Abstract
BACKGROUND AND PURPOSE: Color Doppler sonography (CDS) suffers from such disadvantages as high interobserver variability and problems with data presentation and storage. We therefore performed a prospective trial to assess the role of three-dimensional CDS (3D-CDS) in the evaluation of internal carotid artery stenosis (ICAS).
METHODS: We included 32 consecutive patients with sonographically verified ICAS (30–99%) and 16 asymptomatic volunteers. All underwent CDS and 3D-CDS, and 23 patients also underwent intraarterial angiography. The sensitivity, specificity, positive predictive value, and negative predictive value for the detection of high-grade ICAS were analyzed by three blinded readers, who also graded the percentage of ICAS.
RESULTS: CDS revealed 14 patients with mild to moderate ICAS and 18 patients with high-grade ICAS. Mean sensitivity of 3D-CDS was 81.5%, mean specificity was 98.9%, mean positive predictive value was 97.9%, and mean negative predictive value was 89.9%. 3D-CDS correlated significantly with CDS (mean r = 0.85; P < .001) and angiography (mean r = 0.57; P = .01).
CONCLUSION: 3D-CDS findings correlate with those obtained by CDS and angiography; 3D-CDS also offers excellent interobserver correlation, positive predictive value, and specificity, approaching 100% for the detection of high-grade ICAS.
Cerebral infarction caused by carotid artery atherosclerosis is related to degree of stenosis. Toole and Castaldo (1) have reported that a more hemodynamically disturbed flow has a greater propensity for distal cerebrovascular events. In 1991, the initial published results of the North American Symptomatic Carotid Endarterectomy Trial (NASCET) and the European Carotid Surgery Trial demonstrated that patients having symptomatic high-grade stenoses of the internal carotid artery of 70% or greater who were treated with carotid endarterectomy had much better outcomes than did those treated medically (2, 3). Thus, screening for severe internal carotid artery stenosis (ICAS) has gained considerable clinical importance. Selective intraarterial angiography of the carotid artery is still seen as the reference standard, but it is an invasive method with a morbidity rate of 1–4% that bears a 1% risk of periinterventional stroke (4). Color-flow duplex sonography (CDS) has become the most widely used noninvasive method for assessing extracranial cerebrovascular occlusive disease (5–8), because it avoids the expense and risk of routine arteriography (9, 10). Stenotic lesions are identified and quantified by analyzing Doppler velocity spectra in combination with real-time B-mode and color-flow images of these vessels (11). Several methods have been described to estimate the degree of stenosis on the basis of a number of velocity criteria, but they have shown considerable variation in different publications (5–8, 11–16). It is well recognized that duplex results are highly dependent on the experience of the operator, which underscores the importance of individual evaluation and quality control for each institution (10). Other important disadvantages of CDS include the lack of appropriate data storage and presentation as survey images and the inability of proper quantification in mulitsegmental stenoses (17–19).
Results from preliminary studies reporting observations of different approaches to three-dimensional CDS (3D-CDS) of the carotid arteries have been encouraging (17–27). On the other hand, different limitations—such as the difficult handling of the 3D sensor system, the low quality of the reconstructed images, and the time-consuming process for data reconstruction—have also been reported.
The purpose of this prospective study was therefore to assess the role of 3D-CDS in the evaluation of ICAS. Main objectives were the evaluation of the reconstruction time, the assessment of the interobserver variability in the grading of ICAS, and the correlation of these results with conventional CDS and intraarterial digital subtraction angiography (DSA).
