Abstract
BACKGROUND AND PURPOSE: Postsurgical evaluation of patients with intracranial aneurysms is important because there is a risk of regrowth and subarachnoid hemorrhage from residual aneurysms. We assessed the role and value of 3D angiography in this evaluation.
METHODS: Images from both 3D angiography and digital subtraction angiography (DSA) were preoperatively obtained in 71 consecutive patients with 88 intracranial clipped aneurysms. We analyzed the incidence of postoperative residual aneurysms and compared the 3D angiographic and DSA images in these patients.
RESULTS: With 3D angiography, 37 residual aneurysms were found among 88 clipped aneurysms. These comprised 12 large residual necks and 25 small, dog-eared remnants. DSA demonstrated 12 large, residual necks and only six small, dog-eared remnants. 3D angiography also demonstrated the clips, aneurysms, and parent arteries simultaneously, enabling us to appreciate the clip-aneurysm configuration completely.
CONCLUSION: 3D angiography allowed us to detect more residual aneurysms after surgical clip placement than did conventional DSA, although none of those detected on only 3D angiography were judged to require additional treatment. 3D angiography may provide baseline data for the long-term follow-up of postsurgical aneurysms.
An accurate postoperative assessment of aneurysm obliteration after clip placement of the aneurysm neck is essential in treating patients with intracranial aneurysms. Regrowth and bleeding of aneurysms from a residual neck have been reported (1–3). Postoperative conventional angiography is generally accepted as a standard tool for evaluating aneurysms after neck clip placement because MR and CT angiography have their limitations. The purpose of our study was to assess he value of 3D angiography after intracranial aneurysm clip placement.
Methods
Case Collection
We reviewed 71 consecutive cases with 88 clipped cerebral aneurysms that underwent postoperative angiography with rotational angiography and 3D reconstruction (3D angiography) between December 2001 and September 2003. Follow-up angiography was recommended in all patients who underwent clip placement for intracranial aneurysms; however, some declined. Our series comprised 51 women and 20 men aged 24–74 years. Thirty-three aneurysms were located in the middle cerebral artery, 26 were in the anterior cerebral artery, 23 were in the internal carotid artery (ICA), and six were in the posterior circulation.
The interval between surgery and angiography ranged from 1 day to 13 years. We divided the cases into two groups according to this interval: 1) an immediate follow-up group in which the angiography was performed within a month of surgery and 2) a delayed follow-up group in which the angiography was performed 1 month or more after the operation. Forty-nine cases with 55 clipped aneurysms were allocated to the first group, and 22 cases with 33 clipped aneurysms to the second.
The clips applied were mainly Yasargil clips (Aesculap; Center Valley, PA) made of a cobalt alloy (Phynox) or a titanium alloy. For giant aneurysms, we used Sugita clips (Mizho America; Beverly, MA) made of a cobalt chrome alloy (Elgiloy) or a titanium alloy.
Angiography
Angiographic examinations were performed by using a commercially available biplanar angiographic unit (Integris Allura; Philips Medical Systems, Best, the Netherlands) with an image intensifier matrix of 1024 × 1024. In performing digital subtraction angiography (DSA), we tried to optimize the angle to define the clip–aneurysm relationships as well as possible. Usually, we referenced the angle from rotational angiography.
During rotational angiography, the C arm rotated over a 240° range at a speed of 55° per second for about 4.4 seconds. Contrast medium (Iomeron, 300 mg of iodine per mL; Bracco, Milan, Italy) was injected at a flow rate of 4–5 mL per second, which resulted in total injected volumes of 16–20 mL for each rotational angiographic session in the ICA. A flow rate of 2.5–3.5 mL per second resulted in total injected volumes of 10–14 mL into the vertebral artery. Sometimes, a 1-second exposure delay was applied to allow an aneurysm to be fully filled. Image data were transferred to a workstation (Octane; Silicon Graphics, Mountain Hill, CA) to reconstruct the 3D images with volume (Integris 3D-RA software, release 3.2, Octane; Silicon graphics). We analyzed snapshots, which consisted of six basic views (anterior, posterior, both lateral, superior, and inferior views) and views from arbitrary angles and cutting views to visualize the clip–aneurysm configurations well.
