Abstract
BACKGROUND AND PURPOSE: Desmoplastic infantile tumors (DITs) are rare supratentorial tumors of infancy with a favorable prognosis. Radiologic and histologic features of DIT are misleading, and DIT may be misinterpreted as a malignant lesion. We have studied the usefulness of MR imaging in the diagnosis of these tumors.
METHODS: Between 1995 and 2002, six DITs were diagnosed in young children at our institution. Neuroimaging, age at diagnosis, sex, clinical presentation, symptoms duration, follow-up, and development were studied retrospectively. Contrast-enhanced CT and MR images were available. MR study included T1-, T2-, and postgadolinium T1-weighted sequences in the axial, sagittal, and coronal planes.
RESULTS: These tumors were massive and predominantly cystic, with preferential frontal and parietal involvement. Typically, a DIT appears as a hypointense cystic mass with an isointense peripheral solid component on T1-weighted MR images. The peripheral solid component enhances after gadolinium administration. On T2-weighted MR images, the cystic component is hyperintense and the solid portion isointense or heterogeneous. The cystic portion is usually located deep inside the lesion, whereas its solid portion is peripheral. Meningeal enhancement and thickening adjacent to the solid portion of the tumor, calcifications, bone abnormalities adjacent to the tumor consisting of thinning and deformation were noted in 50% of our cases. Edema was usually absent or moderate. Median follow-up was 32 months, and no recurrence was noted except for one atypical case with incomplete excision, which led to the patient’s death.
CONCLUSION: Despite their malignant appearance, MR imaging features of DIT may help in the diagnosis and obviate unnecessary chemotherapy or radiation therapy.
First described in 1984 by Taratuto et al (1), desmoplastic infantile tumors (DITs) are rare and usually benign lesions affecting infants. They are characterized by a radiologically malignant appearance. Neuroimaging typically shows a massive cystic and solid, well-defined supratentorial lesion. Two histologic forms have been described: desmoplastic infantile astrocytoma (DIA) and desmoplastic infantile ganglioglioma (DIG) (1, 2).
We report a series of six cases of DIAs and DIGs. Clinical, neuroradiologic, and pathologic results and treatment were reviewed, and additional neuroradiologic findings helpful to confirming diagnosis are reported.
Methods
Case Selection and Clinical Findings
Between 1995 and 2002, six DITs were diagnosed in young children at our institution on the basis of pathologic findings. Age at diagnosis, sex, clinical presentation, duration of symptoms before surgical excision, follow-up duration, and development were recorded with a retrospective analysis that is summarized in Table.
Clinical findings
| Finding | Case (No.) |
|||||
|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |
| Sex/age (mo) | M/5 | M/39 | M/5 | F/2.5 | F/48 | M/5 |
| Symptom duration | 3 wk | 1 mo | 2 mo | 2.5 mo | 2 mo | 0 |
| Increasing head circumference | Y | N | N | Y | N | N |
| Seizures | N | Y | Y | N | N | N |
| Increased ICP | Y | N | N | N | Y | N |
| Other symptoms | Skull deformation | . . . | . . . | . . . | Visual field | . . . |
| Treatment | Total excision | Total excision | Subtotal excision | Total excision | Partial excision | Total excision |
| Follow-up | 18 mo, norm dvpt, upper limb paralysis | 14 mo, norm dvpt | 3 y 6 mo, norm dvpt, dysphasia | 24 mo norm dvpt | 13 mo death | 7 y, norm dvpt |
Note.—Yes (Y), no (N), normal development (norm dvpt).
Neuroimaging
In all cases, preoperative CT findings obtained with iodinated contrast material and preoperative and postoperative MR findings were available. MR images were obtained in our institution by use of a 1.5-T unit equipped with a head coil (Vision, Siemens, Erlangen, Germany). The study included T1-weighted (TR = 700 ms, TE = 14 ms, matrix = 144 × 256, flip angle = 70°, field of view = 250 mm, rectangular field of view = 75%, acquisitions = 2), T2-weighted (TR = 5000 ms, TE = 120 ms, matrix = 300 × 512, flip angle = 180°, field of view = 250 mm, rectangular field of view = 75%, acquisitions = 2), and postgadolinium T1-weighted images. All sequences were performed in the axial, sagittal, and coronal planes.
