Abstract
BACKGROUND AND PURPOSE: Atherosclerosis of the major intracranial arteries is an important cause of ischemic stroke. We established measurement criteria to assess percent stenosis of a major intracranial artery (carotid, middle cerebral, vertebral, basilar) and determined the interobserver/intraobserver agreements and interclass/intraclass correlations of these measurements.
METHODS: We defined percent stenosis of an intracranial artery as follows: percent stenosis = [(1 − (Dstenosis/Dnormal))] × 100, where Dstenosis = the diameter of the artery at the site of the most severe stenosis and Dnormal = the diameter of the proximal normal artery. If the proximal segment was diseased, contingency sites were chosen to measure Dnormal: distal artery (second choice), feeding artery (third choice). Using a hand-held digital caliper, three neuroradiologists independently measured Dstenosis and Dnormal of 24 stenotic intracranial arteries. Each observer repeated the readings 4 weeks later. We determined how frequently two observers' measurements of percent stenosis of each of the 24 diseased arteries differed by 10% or less.
RESULTS: Among the three pairs of observers, interobserver agreements were 88% (observer 1 versus observer 2), 79% (observer 1 versus observer 3), 75% (observer 2 versus observer 3) for the first reading and were 75% (observer 1 versus observer 2), 100% (observer 1 versus observer 3), and 71% (observer 2 versus observer 3) for the second reading. Intraobserver agreement for each of the observers was 88%, 83%, and 100%. Interclass correlation was 85% (first reading) and 87% (second reading). Intraclass correlation was 92% (first and second readings combined).
CONCLUSION: This method shows good interobserver and intraobserver agreements for the measurement of intracranial stenosis of a major artery. If validated in subsequent studies, this method may serve as a standard for the measurement of percent stenosis of an intracranial artery.
Atherosclerotic stenosis of the major intracranial arteries is an important cause of ischemic stroke. Currently, there are no standard methods for measuring the severity of intracranial arterial stenosis. The established methods for measuring extracranial carotid stenosis are not suitable for measuring percent stenosis of a major intracranial artery because the intracranial arteries have several branches, they become slightly narrower in their distal portions, and they are often tortuous (1–3). Additionally, extracranial carotid measurement techniques have to contend with the carotid bulb, which has no equivalent in any of the intracranial arteries.
In this study, we established measurement criteria for determining percent stenosis of the major intracranial arteries (carotid, middle cerebral, vertebral, basilar) and determined the interobserver/intraobserver agreements and interclass/intraclass correlations of these measurements.
Methods
Equation for Measuring Intracranial Arterial Stenosis
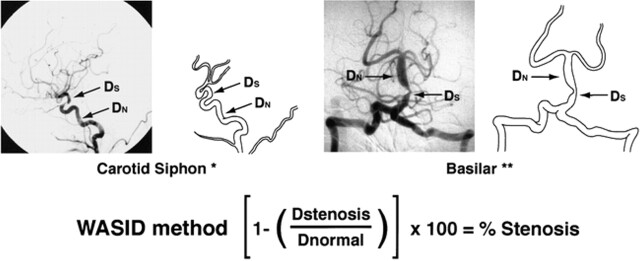
The equation used for determining percent stenosis of a major intracranial artery was as follows: percent stenosis = [(1 − (Dstenosis/Dnormal))] × 100, where Dstenosis = the diameter of the artery at the site of the most severe degree of stenosis and Dnormal = the diameter of the proximal normal artery. Dnormal was determined by the following criteria: for the middle cerebral, intracranial vertebral, and basilar arteries, the diameter of the proximal part of the artery at its widest, non-tortuous, normal segment was chosen (first choice). If the proximal artery was diseased (eg, middle cerebral artery origin stenosis), the diameter of the distal portion of the artery at its widest, parallel, non-tortuous normal segment was substituted (second choice). If the entire intracranial artery was diseased, the most distal, parallel, non-tortuous normal segment of the feeding artery was measured (third choice). For example, if the entire basilar artery was diseased, Dnormal was measured at the most distal, parallel, non-tortuous normal segment of the dominant vertebral artery; if the entire middle cerebral artery was diseased, Dnormal was measured at the most distal, parallel segment of the supraclinoid carotid artery; if the entire intracranial vertebral artery was diseased, Dnormal was measured at the most distal, parallel, non-tortuous normal segment of the extracranial vertebral artery. Because of the variability of vasculature size, slight magnification differences, and subtle differences in anteroposterior projection, the contralateral circulation was not used as the “normal” reference measurements.
