Abstract
BACKGROUND AND PURPOSE: Endovascular therapy is increasingly being used for the treatment of unruptured intracranial aneurysms. Our purpose was to determine the risk of adverse outcomes after contemporary endovascular treatment of unruptured intracranial aneurysms in the United States. Patient, treating physician, and hospital characteristics were tested as potential outcome predictors, with particular attention paid to volume of care.
METHODS: We conducted a retrospective cohort study by using the Nationwide Inpatient Sample, 1996–2000. Multivariate logistic and ordinal regressions were used with end points of mortality, discharge other than to home, length of stay, and total hospital charges.
RESULTS: Four hundred twenty-one patients underwent endovascular treatment at 81 hospitals. The in-hospital mortality rate was 1.7%, and 7.6% were discharged to institutions other than home. Analysis was adjusted for age, sex, race, primary payer, year of treatment, and four variables measuring acuity of treatment and medical comorbidity. Median annual number of unruptured aneurysms treated was nine per hospital and three per treating physician. Higher volume hospitals had fewer adverse outcomes; discharge other than to home occurred after 5.2% of operations at high volume hospitals (>23 admissions per year) compared with 17.6% at low volume hospitals (fewer than four admissions per year) (P < .001). Higher physician volume had a similar effect (0% versus 16.4%, P = .03). The mortality rate was lower at high volume hospitals (1.0% versus 3.7%) but not significantly so. At high volume hospitals, length of stay was shorter (P < .001) and total hospital charges were lower (P < .001).
CONCLUSION: For patients with unruptured aneurysms treated in the United States from 1996 to 2000, endovascular treatment at high volume institutions or by high volume physicians was associated with significantly lower morbidity rates and modestly lower mortality rates. Length of stay was shorter and total hospital charges lower at high volume centers.
A growing body of work suggests that morbidity and mortality rates are lower when complex medical or surgical procedures are performed at high volume centers or by high volume providers (1, 2). For example, in-hospital mortality rates are lower when complex cancer operations (3, 4), cardiovascular operations (5), or peripheral vascular operations (6) are performed at high volume hospitals. Lower rates of adverse outcomes have been documented after surgical repair of unruptured intracranial aneurysms by high volume physicians (7) or at high volume hospitals (8, 9). Overall management adverse outcomes after subarachnoid hemorrhage also are lower at high volume centers (8, 10, 11). However, the volume-outcome relationship for endovascular treatment of unruptured aneurysms has not previously been investigated.
For the present study, we used a national hospital discharge database to study adverse outcomes (in-hospital morbidity and mortality rates) after endovascular treatment of unruptured intracranial aneurysms in relation to patient, treating physician, and hospital characteristics. Specifically, we hypothesized that outcome after endovascular treatment of unruptured aneurysms would be related to hospital and physician case load. We evaluated length of hospital stay and total hospital charges as additional end points.
Methods
We obtained the Nationwide Inpatient Sample (NIS) hospital discharge database for the years 1996 through 2000 from the Healthcare Cost and Utilization Project, Agency for Healthcare Research and Quality, Rockville, MD (12). The NIS is a hospital discharge database that represents approximately 20% of all inpatient admissions to nonfederal hospitals in the United States. For these years, the NIS contains discharge data regarding 100% of discharges from a stratified random sample of nonfederal hospitals in 19 to 28 states to produce a representative 20% subsample of all United States nonfederal hospital discharges. Because the NIS contains data regarding all patients discharged from sampled hospitals during the year regardless of age or payer, it can be used to obtain the annual total volume of specified procedures at individual hospitals. For many states, a code identifying the physician who performed the principal procedure is also included. An overview of the NIS is available at http://www.ahcpr.gov/data/hcup/nisintro.htm.
Inclusion and Exclusion Criteria and Definition of End Points
Five hundred thirty-five discharges were associated with a primary diagnosis of “unruptured intracranial aneurysm” (International Classification of Diseases [ICD]-9-CM code 437.3) and a primary procedure code of “other repair of aneurysm” (ICD-9-CM code 39.52). We excluded 20 patients (3.7%) with a diagnosis of “subarachnoid hemorrhage” (ICD-9-CM code 430) from the cohort, leaving 515 patients potentially eligible for analysis.
The ICD-9-CM code we used to identify endovascular treatment is also sometimes used for open surgical aneurysm treatment other than clipping, such as wrapping, suture, coating with methylmethacrylate, or filipuncture. A validation study that used the University Health System Consortium database suggested that the number of nonendovascular treatments described by using ICD-9-CM code 39.52 was relatively small (13). NIS data contain no independent verification of the nature of the procedure, such as a Current Procedural Terminology code for the principal procedure, so we used patient, treating physician, and hospital characteristics to attempt to remove nonendovascular treatments from the cohort before analysis, as follows. For 214 patients (42%), the database contained a code identifying the physician who performed the procedure. We assumed that procedures coded as 39.52 that were performed by a physician who also performed diagnostic arteriography, noncoronary angioplasty or carotid stent placement, Wada test, or infusion of thrombolytic agents for stroke were actual endovascular treatments. We assumed that procedures coded as 39.52 that were performed by physicians who had performed none of the above procedures that year were actually open procedures. We used these 214 patients, 154 (72%) of whom seemed to have received actual endovascular treatment, as the “test sample” to develop a multivariate model that distinguished cases with a high likelihood of being actual endovascular cases from other cases. The model contained the annual hospital volume of procedures coded as 39.52 (>/<3), whether a diagnostic arteriogram was coded during the admission, the medical comorbidity score, the year of treatment, and the type of admission (emergency, urgent, or elective). The model correctly classified 81% of test sample cases. For cases without identified treating physicians, we used the model to choose which cases would be included in the analysis; we used an estimated P ≥ .5 as the cutoff point to determine whether a case was endovascular and would be included in the study (248 of 301 cases included, 82%). In all, 421 cases (82%) were included in the analysis.
Two primary end points were examined in this study: in-hospital mortality and discharge to institutions other than home. In-hospital mortality was coded directly in the NIS database and was analyzed by using logistic regression. Discharge to institutions other than home was coded on a four-level scale and was analyzed by using ordinal logistic regression, which allows use of the entire spectrum of outcomes rather than simplifying to a single cutoff point with resultant loss of information (14–16). Discharge was coded as death, discharge to a long-term facility, discharge to other facilities, or discharge to home, as follows. NIS data distinguish discharge to long-term facilities (such as skilled nursing facilities) from discharge to intermediate care facilities or other facilities for all states except California and Maryland; for those states, we coded these discharges (0.8% of the total) as discharge to “other facilities.” We counted discharge to home “with home health care” (3.1% of discharges) as discharge to home. Discharge to another acute care hospital was counted as discharge to an institution other than home, not as discharge to a long-term facility.
Length of stay and total hospital charges were coded in NIS data. Ten cases with length of stay of zero (1.9%) were recoded as missing. Length of stay and hospital charge analyses included only patients discharged from hospital alive. Length of stay and hospital charge data were both strongly positively skewed and were analyzed as the logarithmic transforms.
Patient Characteristics
Patient age, sex, race, median income for ZIP code of residence, primary payer (Medicare, Medicaid, private insurance, self-pay, no charge, other), type of admission (emergency, urgent, elective), and admission source (emergency room, transfer from another hospital, transfer from long-term care, and routine) were coded in NIS data. Two cases (0.5%) with admission type coded as “other” were recoded as routine admissions. More than 5% of discharges had missing values for three variables used principally as stratification factors for other analyses, race (27% missing), admission type (28% missing), and whether the principal procedure was performed on the first hospital day (12% missing). When these variables were used as stratification factors, missing values for race and admission type were imputed as follows. Missing race was set to white. Missing admission type was set to “emergency” for admissions whose source was the emergency room, to “urgent” for admissions that were transfers from another hospital, and to “routine” for admissions from other sources. Whether the principal procedure was performed on the first hospital day was not imputed. Sensitivity analyses confirmed that the imputations for race and admission type had no significant effect on the main analyses described below.
