Abstract
BACKGROUND AND PURPOSE:
ICAS is one of the therapeutic options in symptomatic cerebral artery stenosis. iaDSA is the current criterion standard examination after ICAS for the detection of ISR. In this study, we evaluated ivACT as a potential noninvasive follow-up alternative.
MATERIALS AND METHODS:
In 17 cases, ivACT and iaDSA were performed after ICAS. Both procedures were carried out on a flat-panel-detector-equipped angiography system. Postprocessing of ivACT acquisitions was performed on a dedicated workstation producing multiplanar reformations of the stent region and other intracranial arteries. Restenotic lesions were compared with iaDSA measurements. All studies were independently evaluated by 2 experienced neuroradiologists blinded to patients data.
RESULTS:
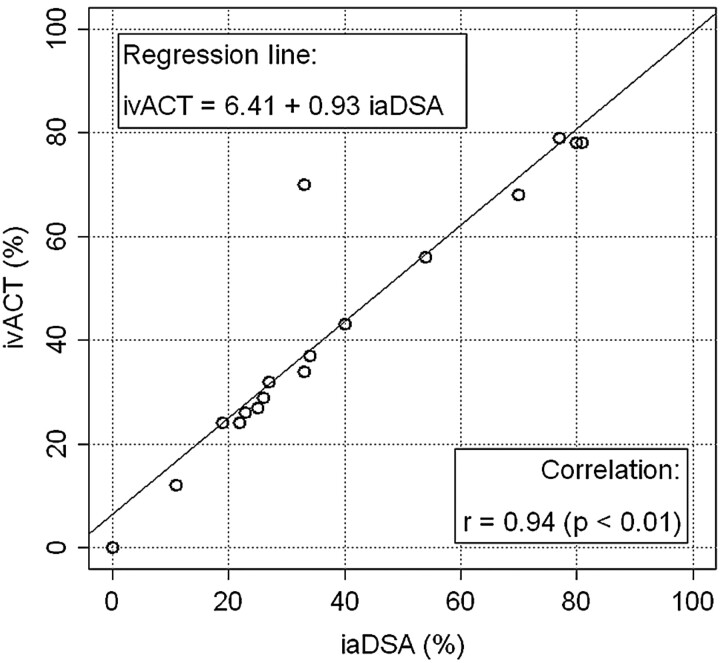
In 5 cases, ISR was diagnosed on iaDSA images. All restenotic lesions were reliably detected (sensitivity, 100%; 95%CI, 48%–100%) and could be correctly quantified on ivACT images in comparison with iaDSA. The neuroradiologists correctly excluded ISR in 11 of 12 lesions after viewing the ivACT examinations (specificity, 92%; 95%CI, 62%–100%). Measurements of ISR on ivACT were highly correlated to iaDSA (Pearson r = 0.94, P < .01).
CONCLUSIONS:
IvACT is a promising noninvasive follow-up examination after ICAS. With its high spatial resolution, it can reliably detect or exclude ISR. Contrary to iaDSA, there is no need for a recovery period after ivACT and the risk of neurologic complications is practically lowered to zero.
ICAS is a viable treatment option in the case of symptomatic intracranial atherosclerotic disease. Due to the high stroke risk under medical treatment, patients with high-grade (>70%) symptomatic intracranial stenosis represent the main target group for alternative endovascular treatment concepts such as ICAS.1 During follow-up, ISR seems to be a major problem, with ≤30% of the patients with ICAS presenting with ISR after 6 months.2,3 The main mechanism of ISR is myointimal hyperplasia. Turk et al2 reported that the rate of ISR was 31.2%, with 31.0% of the lesions being symptomatic. Especially younger patients with intracranial stenosis of the ICA or the M1 segment of the middle cerebral artery have shown a high risk of developing ISR, which may cause stroke in the first 12 months in ≤5% of the cases. Whether restenosis rates are relevant is still under discussion, but it remains undisputable that follow-up is necessary.4,5
Conventional iaDSA is the current criterion standard follow-up examination after ICAS, and as such, it has been primarily used in studies evaluating ISR rates.6,7 Due to its invasive nature, iaDSA carries the risk of neurologic complications, especially in elderly patients with known cardiovascular disease and when fluoroscopic times are ≥10 minutes.8 Recent studies have shown that, within a high-volume neurointerventional department, the risk for neurologic complications during iaDSA is close to zero.9 Nevertheless, the accessibility of a high-volume institution is not always guaranteed for patients having their follow-up after ICAS, and even in high-volume centers, it is impossible to ensure that every iaDSA will be performed by an experienced operator. A new noninvasive technique for the depiction of intracranial vessels after ICAS is the ivACT.10 Buhk et al10 demonstrated, in a small series, that ivACT, in comparison with MDCTA, is a feasible follow-up option for the delineation of in-stent pathologies or the exclusion of ISR. While MDCTA has been proved a reliable tool for screening intracranial artery stenosis,11 in vitro studies have shown that it is insufficient for the assessment of ISR. Trossbach et al12 concluded that it is impossible to visualize the different stenoses subjectively—that is, without using image-analysis software—in a series of in vitro examinations delineating different grades of ISR in 3 and 4 mm stents for intracranial angioplasty. Similar conclusions have been drawn in studies