Measurement of the intracranial carotid artery required a slightly different approach because the caliber of this artery often gets slightly smaller after the origin of the ophthalmic artery, and measuring the normal cavernous portion of the intracranial carotid artery can be difficult because of the tortuosity of these segments. With this in mind, Dnormal for the precavernous, cavernous, and postcavernous stenoses was measured at the widest, non-tortuous, normal portion of the petrous carotid artery that had parallel margins (first choice). If the entire petrous carotid was diseased, the most distal, parallel part of the extracranial internal carotid artery was substituted (second choice). Figure 1 illustrates how these rules were applied for measuring a stenosis of the carotid siphon and basilar artery.
fig 1.
WASID: Warfarin-Aspirin Symptomatic Intracranial Disease. The WASID method is used in the multicenter clinical trial assessing the efficacy of warfarin versus aspirin in cases of symptomatic intracranial stenosis
If tandem intracranial lesions were present (eg, distal vertebral and mid-basilar), percent stenosis of both sites was measured and the more severe stenosis was selected. When a “gap sign” was present (ie, the lumen of the vessel could not be visualized at the site of severe stenosis), Dstenosis could not be measured with calipers. In these cases, percent stenosis was defined as 99% luminal stenosis.
Angiogram Selection, Measuring Technique
After undergoing detailed training in the measurement criteria outlined above, three neuroradiologists independently evaluated 24 angiograms showing stenoses of 24 diseased intracranial arteries: nine carotid, seven middle cerebral, five basilar, and three vertebral arteries. The angiograms were chosen by reviewing the radiology log reports at Emory University Hospital between March and July 1996. Those angiograms with at least 50% stenosis of a major intracranial artery by “eye ball” estimation were selected. None of the neuroradiologists participated in the selection process. The radiographic view (anteroposterior or lateral) that revealed the highest degree of stenosis of each arterial lesion was provided to each reader. After obtaining a single measurement of Dstenosis and Dnormal, the percent diameter stenosis was calculated. Four weeks later, after rules for measuring intracranial stenosis were reinforced, each observer repeated the readings, blinded to their initial measurements.
Using a hand-held electronic caliper, all measurements were made at 90° to the arterial wall, extended point type (Mitutoyo 573–225010). This caliper has two narrow tips that do not obscure the margins of the arterial wall when measurements are obtained. A liquid crystal display provides measurements to 1/100 mm. The accuracy of the caliper is within 0.025 mm. All measurements were obtained on a horizontal lighted view box using a freestanding adjustable lighted magnifying glass (×10).
Statistical Analysis
Measurements of repeatability (measuring the same film by the same reader) and reproducibility (measuring the same film by different readers) were determined by fitting a nested analysis of variance model in which the reader factor was nested within the film factor and the repeated readings of the same film were nested within the reader factor. Both factors, film and reader, were considered as random factors (4, 5).
As a measure of repeatability, we calculated confidence intervals (95%, 90%, 85%) for the difference between the two readings of the same film by the same reader. The interpretation of this measure is that the difference between two readings of the same film by the same reader would differ by no more than this amount, half the width of the confidence interval, 95%, 90%, or 85% of the time.
As a measure of reproducibility, 95%, 90%, and 85% confidence intervals were also calculated for the difference between two readings of the same film by two different readers. Similarly, the interpretation of this measure is that the difference between the two readings of the same film by different readers would differ by no more than this amount, half the width of the confidence interval, 95%, 90%, or 85% of the time.
