Abstract
BACKGROUND AND PURPOSE: Stroke severity in cases of hyperacute carotid ischemic stroke may be related to site of arterial occlusion. We evaluated the relationships between National Institutes of Health Stroke Scale (NIHSS) scores and findings on intra-arterial digital subtraction angiograms (IA-DSA) of patients with ischemic stroke within 6 hr of stroke onset.
METHODS: A total of 43 consecutive patients (38 men and five women; mean age, 69.4 ± 8.7 years) with ischemic stroke in the carotid territory underwent IA-DSA within 6 hr of stroke onset. Baseline NIHSS score was assessed immediately before IA-DSA. Patients were divided into four groups according to site of arterial occlusion: 1) the internal carotid artery (ICA group, n = 10); 2) stem of the middle cerebral artery or stem of the anterior cerebral artery (Stem group, n = 14); 3) branches of middle cerebral artery or anterior cerebral artery (Branch group, n = 11); and 4) no arterial occlusion (Normal group, n = 8).
RESULTS: Mean (±SD) NIHSS score was 14.7 ± 7.4. The interval from stroke onset to IA-DSA study was 205 ± 76 min. NIHSS score was higher in the ICA group (median, 23; range, 6–32) than in the Branch (median, 17; range, 11–25; P = .02) or Normal (median, 15; range, 2–17; P < .001) groups but was not higher than in the Stem group (median, 6; range, 1–11; P = .73). Sensitivity-specificity curve analysis suggested an NIHSS score ≥10 as indicative of arterial occlusion of the carotid system. A total of 96.9% of patients with NIHSS scores ≥10 displayed arterial occlusion, and 63.6% of patients with NIHSS scores <10 displayed no arterial occlusion.
CONCLUSION: NIHSS score is related to site of arterial occlusion in cases of hyperacute carotid ischemic stroke. An NIHSS score of 10 seems to represent the cut-off for discriminating between patients with arterial occlusion and patients without.
The National Institutes of Health Stroke Scale (NIHSS) is a widely used and well-validated neurologic impairment scale, measuring speech and language, cognition, visual field deficits, motor and sensory impairments, and ataxia (1). NIHSS score assessed during the hyperacute phase of stroke strongly predicts the likelihood of patient recovery after stroke and has been used to include or exclude patients from trials of acute stroke therapy, including thrombolysis (2–7). The NIHSS therefore represents a standard part of clinical assessment for patients with acute stroke in many stroke centers.
Fink et al (8) reported a significant correlation between diffusion-weighted MR imaging lesion volume and NIHSS score. Several studies examined relationships between initial NIHSS score and vascular imaging techniques such as ultrasonography (9, 10), CT angiography (11), and MR angiography (12), reporting that a higher NIHSS score was associated with more severe vascular lesions in patients with acute stroke. However, vascular imaging methods have limitations in clearly displaying occlusion or stenosis of the main stem and branches of the middle cerebral artery (MCA) and anterior cerebral artery (ACA). Relationships between NIHSS score and site of arterial occlusion during the hyperacute phase of stroke, therefore, have yet to be accurately determined.
Intra-arterial digital subtraction angiography (IA-DSA) is superior to other methods for detecting the site of arterial occlusion and is considered to represent the gold standard for vascular imaging. During the hyperacute phase of stroke, detailed knowledge of arterial occlusion can be clinically important, particularly regarding thromobolysis (13, 14). The aim of the present study was to evaluate relationships between NIHSS score and IA-DSA findings in patients with ischemic stroke within 6 hr of stroke onset.
Methods
Patients and Techniques
Of the 112 patients admitted to our division within 6 hr of ischemic stroke onset between April 1999 and June 2002, IA-DSA was performed in 43 patients with carotid acute ischemic stroke (38 men and five women; mean age, 69.4 ± 8.7 years). We excluded the patients with posterior circulation strokes because anterior circulation and posterior circulation strokes were thought to be separate entities, with different underlying pathogenesis and natural histories. These 43 patients were enrolled into this study. All patients were assessed by using the NIHSS immediately before IA-DSA. If the patient was aware of symptoms on waking from sleep, time of onset was defined as the last time they were free from symptoms. A modified Rankin Scale (15) score ≥2 before stroke onset was used as an exclusion criterion.
Sex, age, history of stroke or transient ischemic attack, and modified Rankin Scale score were examined, along with vascular risk factors including hypertension, diabetes mellitus, hyperlipidemia, smoking, and potential embolic sources of emboli (atrial fibrillation, patent foramen ovale, left ventricular aneurysm, prosthetic heart valves, infective endocarditis, sick sinus syndrome, dilated cardiomyopathy, and complicated lesions in aortic arch).
