Abstract
The new coronavirus SARS‐CoV‐2 is a global pandemic and a severe public health crisis. SARS‐CoV‐2 is highly contagious and shows high mortality rates, especially in elderly and patients with pre‐existing medical conditions. At the current stage, no effective drugs are available to treat these patients. In this review, we analyse the rationale of targeting RGD‐binding integrins to potentially inhibit viral cell infection and to block TGF‐β activation, which is involved in the severity of several human pathologies, including the complications of severe COVID‐19 cases. Furthermore, we demonstrate the correlation between ACE2 and TGF‐β expression and the possible consequences for severe COVID‐19 infections. Finally, we list approved drugs or drugs in clinical trials for other diseases that also target the RGD‐binding integrins or TGF‐β. These drugs have already shown a good safety profile and, therefore, can be faster brought into a trial to treat COVID‐19 patients.
Keywords: COVID‐19, RGD‐binding integrins, TGF‐β, inflammation, ARDS, cytokines
SARS‐CoV‐2 is highly contagious and shows high mortality rates especially in elderly and patients with pre‐existing medical conditions. In this review, we explain the rationale of targeting RGD‐binding integrins to potentially inhibit viral cell infection and to block TGF‐b activation, which is involved in the severity of several human pathologies, including the complications of severe COVID‐19 cases.
Introduction
The new coronovirus SARS‐CoV‐2 has become a public health challenge worldwide, declared pandemic in March 2020, with millions of affected patients. Dr Marc Lipsitch from Harvard University cautioned that 40–70% of the human population will become infected if no actions are taken. Moreover, it is estimated that about 14% of the patients will develop serious conditions requiring hospitalisation, and approximately 1.4–3.4% will die from this infection, putting an unprecedented strain on healthcare systems. 1 , 2 The leading causes of deaths are acute respiratory distress syndrome (ARDS), septic shock, haemorrhage/coagulopathy, acute heart, liver, kidney injury and secondary bacterial infections. 2 Currently, there are no medications approved to treat this virus. A recent study demonstrated that the most promising drugs (remdesivir, hydroxychloroquine, lopinavir and interferon) appeared to have little or no effect on hospitalised COVID‐19 patients. 3 Some vaccines have finished phase III trials with a good safety profile and more than 90% efficacy (press release websites of Biontech/Pfizer and Moderna). However, as Dr Anthony Fauci and others have estimated, 70% of the world population will need to receive the vaccine to achieve herd immunity, and this will take time. Furthermore, it is not clear whether the vaccine will protect the most vulnerable people; for example, it has been reported that vaccines in elderly population have a lower efficacy to build an immune memory. Therefore, there is still an urgent need to treat these patients with known approved drugs or drugs that are further along the development pipeline.
SARS‐CoV‐2 infects cells via its spike protein, which binds the ACE2 receptor on target cells. The virus enters the cells after the proteolytic cleavage of the spike protein by the transmembrane protease TMPRSS2. Other receptors, for example CD147 and CD26 (DPP4), have also been proposed to be a potential entry point of the virus. 4 , 5 However, no direct binding activity of CD147 to spike has been reported. 6 For CD26, which is the receptor of MERS (Middle East respiratory syndrome) to infect cells, a model of docking analysis predicted a binding of CD26 to the spike protein, but experimental data still need to confirm this bioinformatic approach. 5 We reviewed a novel mutation (K403R) in the spike protein that does not exist in other strains of the coronavirus (Figure 1). 7 , 8 This mutation creates an RGD motif, which could be recognised by integrins. Integrins are cell adhesion receptors that play important roles during pathological processes. Eight out of 24 known integrins recognise the RGD sequence in the natural ligands. 9 The RGD is also a common motif for other types of viruses to infect cells (e.g. Epstein–Barr virus, rotavirus, human cytomegalovirus, Ebola). 10 Therefore, the new RGD motif in SARS‐CoV‐2 could increase the binding potency of ACE2‐positive target cells as well as infecting ACE2‐negative cells. This could also explain why the virus spreads faster and more aggressively than SARS‐CoV‐1, which belongs to the same family of coronaviruses. However, further studies are required to evaluate this hypothesis. SARS‐CoV‐1 was declared an epidemic in 2003 and showed higher fatality rates, but lower infection rates than SARS‐CoV‐2.
Figure 1.
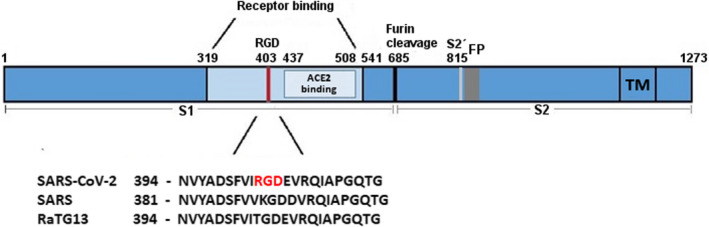
Schematic representation of SARS‐CoV‐2 S‐protein with a focus on the receptor‐binding domain. The sequences of the spike protein of human SARS‐CoV‐2, human SARS‐CoV‐1 (75% similarity) and bat RaTG13 (96% similarity) were aligned using MAFFT. 176 The receptor‐binding domain and the ACE2 receptor‐binding region are coloured in light blue. The RGD motif of SARS‐CoV‐2 is highlighted in red. Numbers refer to the SARS‐CoV‐2 spike protein sequence. FP, fusion peptide; RGD, amino acids arginine‐glycine‐aspartic acid; S1, subunit of spike; S2, subunit of spike (fusion domain); S2´, cleavage site; TM, transmembrane domain.
RGD‐binding integrins are the main regulator for the activation of transforming growth factor beta (TGF‐β). 11 TGF‐β plays an important role in many of the observed complications of severe COVID‐19 patients (discussed below). Furthermore, TGF‐β is increased in patients with pre‐existing medical conditions (discussed below). In this review, we are discussing the rationale of using integrin inhibitors as potential treatment of COVID‐19 patients.
RGD‐binding integrins for SARS‐CoV‐2 cell infection
Several viruses are known to use a RGD motif to bind to the surface of cells, which is crucial for a successful infection. 10 For example, the West Nile virus uses the integrins αvβ1 and αvβ3 for cell entry, 12 , 13 Ebola uses the integrin α5β1, 14 and the Herpes simplex virus type 1 (HSV‐1) interacts with αvβ3. 15 A more detailed list of viruses using the RGD motif for cell entry is reviewed in Hussein et al. 10 Therefore, the RGD motif in the Spike protein of SARS‐CoV‐2 (Figure 1) could be critical in infecting cells through the RGD‐binding integrins. Others have already suggested that the RGD motif of the Spike protein may enhance infection efficiency. 7 , 16 , 17 A recent study further supports the hypothesis that SARS‐CoV‐2 can infect target cells via RGD‐binding integrins. 18 The group used different culture conditions to induce ACE2 expression. They observed that ACE2 was virtually absent in human small intestinal organoids (hSIOs) cells grown in expansion medium and increases dramatically when hSIOs cells were grown in differentiation medium with or without BMP (bone morphogenetic protein) stimulation; nevertheless, both showed similar infection rates. The group suggests that a low level of ACE2 might be sufficient for viral entry. However, the ACE2+ and TMPRSS2+ club cells, which are found in the airway epithelium and are the progenitor cells of the trachea and the bronchiolar region, were neither infected in vitro nor in vivo with Sars‐CoV‐2. 19 Additionally, cell lines that are used for analysing SARS‐CoV‐2 entry via ACE2, for example the monkey kidney epithelial cell line VERO E6, are also expressing RGD‐binding integrins. For VERO E6, α5β 1, αvβ 1 and αvβ 3 have been detected. 20 A recent study showed that the spike protein of SARS‐CoV‐2 binds to both α5β1 and α5β1/hACE2 on VERO E6 cells. 21 Adding the α5β 1 integrin inhibitor ATN‐161, which is in a phase II clinical trial for renal cancer, disrupts SARS‐CoV‐2 infection. Furthermore, pretreating VERO E6 cells with ATN‐161 before adding SARS‐CoV‐2 increased cell viability and decreased cytopathic effects associated with viral infection. A non‐peer review published result by the group of Dr Jenkins (University of Nottingham, UK) showed in a solid‐phase binding assay that the spike S1 subunit potentially also binds αvβ 1, αvβ 5, αvβ 6 and αvβ 8. 22 The binding appears to be 102 times lower than the binding to ACE2. However, RGD specificity still needs to be confirmed and whether this potentially weaker interaction makes the integrins a less promising target needs further evaluation. Interestingly, it has been shown that SARS‐CoV‐2 reduces the expression of ACE2. 23 That seems counterproductive since ACE2 appears to be the main receptor that the virus uses to infect cells. It is known that different viruses use multiple receptors to infect cells. Recently, it has been shown that neuropilin‐1 (NRP‐1) could be such a factor that increases the binding of the spike protein to ACE2 and blocking NRP‐1 reduced viral infection. 24 RGD‐binding integrins could play a similar role, as it has been shown that RGD‐binding integrins α5β1, αvβ5, αvβ6 and αvβ8 are expressed on human airway epithelial cells. 25 In that case, a low expression of ACE2 would still be sufficient for the virus to infect the cells. In conclusion, the acquired RGD motif in the spike protein could potentially promote viral infection in ACE2‐negative cells as well as serving as an additional factor to increase binding and infectivity in ACE2‐positive cells. 18
Involvement of TGF‐β in different pathogeneses
Activation of TGF‐β
RGD‐binding integrins are the main regulator of TGF‐β activation. TGF‐β plays an important role in several biological processes including embryogenesis, tissue regeneration, immune responses and tumorigenesis. Additionally, TGF‐β can also act as a pro‐viral factor. 26 Upregulation of TGF‐β is also involved in mediating different pulmonary diseases, for example bronchial asthma, emphysema, pulmonary fibrosis and lung cancer. 27 Three isoforms of TGF‐β are known (TGF‐β 1, β2 and β3). All three are highly conserved between species, and they have demonstrated similarity in functional properties. However, specific and non‐overlapping functions have been suggested for each isoform. 11 TGF‐β is sequestered as an inactive protein, forming non‐covalently small‐latent complex (SLC) with the latency‐associated peptide (LAP). This complex is bound covalently by disulfide bonds to the latent TGF‐β‐binding protein (LTBP) to form the large latent complex (LLC), which is stored in the extracellular matrix. 11 TGF‐β can be activated through physical processes like acidification, extreme temperature changes and oxidation, by several proteases, for example plasmin, elastase, matrix metalloproteinase (MMP)‐2 and MMP‐9, and by interactions with integrins or thrombospondin. 11 The LAP of TGF‐β1 and TGF‐β3 contain a RGD motif that can bind to at least six αv‐containing integrins (αvβ 1, αvβ 3, αv β5, αv β6, αv β8 and α8 β1). So far, four of them have been shown to activate TGF‐β in vivo (αvb1, αvb5, αvb6 and αvb8). 28 , 29 For the latent TGF‐β2, an alternative mechanism must be responsible for the activation since the LAP of TGF‐β2 does not contain an RGD motif.
