Abstract
BACKGROUND AND PURPOSE: Hemosiderin and white matter lesions are 2 of the most common neurologic complications found on MR imaging that may be related to cranial irradiation and intrathecal methotrexate (MTX) therapy in childhood acute lymphoblastic leukemia (ALL). We evaluated the brains of patients previously treated for ALL with cranial irradiation and intrathecal MTX with MR imaging and tested the hypothesis that these patients have more MR evidence of central nervous system (CNS) injury than control patients who are in complete remission following systemic chemotherapy without cranial irradiation.
METHODS AND MATERIALS: ALL patients recruited from a pediatric cancer center data base were examined at 1.5T by using Tl-weighted, T2-weighted, gradient echo (GE), and fluid-attenuated inversion recovery sequences. Patients treated in childhood for solid extracranial neoplasms were used as controls.
RESULTS: Lesions consistent with old hemorrhage were detected in 23 (55%) of the ALL patients and in none of the control patients (P <.001). Of the 62 hemorrhages detected on the GE images, only 9 (15%) were shown on T2-weighted images. White matter abnormalities were found in 2 ALL patients (5%) and one control patient (5%). Old infarcts were seen in 2 ALL patients (10%).
CONCLUSION: Cerebral hemorrhages related to radiation-induced vascular malformations, most of which are asymptomatic, are much more common in survivors of childhood ALL than previously thought. GE sequencing is more sensitive in detecting hemorrhagic lesions than T2-weighted sequencing and is to be included in imaging protocols for follow-up study of patients after cerebral radiation therapy.
Acute lymphoblastic leukemia (ALL) is the most common malignancy of childhood.1–3 Neurologic complications are common, both during and after completion of therapy. Hemosiderin and white matter lesions are 2 of the most common findings on MR imaging4–8 that may relate to cranial irradiation and intrathecal methotrexate (MTX). Previous MR imaging studies employed mainly spin-echo T2-weighted imaging in their evaluation.
The main purpose of this study was to evaluate the brains of patients previously treated for ALL with cranial irradiation and intrathecal MTX by using T1-weighted, T2-weighted, fluid-attenuated inversion recovery (FLAIR) T2-weighted, and gradient echo (GE) sequences and to test the hypothesis that these patients have more MR evidence of central nervous system (CNS) injury than control patients who are in complete remission following systemic chemotherapy without cranial irradiation.
Materials and Methods
Patients were recruited from our children’s cancer center data base. A senior pediatrician attempted to contact all patients with ALL who had survived more than 5 years from diagnosis. Patients, who had been treated in childhood for solid extracranial neoplasms by using systemic chemotherapy, but not intrathecal therapy or cranial irradiation, were recruited as controls.
Clinical information concerning treatment dates and protocols was obtained by review of medical charts. MR was performed on all patients at 1.5T (Gyroscan NT, Philips, Best, the Netherlands), by using Tl-weighted (TIW) sagittal spin-echo (TR, 500 milliseconds; TE, 15 milliseconds), T2-weighted transverse fast spin-echo (TR, 3300 milliseconds; TE, 100 milliseconds), GE transverse (TR, 300 milliseconds; TE, 30 millisecond; flip angle, 30°), and FLAIR coronal (TR, 8000 milliseconds; TE, 110 milliseconds; TI, 2400 milliseconds) sequences.
Hemorrhages were defined as focal rounded areas of very low signal intensity (due to susceptibility artifact and attributable to the presence of hemosiderin—detected on the T2-weighted or GE sequences—in any part of the brain. The number and location of these lesions were recorded for each patient.
Hyperintense white matter abnormalities seen on the T2-weighted or FLAIR sequences were graded according to a modification of the system of Wilson et al.9 Grade 1 was defined as patchy, mildly increased signal intensity in the periventricular white matter, grade 2 as moderate changes that extend almost to the gray-white junction, sparing the subcortical U-fibers, and grade 3 as severe changes, confluent from the level of the frontal horns to that of the trigones, with or without involvement of the U-fibers.
Old infarcts were diagnosed by the detection of brain parenchymal loss pertaining to arterial territory or discrete lesions with hypointensity on T1-weighted images and hyperintensity on T2-weighted images, and further confirmed by reference to clinical history and previous CT findings.
Fisher’s exact test was used to compare the number of infarcts and hemorrhages detected in the ALL and control groups. Student t test was used for the other comparisons between the groups. Spearman rho (ρ) was used to estimate correlations.