Vascular risk factors were identified as follows: 1) use of antihypertensive agents for hypertension, with systolic blood pressure ≥160 mmHg or diastolic blood pressure ≥95 mmHg at admission for hypertension; 2) use of oral hypoglycemic agents, insulin, or glycosylated hemoglobin >6.4% for diabetes mellitus; 3) use of antihyperlipidemic agents or serum cholesterol level >220 mg/dL for hypercholesterolemia; or 4) history of smoking. To detect potential embolic sources of emboli, all patients underwent 12-lead ECG, 24-hr ECG monitoring, and transthoracic or transesophageal echocardiography.
Informed consent for performance of IA-DSA was obtained from patients and/or their family members. Selective IA-DSA was performed by using a biplane, high resolution angiography system (Angio Rex Super-G and DFP-2000A, Toshiba) with a matrix of 1024 × 1024 pixels. A catheter was inserted into the right brachial artery or femoral artery in accordance with the Seldinger method and then guided to the cerebral arteries for diagnostic four-vessel angiography.
Patients were divided into four groups according to the site of arterial occlusion: 1) occlusion of the internal carotid artery (ICA group); 2) occlusion of the main stem of the MCA or A1 segment of the ACA (Stem group); 3) occlusion of the MCA or ACA branch, including occlusion of M2 or A2 or more distal sites (Branch group); or 4) no arterial occlusion (Normal group). If a patient displayed two or more occluded arteries, the patient was placed in the largest artery group (eg, if occlusions of both the right main trunk of the MCA and the ipsilateral A2 portion were present, the patient was placed in the Stem group).
CT of the brain was performed immediately at admission and 3 days after stroke onset to evaluate ischemic lesions. Within 7 days of stroke onset, MR imaging was performed by using a 1.5-T system (Magnetom Vision, Siemens) equipped with single shot echo-planar imaging to obtain rapid diffusion images. MR imaging studies included axial T1-weighted, axial T2-weighted, and diffusion-weighted sequences (approximately 30 min of imaging time). Imaging parameters were as follows: 4000/103 (TR/TE); matrix, 128 × 128; field of view, 230 mm; section thickness, 4 mm; section gap, 2 mm. Two b values were used (0 and 1000 s/mm2). Diffusion gradients were applied in successive images in each of the x, y, and z directions, and diffusion-weighted images were formed from the mean of these values. Criteria for diagnosis of acute infarcts on diffusion-weighted images included focal hyperintensity judged not to be due to normal anisotropic diffusion or magnetic susceptibility artifact. These lesions were also categorized as cortical, subcortical, or lacunar infarcts according to location.
Statistical analysis was performed by using StatView 5.0 for Windows (SAS Institute, 1998). The χ2 test or Kruskal-Wallis U test was used to compare baseline characteristics among the four groups. Relationships between baseline NIHSS score and site of arterial occlusion were tested by using the Kruskal-Wallis U test, and the differences between NIHSS scores for each group were tested by post hoc analysis under Scheffe’s method. To obtain the NIHSS score as the cut-off point for discriminating between patients with arterial occlusion and those without, a sensitivity and specificity curve was drawn. The study protocol followed all principles outlined in the Declaration of Helsinki.
Results
Of the 43 patients enrolled in this study, 20 underwent IA-DSA within 3 hr of stroke onset. Intervals from stroke onset to arrival at hospital and to IA-DSA study were 88 ± 58 min and 205 ± 76 min, respectively.
The Stem group was the largest (n = 14), with the other groups in descending order being the Branch (n = 11), ICA (n = 10), and Normal (n = 8) groups. Demographic data and clinical features of each group are shown in Table 1. Atrial fibrillation was observed most frequently in the Branch group (P < .014). No other significant differences in baseline characteristics were observed.
