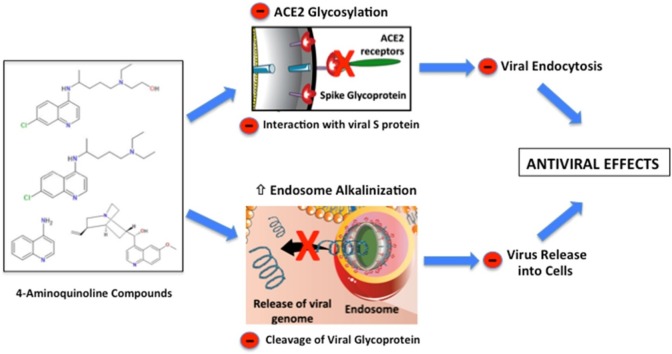
Graphical abstract
Mechanisms of action of 4-aminoquinoline compounds on viral infection.
Keywords: Sars-CoV-2, Antiviral drugs, Chloroquine, Hydroxychloroquine, Endosomal escape, Lysosomotropic drugs
Abstract
In 1918, quinine was used as one of the unscientifically based treatments against the H1N1 virus during the Spanish flu pandemic. Originally, quinine was extracted from the bark of Chinchona trees by South American natives of the Amazon forest, and it has been used to treat fever since the seventeenth century. The recent COVID-19 pandemic caused by Sars-Cov-2 infection has forced researchers to search for ways to prevent and treat this disease. Based on the antiviral potential of two 4-aminoquinoline compounds derived from quinine, known as chloroquine (CQ) and hydroxychloroquine (HCQ), clinical investigations for treating COVID-19 are being conducted worldwide. However, there are some discrepancies among the clinical trial outcomes.Thus, even after one hundred years of quinine use during the Spanish flu pandemic, the antiviral properties promoted by 4-aminoquinoline compounds remain unclear. The underlying molecular mechanisms by which CQ and HCQ inhibit viral replication open up the possibility of developing novel analogs of these drugs to combat COVID-19 and other viruses.
1. Spanish pandemic flu
The Spanish flu did not originate in Spain, as one would expect. Actually, one of the first reported cases of the unusual flu to the U.S. Public Health Service was by a physician in Haskell County, Kansas, in January 1918. Then on March 11 of the same year, more than 100 soldiers from the Fort Riley base had fallen sick. The reported symptoms include fever, sore throat and headache. At that time, knowledge about viruses and disease transmission was lacking and led to a rapid increase in the number of cases. Moreover, physicians/researchers did not know what caused the disease, which was later identified as the influenza virus, H1N1, also known as swine flu.
From the Fort Riley military base, American soldiers carried the disease to other military bases in the USA and eventually to Europe during World War I. The wartime censors of the countries involved in World War I suppressed news of the flu to avoid affecting the morale of the soldiers and the civilian population. However, Spain was one of the European nations that remained neutral during the War and the Spanish media freely reported on the flu in grisly detail. Thus, since the nations at war were undergoing a media blackout, and because Spain was the first to speak openly about the disease, the pandemic became known as the Spanish flu.
From 1918–1919, the Spanish flu claimed the lives of more than 50 million people worldwide. This highly contagious disease did not discriminate, affecting children, healthy adults, the elderly, the rich and poor and even animals (primarily cats and dogs). Similar to the ongoing COVID-19 pandemic, caused by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection, there was no vaccine or pharmacological therapy for Spanish flu patients.
The recommended precautions to prevent the spread of the flu included washing the inside the nose with soap and water every night and morning, forced sneezing in the evenings and mornings followed by deep breathing, not wearing a wrap or scarf around the neck and face for warmth, walking regularly, and eating oatmeal or cereal boiled in water or milk. The population and physicians also used various treatments with no scientific or medical basis such as sliced onions, bloodletting, inhaling fumes, drinking whiskey and taking laxatives, camphor, strychnine and quinine. Concerning quinine, it has been extracted from the bark of the Chinchona tree by South American Indians of the Amazon forest and used to treat fever since the seventeenth century.
2. The 4-aminoquinoline compounds
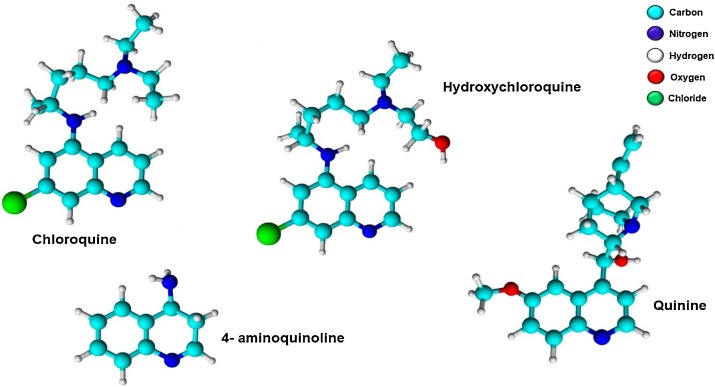
Quinine and its chemical analogs, chloroquine (CQ) and hydroxychloroquine (HCQ), are part of the 4-aminoquinoline family of compounds (Fig. 1 ). Despite the practical use of CQ during the Spanish flu, the antiviral effects of this drug (also known as quinine sulfate) have remained unaddressed for a long time. Interestingly, this medication is perhaps best known for its powerful antimalarial properties rather than its antiviral effects. However, more than one hundred years after the Spanish flupandemic, both CQ and HCQ have come to the forefront of discussions about their antiviral efficacy against Sars-CoV-2.
Fig. 1.
Chemical structures of chloroquine, hydroxychloroquine, quinine, and 4-aminoquinoline.
In the 1970s, several research groups investigated the antiviral properties of natural and synthetic compounds derived from quinine. Indeed, some studies have demonstrated that quinine is effective against viral diseases [[1], [2], [3]]. For example, quinoline derivatives, such as quinine [4], amodiaquine [4,5], primaquine [[4], [5], [6]], quinacrine [4,7] quinidine, pamaquine or plasmoquine, mefloquine [5,8] cinchonidine, camptothecin [9], and ferroquine [5,10], as well as CQ [4,5,8,10], HCQ [5,8] and some metabolites, like desethylchloroquine (a CQ metabolite) and desethylamodiaquine (a metabolite of amodiaquine) [11] have been shown to possess antiviral properties. While quinoline derivatives have been reported to have antiviral properties, recent studies failed to confirm that other derivatives, including halofantrine and lumefantrine, inhibite some viruses [8,12]. There is also in silico and in vitro evidence demonstrating the antiviral activity of CQ and HCQ against SARS-CoV-2 [13,14]. Furthermore, HCQ (EC50 = 0.72 μM) was shown to be more potent than CQ (EC50 = 5.47 μM) in SARS-CoV-2 infected Vero cells [15].
In 1934, CQ (N4-(7-Chloro-4-quinolinyl)-N1,N1-diethyl-1,4-pentanediamine) was first synthesized. This drug has been used to treat malaria and other diseases, such as rheumatoid arthritis, lupus erythematosus, hepatic amebiasis, sarcoidosis, and late cutaneous porphyria [16,17]. Approximately two decades later, CQ was modified to HCQ, which was eventually approved as an antimalarial agent by the Food and Drug Administration (FDA) in 1955. Due to the extensive use of CQ and HCQ by millions of people worldwide, the side effects and toxicities of these medications are well known. Studies have also shown that CQ and HCQ display anti-inflammatory [18] and immunomodulatory properties [19], including attenuated cytokine production by leukocytes that may play a pathogenic role in the progression of viral infections [16].
As shown in Fig. 1, the only difference between CQ and HCQ is the presence of a hydroxyl group at the end of one of the ethyl groups present in the molecule, resulting in HCQ being more water-soluble. Despite both compounds belonging to the 4-aminoquinoline family and having almost identical molecular volumes, there are differences in the intensities of their actions and toxicities.
Pharmacokinetic studies are mostly limited to the treatment of malaria. Both CQ and HCQ are well absorbed when administered orally [20], with a mean absorption half-life of four hours. Moreover, it has been estimated that the bioavailability of CQ is approximately 78 % in an oral solution and 89 % in tablet form [21]. Following absorption, 30–40 % of the drug binds to albumin and α1 glycoprotein. Consequently, CQ and HCQ are extensively distributed to all tissues and high doses of the drugs are required to achieve a given plasma concentration. In rats, the concentration of CQ is higher in the red blood cells (7.3–10.4 times), heart (6.8–184 times) and lung tissue (11.8–450 times) than in the plasma [22].