As previously mentioned, infection with SARS‐CoV‐2 reduces the expression of ACE2. 23 That seems counterproductive since ACE2 appears to be the main receptor that the virus uses to infect cells. Nevertheless, a reduced expression of ACE2 in COVID‐19 patients could be the potential source for TGF‐β. ACE2 usually catalysed Angiotensin II (AngII) to Ang1‐7. Ang1‐7 is a biological active peptide that binds and activates the Mas receptor. This ACE2/Ang1‐7/Mas pathway counterbalances RAAS (renin, angiotensin, aldosterone system), promoting the activation of anti‐inflammatory pathways. 30 Reduced ACE2 leads to an overactive RAAS, which provoked a local vascular inflammation and, through aldosterone, activates TGF‐β production. 31
Another potential source of TGF‐β is different immune cells. Boumaza et al. showed in vitro that SARS‐CoV‐2 efficiently infects monocytes and macrophages, which resulted in the secretion of TGF‐β. 32 There are no reports though that show that this occurs in COVID‐19 patients. In an additional study, TGF‐β1 was increased in two different subsets of CD4+ immune cells of the COVID‐19 patient group compared to the healthy control group. 33 However, the activation of TGF‐β has not been studied during the complete infectious process. Therefore, it is not known whether TGF‐β activation occurs at early or late infection.
ARDS and fibrosis
TGF‐β regulates multiple cellular processes that also play an important role in the development of acute lung injury (ALI)/ARDS; for example, TGF‐β contributes to the alveolar epithelial permeability, fibroblast activation and extracellular matrix remodelling. Increased levels of TGF‐β are associated with impaired alveolar fluid clearance incapable to remove the oedema from alveoli. 34 , 35 , 36 , 37 The overall mortality rate of ARDS is between 30 and 40%. 38 A similar rate has been observed in COVID‐19 patients who develop ARDS. Furthermore, patients with severe conditions also need mechanical ventilation. Different small case series demonstrated that COVID‐19 patients who received mechanical ventilation had a mortality rate between 50 and 81.1%. 39 , 40 The current medical recommendation for these patients is to delay the mechanical ventilation if possible. Interestingly, ARDS patients with a lower TGF‐β level in the bronchoalveolar lavage fluid (BALF) were associated with fewer mechanical ventilation and less intensive care unit (ICU) days. 41 However, the difference was not statistically significant and further research is necessary to evaluate this trend. The activation of TGF‐β increases the endothelial and epithelial permeability leading to the alveolar influx of fluids and proteins that impairs pulmonary gas exchange, which leads to arterial hypoxemia and respiratory failure. 42 , 43 TGF‐β1 is a central mediator of fibrogenesis. As discussed above, SARS‐CoV‐2 infection causes an imbalance in RAAS. A recent review suggested that this imbalance favors lung fibrosis in COVID‐19 patients. 44 The authors also hypothesised that TGF‐β might play a critical role in this process. The release of TGF‐β from injured tissue promotes lung repair, which normally leads to the resolution of infection. However, mice or rats over‐expressing TGF‐β demonstrate severe pulmonary fibrosis. 45 , 46 Reports of patients with influenza A (H1N1) showed elevated levels of TGF‐β1, correlated with the development of pulmonary fibrosis. 47 TGF‐β levels were also markedly increased in SARS patients with ARDS. 48 Zhao et al. showed that the nucleocapsid protein of SARS‐CoV‐1 is responsible for elevated TGF‐β signalling in these patients. 49 The nucleocapsid protein of SARS‐CoV‐2 is over 90% similar to that of SARS‐CoV‐1. 50 Whether it also plays a role for TGF‐β signalling in COVID‐19 patients is unknown. Nevertheless, the activation of TGF‐β leads to the production of fibrin, collagen and matrix metalloproteinases (MMP), which play a critical role in ALI. 51 , 52 , 53 TGF‐β has been also shown to regenerate hyaline cartilage, which contains a large amount of collagen as well as hyaluronan (HA), for the latter through increased expression of hyaluronan synthase 2. 54 A pathological report of a case study of a COVID‐19 patient demonstrated the formation of hyaline membrane—a suggestive sign of early ARDS. 55 HA can absorb a high amount of water, which could explain the accumulation of fluids in the lungs of COVID‐19 patients. 56 However, the association between HA and fluids agglomeration still needs to be confirmed. If so, blocking TGF‐β to inhibit hyaluronan synthase might hold a great promise for COVID‐19 patients.
In response to lung injury as well as inflammation, the surface expression of the epithelial integrins α2β1, α3β1, α5β1, α6β4, αvβ5 and αvβ6 is dramatically upregulated. The upregulation of the integrins α5β1 and αvβ6 is specific to alveolar epithelial cells following injury. 57 Furthermore, activated neutrophils contribute to the development of ventilator‐induced lung injury (VILI) caused by high‐pressure mechanical ventilation and increased TGF‐β expression. In a rat model, ventilator‐induced lung injury is associated with neutrophil infiltration, macrophage activation and upregulated TGF‐β expression. 58 Several in vivo models have shown that blocking TGF‐β via a soluble chimeric TGF‐β receptor or via the corresponding integrins (αv β5, αv β6 and αv β8) can reduce lung injury, 43 , 59 , 60 whereas in the bleomycin model, an overexpression of TGF‐β led to increased apoptosis of airway epithelial cells and increased lung fibrosis. 61 Furthermore, αvβ6−/− mice are protected from LPS and ventilator‐associated lung injury 62 and the pretreatment with antibodies to block αv β5 and αv β6 had additive protective effects against interleukin (IL)‐1β‐induced ALI. 63
Angiogenesis and coagulation
It has been suggested that SARS‐CoV‐2 is not only a respiratory infection but also a haematologic disease because of its significant impact on the haematopoietic system. 64 For instance, the findings of blood clots in COVID‐19 patients and their involvement from deep venous thrombosis in lower extremities to blocked arteries in the brain and lungs, resulting in strokes and pulmonary embolism, are of great concern. 65 , 66 The source of these blood clots has not been defined; however, TGF‐β is known to stimulate the production of Factor XII (FXII). 67 The zymogen FXII stands at the beginning of the coagulation signalling of the intrinsic cascade. 68 Thrombin, which is the last step of the coagulation cascade, mediates the cleavage of fibrinogen to fibrin. The fibrin monomers polymerise and form a fibrin clot that stop the bleeding. FXII knockout mice are largely defective for thrombus formation and are protected from experimental cerebral ischaemia and pulmonary embolism. 69 FXII, thrombin and fibrinogen have also been linked to inflammatory disorders like multiple sclerosis, sepsis, rheumatoid arthritis and colitis. 70 Thrombin cleaves GARP (glycoprotein A repetitions predominant) that is found on the surface of platelets, and the cleaved GARP in cooperation with αvβ8 leads to the release of mature TGF‐β1, creating a positive feedback loop. Blocking αvβ8 on dendritic cells and fibroblasts impairs TGF‐β‐dependent generation of Th17 cells and reduces the inflammatory disease in the lungs. 71 It has been postulated that FXII potentially mediates the development of neuroinflammation via upregulation of neutrophil functions, contributing to macrophage polarisation, and inducing T‐cell differentiation. 72 FXII signalling is also involved in activating pro‐inflammatory pathways that induce chemotaxis of leukocytes and increases vascular permeability. 70
The expression of plasminogen activator inhibitor‐1 (PAI‐1), another important factor in the coagulation process, is induced by TGF‐β. 73 Elevated PAI‐1 has been observed in myocardial infarction and cardiac failure, and the latter is one of the death causes for COVID‐19 patients. 1 , 2 , 74 , 75 Furthermore, Ackermann et al. showed that the expression of the gene for PAI‐1, SERPINE1, is upregulated in COVID‐19 patients. 76 PAI‐1 is a serine‐protease inhibitor and the main inhibitor of the tissue‐type (tPA) and the urinary‐type plasminogen activator (uPA). Both are able to activate plasminogen and, hence, fibrinolysis. Besides the elevated levels of fibrin in COVID‐19 patients and their resulting blood clots, increased levels of the fibrin‐degradation product D‐dimer have been detected. 77 A reduced fibrinolysis can cause thrombosis, pulmonary embolus, and it increases the risk of stroke and heart attack. A case series was performed to treat blood clots with tPA in COVID‐19 patients. However, this treatment only showed temporary improvements. 78
Soluble urokinase‐type plasminogen activator receptor (suPAR) is another factor that is elevated in COVID‐19 patients and may serve as an early predictor of severe respiratory failure in patients with COVID‐19 pneumonia. 79 Elevated suPAR is associated with increased inflammation, disease progression and risk of mortality in several infectious diseases. 80 It is also associated with acute kidney injury in various clinical and experimental contexts. 81 Chang et al. showed that TGF‐β stimulates PAI‐1 and suPAR secretion in a system of repair/regeneration activities of stem cells from apical papilla (SCAP). 82
Besides a widespread thrombosis in patients with COVID‐19, the examination of the lung during an autopsy of 7 deceased COVID‐19 patients also demonstrated increased angiogenesis. 76 Recent evidence has revealed higher vascular endothelial growth factor (VEGF) levels in COVID‐19 patients than in healthy controls. VEGF plays a major role in the formation of new blood vessels, and TGF‐β has been shown to induce angiogenesis through VEGF‐mediated apoptosis. 83 Furthermore, several studies have shown VEGF’s potential key role in the pathogenesis of ALI/ARDS. 84 Additionally, hypoxia‐inducible factor 1 alpha (HIF‐1α) that is also increased in COVID‐19 patients 85 regulates VEGF and, in turn, TGF‐β induces HIF‐1α stabilisation. 86 Blocking RGD‐binding integrins reduces VEGF expression. 87 A clinical trial is evaluating the capacity of anti‐VEGF antibody bevacizumab to inhibit ALI/ARDS as well as to reduce the mortality in severe COVID‐19 patients through the suppression of pulmonary oedema (NCT04275414). In conclusion, blocking TGF‐β can potentially inhibit FXII, PAI‐1, suPAR and VEGF. In conclusion, blocking TGF‐β can potentially inhibit FXII, PAI‐1, suPAR and VEGF, which might prevent and resolve blood clotting and reduce inflammation and vascular permeability.