Analysis of Residual Aneurysms
Two neuroradiologists (M.H.H., B.J.K.) interpreted the DSA and 3D angiographic images, focusing on any residual aneurysms. Each study was not read independently. We classified residual (or recurrent) aneurysms into the two groups: 1) aneurysms with a large residual neck or recurrent aneurysms and 2) aneurysms with a small, dog-ear residue (1) (Figs 1 and 2). We also compared the abilities of DSA and 3D angiography to depict the residual aneurysms.
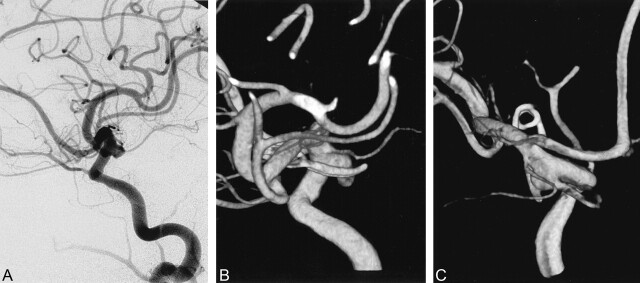
Fig 1.
Case 21. Large residual aneurysm.
A and B, Postoperative DSA image (A) and 3D angiogram (B) obtained 6 months after surgery for a posterior communicating artery aneurysm shows residual aneurysm.
C, Posterior 3D angiogram reveals the relationship of the clip, residual aneurysm, posterior communicating artery, and anterior choroidal artery.
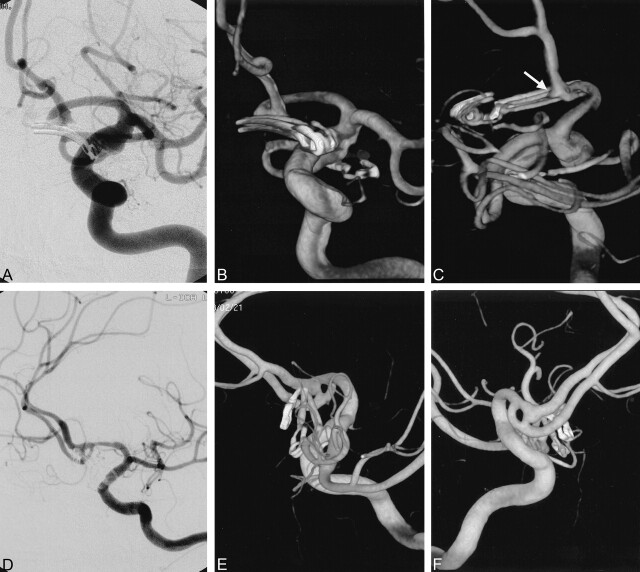
Fig 2.
Small, dog-eared remnant aneurysm.
A—C, Case 24. Postoperative DSA (A) performed 8 days after surgery for anterior and posterior communicating artery aneurysms shows no residual. Anterior 3D angiogram (B) shows the vessels and clips in the two aneurysms well. Posterior 3D angiogram (C) reveals a tiny, dog-ear remnant in the anterior communicating artery region (arrow).
D–F, Case 5. Postoperative DSA (D) of a clipped anterior communicating artery aneurysm shows no definite residual. Anterior oblique and posterior oblique 3D aneurysms (E, F) reveal a residual aneurysm near the ends of the clips and its relationship with a fenestration.
Results
In this series of 71 consecutive cases with 88 clipped cerebral aneurysms, 37 residual (or recurrent) aneurysms (42%) were detected by means of 3D angiography. DSA depicted 18 of the 37 residual aneurysms (Table). Twenty-seven of 55 aneurysms (49%) in the immediate follow-up group proved to have residual portions that the clips did not cover completely. DSA identified 11 of these residual aneurysms. Furthermore, DSA showed only three of 19 small, dog-eared remnants. On the other hand, 3D angiography showed all residual aneurysms (i.e., small dog-eared remnants or large residual necks and/or sacs).