Follow-up comprised MR imaging performed at regular intervals. The patients were followed up for a median of 32 months (range, 13 months to 7 years). Preoperative and postoperative neuroradiologic examinations were reviewed by two radiologists (G.T., H.B.) who analyzed the following anatomic features: location, size, shape, solid and cystic components, mass enhancement, meningeal enhancement, mass effect, edema, and abnormalities within the dome of the skull.
Pathologic Analysis
Surgical specimens were fixed in formalin and paraffin embedded. In each case, histologic examinations included routine histologic and immunohistochemical techniques. Reticulinum stains and immunohistochemical reactions were performed by using the following antibodies: glial fibrillary acidic protein (GFAP), phosphorylated neurofilament, synaptophysin. Proliferation index (MIB-1) was performed in three cases.
Results
Clinical Features
Four male and two female patients were included in the study. Their ages ranged from 3.5 months to 4 years (mean, 17 months). Four patients were younger than 5 months. No one had any notable medical history.
Increasing head circumference occurred in two patients (cases 1 and 4), elevated intracranial pressure in two patients (cases 1 and 5), and seizures in two patients (cases 2 and 3). The other symptoms included skull deformation (case 1) and visual field loss (case 5).
Duration of symptoms ranged from 3 weeks to 2.5 months. In one patient (case 6), tumor discovery was fortuitous after a benign head injury. Mean duration of symptoms was 1.5 months.
Surgical excision was performed in all patients. Macroscopically, total excision was not possible in two patients: a sylvian vessel attachment (case 3) and massive blood loss (case 5) made a total resection impossible (Table). The mass was always strongly adhesive to the dura mater, and surgery was hemorrhagic in every case. No adjuvant therapy was administered.
Neuroimaging
In all patients, a massive peripheral lesion was found in the supratentorial space, with meningeal contact (Figs 1 [case 1] and 2 [case 3]).
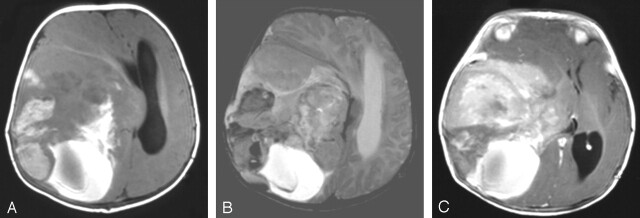
Fig 1.
Case 1. Axial T1- (A), axial T2- (B), and axial postgadolinium T1-weighted (C) MR images show a large supratentorial and heterogeneous tumor involving the frontotemporoparietal lobes with a major mass effect. The solid part of the tumor has two components. The peripheral part is heterogeneous and moderately hyperintense on T1-weighted images and shows low signal intensity on T2-weighted images suggestive of intratumoral bleeding; the medial part is enhanced after gadolinium injection. The posterior cystic part of the lesion is of high signal intensity on T1- and T2-weighted images, which is compatible with a recent bleeding episode. Radiologic appearance is suggestive of a malignant lesion.
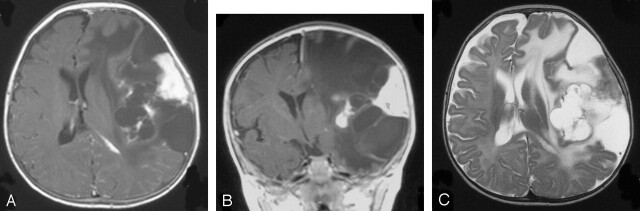
Fig 2.
Case 3. Axial postgadolinium T1- (A), coronal postgadolinium T1- (B), and axial T2-weighted (C) MR images show a typically large supratentorial mass with a peripheral solid and a deep cystic component. The peripheral solid component is enhanced after gadolinium injection (A and B) and is associated with a meningeal thickening and enhancement. Note the major mass effect on the midline and the ventricles.
The tumor occupied at least two hemispheric lobes in four patients (cases 1, 3, and 4, frontotemporoparietal; case 5, frontoparietooccipital) and was limited to a single lobe in two (case 2, temporal; case 6, occipital).
The size (largest diameter) ranged from 45 to 125 mm (mean, 83 mm). Despite their large size, no tumor communicated with the ventricles.
Edema was absent (cases 1, 2, 4, 5) or moderate (cases 3, 6), and mass effect on midline structures was noted in every case except in one patient (case 2).