Acute kidney injury, cytokines, Th17 cells and complement
In addition to the above‐mentioned common complications of severe COVID‐19 cases, other common problems are as follows: pneumonia, sepsis and septic shock, cardiomyopathy and arrhythmia, acute kidney injury (AKI), and several additional complications derived from prolonged hospitalisation, including secondary bacterial infections, gastrointestinal bleeding and critical illness polyneuropathy/myopathy. 2 Special interest of this list of medical complications falls to AKI. Recent work demonstrated that TGF‐β/SMAD2 (homologues of the Drosophila protein, Mothers against decapentaplegic (Mad) and the C. elegans protein Sma) plays a pivotal role in AKI. 88 Liu et al. showed in a mouse model of obstructive nephropathy that the loss of ACE2 enhances renal fibrosis, which is mediated by increased TGF‐β/SMAD signalling. 89 Reduced expression of ACE2 has also been described for SARS‐CoV‐2 infections (see sections above). Furthermore, Breuss et al. observed beta 6 integrin expression in adult lungs and kidneys at focal sites of subclinical inflammation, as well as in a variety of clinical specimens from patients with chronic or acute inflammation of the lungs or kidneys. 90 Blocking the integrin αvβ5 protected rats from AKI. 91 Therefore, TGF‐β might play a pivotal role in the development of AKI in COVID‐19 patients. Moreover, blocking RGD‐binding integrins could prevent AKI.
Patients with severe COVID‐19 infections may present a high level of cytokines (cytokine storm), especially interferon gamma (IFN‐y), tumor necrosis factor alpha (TNF‐a), IL‐17, IL‐8, IL‐1β and IL‐6 are increased. 92 The largest studies so far that analysed 11, 27, 48 and 76 cytokines, respectively, did not measure protein expression of TGF‐β in the serum or BALF. 93 , 94 , 95 , 96 However, in a small study of three patients, mRNA expression of TGF‐β2 was elevated in BALF samples compared to healthy controls. 97 TGF‐β1 was also increased in two different subsets of CD4+ immune cells of the COVID‐19 patient group compared to healthy control. 33 A new study also showed that TGF‐β is increased in the serum of COVID‐19 patients compared to healthy controls. 98 In the case of ALI induced by IL‐1β, pretreating mice with antibodies to block αvβ5 and αvβ6, which inhibits TGF‐β activation, can prevent ALI development, 63 . Type I interferons (IFN) are key cytokines to challenge viral infections. However, in severe COVID‐19 patients IFNs expression are impaired. 99 , 100 In cancer, IFNs can be potent anti‐tumoral agents. Yet, abundant TGF‐β in tumors can limit this IFN‐induced tumor regression. 101 This has also been reported in respiratory viral infections like rhinovirus infections 102 , 103 and respiratory syncytial virus (RSV) infections. 104 , 105 , 106 Another interesting cytokine is IL‐17 because of its broad pro‐inflammatory effect on the induction of cytokines. Mouse experiments with Neisseria gonorrhoeae indicate that IL‐17 suppresses Th1/Th2 immune responses through TGF‐β. Treating mice with anti‐TGF‐antibodies resulted in increased Th1 and Th2 responses and diminished Th17 response, following by an accelerated clearance of N. gonorrhoeae. 107 The main producer of IL‐17 is Th17 cells. 108 Pathologic findings showed an increased concentration of pro‐inflammatory Th17 cells in COVID‐19 patients. 55 , 109 In mice, Th17 cells are induced through TGF‐β and IL‐6, 110 although in humans, it is less clear. It seems that IL‐6 and IL‐1β are the main inducers of human Th17 cells. However, other groups also demonstrated the importance of TGF‐β–inducing Th17 cells. 111 , 112
The complement activation is a key player in the fight against pathogens. However, excessive or unregulated complement activation might be involved in the pathogenesis of ALI and ARDS in COVID‐19 patients. 113 Gu et al. showed that a crosstalk between TGF‐β1 and complement activation augments epithelial injury in pulmonary fibrosis. 114 They suggest that increased TGF‐β levels may crosstalk with the complement cleavage products C3a and C5a and downregulates complement‐inhibitory proteins, which links complement activation to epithelial injury in IPF. Furthermore, C5a triggers the formation of neutrophil extracellular traps (NETs) that are capable of activating platelets to release TGF‐β. NETs are released by neutrophils to halt an infection. They are extracellular webs containing chromatin, microbicidal proteins and oxidant enzymes. However, when NETs are not properly regulated, they have the potential to propagate inflammation and microvascular thrombosis. Recent studies have shown elevated NET release in the serum of COVID‐19 patients and provided clinical evidence of its fundamental role in the pathogenesis of COVID‐19‐related ALI/ARDS and coagulopathy. 115 , 116 Therefore, blocking TGF‐β could be beneficial to prevent complement‐induced ALI as well as coagulopathy.
Kawasaki‐like syndrome in children
Until recently, children were thought to have less severe SARS‐CoV‐2 infections. However, new reports showed that there is an increase of Kawasaki‐like syndrome in infected children. 117 The Kawasaki disease (KD) is an acute and usually self‐limiting vasculitis, which usually affects children < 5 years of age. 118 It has been reported that genetic variants in the TGF‐β pathway genes influence the susceptibility to KD and that TGF‐β may contribute to aneurysm formation. 119 Targeting TGF‐β could therefore be beneficial in preventing KD. However, caution must be taken, because a mouse model of KD using a neutralising pan‐TGF‐β antibody worsened inflammatory‐induced coronary artery lesions. 120 It is possible that using an antibody was not an appropriate strategy to block TGF‐β because it can induce inflammation via Fc receptors (FcR), which are expressed in different types of innate immune cells. Blocking TGF‐β via RGD‐binding integrins or using a specific drug to block particularly one TGF‐β isoform may show a better outcome.
TGF‐β and the potential reason why patients with pre‐existing conditions have a higher risk of developing severe COVID‐19 symptoms
Patients with pre‐existing medical conditions have an increased risk of developing severe COVID‐19 infections. 40 , 121 , 122 The specific reasons are currently unknown. We hypothesise that TGF‐β could be the potential link. TGF‐β plays an important role in the progress of chronic conditions such as diabetes, 123 hypertension, 124 kidney injuries, 125 heart disease, 126 lung diseases (asthma, 127 chronic obstructive pulmonary disease (COPD) 128 and fibrosis 129 ), obese patients 130 and immunocompromised patients (cancer, 131 transplantation 132 , 133 ) as well as in complications associated with elderly patients. 134 , 135 Blocking TGF‐β in animal models was associated with an improved outcome in obesity, 136 diabetes, 136 kidney injuries 91 , 137 and hypertension. 138 , 139 In the case of lung diseases, it has been shown that elevated TGF‐β plays an important role in an exaggerated inflammatory response as well as disease exacerbation. 102 , 104 , 105 , 140 Furthermore, the infection with SARS‐CoV‐2 between men and women seems to be equivalent. Nevertheless, men developed more severe symptoms with higher mortality rates. A new study analysing ACE2 concentrations in the plasma of non‐infected men and women with heart failure demonstrated a higher expression of soluble ACE2 in men, suggesting that the higher soluble ACE2 expression is responsible for the severe symptoms. 141 On the contrary, soluble recombinant human ACE2 is suggested to prevent binding of viral particles to the cell surface of ACE2‐positive cells. Recent in vitro studies have shown that an ACE2 Fc‐fusion protein neutralises SARS‐CoV‐2 and prevents viral cell infection. 142 Another study analysing GTEx and other public data in 30 tissues across thousands of individuals demonstrated that Asian women have a higher ACE2 cell surface expression than Asian men. 143 This study also found an age‐dependent ACE2 decrease as well as a significant ACE2 decrease in type II diabetic patients. Furthermore, they found that ACE2 expression is upregulated by oestrogen. In conclusion, their data suggest that ACE2 might play a protective role in developing severe conditions for COVID‐19 patients. Additionally, the study showed that ACE2 expression is negatively correlated with severe outcome in patients with pre‐existing conditions. The same relationship is observed following age and gender of the patients. Another possible reason for less severe cases observed in women could be that women have a potential more efficient immune system than men. 144 However, we hypothesise that TGF‐β might be the important factor for the difference. Loss of ACE2 expression increases TGF‐β expression in a mouse model, and men show a higher expression of TGF‐β at baseline than women. 89 , 145 Furthermore, TGF‐β induces the expression of αv, α5, β1, β3 and β5 integrins, 146 , 147 creating a positive feedback loop on TGF‐β activation and potentially increases the hypothesised infectivity via RGD‐binding integrins. This hypothesis is further supported by the findings that in severe patients cases, vital tissues with little ACE2 expression are also severely damaged by the SARS‐CoV‐2 infection. 148 Additionally, the infection with SARS‐CoV‐2 reduces ACE2 expression, which, therefore, can increase TGF‐β expression. The reason why women show less severe COVID‐19 infections might be that the female oestrogen suppresses TGF‐β‐induced gene expression, for example type IV collagen. 149 Collagens play an important role in the development of fibrosis. 150 In a mouse kidney injury model, it was also shown that oestradiol can reverse renal injury. 151 A clinical trial just started to use oestrogen as treatment for COVID‐19 patients to evaluate whether oestrogen can reduce the damaging effects of the virus on the lung and other severe symptoms (NCT04359329).
In conclusion, based on the aforementioned evidence, we hypothesise that blocking TGF‐β might be a possible treatment opportunity for COVID‐19 patients with pre‐existing medical conditions to reduce the risk of developing a more severe disease that could potentially lead to death (Figure 2).
Figure 2.
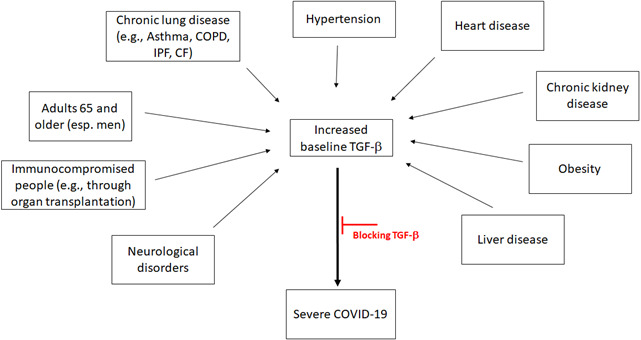
Multiple conditions lead to higher levels TGF‐β. Representation of medical conditions that are associated to high levels of TGF‐β. These can be related to the severe cases of COVID‐19. Specific inhibition of TGF‐β mechanism of action could prevent severe COVID‐19 symptoms. COPD, chronic obstructive pulmonary disease; IPF, idiopathic pulmonary fibrosis; CF, cystic fibrosis.
RGD‐binding integrins
RGD‐binding integrins are heterodimeric proteins composed of two membrane‐spanning subunits. They are part of the superfamily of cell adhesion receptors that recognise their ligand via the RGD motif. Below, we summarise each of the 8 known RGD‐binding integrins on their potential role for SARS‐CoV‐2.