evaluating MDCTA in the assessment of ISR in coronary stents.13 On the other hand, earlier studies have already presented the potential of ACT in the evaluation of small coronary stents. In their study, Mahnken et al14 noted that ACT proved to be superior to MDCT for in vitro visualization of coronary artery stents because the improved spatial resolution of ACT enabled better depiction of the stent lumen. This characteristic of ACT also allows the cross-sectional evaluation of normal or abnormal deployment of small intracranial stents, which is impossible with other imaging modalities such as MDCT, DSA, or MR imaging.15
Other noninvasive techniques allowing the detection of ISR include transcranial duplex sonography and quantitative MR angiography.16,17 However, both of these lack the ability to provide anatomic data of the stent region, and especially duplex sonography is limited due to its dependence on operator experience and the anatomy of the temporal bone window.
The purpose of this study was the evaluation of ivACT in the detection of ISR and the comparison with iaDSA findings in the follow-up of 17 cases after ICAS.
Materials and Methods
Patients
Fourteen patients were treated with ICAS of a symptomatic intracranial artery stenosis from July 2006 to May 2009 (10 men, 4 women; mean age, 60 years; range, 45–75 years). One patient received 2 intracranial stents (petrous ICA and M1), and in another patient, 3 follow-up examinations were performed in the time period mentioned above: the first, 3 months after ICAS; the second and third, 7 and 4 months after the first and second PTA of a recurrent high-grade ISR, respectively. Locations of stenoses, presenting symptoms, and applied stent systems are shown in On-line Table 1. All patients were admitted for standard follow-up within an average of 7 months (range, 3–12 months) after ICAS.
Approval of the local ethics committee and informed patient consent were obtained.
Image Acquisition
IvACT acquisitions and iaDSA examinations were performed on a biplane angiography system equipped with flat panel detectors (Axiom Artis dBA, Siemens, Erlangen, Germany). DSA data included only standard angiography series. For the acquisition of ivACT, we used the DynaCT program of our suite (Siemens) with the following parameters: 20 seconds of rotation; 538 projections; 220° total angle; CTDIw, approximately 35 mGy (manufacturer information); and a 30 × 40 cm detector, which allows the reconstruction of a nontruncated volume of approximately 22 cm (in-plane) and 16 cm (in the z-direction). While planning the DynaCT, we placed the stented segment near the center of the FOV, because a higher image quality is guaranteed near the central plane of the conebeam.18 However, we avoided placing the stented segment exactly in the center of the conebeam because ring artifacts can negatively affect the image quality.19 Postprocessing of the rotational image data to a volume dataset was performed by using dedicated commercial software on a Leonardo medical workstation (InSpace 3D; Siemens). The software includes system-specific algorithms to correct beam-hardening, scattered radiation, truncated projections, and ring artifacts. Reconstruction resulted in a volume dataset of approximately 400 sections with a 512 × 512 matrix and an isotropic spatial resolution of approximately 0.1 × 0.1 × 0.1 mm3. The ivACT datasets were further processed to multiplanar reformations parallel and perpendicular to the stent region with a section thickness of 0.2–0.3 mm and maximum intensity projections of the other intracranial vessels. Before acquisition, 100 mL of iomeprol (Imeron 400; Bracco ALTANA Pharma, Konstanz, Germany) had been injected into a cubital vein at a flow rate of 5 mL/s by using a power injector. The start delay for rotational acquisition was 14–20 seconds, depending on the age and cardiopulmonary status of the patient. During ivACT, the patient was asked to close his or her eyes and to breath-hold during the 20 seconds of the C-arm rotation.