TABLE 1:
Detection of residual aneurysms with DSA and 3DA
| Group* and Residual Aneurysms | DSA | 3D Angiography |
|---|---|---|
| Immediate (27 of 55) | ||
| Small | 3 | 19 |
| Large | 8 | 8 |
| Delayed (10 of 33) | ||
| Small | 1 | 6 |
| Large | 4 | 4 |
| Total (37 of 88) | 16 | 37 |
Immediate is the immediate follow-up group in which angiography was performed within 1 month of surgery, and delayed, delayed follow-up group in which angiography was performed at least 1 month after surgery. Small, is a small, dog-ear remnant aneurysm, and large, a large residual neck and/or sac.
Overall, 13 residual aneurysms were found in the anterior cerebral artery, 11 were in the middle cerebral artery, 10 were in the internal cerebral artery, and three were in the posterior circulation. Twelve large residual neck or recurrent aneurysms were visualized during DSA and 3D angiography. Of 25 small, dog-eared remnants, only four were detected on DSA, whereas 3D angiography depicted all 25.
No further treatments were performed for small, dog-eared remnants. However, four large residual aneurysms were embolized with detachable coils and occluded completely.
The number of clips used in this series was one in 58 cases, two in 24 cases, and three or more in six cases. 3D angiography demonstrated the aneurysm–clip configuration well, irrespective of the type or number of clips applied and the complexities of the aneurysms or clips applied (Figs 3 and 4). In cases of complex aneurysms and multiple clips (as well as with single clips) 3D angiography helped us understand the complex anatomy by providing various angles and by allowing visualization of clips and vessels at the same time. This ability was especially useful for understanding the anatomy of aneurysms with three or more clips.
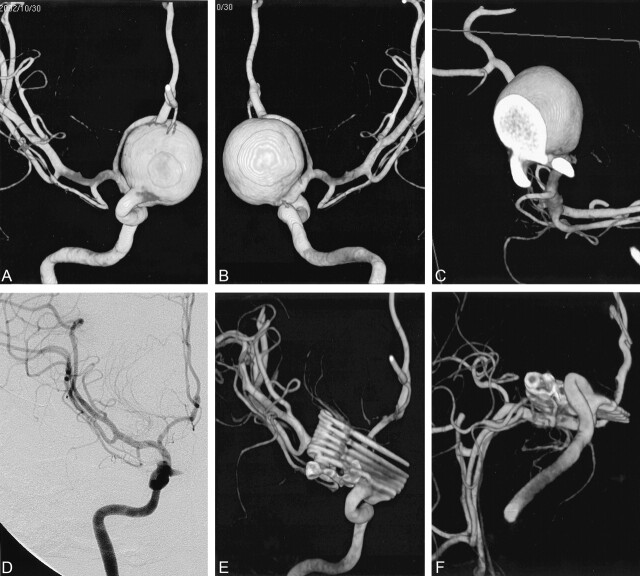
Fig 3.
Case 47. Giant aneurysm of the ICA.
A and B, Anterior and posterior 3D angiograms show the aneurysm in the supraclinoid region, displacing the anterior cerebral artery upward.
C, Section through the aneurysm neck shows its broad nature.
D, Postoperative DSA shows well-reconstructed ICA and small residue in the proximal part of the aneurysm.
E and F, Anterior and inferior postoperative 3D angiograms reveal seven clips, without artifact, used for reconstruction of the ICA and proximal residual aneurysm.
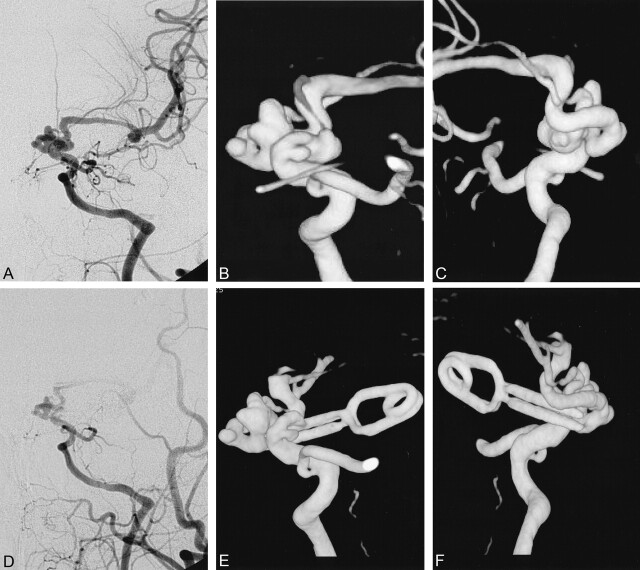
Fig 4.