Methods
Thirty-two consecutive patients (19 male and 13 female) with sonographically verified ICAS of 30–99% were taken from an ongoing carotid study, which was approved by the local ethics committee, and included in this prospective study. Mean age was 70 ± 11 years (range, 44–85 years). Sixteen healthy volunteers (controls) were also included and underwent corresponding procedures. Informed consent was obtained from all patients and control subjects. Bilateral CDS of the common carotid artery (CCA), internal carotid artery (ICA), and external carotid artery (ECA) was performed by an experienced vascular technician by using an Acuson 128 XP scanner with a 5-MHz linear probe (Acuson, Mountain View, CA). The transducer was placed in the longitudinal plane parallel to the carotid artery, the flow imaging window was electronically angled 20° vertical, and the color scale was set at 0.31 m/s maximal mean velocity. Velocity waveforms were obtained routinely from the CCA in the center stream approximately 2–3 cm below the bifurcation and from the ICA within the area of maximal stenosis, according to our local standard protocol, by using an angle of insonation between 50° and 60°. The highest peak systolic velocity (PSV) and the end diastolic velocity (EDV) of blood flow in the CCA and ICA were recorded in m/s. On the basis of these values, we calculated the carotid ratio (CR = PSVICA/PSVCCA), PSVICA/EDVCCA, and EDVICA/EDVCCA for each patient. Percentage of ICAS was then staged on the basis of the cut-off points (Table 1) for highest sensitivity and specificity reported in the publication of Nicolaides et al (15). Quality of conditions for the sonographic assessment was classified into three stages: 1) good visualization of CCA and ICA; 2) high bifurcation or extensive kinking or both and therefore ICA only visible on a short track (<3 cm); and 3) extensive calcification.
TABLE 1:
Cut-off points for the quantification of internal carotid artery stenosis by use of CDS according to Nicolaides et al (15) for minimum false-positive and false-negative tests
| Stenosis (%)* | Duplex Velocity Criteria (m/s) |
||||
|---|---|---|---|---|---|
| PSVICA | EDVICA | PSVICA/PSVCCA | PSVICA/EDVCCA | EDVICA/EDVCCA | |
| <47% | <150 | <80 | <2 | <10 | <2.6 |
| 60% | 150–250 | 80–130 | 2–3.2 | 7–10 | <2.6 |
| 70% | 150–250 | >130 | 3.2–4 | 10–15 | 2.6–5.5 |
| 82% | >250 | >130 | >4 | 15–25 | 2.6–5.5 |
| 90% | >250 | >130 | >4 | >25 | >5.5 |
Diameters determined by angiography according to NASCET criteria.
Note.—CDS indicates color Doppler sonography; PSV, peak systolic velocity; EDV, end diastolic velocity.
After CDS, all patients underwent 3D-CDS of the affected side by using a GE Logiq 700 scanner with a 5–10-MHZ linear probe (GE Sonography Europe, Solingen, Germany). Setting was optimized for color filling of the vessel (long persistence, adequate velocity setting, and color gain) and minimizing wall movement and tissue artifacts. Red color was set for arterial flow. An electromagnetic sensor system (3-D Echotech, Munich, Germany), which enables freehand scanning, was used for the data acquisition from extracranial vessels. The sensor is attached to a standard probe to monitor the position in space. The scanner was connected to a personal computer, and video data were transferred via a frame-grabber. 3-D EchoTech software (3D Freescan), a commercially available software, was used to monitor, store, and analyze three-dimensional data. To avoid pulsatility artifacts, electrocardiography gating was used (delay from R was optimized for each patient according to his heart rate), and one image was stored from each cardiac cycle. Maximum time for data acquisition was limited to 200 frames, which is a theoretical maximum of approximately 3 minutes 20 seconds (at a pulse rate of 60 seconds). All patients and volunteers underwent 3D-CDS examination of CCA, ICA, and ECA. The probe was placed in longitudinal plane, and we then scanned the entire volume of the vessels from lateral to medial starting in a first step at the supraclavicular region then followed by the same procedure in a more cephalad position. The first volunteer and the first two patients also underwent a second examination to ensure reproducibility of data reconstruction. Images were reconstructed as color multi-intensity- projection after elimination of grayscale information. The period necessary for complete 3D reconstruction was recorded for each patient. Quality of the reconstructed 3D images was classified into three stages: 1) perfect or almost perfect image with complete color filling and contours of CCA, ICA, and ECA; 2) either incomplete contours or color filling of ECA; and 3) incomplete contours and incomplete color filling of ICA because of kinking or extensive calcification. The second image reconstructions (n = 3) were judged for the presence of corresponding findings from the first scans. Evaluation of ICAS was performed by three independent observers, all blinded to the results of each other and the results of other imaging modalities. The diameter of maximum stenosis in the ICA was measured and compared with the diameter of the normal-appearing ICA distally. Graduation of stenosis was based on the NASCET criteria (3). One of these observers is a specialist in CDS (A.F.), one in DSA (A.D.), and one for both techniques (M.H.).