αvβ1
The integrin αvβ1 is highly expressed on activated fibroblasts in the lung and directly binds to the latency‐associated peptide of TGFβ1 to mediate TGFβ1 activation. αv and β1 integrins can also be induced by TGF‐β, creating a positive feedback loop. 146 A therapeutic delivery of an αvβ1 inhibitor has been shown to attenuate bleomycin‐induced pulmonary fibrosis. 29 Furthermore, inhibiting this integrin in a kidney injury‐induced mouse model ameliorates renal failure and fibrosis. 152 These results make αvβ1 a potential target in the treatment for severe COVID‐19 infections where these complications have also been observed.
αvβ3
αvβ3 plays a fundamental role in neovascularisation. This integrin is elevated on endothelial cells during wound angiogenesis, tumor angiogenesis and inflammation. 153 , 154 αvβ3 is also upregulated on epithelial cells and can activate TGF‐β in scleroderma fibroblasts, even though it binds weaker to latent TGF‐β than to αvβ6 and αvβ8. 155 , 156 Furthermore, TGF‐β increases the expression of αv and β3 integrins. 146 Of note, excessive blood clot formation was also demonstrated in COVID‐19 patients. 65 Furthermore, Ackermann et al. demonstrated increased angiogenesis in the lung of deceased COVID‐19 patients. 76 There is evidence that some COVID‐19 patients show an increased level of VEGF compared to healthy controls. For this reason, a clinical trial using bevacizumab, an anti‐VEGF‐A antibody, was initiated for COVID‐19 patients (NCT04275414 and NCT04305106). As mentioned above, a small case series was started to treat the blood clots with tPA. 78
αvβ5
αvβ5 plays a role in several biologic processes, for example tumor angiogenesis, phagocytosis, fatty acid uptake and retinal pigment epithelium homeostasis. 157 , 158 , 159 αvβ5 is also involved in vascular permeability, and blocking αvβ5 inhibits vascular leakage in mouse models of ALI, sepsis and AKI. 63 , 91 , 160 Similar to αvβ3, αvβ5 is also upregulated in the dermal epithelium of patients with systemic sclerosis and activates TGF‐β in scleroderma fibroblasts. 155 Both subunits of αvβ5 can be upregulated through TGF‐β. 146
αvβ6
The integrin αvβ6 is highly expressed at high levels during embryogenesis in epithelial cells of the developing lung, but downregulated in healthy adults. 90 , 161 However, during epithelial injuries, αvβ6 is highly upregulated. 90 , 162 As mentioned earlier, blocking avb6 interleukin can reduce lung injury and IL‐1β‐induced ALI. 43 , 59 , 60 , 63
αvβ8
αvβ8 is expressed in the lung by epithelial cells and fibroblasts, and its expression is increased in airway fibroblasts of COPD patients. 163 Compared to the other RGD‐binding integrins that activate TGF‐β through a conformational change in the LAP protein, αvβ8 activates TGF‐β through a proteolytic cleavage of LAP. 156 Asthmatic children demonstrate a higher expression of epithelial αvβ8 than control children 164 Kitamura et al. demonstrated that αvβ8‐mediated TGF‐β activation is important for the development of airway fibrosis and inflammation and that blocking αvβ8 represents a strategy to treat fibroinflammatory airway diseases. 128 , 165
αIIbβ3
αIIbβ3 is the major integrin expressed on the surface of platelets where it plays a critical role in platelet aggregation and blood clotting. 166 αIIbβ3 plays also a role in stroke and myocardial ischaemia. 167 , 168 Several drugs are approved to prevent platelet aggregation and thrombus formation (Table 1), incidences that are also reported in COVID‐19 patients. There are no reports showing that αIIbβ3 binds or activates latent TGF‐β.
Table 1.
Drugs against RGD‐binding integrins and against TGF‐β
| Drug | Target | Disease | Clinical Stage | Company |
|---|---|---|---|---|
| A: Chemical or peptide/protein based drugs | ||||
| Abciximab ( a ) | α IIbβ3 ( b ) | Thrombosis | approved | Janssen Biologics |
| Tirofiban | α IIbβ3 | Thrombosis | approved | Medicure Pharma |
| Intrifiban | α IIbβ3 | Thrombosis | approved | Millennium Pharmaceuticals |
| GSK3008348 | α vβ6 | idiopathic pulmonary fibrosis (IPF) | Phase I | GSK |
| PLN‐74809 | α vβ1 and α vβ6 | IPF | Phase IIa | Pliant |
| THR‐687 | pan RGD integrin inhibitor | Diabetic macular edema | Phase II planned | Oxurion NV |
| GLPG0187 | α vβ1, α vβ3, α vβ5, α vβ6, and α 5β1 | Solid tumors | Phase I | Galapagos NV |
| IDL2965 | α vβ1, α vβ3, and α vβ6 | IPF | Phase IIa | Indalo Therapeutics |
| Risuteganib | α 5β1, α vβ1, α vβ3, and α vβ5 | Dry age‐related macular degeneration, diabetic macular edema | Phase IIa | Allegro |
| MK‐0429 | αvβ1, but has also been shown to block α 5β1, α vβ3, α vβ5, α vβ6, α vβ8, α 5β1139 | Prostate cancer, Post‐Menopausal Osteoporosis | Phase I, II | Merck Sharp & Dohme |
| Cilengitide | α 5β1, α vβ3 and α vβ5 | Solid tumors | Phase II/III | Merck KGaA |
| ATN161 | α 5β1 | Renal cancer, glioma | Phase II | Attenuon |
| B: Antibodies against RGD‐binding integrins | ||||
| Intetumumab | α vβ1, α vβ3, α vβ5, and α vβ6 | Prostate cancer, melanoma | Phase II | Centocor, Inc. |
| BG00011 (STX‐100) | α vβ6 | IPF | Phase II | Biogen |
| Abituzumab (EMD525797) | α vβ1, α vβ3, α vβ5, α vβ6, and α vβ8 | Prostate cancer, colorectal cancer | Phase II | Merck KGaA |
| C: TGF‐β inhibitors | ||||
| OT‐101 (Trabedersen) | TGF‐β2 | Solid cancer/ COVID‐19 | Phase III/IND filed for phase II study | Mateon Therapeutics |
| Galunisertib | TGF‐beta receptor type‐1 (TGF‐βR1) | Myelodysplastic syndrome and solid tumors | Phase II/III | Eli Lilly & Company (Lilly) |
| TEW‐7197 | TGF‐βR1 | Solid tumors | Phase I | MedPacto Inc. |
| LY3022859 | TGF‐βR2 | Advanced solid tumors | Phase I | Lilly |
| LY2157299 | TGF‐βR2 | Hepatocellular carcinoma | Phase II | Lilly |
| LY2382770 | TGF‐β1 | Diabetic kidney disease, diabetic nephropathy, diabetic glomerulosclerosis | Phase II | Lilly |
| Fresolimumab (GC‐1008) | Pan TGF‐β | Systemic sclerosis, focal segmental glomerulosclerosis, myelofibrosis, and solid tumors | Phase II | Genzyme |
| Luspartecept | TGF‐β superfamily inhibitor | β‐thalassaemia; anaemia in patients with MDS | Phase II | Acceleron Pharma |
| NIS793 | Pan TGF‐β | Solid tumors | Phase I | Novartis |
| LY2382770 | TGF‐β1 | Diabetic kidney disease (fibrosis) | Phase II | Lilly & |
| CAT‐192 | TGF‐β1 | Systemic sclerosis scleroderma | Phase I/II | Genzyme |
| AVID200 | TGF‐β1 & ‐β3 | Scleroderma, myelofibrosis, solid tumors | Phase I | Icahn School of Medicine at Mount Sinai |
IND, Investigational New Drug.
Abciximab is a Fab fragment. We listed it in this table because it is not a full length antibody.
We only show approved drugs against αIIbβ3. There are more inhibitors in different stages of clinical trials.
α5β1
α5β1 is expressed in the foetal lung mesenchyme, but not lung epithelium. Several studies with α5β1 antagonists suggest that it plays a critical role in wound repair, inflammation and tumor angiogenesis. 169 , 170 Furthermore, TGF‐β treatment can increase the expression of both subunits. 146 , 147 A recent publication showed that the gene of α5β1 is upregulated in COVID‐19 patients. 76 Of particular interest concerning COVID‐19 is the study showing that blocking α5β1 can also reduce severe airway hyperresponsiveness in a mouse model of asthma. 171
α8β1
The α8β1 integrin is expressed on human intestinal epithelial crypt cells. 172 It is also present on alveolar interstitial cells and smooth muscle cells in the lung parenchyma and is upregulated during pulmonary and hepatic fibrosis. 173 Furthermore, T regulatory cells express high amounts of the α8β1 integrin, which enables them to activate latent TGF‐β. 174
Potential drugs to block TGF‐β directly or via RGD‐binding integrins
A variety of preclinical studies have demonstrated that blocking integrins can, for example, prevent pulmonary fibrosis and protect against ALI. There are several medications for different diseases available that target different RGD‐binding integrins and that have been approved or are in clinical trials (Table 1a). From this group of potential drugs, GSK3008348 is particularly interesting. It is the first inhaled inhibitor of the integrin αvβ6. A phase I trial for idiopathic pulmonary fibrosis (IPF) has been shown to be safe. Another interesting drug is the α5β1 inhibitor ATN161, which has shown in vitro that it can inhibit SARS‐CoV‐2 infections (see section above). Our literature review supports the idea that full‐length antibodies’ treatment should be taken carefully because of their potential to further stimulate inflammation through binding to FcRs. However, due to the urgency to find a treatment, they are also listed here (Table 1b).
Targeting TGF‐β systemically bears some risks as explained below. However, considering the severity of some COVID‐19 cases these drugs could be beneficial for this group of patients. An Investigational New Drug (IND) has been filed for a phase II clinical trial using a TGF‐β2 antisense drug (OT‐101) to treat COVID‐19 patients. The rationale for this trial was based on the significant negative correlation between TGF‐β levels in BALF samples from ARDS patients and ventilator‐free days and ICU‐free days and that lower TGF‐β levels correlated with better survival outcome in ARDS patients. 175 Here, we present several TGF‐β inhibitors that are in clinical trial for different diseases, for example receptor kinase inhibitors, neutralising antibodies and ligand traps (Table 1c).
Potential risks
Blocking TGF‐β has a great potential to treat COVID‐19 patients. However, a systemic anti‐TGF‐β treatment could increase severe adverse events because of its involvement in several biological processes. A partial block might be preferred. This can be accomplished through blocking RGD‐binding integrins. This would resemble the body’s natural mechanism of locally TGF‐β activation, and it has, therefore, the potential to return to a homeostatic state without the systemic side effects that have been seen with a systemic blockage of activated TGF‐β.