To assess the effect of general medical comorbidity, the set of 30 medical comorbidity markers described by Elixhauser et al (17), excluding the two specific neurologic comorbidity variables (“paralysis” and “other neurologic deficit”), were calculated by using Agency for Healthcare Research and Quality software (www.ahcpr.gov/data/hcup/comorbid.htm) and summed to render a single comorbidity score with possible values between 0 and 28. We identified potential markers of specific adverse outcomes of care for patients with unruptured aneurysms that occurred in at least 2% of the 421 eligible patients: postoperative neurologic complications, including infarction or hemorrhage (ICD-9-CM codes 997.00–997.09); occlusion of acerebral artery, with or without infarction (ICD-9-CM codes 434.0–434.9); hydrocephalus (ICD-9-CM codes 331.3–331.4) or the performance of a ventriculostomy (ICD-9-CM code 02.2); placement of an endotracheal tube (ICD-9-CM code 96.04) or mechanical ventilation (ICD-9-CM codes 96.70–96.72); performance of a gastrostomy (ICD-9-CM codes 43.11–43.19); and hematoma complicating a procedure (ICD-9-CM codes 998.1–998.13).
Provider and Hospital Characteristics
Hospital region (Northeast, Midwest, South, or West), location (rural or urban), teaching status, and bed size (small, medium, large) were coded in NIS data. We derived hospital and physician volumes of surgical (“clipping”) or endovascular (“coiling”) treatment of unruptured intracranial aneurysms for each year by counting the cases for each identified treating physician and hospital in the database with the primary procedures coded as 39.51 and 39.52, respectively. We also counted the total number of aneurysms clipped, whether ruptured or unruptured, for each hospital and treating physician by combining the number of discharges with primary or secondary procedure codes of 39.51 with a primary diagnosis of 430 (subarachnoid hemorrhage) or 437.3 (“unruptured cerebral aneurysm”) in the NIS database for each year. Hospital volumes of endovascular treatment of aneurysms were calculated in the same way, using procedure code 39.52 (“other aneurysm repair”) instead of 39.51. Because all measures of hospital and physician volume were positively skewed, the logarithmic transforms were used when measures of volume were entered into regression models.
Statistical Methods
Statistical methods included Fisher’s exact test, the Wilcoxon rank test, and Spearman rank correlation and loglinear, ordinary logistic, and proportional-odds ordinal logistic regression (18–20). To adjust for possible clustering of similar outcomes within hospitals, which could cause falsely inflated estimates of the statistical significance of regression coefficients in the regression analyses, a sandwich variance-covariance matrix was estimated from the data by using Huber-White variance estimator methods, with adjustment for clustering by hospital (20). The main analysis was also repeated by using a bootstrap-estimated variance-covariance matrix with cluster-based resampling (20) and with a hierarchical ordinal logistic regression model (21); results were similar to those obtained by using the other method and are not separately reported. “Optimal” cutoff points for hospital and treating physician volume were chosen by examining all possible cutoff points and choosing the one that minimized the P value for the statistical model. Because the P values for cutoff points so chosen are spuriously low, a correction factor was applied (22, 23), with 5% of potential cutoff points omitted at either end of the data range. For confidence intervals for the cutoff points, the cutoff point calculation was bootstrapped (1000 repetitions) and the 2.5 and 97.5 percentile values for the cutoff points were chosen (24). Length of stay and hospital charges were analyzed as logarithmic transforms by using least-squares regression corrected for clustering, as described above. Calculations were performed by using SAS (version 8.2; SAS Institute, Cary, NC), S-plus (version 3.3 for Windows; Insightful, Inc., Seattle, WA) with the Hmisc and Design modeling function software libraries presented by Harrell (20, 25), and HLM 5 (version 5.04; Scientific Software International, Lincolnwood, IL). All P values are two-tailed.
Results
Four hundred twenty-one patients who had been treated at 81 hospitals by 75 identified physicians were included in the analysis. Clinical characteristics are summarized in Table 1. Most patients were white, female, between 45 and 65 years old, and had admissions classified as routine. Seven patients died (1.7%; 95% confidence interval, 0.7% to 3.4%), 10 were discharged to skilled nursing facilities (2.4%; 95% confidence interval, 1.1% to 4.3%), 22 were discharged to other facilities (5.2%; 95% confidence interval, 3.3% to 7.8%), and 382 were discharged to home (90.7%; 95% confidence interval, 87.6% to 93.3%).
TABLE 1:
Clinical characteristics of 421 patients who underwent endovascular treatment of unruptured intracranial aneurysms
| Age (yr) (n = 420) | |
| Mean | 56 |
| Median | 56 |
| Interquartile range | 47–67 |
| Range | 15–88 |
| Female sex (n = 421) | 326 (77%) |
| Race (n = 307) | |
| White | 237 (77%) |
| Black | 24 (8%) |
| Hispanic | 22 (7%) |
| Asian/Pacific Islands | 10 (3%) |
| Native American | 3 (1%) |
| Other | 11 (4%) |
| Median household income for ZIP code of residence (n = 405) | |
| <$25,000 | 36 (9%) |
| $25,000–$34,999 | 117 (29%) |
| $35,000–$44,999 | 122 (30%) |
| >$45,000 | 130 (32%) |
| Primary payer (n = 421) | |
| Medicare | 113 (27%) |
| Medicaid | 28 (7%) |
| Private insurance | 257 (61%) |
| Self-pay | 6 (1%) |
| No charge | 2 (0.5%) |
| Other | 15 (4%) |
| Admission type (n = 279) | |
| Emergency | 19 (7%) |
| Urgent | 42 (15%) |
| Routine | 218 (78%) |
| Admission source (n = 417) | |
| Emergency ward | 7 (2%) |
| Transfer from acute care hospital | 21 (5%) |
| Transfer from long term care | 11 (3%) |
| Routine | 378 (91%) |
| Year of treatment | |
| 1996 | 30 (7%) |
| 1997 | 82 (19%) |
| 1998 | 57 (14%) |
| 1999 | 125 (30%) |
| 2000 | 127 (30%) |
Patient Characteristics and Outcome
We examined six demographic variables as predictors of outcome: age, sex, race, primary payer, year of treatment, and median household income in the ZIP code of the patient’s residence (Table 2). Older age significantly predicted both death and discharge other than to home (P = .002 for both). Both female sex and private insurance as the primary payer for care were associated with significantly lower odds of discharge other than to home (P = .01 for both). No other demographic variable was significantly associated with both primary end points. Age, sex, race (white/nonwhite), primary payer (private insurance/other), hospital region, and year of treatment were included in all models described below.
TABLE 2:
Effect of demographic variables on outcome
| Odds of Death | Odds of Worse Outcome at Hospital Discharge (Four-Level Scale) | |
|---|---|---|
| Age (per decade) | 2.04 (1.29–3.24) | 1.58 (1.19–2.11) |
| P = 0.002 | P = 0.002 | |
| Female gender | 0.21 (0.06–0.76) | 0.41 (0.21–0.80) |
| P = 0.02 | P = 0.01 | |
| Race | ||
| White versus nonwhite | 1.49 (0.14–15) | 1.95 (0.76–5.00) |
| P = 0.7 | P = 0.16 | |
| Black versus nonblack | (No deaths)* | 0.39 (0.05–2.82) |
| P = 0.4 | ||
| Primary payer | ||
| Medicaid versus other | (No deaths)* | 2.22 (0.89–5.58) |
| P = 0.09 | ||
| Private insurance versus other | (No deaths)* | 0.36 (0.16–0.80) |
| P = 0.01 | ||
| Year of treatment | 1.005 (0.53–1.88) | 0.83 (0.62–1.11) |
| P = 0.99 | P = 0.2 | |
| Median income in ZIP code of residence | 0.64 (0.33–1.25) | 0.72 (0.52–1.00) |
| P = 0.2 | P = 0.05 |
Regression coefficients for these variables were undefined because all deaths occurred in one subgroup.
Four variables related to acuity of care and general comorbidity were examined: admission type (emergency, urgent, elective); admission source (emergency room, transfer from another hospital, routine); whether the endovascular procedure was performed on the day of admission; and a general medical comorbidity score. These variables were tested in models including a multivariate adjustment for age, sex, race, primary payer, and year of treatment. Admission type and source and whether the procedure was performed on the first hospital day were significant (P < .05) in models predicting discharge other than to home, and a trend toward significance for the summed comorbidity score also was observed (P = .07) (Table 3). None of these variables were significant predictors of mortality.