TABLE 1:
Clinical characteristics of all patients
| Group | ICA | Stem | Branch | Normal | P |
|---|---|---|---|---|---|
| Number of patients | 10 | 14 | 11 | 8 | |
| Age, median (range) (yr) | 71 (48–78) | 68 (56–82) | 74 (57–86) | 64 (55–84) | 0.671 |
| Sex, male | 10 (100%) | 12 (86%) | 9 (82%) | 7 (88%) | 0.598† |
| Hypertension | 8 (80%) | 9 (64%) | 5 (45%) | 8 (100%) | 0.065 |
| Diabetes mellitus | 2 (20%) | 5 (36%) | 4 (36%) | 1 (13%) | 0.405 |
| Hyperlipidemia | 5 (50%) | 4 (29%) | 4 (36%) | 6 (75%) | 0.181 |
| Smoking | 6 (60%) | 8 (57%) | 2 (18%) | 3 (38%) | 0.163 |
| Atrial fibrillation | 5 (50%) | 10 (71%) | 9 (82%) | 1 (13%) | 0.014 |
| Potential emboligenic diseases | 2 (20%) | 3 (21%) | 3 (27%) | 4 (50%) | 0.467 |
| Patent foramen ovale | 0 | 2 | 2 | 2 | |
| Left ventricular aneurysm | 0 | 1 | 0 | 0 | |
| Complicated lesion in aorta | 2 | 0 | 1 | 2 | |
| History of stroke/transient ischemic attack | 1 (10%) | 1 (7%) | 2 (18%) | 1 (13%) | 0.858 |
| Interval from stroke onset to angiography mean ± SD (min) | 173 ± 75 (150) | 211 ± 86 (194) | 223 ± 67 (185) | 223 ± 76 (199) | 0.272† |
| Affected side (right/left) | 6/4 | 8/6 | 3/8 | 3/5 | 0.353 |
* Analyzed by using the χ2 test.
Analyzed by using the Kruskal-Wallis U test.
In the ICA group, seven patients displayed ICA occlusion. For one, occlusion of both the ICA and ipsilateral ACA A2 portion was show, and for the remaining two, both ICA occlusion and ipsilateral MCA stem occlusion was shown, despite good collateral flow from the contralateral ACA via the anterior communicating artery. In the Stem group, 11 patients displayed MCA stem occlusion, one had bilateral MCA stem occlusion, one had both MCA stem occlusion and ipsilateral A2 occlusion, and the remaining had both MCA stem occlusion and occlusion of the distal site of ipsilateral ACA. In the Branch group, five patients had MCA M2 branch occlusion, two had M3 branch occlusion, two had both M2 and A2 portion occlusion, one had M2 and A4 portion occlusions, and the other one displayed M4 and A3 portion occlusions.
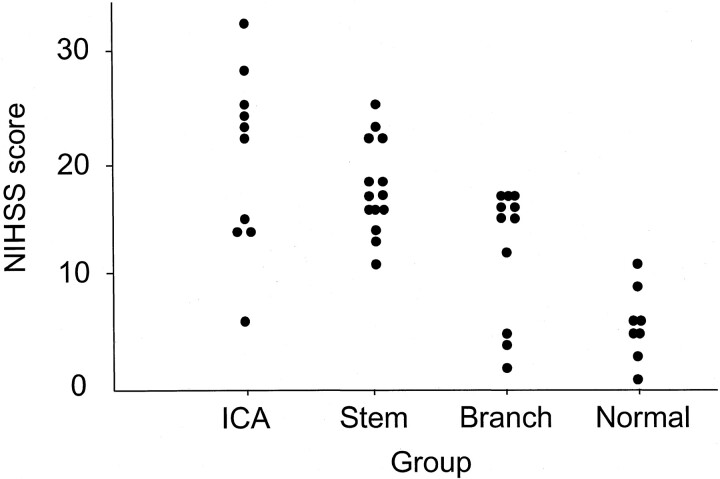
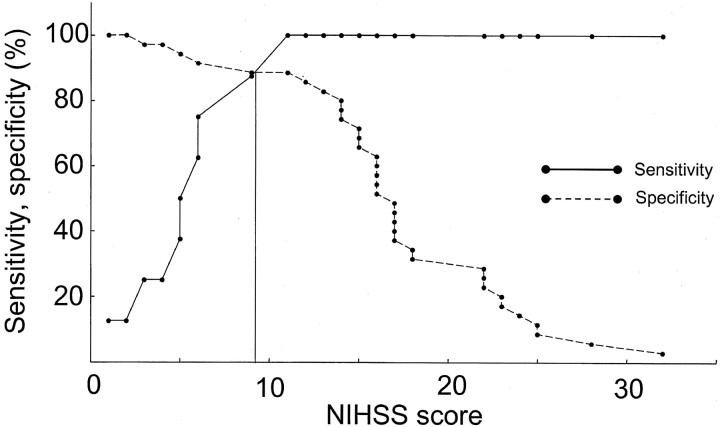
The median NIHSS score was 16 (range, 1–32). No significant differences were observed between NIHSS scores of patients with left- and right-sided stroke (P = .874, Mann-Whitney U test). NIHSS score was higher in the ICA group (median, 23; range, 6–32) than in the Branch (median, 15; range, 2–17; P = .02) or Normal (median, 6; range, 1–11; P < .001) groups but not higher than in the Stem group (median, 17; range, 11–25; P = .73). Patients in the Stem group displayed higher NIHSS scores than those in the Normal group (P < .001). In sensitivity-specificity curve analysis for predicting arterial occlusion, the optimal threshold value of the NIHSS score was 10 (Figs 1 and 2). Using an NIHSS score of 10 as the cut-off, sensitivity, specificity, positive predictive value, and negative predictive value for any arterial occlusion were 88.6%, 87.5%, 63.6%, and 96.9%, respectively.