Interobsever agreement for the detection of hemorrhages on GE images was assessed by (1) overall proportion of agreement on dichotomized results (positive or negative), (2) unweighted kappa for 4 categories (0 = no hemorrhage; 1 = 1–3 hemorrhagic foci; 2 = 4–6 hemorrhagic foci; 3 = 7 or more hemorrhagic foci). Two experienced radiologists individually and independently assessed these images, and the final interpretation of discrepant results was by consensus agreement.
Informed consent was obtained from patients or their guardians, as appropriate. The study was approved by the local institutional ethics committee.
Results
Forty-two ALL patients treated at our center, and diagnosed more than 5 years previously, agreed to take part in this study. Twenty-two control patients were recruited. The 2 groups were similar with respect to sex and age at diagnosis (Table 1), though the mean interval from diagnosis and mean age at examination were longer for the ALL patients.
Table 1:
Clinical characteristic of ALL and control patients
| ALL Patients | Control Patients | Student t test | |
|---|---|---|---|
| Number | 42 | 22 | |
| Number (%) male | 27 (64) | 15 (68) | |
| Age at examination (y) | |||
| Range | 6.9–27.6 | 7.2–31.8 | P = .16 |
| Mean ± SD | 17.4 ± 4.6 | 15.4 ± 5.5 | |
| Age at diagnosis (y) | |||
| Range | 1.2–13.7 | 0.5–13.0 | P = .46 |
| Mean ± SD | 5.2 ± 2.9 | 5.9 ± 4.0 | |
| Interval from diagnosis (y) | |||
| Range | 5.0–18.8 | 5.6–20.4 | P = .015 |
| Mean ± SD | 12.2 ± 3.6 | 9.5 ± 4.2 |
Note:—ALL indicates acute lymphoblastic leukemia.
Forty of the ALL patients had been treated with cranial irradiation (at least 18 Gy) and intrathecal MTX, as well as systemic chemotherapy according to various protocols. Seven patients received >18 Gy (range, 22.8–36 Gy; mean, 25.7 Gy). Two had been treated with intrathecal MTX and systemic chemotherapy but had not received cranial irradiation.
Ten patients received >72 mg (range, 125–300 mg; mean, 210 mg) intrathecal MTX, and 31 had 72 or 60 mg intrathecal MTX.
The higher dose of radiation and intrathecal MTX was for patients with blast cells on CSF cytology with MTX dose adjusted further by serial CSF cytology. None had undergone bone marrow transplantation.
The control patients had been treated for Wilms tumor (6), Hodgkin disease (3), osteosarcoma (3), Langerhans cell histiocytosis (LCH; 3), rhabdomyosarcoma (2), neuroblastoma (2), Ewing sarcoma (1), chronic myeloid leukemia (1), and ovarian sex cord tumor (1), by using various chemotherapy protocols. None had been treated with intrathecal chemotherapy or cranial irradiation.
In the measurement of interobsever agreement the overall proportion of agreement in the diagnosis of presence or absence of hemorrhage on GE imaging was 0.93; and the unweighted kappa for the 4 categories of number of hemorrhages was 0.89.
Sixty-two lesions consistent with old hemorrhages were detected in 23 (55%) of the ALL patients (Fig 1). The maximum number of 10 foci of hemorrhages detected is 10, and the median is 1 for those positive for hemorrhage. Most hemorrhages were located in the cerebral hemispheres (Table 2). No hemorrhage was detected in the control patients (P < .001) or the 2 ALL patients who did not undergo cranial irradiation. There was a history of a neurologic event in only one of the affected patients, but this was almost certainly due to an established cerebral infarct shown on the MR imaging and previous CT. Of the 62 hemorrhages detected on the GE images, 9 (15%) were shown on T2-weighted, 7 (11%) on FLAIR, and 3 (5%) on T1-weighted images (Table 3). There was no hemorrhage detected on T1-weighted, T2-weighted, or FLAIR images that was not shown on GE images.