Previous work has shown that both drugs also bind strongly to melanin [23]. In arthritic patients, CQ remained in the skin for 6–7 months after cessation of the therapy, indicative of a long-term reservoir. Liver metabolism mediated by cytochrome P450 followed by renal excretion (21–47 % is excreted modified) is the principal route by which CQ and HCQ are removed from the body [24]. Combining the wide tissue distribution with a slow elimination rate can result in a long half-life (40–50 days). The chiraliy of these molecules is an important determinant of several pharmacological characterists [25,26]. Furthermore, R-enantiomers, S-enantiomers, and racemates of CQ and HCQ show differences in metabolism, excretion, and biological activity [27,28]. Drug pharmacodynamics is affected due to different stereochemistries and chiral mixtures of CQ and HCQ [29]. In 2000, Tucker was the first author to introduce the chiral switch concept [30] and D’Acquarica and Agranat [25], by using this concept, proposed that replacing CQ or HCQ racemates with single enantiomers for the COVID-19 treatment might be a good strategy to improve desired pharmacological effects. In addition, Lentini et al. [31] are the first authors to suggest the use of single CQ enantiomers in COVID-19 patients to avoid cardiac adverse events during the racemate administration, including hERG blocking and prolonged QT syndrome, thus improving the safety and efficacy profiles of these medications. Another potential severe side effect is the retinopathy that seems to be related with the accumulation of an enantiomer of HCQ in the ocular tissue due to the prolonged use of this drug [25]. In the same way, using the chiral switch strategy can help to decrease the risk of this side effect.
Concerning specific interactions, CQ has been shown to block potassium channels by inhibiting ERG [32]. Additionally, CQ may prolong the QT interval, causing the potentially lethal long QT syndrome (LQTS) [33]. Thus, the CQ interaction could occur at three different points, including cation-π and π-stacking interactions with proteic subunits lining the pore, such as Tyr-652 and Phe-656, and also with Ser-649 of the hERG chanel, displaying stereoselectivity cardiac activity [34,35]. Recently, one CQ enantiomer was shown to have a reduced effect on cardiac function, possibly due to weaker interactions resulting in less hERG inhibition [31]. In a recent study using a protein-protein interaction map, Gordon et al. [36] found that CQ is a potential SARS-CoV-2 inhibitor by binding to the host Sigma-1 receptor. Further studies are necessary to evaluate the stereoselectivity of different CQ and HCQ enantiomers on this inhibitory signaling pathway.
3. Mechanism of 4-aminoquinolines as antiviral drugs
3.1. Mechanism of endocytosis and endosomal escape of Sars-CoV-2 as a basis for understanding the antiviral activity of 4-aminoquinolines
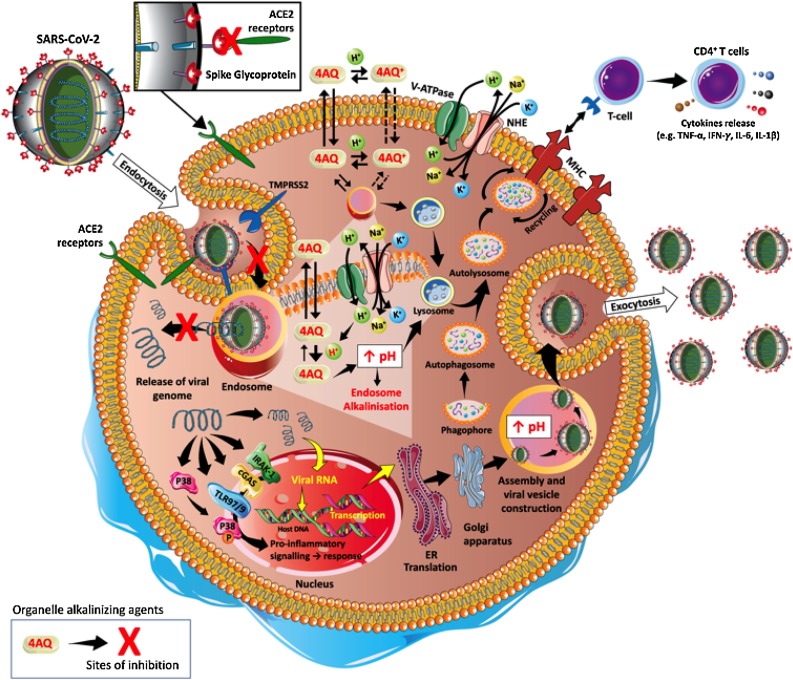
It is well known that SARS-CoV-2 is an RNA enveloped virus that uses its spike (S) protein to bind to the angiotensin-converting enzyme 2 (ACE-2) receptor on the surface of human macrophages, monocytes, and dendritic cells [19]. After interaction with the host cell, the virus-receptor complex is internalized into vesicles through the endosomal/lysosomal pathway [5]. This entry mechanism allows the virus to access the target cell and shields its genetic material from detection by the immune system [37]. Furthermore, endosomal/lysosomal acidification is necessary for viral glycoprotein cleavage and RNA release, essential steps for viral replication [38]. However, the enveloped virus must escape from the endosomal/lysosome compartment to be recycled back into the extracellular space or face degradation by the harsh lysosomal environment [37]. The endosomal escape mechanism involves the fusion of proteins embedded in the membrane of enveloped viruses with the endosomal membrane and the release of their genomic content into the cytoplasm [39]. The main virus-induced molecular mechanisms, specifically related to SARS-CoV-2, are depicted in Fig. 2 .
Fig. 2.
Mechanism of 4-aminoquinolines as antiviral drugs. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a single-stranded RNA-enveloped virus with the ability to infect host/human cells. This is dependent on the binding of its structural spike glycoproteins to angiotensin-converting enzyme 2 (ACE-2) receptors present on the surface of human cells. Once this process is initiated, the 2 transmembrane serine protease (TMPRSS2) primes the S-protein to facilitate the viral entry into the host cell through endocytosis pathway. Once internalized into the endosomes, SARS-CoV-2 efficiently delivers and spreads the viral nucleocapsid into several intracellular compartments. During the process of viral replication and host infection, a severe inflammatory cascade is activated by intracellular proteins, such as interleukin receptor-associated kinase-1 (IRAK-1), toll-like receptor 7/9 (TLR7/9), cyclic GMP-AMP synthase (cGAS) and phosphorylation of P38 mitogen-activated protein kinases (P38). Viral replication and host infection could be prevented by 4-aminoquinolines in the non-protonated form (4AQ) and protonated form (4AQ+). 4AQ diffuse passively across cell membranes, reach endosomes, lysosomes, and Golgi vesicles where they are converted to 4AQ+ increasing the pH. This alkalinization prevents the proteolytic cleavage of viral glycoprotein and the fusion of proteins that are embedded in the membrane of enveloped viruses with the endosomal membrane. Consequently, there is an inhibition of the genomic release (RNA) into the cytoplasm and viral replication. Other abbreviations: CD4+: Cluster of differentiation 4; ER: Endoplasmic reticulum; H+ : Proton; IFN-γ: Interferon gamma; IL-1β: Interleukin 1β; IL-6 Interleukin 6; K+ : Potassium ion; MHC: Major histocompatibility complex; Na+: Sodium ion; NHE: Activation of sodium/hydrogen exchanger; TNF-α: Tumour Necrosis Factor-α; V-ATPase: Vacuolar Na+, K+-ATPase.
There is evidence that CQ and HCQ block the uptake of the virion by inhibiting the glycosylation of the ACE-2 receptor in the plasma membrane [40,41]. However, the alkalinization of endosomes and lysosomes appears to the primary mechanism by which these substances exert their antiviral effects. Endocytosis is a process in which cells take up and internalize macromolecules (damaged proteins, lipoproteins, antigens and others) via specific cell-surface receptors and fuse these components to preexisting endosomes [42]. The primary pH regulator of endocytic compartments is the proton translocating vacuolar vacuolar-type H + ATPase (V-ATPase), which pumps protons and generates an acidic endosomal lumen (pH≈6.0) [37,42]. Consequently, the acidic internal pH promotes the dissociation of the ligands from the receptors and the cleavage of the viral glycoproteins by endosomal proteases [38,43]. Indeed, without endosomal acidification and cleavage processes, subsequent viral replication and infection are abrogated [44,45]. The remaining endocytic compartment becomes a lysosome and it is further acidified to a pH range (between 4 and 5) optimal for lysosomal proteases. Notably, lysosomal enzymes present low activity at neutral pH (e.g., in the cytoplasm), thus representing a protective mechanism in the event of lysosomal leakage [46].