At CT, the lesion appeared heterogeneous with a cystic hypoattenuating portion and a solid portion enhanced after iodine injection in four patients (cases 1, 3, 4, and 6). In two patients (cases 2 and 5) the tumor was solid, and in three patients (cases 1, 2, and 5) calcifications were noted. On T1-weighted images and T2-weighted images (Fig 2C [case 3]), the lesion appeared to be predominantly cystic in four patients (cases 1, 3, 4, and 6). In one patient, the tumor was predominantly solid with small cysts (case 2), and in one patient the tumor was initially solid (case 5), but on follow-up, cyst occurrence was noted. Cysts were isointense on T1-weighted images and isointense on T2-weighted images relative to CSF except in one patient (case 1) in whom the lesion showed higher signal intensity relative to CSF on T1-weighted images.
Gadolinium injection induced the enhancement of the solid portion of the tumor in all cases (Fig 2A and B [case 3]). The latter had a peripheral location, and meningeal enhancement was noted. In three cases (cases 2, 4, and 5), meningeal enhancement was found adjacent to the tumor (Fig 2A and B [case 3]). In all cases, the mass (cyst + solid portion) showed well-defined margins. The bone adjacent to the tumor was thin or deformed in three cases (cases 1, 4, and 5).
Histologic and Immunohistochemical Findings
Various components were found, and histologic appearance was polymorphic. In all cases, fusiform astrocytic cells forming either a fascicle or storiform or whorled pattern were found (Fig 3). Glial cells express GFAP and vimentin. They were dissociated from each other by edema or hemorrhagic areas. In four cases (cases 1, 2, 3, 6), neuronal heterogeneous elements ranging from small to big cells were found. Neuronal differentiation is characterized by synaptophysin and neurofilament protein staining (Fig 4). Reticulin stain was positive in all the cases and revealed predominantly in a fascicle pattern. Vascularization was variable. In two cases (cases 1 and 5), it was profuse but without endothelial proliferation. Case 5 was atypical, showing mitosis, necrosis, and giant multinucleate cells. MIB-1, realized in three cases (cases 1, 2, and 4), varied from 2–5%.
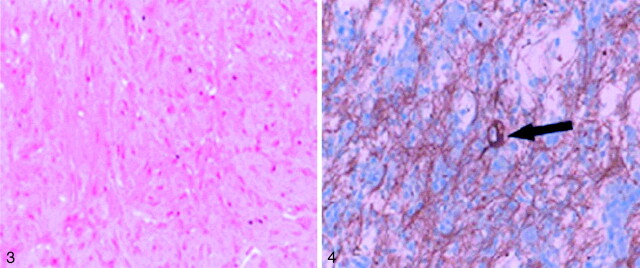
Fig 3.
Standard hematoxylin coloration (magnification ×250) shows the fusiform astrocytic cells forming a dense fascicle pattern.
Fig 4.
DIG with marked neurofilament immunostain (magnification ×400) due to the presence of neural cells (black arrow).
Follow-up
The follow-up duration ranged from 13 months to 7 years, with a mean of 32 months. To date, five patients are alive. One patient (case 5) died after 13 months. In this case, the excision was partial and tumorrecurrence noted despite chemotherapy and radiation therapy. The other children (cases 1–4 and 6) showed normal development, but two patients (cases 1 and 3) had focal deficiency: upper limb paralysis (case 1) and dysphasia (case 3). Two patients (cases 2 and 4) still adhere to an anticonvulsive treatment regimen.
Discussion
In our series, MR and CT findings are helpful in the diagnosis of DITs. The tumor appears as a massive, cystic supratentorial lesion. In addition, the cystic portion, uni- or multilocular, seems usually to be located deep inside the lesion, whereas its solid portion is peripheral (3). No communication between the deep cysts and the lateral ventricle was detected (2–4). Meningeal enhancement and thickening adjacent to the solid portion of the tumor were noted in 50% of our cases. Some cases of bone abnormalities adjacent to a tumor consist of skull bossing, thinning, and deformation that have been reported in the literature (2, 3, 5–9) were present in 50% of our patients. We noted calcifications in 50% of our cases, a sign that, to our knowledge, has been reported by only one other author (10). Typically, DIT appears as a hypointense cystic mass with an isointense peripheral solid component on T1-weighted MR images. The peripheral solid component enhances with gadolinium administration. Serra et al (11) noted the conspicuous lack of enhancement surrounding the circumference of the cysts. On T2-weighted MR images, the cystic component is hyperintense and the solid portion isointense or heterogeneous. Edema is usually absent or moderate (3). Desmoplastic tumors are superficial lesions with frequent dura mater adherent portions. At least in infants, these findings seem to be characteristic. Therefore, the diagnosis of DIT can be proposed despite these malignant appearances.