TABLE 3:
Effect of variables reflecting acuity and comorbidity on outcome
| Odds of Death | Odds of Worse Outcome at Hospital Discharge (Four-Level Scale) | |
|---|---|---|
| Admission type | ||
| (Emergency versus urgent versus elective) | 1.38 (0.35–5.42) | 1.84 (1.10–3.08) |
| P = 0.6 | P = 0.02 | |
| Admission source | ||
| Emergency room versus routine | (No deaths)* | 4.90 (1.66–14.4) |
| P = 0.009 | ||
| Transfer from another hospital versus routine | (No deaths)* | 2.85 (0.89–9.1) |
| P = 0.08 | ||
| Surgery not done on day of admission to hospital | 2.11 (0.33–13.5) | 4.87 (2.08–11.4) |
| P = 0.4 | P < 0.001 | |
| Summed comorbidity score (per point) | 1.08 (0.58–2.02) | 1.26 (0.98–1.61) |
| P = 0.8 | P = 0.07 |
Regression coefficients for these variables were undefined because all deaths occurred in one subgroup.
Hospital and Treating Physician Characteristics and Outcome
Patients were treated at 81 hospitals. For 173 patients (41% of the total), 75 treating physicians were identified in the database. Hospitals and physicians varied considerably in the volume of unruptured and ruptured aneurysms treated annually. Analyzed on a per-patient basis, the median number of unruptured aneurysms to undergo endovascular treatment that year was nine per hospital (range, 1–61 aneurysms) or three per physician (range, 1–9 aneurysms). Counting both ruptured and unruptured aneurysms, the median number treated per hospital was 15 (range, 1–115 aneurysms) and the median per physician was three (range, 1–13 aneurysms). (Hospital treatment volumes seem disproportionately larger than physician volumes because the largest volume hospitals were located in states that did not provide treating physician identifier codes.) For 52 patients (12%), no other endovascular treatment of unruptured aneurysms was reported during that year at their hospital, and for 55 patients (32%), no other unruptured aneurysm was reported treated that year by their treating physician. Hospitals had substantial volumes of aneurysms clipped and substantial volumes of endovascular services provided. Per hospital, the median annual number of unruptured aneurysms clipped was 15 (range, 0–77 aneurysms) and the median total number of ruptured and unruptured aneurysms clipped was 45 (range, 0–127 aneurysms) in addition to the above-described endovascular treatments. Table 4 shows clinical characteristics of patients treated at hospitals in the lowest and highest volume quartiles for volume of endovascular treatment of unruptured aneurysms (one to three cases per year and >24 cases per year, respectively).
TABLE 4:
Patient characteristics in high- and low-volume hospitals
| Low Volume (1–3/yr) | High Volume (>24/yr) | |
|---|---|---|
| Age (yr) (median) | 58 | 54 |
| Female | 82/108 (76%) | 75/96 (78%) |
| Race | ||
| White | 70/86 (81%) | 68/96 (71%) |
| Black | 9/86 (10%) | 9/96 (10%) |
| Primary payer | ||
| Medicare | 34/108 (31%) | 26/96 (27%) |
| Medicaid | 8/108 (8%) | 9/96 (9%) |
| Private insurance | 57/108 (53%) | 57/96 (59%) |
| Median income in ZIP code of residence | ||
| <$25,000 | 13/105 (12%) | 12/89 (13%) |
| $25,000–$34,999 | 33/105 (31%) | 27/89 (28%) |
| $35,000–$44,999 | 23/105 (22%) | 28/89 (31%) |
| >$45,000 | 36/105 (34%) | 22/89 (25%) |
| Rural versus urban (hospital location used as proxy) | ||
| Rural | 3/108 (3%) | 0/96 (0%) |
| Admission type | ||
| Emergency | 6/101 (6%) | 0/96 (0%) |
| Urgent | 17/101 (17%) | 4/96 (4%) |
| Routine | 78/101 (77%) | 92/96 (92%) |
| Admission source | ||
| Emergency ward | 3/104 (3%) | 0/96 (0%) |
| Transfer from acute care hospital | 4/104 (4%) | 4/96 (4%) |
| Transfer from long-term care | 8/108 (7%) | 1/96 (1%) |
| Routine | 89/108 (82%) | 91/96 (95%) |
We tested the effects of hospital and physician volume of intracranial aneurysm treatment on outcome in several ways: by hospital or physician volume of unruptured aneurysm coiling or any aneurysm coiling, by total hospital aneurysm treatment volume (clipping plus coiling), and by hospital clipping volume (unruptured or any aneurysm) (Table 5). Only one volume measure was significantly related to in-hospital mortality: total hospital volume of unruptured aneurysm care (coiling plus clipping). The odds ratio for mortality associated with a 10-fold increase in hospital volume of care (approximately the difference between the highest and lowest volume quartiles) was 0.20 (95% confidence interval, 0.04 to 0.91; P = .04).
TABLE 5:
Effect of hospital and physician volume on outcome
| Odds of Worse Outcome at Hospital Discharge (Four-Level Scale) | |
|---|---|
| Hospital volume-unruptured aneurysm coiling | 0.28 (0.14–0.54) P < 0.001 |
| Hospital volume-all aneurysm coiling | 0.34 (0.18–0.63) P < 0.001 |
| Hospital volume-unruptured aneurysm, coiling or clipping | 0.29 (0.11–0.77) P = 0.01 |
| Hospital volume-all aneurysm, coiling or clipping | 0.34 (0.12–0.95) P = 0.04 |
| Physician volume-unruptured aneurysm coiling | 0.11 (0.02–0.80) P = 0.03 |
| Physician volume-all aneurysm coiling | 0.10 (0.02–0.63) P = 0.01 |
| Hospital clipping-unruptured aneurysm volume | 0.49 (0.13–1.84) P = 0.3 |
| Hospital clipping-any aneurysm volume | 0.51 (0.14–1.88) P = 0.3 |
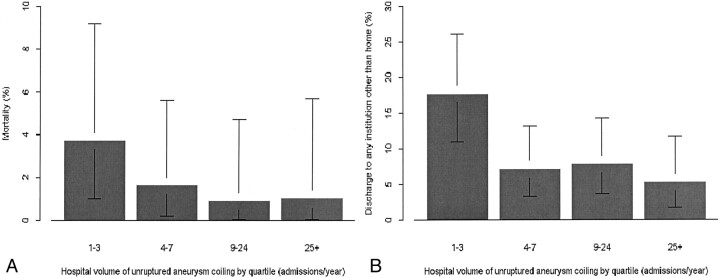
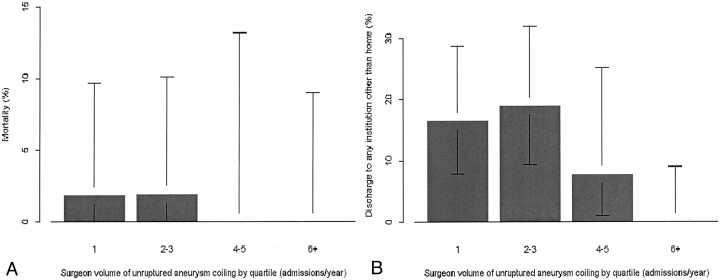
For all measures of volume of endovascular treatment of aneurysms, whether for hospital or physician, a significant relationship was observed between higher volume of cases and better outcome for discharge other than to home. For hospital volume of endovascular treatment of aneurysms, whether unruptured aneurysms only or both ruptured and unruptured aneurysms, the correlation was highly significant (P < .001 for both, with odds ratios of 0.28 and 0.34, respectively, for a 10-fold increase in volume). Figure 1 shows the relationship between volume and outcome (mortality or discharge other than to home) for hospital volume of unruptured aneurysm treatment, and Figure 2 shows the relationship between treating physician volume of unruptured aneurysm treatment and outcome. Measures of hospital surgical clipping volume were not significantly related to outcome for patients who underwent endovascular treatment.
Fig 1.
Bar graphs show probability of in-hospital mortality (A) and discharge other than to home (B) in relation to hospital volume of endovascular treatment of unruptured intracranial aneurysms, by quartile. Error bars, 95% confidence intervals.