Image Analysis
Two neuroradiologists (M.-N.P., A.X.) independently performed the image viewing and rating on the above-mentioned Leonardo medical workstation by using the Warfarin-Aspirin Symptomatic Intracranial Disease study technique.1 As in the study of Albuquerque et al,6 ISR was defined as >50% stenosis within or adjacent (within 5 mm) to the stent as well as >20% absolute luminal loss at follow-up imaging. Moreover, all restenotic lesions, even those not fulfilling the criteria of ISR, were categorized by using the modified Mehran classification system.6 This system, originally developed to describe ISR after coronary PTA with stent placement, divides ISR into 4 subgroups: class I, a focal group with lesions involving <50% of the stented segment; class II, a diffuse intrastent group (>50% of the stented segment); class III, a proliferative group with lesions expanding beyond the confines of the stent; and class IV, a complete stent occlusion group. Class I lesions can be further categorized IA, IB, or IC cases. ISR cases were then compared with the original lesions presented for treatment.
Statistical Analysis
The mean value of each pair of measurements (rater 1, rater 2) was then calculated and used for further statistical analysis. The correlation between ISR percentage measures on ivACT and iaDSA examinations was assessed by the Pearson correlation coefficient r. Additionally, a simple linear regression of ivACT versus iaDSA was performed. Considering iaDSA as the criterion standard for the detection of ISR, we calculated empiric sensitivity and specificity, as well as the positive and negative predictive values for ivACT as a detection method. Statistical significance was assumed for P values < .05. All analyses were conducted with the free software R (version 2.8, http://www.r-project.org).
Results
Follow-up imaging with iaDSA and ivACT was obtained for 17 stenotic lesions. Measurements and descriptive results can be seen in On-line Table 2. Nine lesions had been treated with a self-expanding intracranial nitinol stent (Wingspan; Boston Scientific, Natick, Massachusetts), 4 with a relatively new balloon-mounted stent designed for intracranial use (Pharos; Micrus Endovascular, Renens, Switzerland), and another 4 lesions with a common coronary stent (Driver RX, Medtronic, Minneapolis, Minnesota). IaDSA excluded ISR in 12 of 17 lesions (70.6%). Five lesions (29.4%) developed ISR during the follow-up interval, and 4 of them were treated successfully with another PTA. The patient in case 4 developed a mild ISR with 54% restenosis (Δ = 22%), for which angioplasty was not considered necessary. At follow-up, 2 of the 5 ISR lesions were symptomatic with a TIA, while the rest presented no new symptoms. The average change of stenosis percentage at follow-up compared with measurements before ICAS documented an absolute reduction of 40% (range, −90% to +9%); compared with the percentages of stenoses directly after ICAS, an average increase of 21% could be confirmed with iaDSA after the follow-up interval (range, 0%–63%). With the modified Mehran system, a restenotic pattern could be described in 9 of 17 lesions (though only 5 fulfilled the criteria for an ISR). Seven of 9 restenotic lesions were characterized as focal lesions involving <50% of the stented segment (class I), of which 5 were group IB and 1, group IA and IC, respectively. Two lesions demonstrated diffuse stenosis (class II), but there were no proliferative lesions (class III) or complete stent occlusions detected.
IvACT was of diagnostic quality in all 17 cases examined. No motion artifacts−related problems were encountered, and sufficient delineation of the intracranial vessels and the stented segment was demonstrated. Only the radiopaque stent markers of the Wingspan stent proved to be a problem because a very short segment could not be delineated due to streak artifacts. The confines of the other stents could be depicted without any limitations due to metal artifacts. The neuroradiologists correctly excluded ISR in 11 of 12 lesions after viewing the ivACT examinations (specificity, 92%; 95% CI, 62%–100%). The cases presented in Fig 1 illustrate 2 examples in which ISR was correctly excluded in ivACT, in accordance with iaDSA measurements. With ivACT, there was 1 false-positive diagnosis of ISR (case 2). In this case, a Wingspan stent (2.5 × 15 mm) had been placed in a symptomatic stenosis of the right M1 segment and was expanded to 1.8 mm. On follow-up imaging, a 70% ISR was detected in ivACT, whereas iaDSA excluded an ISR with 33% residual stenosis (ΔafterICAS = 0%, Fig 2). All restenotic lesions were sufficiently depicted in ivACT (sensitivity, 100%; 95% CI, 48%–100%) and could be correctly quantified and categorized by using the modified Mehran system. Thus, a positive predictive value of 83% (95% CI, 36%–100%) and a negative predictive value of 100% (95% CI, 71%–100%) could be achieved with ivACT.