Additional DSA was performed according to the protocol of the ongoing carotid study, which served as the selection pool for our patients. In total, 23 patients (all 18 with ICAS >70% according to CDS and five with ICAS 30–70%) also underwent DSA. After femoral puncture and selective catheterization of the carotid arteries, 8 mL of contrast medium (Visipaque 240, Amersham, Vienna, Austria) were injected with a flow rate of 7 mL/s and views were taken in at least two planes. The diameter of maximum stenosis in the ICA was measured and compared with the diameter of the normal-appearing ICA distally. Graduation of stenosis was based on the NASCET criteria (3). The observer was blinded to the results of CDS and 3D-CDS. We also evaluated the rate of angiography-related neurologic complications and of complications (bleedings, false aneurysms) at the puncture site.
For statistical analysis, we used SPSS 10.0.5 statistical software (SPSS, Chicago, IL). Numeric values are expressed either as mean ± SD with minimum and maximum in parenthesis or as median with the interquartile range (IQR) in parenthesis. In a first step, images of CDS and 3D-CDS findings were classified as indicating controls (0%), mild and moderate ICAS (25%–70%), and high-grade ICAS (>70%). In a second step, results of CDS, 3D-CDS, and DSA were staged in percentage classes (30–39%, 40–49%, etc). Spearman’s correlation coefficient (r) was used for correlation analysis of percentage stenosis. Kappa statistics were used to state the interobserver correlation for the detection of high-grade ICAS with 3D-CDS. For all tests, a P value <.05 was considered a statistically significant difference.
Results
CDS was performed in all 48 subjects and revealed 16 healthy volunteers, 14 patients with mild or moderate ICAS, and 18 patients with high-grade ICAS. Four patients had ICAS of <47%, five of 50–59%, five of 60–69%, four of 70–82%, and 14 of >82%. Quality of assessment in patients was staged as follows: 1) 12 (37.5%), 2) 10 (31.3%), and 3) 10 (31.2%); as expected, inferior conditions were more common among patients with high-grade ICAS than among those with mild and moderate ICAS (P = .02).
DSA was performed in 23 patients; 14 (60.9%) had high-grade and nine (39.1%) had mild or moderate ICAS. Four patients had ICAS <47%, one of 50–59%, three of 60–69%, seven of 70–82%, and eight of >82%. ICAS percentage calculated on the basis of DSA findings correlated significantly (r = 0.72; P = .01) with those of CDS, although CDS overestimated ICAS in 13 (56.5%) of 23 patients with a median difference of 10% (IQR, 10%). One patient had a false aneurysm at the puncture site, which was treated by sonography-guided compression therapy. No neurologic complications resulted, and no patient died periinterventionally.