Fig 2.
Bar graphs show probability of in-hospital mortality (A) and discharge other than to home (B) in relation to treating physician volume of endovascular treatment of unruptured intracranial aneurysms, by quartile. Error bars, 95% confidence intervals.
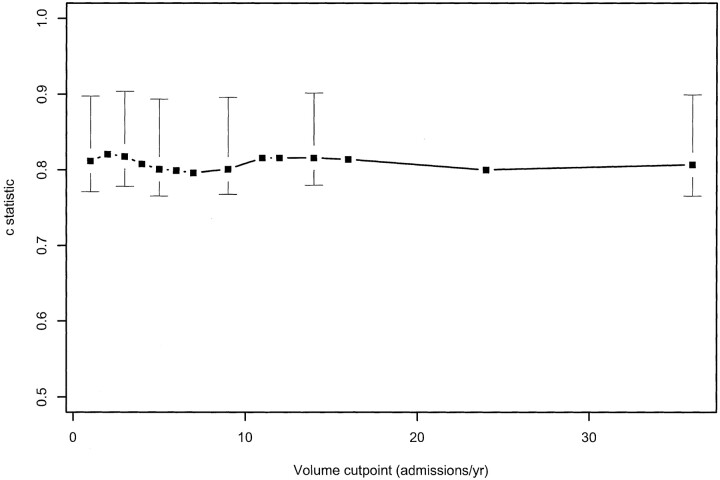
We tested whether a two-tier volume variable (ie, classifying hospitals as high or low volume) was supported by the data by choosing a statistically optimal cutoff point and testing its significance. Based on univariate analysis, hospitals where more than two unruptured aneurysms were coiled annually had better outcomes (odds ratio, 0.31; adjusted P = .03). Based on multivariate analysis, the optimal cutoff point was three endovascular treatments per year (adjusted P = .07), with a 95% confidence interval for the cutoff point of one to 36 endovascular procedures per year (the entire range of the data). Figure 3 shows the c statistic (a generalization of the area under the receiver operating characteristic curve that is uniquely defined for the ordinal logistic regression model) for all possible outcome cutoff point, with 95% confidence intervals (18). Because the c statistic for every cutoff point is within the 95% confidence interval for the “best” cutoff point, the data failed to support the existence of a single caseload under which patient outcomes are evidently worse. No single cutoff point was statistically significant for physician volume (annual volume of endovascular treatment of unruptured aneurysms).
Fig 3.
c statistic (a generalization of the area under the receiver operating characteristic curve for ordinal models) plotted against cutoff points to divide high volume providers from low volume providers. The c statistic for all possible cutoff points is within the 95% confidence interval for the best cutoff point, indicating that the data fail to support a unique caseload value under which patient outcomes are evidently inferior.
We examined three hospital characteristics as potential predictors of outcome (discharge other than to home): hospital bed size, teaching status, and location (rural versus urban). Hospital size was classified as large for 87% of patients, medium for 8%, and small for 5%. Most endovascular procedures (93%) were performed at a teaching hospital, and 99% were performed at an urban hospital. After adjustment for hospital volume of unruptured aneurysm surgery, none of these three factors was a statistically significant predictor of outcome (P > .8 for all).
We examined several types of adverse outcome for correlation with hospital volume. None of the following clinical diagnoses or procedures was significantly correlated with hospital volume of unruptured aneurysm clipping: postoperative neurologic complication (coded in 5.0% of patients), occlusion of a cerebral artery (8.8%), hydrocephalus (2.6%), or hematoma (3.8%). Both ventriculostomy (3.3%, P = .02) and postoperative ventilation (3.3%, P < .001) were more common at low volume hospitals. (All of these diagnoses except occlusion of a cerebral artery were highly significant predictors of discharge other than to home in multivariate models, and ventriculostomy and hydrocephalus were significant predictors of mortality.)
Access to High Volume Hospitals and Physicians
We tested demographic variables as possible predictors of access to high volume hospitals or physicians. Patients from higher income areas of residence were treated at higher volume hospitals. The median hospital volume of endovascular treatment for patients from the two higher quartiles of median income in ZIP code of residence was nine, versus seven for those from the two lowest quartiles (P = .045). Using hospital location as a surrogate for patient location, rural patients were treated at significantly lower volume hospitals (median hospital volume, rural versus urban, one versus 9; P = .008). Teaching status was also a highly significant predictor of larger hospital volume of endovascular unruptured aneurysm treatments (P < .001). The following variables were not significantly correlated with treating hospital volume of care: patient age, sex, race (coded as white versus nonwhite or as black versus nonblack), and primary payer (coded as Medicaid versus other or as private insurance versus other). No variable examined was significantly correlated with treating physician volume of care.
Length of Stay and Hospital Charges in Relation to Hospital Volume
We investigated secondary end points of length of stay and total hospital charges in relation to hospital volume of endovascular treatment of unruptured aneurysms. These analyses included only 294 patients with routine elective admissions who underwent their procedure on the first hospital day and were discharged alive. Because length of stay decreased during the study period (P = .09) and hospital charges increased (P < .001), length of stay and hospital charge analyses were stratified by year of treatment in addition to the demographic, acuity, and comorbidity variables described above.
Length of stay was significantly shorter at high volume hospitals (P < .001). Median length of stay was 3 days for hospitals with fewer than eight annual endovascular procedures and 2 days for hospitals with eight or more. The difference remained statistically significant when the 1%, 5%, or 10% longest hospital stays were excluded from analysis, indicating that outliers were not responsible for the difference in length of stay. Using length of stay longer than 6 days as the definition of an outlier (the 90th percentile value in the group as a whole), 15% of patients treated at low volume hospitals (fewer than eight annual procedures) and 8% of patients treated at high volume hospitals (more than seven annual procedures) were outliers.
Total hospital charges were significantly lower at high volume hospitals (P < .001). Median total hospital charges were $26,069 for hospitals with fewer than eight annual endovascular procedures and $17,274 for hospitals with eight or more. A 10-fold increase in hospital volume, approximately the equivalent of the difference between the highest and lowest hospital volume quartiles, was associated with 39% lower charges (95% confidence interval, 24% to 61%). The difference remained significant when the most expensive 1%, 5%, or 10% of admissions were excluded from the analysis, indicating that outliers were not responsible for the difference in charges. Using total hospital charge more than $50,007 (the 90th percentile value in the group as a whole) as the definition of an outlier, 22% of patients treated at low volume hospitals (fewer than eight annual procedures) and 7% of patients treated at high volume hospitals (more than seven annual procedures) were outliers.
Discussion
We studied a representative sample of 421 patients with unruptured aneurysms who underwent endovascular treatment in the United States between 1996 and 2000. This group represents one of the largest cohorts of patients with unruptured aneurysms treated with endovascular techniques reported to date and is approximately one-fifth of all patients treated in the United States during that time period. The overall in-hospital mortality rate was 1.7% and the rate of discharge dead or to an institution other than home was 9.3%. Higher volume hospitals and endovascular physicians had lower rates of discharge other than to home, but no significant difference was observed between the mortality rates achieved by high and low volume hospitals and physicians.
This study was based on a national hospital discharge database, a data source with unique advantages and some severe shortcomings. The main advantage of the NIS database is its comprehensive sampling of the entire United States nonfederal hospital community. Other groups have used Medicare data (11), single state data from California (10, 26) or New York (8), data from consortia of university hospitals or specialized centers (13, 27, 28), or surveys of published data from the literature (29–31) to gain a broader perspective regarding the results of aneurysm treatment than is possible with single institution studies. Medicare data, although nationally comprehensive, include only the oldest patients with unruptured aneurysms; only approximately one-quarter of the patients we studied had Medicare insurance. Single state data may not be representative of broader geographic regions because of concentrations of expertise in specialized services, such as endovascular treatment, within individual states. In addition, single states typically have only a few high volume centers, and idiosyncratic results or patterns of care at these institutions can cause bias. Results from university hospitals and specialized centers are likely to be skewed toward high volume institutions and physicians and are likely to poorly represent community hospital results. Literature sources and synthetic reviews are liable to publication bias, the selective publication of unusually favorable results that may not fairly represent community practice in the broader sense (32–34). Because the NIS contains a complete representation of all cases discharged from a random sample of United States hospitals, no selection bias occurs on an individual or institutional level. In addition, the complete representation of a year’s discharges from each institution allows an accurate accounting of hospital volume of care, unlike databases that capture only a fraction of hospital discharges (such as Medicare data).