Case 65. Dolichocarotid aneurysms.
A–C, Anterior and posterior preoperative DSA and 3D angiographic views show a complex aneurysms involving the distal ICA.
D, On postoperative DSA, it is hard to see which part of the aneurysms is clipped.
E and F, Anterior and posterior 3D angiograms reveal that the lateral part of the aneurysms is clipped, and part of ICA is included within the end of the clip.
Discussion
By simultaneously rotating the X-ray tube and image intensifier during the intra-arterial injection of contrast media, rotational angiography and 3D reconstructed images enables us to obtain accurate information regarding the cerebral arteries and their branches; thus, we can visualize them from different perspectives, avoiding vessel overlap (4–8). One study (8) demonstrated excellent correlation of 3D angiographic images with surgical findings in intracranial aneurysms. To our knowledge, ours is the first report on the application of 3D angiography for the evaluation of postoperative intracranial aneurysms.
The evaluation of the result of aneurysm clip placement is important because the complete exclusion of aneurysms from the circulation is the aim of the operation. Moreover, reports have described rebleeding and the enlargement of incompletely clipped aneurysms after surgery (1–3). Sindou et al (3) suggested a classification scheme for residual aneurysms: grade I, less than 50% neck implantation; grade II, more than 50% neck implantation; grade III, residual lobe from a multilobulated sac; grade IV, residual portion of the sac less than 75% of the aneurysmal size; and grade V, residual portion of the sac more than 75% of the aneurysmal size. The suggested causes of incomplete clip placement were: 1) insufficient exposure of the aneurysm neck due to incomplete removal of the anterior clinoid process, 2) a polylobulated sac with a large neck implanted on a dysplastic arterial segment, 3) a large neck partly implanted on the infundibulum, 4) a large dysplastic neck in close contact with perforators, and 5) a large atheromatous neck (3).
The reported incidences of residual aneurysms are between 4% and 8% (1, 3, 9–12), and the risk of bleeding from residual aneurysm is believed to be 1–2% (1, 9). The incidence of recurrent aneurysm after successful clip placement has been reported to be 1.5–2.9% (1, 13).
Conventional angiography has been regarded as the criterion standard for the postoperative evaluation of clipped aneurysms. However, the detection of a small remnant of an aneurysm is often difficult because of the clip mass and because of overlap with cerebral arteries. In our series, the incidence of residual aneurysm after surgery was 42% (37 of 88 aneurysms clipped). In the immediate follow-up group, the rate was 49% (27 of 55 aneurysms clipped), which was far higher than incidences reported earlier (1, 3, 9–12). This increased rate of detecting residual aneurysms may be attributed to the excellent image quality and the various viewing angles provided by 3D angiography. In the present series, DSA depicted fewer than half (i.e., 18) of the 37 residual aneurysms demonstrated on 3D angiography and only four of 25 small, dog-eared remnants, even though the DSA studies were optimized by rotation angiograms, referencing adequate angles from the latter. It is possible that the DSA examinations would have demonstrated even fewer of the residual aneurysms without the optimization. 3D angiography allows the angles necessary to check residual aneurysms, and it allows avoidance of overlapping cerebral vessels. If required, overlapping vessels can be deleted from the images to allow more accurate definition of the aneurysmal residue. Moreover, 3D angiography demonstrated cerebral vessels, the aneurysmal residue, and the clips simultaneously. Thus, the clip–aneurysm configuration can be appreciated in its entirety, especially in cases of complex aneurysms and multiple clips.