As mentioned above, anti‐integrin antibodies are not the first choice because latent TGF‐β can be also stored at the surface of cells. Therefore, an anti‐TGF‐β antibody could activate the complement system against the targeted cell as well as to stimulate further inflammation through the interaction with the FcR‐expressing immune cells. The option of using novel antibody formats, such as Fc‐less or Fc‐silent antibodies, is promising, and it deserves to be explored. Systemic blockade of TGF‐β through, for example a pan integrin inhibitor, also could increase the risk of adverse events similar to a systemic inhibition of activated TGF‐β. Therefore, blocking just the specific RGD‐integrins or a local delivery via an inhaler (see GSK3008348) might be preferred. The inhalation delivery has the advantage that the drug is directly delivered to the infection area in the lungs. Furthermore, part of the drug can still enter the bloodstream to reach other parts of the body that are damaged through elevated TGF‐β, for example the kidneys.
Open questions
Only three studies so far have showed elevated TGF‐β in COVID‐19 patients. One study measured mRNA expression in BALF, another study revealed elevated TGF‐β in two different types of immune cells (see section above), and the third and most recent study showed increased TGF‐β in the serum of COVID‐19 patients. 98 However, it is not clear whether TGF‐β is the driver of the severity of COVID‐19 patients or whether it is a consequence of it. Further studies are necessary to analyse these alternatives. Nevertheless, blocking TGF‐β could still be beneficial to reduce or prevent complications associated with severe COVID‐19 infection and it deserves further investigation.
Conclusion
Considering that high levels of TGF‐β expression are involved in several pathologies including the complications of severe COVID‐19 outcome and that two studies revealed increased TGF‐β in COVID‐19 patients, we highly promote to include RGD‐binding integrin inhibitors in clinical trials as potential treatment for COVID‐19 patients (Figure 3). Blocking RGD‐binding integrins could also be beneficial for COVID‐19 patients with pre‐existing conditions since these patients have already increased TGF‐β at basal levels and blocking further TGF‐β activation could potentially reduce the risk of developing a more severe disease, including death (Figure 2).
Figure 3.
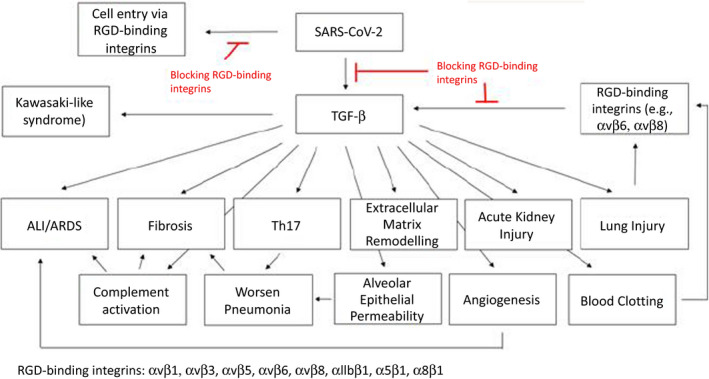
Scheme representing the potential effect of RGD‐binding integrin inhibitors to avoid the common complications derived from SARS‐CoV‐2 infection.
Furthermore, taking in consideration that there are active clinical trials using the mentioned drugs that inhibit TGF‐β directly or via the RGD‐binding integrins, it would be important to analyse whether these patients have a milder COVID‐19 disease course than comparable patients who take other medications.
Conflicts of Interest
The authors declare no competing interests.
Author Contribution
Ingrid Carvacho: Validation; Visualization; Writing‐review & editing. Matthias Piesche: Conceptualization; Formal analysis; Resources; Supervision; Validation; Visualization; Writing‐original draft; Writing‐review & editing.
Acknowledgments
The authors thank Dr Girija Goyal (Wyss Institute, Harvard University, USA) and Dr Luis Álvarez‐Vallina (12 de Octubre University Hospital, Spain) for important comments and suggestions on the manuscript. We further thank Nolan Piesche‐Carvacho for his fundamental contribution to our distraction during the pandemic.
References
- 1. Zhou F, Yu T, Du R et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet 2020; 395: 1054–1062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. CDC . Interim Clinical Guidance for Management of Patients with Confirmed Coronavirus Disease (COVID‐19). CDC at https://www.cdc.gov/coronavirus/2019‐ncov/hcp/clinical‐guidance‐management‐patients.html (2020).
- 3. Consortium WHOST , Pan H, Peto R et al. Repurposed Antiviral Drugs for Covid‐19 ‐ Interim WHO Solidarity Trial Results. N Engl J Med 2020; NEJMoa2023184. 10.1056/NEJMoa2023184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Wang K, Chen W, Zhou YS et al.SARS‐CoV‐2 invades host cells via a novel route: CD147‐spike protein. biorxiv Preprint. 10.1101/2020.03.14.988345. [DOI]
- 5. Vankadari N, Wilce JA. Emerging WuHan (COVID‐19) coronavirus: glycan shield and structure prediction of spike glycoprotein and its interaction with human CD26. Emerg Microbes Infect 2020; 9: 601–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Shilts J, Wright GJ.No evidence for basigin/CD147 as a direct SARS‐CoV‐2 spike binding receptor. biorxiv Preprint. 10.1101/2020.07.25.221036. [DOI] [PMC free article] [PubMed]
- 7. Sigrist CJ, Bridge A, Le Mercier P. A potential role for integrins in host cell entry by SARS‐CoV‐2. Antiviral Res 2020; 177: 104759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Sun C, Chen L, Yang J et al. SARS‐CoV‐2 and SARS‐CoV Spike‐RBD structure and receptor binding comparison and potential implications on neutralizing antibody and vaccine development. Biorxiv Preprint 2020. 10.1101/2020.02.16.951723. [DOI] [Google Scholar]
- 9. Hynes RO. Integrins: bidirectional, allosteric signaling machines. Cell 2002; 110: 673–687. [DOI] [PubMed] [Google Scholar]
- 10. Hussein HA, Walker LR, Abdel‐Raouf UM, Desouky SA, Montasser AK, Akula SM. Beyond RGD: virus interactions with integrins. Arch Virol 2015; 160: 2669–2681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Worthington JJ, Klementowicz JE, Travis MA. TGFβ: a sleeping giant awoken by integrins. Trends Biochem Sci 2011; 36: 47–54. [DOI] [PubMed] [Google Scholar]
- 12. Schmidt K, Keller M, Bader BL et al. Integrins modulate the infection efficiency of West Nile virus into cells. J Gen Virol 2013; 94: 1723–1733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chu JJ, Ng ML. Interaction of West Nile virus with αvβ3 integrin mediates virus entry into cells. J Biol Chem 2004; 279: 54533–54541. [DOI] [PubMed] [Google Scholar]
- 14. Schornberg KL, Shoemaker CJ, Dube D et al. α5β1‐integrin controls ebolavirus entry by regulating endosomal cathepsins. Proc Natl Acad Sciences USA 2009; 106: 8003–8008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Parry C, Bell S, Minson T, Browne H. Herpes simplex virus type 1 glycoprotein H binds to αvβ3 integrins. J Gen Virol 2005; 86: 7–10. [DOI] [PubMed] [Google Scholar]
- 16. Tresoldi I, Sangiuolo CF, Manzari V, Modesti A. SARS‐COV‐2 and infectivity: Possible increase in infectivity associated to integrin motif expression. J Med Virol 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Yan S, Sun H, Bu X, Wan G. New Strategy for COVID‐19: An Evolutionary Role for RGD Motif in SARS‐CoV‐2 and Potential Inhibitors for Virus Infection. Front Pharmacol 2020; 11: 912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Lamers MM, Beumer J, van der Vaart J et al. SARS‐CoV‐2 productively infects human gut enterocytes. Science 2020;369: 50–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hou YJ, Okuda K, Edwards CE et al. SARS‐CoV‐2 Reverse Genetics Reveals a Variable Infection Gradient in the Respiratory Tract. Cell 2020; 182: 429–446.e414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Wei Y, Zhang Y, Cai H et al. Roles of the putative integrin‐binding motif of the human metapneumovirus fusion (f) protein in cell‐cell fusion, viral infectivity, and pathogenesis. J Virol 2014; 88: 4338–4352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Beddingfield B, Iwanaga N, Chapagain P et al. The Integrin Binding Peptide, ATN‐161, as a Novel Therapy for SARS‐CoV‐2 Infection. JACC Basic Transl Sci 2020. 10.1016/j.jacbts.2020.10.003. Online ahead of print. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Calver J, John A, Jenkins G.Solid phase binding assay of SARS‐CoV‐2 Spike protein to binding to four RGD. Preprint at https://wwwnottinghamcrginfo/post/solid‐phase‐binding‐assay‐of‐sars‐cov‐2‐spike‐protein‐to‐binding‐to‐four‐rgd (2020).