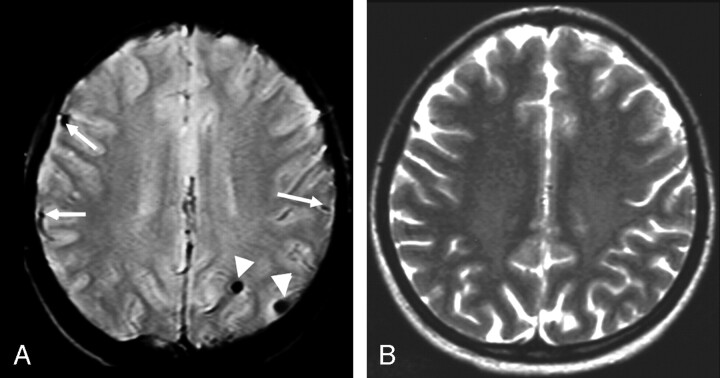
Fig 1.
Twenty-year-old woman diagnosed 14 years previously and treated with intrathecal MTX (total dose = 72 mg) and cranial radiation therapy (18 Gy). A, Transverse gradient echo image shows 2 lesions compatible with old hemorrhages (arrowheads). The structures indicated by the arrows are blood vessels. B, Transverse T2-weighted fast spin-echo image at the same level appears normal.
Table 2:
Location of hemorrhages detected at gradient echo imaging in 42 long-term survivors of acute lymphoblastic leukemia
| Region | Number (%) | Subregion | Number |
|---|---|---|---|
| Cerebral hemispheres | 51 (82%) | Frontal lobe | 23 |
| Parietal lobe | 10 | ||
| Temporal lobe | 10 | ||
| Occipital lobe | 6 | ||
| Insula | 2 | ||
| Deep brain structures | 5 (8%) | Corpus callosum | 2 |
| Pineal | 1 | ||
| Thalamus | 1 | ||
| Caudate nucleus | 1 | ||
| Posterior fossa | 6 (10%) | Cerebellum | 5 |
| Brain stem | 1 | ||
| Total | 62 (100%) |
Table 3:
Detection of hemorrhages by different imaging sequences
| Sequence | No. of hemorrhagic foci detected |
|---|---|
| Gradient echo | 62 |
| T2-weighted spin echo | 9 |
| Fluid-attenuated inversion recovery | 7 |
| T1-weighted spin echo | 3 |
In the group of ALL patients, there was a significant correlation between number of hemorrhages and the radiation dose (r = 0.44; P =.004) but not between number of hemorrhages and dose of intrathecal MTX (r = 0.04; P =.8). There was a correlation between number of hemorrhages and time since diagnosis (ρ = 0.391; P =.013). In the relatively homogeneous group of 30 patients treated with the lower dose of 18 Gy of cranial radiation therapy and a total of 72 mg or less of intrathecal MTX, however, the correlation was weaker (ρ = 0.274; P =.135). There was also no correlation in this group between age at diagnosis and the number of hemorrhages (P =.973).
White matter abnormalities (Fig 2) were found in 2 ALL patients (one grade 1 and one grade 2), both treated with a high total dose of intrathecal MTX (≥300 mg), and one control patient (grade 1), who had been treated with high-dose intravenous MTX for osteosarcoma.
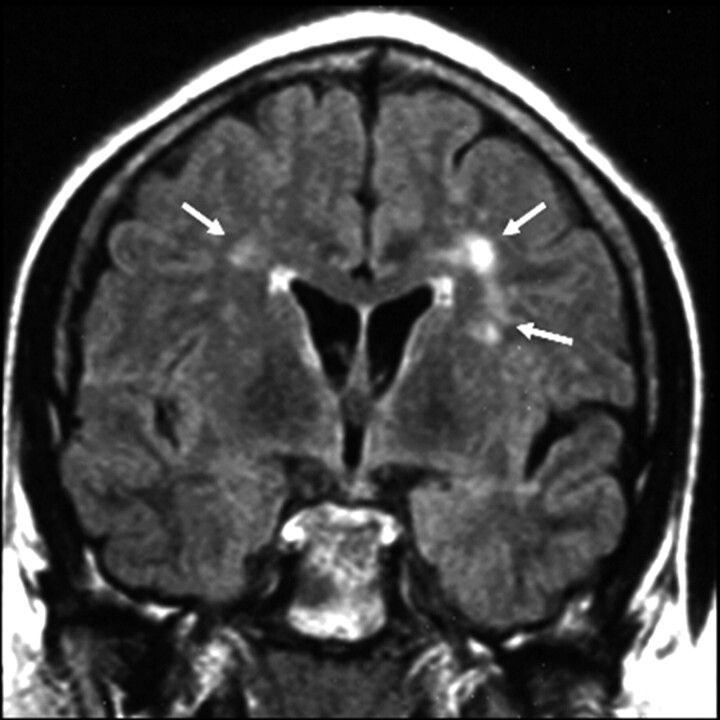
Fig 2.