3D-CDS was performed in all 48 subjects. Median reconstruction time was 6.4 minutes (IQR, 3.7). As a first step, the reproducibility of data reconstruction was evaluated. In both patients and healthy volunteers, all typical findings of the first assessment and reconstruction could be identified in the second series of both observers in consensus; an example is shown in Figure 1. Results of the quality assessment of reconstructed 3D-CDS were 21 (43.8%) patients in category 1, 11 (22.9%) patients in category 2, and 16 (33.3%) patients in category 3. Results in detail of the assessment of categories (healthy volunteers, 30%–69% and 70%–99%) distributed to each observer for 3D-CDS versus CDS can be seen in Table 2. Kappa values, stating the perfect interobserver correlation for the detection of high-grade ICAS with 3D-CDS, were as follows: 0.9 for observer 1 versus observer 2, and 0.8 for observer 3 versus observer 1 and also versus observer 2. On the basis of these results, we evaluated sensitivity, specificity, positive predictive value, and negative predictive value for each observer by using CDS as the reference standard, which are shown in Table 3. Correlation analysis of the percentage of ICAS of 3D-CDS versus CDS revealed a correlation coefficient of r = 0.85 (P < .001) for observer 1, r = 0.84 (P < .001) for observer 2, and r = 0.85 (P < .001) for observer 3. Median difference compared with CDS was 0% for each observer, with an IQR of 20%, 30%, and 10%. In patients with high-grade ICAS, the quality of reconstructed images was not significantly inferior compared with those of healthy volunteers and those with mild to moderate ICAS (P = .2). Examples of the correlation of 3D-CDS versus DSA in a patient with high-grade ICAS and a patient with moderate ICAS are shown in Figure 2 and 3. Correlation analysis of percentage of ICAS versus DSA revealed a correlation coefficient of r = 0.52 (P = .01), r = 0.7 (P < .001), and r = 0.5 (P = .02). 3D-CDS overestimated ICAS compared with DSA with a median difference of 10% (IQR 20%), 0% (10%), and 0% (30%).
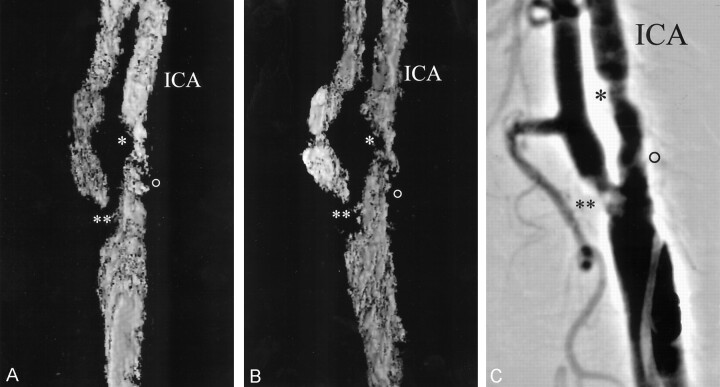
Fig 1.
Evaluation of reconstruction accuracy of 3D-CDS compared with that of DSA in a patient with moderate ICAS.
Note corresponding findings in two reconstructed color (shown here in black and white) multi-intensity-projection 3D images (A and B) and a DSA image (C) indicate excellent reproducibility. The single asterisk denotes moderate ICAS, the double asterisk denotes high-grade external carotid artery stenosis, and the open circle denotes mild ICAS.
TABLE 2:
CDS and 3D-CDS classification of internal carotid artery stenosis in patients and controls determined among three observers
| CDS | 3D-CDS (Observer 1) | 3D-CDS (Observer 2) | 3D-CDS (Observer 3) | |
|---|---|---|---|---|
| Controls | 16 (33.3%) | 10 (20.8%) | 8 (16.7%) | 12 (25%) |
| ICAS, 30–69% | 14 (29.2%) | 22 (45.8%) | 26 (54.2%) | 20 (41.7%) |
| ICAS, 70–99% | 18 (37.5%) | 16 (33.3%) | 14 (29.2%) | 16 (33.3%) |
Note.—CDS indicates color Doppler sonography; 3D-CDS, three-dimensional color Doppler sonography; ICAS, internal carotid artery stenosis.