DITs are large, with a reported median size reaching 80 mm. Plurilobar invasion is usual. The tumor involves preferentially the frontal and parietal lobes (3). The occipital lobe is usually spared, and its invasion has been described just once (11). The sheer size of these lesions and the relative lack of symptoms in children with such masses are most notable. This suggests a slow growth of the tumors and probable congenital origin (2, 3, 12).
The male-female ratio is 1.7:1 (13). DITs are generally found in the first 18 months of a patient’s life (13). Analysis of the literature shows the median age at diagnosis to be more precocious at about 5 or 6 months. In terms of clinical presentation, 11 cases are described in children from 18 months to 9 years of age (3, 14–20), and four cases of noninfantile desmoplastic tumors are described (12, 21, 22). In our series, a child was 4 years old at diagnosis and another 3.5 years old. This tumor is not exclusively seen in pediatric patients; it may be diagnosed in older patients. The clinical presentation of DIT is stereotyped: its large size in pediatric patients results in increasing head circumference, tense and bulging fontanelles, paresis, and seizures. These tumors have a favorable prognosis, but recurrence and death have been noted in some atypical cases.
With respect to atypical radiologic features, two cases of nonhemispheric tumors have been described. They were located in the ventral diencephalon (23) and in the suprasellar and hypothalamic region (24). Their development was atypical, leading to the patients’ deaths. In the second case, the tumor was solid, and metastases were found at presentation. Other cases of solid tumor with a long survival have been described in the literature (7). Sperner et al (19) described a macroscopically solid temporal mass, but cystic parts of the tumor were detected histologically. Tseng et al (17) described a solid temporal tumor that became cystic during its evolution. In our series, a similar evolution pattern has been found in one patient (case 5): after resection of the solid part of the lesion, cysts appeared secondarily. The tumoral behavior and the clinical evolution were atypical, and the patient died after 13 months.
The radiologic differential diagnoses of DIT include primitive neuroectodermal tumor, ependymoma, ganglioglioma, and dysembryoplastic neuroepithelial tumor (3).
Primitive neuroectodermal tumors may have a prominent cystic component, but usually this tumor lies deep in the white water and the solid component is less peripheral. Supratentorial ependymoma has a parietal predilection, but with a paraventricular location (3). Ganglioglioma is usually located within the temporal lobe (16). It is smaller than a DIT, with both a cystic and a solid part. Dysembryoplastic neuroepithelial tumor is usually a cortical supratentorial tumoral lesion without perilesional edema. Mass effect is absent, and a cystic component is occasionally found (25).
The DIT group include both DIA and DIG. A consistent feature of these tumors is desmoplasia associated with cellular areas (15). Desmoplasia is defined by augmentation or modification of conjunctive tissues related to the presence of neoplastic cells with the formation of a collagen-rich extracellular matrix. The densely desmoplastic areas tend to have a well-defined border with the adjacent brain, whereas the areas of high neural cellularity show focal microinvasion of the brain interface (4). In the cellular areas, four neoplastic cell types are encountered; the most frequently found cell type is the astrocytic tumor cell. The leptomeningeal portion of the mass consists of spindled elongated astrocytic cells that are arranged in a fasciculated pattern mimicking a mesenchymal tumor (14, 26). Neural cells represent the second cell type within the tumor and show variable degrees of maturation (9). This cell type allows differentiation between DIG and DIA. Neural cells are specific of DIG. More primitive cells corresponding to neuroepithelial cells are commonly present and account for approximately 5% of the tumor cells (27). The fourth cell type described is the Schwann cell (7, 9), but the presence of this cell type is still controversial.
Mitotic activity and necrosis are uncommon (4). In some cases, angiomatoid vessels are found, but microvascular proliferation is not obvious (28, 29). Because histologic features on brain biopsies could have similarities with highly aggressive lesions, the interpretation of these findings must be done with care to avoid unnecessary chemotherapy or radiation therapy (13, 30).
DITs represent a formidable surgical challenge and are associated with a good prognosis after surgery (1, 4, 27, 31–33). Complete surgical excision is possible in 70% of the cases reported in the literature. When performed, no recurrence is noted and no further treatment is necessary. In case of incomplete resection, favorable development is noted in 73% of the patients and adjuvant treatment must be discussed on a case-by-case basis.