Twenty-seven-year-old man diagnosed 18 years previously and treated with intrathecal MTX (total dose = 300 mg) and cranial radiation therapy (24 Gy). On coronal FLAIR image, there is grade 1 leukoencephalopathy (arrows), defined as patchy, mildly increased signal intensity in the periventricular white matter, without involvement of the subcortical U-fibers.
Old infarcts were seen in 4 ALL patients (10%). All of these had been previously diagnosed on the basis of symptoms and prior imaging. No infarcts were demonstrated in the control patients (P =.29).
One nonspecific focal lesion was found in an ALL patient. One control patient, treated 15 years previously for LCH, was found to have a mass in the cerebellar vermis. Biopsy of this lesion produced tissue consistent with LCH. With these exceptions, no tumor was identified.
Discussion
Overall long-term survival in ALL is about 70%, though for individual patients this varies from about 40% to 90%, depending on prognostic features at diagnosis and early response to therapy.10–13 CNS relapse, however, formerly occurred in at least 50% of patients.14–16 This phenomenon led to the introduction of CNS prophylaxis for children with ALL. The most commonly used method in the 1970s and 1980s involved cranial irradiation, originally to a dose of 24 Gy but later to 18 Gy,17 and intrathecal chemotherapy with MTX. This treatment reduces the rate of isolated CNS relapse to 5%–10%,14,18 but is associated with various forms of damage to normal brain tissue, including leukoencephalopathy, mineralizing microangiopathy (MMA), and the development of secondary tumors. Cranial irradiation appears to be an important cause of long-term neuropsychological impairment.19,20 Recent protocols have used risk stratification to avoid cranial irradiation in children with standard and intermediate-risk ALL,21–24 and to reduce the dose to 12 Gy in those with high-risk ALL, without compromising event-free survival.25,26
Several studies have used MR imaging to detect neurologic complications in patients treated for ALL.5,6,8,27–37 A wide range of results has been reported. MR evidence of leukoencephalopathy has been reported in about 40% of patients during therapy6,9,33,35 and 0%–31% of patients in long-term remission.7,31,32,39
Evidence of old hemorrhage, presumably related to radiation-induced vascular malformations,27,29,32 which could be large arterial or cryptic vascular malformation, has been reported in 6.5%–20% of long-term survivors.27,36 Previous studies, however, have relied on spin-echo sequences, which are not particularly sensitive to the presence of hemosiderin. Most published studies have failed to include control patients. It is therefore not clear whether the abnormalities detected are due to ALL itself, to systemic chemotherapy, or to prophylactic CNS therapy.
In our study, the use of a GE MR image revealed a high prevalence (55%) of lesions consistent with old cerebral hemorrhage in survivors of ALL. Hemorrhages were present in 58% of the 40 patients who had been treated with cranial irradiation. If we had used only the spin-echo T2-weighted sequence, 9 hemorrhages, at most, would have been found in 8 (19%) of the ALL patients. The lower sensitivity of spin-echo T2-weighted imaging in the detection of hemorrhages was also illustrated by a retrospective review that found symptomatic cerebral hemorrhages on T2-weighted imaging in 7% of a group of children whose treatment for ALL had included cranial irradiation.27 A more recent retrospective study in children after cranial irradiation for heterogeneous malignancies comprising ALL, acute myeloid leukemia, and intracranial tumors also employed T2-weighted fast spin-echo imaging for the evaluation of telangiectasia and reported an incidence of 20% (18 of 90 children) of telangiectasia.36 We suspect that the use of the more sensitive GE sequence accounts for the high prevalence of apparently asymptomatic old hemorrhages in our patients and the use of T2-weighted images only would result in an underestimation of telangiectasia manifested as cerebral hemorrhages.
In a study with a cross-sectional profile as in the current one, whether hemorrhages may remain positive ad infinitum on GE imaging cannot be addressed and a future longitudinal study is necessary to answer the question.