TABLE 3:
Sensitivity, specificity, positive predictive values, and negative predictive values for the detection of high-grade internal carotid artery stenosis of 3D-CDS (CDS was criterion standard for all three observers)
| Sensitivity | Specificity | Positive Predictive Value | Negative Predictive Value | |
|---|---|---|---|---|
| Observer 1 | 83.3% (15/18) | 96.7% (29/30) | 93.8% (15/16) | 90.6% (29/32) |
| Observer 2 | 77.8% (14/18) | 100% (30/30) | 100% (14/14) | 88.2% (30/34) |
| Observer 3 | 83.3% (15/18) | 100% (30/30) | 100% (15/15) | 90.9% (30/33) |
Note.—CDS indicates color Doppler sonography; 3D-CDS, three-dimensional color Doppler sonography.
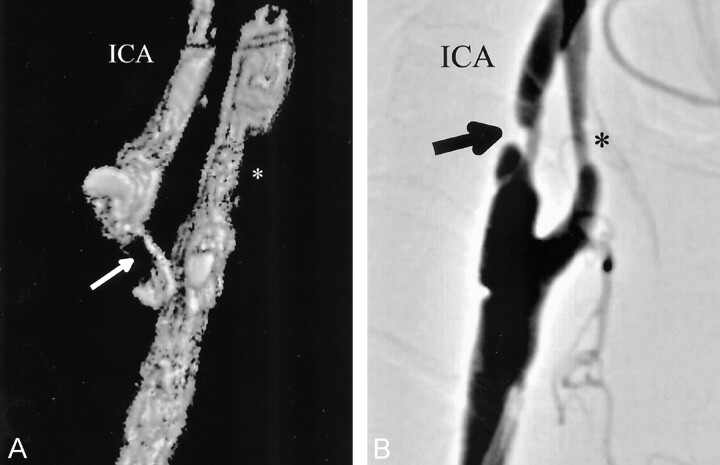
Fig 2.
Comparison of 3D-CDS (A) and DSA (B) in a patient with high-grade (>82%) ICAS. Stenosis is marked with arrows. An asterisk marks mild ECA stenosis
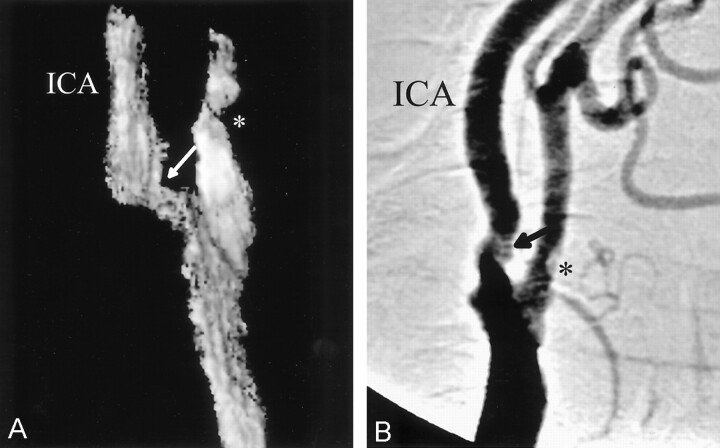
Fig 3.
Comparison of 3D-CDS (A) and DSA (B) in a patient with moderate (50–59%) ICAS. Stenosis is marked with arrows. An asterisk marks mild external carotid artery stenosis
Discussion
The practice of performing carotid endarterectomy without arteriography is a growing trend in vascular surgery and has been shown to be a safe practice, depending heavily on reliable sonography results (13, 20). On the other hand, clinicians caring for patients with carotid artery disease should be aware that limitations of this technique are the high dependency on the experience of the investigator and problems with data presentation and storage (19); however, it is especially the case that the interdisciplinary approach to carotid endarterectomy—involving neurologists, vascular surgeons, angiologists, and radiologists—is highly dependent on the presentation of reliable results of imaging techniques. The preferred form—especially focusing on possible later surgery or stent implantation—is survey images; therefore, MR and CT angiographies are often used in this indication, knowing that they also suffer from different limitations as a long examination time, the need for expensive equipment, and the administration of contrast media (22, 28, 29). Therefore, 3D-CDS has been proposed as a new and noninvasive tool in the investigation of ICAS.