Conclusion
DITs are rare and usually benign lesions. These tumors represent a formidable surgical challenge regarding their size, dural attachment, and hemorrhagic risk. Radiologists and pathologists unfamiliar with these lesions may interpret the radiologic and histologic features as highly aggressive, resulting in unnecessary chemotherapy or radiation therapy. We have reported herein the main MR imaging features of these tumors. Some additional neuroradiologic features including a solid superficial portion, a meningeal enhancement and thickening adjacent to the solid portion of the tumor, and calcifications may help in the diagnosis.
References
- 1.Taratuto AL, Monges J, Lylyk P, Leiguarda R. Superficial cerebral astrocytoma attached to dura: report of six cases in infants. Cancer 1984;54:2505–2512. [DOI] [PubMed] [Google Scholar]
- 2.VandenBerg SR, May EE, Rubinstein LJ, et al. Desmoplastic supratentorial neuroepithelial tumors of infancy with divergent differentiation potential (“desmoplastic infantile gangliogliomas”): report on 11 cases of a distinctive embryonal tumor with favorable prognosis. J Neurosurg 1987;66:58–71. [DOI] [PubMed] [Google Scholar]
- 3.Rypens F, Esteban MJ, Lellouch-Tubiana A, et al. Desmoplastic supratentorial neuroepithelial tumours of childhood: imaging in 5 patients. Neuroradiology 1996;38:S165–S168 [DOI] [PubMed] [Google Scholar]
- 4.VandenBerg SR. Desmoplastic infantile ganglioglioma and desmoplastic cerebral astrocytoma of infancy. Brain Pathol 1993;3:275–281 [DOI] [PubMed] [Google Scholar]
- 5.Paulus W, Schlote W, Perentes E, et al. Desmoplastic supratentorial neuroepithelial tumours of infancy. Histopathology 1992;21:43–49 [DOI] [PubMed] [Google Scholar]
- 6.Tenreiro-Picon OR, Kamath SV, Knorr JR, et al. Desmoplastic infantile ganglioglioma: CT and MRI features. Pediatr Radiol 1995;25:540–543 [DOI] [PubMed] [Google Scholar]
- 7.Rushing EJ, Rorke LB, Sutton L. Problems in the nosology of desmoplastic tumors of childhood. Pediatr Neurosurg 1993;19:57–62 [DOI] [PubMed] [Google Scholar]
- 8.Rothman S, Sharon N, Shiffer J, et al. Desmoplastic infantile ganglioglioma. Acta Oncol 1997;36:655–657. [DOI] [PubMed] [Google Scholar]
- 9.Ng THK, Fung CF, Ma LT. The pathological spectrum of desmoplastic infantile gangliogliomas. Histopathology 1990;16:235–241 [DOI] [PubMed] [Google Scholar]
- 10.Dunnick NR. Image interpretation session: 1999: desmoplastic infantile ganglioglioma. Radiographics 2000;20:276–278 [PubMed] [Google Scholar]
- 11.Serra A, Strain J, Ruyle S. Desmoplastic cerebral astrocytoma of infancy: report and review of the imaging characteristics. AJR Am J Roentgenol 1996;166:1459–1461 [DOI] [PubMed] [Google Scholar]
- 12.Kuchelmeister K, Bergmann M, von Wild K, et al. Desmoplastic ganglioglioma: report of two non-infantile cases. Acta Neuropathol (Berlin) 1993;85:199–204 [DOI] [PubMed] [Google Scholar]
- 13.Taratuto AL, Vandenberg SR, Rorke LB. Desmoplastic infantile astrocytoma and ganglioglioma. In: Kleihues P, Cavenee WK, eds. Pathology and genetics: tumors of the nervous system. Lyon, France: IARC Press2000;:99–102
- 14.Mallucci C, Lellouch-Tubiana A, Salazar C, et al. The management of desmoplastic neuroepithelial tumours in childhood. Childs Nerv Syst 2000;16:8–14 [DOI] [PubMed] [Google Scholar]
- 15.Rout P, Santosh V, Mahadevan A, et al. Desmoplastic infantile ganglioglioma: clinical and immunohistological study of four cases. Childs Nerv Syst 2002;18:463–467 [DOI] [PubMed] [Google Scholar]