These hypointense foci do not appear to be due to calcification, for 2 reasons. First, their distribution is clearly different from that of the calcification seen in MMA, which, in addition to involving the deep layers of the cortex and subcortical U-fibers,40 has a strong predilection for the lentiform nuclei.40,41 None of the 62 hypointense lesions we identified occurred in the lentiform nuclei, though one occurred in the head of the caudate nucleus and one in the thalamus. Second, the lesions we found were all rounded in shape, whereas in MMA the calcification is typically elongated (corresponding to the configuration of the Cortex) at CT or MR,34 and at autopsy.42 Calcifications have been shown to correspond to hyperintense foci on T1-weighted MR imaging.30 None of the hypointense foci on GE images were shown as hyperintense foci on T1-weighted images. Some of the hemorrhages may, however, have occurred in areas affected by MMA as occlusion of small blood vessel lumen is a histologic feature of MMA. In the multicenter study by Hertzberg et al,34 intracranial calcification was detected in 4 of 49 (8.2%) ALL survivors who had CT. We agree with Kingma et al31 that it would not be ethical to perform CT in these patients, and are therefore unable to prove or disprove this.
All of the hemorrhages detected in this study were apparently asymptomatic so the immediate clinical relevance is limited. These hemorrhages, however, may be markers for future bleeding. After completion of the study, one of the ALL patients who had been shown to have 3 hemorrhages presented with neurologic symptoms. CT showed a small new thalamic hemorrhage, and no evidence of cerebral calcification. Sequential MR examinations showed the evolution during a 2-month period of a hypointense lesion very similar to the 3 pre-existing hemorrhages.
As the presence of hemorrhage may relate to a change of the global microenvironment, future studies involving the assessment of neuro-cognitive function, which is known to be affected by cranial irradiation, would be valuable in interpreting the importance of these hemorrhages.
Old hemorrhages detected after therapy for ALL have probably occurred in association with acquired vascular malformations,28 which may result from venous occlusion due to radiation-induced endothelial injury.28,29 Subclinical hemorrhage appears to be a common phenomenon in cryptic vascular malformations.28,43,44 The absence of old hemorrhages in control patients, treated with various forms of systemic chemotherapy for extracranial-neoplastic diseases, is consistent with this hypothesis. The association of radiation-induced injury and vascular malformation has also been implicated by the development of symptomatic or asymptomatic vascular malformations a few years after cranial irradiation treatment in children with brain tumors.28 The current study supports the induction of vascular malformation by radiation therapy by demonstrating a significant correlation between the number of hemorrhages and the radiation dose.
Comparison of the occurrence of hemorrhages in the present group of ALL survivors and new group of patients being treated with stratified radiation therapy would be valuable to further elucidate the effect of radiation on inducing these vascular abnormalities.
The number of hemorrhages appears to increase with time from diagnosis, even in an apparently homogeneous treatment group. This may be due to a tendency to accumulate hemorrhages over time or the emergence of new vascular malformations with time.
Only 2 ALL patients (5%) showed evidence of leukoencephalopathy. The patient with grade 1 changes (Fig 2) had been treated with 300 mg of intrathecal MTX and cranial irradiation to 24 Gy. The patient with grade 2 changes had received 372 mg and 18 Gy. The small numbers precluded any analysis of the relative sensitivity of T2-weighted and FLAIR sequences. The wide range of reported prevalence of leukoencephalopathy in children cured of ALL7,31,32,38,39 has not been fully explained. The variation may be partly explained by the different interpretation of white matter hyperintensities on T2-weighted images by different researchers. Mimics of leukoencephalopathy include symmetrical areas of mild hyperintensity in the peritrigonal deep white matter in young children and hyperintense perivascular spaces on T2-weighted images. In the current study, attention to the typical symmetrical distribution of the former and demonstration of low signal intensity on FLAIR images of the latter may have contributed to the relatively low incidence of leukoencephalopathy; however, we believe it is the long interval (range, 5–19 years; mean, 12.8 years) between diagnosis and MR imaging in our ALL survivors compared with those of most previous studies8,34,36 that is the main reason for the lower incidence of leukoencephalopathy in the current study. Disappearance of white matter changes on MR imaging in ALL survivors with time has been documented elsewhere.33,37
No clear relationship exists in the published studies between the dose of MTX given and the incidence of leukoencephalopathy. Subclinical leukoencephalopathy is common in children with osteosarcoma treated with high-dose intravenous MTX,45 and suggests that MTX, not cranial irradiation, is the most important cause of leukoencephalopathy.