The results of our study indicate that 3D-CDS is an accurate technique for the detection of high-grade ICAS by providing a positive predictive value and specificity that approaches 100%. Median difference between 3D-CDS compared with CDS and DSA was 0%–10%, as assessed by all three observers. The results of the present study correspond well to those published in the literature, where a sensitivity of 90–100% and a specificity of 100% for the detection of high-grade ICAS has been reported (18–20). Our results concerning correlation analysis compared with CDS show inferior results −r = 0.85 in our study compared with r = 0.98 of Keberle et al (18, 19). The main difference compared with the studies of Keberle et al is that we used CDS and 3D-CDS reconstructions compared with power-Doppler sonography (PDS)-based 3D surveys. Advantages of PDS are that it is less dependent on the operator, because results are not influenced by the flow direction and the Doppler angle and the improved visualization of the continuity of blood flow in arterial stenoses (18, 30). On the other hand, the major limitation of PDS is the lack of any information about the hemodynamics in the vessel; thus, PDS and CDS are complementary techniques that should be used together (30). When comparing these facts, especially focusing on later 3D-reconstructed surveys, we decided to rely on 3D-CDS, because hemodynamic as well as morphologic information can be obtained (25). However, correlation to DSA, a technique that only depicts morphology, reveals inferior results. In our study a coefficient of r = 0.5–0.7 was comparable to r = 0.97 reported by Keberle et al (18). The type of statistical analysis used must be considered when presenting and interpreting 3D-CDS surveys. These surveys are especially useful to vascular surgeons for an exact preparation before carotid endarterectomy or stent implantation. 3D-CDS may increase diagnostic confidence, because it provides the possibility for the visualization of the stenosis from different orientations, not limited to a single plane, by rotating the image interactively (17, 21, 26).
The main disadvantage of 3D-CDS is the major problem of all sonography techniques especially compared with DSA—the inability to get signals in patients with severe calcified atherosclerotic plaques and severely kinking vessels. In the present study, about one third of reconstructed images had one of these limitations, but finally it was possible to grade ICAS in all individuals. The most problematic area in our experience was the proximal ECA, whereas ICA was well visualized in nearly every patient. An explanation for the inadequate imaging of the ECA might be the fact that we used an optimized longitudinal probe position for the visualization of the ICA; therefore, the angle of the proximal ECA often approximates 90°. Compared with CDS, 3D-CDS has the advantage that the observer may scan the vessel volume from different insonation positions. The computer software stores all images for the later reconstruction, an important fact, especially in cases of calcification to reduce artifacts from acoustic shadowing (21). This fact was also discussed by Keberle et al (18), who proposed that in all 3D assessments two complete data sets—one from an anterior and one from a lateral view—should be obtained to minimize possible filling defects. With this approach, they achieved satisfactory quality in more than 90% of reconstructed images. In the present study, we obtained—except those cases where date reproducibility was evaluated—only one data set. As stated above, however, the distal CCA, the carotid bulb, and the ICA were visible in all patients.
A further limitation is the differing lumen diameter of the carotid arteries in the cardiac cycle, which leads to motion artifacts, because of curved vessel contours and which also occurs with the use of MR and CT. In the present study, we solved this problem by using electrocardiography gating to ensure the best possible reconstruction quality. One problem of 3D-CDS that has been reported in the literature could be disproved by the current study: the time-consuming process of data acquisition and reconstruction (21). In our trial, data acquisition time was limited to 200 frames at one frame per cardiac cycle. Median reconstruction time was 6.4 minutes, so the time needed for a complete 3D data set was about 10 minutes, in our opinion a sound time investment in view of the advantages of 3D surveys as stated above.