Interestingly, some studies have been reported that omeprazole has a similar effect when compared to HCQ by blocking the proton pump on parasitic vacuoles and phagolysosomes, which provide in vitro antimalarial activity, as well as in vivo antileishmanial activity. Thus, the increased endo lysosomal pH occurs due to the H+/K+ATPase inhibition in gastric parietal cells, and suppression of the same pump in lysosomal membranes [47,48]. Kochar et al. [48] tested the in vivo efficacy of rifampicin (1200 mg/day) and omeprazole (20 mg), per 6 weeks, in 50 patients with anthroponotic cutaneous leishmaniasis. They found high efficacy with low toxicity, suggesting this intervention is a good alternative to treat leishmaniasis. Moreover, all patients showed a good drug tolerance without any side effects.
In general, macromolecules present in endosome/lysosome compartments are subjected to enzymatic degradation; however, viruses have evolved to take advantage of the lysosomal proteases [49], which promotes the release of the replication-competent viral genome in the host cell [5]. In this sense, the dysregulation of endosomal and lysosomal acidification and, consequently, their acidic pH-dependent proteases could be a highly effective pharmacological strategy for combating viruses, including Sars-CoV-2.
3.2. Mechanisms of alkalinization of endosomes and lysosomes as a basis for understanding the antiviral effects of 4-aminoquinolines
The term "lysosomotropic" was first used by Duve et al. De Duve, De Barsy, Poole and Tulkens [50] to designate all substances that are selectively taken up into lysosomes, irrespective of their chemical nature or uptake mechanism. However, since lysosomes and other cell compartments take up these substances, we will use the term "organelle alkalinizing agents" (OAAs).
In general, OAAs are weak bases with lipophilic properties. They are freely membrane-permeable in the non-protonated form but become less permeable when protonated (positively charged). Thus, in the non-protonated form, OAAs passively diffuse across cell membranes until becoming protonated and trapped, in a manner inversely proportional to the pH, according to the Henderson-Hasselbach equation, inside acidic intracellular compartments [50]. Thus, the concentration of these weak bases is increased in organelles with a low pH, such as endosomes, lysosomes, and Golgi vesicles, consequently leading to a rise in the pH [51].
Both HCQ and CQ are considered to be weak bases. HCQ contains three main functional groups with pK a values of <4.0, 8.3, and 9.7, two of which would be protonated at pH 7.4, and CQ has three with pK a values of 4.0, 8.4, and 10.2 [23]. The non-protonated form of CQ spontaneously and rapidly diffuses across cell membranes until it reaches endosomes, lysosomes, and Golgi vesicles, where CQ will become protonated, increasing the internal pH. This pH change impairs macromolecule assembly in endosomes, post-translational modifications in Golgi vesicles and acidic hydrolase-mediated protein degradation in lysosomes [5].
The kinetics and thermodynamics of CQ and HCQ transport across the human erythrocyte membrane have been studied [52]. It was found that the permeability coefficient of the unionized species of CQ (2.0 cm/sec at 25 °C) was much higher (about 50 times) than of HCQ (0.039 cm/sec at 25 °C). Despite this discrepancy, these two drugs exhibit similar apparent activation energies for transport. The authors concluded that interactions with the hydrogen bonding groups within the plasma membrane modulate the membrane transport kinetics of these drugs.
Low levels of OAAs (μM range) in the acidic organelles are sufficient at inhibiting the proteolytic cleavage of viral proteins [16]. However, the ability of OAAs to promote a rise in pH varies from compound to compound. For example, CQ is 10 times more potent than tributylamine > methylamine > triethylamine and benzylamine. It is plausible that these variations account for the lack of antiviral activity displayed by weak bases like atropine, eserine, and propranolol [49].
Previous studies have demonstrated that high concentrations of OAAs promote intense osmotic swelling, cytoplasmic vacuolation (fusion of lysosomes) and protonated base leakage. For example, CQ leads to vacuolation and autophagy activation in plasmodia-infected erythrocytes [53], which may explain why long-term CQ use sometimes results in retinal damage and other neurological side effects. However, since CQ and other 4-aminoquinolines directly inhibit the pH-dependent viral replication steps, short-term administration of OAAs may represent a viable strategy for attenuating the proliferative activity of the flaviviruses, retroviruses and coronaviruses [16].
According to Al-Bari Al‐Bari [5], the effectiveness of CQ analogs in the treatment of Chikungunya virus depends on the stage and severity of the disease. Thus, to maximize the antiviral effect of these drugs, the moment the treatment starts, dosage and duration must be considered to achieve steady-state plasma levels that can inhibit the viral infection. It is not unreasonable to speculate that Al-Baris's opinion about the efficacy of CQ and Chikungunyavirus [[54], [55], [56]] is also valid for Sars-CoV-2.
In addition to 4-aminoquinoline compounds, other OAAs with antiviral properties, including macrolide antibiotics (clarithromycin, bafilomycin, erythromycin, and azithromycin) and the non-steroidal anti-inflammatory drug indomethacin, have been reported [51].
It is worth mentioning that vacuolar-type H+-ATPase (V-ATPase) inhibition represents another pathway for promoting endosomal and lysosomal alkalinization. Antiviral activity was previously shown with omeprazole, esomeprazole [57], and bafilomycin A1, a specific inhibitor of V-ATPase [58] in vitro for several viruses, but not for Sars-CoV-2.
4. CQ and HCQ use in COVID-19 patients
Zhou et al. Zhou, Dai and Tong [59] proposed that COVID-19 patients respond better to HCQ than CQ. However, a systematic review on the use of CQ and HCQ in COVID-19, involving 65 clinical trials, some still in progress, and 159,669 patients worldwide, reported that the efficacy and safety of CQ and HCQ are still uncertain and that the routine use of these drugs is not recommended until their risks and benefits are more thoroughly evaluated [60]. Similar results and conclusions were also reached by Touret and Lamballerie Touret and de Lamballerie [61].
However, some studies support the use of HCQ for the treatment of COVID-19 patients. For example, Gautret et al. Gautret, Lagier, Parola, Meddeb, Mailhe, Doudier, Courjon, Giordanengo, Vieira and Dupont [62] treated COVID-19 patients with 200 mg HCQ, three times per day, for 10 days, and evaluated the viral load daily. They found that HCQ reduces the Sars-CoV-2 viral load, with most HCQ-treated patients presenting negative PCR results on day 6, a significant improvement over the control group. Additionally, when HCQ was combined with azithromycin, 100 % of the patients tested negative for the Sars-CoV-2 on day 6. It is important to point out that this study had a small sample size (16 control patients and 20 with HCQ alone or in combination with azithromycin). Moreover, six patients from the HCQ group were not followed-up for different reasons. Three patients from this group were transferred to an intensive care therapy unit, one due to side effects (nausea); one was a virus-discharged patient, and one passed away.
In another study, Gautret et al. Gautret, Lagier, Parola, Meddeb, Sevestre, Mailhe, Doudier, Aubry, Amrane and Seng [63] investigated the efficacy of HCQ (200 mg, three times per day) combined with azithromycin (500 mg on day 1 and 250 mg on days 2−5) for the treatment of COVID-19 patients for 3–6 days. The authors observed that approximately 80 % of patients recovered and were discharged from the hospital after the treatment and 83 % had negative PCR results for COVID-19 on day 7, 93 % on day 8 and 100 % on day 12.
Furthermore, in a French clinical study, Million et al. Million, Lagier, Gautret, Colson, Fournier, Amrane, Hocquart, Mailhe, Esteves-Vieira and Doudier [64] prospectively evaluated 1061 COVID-19 positive patients treated for at least three days. In the early stage of the disease, the patients received HCQ (200 mg three times daily for ten days) and azithromycin (500 mg on the first day followed by 250 mg daily for four days). The authors reported that 91.7 % of the patients tested negative for the virus. A poor clinical outcome occurred for 4.3 %, and 0.75 % died (74–95 years old). While this study did not include a control group for comparison, these human studies suggest that HCQ increased the elimination of viral charge, decreased duration of symptoms (fever, cough), and reduced pneumonia aggravation.