Conclusion
In conclusion, cerebral hemorrhages are more frequent in survivors of childhood ALL than previously thought and the hemorrhages detected in this study are apparently all asymptomatic. Their occurrence is probably related to vascular malformations induced by radiation therapy. GE sequence is more sensitive in detecting hemorrhagic lesions resulting in more hemorrhages being detected than by using T2-weighted sequence and should be included in imaging protocols for follow-up study of patients after cerebral radiation therapy.
References
- 1.Miller RW, Young JL Jr, Novakovic B. Childhood cancer. Cancer 1994;75:395–405 [DOI] [PubMed] [Google Scholar]
- 2.Grovas A, Fremgen A, Rauck A, et al. The National Cancer Data Base report on patterns of childhood cancers in the United States. Cancer. 1997;80:2321–32 [DOI] [PubMed] [Google Scholar]
- 3.Merrill RM, Capocaccia R, Feuer EJ, et al. Cancer prevalence estimates based on tumour registry data in the Surveillance, Epidemiology, and End Results (SEER) Program. Int J Epidemiol. 2000;29:197–207 [DOI] [PubMed] [Google Scholar]
- 4.Brecher ML, Berger P, Freeman AI, et al. Computerized tomography scan findings in children with acute lymphocytic leukemia treated with three different methods of central nervous system prophylaxis. Cancer 1985;56:2430–33 [DOI] [PubMed] [Google Scholar]
- 5.Asato R, Akiyama Y, Ito M, et al. Nuclear magnetic resonance abnormalities of the cerebral white matter in children with acute lymphoblastic leukemia and malignant lymphoma during and after central nervous system prophylactic treatment with intrathecal methotrexate. Cancer 1992;70:1997–2004 [DOI] [PubMed] [Google Scholar]
- 6.Matsumoto K, Takahashi S, Sato A, et al. Leukoencephalopathy in childhood hematopoietic neoplasm caused by moderate-dose methotrexate and prophylactic cranial radiotherapy-an MR analysis. Int J Radiat Oncol Biol Phys 1995;32:913–18 [DOI] [PubMed] [Google Scholar]
- 7.Laitt RD. Chambers EJ. Goddard PR, et al. Magnetic resonance imaging and magnetic resonance angiography in long term survivors of acute lymphoblastic leukemia treated with cranial irradiation. Cancer 1995;76:1846–52 [DOI] [PubMed] [Google Scholar]
- 8.Harila-Saari AH, Paakko EL, Vainionpaa LK, et al. A longitudinal magnetic resonance imaging study of the brain in survivors in childhood acute lymphoblastic leukemia. Cancer 1998;83:2608–17 [PubMed] [Google Scholar]
- 9.Wilson DA, Nitschke R, Bowman ME, et al. Transient white matter changes on MR images in children undergoing chemotherapy for acute lymphoblastic leukemia: correlation with neuropsychologic deficiencies. Radiology 1991;180:205–09 [DOI] [PubMed] [Google Scholar]
- 10.Chessells JM, Hardisty RM, Richards S. Long survival in childhood lymphoblastic leukaemia. Br J Cancer 1987;55:315–19 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Forestier E, Johansson B, Gustafsson G, et al. Prognostic impact of karyotypic findings in childhood acute lymphoblastic leukaemia: a Nordic series comparing two treatment periods. For the Nordic Society of Paediatric Haematology and Oncology (NOPHO) Leukemia Cytogenetic Study Group. Br J Haematol. 2000;110:147–53 [DOI] [PubMed] [Google Scholar]
- 12.Donadieu J, Hill C. Early response to chemotherapy as a prognostic factor in childhood acute lymphoblastic leukaemia: a methodological review. Br J Haematol 2001;115:34–45 [DOI] [PubMed] [Google Scholar]
- 13.Donadieu J, Auclerc MF, Baruchel A, et al. Critical study of prognostic factors in childhood acute lymphoblastic leukaemia: differences in outcome are poorly explained by the most significant prognostic variables. Fralle group. French Acute Lymphoblastic Leukaemia Study Group. Br J Haematol 1998;102:729–39 [DOI] [PubMed] [Google Scholar]