Conclusion
3D-CDS is an accurate and reliable method for the detection of high-grade ICAS, providing a positive predictive value and sensitivity approaching 100%. Three-dimensional data sets can be acquired and reconstructed within 10 minutes, giving the opportunity for complete data storage and the presentation as 3D volume surveys, facts that are especially essential in the interdisciplinary approach to carotid endarterectomy or stent implantation.
References
- 1.Toole JF, Castaldo JE. Accurate measurement of carotid stenosis. J Neuroimaging 1994;4:222–230 [DOI] [PubMed] [Google Scholar]
- 2.European Carotid Surgery Trialists Collaborative Group. MRC European Carotid Surgery Trial: interim results for symptomatic patients with severe (70–99%) or with mild (0–29%) carotid stenosis. Lancet 1991;337:1235–1243 [PubMed] [Google Scholar]
- 3.North American Symptomatic Carotid Endarterectomy Trial Collaboratiors. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Eng J Med 1991;325:445–453 [DOI] [PubMed] [Google Scholar]
- 4.Asymptomatic Carotid Atherosclerosis Study Collaborators. Endarterectomy for asymptomatic carotid artery stenosis. JAMA 1995;273:1421–1428 [PubMed] [Google Scholar]
- 5.Staikov IN, Arnold M, Mattle HP, et al. Comparison of the ECST, CC and NASCET grading methods and ultrasound for assessing carotid stenosis. J Neurol 2000;247:681–686 [DOI] [PubMed] [Google Scholar]
- 6.Hood DB, Mattos MA, Mansour A, et al. Prospective evaluation of new duplex criteria to identify 70% internal carotid artery stenosis. J Vasc Surg 1996;23:254–262 [DOI] [PubMed] [Google Scholar]
- 7.Hunink MGM, Polak JF, Barlan MM, O’Leary DH. Detection and quantification of carotid artery stenosis: efficacy of various doppler velocity parameters. AJR Am J Roentgenol 1993;160:619–625 [DOI] [PubMed] [Google Scholar]
- 8.Padayachee TS, Cox TCS, Modaresi KB, et al. The measurement of internal carotid artery stenosis: comparison of duplex with digital subtraction angiography. Eur J Vasc Endovasc Surg 1997;13:180–185 [DOI] [PubMed] [Google Scholar]
- 9.Bell PRF. Preoperative angiography is outdated. BMJ 1995;310:1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Curley PJ, Norrie L, Nocholson A, et al. Accuracy of carotid duplex is laboratory specific and must be determined by internal audit. Eur J Vasc Endovasc Surg 1998;15:511–514 [DOI] [PubMed] [Google Scholar]
- 11.Carpenter JP, Lexa FJ, Davis JT. Determination of sixty percent or greater carotid artery stenosis by duplex Doppler ultrasonography. J Vasc Surg 1995;22:697–705 [DOI] [PubMed] [Google Scholar]
- 12.Carpenter JP, Lexa FJ, Davis JT. Determination of duplex Doppler ultrasound criteria appropriate to the North American Symptomatic Carotid Endarterectomy Trial. Stroke 1996;27:695–699 [DOI] [PubMed] [Google Scholar]
- 13.Chen JC, Salvian AJ, Taylor DC, et al. Predictive ability of duplex ultrasonography for internal carotid artery stenosis of 70%–99%: a comparative study. Ann Vasc Surg 1998;12:244–247 [DOI] [PubMed] [Google Scholar]
- 14.Moneta GL, Edwards JM, Chitwood RW, et al. Correlation of North American Symptomatic Carotid Endarterectomy Trial (NASCET) angiographic definition of 70% to 99% internal carotid artery stenosis with duplex scanning. J Vasc Surg 1993;17:152–159 [DOI] [PubMed] [Google Scholar]