Additionally, Chen et al. Chen, Hu, Zhang, Jiang, Han, Yan, Zhuang, Hu and Zhang [65] studied 62 patients with COVID-19, randomly and equally divided into two groups: control and HCQ (400 mg per day, for 5 days). The authors reported that HCQ significantly decreased clinical recovery time (fever and cough) and improved pneumonia (80.6 % in the HCQ group versus 54.8 % in the control group).
In contrast, Tang et al. Tang, Cao, Han, Wang, Chen, Sun, Wu, Xiao, Liu and Chen [66] did not observe any clinical improvements in COVID-19 patients treated with HCQ. Additionally, Rosenberg et al. Rosenberg, Dufort, Udo, Wilberschied, Kumar, Tesoriero, Weinberg, Kirkwood, Muse and DeHovitz [67] evaluated 1438 hospitalized COVID-19 patients treated with HCQ, azithromycin, or combined therapy and found that none of these therapeutic approaches significantly reduced in-hospital mortality. Molina et al. Molina, Delaugerre, Le Goff, Mela-Lima, Ponscarme, Goldwirt and de Castro [68] found that 8 out of 11 COVID-19 patients still tested positive after 5−6 days of treatment with HCQ and azithromycin. Combining HCQ with lopinavir and ritonavir, in the absence or presence of INFβ-1b, also did not alter the clinical evolution of 92 patients with severe COVID-19 [69]. Huang et al. Huang, Tang, Pang, Li, Ma, Lu, Shu, You, Chen and Liang [70] reported similar results in moderate to severe COVID-19 patients treated with CQ (n = 10) or lopinavir/ritonavir (n = 12). On the other hand, Dastan et al. Dastan, Nadji, Saffaei, Marjani, Moniri, Jamaati, Hashemian, Shiva, Abedini and Varahram [71] concluded that the HCQ combined with lopinavir/ritonar/INF β-1b has to be considered, based on the findings in 20 patients with COVID-19. Furthermore, Boulware et al. Boulware, Pullen, Bangdiwala, Pastick, Lofgren, Okafor, Skipper, Nascene, Nicol and Abassi [72] evaluated 719 participants with a high risk of COVID-19 exposure and found no significant effect of HCQ on the disease prevention.
Detailed protocols, doses, and outcomes of the studies are described in Table 1 . Although the literature shows significant progress in understanding the antiviral effects of CQ and HCQ, there is a lack of clinical research to undeniably support the use of these drugs in COVID-19 patients.
Table 1.
Summary of the findings of studies on chloroquine and hydroxychloroquine in COVID-19 patients.
| Authors, groups, and duration of the study | Patients information | Drugs and treatment protocol | Treatment interruption or adverse effects | Main effects (intervention vs. control) |
|---|---|---|---|---|
| Gautret et al. [62,76] |
|
HCQ:3 × 200 mg per day | 3 transferred to ICU; 1 left the hospital; 1 due to nausea, 1 died |
|
| Groups: Control n= 16 | ATM: day 1 = 500 mg | |||
| HCQ n= 20 (6 plus ATM) | days 2–5 = 250 mg | |||
| Duration: 10 days | ||||
| Gautret et al. [63] |
|
HCQ: 3 × 200 mg per day | 1 due to drug interaction |
|
| Group: HCQ + ATM n= 80 | ATM: day 1 = 500 mg: | |||
| Duration: 3-6 days | days 2–5 = 250 mg | |||
| Million et al. [64] |
|
HCQ: 3 × 200 mg per day (for 10 days) | 10 transferred to ICU (2 died); 6 died (conventional hospital units); 3 due to abdominal pain, urticaria, erythematous and bullous rash |
|
| Group: HCQ + ATM n= 1061 | ||||
| ATM: day 1= 500 mg | ||||
| Duration: 3 days | ||||
| (retrospective study) | Days 2–5= 250 mg | |||
| Tang et al. [66] |
|
HCQ: 1200 mg per day (day 1–3); 800 mg per day (Days 4-11 in md and days 4-18 in sd) | 30 % of HCQ: adverse effects |
|
| Group: standard care n= 75 | (10 % diarrhea); 7 % standard care: adverse effects | |||
| HCQ n=75 | ||||
| Duration: 14-21 days | ||||
| Rosenberg et al.[67] |
|
HCQ: doses between 200 to 600 mg (90 % 400 mg in initial prescription, 70 % twice a day) | Abnormal ECG findings, mainly arrhythmia without significant differences between the groups; Diarrhea 11.6 % HCQ+ATM, 17 % only HCQ, 8.5 % only ATM vs. 7.2 % neither drug |
|
| ||||
| HCQ + ATM n = 735 | ATM: 220 to 500 mg (92 % 500 mg in initial prescription, 75.4 % once a day) | |||
| Only HCQ n = 271 | ||||
| Only ATM n = 211 | ||||
| (retrospective study) | ||||
| Chen et al. [65] |
|
HCQ: 400 mg per day | 1 due to headache |
|
| Groups: Control n = 31 | ||||
| HCQ n = 31 | ||||
| Duration: 5 days | ||||
| Geleris et al.[83] |
|
HCQ day 1: 2 × 600 mg; | 180 patients were intubated |
|
| HCQ monotherapy | 1 × 400 mg Days 2 – 5. | 166 patients died | ||
| n=1376 | ||||
| Median duration: 22.5 days | ||||
| Mercuro et al. [80] |
|
HCQ day 1: 2 × 400 mg | 10 patients discontinued treatment due to nausea, hypoglycemia, and 1 case of torsades de pointes. | Only QT interval was evaluated |
| HCQ monotherapy n=37 | 400 mg Days 2–5. | |||
| ATM: not described |
|
|||
| HCQ+ATM n=53 | ||||
| Borba et al. [82] |
|
High CQ: 2 × 600 mg for 10 days or low CQ day 1:2 × 450 mg; 450 mg Days 2–5 All received: ceftriaxone 2 × 1 g for 7 days + AZT 1 × 500 mg for 5 days |
1 developed rhabdomyolysis |
|
| CQ monotherapy | ||||
| n=81 | ||||
| Duration: 13-28 days | ||||
| 2 ventricular tachycardia | ||||
| Osetalmivir: 2× 75 mg for 5 days, when influenza was suspected. | ||||
| Boulware et al. [72] |
|
HQC day 1: 1 × 800 mg; |
|
|
| Groups: | 6 to 8 hours later: 600 mg; | |||
| 4 × 600 mg days 2-4. | ||||
| Control n= 407 | ||||
| HQC n= 414 | ||||
| Duration: 5 days | ||||
| Davoudi-Monfared et al. [69] |
|
INFβ-1a: 3× 44μg/ml was subcutaneously injected per week for 2 weeks; |
|
|
| Groups: | ||||
| HCQ day: 2 × 400 mg (first day) | ||||
| 2 × 200 mg per day | ||||
| Control (HC+lopinavir- ritonavir) n =46 | ||||
| Lopinavir-ritonavir: 1× 400 mg (first day) | ||||
| INFβ-1a + HCQ + lopinavir-ritonavir n= 46 | ||||
| 1× 100mg per day | ||||
| oratazanavir-ritonavir: 1× 300mg (first day) | ||||
| Duration: 2 weeks | ||||
| 100 mg per day | ||||
| Duration: 7 to 10 days | ||||
| Dastan et al. [71] Group: |
|
INFβ-1a: 44μg/ml was subcutaneously injected (initiated on day one and administrated every other day until day 10); |
|
|
| INF β 1-a + HCQ + lopinavir/ritonavir n= 20 | ||||
| HCQ: 200mg | ||||
| Duration: 10 days | Lopinavir/ritonavir: 200/50mg | |||
| Duration: 5 days | ||||
| Huang et al. [84] |
|
CQ: 2×500mg |
|
|
| Lopinavir/ritonavir:2× 400/100mg | ||||
| Groups: | ||||
| Duration: 10 days | ||||
| CQ n= 10 | ||||
| Lopinavir/ritonavir n= 12 | ||||
| Duration: 10 days | ||||
| Molina et al. [68] |
|
HCQ: 1 × 600 mg per day |
|
|
| Duration: 10 days | ||||
| Group : | ATM day 1: 1 × 500 mg | |||
| Day 2 to 5: 1 × 250 mg | ||||
| HCQ + ATM n= 11 | ||||
| Duration: 5 to 10 days |
ATM: azithromycin; CQ: chloroquine; HCQ: hydroxychloroquine; LRTI: lower respiratory tract infections (LRTI); URTI: upper respiratory tract infection; ICU, intensive care unit; ID infectious disease unit.