Conventional angiography is not without risk. In a review article, cerebral angiography reportedly has a 1% risk of a disabling neurologic deficit and a <0.1% risk of mortality (14), which has prompted the development of noninvasive means for evaluating intracranial aneurysms (15–19). Despite efforts made, the role of MR angiography and CT angiography appear to be limited to the pretreatment evaluation of cerebral aneurysms at best. Postsurgical evaluation of intracranial aneurysms with MR angiography has never been satisfactory because of metal artifact. Moreover, flow void artifacts in MR angiography may erroneously suggest severe stenosis of the parent artery. In addition, there is a concern about possible clip motion with MR angiography, and vascular injury due to clip movement might bring about a fatal outcome (20). Currently, aneurysm clips made of pure titanium are available, and they are known to display no translational attraction on MR imaging systems, unlike those made of stainless steel alloys, cobalt alloys, or cobalt chrome alloys (21). However, pure titanium clips are not without susceptibility artifacts or radiofrequency shielding effects. Recently, efforts have been made to reduce metal artifacts on MR images, especially in the field of vascular stents (22–24); however, these are not applicable to the postsurgical evaluation of intracranial aneurysms.
Several articles regarding the evaluation of intracranial aneurysms with CT angiography have been published (25–34). However, to our knowledge, only one report commented on its use for the follow-up of postsurgical patients (35). Severe beam-hardening artifact renders CT angiograms diagnostically useless. Fortunately, Cho et al (35) reduced artifacts and improved the quality of CT angiography images by using only titanium clips.
Postsurgical evaluation of patients with intracranial aneurysms is a starting point for their long-term follow-up, and 3D angiography provides the most accurate and valuable information in this respect. Therefore, 3D angiography may become the criterion standard for evaluating postsurgical intracranial aneurysms, although current 3D angiography seems to have no diagnostic effect with regard to retreatment because all retreated aneurysms were also found with DSA. Reports regarding the long-term outcome of small, dog-eared remnants found on 3D angiography are anticipated.
Conclusion
Tiny dog-eared remnants are well visualized on 3D angiography, and the absence of a residual aneurysm can be confirmed by using its facility to view various angles. Moreover, 3D angiography facilitates understanding of clip–aneurysm configurations in complex aneurysms and in those involving multiple clips. Therefore, postoperative angiography may provide ample information if it is combined with 3D angiography, and this technique has a role postoperatively in patients with cerebral aneurysms. 3D angiography also allowed the detection of more residual aneurysms after surgical clip placement than did conventional DSA, although none of those detected on only 3D angiography were judged to require additional treatment. We believe that 3D angiography may provide baseline data for long-term follow-up of postsurgical aneurysms. 3D angiography may become the criterion standard for evaluating postsurgical intracranial aneurysms.
References
- 1.David CA, Vishteh AG, Spetzler RF, Lemole M, Lawton MT, Partovi S. Late angiographic follow-up review of surgically treated aneurysms. J Neurosurg 1999;91:396–401 [DOI] [PubMed] [Google Scholar]
- 2.Rauzzino MJ, Quinn CM, Fischer WS. Angiography after aneurysm surgery: indications for “selective” angiography. Surg Neurol 1998;49:32–41 [DOI] [PubMed] [Google Scholar]