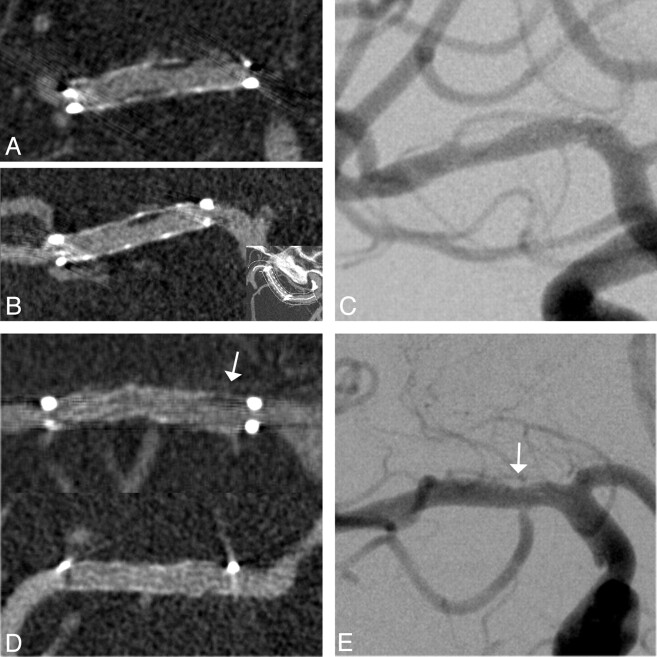
Fig 1.
Follow-up images of a 56-year-old woman 7 months after ICAS of a 76% middle cerebral artery stenosis. A and B, Excellent delineation of in-stent anatomy on coronal (A) and curved planar (B) ivACT reformations allows exclusion of a high-grade ISR and detection of low-grade neointimal hyperplasia within the body of the stent. C, The same findings are demonstrated on the iaDSA image. D and E, Another example of a low-grade restenotic lesion (25%), which is depicted in the proximal stent portion (arrows) on both ivACT (D) and a standard iaDSA image (E). This 61-year-old woman was admitted to standard follow-up without any new symptoms after ICAS of a symptomatic 79% middle cerebral artery stenosis. At the confines of the Wingspan stent, streak artifacts limit the image quality on ivACT reformations.
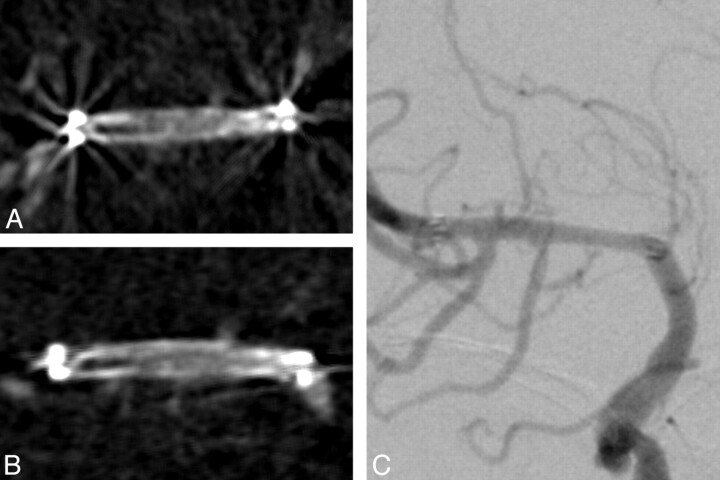
Fig 2.
ISR is falsely diagnosed by both raters on transversal (A) and coronal (B) ivACT reformations. C, On both images, a restenotic lesion is delineated at the distal stent portion. ISR can be excluded by iaDSA. The patient was admitted 2 months earlier than his planned follow-up appointment, despite no new symptoms, due to suspected restenosis on transcranial duplex sonography.
Figure 3 shows the follow-up images of a 69-year-old male patient. Seven months after ICAS of a symptomatic BA stenosis, the patient was admitted for standard follow-up imaging without any new symptoms. IvACT revealed a high-grade ISR within the applied Pharos stent (78% ISR, class II), which was verified by iaDSA, with which an 81% ISR could be depicted. ISR quantification on ivACT images was highly correlated to iaDSA (Pearson r = 0.94, P < .01). Therefore, we built a simple linear regression model of both parameters, the estimated coefficients of which are depicted in Fig 4.
Fig 3.