A variety of adverse side effects and activities have been reported for CQ and HCQ. Due to lower tissue/cell accumulation, HCQ is considered less toxic than CQ [41,73,74], and it is well known that retinal toxicity is a side effect of 4-aminoquinoline compounds. Mukwikwi et al. Mukwikwi, Pineau, Vinet, Clarke, Nashi, Kalache, Grenier and Bernatsky [75] investigated retinal complications in COVID-19 patients treated with HCQ and CQ and found that 5.5 % develop retinal toxicity. Interestingly, when HCQ was administered as an antimalarial therapy, low risk for retinal disease or maculopathy was observed when patients received 200−400 mg/day during follow-up at five years. However, the risk significantly increased when the patients were treated with doses higher than 400 mg/day, for a prolonged duration (>5 years) [76]. Li et al. Huang, Tang, Pang, Li, Ma, Lu, Shu, You, Chen and Liang [70] reported that CQ and HCQ had an immune-suppressive effect leading to decreased immunity. The authors recommended that these drugs should be administered only to COVID-19 patients in the early stages of the disease and presenting mild symptoms. Others reported that HCQ has anti-inflammatory and antiviral properties, with few side effects [70,77].
Among the well-known adverse events associated with CQ and HCQ are cardiac disorders. The QT-prolonging effect of CQ is modest and, in general, it does not result in a clinically significant QT-prolongation in patients with LQTS. On the other hand, no considerable effects on ECG parameters have been related to HCQ. Combining HCQ or CQ with other drugs (such as ritonavir plus lopinavir, azithromycin, and remdesivir) can result in higher plasma levels of 4-aminoquinoline compounds, along with a significantly prolonged QT-interval [78]. In an observational study involving patients with COVID-19 admitted to the hospital, Geleris et al. Geleris, Sun, Platt, Zucker, Baldwin, Hripcsak, Labella, Manson, Kubin and Barr [79] described that HCQ administration is not associated with increased risk of intubation or death. In turn, Mercuro et al. Mercuro, Yen, Shim, Maher, McCoy, Zimetbaum and Gold [80] reported that patients with COVID-19 who received HCQ to treat pneumonia show a high risk of QTc prolongation, and concurrent treatment with azithromycin is associated with more significant changes in the QT. Recentely, Giudicessi et al. [81] described a guidance for the treatment of COVID-19 patients with possible pharmacotherapies based on their potential QT-prolonged effect, especially CQ and HCQ. The authors considered several factors, including QT value, age, risk comorbid contitions, patient respiratory requirement, and eletrolyte levels (Ca2+, Mg2+, and K+) [81].
Interestingly, a prospective study conducted by Borba et al. Borba, Val, Sampaio, Alexandre, Melo, Brito, Mourão, Brito-Sousa, Baía-da-Silva and Guerra [82], in Manaus, Amazonas, Brazil, was the first randomized controlled clinical trial that evaluated CQ at high (600 mg) and low (400 mg) doses for treating critically ill COVID-19 patients. The authors recommended not using higher doses of the drug (recognized as safe in clinical protocols for other diseases) to treat critically ill patients due to high mortality. Accordingly, the FDA and NIH do not recommend using 600 mg of CQ in clinical studies.
In summary, the data concerning the efficacy and toxicity of CQ and HCQ in patients with COVID-19 is inconclusive. Much of this uncertainty revolves around when they should be administered, the limit between the therapeutic dose and the toxic dose of these drugs in hospitalized COVID-19 patients and the treatment duration.
5. Perspectives and inferences
After a critical analysis of the current scientific data concerning the pharmacological mechanism of 4-aminoquinolines in the treatment of several viruses, including COVID-19, we concluded that there are important points that require attention:
-
1
Since the clinical antiviral evaluation of 4-aminoquinoline compounds against Sars-CoV-2 is restricted to CQ and HCQ, it is necessary to expand these studies to other available natural and synthetic aminoquinolines (i.e., quinine, quinidine, mefloquine, amodiaquine, primaquine, quinacrine, pamaquine, plasmoquine, cinchonidine, camptothecin, halofantrine, lumefantrine, ferroquine), as well as their metabolites (desethylchloroquine, bisdesethylchloroquine, and desethylhydroxy-chloroquine).
-
2
While CQ and HCQ are potentially useful, inexpensive, and universally available candidates to treat viruses, the antiviral efficacy against SARS-CoV-2 is still inconclusive.
-
3
The understanding of the CQ and HCQ chiraliy is crucial to determinate antiviral activity and toxicity of different related molecules in terms of stereoselective pharmacokinetics and pharmacodynamics properties (racemic vs. R-enantiomers vs. S-enantiomers), salts (sulfate vs. phosphate; monophosphate vs.diphosphate), dosing regimens, routes of administration, and new pharmaceutical formulations.
-
4
Considering the long half-life, chronic toxicity, drug resistance, and absence of prophylactic effects against COVID-19, the use of CQ and HCQ should be investigated for short periods (7–10 days) with monitoring of QT-interval, risk comorbid factors, and eletrolyte control.
-
5
Based on the in vitro antiviral effects and the acute and chronic toxicity, HCQ should be preferentially administered instead of CQ. Neither drug is recommended for self-treatment or critically ill patients.
-
6
Based on the modest clinical efficacy of CQ and HCQ to prevent or treat COVID-19 and several other viruses, a potential clinical evaluation of CQ or HCQ associated with other antiviral drugs should be considered.
-
7
Based on the "trojan horse strategy" of the virus and the pharmacological action of 4-aminoquinolines "keeping the soldiers (virus) trapped inside the horse (vesicles)", the efficacy of the treatment may be considered/investigated only in the early stages of infection. Thus accounting for lack of efficacy" after the soldiers (virus) leave the horse (vesicles) and dominate the city (cell)" during later stages of infection. In this case, viral replication inhibition allows time for the adaptive immune response against infection, preventing hospitalization.
6. Remarks
Quinine was used in 1918 during the Spanish Flu, and CQ and HCQ are now considered for the treatment of COVID-19. However, even after one hundred years, 4-aminoquinoline compounds are still considered unconfirmed, but promising antiviral agents.
Declaration of Competing Interest
The authors report no declarations of interest.
Acknowledgments
The authors would like to thank Dr. Philip Newsholme, School of Pharmacy and Biomedical Sciences, CHIRI Biosciences, Curtin University, Perth, Western Australia, Australia, for revising the manuscript, and to Dr. Renato Padovese, Cruzeiro do Sul University, Sao Paulo, Brazil, for the constant academic and finantial support. The authors acknowledge grants from Sao Paulo Research Foundation (FAPESP, Brazil, grant IDs 2019/25936-5 and 2018/09868-7), Coordination for the Improvement of Higher Education Personnel (CAPES, Brazil, grant ID 88881.068515/2014-01), National Council for Scientific and Technological Development (CNPq, Brazil, grant ID 303753/2015-3), Guggenheim Foundation (GF, USA, grant ID John Simon Fellow 2008), and Pro-Rectory of Post-Graduate and Research of the Cruzeiro do Sul University (PRPGP/Cruzeiro do Sul, Brazil, grant ID 2020).