The drawbacks of administrative data should be clearly borne in mind in evaluating our results. First, the data available to us included only data obtained during the index hospitalization. Because individual patients cannot be identified or followed after discharge, outcome measures other than discharge status (such as 30-day mortality or 6-month functional outcome) are not available. In addition, patients readmitted for subsequent treatment of incompletely occluded aneurysms were (unavoidably) analyzed in this study as new patients. These procedures typically have low risk and are associated with short hospital stays, potentially introducing a bias toward better apparent results for centers and physicians with low rates of complete occlusion achieved by first treatment. Additionally, some patients may have undergone treatment of more than one aneurysm during a single admission. Second, only data obtained for all hospital patients are included in the database. Aneurysm characteristics such as size, location, and geometry are used by physicians to judge the risk of treatment for individual patients (35, 36), but we had no access to this information. Third, some data that were available to us are susceptible to multiple interpretations: some diagnoses could represent a preexisting condition, a new symptom of an unruptured aneurysm, or a complication of care (37). For example, a coded occlusion of a cerebral artery could represent a premorbid condition, a result of previous treatment of a second aneurysm, or a new endovascular complication. This ambiguity complicates the use of administrative databases to study carotid endarterectomy, for which “stroke” is both a major presenting symptom and an important potential iatrogenic complication but probably has less effect on our results because preadmission morbidity is relatively uncommon for unruptured aneurysms (38, 39). Finally, coding of important presenting symptoms and signs, medical comorbidity, and treatment complications are almost certain to be incomplete, as previous validation studies using similar databases have shown (37, 40–44). This tends to reduce study sensitivity to the effects these factors have on patient outcome (41).
Another disadvantage of our study, although due to the use of an administrative database, is specific to the topic of endovascular treatment of aneurysms. The ICD-9-CM code used to identify cases (code 39.52) is also used for open surgical aneurysm treatments other than clipping, the most common of which is aneurysm wrapping. Other groups have suggested that surgical treatment of unruptured aneurysms is associated with higher mortality rates, short-term morbidity, longer hospital stays, and higher charges than are associated with endovascular treatment (13, 36, 45). If so, an admixture of such cases could potentially bias our results. Because aneurysm wrapping is a relatively uncommon treatment (7, 36, 38), its use at hospitals where endovascular treatment is not actually performed could result in spurious “low volume hospitals” and “low volume physicians” in our data. One approach to this problem, used by Johnston et al (36) in a study that included validation by using a chart review, is to reclassify cases based on most procedures performed by the treating physician. However, for more than half of the cases in our sample, the treating physician was not identified and the terms of use of the NIS database forbade the use of chart review or other direct validation methods.
We used a two-stage approach to attempt to eliminate open surgical cases from our data. In the first stage, for cases in which the treating physician was identified (by a unique, masked identifying code), we used the performance of other procedures that include angiography to classify physicians as those who perform angiography and those who do not. We assumed procedures coded as 39.52 performed by physicians who perform angiography constituted true endovascular treatment of aneurysms. (Coding errors could have introduced bias if diagnostic angiography performed by another physician was misattributed to an admitting physician who does not perform angiography). Having defined a “gold standard” in those cases with identified treating physicians, we developed a multivariate logistic model by using only patient- and hospital-level characteristics that classified true cases and spurious cases with good accuracy. In the second stage, this model was applied to cases without an identified treating physician to identify those likely to be true endovascular treatments. These were combined with the cases with identified treating physicians who performed other angiographic procedures to produce the final group considered eligible for analysis.
Considering the possibility of introducing an important bias through misclassification of cases, we used several approaches to test our results. First, we hypothesized that transfusions (ICD-9-CM code 99.04) would be more frequent after surgical than after endovascular treatment. Reported rates of perioperative blood transfusion for unruptured aneurysm clipping range from 13% to 21% in single institution studies (46, 47). This code was not used in the classification model and provides an independent test of validity. Transfusions were six times more common in admissions we classified as ineligible compared with eligible cases (4.3% versus 0.7%, P = .03). Second, we repeated the main analysis by using only the 152 cases with identified treating physicians who performed other angiographic procedures. Hospital volume remained significant (P = .04) in this group, with an odds ratio of 0.19 (comparable with the odds ratio of 0.29 for the entire group of 421 cases). Third, we used the classification model alone (ignoring whether identified treating physicians had performed other angiographic procedures) to determine eligible cases in the entire study sample, potentially removing misclassified cases in the group with identified treating physicians. Hospital volume was significant (P = .02) in these 354 eligible cases, with an odds ratio of 0.34. Finally, we repeated the analysis excluding 21 cases that had undergone secondary procedures at any time during their hospital stay that included an open cranial operation; hospital volume again remained a significant predictor of outcome (P = .005).
Mortality and Discharge Other Than to Home
Comparison of our series with other large series of unruptured aneurysm coiling shows that our patient population was similar with respect to age, percent of female patients, race, and percent of emergency admissions and transfers from other health care institutions (13, 26, 36, 45, 48–52). Although some patient-level covariates, such as female sex and private insurance, were significant predictors of mortality and/or outcome at discharge in univariate models, only procedure timing (first hospital day versus other) remained significant in the full multivariate outcome model.
In-hospital mortality in our patient cohort was 1.7%, which is slightly higher than rates reported in most other series. In one population-based series, the in-hospital mortality rate after unruptured aneurysm coiling in California from 1990 through 1998 was 0.5% (26). Mortality rates in reports from specialized centers also have been low: the in-hospital mortality rate at a group of university hospitals from 1994 through 1997 was 0.4% (36), and no “procedure-related deaths” occurred based on a meta-analysis of 90 cases drawn from reports published before March 1997 (although 1.4% of patients died overall) (31). The in-hospital mortality rate was 2% at the University of California, San Francisco, from 1990 to 1997 (36), and several large single institution studies have reported zero mortality (48, 50, 52, 53).
Procedure-related morbidity has not been uniformly defined in previous reports of endovascular treatment of aneurysms. Studies based on discharge databases have typically classified all discharges other than to home as adverse outcomes (10, 13, 26, 27, 45, 54) without distinguishing between discharges to long-term facilities, such as skilled nursing facilities, from discharges to short-term facilities, such as rehabilitation hospitals. Counting all discharges other than an immediate return to home as equally severe “adverse events” ignores the prognostic judgment implied in the choice between short- and long-term placement. If a single cutoff point must be chosen, a more appropriate one might be “death or discharge to a skilled nursing facility,” because this implies a judgment by treating physicians and consulting professionals that a return to independent life is unlikely. Our use of ordinal logistic regression allows all the information inherent in a multilevel ordered outcome to be preserved, rather than collapsing outcome categories with consequent loss of information. Although detailed functional scores assessed 1 month to 1 year after treatment, as used by others (7, 28, 36), would clearly be preferable to the outcome scale we used, our database did not include this information.
In our study, the rate of discharge to a skilled nursing facility was 2.4% and a total of 9.3% of patients were not discharged directly to home. Johnston et al (26) reported that 9.7% of patients who underwent endovascular treatment of unruptured aneurysms in California from 1990 to 1998 were not discharged directly to home, and the corresponding rate for 60 university hospitals belonging to the University Health System Consortium was 10.6% from 1994 to 1997 (13). At discharge from the University of California, San Francisco, from 1990 to 1997, 4% of patients had Rankin scores of 3% to 5% and 13% had Glasgow Outcome Scale scores corresponding to moderate or severe disability or death (36). Significant deficits still present 1 month after open surgical treatment of unruptured aneurysms are likely to persist for at least 1 year. However, the persistence of deficits after endovascular treatment of unruptured aneurysms that are sufficiently mild at hospital discharge to allow discharge to a short-term rehabilitation facility has not been well defined. In most single institution studies, morbidity has been described in terms of complication rates, variously defined but generally reported to be approximately 5% of patients (48, 50, 52). This is comparable with the 5.0% of patients in the present series for whom a “neurologic complication of treatment” was coded.
