii.2020.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kopf M., Baumann H., Freer G., Freudenberg M., Lamers M., Kishimoto T. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature. 1994;368(6469):339–342. doi: 10.1038/368339a0. [DOI] [PubMed] [Google Scholar]
- Long Q.X., Tang X.J., Shi Q.L., Li Q., Deng H.J., Yuan J. Clinical and immunological assessment of asymptomatic SARS-CoV-2 infections. Nat Med. 2020;26(8):1200–1204. doi: 10.1038/s41591-020-0965-6. [DOI] [PubMed] [Google Scholar]
- Maeda K., Mehta H., Drevets D.A., Coggeshall K.M. IL-6 increases B-cell IgG production in a feed-forward proinflammatory mechanism to skew hematopoiesis and elevate myeloid production. Blood. 2010;115(23):4699–4706. doi: 10.1182/blood-2009-07-230631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masiá M., Fernández-González M., Padilla S., Ortega P., García J.A., Agulló V. Impact of interleukin-6 blockade with tocilizumab on SARS-CoV-2 viral kinetics and antibody responses in patients with COVID-19: a prospective cohort study. EBioMedicine. 2020;60 doi: 10.1016/j.ebiom.2020.102999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masiá M., Fernández-González M., García J.A., Padilla S., Gutiérrez F. Lack of detrimental effect of corticosteroids on antibody responses to SARS-CoV-2 and viral clearance in patients hospitalized with COVID-19. J Infect. 2021;82(3):414–451. doi: 10.1016/j.jinf.2020.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori S., Ueki Y., Hirakata N., Oribe M., Hidaka T., Oishi K. Impact of tocilizumab therapy on antibody response to influenza vaccine in patients with rheumatoid arthritis. Ann Rheum Dis. 2012;71(12):2006–2010. doi: 10.1136/annrheumdis-2012-201950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muraguchi A., Hirano T., Tang B., Matsuda T., Horii Y., Nakajima K. The essential role of B cell stimulatory factor 2 (BSF-2/IL-6) for the terminal differentiation of B cells. J Exp Med. 1988;167(2):332–344. doi: 10.1084/jem.167.2.332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olnes M.J., Kotliarov Y., Biancotto A., Cheung F., Chen J., Shi R. CHI Consortium. Effects of systemically administered hydrocortisone on the human immunome. Sci Rep. 2016;6 doi: 10.1038/srep23002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poland G.A., Ovsyannikova I.G., Kennedy R.B. SARS-CoV-2 immunity: review and applications to phase 3 vaccine candidates. Lancet. 2020;396(10262):1595–1606. doi: 10.1016/S0140-6736(20)32137-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quartier P., Allantaz F., Cimaz R., Pillet P., Messiaen C., Bardin C. A multicentre, randomised, double-blind, placebo-controlled trial with the interleukin-1 receptor antagonist anakinra in patients with systemic-onset juvenile idiopathic arthritis (ANAJIS trial) Ann Rheum Dis. 2011;70(5):747–754. doi: 10.1136/ard.2010.134254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RECOVERY Collaborative Group, Horby P., Lim W.S., Emberson J.R., Mafham M., Bell J.L. Dexamethasone in Hospitalized Patients with Covid-19. N Engl J Med. 2021;384(8):693–704. doi: 10.1056/NEJMoa2021436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riva G., Nasillo V., Tagliafico E., Trenti T., Luppi M. COVID-19: room for treating T cell exhaustion? Crit Care. 2020;24(1):229. doi: 10.1186/s13054-020-02960-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roll P., Muhammad K., Schumann M., Kleinert S., Einsele H., Dorner T. In vivo effects of the anti–interleukin-6 receptor inhibitor tocilizumab on the B cell compartment. Arthritis Rheum. 2011;63(5):1255–1264. doi: 10.1002/art.30242. [DOI] [PubMed] [Google Scholar]
- Rydyznski Moderbacher C., Ramirez S.I., Dan J.M., Grifoni A., Hastie K.M., Weiskopf D. Antigen-specific adaptive immunity to SARS-CoV-2 in acute COVID-19 and associations with age and disease severity. Cell. 2020;183(4):996–1012.e19. doi: 10.1016/j.cell.2020.09.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slade J.D., Hepburn B. Prednisone-induced alterations of circulating human lymphocyte subsets. J Lab Clin Med. 1983;101(3):479–487. [PubMed] [Google Scholar]
- Stone J.H., Frigault M.J., Serling-Boyd N.J., Fernandes A.D., Harvey L., Foulkes A.S. Efficacy of tocilizumab in patients hospitalized with Covid-19. N Engl J Med. 2020;383(24):2333–2344. doi: 10.1056/NEJMoa2028836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toniati P., Piva S., Cattalini M., Garrafa E., Regola F., Castelli F. Tocilizumab for the treatment of severe COVID-19 pneumonia with hyperinflammatory syndrome and acute respiratory failure: a single center study of 100 patients in Brescia, Italy. Autoimmun Rev. 2020;19(7) doi: 10.1016/j.autrev.2020.102568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van der Poll T., Wiersinga W.J. Sepsis and septic shock. In: Bennett J.E., Dolin R., Blaser M.J., editors. Mandell, Douglas, and Bennett’s principles and practice of infectious diseases. Elsevier; Philadelphia: 2020. pp. 1005–1008. [Google Scholar]
- Wajnberg A., Mansour M., Leven E., Bouvier N.M., Patel G., Firpo-Betancourt A. Humoral response and PCR positivity in patients with COVID-19 in the New York City region, USA: an observational study. Lancet Microbe. 2020;1(7):e283–9. doi: 10.1016/S2666-5247(20)30120-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wajnberg A., Amanat F., Firpo A., Altman D.R., Bailey M.J., Mansour M. Robust neutralizing antibodies to SARS-CoV-2 infection persist for months. Science. 2020;370(6521):1227–1230. doi: 10.1126/science.abd7728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization (WHO) 2020. WHO COVID-19 case definition.https://www.who.int/publications/i/item/WHO-2019-nCoV-Surveillance_Case_Definition-2020.2 [Google Scholar]