References
- 1.Baroni A., Paoletti I., Ruocco E., Ayala F., Corrado F., Wolf R., Tufano M., Donnarumma G. Antiviral effects of quinine sulfate on HSV-1 HaCat cells infected: analysis of the molecular mechanisms involved. J. Dermatol. Sci. 2007;47(3):253–255. doi: 10.1016/j.jdermsci.2007.05.009. [DOI] [PubMed] [Google Scholar]
- 2.Malakar S., Sreelatha L., Dechtawewat T., Noisakran S., Yenchitsomanus P.-t., Chu J.J.H., Limjindaporn T. Drug repurposing of quinine as antiviral against dengue virus infection. Virus Res. 2018;255:171–178. doi: 10.1016/j.virusres.2018.07.018. [DOI] [PubMed] [Google Scholar]
- 3.McDonagh P., Sheehy P.A., Fawcett A., Norris J.M. Antiviral effect of mefloquine on feline calicivirus in vitro. Vet. Microbiol. 2015;176(3–4):370–377. doi: 10.1016/j.vetmic.2015.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Marois I., Cloutier A., Meunier I., Weingartl H.M., Cantin A.M., Richter M.V. Inhibition of influenza virus replication by targeting broad host cell pathways. PLoS One. 2014;9(10) doi: 10.1371/journal.pone.0110631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Al‐Bari M.A.A. Targeting endosomal acidification by chloroquine analogs as a promising strategy for the treatment of emerging viral diseases. Pharmacol. Res. Perspect. 2017;5(1) doi: 10.1002/prp2.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gláz E.T., Szolgay E., Stöger I., Tálas M. Antiviral activity and induction of interferon-like substance by quinacrine and acranil. Antimicrob. Agents Chemother. 1973;3(5):537–541. doi: 10.1128/aac.3.5.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Glaz E., Szolgay E., Stöger I., Tálas M. Antiviral activity and induction of interferon-like substance by quinacrine and acranil. Antimicrob. Agents Chemother. 1973;3(5):537–541. doi: 10.1128/aac.3.5.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.D’Alessandro S., Scaccabarozzi D., Signorini L., Perego F., Ilboudo D.P., Ferrante P., Delbue S. The use of antimalarial drugs against viral infection. Microorganisms. 2020;8(1):85. doi: 10.3390/microorganisms8010085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cordell G.A. 1º ed. 1993. The Alkaloids: Chemistry and Pharmacology. [Google Scholar]
- 10.Vausselin T., Calland N., Belouzard S., Descamps V., Douam F., Helle F., François C., Lavillette D., Duverlie G., Wahid A. The antimalarial ferroquine is an inhibitor of hepatitis C virus. Hepatology. 2013;58(1):86–97. doi: 10.1002/hep.26273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Han Y., Pham H.T., Xu H., Quan Y., Mesplède T. Antimalarial drugs and their metabolites are potent Zika virus inhibitors. J. Med. Virol. 2019;91(7):1182–1190. doi: 10.1002/jmv.25440. [DOI] [PubMed] [Google Scholar]
- 12.Mazzon M., Ortega-Prieto A.M., Imrie D., Luft C., Hess L., Czieso S., Grove J., Skelton J.K., Farleigh L., Bugert J.J. Identification of broad-spectrum antiviral compounds by targeting viral entry. Viruses. 2019;11(2):176. doi: 10.3390/v11020176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hashem A.M., Alghamdi B.S., Algaissi A.A., Alshehri F.S., Bukhari A., Alfaleh M.A., Memish Z.A. Therapeutic use of chloroquine and hydroxychloroquine in COVID-19 and other viral infections: a narrative review. Travel Med. Infect. Dis. 2020 doi: 10.1016/j.tmaid.2020.101735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Braz H.L.B., Silveira J.A.M., Marinho A.D., de Moraes M.E.A., Moraes Filho M.O., Monteiro H.S.A., Jorge R.J.B. In silico study of azithromycin, chloroquine and hydroxychloroquine and their potential mechanisms of action against SARS-CoV-2 infection. Int. J. Antimicrob. Agents. 2020;56(3) doi: 10.1016/j.ijantimicag.2020.106119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yao X., Ye F., Zhang M., Cui C., Huang B., Niu P., Liu X., Zhao L., Dong E., Song C. In vitro antiviral activity and projection of optimized dosing design of hydroxychloroquine for the treatment of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) Clin. Infect. Dis. 2020;71(15):732–739. doi: 10.1093/cid/ciaa237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Savarino A., Di Trani L., Donatelli I., Cauda R., Cassone A. New insights into the antiviral effects of chloroquine. Lancet Infect. Dis. 2006;6(2):67–69. doi: 10.1016/S1473-3099(06)70361-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fox R.I. Elsevier; 1993. Mechanism of Action of Hydroxychloroquine As an Antirheumatic Drug, Seminars in Arthritis and Rheumatism; pp. 82–91. [DOI] [PubMed] [Google Scholar]
- 18.Whitehouse M., Boström H. Biochemical properties of anti-inflammatory drugs—VI: the effects of chloroquine (resochin), mepacrine (quinacrine) and some of their potential metabolites on cartilage metabolism and oxidative phosphorylation. Biochem. Pharmacol. 1965;14(8):1173–1184. doi: 10.1016/0006-2952(65)90295-9. [DOI] [PubMed] [Google Scholar]
- 19.Sanders J.M., Monogue M.L., Jodlowski T.Z., Cutrell J.B. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;323(18):1824–1836. doi: 10.1001/jama.2020.6019. [DOI] [PubMed] [Google Scholar]
- 20.McChesney E.W. Animal toxicity and pharmacokinetics of hydroxychloroquine sulfate. Am. J. Med. 1983;75(1):11–18. doi: 10.1016/0002-9343(83)91265-2. [DOI] [PubMed] [Google Scholar]
- 21.Smit C., Peeters M.Y., van den Anker J.N., Knibbe C.A. Chloroquine for SARS-CoV-2: implications of its unique pharmacokinetic and safety properties. Clin. Pharmacokinet. 2020:1. doi: 10.1007/s40262-020-00891-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Adelusi S., Salako L. Kinetics of the distribution and elimination of chloroquine in the rat. Gen. Pharmacol. 1982;13(5):433. doi: 10.1016/0306-3623(82)90110-0. [DOI] [PubMed] [Google Scholar]
- 23.Schroeder R., Gerber J. Chloroquine and hydroxychloroquine binding to melanin: some possible consequences for pathologies. Toxicol. Rep. 2014;1:963–968. doi: 10.1016/j.toxrep.2014.10.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Furst D.E. Pharmacokinetics of hydroxychloroquine and chloroquine during treatment of rheumatic diseases. Lupus. 1996;5(1_suppl):11–15. [PubMed] [Google Scholar]
- 25.D’Acquarica I., Agranat I. Chiral switches of chloroquine and hydroxychloroquine: potential drugs to treat COVID-19. Drug Discov. Today. 2020;25(7):1121–1123. doi: 10.1016/j.drudis.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ducharme J., Fieger H., Ducharme M.P., Khalil S.K., Wainer I.W. Enantioselective disposition of hydroxychloroquine after a single oral dose of the racemate to healthy subjects. Br. J. Clin. Pharmacol. 1995;40(2):127–133. doi: 10.1111/j.1365-2125.1995.tb05768.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Haberkorn A., Kraft H., Blaschke G. Antimalarial activity of the optical isomers of chloroquine diphosphate. Tropenmedizin und Parasitologie. 1979;30(3):308–312. [PubMed] [Google Scholar]
- 28.Ofori-Adjei D., Ericsson Ö., Lindström B., Hermansson J., Adjepon-Yamoah K., Sjöqvist F. Enantioselective analysis of chloroquine and desethylchloroquine after oral administration of racemic chloroquine. Ther. Drug Monit. 1986;8(4):457–461. doi: 10.1097/00007691-198612000-00014. [DOI] [PubMed] [Google Scholar]
- 29.McConathy J., Owens M.J. Stereochemistry in drug action. Prim. Care Companion J. Clin. Psychiatry. 2003;5(2):70. doi: 10.4088/pcc.v05n0202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tucker G.T. Chiral switches. Lancet. 2000;355(9209):1085–1087. doi: 10.1016/S0140-6736(00)02047-X. [DOI] [PubMed] [Google Scholar]
- 31.Lentini G., Cavalluzzi M.M., Habtemariam S. COVID-19, chloroquine repurposing, and cardiac safety concern: chirality might help. Molecules. 2020;25(8):1834. doi: 10.3390/molecules25081834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sánchez-Chapula J.A., Navarro-Polanco R.A., Culberson C., Chen J., Sanguinetti M.C. Molecular determinants of voltage-dependent human ether-a-go-go related gene (HERG) K+ channel block. J. Biol. Chem. 2002;277(26):23587–23595. doi: 10.1074/jbc.M200448200. [DOI] [PubMed] [Google Scholar]