- 3.Sindou M, Acevedo JC, Turjman F. Aneurysmal remnants after microsurgical clipping: classification and results from a prospective angiographic study (in a consecutive series of 305 operated intracranial aneurysms). Acta Neurochir (Wien) 1998;140:1153–1159 [DOI] [PubMed] [Google Scholar]
- 4.Abe T, Hirohata M, Tanaka N, et al. Clinical benefits of rotational 3D angiography in endovascular treatment of ruptured cerebral aneurysm. AJNR Am J Neuroradiol 2002;23:686–688 [PMC free article] [PubMed] [Google Scholar]
- 5.Kiyosue H, Okahara M, Tanoue S, Nakamura T, Nagatomi H, Mori H. Detection of the residual lumen of intracranial aneurysms immediately after coil embolization by three-dimensional digital subtraction angiographic virtual endoscopic imaging. Neurosurgery 2002;50:476–48511841714 [Google Scholar]
- 6.Okahara M, Kiyosue H, Yamashita M, et al. Diagnostic accuracy of magnetic resonance angiography for cerebral aneurysms in correlation with 3D-digital subtraction angiographic images: a study of 133 aneurysms. Stroke 2002;33:1803–1808 [DOI] [PubMed] [Google Scholar]
- 7.Sugahara T, Korogi Y, Nakashima K, Hamatake S, Honda S, Takahashi M. Comparison of 2D and 3D digital subtraction angiography in evaluation of intracranial aneurysms. AJNR Am J Neuroradiol 2002;23:1545–1552 [PMC free article] [PubMed] [Google Scholar]
- 8.Tanoue S, Kiyosue H, Kenai H, Nakamura T, Yamashita M, Mori H. Three-dimensional reconstructed images after rotational angiography in the evaluation of intracranial aneurysms: surgical correlation. Neurosurgery 2000;47:866–871 [DOI] [PubMed] [Google Scholar]
- 9.Feuerberg I, Lindquist CH. Natural history of postoperative aneurysm rests. J Neurosurg 1987;66:30–34 [DOI] [PubMed] [Google Scholar]
- 10.Macdonald RL, Wallace MC, Kestle JR. Role of angiography following aneurysm surgery. J Neurosurg 1993;79:826–832 [DOI] [PubMed] [Google Scholar]
- 11.Lin T, Fox AJ, Drake CG. Regrowth of aneurysm sacs from residual neck following aneurysm clipping. J Neurosurg 1989;70:556–560 [DOI] [PubMed] [Google Scholar]
- 12.Thornton J, Bashir Q, Aletich VA, Bebrum GM, Ausman JI, Charbel FT. What percentage of surgically clipped aneurysms have residual necks? Neurosurgery 2000;46:1294–1300 [DOI] [PubMed] [Google Scholar]
- 13.Tsutsumi K, Ueki K, Morita A, Usui M, Kirino T. Risk of aneurysm recurrence in patients with clipped cerebral aneurysms. Results of long-term follow-up angiography. Stroke 2001;32:1191–1194 [DOI] [PubMed] [Google Scholar]
- 14.Hankey GJ, Warlow CP, Sellar RJ. Cerebral angiographic risk in mild cerebrovascular disease. Stroke 1990;21:209–222 [DOI] [PubMed] [Google Scholar]
- 15.Adams WM, Laitt RD, Jackson A. The role of MR angiography in the pretreatment assessment of intracranial aneurysms: a comparative study. AJNR Am J Neuroradiol 2000;21:1618–1628 [PMC free article] [PubMed] [Google Scholar]
- 16.Maeder PP, Meuli RA, de Tribolet N. Three-dimensional volume rendering for magnetic resonance angiography in the screening and preoperative workup of intracranial aneurysms. J Neurosurg 1996;85:1050–1055 [DOI] [PubMed] [Google Scholar]
- 17.van Gelder JM. Computed tomographic angiography for detecting cerebral aneurysms: implications of aneurysm size distribution for the sensitivity, specificity, and likelihood ratios. Neurosurgery 2003;53:597–606 [DOI] [PubMed] [Google Scholar]
- 18.Villablanca JP, Martin N, Jahan R, et al. Volume-rendered helical computerized tomography angiography in the detection and characterization of intracranial aneurysms. J Neurosurg 2000;93:254–264 [DOI] [PubMed] [Google Scholar]
- 19.Watanabe Z, Kikuchi Y, Izaki K, et al. The usefulness of 3D MR angiography in surgery for ruptured cerebral aneurysms. Surg Neurol 2001;55:259–264 [DOI] [PubMed] [Google Scholar]