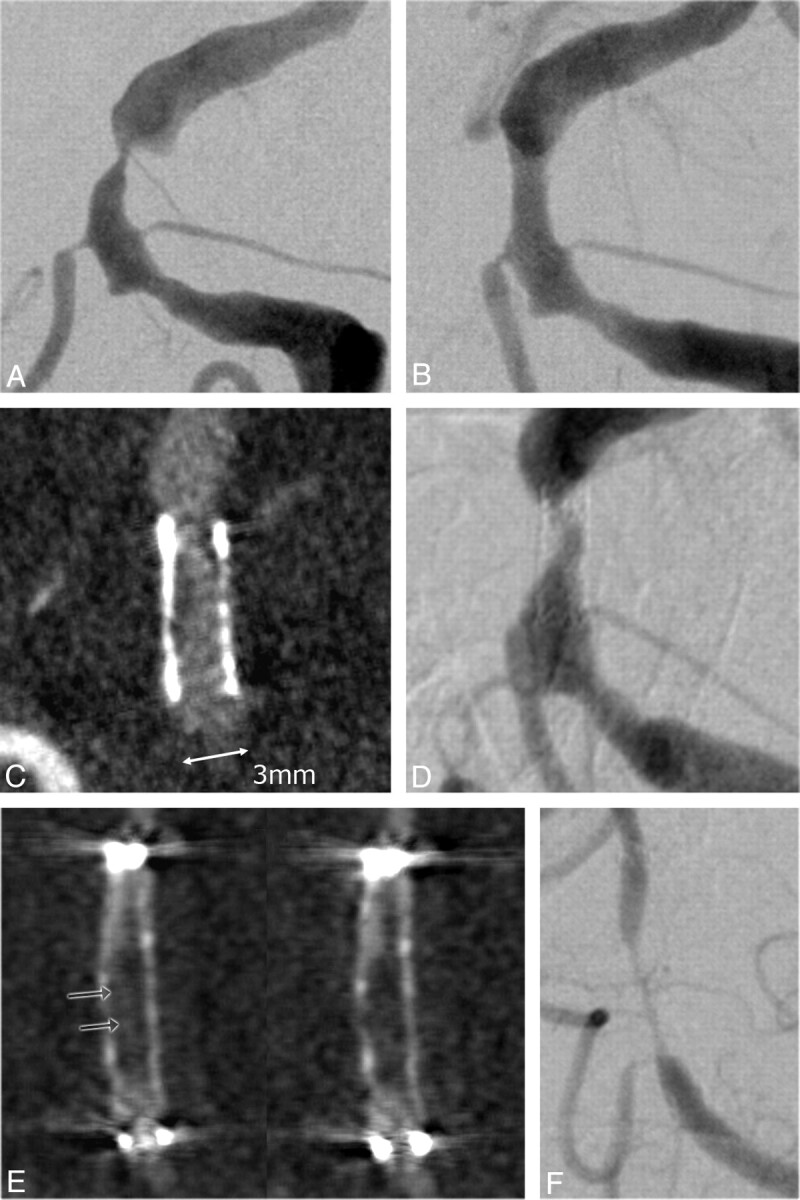
Images of a 69-year-old patient before (A) and after (B) ICAS of a high-grade (85%) symptomatic BA stenosis. The patient was admitted to follow-up without any new symptoms 7 months after stent placement. C and D, ISR is depicted on ivACT reformations (C), a finding that is confirmed on iaDSA (D). The stenosis percentage on ivACT and iaDSA correlates excellently. E and F, In another example of a 45-year-old woman, high-grade ISR can be reliably diagnosed on ivACT (E) due to extensive neointimal hyperplasia. The residual filiform lumen within the stent can be correctly measured on ivACT reformations (E, arrows), compared with iaDSA images (F).
Fig 4.
Scatterplot of ivACT (%) versus iaDSA (%) plus an estimated regression line.