- 15.Nicolaides AN, Shifrin EG, Bradbury A, et al. Angiographic and duplex grading of internal carotid stenosis: can we overcome the confusion? J Endovasc Surg 1996;3:158–165 [DOI] [PubMed] [Google Scholar]
- 16.Winkelaar GB, Chen JC, Salvian AJ, et al. New duplex ultrasound scan criteria for managing symptomatic 50% or greater carotid stenosis. J Vasc Surg 1999;29:986–994 [DOI] [PubMed] [Google Scholar]
- 17.Gou Z, Fenster A. Three-dimensional power Doppler imaging: a phantom study to quantify vessel stenosis. Ultrasound Med Biol 1996;22:1059–1069 [DOI] [PubMed] [Google Scholar]
- 18.Keberle M, Jenett M, Wittenberg G, et al. Comparison of 3D-power-Doppler-ultrasound, color doppler ultrasound, and digital subtraction angiography in carotid artery disease [in German]. Rofo Fortschr Röntgenstr 2001;173:133–138 [DOI] [PubMed] [Google Scholar]
- 19.Keberle M, Jenett M, Beissert M, et al. Three-dimensional power Doppler sonography in screening for carotid artery disease. J Clin Ultrasound 2000;28:441–451 [DOI] [PubMed] [Google Scholar]
- 20.Bendick PJ, Brown W, Hernandez D, Glover JL, Bove PG. Three-dimensional vascular imaging using Doppler ultrasound. Am J Surg 1998;176:183–187 [DOI] [PubMed] [Google Scholar]
- 21.Campani R, Bottinelli O, Calliada F, Coscia D. The latest in ultrasound: three-dimensional imaging. Part II. Eur J Radiol 1998;27:183–187 [DOI] [PubMed] [Google Scholar]
- 22.Delcker A, Schürks M, Polz H. Development and applications of 4-D ultrasound (dynamic 3-D) in neurosonology. J Neuroimaging 1999;9:229–234 [DOI] [PubMed] [Google Scholar]
- 23.Downey DB, Fenster A. Vascular imaging with a three-dimensional power Doppler system. AJR Am J Roentgenol 1995;165:665–668 [DOI] [PubMed] [Google Scholar]
- 24.Griewing B, Schminke U, Morgenstern C, et al. Three-dimensional ultrasound angiography (power mode) for the quantification of carotid artery atherosclerosis. J Neuroimaging 1997;7:40–45 [DOI] [PubMed] [Google Scholar]
- 25.Delcker A, Diener C. Quantification of atherosclerotic plaques in carotid arteries by three-dimensional ultrasound. Br J Radiol 1994;67:672–678 [DOI] [PubMed] [Google Scholar]
- 26.Pretorius DH, Nelson TR, Jaffe JS. 3-dimensional sonographic analysis based on color flow Doppler and gray scale image data: a preliminary report. J Ultrasound Med 1992;11:225–232 [DOI] [PubMed] [Google Scholar]
- 27.Trattnig S, Braunschweig R, Fleiter T. Dynamic 3-D reconstruction of peripheral vessels via colour-coded Doppler sonography [in German]. Ultraschall Med 1993;14:85–89 [DOI] [PubMed] [Google Scholar]
- 28.Wetzel S, Bongartz G. MR angiography: supraaortic vessels. Eur J Radiol 1999;9:1277–1284 [DOI] [PubMed] [Google Scholar]
- 29.Leclerc X, Godefroy O, Lucas C, et al. Internal carotid arterial stenosis: CT angiography with volume rendering. Radiology 1999;210:673–682 [DOI] [PubMed] [Google Scholar]
- 30.Steinke W, Ries S, Artemis N, Schwartz A, Hennerici M. Power Doppler imaging of carotid artery stenosis: comparison with color Doppler flow imaging and angiography. Stroke 1997;28:1981–1987 [DOI] [PubMed] [Google Scholar]