- 20.Klucznik RP, Carrier DA, Pyka R, Haid RW. Placement of a ferromagnetic aneurysm clip in a magnetic field with a fatal outcome. Radiology 1993;187:855–856 [DOI] [PubMed] [Google Scholar]
- 21.Shellock FG, Tkach JA, Ruggieri PM, Masaryk TJ, Rasmussen PA. Aneurysm clips: evaluation of magnetic field interactions and translational attraction by use of “long-bore” and “short-bore” 3.0-T MR imaging systems. AJNR Am J Neuroradiol 2003;24:463–471 [PMC free article] [PubMed] [Google Scholar]
- 22.Bartels LW, Smits HFM, Bakker CJG, Viergever MA. MR imaging of vascular stents: effects of susceptibility, flow, and radiofrequency eddy currents. J Vasc Interv Radiol 2001;12:365–371 [DOI] [PubMed] [Google Scholar]
- 23.Lee MJ, Janzen DL, Munk PL, MacKay A, Wiang Q, McGowen A. Quantitative assessment of an MR technique for reducing metal artifact: application to spin-echo imaging in a phantom. Skel Radiol 2001;30:398–401 [DOI] [PubMed] [Google Scholar]
- 24.Spuentrup E, Rueben A, Stuber M, Gunther RW, Buecker A. Metallic renal artery MR imaging stent: artifact-free lumen visualization with projection and standard renal MR angiography. Radiology 2003;227:897–902 [DOI] [PubMed] [Google Scholar]
- 25.Anderson GB, Findlay JM, Steinke DE, Achforth R. Experience with computed tomographic angiography for the detection of intracranial aneurysms in the setting of acute subarachnoid hemorrhage. Neurosurgery 1997;41:522–528 [DOI] [PubMed] [Google Scholar]
- 26.Anderson GB, Steinke DE, Petruk KC, Achforth R, Findlay JM. Computed tomographic angiography versus digital subtraction angiography for the diagnosis and early treatment of ruptured intracranial aneurysms. Neurosurgery 1999;45:1315–1322 [DOI] [PubMed] [Google Scholar]
- 27.Chappell ET, Noure FC, Good MC. Comparison of computed tomographic angiography with digital subtraction angiography in the diagnosis of cerebral aneurysms: a meta-analysis. Neurosurgery 2003;52:624–631 [DOI] [PubMed] [Google Scholar]
- 28.Harrison MJ, Johnson BA, Gardner GM, Welling BG. Preliminary results on the management of unruptured intracranial aneurysms with magnetic resonance angiography and computed tomographic angiography. Neurosurgery 1997;40:947–957 [DOI] [PubMed] [Google Scholar]
- 29.Hope JKA, Wilson JL, Thomson FJ. Three-dimensional CT angiography in the detection and characterization of intracranial berry aneurysms. AJNR Am J Neuroradiol 1996;17:439–445 [PMC free article] [PubMed] [Google Scholar]
- 30.Hsiang JNK, Liang EY, Lam JMK, Zhu X, Poon WS. The role of computed tomographic angiography in the diagnosis of intracranial aneurysms and emergent aneurysm clipping. Neurosurgery 1996;38:481–487 [DOI] [PubMed] [Google Scholar]
- 31.Pedersen HK, Bakke SJ, Hald JK, et al. CTA in patients with acute subarachnoid hemorrhage: a comparative study with selective, digital angiography and blinded, independent review. Acta Radiol 2001;42:43–49 [PubMed] [Google Scholar]
- 32.Preda L, Gaetani P, Rodriguez y Baena R, et al. Spiral CT angiography and surgical correlations in the evaluation of intracranial aneurysms. Eur Radiol 1998;8:739–745 [DOI] [PubMed] [Google Scholar]
- 33.Villablanca JP, Jahan R, Hooshi P, et al. Detection and characterization of very small cerebral aneurysms by using 2D and 3D helical CT angiography. AJNR Am J Neuroradiol 2002;23:1187–1198 [PMC free article] [PubMed] [Google Scholar]
- 34.Zouaoui A, Sahel M, Marro B, et al. Three-dimensional computed tomographic angiography in detection of cerebral aneurysms in acute subarachnoid hemorrhage. Neurosurgery 1997;41:125–130 [DOI] [PubMed] [Google Scholar]
- 35.Cho CB, Kim JK, Yoo DS, Cho KS, Kim DS, Huh PW. Usefulness of angiographic three-dimensional reconstruction of spiral computed tomography images for postoperative evaluation after aneurysm clipping with titanium clips. J Korean Neurosurg Soc 2003;34:299–302 [Google Scholar]