Discussion
ICAS is a promising new therapeutic option in patients with symptomatic intracranial atherosclerotic disease, especially when stenotic lesions are refractory to medical treatment.3,7,20 However, ISR rates of ≤31% have been reported in the past, proving the necessity for reliable follow-up imaging. The current criterion standard, iaDSA, has known issues due to its invasive nature, carrying the risks of neurologic or other periprocedural complications.8
Noninvasive follow-up imaging of small intracranial stents for the detection of ISR has not been very successful to date. Commonly used stainless steel coronary stents cause metal artifacts on CT and significant signal intensity loss on MR images. Nitinol stents are more compatible with MR imaging, but exact depiction of the in-stent region and exact measurement of ISR are not possible. Duplex sonography is another noninvasive follow-up examination for the detection of ISR.16 However, the absence of a sufficient temporal bone window, the investigator dependency, and its substantial limitations in the assessment of the petrous segment of the ICA or the BA restrict the use of duplex sonography as a first-line follow-up technique. Small series have shown the feasibility of MDCTA as a follow-up after ICAS,21 but larger in vitro studies have concluded that it is insufficient for the exact quantification of ISR in small vessel stents.12 Similar results have been shown in studies evaluating MDCTA in the assessment of ISR in small coronary stents.13
On the contrary, ACT has proved to be a technique with performance characteristics suitable for imaging small high-contrast structures such as intracranial stents.22 Due to its superior spatial resolution compared with MDCT, ACT has already proved to be better for the depiction of self-expandable intracranial stents or other small objects, such as cochlear implants.15,23 The slightly inferior contrast resolution leads to insufficient detection of ischemic lesions on ACT compared with MDCT,24 but in high-contrast imaging (such as ivACT and MDCTA), this minor difference does not have a significant influence on image quality. Also, contrary to MDCT, reconstruction of ACT rotational data results in volume datasets with isotropic voxel size, meaning sufficient image quality on multiplanar reformations even if the stent is not aligned to the conebeam axis (vessels with curved shape, irregular walls, and so forth, Fig 1).12,25 The addition of intravenous contrast medium has created a “mixture” with the ability of sufficient depiction of ISR.10 This potential is very important if one takes into account the increased use of intracranial stents, not only for revascularization of stenotic vessels but also for treating cerebral aneurysms.26,27 Moreover, the use of new flat detector technologies and new reconstruction algorithms may, in the near future, allow dynamic perfusion imaging to be extracted from the same ivACT dataset used for angiographic imaging. This would facilitate the assessment of hemodynamic relevance of the stenotic lesion in addition to quantification of restenosis.28
In our study, ivACT showed a high sensitivity and specificity for the detection of ISR compared with iaDSA. We could successfully detect and quantify ISR on ACT images in all 5 patients presenting an ISR at follow-up iaDSA. Additionally, in 11 of 12 cases without an ISR, ivACT sufficiently excluded a restenotic lesion. In the majority of cases, these were stents with diameters of 2.75–4 mm. In only 1 case, both neuroradiologists falsely diagnosed an ISR on ACT images. A possible explanation could be the size of this stent because it was a 2.5-mm Wingspan stent self-expanded to a diameter of 1.8 mm, as well as the streak artifacts near the confines of the stent (Fig 2).
Regarding the radiation dose, the use of a 20-second ACT protocol results in a CTDIw value ∼ 3 5 mGy, which is comparable with or even lower than the usual MDCTA or conventional cranial CT protocols (∼60 mGy).29,30 The dose could be further reduced with the application of a 5-second (CTDIw ∼ 9 mGy) or an 8-second ACT protocol. However, longer rotating time is currently necessary to achieve sufficient contrast resolution for the depiction of ISR. Kyriakou et al31 report in their publication that “the sampling artifacts, originating from the small number of projections in high speed (5 seconds), are quite distinct, and in high-speed protocols low tissue resolution is nearly nonexistent.” In contrast to other studies that have used a high-dose 20-second ACT protocol with a CTDIw ∼ 75 mGy, we used the low-dose protocol solely in our study.23,31
The amount of contrast media used for ivACT in our study is higher than the amount being used in a conventional MDCTA (∼60 mL in our department). The reason for this difference is the necessity of a longer acquisition time with the ACT protocol. To achieve an adequate contrast throughout the whole rotation, one must inject large amounts of contrast media continuously. We are currently using a modified protocol with 90 mL of contrast medium followed by 30 mL of saline flush, but a definite solution to the contrast dosage issue will be the deployment, in the future, of a shorter rotation protocol, with contrast and spatial resolution comparable with the currently used 20-second ACT.
The limitations of this study include its small sample size, the absence of bolus-tracking for ivACT, and the metal artifacts around the markers of the Wingspan stent. Larger studies have to be conducted to verify the value of ivACT in the follow-up imaging after ICAS. Although the first results are very promising, the absence of a bolus-tracking method after intravenous contrast medium administration could sometimes result in an insufficient depiction of intracranial vessels. This would mean repeated examinations and increased overall radiation and contrast dosage for the patient. We are currently working with the manufacturer on this issue, and we think that a viable method of bolus-tracking will be available in the months to come. Regarding the streak artifacts at the confines of the Wingspan stent, one should consider that an ISR that includes the confines of a stent is an infrequent one (modified Mehran IA).6 Moreover, with the development of new metal artifacts-reduction algorithms for ACT, even the marker region of the Wingspan stent may be visible without any streak artifacts in the near future.32
Conclusions
IvACT is a promising new noninvasive follow-up tool after ICAS. In our sample, we could detect ISR on ivACT images with a high sensitivity and specificity. Further research is required to conclude that ivACT could replace iaDSA as a standard follow-up examination after ICAS.