- 14.Schroeder H, Garwicz S, Kristinsson J, et al. Outcome after first relapse in children with acute lymphoblastic leukemia: a population-based study of 315 patients from the Nordic Society of Pediatric Hematology and Oncology. Med Pediatr Onco1 1995;25:372–78 [DOI] [PubMed] [Google Scholar]
- 15.Evans AE, Gilbert ES, Zandstra R. The increasing incidence of central nervous system leukemia in children (Children’s Cancer Study Group A). Cancer 1970;26:404–09 [DOI] [PubMed] [Google Scholar]
- 16.West RJ, Graham-Pole J, Hardisty RM, et al. Factors in pathogenesis of central-nervous-system leukaemia. BMJ 1972;3:311–14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nesbit ME Jr, Sather HN, Robison LL, et al. Presymptomatic central nervous system therapy in previously untreated childhood acute lymphoblastic leukaemia: comparison of 1800 rad and 2400 rad: a report for Children’s Cancer Study Group. Lancet 1981;1:461–66 [DOI] [PubMed] [Google Scholar]
- 18.Eden OB, Lilleyman JS, Richards S, et al. Results of Medical Research Council Childhood Leukaemia Trial UKALL VIII: report to the Medical Research Council on behalf of the Working Party on Leukaemia in Childhood. Br J Haematol 1991;78:187–96 [DOI] [PubMed] [Google Scholar]
- 19.Hill JM, Kornblith AB, Jones D, et al. A comparative study of the long term psychosocial functioning of childhood acute lymphoblastic leukemia survivors treated by intrathecal methotrexate with or without cranial radiation. Cancer 1998;82:208–18 [PubMed] [Google Scholar]
- 20.Bleyer WA. Central nervous system leukemia. Pediatr Clin North Am 1988;35:789–814 [DOI] [PubMed] [Google Scholar]
- 21.Nachman J, Sather HN, Cherlow JM, et al. Response of children with high-risk acute lymphoblastic leukemia treated with and without cranial irradiation: a report from the Children’s Cancer Group. J Clin Oncol 1998;16:920–30 [DOI] [PubMed] [Google Scholar]
- 22.Hill FG, Richards S, Gibson B, et al. The UK Medical Research Council Working Party on Childhood Leukaemia: successful treatment without cranial radiotherapy of children receiving intensified chemotherapy for acute lymphoblastic leukaemia: results of the risk-stratified randomized central nervous system treatment trial MRC UKALL XI (ISRC TN 16757172). Br J Haematol 2004;124:33–46 [DOI] [PubMed] [Google Scholar]
- 23.Veerman AJ, Hahlen K, Kamps WA, et al. High cure rate with a moderately intensive treatment regimen in non-high-risk childhood acute lymphoblastic leukemia: results of protocol ALL VI from the Dutch Childhood Leukemia Study Group. J Clin Oncol 1996;14:911–18 [DOI] [PubMed] [Google Scholar]
- 24.Kamps WA, Bokkerink JP, Hahlen K, et al. Intensive treatment of children with acute lymphoblastic leukemia according to ALL-BFM-86 without cranial radiotherapy: results of Dutch Childhood Leukemia Study Group Protocol ALL-7 (1988–1991). Blood 1999;94:1226–36 [PubMed] [Google Scholar]
- 25.Schrappe M, Reiter A, Ludwig WD, et al. Improved outcome in childhood acute lymphoblastic leukemia despite reduced use of anthracyclines and cranial radiotherapy: results of trial ALL-BFM 90. German-Austrian-Swiss ALL-BFM Study Group. Blood. 2000;95:3310–22 [PubMed] [Google Scholar]
- 26.Schrappe M, Reiter A, Zimmermann M, et al. Long-term results of four consecutive trials in childhood ALL performed by the ALL-BFM study group from 1981 to 1995. Berlin-Frankfurt-Munster. Leukemia. 2000;14:2205–22 [DOI] [PubMed] [Google Scholar]
- 27.Humpl T, Bruhl K, Bohl J, et al. Cerebral haemorrhage in long-term survivors of childhood acute lymphoblastic leukemia. Eur J Pediatr 1997;156:367–70 [DOI] [PubMed] [Google Scholar]
- 28.Gaensler EHL, Dillon WP, Edwards MSB, et al. Radiation-induced telangiectasia in the brain simulates cryptic vascular malformations at MR imaging. Radiology 1994;193:629–36 [DOI] [PubMed] [Google Scholar]