- 23. Gheblawi M, Wang K, Viveiros A et al. Angiotensin‐Converting Enzyme 2: SARS‐CoV‐2 Receptor and Regulator of the Renin‐Angiotensin System: Celebrating the 20th Anniversary of the Discovery of ACE2. Circ Res 2020; 126: 1456–1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Cantuti‐Castelvetri L, Ojha R, Pedro LD et al. Neuropilin‐1 facilitates SARS‐CoV‐2 cell entry and infectivity. Science 2020; 370: 856–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Teoh CM, Tan SS, Tran T. Integrins as therapeutic targets for respiratory diseases. Curr Mol Med 2015; 15: 714–734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Denney L, Branchett W, Gregory LG, Oliver RA, Lloyd CM. Epithelial‐derived TGF‐β1 acts as a pro‐viral factor in the lung during influenza A infection. Mucosal Immunol 2018; 11: 523–535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Saito A, Horie M, Nagase T. TGF‐β Signaling in Lung Health and Disease. Int J Mol Sci 2018; 19:2460. 10.3390/ijms19082460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Tatler AL, Jenkins G. TGF‐β activation and lung fibrosis. Proc Am Thorac Soc 2012; 9: 130–136. [DOI] [PubMed] [Google Scholar]
- 29. Reed NI, Jo H, Chen C et al. The αvβ1 integrin plays a critical in vivo role in tissue fibrosis. Sci Transl Med 2015; 7: 288ra279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Brojakowska A, Narula J, Shimony R, Bander J. Clinical implications of SARS‐CoV‐2 interaction with renin angiotensin system: JACC review topic of the week. J Am Coll Cardiol 2020; 75: 3085–3095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Han JS, Choi BS, Yang CW, Kim YS. Aldosterone‐induced TGF‐β1 expression is regulated by mitogen‐activated protein kinases and activator protein‐1 in mesangial cells. J Korean Med Sci 2009; 24(Suppl): S195–203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Boumaza A, Gay L, Mezouar S et al.Monocytes and macrophages, targets of SARS‐CoV‐2: the clue for Covid‐19 immunoparalysis. Preprint at https://wwwbiorxivorg/content/10.1101/2020.09.17.300996v1 2020, ( 10.1101/2020.09.17.300996). [DOI]
- 33. Wang W, Su B, Pang L et al. High‐dimensional immune profiling by mass cytometry revealed immunosuppression and dysfunction of immunity in COVID‐19 patients. Cell Mol Immunol 2020; 17: 650–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Overgaard CE, Schlingmann B, Dorsainvil White S et al. The relative balance of GM‐CSF and TGF‐β1 regulates lung epithelial barrier function. Am J Physiol Lung Cell Mol Physiol 2015; 308: L1212–1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Quesnel C, Nardelli L, Piednoir P et al. Alveolar fibroblasts in acute lung injury: biological behaviour and clinical relevance. Eur Respir J 2010; 35: 1312–1321. [DOI] [PubMed] [Google Scholar]
- 36. Ito JT, Lourenco JD, Righetti RF, Tiberio I, Prado CM, Lopes F. Extracellular matrix component remodeling in respiratory diseases: what has been found in clinical and experimental studies? Cells 2019; 8: 342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Fahy RJ, Lichtenberger F, McKeegan CB, Nuovo GJ, Marsh CB, Wewers MD. The acute respiratory distress syndrome: a role for transforming growth factor‐β 1. Am J Respir Cell Mol Biol 2003; 28: 499–503. [DOI] [PubMed] [Google Scholar]
- 38. Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med 2000; 342: 1334–1349. [DOI] [PubMed] [Google Scholar]
- 39. Bhatraju PK, Ghassemieh BJ, Nichols M et al. Covid‐19 in Critically Ill Patients in the Seattle Region ‐ Case Series. N Engl J Med 2020; 382: 2012–2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Yang X, Yu Y, Xu J et al. Clinical course and outcomes of critically ill patients with SARS‐CoV‐2 pneumonia in Wuhan, China: a single‐centered, retrospective, observational study. Lancet Respir Med 2020; 8: 475–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Budinger GR, Chandel NS, Donnelly HK, Eisenbart J, Oberoi M, Jain M. Active transforming growth factor‐β1 activates the procollagen I promoter in patients with acute lung injury. Intensive Care Med 2005; 31: 121–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Hurst VI, Goldberg PL, Minnear FL, Heimark RL, Vincent PA. Rearrangement of adherens junctions by transforming growth factor‐β1: role of contraction. Am J Physiol 1999; 276: L582–595. [DOI] [PubMed] [Google Scholar]
- 43. Pittet JF, Griffiths MJ, Geiser T et al. TGF‐β is a critical mediator of acute lung injury. J Clin Investig 2001; 107: 1537–1544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Delpino MV, Quarleri J. SARS‐CoV‐2 pathogenesis: imbalance in the renin‐angiotensin system favors lung fibrosis. Front Cell Infect Microbiol 2020; 10: 340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Sime PJ, Xing Z, Graham FL, Csaky KG, Gauldie J. Adenovector‐mediated gene transfer of active transforming growth factor‐β1 induces prolonged severe fibrosis in rat lung. J Clin Investig 1997; 100: 768–776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Sanderson N, Factor V, Nagy P et al. Hepatic expression of mature transforming growth factor β 1 in transgenic mice results in multiple tissue lesions. Proc Natl Acad Sciences USA 1995; 92: 2572–2576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Wen Y, Deng BC, Zhou Y et al. Immunological features in patients with pneumonitis due to influenza A H1N1 infection. J Investig Allergol Clin Immunol 2011; 21: 44–50. [PubMed] [Google Scholar]
- 48. Lee CH, Chen RF, Liu JW et al. Altered p38 mitogen‐activated protein kinase expression in different leukocytes with increment of immunosuppressive mediators in patients with severe acute respiratory syndrome. J Immunol 2004; 172: 7841–7847. [DOI] [PubMed] [Google Scholar]
- 49. Zhao X, Nicholls JM, Chen YG. Severe acute respiratory syndrome‐associated coronavirus nucleocapsid protein interacts with Smad3 and modulates transforming growth factor‐β signaling. J Biol Chem 2008; 283: 3272–3280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Tilocca B, Soggiu A, Sanguinetti M et al. Comparative computational analysis of SARS‐CoV‐2 nucleocapsid protein epitopes in taxonomically related coronaviruses. Microbes Infect 2020; 22: 188–194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Idell S. Extravascular coagulation and fibrin deposition in acute lung injury. New Horiz 1994; 2: 566–574. [PubMed] [Google Scholar]
- 52. Dos Santos CC. Advances in mechanisms of repair and remodelling in acute lung injury. Intensive Care Med 2008; 34: 619–630. [DOI] [PubMed] [Google Scholar]
- 53. Fligiel SE, Standiford T, Fligiel HM et al. Matrix metalloproteinases and matrix metalloproteinase inhibitors in acute lung injury. Hum Pathol 2006; 37: 422–430. [DOI] [PubMed] [Google Scholar]
- 54. Ongchai S, Somnoo O, Kongdang P, Peansukmanee S, Tangyuenyong S. TGF‐β1 upregulates the expression of hyaluronan synthase 2 and hyaluronan synthesis in culture models of equine articular chondrocytes. J Vet Sci 2018; 19: 735–743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Xu Z, Shi L, Wang Y et al. Pathological findings of COVID‐19 associated with acute respiratory distress syndrome. Lancet Respir Med 2020; 8: 420–422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Shi Y, Wang Y, Shao C et al. COVID‐19 infection: the perspectives on immune responses. Cell Death Differ 2020; 27: 1451–1454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Pilewski JM, Latoche JD, Arcasoy SM, Albelda SM. Expression of integrin cell adhesion receptors during human airway epithelial repair in vivo . Am J Physiol 1997; 273: L256–263. [DOI] [PubMed] [Google Scholar]
- 58. Imanaka H, Shimaoka M, Matsuura N, Nishimura M, Ohta N, Kiyono H. Ventilator‐induced lung injury is associated with neutrophil infiltration, macrophage activation, and TGF‐β 1 mRNA upregulation in rat lungs. Anesth Analg 2001; 92: 428–436. [DOI] [PubMed] [Google Scholar]
- 59. Puthawala K, Hadjiangelis N, Jacoby SC et al. Inhibition of integrin αvβ6, an activator of latent transforming growth factor‐β, prevents radiation‐induced lung fibrosis. Am J Respir Crit Care Med 2008; 177: 82–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Hahm K, Lukashev ME, Luo Y et al. αvβ6 integrin regulates renal fibrosis and inflammation in Alport mouse. Am J Pathol 2007; 170: 110–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Lee CG, Cho SJ, Kang MJ et al . Early growth response gene 1‐mediated apoptosis is essential for transforming growth factor β1‐induced pulmonary fibrosis. J Exp Med 2004; 200: 377–389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Jenkins RG, Su X, Su G et al. Ligation of protease‐activated receptor 1 enhances αvβ6 integrin‐dependent TGF‐β activation and promotes acute lung injury. J Clin Investig 2006; 116: 1606–1614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Ganter MT, Roux J, Miyazawa B et al. Interleukin‐1β causes acute lung injury via αvβ5 and αvβ6 integrin‐dependent mechanisms. Circ Res 2008; 102: 804–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Debuc B, Smadja DM. Is COVID‐19 a new hematologic disease? Stem Cell Rev Rep 2020; 12: 1–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Magro C, Mulvey JJ, Berlin D et al. Complement associated microvascular injury and thrombosis in the pathogenesis of severe COVID‐19 infection: a report of five cases. Transl Res 2020; 220: 1–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Lodigiani C, Iapichino G, Carenzo L et al. Venous and arterial thromboembolic complications in COVID‐19 patients admitted to an academic hospital in Milan, Italy. Thromb Res 2020; 191: 9–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67. Jablonska E, Markart P, Zakrzewicz D, Preissner KT, Wygrecka M. Transforming growth factor‐β1 induces expression of human coagulation factor XII via Smad3 and JNK signaling pathways in human lung fibroblasts. J Biol Chem 2010; 285: 11638–11651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Samuel M, Pixley RA, Villanueva MA, Colman RW, Villanueva GB. Human factor XII (Hageman factor) autoactivation by dextran sulfate. Circular dichroism, fluorescence, and ultraviolet difference spectroscopic studies. J Biol Chem 1992; 267: 19691–19697. [PubMed] [Google Scholar]
- 69. Renne T, Schmaier AH, Nickel KF, Blomback M, Maas C. In vivo roles of factor XII. Blood 2012; 120: 4296–4303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Gobel K, Eichler S, Wiendl H, Chavakis T, Kleinschnitz C, Meuth SG. The coagulation factors fibrinogen, thrombin, and factor XII in inflammatory disorders‐a systematic review. Front Immunol 2018; 9: 1731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Nolte M, Margadant C. Controlling Immunity and Inflammation through Integrin‐Dependent Regulation of TGF‐β. Trends Cell Biol 2020; 30: 49–59. [DOI] [PubMed] [Google Scholar]
- 72. Renne T, Stavrou EX. Roles of Factor XII in Innate Immunity. Front Immunol 2019; 10: 2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Dong C, Zhu S, Wang T, Yoon W, Goldschmidt‐Clermont PJ. Upregulation of PAI‐1 is mediated through TGF‐β/Smad pathway in transplant arteriopathy. J Heart Lung Transplant 2002; 21: 999–1008. [DOI] [PubMed] [Google Scholar]