Supplementary Material
Abbreviations
- A2
second segment of anterior cerebral artery
- ACT
angiographic CT
- BA
basilar artery
- C3
third segment of ICA
- CI
confidence interval
- CTDIw
weighted CT dose index
- ΔDSA%
change of stenosis percentage at follow-up iaDSA compared with measurements on iaDSA after stenting
- DSApost
digital subtraction angiography after stenting
- DSApre
digital subtraction angiography before stenting
- DWI
diffusion-weighted imaging
- IA
focal lesion involving the end of the stent
- iaDSA
intra-arterial digital subtraction angiography
- IB
focal lesion involving the body of the stent
- IC
multiple restenotic foci involving <50% of the stented segment
- ICA
internal carotid artery
- ICAS
intracranial artery angioplasty and stenting
- II
diffuse intrastent lesion
- ISR
in-stent restenosis
- ivACT
ACT after intravenous contrast medium application
- M1
first segment of middle cerebral artery
- MDCTA
multidetector CT angiography
- PTA
percutaneous transluminal angioplasty
- rePTA
repeated PTA after ICAS
- TIA
transient ischemic attack
- V4
intracranial segment of vertebral artery
Footnotes
Paper previously presented at: 34th Annual Meeting of the European Society of Neuroradiology, September 17–20 2009; Athens, Greece. Preliminary results after 10 cases.
Indicates article with supplemental on-line tables.
References
- 1. Chimowitz MI, Lynn MJ, Howlett-Smith H, et al. Comparison of warfarin and aspirin for symptomatic intracranial arterial stenosis. N Engl J Med 2005;352:1305–16 [DOI] [PubMed] [Google Scholar]
- 2. Turk AS, Levy EI, Albuquerque FC, et al. Influence of patient age and stenosis location on Wingspan in-stent restenosis. AJNR Am J Neuroradiol 2008;29:23–27. Epub 2007 Nov 7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Levy EI, Turk AS, Albuquerque FC, et al. Wingspan in-stent restenosis and thrombosis: incidence, clinical presentation, and management. Neurosurgery 2007;61:644–50, discussion 650–51 [DOI] [PubMed] [Google Scholar]
- 4. Derdeyn CP, Chimowitz MI. Re: Turk et al and the “how do we spin Wingspan? ” commentary. AJNR Am J Neuroradiol 2008;29:e69; author reply e70. Epub 2008 Apr 3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Gröschel K, Schnaudigel S, Pilgram SM, et al. A systematic review on outcome after stenting for intracranial atherosclerosis. Stroke 2009;40:e340–47 [DOI] [PubMed] [Google Scholar]
- 6. Albuquerque FC, Levy EI, Turk AS, et al. Angiographic patterns of Wingspan in-stent restenosis. Neurosurgery 2008;63:23–27, discussion 27–28 [DOI] [PubMed] [Google Scholar]
- 7. SSYLVIA Study Investigators. Stenting of Symptomatic Atherosclerotic Lesions in the Vertebral or Intracranial Arteries (SSYLVIA): study results. Stroke 2004;35:1388–92 [DOI] [PubMed] [Google Scholar]
- 8. Willinsky RA, Taylor SM, TerBrugge K, et al. Neurologic complications of cerebral angiography: prospective analysis of 2,899 procedures and review of the literature. Radiology 2003;227:522–28 [DOI] [PubMed] [Google Scholar]
- 9. Thiex R, Norbash AM, Frerichs KU. The safety of dedicated-team catheter-based diagnostic cerebral angiography in the era of advanced noninvasive imaging. AJNR Am J Neuroradiol 2010;31:230–34 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Buhk JH, Lingor P, Knauth M. Angiographic CT with intravenous administration of contrast medium is a noninvasive option for follow-up after intracranial stenting. Neuroradiology 2008;50:349–54 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Nguyen-Huynh MN, Wintermark M, English J, et al. How accurate is CT angiography in evaluating intracranial atherosclerotic disease? Stroke 2008;39:1184–88 [DOI] [PubMed] [Google Scholar]
- 12. Trossbach M, Hartmann M, Braun C, et al. Small vessel stents for intracranial angioplasty: in vitro evaluation of in-stent stenoses using CT angiography. Neuroradiology 2004;46:459–63 [DOI] [PubMed] [Google Scholar]