- 29.Pozzati E, Giangaspero F, Marliani F, et al. Occult cerebrovascular malformations after irradiation. Neurosurgery 1996;39:677–84 [DOI] [PubMed] [Google Scholar]
- 30.Iuvone L, Mariotti P, Colosimo C, et al. Long-term cognitive outcome, brain computed tomography scan, and magnetic resonance imaging in children cured for acute lymphoblastic leukemia. Cancer 2002;95:2562–70 [DOI] [PubMed] [Google Scholar]
- 31.Kingma A. Mooyaart EL. Kamps WA, et al. Magnetic resonance imaging of the brain and neuropsychological evaluation in children treated for acute lymphoblastic leukemia at a young age. Am J Pediatr Hematol-Oncol 1993;15:231–38 [DOI] [PubMed] [Google Scholar]
- 32.Paakko E, Talvensaari K, Pyhtinen J, et al. Late cranial MRI after cranial irradiation in survivors of childhood cancer. Neuroradiology 1994;36:652–55 [DOI] [PubMed] [Google Scholar]
- 33.Paakko E, Vainionpaa L, Pyhtinen J, et al. Minor changes on cranial MRI during treatment in children with acute lymphoblastic leukemia. Neuroradiology. 1996;38:264–68 [DOI] [PubMed] [Google Scholar]
- 34.Hertzberg J, Huk WJ, Ueberall MA, et al. CNS late effects after ALL therapy in Childhood. Part I. neuroradiological findings in long-term survivors of childhood ALL: an evaluation of the interferences between morphology and neuropsychological performance. Med Pedatr Oncol 1997;28:387–400 [DOI] [PubMed] [Google Scholar]
- 35.Paakko E, Harila-Saari A, Vanionpaa L, et al. White matter changes on MRI during treatment in children with acute lymphoblastic leukemia: correlation with neuropsychological findings. Med Pediatr Oncol 2000;35:456–61 [DOI] [PubMed] [Google Scholar]
- 36.Koike S, Aida N, Hata M, et al. Asymptomatic radiation-induced telangiectasia in children after cranial irradiation: frequency, latency, and dose relation. Radiology 2004;230:93–99 [DOI] [PubMed] [Google Scholar]
- 37.Chu WC, Chik KW, Chan YL, et al. White matter and cerebral metabolite changes in children undergoing treatment for acute lymphoblastic leukemia: longitudinal study with MR imaging and 1H MR spectroscopy Radiology 2003;229:659–69 [DOI] [PubMed] [Google Scholar]
- 38.Kramer JH, Norman D, Brant-Zawadzki, et al. Absence of white matter changes on magnetic resonance imaging in children treated with CNS prophylaxis therapy for leukemia. Cancer 1988;61:928–30 [DOI] [PubMed] [Google Scholar]
- 39.Seidel H, Nygaard R, Haave I, Moe PJ. Magnetic resonance imaging and neurological evaluation after treatment with high-dose methotrexate for acute lymphocytic leukaemia in young children. Acta Paediatr 1996;85:450–53 [DOI] [PubMed] [Google Scholar]
- 40.Price RA. Histopathology of CNS leukemia and complications of therapy. Am J Pediatr Hematol-Oncol 1979;1:21–30 [PubMed] [Google Scholar]
- 41.Price RA. Birdwell DA. The central nervous system in childhood leukemia. III. Mineralizing microangiopathy and dystrophic calcification. Cancer 1978;42:717–28 [DOI] [PubMed] [Google Scholar]
- 42.Langer T, Martus P, Ottensmeier H, et al. CNS late-effects after ALL therapy in childhood. Part III. Neuropsychological performance in long-term survivors of childhood ALL: impairments of concentration, attention, and memory. Med Pediatr Oncol 2002;38:320–28 [DOI] [PubMed] [Google Scholar]
- 43.Wilkins RH. Natural history of intracranial vascular malformations: a review. Neurosurgery 1985;16:421–30 [DOI] [PubMed] [Google Scholar]
- 44.Kim DS, Park YG, Choi JU, et al. An analysis of the natural history of cavernous malformations. Surg Neurol 1997;48:9–17 [DOI] [PubMed] [Google Scholar]
- 45.Lien HR, Blomlie V, Saeter G, et al. Osteogenic sarcoma: MR signal abnormalities of the brain in asymptomatic patients treated with high-dose methotrexate. Radiology 1991;179:547–50 [DOI] [PubMed] [Google Scholar]