Our decision to exclude 20 patients with a coded diagnosis of subarachnoid hemorrhage from analysis may have resulted in lower morbidity and mortality rates than those actually experienced, because aneurysm rupture during attempted coiling is a recognized treatment complication that can have severe consequences (55–57). Morbidity and mortality rates for patients with this diagnosis were significantly higher than for other patients (P < .001), as were reported rates of neurologic complications of treatment (25% versus 5%, P = .001). Unfortunately, a coded diagnosis in the NIS database can represent medical history, new diagnosis, or an event that occurs during current admission (37). Most of the patients with this diagnosis in our cohort (65%) had emergent or urgent admissions, compared with 18% for other patients. This suggests that most patients with this diagnosis actually had ruptured aneurysms at the time of hospital admission and were appropriately excluded from the cohort.
We defined several patient-related risk factors for in-hospital mortality, discharge other than to home, or both: age, male sex, primary insurance other than private, emergency or urgent hospital admission, admission through the emergency room or as a transfer from another hospital, and whether the endovascular procedure was performed on the first hospital day. Our database lacked information most endovascular surgeons consider vital in estimating the risk of endovascular treatment for a specific unruptured aneurysm, such as aneurysm size, location, and geometry (36). However, it should be noted that our multivariate model accurately predicted adverse outcomes by using information that is lacking in most reported series of unruptured aneurysms undergoing endovascular treatment. The best model would probably combine detailed aneurysm-specific information with patient-related information more typically included in hospital discharge databases.
Volume of Care and Outcome
Since the early work of Luft et al (1) and others (58–60) in the 1970s and 1980s, multiple studies have documented better medical outcomes at high volume centers and with high volume providers. Within neurosurgery, this relationship has been studied extensively for carotid endarterectomy (5, 61–65); other studies focus on pediatric brain tumor surgery (66, 67) and neurotrauma (68). Better short-term outcomes after endovascular treatment performed at high volume institutions or by high volume operators have been documented repeatedly for percutaneous coronary artery angioplasty and/or coronary artery stent placement (69–76).
Outcome after treatment for both ruptured and unruptured intracranial aneurysms has also been studied in relation to hospital volume of care (8–11, 13, 26, 27, 35, 36, 60, 77, 78). In most reports, patient outcomes were better at high volume institutions, although one study found higher mortality rates at high volume hospitals (60) (attributed to referral of more severe cases to specialized centers) and some studies found no effect of volume or surgical experience on outcome (77, 78).
Fewer studies have focused specifically on treatment of unruptured aneurysms and hospital volume, and none are directly comparable with our study. Solomon et al (8) examined mortality in 1604 patients who underwent surgical clipping of unruptured aneurysms at New York hospitals from 1987 through 1993. Mortality was lower at high volume hospitals. An important series of studies by Johnston et al (13, 26, 27, 35, 36) focused on outcomes of endovascular and surgical treatment of unruptured cerebral aneurysms, finding that short-term patient outcomes were better after endovascular treatment and that overall outcomes were better at hospitals where a higher proportion of patients underwent endovascular treatment. However, the relation between absolute hospital volume of endovascular treatment and the outcome of patients treated by using endovascular means was not reported.
Volume of care for individual treating physicians was also correlated with better patient outcome in our study. Similar findings have been reported for cardiologist volume of care and short-term outcome after coronary artery angioplasty (69, 71, 73). We know of no direct parallel between our finding and those of other studies of endovascular treatment of aneurysms, although the “learning curve” for aneurysm coiling has been previously studied. Singh et al (51) reported that complications occurred in 53% of the first five patients treated with detachable coils by each of three endovascular surgeons, compared with 10% of later cases (P < .001). On an institutional level, two groups have reported better short-term outcomes after the first 50 (79) or 100 cases (80) treated at single institutions.
We also found significant relationships between two secondary end points and hospital volume of endovascular treatment of aneurysms: higher volume hospitals had shorter lengths of stay and lower total charges. In an analysis of all unruptured aneurysms treated with surgery or endovascular techniques in California from 1990 through 1998, treatment volume was not an independent predictor of length of stay or hospital charges, although 80% of patients had undergone surgery (26).
Because we found better patient outcomes at high volume centers, in association with lower total hospital charges, we investigated the presence of a cutoff point that might divide centers into high and low volume groups with significantly different outcomes. Such a cutoff point could potentially assist patients (or insurers) seeking high quality care for unruptured intracranial aneurysms. Our analysis did not support a single significant cutoff point, with an adjusted P value of 0.07 for the best cutoff point in multivariate analysis and a confidence interval for the best cutoff point that included the entire range of possible choices (from 1 to 36 annual procedures). However, from the standpoint of an individual patient seeking high quality care, our results do support the selection of a high volume physician at a high volume center. Such care is most likely to be found at urban teaching institutions.
Successful treatment of unruptured intracranial aneurysms might be defined as comprising protection from future risk of hemorrhage, alleviation of any neurologic symptoms present at diagnosis, and freedom from new deficits that result from the treatment. Our study addresses only one of these three elements. It is intuitively appealing to suggest that other quality measures, in addition to short-term outcome, also will be better for patients treated at high volume centers. Because the database we used does not allow tracking of individual patients after discharge, we could not investigate long-term end points, such as recurrent hemorrhage rates or return to independent function for patients discharged to rehabilitation centers. Short-term surrogate end points for protection from hemorrhage, such as degree of arteriographic occlusion at the time of initial procedure, are not contained in NIS data. Answering questions regarding long-term outcome will require data sources other than the NIS and will pose significant challenges for future investigators.
Conclusion
Based on a large cohort of patients with unruptured aneurysms treated with endovascular techniques in the United States from 1996 through 2000, our analysis suggests that outcomes at the time of discharge were better at high volume centers, although we found no significant difference in mortality rates. We found that patients treated by high volume physicians also had better outcomes at the time of discharge and that hospital stays were significantly shorter and costs of treatment were significantly lower at high volume institutions. Our data source did not extend to longer-term end points, such as functional status or rebleeding rates. If our findings are confirmed and extended by further studies, they will be noteworthy for patients with unruptured aneurysms seeking quality endovascular treatment and for their physicians and insurers.