- 16.Hasegawa Y, Hayabuchi Y, Namba I, et al. Cytologic features of desmoplastic infantile ganglioglioma: a report of two cases. Acta Cytol 2001;45:1037–1042 [DOI] [PubMed] [Google Scholar]
- 17.Tseng JH, Tseng MY, Kuo MF, et al. Chronological changes on magnetic resonance images in a case of desmoplastic infantile ganglioglioma. Pediatr Neurosurg 2002;36:29–32 [DOI] [PubMed] [Google Scholar]
- 18.Kurose A, Beppu T, Miura Y, et al. Desmoplastic cerebral astrocytoma of infancy intermingling with atypical glial cells. Pathol Int 2000;50:744–749 [DOI] [PubMed] [Google Scholar]
- 19.Sperner J, Gottschalk J, Neumann K, et al. Clinical, radiological and histological findings in desmoplastic infantile ganglioglioma. Childs Nerv Syst 1994;10:458–462; discussion 462–463 [DOI] [PubMed] [Google Scholar]
- 20.Park JP, Dossu JR, Rhodes CH. Telomere associations in desmoplastic infantile ganglioglioma. Cancer Genet Cytogenet 1996;92:4–7 [DOI] [PubMed] [Google Scholar]
- 21.Galatioto S, Gullotta F. Desmoplastic non-infantile ganglioglioma. J Neurosurg Sci 1996;40:235–238 [PubMed] [Google Scholar]
- 22.Marti A, Almostarchid B, Maher M, Saidi A. Desmoplastic non-infantile ganglioglioma: case report. J Neurosurg Sci 2000;44:150–154 [PubMed] [Google Scholar]
- 23.Komori T, Scheithauer BW, Parisi JE, et al. Mixed conventional and desmoplastic infantile ganglioglioma: an autopsied case with 6-year follow-up. Mod Pathol 2001;14:720–726. [DOI] [PubMed] [Google Scholar]
- 24.Setty SN, Miller DC, Camras L et al. Desmoplastic infantile astrocytoma with metastases at presentation. Mod Pathol 1997;10:945–951 [PubMed] [Google Scholar]
- 25.Fernandez C, Girard N, Paz Paredes A, et al. The usefulness of MR imaging in the diagnosis of dysembryoplastic neuroepithelial tumor in children: a study of 14 cases. AJNR Am J Neuroradiol 2003;24:829–834. [PMC free article] [PubMed] [Google Scholar]
- 26.Craver RD, Nadell J, Nelson JS. Desmoplastic infantile ganglioglioma. Pediatr Dev Pathol 1999;2:582–587. [DOI] [PubMed] [Google Scholar]
- 27.Vandenberg SR Desmoplastic infantile ganglioglioma: a clinicopathologic review of sixteen cases. Brain Tumor Pathol 1991;8:25–31. [Google Scholar]
- 28.De Chadarevian JP, Pattisapu JV, Faerber EN. Desmoplastic cerebral astrocytoma of infancy: light microscopy, immunocytochemistry, and ultrastructure. Cancer 1990;66:173–179 [DOI] [PubMed] [Google Scholar]
- 29.Louis DN, von Deimling A, Dickersin GR, et al. Desmoplastic cerebral astrocytomas of infancy: a histopathologic, immunohistochemical, ultrastructural, and molecular genetic study. Hum Pathol 1992;23:1402–1409 [DOI] [PubMed] [Google Scholar]
- 30.Duffner PK, Burger PC, Cohen ME, et al. Desmoplastic infantile gangliogliomas: an approach to therapy. Neurosurgery 1994;34:583–589 [DOI] [PubMed] [Google Scholar]
- 31.Rodriguez-Morales EL, Correa Rivas MS, Colon CE. Desmoplastic astrocytoma of infancy: a case report with histopathologic and immunohistochemistry profile. P R Health Sci J 2002;21:129–132 [PubMed] [Google Scholar]
- 32.Finizio FS. CT and MRI aspects of supratentorial hemispheric tumors of childhood and adolescence. Childs Nerv Syst 1995;11:559–567 [DOI] [PubMed] [Google Scholar]
- 33.Martin DS, Levy B, Awwad EE, Pittman T. Desmoplastic infantile ganglioglioma: CT and MR features. AJNR Am J Neuroradiol. 1991;12:1195–1197 [PMC free article] [PubMed] [Google Scholar]