- 13. Haraldsdottir S, Gudnason T, Sigurdsson AF, et al. Diagnostic accuracy of 64-slice multidetector CT for detection of in-stent restenosis in an unselected, consecutive patient population. Eur J Radiol 2009. June 29. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 14. Mahnken AH, Seyfarth T, Flohr T, et al. Flat-panel detector computed tomography for the assessment of coronary artery stents: phantom study in comparison with 16-slice spiral computed tomography. Invest Radiol 2005;40:8–13 [PubMed] [Google Scholar]
- 15. Ebrahimi N, Claus B, Lee CY, et al. Stent conformity in curved vascular models with simulated aneurysm necks using flat-panel CT: an in vitro study. AJNR Am J Neuroradiol 2007;28:823–29 [PMC free article] [PubMed] [Google Scholar]
- 16. Gröschel K, Schnaudigel S, Buhk JH, et al. Intracranial stent restenosis diagnosed on routine duplex follow-up investigation. AJNR Am J Neuroradiol 2008;29:e65; author reply e66. Epub 2008 Jul 18 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Prabhakaran S, Warrior L, Wells KR, et al. The utility of quantitative magnetic resonance angiography in the assessment of intracranial in-stent stenosis. Stroke 2009;40:991–93 [DOI] [PubMed] [Google Scholar]
- 18. Kyriakou Y, Struffert T, Dörfler A, et al. Basic principles of flat detector computed tomography (FD-CT) [in German]. Radiologe 2009;49:811–19 [DOI] [PubMed] [Google Scholar]
- 19. Prell D, Kyriakou Y, Kalender WA. Comparison of ring artifact correction methods for flat-detector CT. Phys Med Biol 2009;54:3881–95 [DOI] [PubMed] [Google Scholar]
- 20. Berkefeld J, Zanella FE. Intracranial stenting of atherosclerotic stenoses: current status and perspectives. Klin Neuroradiol 2009;19:38–44 [DOI] [PubMed] [Google Scholar]
- 21. Schlötzer W, Huber R, Schmitz BL.. Stent-assisted intracranial angioplasty: potentials and limitations of pre- and postinterventional CT angiography [in German]. RöFo 2009;181:121–28. Epub 2009 Jan 27 [DOI] [PubMed] [Google Scholar]
- 22. Benndorf G, Strother CM, Claus B, et al. Angiographic CT in cerebrovascular stenting. AJNR Am J Neuroradiol 2005;26:1813–18 [PMC free article] [PubMed] [Google Scholar]
- 23. Struffert T, Hertel V, Kyriakou Y, et al. Imaging of cochlear implant electrode array with flat-detector CT and conventional multislice CT: comparison of image quality and radiation dose. Acta Otolaryngol 2010;130:443–52 [DOI] [PubMed] [Google Scholar]
- 24. Psychogios MN, Buhk JH, Schramm P, et al. Feasibility of angiographic CT in peri-interventional diagnostic imaging: a comparative study with multidetector CT. AJNR Am J Neuroradiol 2010;31:1226–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kalender WA, Kyriakou Y. Flat-detector computed tomography (FD-CT). Eur Radiol 2007;17:2767–79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Sadasivan C, Cesar L, Seong J, et al. An original flow diversion device for the treatment of intracranial aneurysms: evaluation in the rabbit elastase-induced model. Stroke 2009;40:952–58 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Wakhloo AK, Mandell J, Gounis MJ, et al. Stent-assisted reconstructive endovascular repair of cranial fusiform atherosclerotic and dissecting aneurysms: long-term clinical and angiographic follow-up. Stroke 2008;39:3288–96 [DOI] [PubMed] [Google Scholar]
- 28. Bley T, Strother CM, Pulfer K, et al. C-arm CT measurement of cerebral blood volume in ischemic stroke: an experimental study in canines. AJNR Am J Neuroradiol 2010;31:536–40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Van der Molen AJ, Veldkamp WJ, Geleijns J. 16-slice CT: achievable effective doses of common protocols in comparison with recent CT dose surveys. Br J Radiol 2007;80:248–55. Epub 2006 Oct 12 [DOI] [PubMed] [Google Scholar]
- 30. Bundesamt fuer Strahlenschutz, Diagnostische Referenzwerte fuer radiologische Untersuchungen. Bundesanzeiger 2003;143:17503–10 [Google Scholar]
- 31. Kyriakou Y, Richter G, Dorfler A, et al. Neuroradiologic applications with routine C-arm flat panel detector CT: evaluation of patient dose measurements. AJNR Am J Neuroradiol 2008;29:1930–36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Prell D, Kyriakou Y, Struffert T, et al. Metal artifact reduction for clipping and coiling in interventional C-arm CT. AJNR Am J Neuroradiol 2010;31:634–39. Epub 2009 Nov 26 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.