- 74. Hamsten A, Wiman B, de Faire U, Blomback M. Increased plasma levels of a rapid inhibitor of tissue plasminogen activator in young survivors of myocardial infarction. N Engl J Med 1985; 313: 1557–1563. [DOI] [PubMed] [Google Scholar]
- 75. Lijnen HR, Collen D. Impaired fibrinolysis and the risk for coronary heart disease. Circulation 1996; 94: 2052–2054. [DOI] [PubMed] [Google Scholar]
- 76. Ackermann M, Verleden SE, Kuehnel M et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid‐19. N Engl J Med 2020; 383: 120–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Han H, Yang L, Liu R et al. Prominent changes in blood coagulation of patients with SARS‐CoV‐2 infection. Clin Chem Lab Med 2020; 58: 1116–1120. [DOI] [PubMed] [Google Scholar]
- 78. Wang J, Hajizadeh N, Moore EE et al. Tissue plasminogen activator (tPA) treatment for COVID‐19 associated acute respiratory distress syndrome (ARDS): A case series. J Thromb Haemost 2020; 18: 1752–1755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Rovina N, Akinosoglou K, Eugen‐Olsen J, Hayek S, Reiser J, Giamarellos‐Bourboulis EJ. Soluble urokinase plasminogen activator receptor (suPAR) as an early predictor of severe respiratory failure in patients with COVID‐19 pneumonia. Crit Care 2020; 24: 187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Haupt TH, Petersen J, Ellekilde G et al. Plasma suPAR levels are associated with mortality, admission time, and Charlson Comorbidity Index in the acutely admitted medical patient: a prospective observational study. Crit Care 2012; 16: R130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Hayek SS, Leaf DE, Samman Tahhan A et al. Soluble Urokinase Receptor and Acute Kidney Injury. N Engl J Med 2020; 382: 416–426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Chang MC, Chang HH, Hsieh WC et al. Effects of transforming growth factor‐β1 on plasminogen activation in stem cells from the apical papilla: role of activating receptor‐like kinase 5/Smad2 and mitogen‐activated protein kinase kinase (MEK)/extracellular signal‐regulated kinase (ERK) signalling. Int Endod J 2020; 53: 647–659. [DOI] [PubMed] [Google Scholar]
- 83. Ferrari G, Cook BD, Terushkin V, Pintucci G, Mignatti P. Transforming growth factor‐β 1 (TGF‐β1) induces angiogenesis through vascular endothelial growth factor (VEGF)‐mediated apoptosis. J Cell Physiol 2009; 219: 449–458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Medford AR, Millar AB. Vascular endothelial growth factor (VEGF) in acute lung injury (ALI) and acute respiratory distress syndrome (ARDS): paradox or paradigm? Thorax 2006; 61: 621–626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. McElvaney OJ, McEvoy N, McElvaney OF et al. Characterization of the inflammatory response to severe COVID‐19 illness. Am J Respir Crit Care Med 2020; 202: 812–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. McMahon S, Charbonneau M, Grandmont S, Richard DE, Dubois CM. Transforming growth factor β1 induces hypoxia‐inducible factor‐1 stabilization through selective inhibition of PHD2 expression. J Biol Chem 2006; 281: 24171–24181. [DOI] [PubMed] [Google Scholar]
- 87. Wilkinson‐Berka JL, Jones D, Taylor G et al. SB‐267268, a nonpeptidic antagonist of αvβ3 and αvβ5 integrins, reduces angiogenesis and VEGF expression in a mouse model of retinopathy of prematurity. Invest Ophthalmol Vis Sci 2006; 47: 1600–1605. [DOI] [PubMed] [Google Scholar]
- 88. Yang Q, Ren GL, Wei B et al. Conditional knockout of TGF‐βRII /Smad2 signals protects against acute renal injury by alleviating cell necroptosis, apoptosis and inflammation. Theranostics 2019; 9: 8277–8293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Liu Z, Huang XR, Chen HY, Penninger JM, Lan HY. Loss of angiotensin‐converting enzyme 2 enhances TGF‐β/Smad‐mediated renal fibrosis and NF‐kappaB‐driven renal inflammation in a mouse model of obstructive nephropathy. Lab Invest 2012; 92: 650–661. [DOI] [PubMed] [Google Scholar]
- 90. Breuss JM, Gallo J, DeLisser HM et al. Expression of the β 6 integrin subunit in development, neoplasia and tissue repair suggests a role in epithelial remodeling. J Cell Sci 1995; 108(Pt 6): 2241–2251. [DOI] [PubMed] [Google Scholar]
- 91. McCurley A, Alimperti S, Campos‐Bilderback SB et al. Inhibition of αvβ5 integrin attenuates vascular permeability and protects against renal ischemia‐reperfusion injury. J Am Soc Nephrol 2017; 28: 1741–1752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92. Arnaldez FI, O'Day SJ, Drake CG et al. The Society for Immunotherapy of Cancer perspective on regulation of interleukin‐6 signaling in COVID‐19‐related systemic inflammatory response. J Immunother Cancer 2020; 8: e000930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Huang C, Wang Y, Li X et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China. Lancet 2020; 395: 497–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Chi Y, Ge Y, Wu B et al. Serum Cytokine and Chemokine profile in Relation to the Severity of Coronavirus disease 2019 (COVID‐19) in China. J Infect Dis 2020; 222: 746–754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Zhang X, Tan Y, Ling Y et al. Viral and host factors related to the clinical outcome of COVID‐19. Nature 2020; 583: 437–440. [DOI] [PubMed] [Google Scholar]
- 96. Wilson JG, Simpson LJ, Ferreira AM et al. Cytokine profile in plasma of severe COVID‐19 does not differ from ARDS and sepsis. JCI Insight 2020; 5: e140289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Xiong Y, Liu Y, Cao L et al. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID‐19 patients. Emerg Microbes Infect 2020; 9: 761–770. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98. Ferreira‐Gomes M, Kruglov A, Durek P et al. In severe COVID‐19, SARS‐CoV‐2 induces a chronic, TGF‐β‐dominated adaptive immune response. Medrxiv Preprint 2020. 10.1101/2020.09.04.20188169. [DOI]
- 99. Hadjadj J, Yatim N, Barnabei L et al. Impaired type I interferon activity and inflammatory responses in severe COVID‐19 patients. Science 2020; 369: 718–724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Blanco‐Melo D, Nilsson‐Payant BE, Liu WC et al. Imbalanced Host Response to SARS‐CoV‐2 Drives Development of COVID‐19. Cell 2020; 181: 1036–1045.e1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101. Guerin MV, Regnier F, Feuillet V et al. TGFβ blocks IFN α/β release and tumor rejection in spontaneous mammary tumors. Nat Commun 2019; 10: 4131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Bedke N, Sammut D, Green B et al. Transforming growth factor‐β promotes rhinovirus replication in bronchial epithelial cells by suppressing the innate immune response. PLoS One 2012; 7: e44580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Thomas BJ, Lindsay M, Dagher H et al. Transforming growth factor‐β enhances rhinovirus infection by diminishing early innate responses. Am J Respir Cell Mol Biol 2009; 41: 339–347. [DOI] [PubMed] [Google Scholar]
- 104. Grunwell JR, Yeligar SM, Stephenson S et al. TGF‐β1 Suppresses the Type I IFN Response and Induces Mitochondrial Dysfunction in Alveolar Macrophages. J Immunol 2018; 200: 2115–2128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. McCann KL, Imani F. Transforming growth factor β enhances respiratory syncytial virus replication and tumor necrosis factor α induction in human epithelial cells. J Virol 2007; 81: 2880–2886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Thornburg NJ, Shepherd B, Crowe JE Jr. Transforming growth factor β is a major regulator of human neonatal immune responses following respiratory syncytial virus infection. J Virol 2010; 84: 12895–12902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Liu Y, Russell MW. Diversion of the immune response to Neisseria gonorrhoeae from Th17 to Th1/Th2 by treatment with anti‐transforming growth factor β antibody generates immunological memory and protective immunity. MBio 2011; 2: e00095‐11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108. Voo KS, Wang YH, Santori FR et al. Identification of IL‐17‐producing FOXP3+ regulatory T cells in humans. Proc Natl Acad Sciences USA 2009; 106: 4793–4798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Wu D, Yang XO. TH17 responses in cytokine storm of COVID‐19: An emerging target of JAK2 inhibitor Fedratinib. J Microbiol Immunol Infect 2020; 53: 368–370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110. McGeachy MJ, Bak‐Jensen KS, Chen Y et al. TGF‐β and IL‐6 drive the production of IL‐17 and IL‐10 by T cells and restrain T(H)‐17 cell‐mediated pathology. Nat Immunol 2007; 8: 1390–1397. [DOI] [PubMed] [Google Scholar]
- 111. Wang J, Huizinga TW, Toes RE. De novo generation and enhanced suppression of human CD4+CD25+ regulatory T cells by retinoic acid. J Immunol 2009; 183: 4119–4126. [DOI] [PubMed] [Google Scholar]
- 112. Hatton RD. TGF‐β in Th17 cell development: the truth is out there. Immunity 2011; 34: 288–290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Risitano AM, Mastellos DC, Huber‐Lang M et al. Complement as a target in COVID‐19? Nat Rev Immunol 2020; 20: 343–344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Gu H, Mickler EA, Cummings OW et al. Crosstalk between TGF‐β1 and complement activation augments epithelial injury in pulmonary fibrosis. FASEB J 2014; 28: 4223–4234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Zuo Y, Yalavarthi S, Shi H et al. Neutrophil extracellular traps in COVID‐19. JCI Insight 2020; 5: e138999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Middleton EA, He XY, Denorme F et al. Neutrophil extracellular traps (NETs) contribute to immunothrombosis in COVID‐19 acute respiratory distress syndrome. Blood 2020; 136: 1169–1179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Verdoni L, Mazza A, Gervasoni A et al. An outbreak of severe Kawasaki‐like disease at the Italian epicentre of the SARS‐CoV‐2 epidemic: an observational cohort study. Lancet 2020; 395: 1771–1778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118. Kawasaki T, Kosaki F, Okawa S, Shigematsu I, Yanagawa H. A new infantile acute febrile mucocutaneous lymph node syndrome (MLNS) prevailing in Japan. Pediatrics 1974; 54: 271–276. [PubMed] [Google Scholar]
- 119. Shimizu C, Jain S, Davila S et al. Transforming growth factor‐β signaling pathway in patients with Kawasaki disease. Circ Cardiovasc Genet 2011; 4: 16–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Alvira CM, Guignabert C, Kim YM et al. Inhibition of transforming growth factor β worsens elastin degradation in a murine model of Kawasaki disease. Am J Pathol 2011; 178: 1210–1220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Guan WJ, Ni ZY, Hu Y et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382: 1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122. Zhang JJ, Dong X, Cao YY et al. Clinical characteristics of 140 patients infected with SARS‐CoV‐2 in Wuhan. China. Allergy 2020; 75: 1730–1741. [DOI] [PubMed] [Google Scholar]