- 33.Rodríguez-Menchaca A.A., Navarro-Polanco R.A., Ferrer-Villada T., Rupp J., Sachse F.B., Tristani-Firouzi M., Sánchez-Chapula J.A. The molecular basis of chloroquine block of the inward rectifier Kir2. 1 channel. Proc. Natl. Acad. Sci. 2008;105(4):1364–1368. doi: 10.1073/pnas.0708153105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Polak S., Wiśniowska B., Brandys J. Collation, assessment and analysis of literature in vitro data on hERG receptor blocking potency for subsequent modeling of drugs’ cardiotoxic properties. J. Appl. Toxicol. 2009;29(3):183–206. doi: 10.1002/jat.1395. [DOI] [PubMed] [Google Scholar]
- 35.Thai K.-M., Ecker G.F. Predictive models for HERG channel blockers: ligand-based and structure-based approaches. Curr. Med. Chem. 2007;14(28):3003–3026. doi: 10.2174/092986707782794087. [DOI] [PubMed] [Google Scholar]
- 36.Gordon D.E., Jang G.M., Bouhaddou M., Xu J., Obernier K., O’Meara M.J., Guo J.Z., Swaney D.L., Tummino T.A., Hüttenhain R., Kaake R.M., Richards A.L., Tutuncuoglu B., Foussard H., Batra J., Haas K., Modak M., Kim M., Haas P., Polacco B.J., Braberg H., Fabius J.M., Eckhardt M., Soucheray M., Bennett M.J., Cakir M., McGregor M.J., Li Q., Naing Z.Z.C., Zhou Y., Peng S., Kirby I.T., Melnyk J.E., Chorba J.S., Lou K., Dai S.A., Shen W., Shi Y., Zhang Z., Barrio-Hernandez I., Memon D., Hernandez-Armenta C., Mathy C.J.P., Perica T., Pilla K.B., Ganesan S.J., Saltzberg D.J., Ramachandran R., Liu X., Rosenthal S.B., Calviello L., Venkataramanan S., Lin Y., Wankowicz S.A., Bohn M., Trenker R., Young J.M., Cavero D., Hiatt J., Roth T., Rathore U., Subramanian A., Noack J., Hubert M., Roesch F., Vallet T., Meyer B., White K.M., Miorin L., Agard D., Emerman M., Ruggero D., García-Sastre A., Jura N., von Zastrow M., Taunton J., Schwartz O., Vignuzzi M., d’Enfert C., Mukherjee S., Jacobson M., Malik H.S., Fujimori D.G., Ideker T., Craik C.S., Floor S., Fraser J.S., Gross J., Sali A., Kortemme T., Beltrao P., Shokat K., Shoichet B.K., Krogan N.J. A SARS-CoV-2-human protein-protein interaction map reveals drug targets and potential drug-repurposing. bioRxiV (preprint) 2020:1–45. [Google Scholar]
- 37.Staring J., Raaben M., Brummelkamp T.R. Viral escape from endosomes and host detection at a glance. J. Cell. Sci. 2018;131(15) doi: 10.1242/jcs.216259. [DOI] [PubMed] [Google Scholar]
- 38.Gramberg T., Soilleux E., Fisch T., Lalor P.F., Hofmann H., Wheeldon S., Cotterill A., Wegele A., Winkler T., Adams D.H. Interactions of LSECtin and DC-SIGN/DC-SIGNR with viral ligands: differential pH dependence, internalization and virion binding. Virology. 2008;373(1):189–201. doi: 10.1016/j.virol.2007.11.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Harrison S.C. Viral membrane fusion. Virology. 2015;479:498–507. doi: 10.1016/j.virol.2015.03.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Vincent M.J., Bergeron E., Benjannet S., Erickson B.R., Rollin P.E., Ksiazek T.G., Seidah N.G., Nichol S.T. Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. Virol. J. 2005;2(1):1–10. doi: 10.1186/1743-422X-2-69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu J., Cao R., Xu M., Wang X., Zhang H., Hu H., Li Y., Hu Z., Zhong W., Wang M. Hydroxychloroquine, a less toxic derivative of chloroquine, is effective in inhibiting SARS-CoV-2 infection in vitro. Cell Discov. 2020;6(1):1–4. doi: 10.1038/s41421-020-0156-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Cain C.C., Sipe D.M., Murphy R.F. Regulation of endocytic pH by the Na+, K+-ATPase in living cells. Proc. Natl. Acad. Sci. 1989;86(2):544–548. doi: 10.1073/pnas.86.2.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chandran K., Sullivan N.J., Felbor U., Whelan S.P., Cunningham J.M. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science. 2005;308(5728):1643–1645. doi: 10.1126/science.1110656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Martinson J.A., Montoya C.J., Usuga X., Ronquillo R., Landay A.L., Desai S.N. Chloroquine modulates HIV-1-induced plasmacytoid dendritic cell alpha interferon: implication for T-cell activation. Antimicrob. Agents Chemother. 2010;54(2):871–881. doi: 10.1128/AAC.01246-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Shivanna V., Kim Y., Chang K.-O. Endosomal acidification and cathepsin L activity is required for calicivirus replication. Virology. 2014;464:287–295. doi: 10.1016/j.virol.2014.07.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Villamil Giraldo A.M., Appelqvist H., Ederth T., Öllinger K. Lysosomotropic agents: impact on lysosomal membrane permeabilization and cell death. Biochem. Soc. Trans. 2014;42(5):1460–1464. doi: 10.1042/BST20140145. [DOI] [PubMed] [Google Scholar]
- 47.Skinner-Adams T., Davis T.M. Synergistic in vitro antimalarial activity of omeprazole and quinine. Antimicrob. Agents Chemother. 1999;43(5):1304–1306. doi: 10.1128/aac.43.5.1304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kochar D.K., Saini G., Kochar S.K., Sirohi P., Bumb R.A., Mehta R.D., Purohit S.K. A double blind, randomised placebo controlled trial of rifampicin with omeprazole in the treatment of human cutaneous leishmaniasis. J. Vector Borne Dis. 2006;43(4):161–167. [PubMed] [Google Scholar]
- 49.Ohkuma S., Poole B. Cytoplasmic vacuolation of mouse peritoneal macrophages and the uptake into lysosomes of weakly basic substances. J. Cell Biol. 1981;90(3):656–664. doi: 10.1083/jcb.90.3.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.De Duve C., De Barsy T., Poole B., Tulkens P. Lysosomotropic agents. Biochem. Pharmacol. 1974;23(18):2495–2531. doi: 10.1016/0006-2952(74)90174-9. [DOI] [PubMed] [Google Scholar]
- 51.Homolak J., Kodvanj I. Widely available lysosome targeting agents should be considered as a potential therapy for COVID-19. Int. J. Antimicrob. Agents. 2020 doi: 10.1016/j.ijantimicag.2020.106044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ferrari V., Cutler D.J. Kinetics and thermodynamics of chloroquine and hydroxychloroquine transport across the human erythrocyte membrane. Biochem. Pharmacol. 1991;41(1):23–30. doi: 10.1016/0006-2952(91)90006-q. [DOI] [PubMed] [Google Scholar]
- 53.Hain A.U., Bosch J. Autophagy in plasmodium, a multifunctional pathway? Comput. Struct. Biotechnol. J. 2013;8(11) doi: 10.5936/csbj.201308002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Lamballerie X.D., Boisson V., Reynier J.-C., Enault S., Charrel R.N., Flahault A., Roques P., Grand R.L. On chikungunya acute infection and chloroquine treatment. Vector-Borne Zoonotic Dis. 2008;8(6):837–840. doi: 10.1089/vbz.2008.0049. [DOI] [PubMed] [Google Scholar]
- 55.Kaur P., Chu J.J.H. Chikungunya virus: an update on antiviral development and challenges. Drug Discov. Today. 2013;18(19–20):969–983. doi: 10.1016/j.drudis.2013.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Stock I. Chikungunya fever–expanded distribution of a re-emerging tropical infectious disease. Medizinische Monatsschrift fur Pharmazeuten. 2009;32(1):17–26. [PubMed] [Google Scholar]
- 57.Long J., Wright E., Molesti E., Temperton N., Barclay W. Antiviral therapies against Ebola and other emerging viral diseases using existing medicines that block virus entry. F1000Research. 2015;4 doi: 10.12688/f1000research.6085.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Yeganeh B., Ghavami S., Kroeker A.L., Mahood T.H., Stelmack G.L., Klonisch T., Coombs K.M., Halayko A.J. Suppression of influenza A virus replication in human lung epithelial cells by noncytotoxic concentrations bafilomycin A1. Am. J. Physiol. Lung Cell. Mol. Physiol. 2015;308(3):L270–L286. doi: 10.1152/ajplung.00011.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhou D., Dai S.-M., Tong Q. COVID-19: a recommendation to examine the effect of hydroxychloroquine in preventing infection and progression. J. Antimicrob. Chemother. 2020 doi: 10.1093/jac/dkaa114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Pacheco R.L., Pachito D.V., Bagattini Â.M., Riera R. 2020. HIDROXICLOROQUINA E CLOROQUINA PARA INFECÇÃO POR COVID-19. Revisão sistemática rápida–atualização 10/04. [Google Scholar]