Fig 1.
Distribution of the NIHSS score of four groups.
Fig 2.
Sensitivity-specificity curve analysis for predicting arterial occlusion. The optimal threshold value of the NIHSS score was 10.
MR imaging revealed 33 cortical infarcts, six subcortical infarcts, one lacunar infarct, and no ischemic lesions in three patients. Cortical and subcortical infarcts were present in seven and three of the 10 ICA group patients, respectively, 14 and 0 of the 14 MCA group patients, respectively, 10 and 0 of the 11 Branch group patients, respectively, and two and three of the eight Normal group patients, respectively. Lacunar infarct was observed in only one patient from the Normal group. One patient in the Branch group and three in the Normal group displayed no fresh lesions.
Discussion
Our study showed that an NIHSS score ≥10 represented the optimal value for predicting arterial occlusion in patients within 6 hr of stroke onset; 31 (97.0%) of 32 patients with NIHSS score ≥10 displayed arterial occlusion. Lewandowski et al (4) studied patients with stroke within 3 hr of stroke onset in the Emergency Management of Stroke bridging trial by using angiography. In that study, 17 (77.3%) of 22 patients with NIHSS scores ≥10 displayed occluded arteries in the carotid system. This minor discrepancy may be attributable to the differing interval between stroke onset and vascular imaging and to the methods to evaluate occlusive lesions.
Some studies have reported no significant differences in NIHSS scores between patients with MCA trunk occlusion and ICA/MCA tandem occlusion, a result that is compatible with the present results (9, 16). When the ICA is occluded, the severity of neurologic deficit is contingent on collateral blood flow through the anterior communicating or leptomeningial arteries from the ACA or posterior cerebral artery.
Our study included a small number of small artery diseases presenting as lacunar syndrome. We do not frequently perform IA-DSA but MRA to evaluate occlusive lesions in patients with lacunar stroke. A previous study reported that 205 (67.0%) of 306 patients with small artery disease had NIHSS scores of 0 to 6 (2). Patients with small artery disease may therefore be likely to achieve NIHSS scores <10.
In the present study, only one (3.0%) of 33 patients with NIHSS scores ≥10 displayed no arterial occlusion. In this case, neurologic symptoms improved immediately after angiography; this was attributed to spontaneous reopening of the occluded artery immediately before IA-DSA. Conversely, four (36.4%) of 11 patients with NIHSS scores <10 displayed arterial occlusions. Naylor et al (17) reported that patients with hyperacute stroke with MCA or ICA occlusions may occasionally display mild stroke severity or mimic lacunar events. Of the four patients, angiography in one case revealed occlusion of the right ICA origin and ipsilateral ACA and good collateral blood flow through the anterior communicating artery from the contralateral ICA system to the ipsilateral MCA. MR imaging of the brain revealed only a small infarct in ACA territory. In the remaining three patients with MCA branch occlusion, good leptomeningeal collateral blood supply from the ACA or posterior cerebral artery was present, and MR imaging revealed small infarcts. The mild neurologic deficits in these cases may therefore be due to good collateral flow.
The present study displayed some limitations. We did not perform IA-DSA for all patients with stroke within 6 hr of onset. In particular, IA-DSA was infrequently performed for patients older than 80 years and patients with lacunar stroke. This represents a source of selection bias in the study.
In conclusion, NIHSS score is associated with site of arterial occlusion in patients with hyperacute carotid ischemic stroke. An NIHSS score ≥10 is predictive of arterial occlusion in hyperacute ischemic stroke within 6 hr of onset.