- 123. Herder C, Zierer A, Koenig W, Roden M, Meisinger C, Thorand B. Transforming growth factor‐β1 and incident type 2 diabetes: results from the MONICA/KORA case‐cohort study, 1984–2002. Diabetes Care 2009; 32: 1921–1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Zaiman AL, Podowski M, Medicherla S et al. Role of the TGF‐β/Alk5 signaling pathway in monocrotaline‐induced pulmonary hypertension. Am J Respir Crit Care Med 2008; 177: 896–905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Meng XM, Nikolic‐Paterson DJ, Lan HY. TGF‐β: the master regulator of fibrosis. Nat Rev Nephrol 2016; 12: 325–338. [DOI] [PubMed] [Google Scholar]
- 126. Dobaczewski M, Chen W, Frangogiannis NG. Transforming growth factor (TGF)‐ β signaling in cardiac remodeling. J Mol Cell Cardiol 2011; 51: 600–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Boxall C, Holgate ST, Davies DE. The contribution of transforming growth factor‐β and epidermal growth factor signalling to airway remodelling in chronic asthma. Eur Respir J 2006; 27: 208–229. [DOI] [PubMed] [Google Scholar]
- 128. Kitamura H, Cambier S, Somanath S et al. Mouse and human lung fibroblasts regulate dendritic cell trafficking, airway inflammation, and fibrosis through integrin αvβ8‐mediated activation of TGF‐β. J Clin Investig 2011; 121: 2863–2875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Fernandez IE, Eickelberg O. The impact of TGF‐β on lung fibrosis: from targeting to biomarkers. Proc Am Thorac Soc 2012; 9: 111–116. [DOI] [PubMed] [Google Scholar]
- 130. Fain JN, Tichansky DS, Madan AK. Transforming growth factor β1 release by human adipose tissue is enhanced in obesity. Metabolism 2005; 54: 1546–1551. [DOI] [PubMed] [Google Scholar]
- 131. Colak S, Ten Dijke P. Targeting TGF‐β signaling in cancer. Trends Cancer 2017; 3: 56–71. [DOI] [PubMed] [Google Scholar]
- 132. Matl I, Viklicky O, Voska L, Lodererova A, Vitko S. The effect of different immunosuppressive regimens on TGF‐β1 expression in kidney transplant patients. Transpl Int 2005; 18: 668–671. [DOI] [PubMed] [Google Scholar]
- 133. Iwashima M, Love R. Potential of targeting TGF‐β for organ transplant patients. Future Med Chem 2013; 5: 281–289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134. Forsey RJ, Thompson JM, Ernerudh J et al. Plasma cytokine profiles in elderly humans. Mech Ageing Dev 2003; 124: 487–493. [DOI] [PubMed] [Google Scholar]
- 135. Senatorov VV Jr, Friedman AR, Milikovsky DZ et al. Blood‐brain barrier dysfunction in aging induces hyperactivation of TGFβ signaling and chronic yet reversible neural dysfunction. Sci Transl Med 2019; 11. [DOI] [PubMed] [Google Scholar]
- 136. Yadav H, Quijano C, Kamaraju AK et al. Protection from obesity and diabetes by blockade of TGF‐β/Smad3 signaling. Cell Metab 2011; 14: 67–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137. Basta J, Robbins L, Stout L, Prinsen MJ, Griggs DW, Rauchman M. Pharmacologic inhibition of RGD‐binding integrins ameliorates fibrosis and improves function following kidney injury. Physiol Rep 2020; 8: e14329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Gordon KJ, Blobe GC. Role of transforming growth factor‐β superfamily signaling pathways in human disease. Biochim Biophys Acta 2008; 1782: 197–228. [DOI] [PubMed] [Google Scholar]
- 139. Lavoie P, Robitaille G, Agharazii M, Ledbetter S, Lebel M, Lariviere R. Neutralization of transforming growth factor‐β attenuates hypertension and prevents renal injury in uremic rats. J Hypertens 2005; 23: 1895–1903. [DOI] [PubMed] [Google Scholar]
- 140. Thomas BJ, Kan OK, Loveland KL, Elias JA, Bardin PG. In the shadow of fibrosis: innate immune suppression mediated by transforming growth factor‐β. Am J Respir Cell Mol Biol 2016; 55: 759–766. [DOI] [PubMed] [Google Scholar]
- 141. Sama IE, Ravera A, Santema BT et al. Circulating plasma concentrations of angiotensin‐converting enzyme 2 in men and women with heart failure and effects of renin‐angiotensin‐aldosterone inhibitors. Eur Heart J 2020; 41: 1810–1817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Lei C, Qian K, Li T et al. Neutralization of SARS‐CoV‐2 spike pseudotyped virus by recombinant ACE2‐Ig. Nat Commun 2020; 11: 2070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143. Chen J, Jiang Q, Xia X et al. Individual variation of the SARS‐CoV‐2 receptor ACE2 gene expression and regulation. Aging Cell 2020; 19: e13168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Yeretssian G, Doiron K, Shao W et al. Gender differences in expression of the human caspase‐12 long variant determines susceptibility to Listeria monocytogenes infection. Proc Natl Acad Sciences USA 2009; 106: 9016–9020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145. Lin Y, Nakachi K, Ito Y et al. Variations in serum transforming growth factor‐β1 levels with gender, age and lifestyle factors of healthy Japanese adults. Dis Markers 2009; 27: 23–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146. Salvo E, Garasa S, Dotor J et al. Combined targeting of TGF‐β1 and integrin β3 impairs lymph node metastasis in a mouse model of non‐small‐cell lung cancer. Mol Cancer 2014; 13: 112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147. Nesti LJ, Caterson EJ, Wang M et al. TGF‐β1 calcium signaling increases α5 integrin expression in osteoblasts. J Orthop Res 2002; 20: 1042–1049. [DOI] [PubMed] [Google Scholar]
- 148. Chai X, Hu L, Zhang Y et al. Specific ACE2 Expression in Cholangiocytes May Cause Liver Damage After 2019‐nCoV Infection. biorxiv Preprint 2020. 10.1101/2020.02.03.931766. [DOI]
- 149. Diamond‐Stanic MK, You YH, Sharma K. Sugar, sex, and TGF‐β in diabetic nephropathy. Semin Nephrol 2012; 32: 261–268. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Wynn TA. Cellular and molecular mechanisms of fibrosis. J Pathol 2008; 214: 199–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151. Blush J, Lei J, Ju W, Silbiger S, Pullman J, Neugarten J. Estradiol reverses renal injury in Alb/TGF‐β1 transgenic mice. Kidney Int 2004; 66: 2148–2154. [DOI] [PubMed] [Google Scholar]
- 152. Chang Y, Lau WL, Jo H et al. Pharmacologic Blockade of αvβ1 Integrin Ameliorates Renal Failure and Fibrosis In Vivo. J Am Soc Nephrol 2017; 28: 1998–2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Brooks PC, Clark RA, Cheresh DA . Requirement of vascular integrin αvβ3 for angiogenesis. Science 1994; 264: 569–571. [DOI] [PubMed] [Google Scholar]
- 154. Van Waes C. Cell adhesion and regulatory molecules involved in tumor formation, hemostasis, and wound healing. Head Neck 1995; 17: 140–147. [DOI] [PubMed] [Google Scholar]
- 155. Asano Y, Ihn H, Yamane K, Jinnin M, Mimura Y, Tamaki K. Increased expression of integrin αvβ3 contributes to the establishment of autocrine TGF‐β signaling in scleroderma fibroblasts. J Immunol 2005; 175: 7708–7718. [DOI] [PubMed] [Google Scholar]
- 156. Mu D, Cambier S, Fjellbirkeland L et al. The integrin αvβ8 mediates epithelial homeostasis through MT1‐MMP‐dependent activation of TGF‐β1. J Cell Biol 2002; 157: 493–507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Eliceiri BP, Cheresh DA. The role of αv integrins during angiogenesis: insights into potential mechanisms of action and clinical development. J Clin Investig 1999; 103: 1227–1230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Khalifeh‐Soltani A, McKleroy W, Sakuma S et al. Mfge8 promotes obesity by mediating the uptake of dietary fats and serum fatty acids. Nat Med 2014; 20: 175–183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159. Nandrot EF, Kim Y, Brodie SE, Huang X, Sheppard D, Finnemann SC. Loss of synchronized retinal phagocytosis and age‐related blindness in mice lacking αvβ5 integrin. J Exp Med 2004; 200: 1539–1545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160. Su G, Atakilit A, Li JT et al. Effective treatment of mouse sepsis with an inhibitory antibody targeting integrin αvβ5. Crit Care Med 2013; 41: 546–553. [DOI] [PubMed] [Google Scholar]
- 161. Breuss JM, Gillett N, Lu L, Sheppard D, Pytela R. Restricted distribution of integrin β6 mRNA in primate epithelial tissues. J Histochem Cytochem 1993; 41: 1521–1527. [DOI] [PubMed] [Google Scholar]
- 162. Xu MY, Porte J, Knox AJ et al. Lysophosphatidic acid induces αvβ6 integrin‐mediated TGF‐β activation via the LPA2 receptor and the small G protein G α (q). Am J Pathol 2009; 174: 1264–1279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163. Araya J, Cambier S, Markovics JA et al. Squamous metaplasia amplifies pathologic epithelial‐mesenchymal interactions in COPD patients. J Clin Investig 2007; 117: 3551–3562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Ling KM, Sutanto EN, Iosifidis T et al. Reduced transforming growth factor β1 (TGF‐β1) in the repair of airway epithelial cells of children with asthma. Respirology 2016; 21: 1219–1226. [DOI] [PubMed] [Google Scholar]
- 165. Minagawa S, Lou J, Seed RI et al. Selective targeting of TGF‐β activation to treat fibroinflammatory airway disease. Sci Transl Med 2014; 6: 241ra279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166. Ma YQ, Qin J, Plow EF. Platelet integrin αIIbβ3: activation mechanisms. J Thromb Haemost 2007; 5: 1345–1352. [DOI] [PubMed] [Google Scholar]
- 167. Ciccone A, Motto C, Abraha I, Cozzolino F, Santilli I. Glycoprotein IIb‐IIIa inhibitors for acute ischaemic stroke. Cochrane Database Syst Rev 2014; CD005208. [DOI] [PubMed] [Google Scholar]
- 168. Centurión OA. Current Role of Platelet Glycoprotein IIb/IIIa Inhibition in the Therapeutic Management of Acute Coronary Syndromes in the Stent Era. J Cardiol Curr Res 2016; 5: 00175. [Google Scholar]
- 169. Jakhu H, Gill G, Singh A. Role of integrins in wound repair and its periodontal implications. J Oral Biol Craniofac Res 2018; 8: 122–125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Sui A, Zhong Y, Demetriades AM et al. Inhibition of integrin α5β1 ameliorates VEGF‐induced retinal neovascularization and leakage by suppressing NLRP3 inflammasome signaling in a mouse model. Graefes Arch Clin Exp Ophthalmol 2018; 256: 951–961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171. Sundaram A, Chen C, Khalifeh‐Soltani A et al. Targeting integrin α5β1 ameliorates severe airway hyperresponsiveness in experimental asthma. J Clin Investig 2017; 127: 365–374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172. Benoit YD, Larrivee JF, Groulx JF, Stankova J, Vachon PH, Beaulieu JF. Integrin α8β1 confers anoikis susceptibility to human intestinal epithelial crypt cells. Biochem Biophys Res Commun 2010; 399: 434–439. [DOI] [PubMed] [Google Scholar]
- 173. Levine D, Rockey DC, Milner TA, Breuss JM, Fallon JT, Schnapp LM. Expression of the integrin α8β1 during pulmonary and hepatic fibrosis. Am J Pathol 2000; 156: 1927–1935. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174. Worthington JJ, Kelly A, Smedley C et al. Integrin αvβ8‐Mediated TGF‐β Activation by Effector Regulatory T Cells Is Essential for Suppression of T‐Cell‐Mediated Inflammation. Immunity 2015; 42: 903–915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Uckun FM, Trieu V. Medical‐scientific rationale for a randomized, placebo‐controlled, phase 2 study of trabedersen/OT‐101 in COVID‐19 patients with hypoxemic respiratory failure. Anna pul and Cri Car Med 2020; 3: 01–09. [Google Scholar]
- 176. Katoh K, Rozewicki J, Yamada KD. MAFFT online service: multiple sequence alignment, interactive sequence choice and visualization. Brief Bioinform 2019; 20: 1160–1166. [DOI] [PMC free article] [PubMed] [Google Scholar]