References
- 1.Luft HS, Garnick DW, Mark DH, McPhee SJ. Hospital Volume, Physician Volume, and Patient Outcomes. Ann Arbor: Health Administration Press Perspectives;1990
- 2.Hewitt M. Interpreting the Volume-Outcome Relationship in the Context of Health Care Quality: Workshop Summary. Washington: National Academy Press;2000
- 3.Begg CB, Cramer LD, Hoskins WJ, Brennan MF. Impact of hospital volume on operative mortality for major cancer surgery. JAMA 1998;280:1747–1751 [DOI] [PubMed] [Google Scholar]
- 4.Hillner BE, Smith TJ, Desch CE. Hospital and physician volume or specialization and outcomes in cancer treatment: importance in quality of cancer care. J Clin Oncol 2000;18:2327–2340 [DOI] [PubMed] [Google Scholar]
- 5.Birkmeyer JD, Siewers AE, Finlayson EV, et al. Hospital volume and surgical mortality in the United States. N Engl J Med 2002;346:1128–1137 [DOI] [PubMed] [Google Scholar]
- 6.Cronenwett JL, Birkmeyer JD, Cronenwett JL, Birkmeyer JD. The Dartmouth Atlas of Vascular Health Care. Chicago: American Hospital Publishing;2000 [PubMed]
- 7.Chyatte D, Porterfield R. Functional outcome after repair of unruptured intracranial aneurysms. J Neurosurg 2001;94:417–421 [DOI] [PubMed] [Google Scholar]
- 8.Solomon RA, Mayer SA, Tarmey JJ. Relationship between the volume of craniotomies for cerebral aneurysm performed at New York state hospitals and in-hospital mortality. Stroke 1996;27:13–17 [DOI] [PubMed] [Google Scholar]
- 9.Barker FG II, Amin-Hanjani S, Butler WE, Ogilvy CS, Carter BS . In-hospital mortality and morbidity after surgical treatment of unruptured intracranial aneurysms in the United States, 1996–2000: the effect of hospital and surgeon volume. Neurosurgery 2003;52:995–1009 [PubMed] [Google Scholar]
- 10.Bardach NS, Zhao S, Gress DR, Lawton MT, Johnston SC. Association between subarachnoid hemorrhage outcomes and number of cases treated at California hospitals. Stroke 2002;33:1851–1856 [DOI] [PubMed] [Google Scholar]
- 11.Taylor CL, Yuan Z, Selman WR, Ratcheson RA, Rimm AA. Mortality rates, hospital length of stay, and the cost of treating subarachnoid hemorrhage in older patients: institutional and geographical differences. J Neurosurg 1997;86:583–588 [DOI] [PubMed] [Google Scholar]
- 12.Steiner C, Elixhauser A, Schnaier J. The healthcare cost and utilization project: an overview. Eff Clin Pract 2002;5:143–151 [PubMed] [Google Scholar]
- 13.Johnston SC, Dudley RA, Gress DR, Ono L. Surgical and endovascular treatment of unruptured cerebral aneurysms at university hospitals. Neurology 1999;52:1799–1805 [DOI] [PubMed] [Google Scholar]
- 14.Moses LE, Emerson JD, Hosseini H. Analyzing data from ordered categories. In Bailar JC III, Mosteller F, eds. Medical Uses of Statistics. Boston: New England Journal of Medicine Books;1992. :259–279 [DOI] [PubMed]
- 15.McCullagh P, Nelder JA. Generalized Linear Models. 2nd ed. London: Chapman & Hall;1989
- 16.Strömberg U. Collapsing ordered outcome categories: a note of concern. Am J Epidemiol 1996;144:421–424 [DOI] [PubMed] [Google Scholar]
- 17.Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care 1998;36:8–27 [DOI] [PubMed] [Google Scholar]
- 18.Hosmer DW Jr, Lemeshow S. Applied Logistic Regression. 2nd ed. New York: John Wiley & Sons;2000
- 19.McCullagh P. Regression models for ordinal data. J R Stat Soc [Ser B] 1980;42:109–142 [Google Scholar]
- 20.Harrell FE Jr. Regression Modeling Strategies: With Applications to Linear Models, Logistic Regression, and Survival Analysis. New York: Springer-Verlag;2001
- 21.Raudenbush S, Bryk A, Cheong YF, Congdon R. HLM 5: Hierarchical Linear and Nonlinear Modeling. Lincolnwood: Scientific Software International;2001
- 22.Altman DG, Lausen B, Sauerbrei W, Schumacher M. Dangers of using “optimal” cutpoints in the evaluation of prognostic factors. J Natl Cancer Inst 1994;86:829–835 [DOI] [PubMed] [Google Scholar]
- 23.Mazumdar M, Glassman JR. Categorizing a prognostic variable: review of methods, code for easy implementation and applications to decision-making about cancer treatments. Stat Med 2000;19:113–132 [DOI] [PubMed] [Google Scholar]
- 24.Efron B, Tibshirani RJ. An Introduction to the Bootstrap. London: Chapman & Hall;1993
- 25.Harrell FE Jr. Hmisc and Design libraries for S-Plus for Windows. 2000; Software and electronic documentation available from http://hesweb1.med.virginia.edu/biostat/s/splus.html
- 26.Johnston SC, Zhao S, Dudley RA, Berman MF, Gress DR. Treatment of unruptured cerebral aneurysms in California. Stroke 2001;32:597–605 [DOI] [PubMed] [Google Scholar]
- 27.Johnston SC. Effect of endovascular services and hospital volume on cerebral aneurysm treatment outcomes. Stroke 2000;31:111–117 [DOI] [PubMed] [Google Scholar]
- 28.International Study of Unruptured Intracranial Aneurysms Investigators. Unruptured intracranial aneurysms: risk of rupture and risks of surgical intervention. N Engl J Med 1998;339:1725–1733 [DOI] [PubMed] [Google Scholar]
- 29.King JT Jr, Berlin JA, Flamm ES. Morbidity and mortality from elective surgery for asymptomatic, unruptured, intracranial aneurysms: a meta-analysis. J Neurosurg 1994;81:837–842 [DOI] [PubMed] [Google Scholar]
- 30.Raaymakers TW, Rinkel GJ, Limburg M, Algra A. Mortality and morbidity of surgery for unruptured intracranial aneurysms: a meta-analysis. Stroke 1998;29:1531–1538 [DOI] [PubMed] [Google Scholar]
- 31.Brilstra EH, Rinkel GJ, van der Graaf Y, van Rooij WJ, Algra A. Treatment of intracranial aneurysms by embolization with coils: a systematic review. Stroke 1999;30:470–476 [DOI] [PubMed] [Google Scholar]
- 32.Shaheen NJ, Crosby MA, Bozymski EM, Sandler RS. Is there publication bias in the reporting of cancer risk in Barrett’s esophagus? Gastroenterology 2000;119:333–338 [DOI] [PubMed] [Google Scholar]
- 33.Song F, Eastwood AJ, Gilbody S, Duley L, Sutton AJ. Publication and related biases. Health Technol Assess 2000;4:1–115 [PubMed] [Google Scholar]
- 34.Sutton AJ, Duval SJ, Tweedie RL, Abrams KR, Jones DR. Empirical assessment of effect of publication bias on meta-analyses. BMJ 2000;320:1574–1577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Johnston SC. Identifying confounding by indication through blinded prospective review. Am J Epidemiol 2001;154:276–284 [DOI] [PubMed] [Google Scholar]
- 36.Johnston SC, Wilson CB, Halbach VV, et al. Endovascular and surgical treatment of unruptured cerebral aneurysms: comparison of risks. Ann Neurol 2000;48:11–19 [DOI] [PubMed] [Google Scholar]
- 37.Lawthers AG, McCarthy EP, Davis RB, Peterson LE, Palmer RH, Iezzoni LI. Identification of in-hospital complications from claims data: is it valid? Med Care 2000;38:785–795 [DOI] [PubMed] [Google Scholar]
- 38.Solomon RA, Fink ME, Pile-Spellman J. Surgical management of unruptured intracranial aneurysms. J Neurosurg 1994;80:440–446 [DOI] [PubMed] [Google Scholar]
- 39.Friedman JA, Piepgras DG, Pichelmann MA, Hansen KK, Brown RD Jr, Wiebers DO. Small cerebral aneurysms presenting with symptoms other than rupture. Neurology 2001;57:1212–1216 [DOI] [PubMed] [Google Scholar]
- 40.Best WR, Khuri SF, Phelan M, et al. Identifying patient preoperative risk factors and postoperative adverse events in administrative databases: results from the Department of Veterans Affairs National Surgical Quality Improvement Program. J Am Coll Surg 2002;194:257–266 [DOI] [PubMed] [Google Scholar]
- 41.Romano PS, Roos LL, Luft HS, Jollis JG, Doliszny K. A comparison of administrative versus clinical data: coronary artery bypass surgery as an example: Ischemic Heart Disease Patient Outcomes Research Team. J Clin Epidemiol 1994;47:249–260 [DOI] [PubMed] [Google Scholar]
- 42.McCarthy EP, Iezzoni LI, Davis RB, et al. Does clinical evidence support ICD-9-CM diagnosis coding of complications? Med Care 2000;38:868–876 [DOI] [PubMed] [Google Scholar]