- 61.Touret F., de Lamballerie X. Of chloroquine and COVID-19. Antiviral Res. 2020 doi: 10.1016/j.antiviral.2020.104762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gautret P., Lagier J.-C., Parola P., Meddeb L., Mailhe M., Doudier B., Courjon J., Giordanengo V., Vieira V.E., Dupont H.T. Hydroxychloroquine and azithromycin as a treatment of COVID-19: results of an open-label non-randomized clinical trial. Int. J. Antimicrob. Agents. 2020 doi: 10.1016/j.ijantimicag.2020.105949. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 63.Gautret P., Lagier J.-C., Parola P., Meddeb L., Sevestre J., Mailhe M., Doudier B., Aubry C., Amrane S., Seng P. Clinical and microbiological effect of a combination of hydroxychloroquine and azithromycin in 80 COVID-19 patients with at least a six-day follow up: a pilot observational study. Travel Med. Infect. Dis. 2020 doi: 10.1016/j.tmaid.2020.101663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Million M., Lagier J.-C., Gautret P., Colson P., Fournier P.-E., Amrane S., Hocquart M., Mailhe M., Esteves-Vieira V., Doudier B. Full-length title: Early treatment of COVID-19 patients with hydroxychloroquine and azithromycin: a retrospective analysis of 1061 cases in Marseille, France. Travel Med. Infect. Dis. 2020 doi: 10.1016/j.tmaid.2020.101738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Chen Z., Hu J., Zhang Z., Jiang S., Han S., Yan D., Zhuang R., Hu B., Zhang Z. Efficacy of hydroxychloroquine in patients with COVID-19: results of a randomized clinical trial. medRxiv (preprint) 2020:1–11. [Google Scholar]
- 66.Tang W., Cao Z., Han M., Wang Z., Chen J., Sun W., Wu Y., Xiao W., Liu S., Chen E. Hydroxychloroquine in patients with mainly mild to moderate coronavirus disease 2019: open label, randomised controlled trial. BMJ. 2020;369 doi: 10.1136/bmj.m1849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Rosenberg E.S., Dufort E.M., Udo T., Wilberschied L.A., Kumar J., Tesoriero J., Weinberg P., Kirkwood J., Muse A., DeHovitz J. Association of treatment with hydroxychloroquine or azithromycin with in-hospital mortality in patients with COVID-19 in New York state. J. Emerg. Med. 2020;59(2):333–338. doi: 10.1001/jama.2020.8630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Molina J.M., Delaugerre C., Le Goff J., Mela-Lima B., Ponscarme D., Goldwirt L., de Castro N. No evidence of rapid antiviral clearance or clinical benefit with the combination of hydroxychloroquine and azithromycin in patients with severe COVID-19 infection. Med. Mal. Infect. 2020;50(384):30085–30088. doi: 10.1016/j.medmal.2020.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Davoudi-Monfared E., Rahmani H., Khalili H., Hajiabdolbaghi M., Salehi M., Abbasian L., Kazemzadeh H., Yekaninejad M.S. A randomized clinical trial of the efficacy and safety of interferon β-1a in treatment of severe COVID-19. Antimicrob. Agents Chemother. 2020;64(9) doi: 10.1128/AAC.01061-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Huang M., Tang T., Pang P., Li M., Ma R., Lu J., Shu J., You Y., Chen B., Liang J. Treating COVID-19 with chloroquine. J. Mol. Cell Biol. 2020;12(4):322–325. doi: 10.1093/jmcb/mjaa014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Dastan F., Nadji S.A., Saffaei A., Marjani M., Moniri A., Jamaati H., Hashemian S.M., Shiva P.B., Abedini A., Varahram M. Subcutaneous administration of interferon beta-1a for COVID-19: a non-controlled prospective trial. Int. Immunopharmacol. 2020 doi: 10.1016/j.intimp.2020.106688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Boulware D.R., Pullen M.F., Bangdiwala A.S., Pastick K.A., Lofgren S.M., Okafor E.C., Skipper C.P., Nascene A.A., Nicol M.R., Abassi M. A randomized trial of hydroxychloroquine as postexposure prophylaxis for Covid-19. N. Engl. J. Med. 2020 doi: 10.1056/NEJMoa2016638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chatre C., Roubille F., Vernhet H., Jorgensen C., Pers Y.-M. Cardiac complications attributed to chloroquine and hydroxychloroquine: a systematic review of the literature. Drug Saf. 2018;41(10):919–931. doi: 10.1007/s40264-018-0689-4. [DOI] [PubMed] [Google Scholar]
- 74.Jorge A., Ung C., Young L.H., Melles R.B., Choi H.K. Hydroxychloroquine retinopathy—Implications of research advances for rheumatology care. Nat. Rev. Rheumatol. 2018;14(12):693–703. doi: 10.1038/s41584-018-0111-8. [DOI] [PubMed] [Google Scholar]
- 75.Mukwikwi E.-R., Pineau C.A., Vinet E., Clarke A.E., Nashi E., Kalache F., Grenier L.-P., Bernatsky S. Retinal complications in patients with systemic lupus erythematosus treated with antimalarial drugs. J. Rheumatol. 2020;47(4):553–556. doi: 10.3899/jrheum.181102. [DOI] [PubMed] [Google Scholar]
- 76.Geamnu A., Popa-Cherecheanu A., Marinescu B., Geamanu C., Voinea L. Retinal toxicity associated with chronic exposure to hydroxychloroquine and its ocular screening. Rev. J. Med. Life. 2014;7(3):322. [PMC free article] [PubMed] [Google Scholar]
- 77.Sharma H.B., Vanapalli K.R., Cheela V.S., Ranjan V.P., Jaglan A.K., Dubey B., Goel S., Bhattacharya J. Challenges, opportunities, and innovations for effective solid waste management during and post COVID-19 pandemic. Resour. Conserv. Recycl. 2020;162 doi: 10.1016/j.resconrec.2020.105052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Wu C.-I., Postema P.G., Arbelo E., Behr E.R., Bezzina C.R., Napolitano C., Robyns T., Probst V., Schulze-Bahr E., Remme C.A. SARS-CoV-2, COVID-19 and inherited arrhythmia syndromes. Heart Rhythm. 2020;17(9):1456–1462. doi: 10.1016/j.hrthm.2020.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Geleris J., Sun Y., Platt J., Zucker J., Baldwin M., Hripcsak G., Labella A., Manson D.K., Kubin C., Barr R.G. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N. Engl. J. Med. 2020;382(25):2411–2418. doi: 10.1056/NEJMoa2012410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Mercuro N.J., Yen C.F., Shim D.J., Maher T.R., McCoy C.M., Zimetbaum P.J., Gold H.S. Risk of QT interval prolongation associated with use of hydroxychloroquine with or without concomitant azithromycin among hospitalized patients testing positive for coronavirus disease 2019 (COVID-19) JAMA Cardiol. 2020;5(9):1036–1041. doi: 10.1001/jamacardio.2020.1834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Giudicessi J.R., Noseworthy P.A., Friedman P.A., Ackerman M.J. Urgent guidance for navigating and circumventing the QTc-prolonging and torsadogenic potential of possible pharmacotherapies for coronavirus disease 19 (COVID-19) Mayo Clin. Proc. 2020;95(6):1213–1221. doi: 10.1016/j.mayocp.2020.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Borba M.G.S., Val F.F.A., Sampaio V.S., Alexandre M.A.A., Melo G.C., Brito M., Mourão M.P.G., Brito-Sousa J.D., Baía-da-Silva D., Guerra M.V.F. Effect of high vs low doses of chloroquine diphosphate as adjunctive therapy for patients hospitalized with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection: a randomized clinical trial. JAMA Network Open. 2020;3(4) doi: 10.1001/jamanetworkopen.2020.8857. e208857–e208857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Geleris J., Sun Y., Platt J., Zucker J., Baldwin M., Hripcsak G., Labella A., Manson D.K., Kubin C., Barr R.G., Sobieszczyk M.E., Schluger N.W. Observational study of hydroxychloroquine in hospitalized patients with Covid-19. N. Engl. J. Med. 2020;382(25):2411–2418. doi: 10.1056/NEJMoa2012410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Huang H., Zhang M., Chen C., Zhang H., Wei Y., Tian J., Shang J., Deng Y., Du A., Dai H. Clinical characteristics of COVID-19 in patients with preexisting ILD: a retrospective study in a single center in Wuhan, China. J. Med. Virol. 2020;92(11):2742–2750. doi: 10.1002/jmv.26174. [DOI] [PMC free article] [PubMed] [Google Scholar]