The analysis of variance model also provided estimates of the interclass and intraclass correlation coefficients. As a descriptive measure of repeatability, we calculated the percent of films for which the two readings of a film by the same reader were within 10% of each other (intraobserver variation). Similarly, for reproducibility, we calculated the percent of films for which the two readings of the same film by different readers were within 10% of each other (interobserver variation). This was done separately for each pairing of the three readers for both the first and second readings.
Results
Repeatability
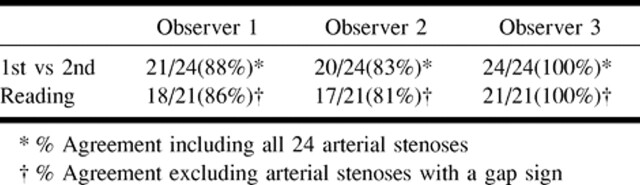
Among the three readers, intraobserver agreement or the percent of films in which the difference in percent stenosis between the first and second reading of a film by the same reader were less than 10% varied from 83% to 100% (81% to 100% excluding gap sign films) (Table 1). The intraclass correlation coefficient was 0.93. The estimate of the standard deviation of repeated readings of a film by the same reader based on the analysis of variance model was 3.9.
TABLE 1:
Intraobserver agreement (% stenosis within 10%)
Based on confidence intervals of 95%, 90%, and 85%, estimates of repeatability were 10.8%, 9%, and 6.8%, respectively. This indicated that 95%, 90%, and 85% of the time, we would expect the difference in measurements of percent stenosis between repeated measurements of the same angiogram by the same reader to be less than 10.8%, 9%, and 6.8%, respectively.
Reproducibility
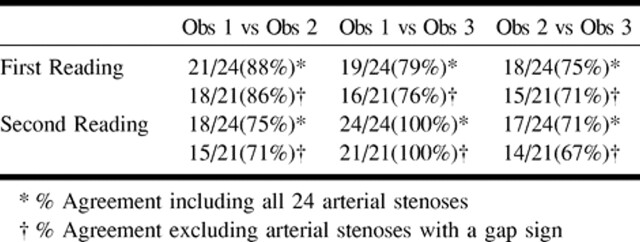
Among the pairings of the three readers, interobserver agreement or the percent of films in which the difference in percent stenosis measured by two readers was less than 10% varied from 71% to 100% (67% to 100% excluding gap sign films) (Table 2). The interclass correlation coefficient among the three readers was .85 for the first reading and .87 for the second reading.
TABLE 2:
Interobserver agreement (% stenosis within 10%)
Based on confidence intervals of 95%, 90%, and 85%, estimates of repeatability were 15.7%, 13.2%, and 11.7%, respectively. This indicated that 95%, 90%, and 85% of the time we would expect the difference in measurements of percent stenosis of the same film by different readers to be less than 15.7%, 13.2%, and 11.7%, respectively.
Discussion
Angiographic measurement standards for determining percent stenosis of the extracranial internal carotid artery are well established (1–3, 6). These methods are routinely used in clinical practice to identify patients who may benefit from carotid endarterectomy. Currently, there is no equivalent method for measuring percent stenosis of the major intracranial arteries. This may be because until recently, there were limited data on the impact of the severity of intracranial stenosis on the risk of ischemic stroke. Recent studies suggest that the annual risk of stroke in patients with at least 50% stenosis of a major intracranial artery is 4% to 10% (7). Moreover, patients with severe intracranial stenosis (70% to 99%) have a higher risk of stroke than do patients with moderate intracranial stenosis (50% to 69%) (7). If the prognosis of intracranial arterial stenosis and the choice of therapy (eg, anticoagulation, angioplasty) for these patients are clearly shown to be based on the severity of intracranial stenosis, a reproducible and repeatable method for measuring percent stenosis of the major intracranial arteries will be required. In our study, intracranial arterial stenoses of more than 50% were chosen because of the significance of the stroke risk of high-grade intracranial stenosis as compared with lesions of lesser severity. Future studies may address low- and moderate-grade intracranial stenoses.