References
- 1.Brott T, Adams HP Jr, Olinger CP, et al. Measurements of acute cerebral infarction: a clinical examination scale. Stroke 1989;20:864–870 [DOI] [PubMed] [Google Scholar]
- 2.Adams HP Jr, Davis PH, Leira EC, et al. Baseline NIH Stroke Scale score strongly predicts outcome after stroke: a report of the Trial of Org 10172 in Acute Stroke Treatment (TOAST). Neurology 1999;53:126–131 [DOI] [PubMed] [Google Scholar]
- 3.DeGraba TJ, Hallenbeck JM, Pettigrew KD, Dutka AJ, Kelly BJ. Progression in acute stroke: value of the initial NIH stroke scale score on patient stratification in future trials. Stroke 1999;30:1208–1212 [DOI] [PubMed] [Google Scholar]
- 4.Lewandowski CA, Frankel M, Tomsick TA, et al. Combined intravenous and intra-arterial r-TPA versus intra-arterial therapy of acute ischemic stroke: Emergency Management of Stroke (EMS) Bridging Trial. Stroke 1999;30:2598–2605 [DOI] [PubMed] [Google Scholar]
- 5.Arnold M, Schroth G, Nedeltchev K, et al. Intra-arterial thrombolysis in 100 patients with acute stroke due to middle cerebral artery occlusion. Stroke 2002;33:1828–1833 [DOI] [PubMed] [Google Scholar]
- 6.Ernst R, Pancioli A, Tomsick T, et al. Combined intravenous and intra-arterial recombinant tissue plasminogen activator in acute ischemic stroke. Stroke 2000;31:2552–2557 [DOI] [PubMed] [Google Scholar]
- 7.Roberts HC, Dillon WP, Furlan AJ, et al. Computed tomographic findings in patients undergoing intra-arterial thrombolysis for acute ischemic stroke due to middle cerebral artery occlusion: results from the PROACT II Trial. Stroke 2002;33:1557–1565 [DOI] [PubMed] [Google Scholar]
- 8.Fink JN, Selim MH, Kumar S, et al. The association of National Institutes of Health Stroke Scale scores and acute magnetic resonance imaging stroke volume equal for patients with right- and left-hemisphere ischemic stroke? Stroke 2002;33:954–958 [DOI] [PubMed] [Google Scholar]
- 9.El-Mitwalli A, Saad M, Christou I, Malkoff M, Alexandrov AV. Clinical and sonographic patterns of tandem internal carotid artery/middle cerebral artery occlusion in tissue plasminogen activator-treated patients. Stroke 2002;33:99–102 [DOI] [PubMed] [Google Scholar]
- 10.Koga M, Kimura K, Minematsu K, Yamaguchi T. Ultrasonographic prediction of patients’ outcome in hyperacute ischemic stroke. Eur J Ultrasound 2002;15:1–8 [DOI] [PubMed] [Google Scholar]
- 11.Verro P, Tanenbaum LN, Borden NM, Sen S, Eshkar N. CT angiography in acute ischemic stroke: preliminary results. Stroke 2002;33:276–278 [DOI] [PubMed] [Google Scholar]
- 12.Derex L, Nighoghossian N, Hermier M, Adeleine P, Froment JC, Trouillas P. Early detection of cerebral artery occlusion on magnetic resonance angiography: predictive value of the baseline NIHSS score and impact on neurological outcome. Cerebrovasc Dis 2002;13:225–229 [DOI] [PubMed] [Google Scholar]
- 13.Bozzao L, Fantozzi LM, Bastianello S, et al. Ischaemic supratentorial stroke: angiographic findings in patients examined in the very early phase. J Neurol 1989;236:340–342 [DOI] [PubMed] [Google Scholar]
- 14.Bozzao L, Fantozzi LM, Bastianello S, Bozzao A, Fieschi C. Occlusion of the extracranial internal carotid artery in the acute stroke; angiographic findings within 6 hours. Acta Neurochir (Wien) 1989;100:39–42 [DOI] [PubMed] [Google Scholar]
- 15.Swieten JC, Koudstaal PJ, Visser MC, Schouten HJ, Gijn J. Interobserver agreement for the assessment of handicap in stroke patients. Stroke 1988;19:604–607 [DOI] [PubMed] [Google Scholar]
- 16.Linfante I, Linas RH, Selim M, et al. Clinical and vascular outcome in internal carotid artery versus middle cerebral artery occlusions after intravenous tissue plasminogen activator. Stroke 2002;33:2066–2071 [DOI] [PubMed] [Google Scholar]
- 17.Naylor AR, Sandercock PA, Sellar RJ, Warlow CP. Patterns of vascular pathology in acute, first-ever cerebral infarction. Scott Med J 1993;38:41–44. [DOI] [PubMed] [Google Scholar]