- 43.Iezzoni LI. Assessing quality using administrative data. Ann Intern Med 1997;127:666–674 [DOI] [PubMed] [Google Scholar]
- 44.Iezzoni LI, Daley J, Heeren T, et al. Identifying complications of care using administrative data. Med Care 1994;32:700–715 [DOI] [PubMed] [Google Scholar]
- 45.Higashida RT, Justason BJ, Clark M, et al. Endovascular treatment of unruptured cerebral aneurysms may lead to improved outcomes and lower hospital resource use [abst]. Presented at the 41st Annual Meeting of the American Society of Neuroradiology, Vancouver, B.C., Canada,2002
- 46.Couture DE, Ellegala DB, Dumont AS, Mintz PD, Kassell NF. Blood use in cerebrovascular neurosurgery. Stroke 2002;33:994–997 [DOI] [PubMed] [Google Scholar]
- 47.Le Roux PD, Elliott JP, Winn HR. Blood transfusion during aneurysm surgery. Neurosurgery 2001;49:1068–1075 [DOI] [PubMed] [Google Scholar]
- 48.Goddard AJ, Annesley-Williams D, Gholkar A. Endovascular management of unruptured intracranial aneurysms: does outcome justify treatment? J Neurol Neurosurg Psychiatry 2002;72:485–490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Regli L, Uske A, de Tribolet N. Endovascular coil placement compared with surgical clipping for the treatment of unruptured middle cerebral artery aneurysms: a consecutive series. J Neurosurg 1999;90:1025–1030 [DOI] [PubMed] [Google Scholar]
- 50.Roy D, Milot G, Raymond J. Endovascular treatment of unruptured aneurysms. Stroke 2001;32:1998–2004 [DOI] [PubMed] [Google Scholar]
- 51.Singh V, Gress DR, Higashida RT, Dowd CF, Halbach VV, Johnston SC. The learning curve for coil embolization of unruptured intracranial aneurysms. AJNR Am J Neuroradiol 2002;23:768–771 [PMC free article] [PubMed] [Google Scholar]
- 52.Wanke I, Doerfler A, Dietrich U, et al. Endovascular treatment of unruptured intracranial aneurysms. AJNR Am J Neuroradiol 2002;23:756–761 [PMC free article] [PubMed] [Google Scholar]
- 53.Ng P, Khangure MS, Phatouros CC, Bynevelt M, ApSimon H, McAuliffe W. Endovascular treatment of intracranial aneurysms with Guglielmi detachable coils: analysis of midterm angiographic and clinical outcomes. Stroke 2002;33:210–217 [DOI] [PubMed] [Google Scholar]
- 54.Johnston SC. Combining ecological and individual variables to reduce confounding by indication: case study: subarachnoid hemorrhage treatment. J Clin Epidemiol 2000;53:1236–1241 [DOI] [PubMed] [Google Scholar]
- 55.Doerfler A, Wanke I, Egelhof T, et al. Aneurysmal rupture during embolization with Guglielmi detachable coils: causes, management, and outcome. AJNR Am J Neuroradiol 2001;22:1825–1832 [PMC free article] [PubMed] [Google Scholar]
- 56.Levy E, Koebbe CJ, Horowitz MB, et al. Rupture of intracranial aneurysms during endovascular coiling: management and outcomes. Neurosurgery 2001;49:807–813 [DOI] [PubMed] [Google Scholar]
- 57.McDougall CG, Halbach VV, Dowd CF, Higashida RT, Larsen DW, Hieshima GB. Causes and management of aneurysmal hemorrhage occurring during embolization with Guglielmi detachable coils. J Neurosurg 1998;89:87–92 [DOI] [PubMed] [Google Scholar]
- 58.Hughes RG, Hunt SS, Luft HS. Effects of surgeon volume and hospital volume on quality of care in hospitals. Med Care 1987;25:489–503 [DOI] [PubMed] [Google Scholar]
- 59.Luft HS, Bunker JP, Enthoven AC. Should operations be regionalized? the empirical relation between surgical volume and mortality. N Engl J Med 1979;301:1364–1369 [DOI] [PubMed] [Google Scholar]
- 60.Luft HS, Hunt SS, Maerki SC. The volume-outcome relationship: practice-makes-perfect or selective-referral patterns? Health Serv Res 1987;22:157–182 [PMC free article] [PubMed] [Google Scholar]
- 61.Finlayson EV, Birkmeyer JD. Outcomes in vascular surgery: volume versus certification. Surgery 2001;130:897–898 [DOI] [PubMed] [Google Scholar]
- 62.Hannan EL, Popp AJ, Feustel P, et al. Association of surgical specialty and processes of care with patient outcomes for carotid endarterectomy. Stroke 2001;32:2890–2897 [DOI] [PubMed] [Google Scholar]
- 63.O’Neill L, Lanska DJ, Hartz A. Surgeon characteristics associated with mortality and morbidity following carotid endarterectomy. Neurology 2000;55:773–781 [DOI] [PubMed] [Google Scholar]
- 64.Dudley RA, Johansen KL, Brand R, Rennie DJ, Milstein A. Selective referral to high-volume hospitals: estimating potentially avoidable deaths. JAMA 2000;283:1159–1166 [DOI] [PubMed] [Google Scholar]
- 65.Wennberg DE, Lucas FL, Birkmeyer JD, Bredenberg CE, Fisher ES. Variation in carotid endarterectomy mortality in the Medicare population: trial hospitals, volume, and patient characteristics. JAMA 1998;279:1278–1281 [DOI] [PubMed] [Google Scholar]
- 66.Albright AL, Sposto R, Holmes E, et al. Correlation of neurosurgical subspecialization with outcomes in children with malignant brain tumors. Neurosurgery 2000;47:879–877 [DOI] [PubMed] [Google Scholar]
- 67.Sanford RA. Craniopharyngioma: results of survey of the American Society of Pediatric Neurosurgery. Pediatr Neurosurg 1994;21:39–43 [DOI] [PubMed] [Google Scholar]
- 68.Kagan RJ, Baker RJ. The impact of the volume of neurotrauma experience on mortality after head injury. Am Surg 1994;60:394–400 [PubMed] [Google Scholar]
- 69.Vakili BA, Kaplan R, Brown DL. Volume-outcome relation for physicians and hospitals performing angioplasty for acute myocardial infarction in New York state. Circulation 2001;104:2171–2176 [DOI] [PubMed] [Google Scholar]
- 70.Birkmeyer JD, Finlayson EV, Birkmeyer CM. Volume standards for high-risk surgical procedures: potential benefits of the Leapfrog initiative. Surgery 2001;130:415–422 [DOI] [PubMed] [Google Scholar]
- 71.McGrath PD, Wennberg DE, Dickens JD Jr, et al. Relation between operator and hospital volume and outcomes following percutaneous coronary interventions in the era of the coronary stent. JAMA 2000;284:3139–3144 [DOI] [PubMed] [Google Scholar]
- 72.Kimmel SE, Sauer WH, Brensinger C, Hirshfeld J, Haber HL, Localio AR. Relationship between coronary angioplasty laboratory volume and outcomes after hospital discharge. Am Heart J 2002;143:833–840 [DOI] [PubMed] [Google Scholar]
- 73.Hannan EL, Racz M, Ryan TJ, et al. Coronary angioplasty volume-outcome relationships for hospitals and cardiologists. JAMA 1997;277:892–898 [PubMed] [Google Scholar]
- 74.Phillips KA, Luft HS, Ritchie JL. The association of hospital volumes of percutaneous transluminal coronary angioplasty with adverse outcomes, length of stay, and charges in California. Med Care 1995;33:502–514 [DOI] [PubMed] [Google Scholar]
- 75.Kimmel SE, Berlin JA, Laskey WK. The relationship between coronary angioplasty procedure volume and major complications. JAMA 1995;274:1137–1142 [PubMed] [Google Scholar]
- 76.Ritchie JL, Phillips KA, Luft HS. Coronary angioplasty: statewide experience in California. Circulation 1993;88:2735–2743 [DOI] [PubMed] [Google Scholar]
- 77.Kassell NF, Torner JC, Haley EC Jr, Jane JA, Adams HP, Kongable GL. The International Cooperative Study on the Timing of Aneurysm Surgery: part 1. overall management results. J Neurosurg 1990;73:18–36 [DOI] [PubMed] [Google Scholar]
- 78.Taylor B, Harries P, Bullock R. Factors affecting outcome after surgery for intracranial aneurysm in Glasgow. Br J Neurosurg 1991;5:591–600 [DOI] [PubMed] [Google Scholar]
- 79.Murayama Y, Viñuela F, Duckwiler GR, Gobin YP, Guglielmi G. Embolization of incidental cerebral aneurysms by using the Guglielmi detachable coil system. J Neurosurg 1999;90:207–214 [DOI] [PubMed] [Google Scholar]
- 80.Malisch TW, Guglielmi G, Viñuela F, et al. Intracranial aneurysms treated with the Guglielmi detachable coil: midterm clinical results in a consecutive series of 100 patients. J Neurosurg 1997;87:176–183 [DOI] [PubMed] [Google Scholar]