The method developed in this study seems to fulfill these criteria. Using a narrow agreement range (ie, within 10% of each reader's measurement of percent stenosis), interobserver agreements for the three readers ranged from 71% to 100% and intraobserver agreements ranged from 83% to 100%. These rates of observer agreements are similar to those reported for the extracranial carotid artery (8) and exceed those reported for the coronary arteries (9). In a study assessing measurement of extracranial carotid stenosis, Rothwell et al (8) reported interobserver agreements of percent stenosis within 10% of two readers' measurements as 70% for the European Carotid Surgery Trial (ECST) method, 74% for the North American Symptomatic Carotid Endarterectomy Trial (NASCET) method, and 75% for the Common Carotid method. Zir et al (9) reported a low interobserver agreement of four coronary angiographers in the interpretation of coronary artery stenosis. In this study, interobserver agreement for proximal or mid-left anterior descending stenosis (within 10%) ranged from 0% to 40%. The four observers in this study agreed regarding the presence of at least 50% stenosis in only 45% of left anterior descending lesions and 65% of right coronary artery lesions. The authors reported that a major reason for the poor results was disagreement regarding where to measure the normal vessel, Dnormal. Thus, development of specific criteria for measuring Dnormal was suggested as one method of reducing interobserver variability. Hence, in the present study, we defined specific rules for determining the specific site at which to measure Dnormal. Moreover, the three participating neuroradiologists in the present study underwent extensive training with reinforcement of the rules before obtaining their second measurements. We suspect that the success of this measuring technique depends in large portion on ability to follow these rules.
This study has shown that obtaining reproducible measurements of percent stenosis of an intracranial artery by using conventional cerebral angiography is possible using standardized measurement criteria. If validated in further studies, this method may serve as a standard for the measurement of percent stenosis of an intracranial artery obtained using conventional angiography.
Footnotes
Address reprint requests to Owen B. Samuels, MD, Emory University, Department of Neurosurgery, 1365 Clifton Road N.E., Clinic Building B, Suite 6400, Atlanta, GA 30322.
References
- 1. European Carotid Surgery Trialists' Collaborative Group. MRC European Carotid Surgery Trial: interim results for symptomatic patients with severe (70–99%) or with mild (0–29%) carotid stenosis. Lancet 1991;337:1235-1243 [PubMed] [Google Scholar]
- 2. North American Symptomatic Carotid Endarterectomy Trial Collaborators. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med 1991;325:445-453 [DOI] [PubMed] [Google Scholar]
- 3.Williama M, Nicoliades A. Predicting the normal dimensions of the internal and external carotid arteries from the diameter of the common carotid. Eur J Vasc Surg 1987;1:91-96 [DOI] [PubMed] [Google Scholar]
- 4.Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986;1:307-310 [PubMed] [Google Scholar]
- 5.Alterman DG, Bland JM. Measurement in medicine: the analysis of method comparison studies. Statistician 1983;307-317
- 6.Fox AJ. How to measure carotid stenosis. Radiology 1993;186:316-318 [DOI] [PubMed] [Google Scholar]
- 7.Chimowitz MI, Kokkinos J, Strong J, et al. The Warfarin-Aspirin Symptomatic Intracranial Disease Study. Neurology 1995;45:1488-1493 [DOI] [PubMed] [Google Scholar]
- 8.Rothwell PM, Gibson RJ, Slattery J, Warlow CP, for the European Carotid Surgery Trialists' Collaborative Group Prognostic value and reproducibility of measurements of carotid stenosis: a comparison of three methods on 1001 angiograms. Stroke 1994;25:2440-2444 [DOI] [PubMed] [Google Scholar]
- 9.Zir LM, Miller SW, Dinsmore RE, Gilbert JP, Harthorne JW. Interobserver variability in coronary angiography. Circulation 1976;53:627-632 [DOI] [PubMed] [Google Scholar]