Abstract
BACKGROUND AND PURPOSE: The imaging appearance of facial nerve schwannomas (FNSs) has been described as an enhancing tubular mass (using T1-enhanced MR) within an enlarged facial nerve canal (using CT). The purpose of this study is to identify how often the FNS imaging findings conform to this description and determine whether there are underlying anatomic explanations for the discrepant imaging appearances identified.
MATERIALS AND METHODS: The clinical, pathologic, and radiologic records of 24 FNS in 23 patients were retrospectively reviewed. Each FNS was evaluated for location along the facial nerve. The lesions were cataloged by facial nerve segment with the imaging characteristics of each segment described.
RESULTS: The average age at time of first imaging was 39 years (age range, 10–70 years). Eighteen (71%) of the 24 FNSs were pathologically confirmed, while the others were determined intraoperatively or diagnostically by the presence of both enlargement of the facial nerve canal and enhancement on contrast-enhanced T1 MR examination. The most common location was in the geniculate fossa (83%), followed by the labyrinthine and tympanic segments of the facial nerve (both 54%). The most common clinical presentation was facial neuropathy (42%).
CONCLUSION: The classic description of FNS on enhanced T1 MR is that of a well-circumscribed fusiform enhancing mass along the course of the intratemporal facial nerve with bone algorithm CT showing sharply defined bony canal enlargement. Modern imaging techniques, however, demonstrate the importance of the surrounding anatomic landscape, leading to various imaging appearances. Lesions traversing the labyrinthine segment can demonstrate a dumbbell appearance. When FNSs track along the greater superficial petrosal nerve, they may present as a round mass projecting up into the middle cranial fossa. FNS of the tympanic segment of the facial nerve preferentially pedunculate into the middle ear cavity, clinically presenting as a middle ear mass. When the mastoid segment of the facial nerve is involved, irregular and “invasive“ tumor margins seen on MR can be explained on CT as tumor breaking into surrounding mastoid air cells.
Facial nerve schwannomas (FNSs) are rare lesions that can arise anywhere along the course of the facial nerve, from its origin in the cerebellopontine angle to its extracranial ramifications in the parotid space of the extracranial head and neck.1,2 The lack of a large radiologic series of these lesions has resulted in a uniform imaging description of an enhancing tubular mass (using MR imaging) in a smoothly enlarged facial nerve canal (using CT).3 This retrospective review of the clinical and radiologic records of 24 FNSs was undertaken to determine whether this classic radiologic description accurately applies to all FNSs.
Although the classic description of FNS was identified, 5 additional distinct imaging appearances were seen along the course of the facial nerve. Cerebellopontine angle-internal auditory canal (CPA-IAC) FNSs were indistinguishable from acoustic schwannomas if they did not enter the labyrinthine segment of the facial nerve canal. FNSs centered in the geniculate fossa most commonly conformed to the classic tubular description of the lesion or were seen as round masses enlarging only the fossa itself. When a FNS extended along the greater superficial petrosal nerve, a round middle cranial fossa extra-axial mass was seen. A tympanic segment FNS often lobulated into the middle ear cavity, losing its tubular configuration. Finally, a mastoid segment FNS could break into adjacent mastoid air cells, thereby appearing as an aggressive tumor, especially on MR imaging.
Materials and Methods
The clinical and pathologic records and imaging studies of 23 patients with 24 FNSs within the temporal bone were retrospectively reviewed. One patient presented with bilateral lesions and had a known history of neurofibromatosis type II. Seventeen (71%) of these 24 lesions were pathologically confirmed, while the others were determined intraoperatively or diagnostically by the presence of both enlargement of the facial nerve canal and enhancement on contrast-enhanced MR imaging examinations. The imaging examinations were collected from 1986 to 2000 and consisted of CT (18 studies; 75%), MR imaging (17 studies; 77%), and high-resolution MR imaging with thin sections through the temporal bones (11 cases; 46%). In 12 cases, both CT and MR imaging were performed.
The involved portions of the facial nerve by these lesions were divided into the following 8 segments: cerebellopontine angle cistern segment, internal auditory canal segment, labyrinthine segment, geniculate fossa, greater superficial petrosal nerve involvement, tympanic segment, mastoid segment, and extracranial segment.
Results
Table 1 lists the location of the FNSs and basic clinical data of the cases. The average age at time of imaging was 39 years (range, 10 –70), with 17 (71%) of the patients between 30 and 63 years of age. A slight male predominance (14 of 24 cases; 58%) was observed. Two of the patients (3 lesions) had a known history of neurofibromatosis type II. Twenty-one of the 24 lesions (88%) in this series involved at least 2 contiguous segments of the facial nerve, while 17 lesions (71%) involved at least 3 contiguous segments.
The most common clinical presentation was peripheral facial nerve neuropathy (10 cases; 42%), and this dysfunction may encompass weakness, focal twitch, and/or full hemifacial spasm. Other clinical findings included sensorineural hearing loss in 7 cases (29%), conductive hearing loss in 4 cases (17%), clinical presentation with a middle ear mass in 3 cases (13%), and vestibular findings in 3 cases (13%)—these vestibular symptoms were not convincingly attributable to the facial nerve.
The involved portions of the facial nerve included the following 7 segments: cerebellopontine angle-internal auditory canal (12 lesions; 50%), labyrinthine (13; 54%), geniculate fossa (20; 83%), greater superficial petrosal nerve (6; 25%), tympanic (13; 54%), and mastoid (8; 33%).
Of the 12 cases involving the CPA-IAC segment, 11 (92%) also had extension into the labyrinthine segment, and 10 (83%) cases continued into the geniculate fossa (Figs 1 and 2). Four (33%) cases had continued extension into the tympanic segment of the facial nerve. Most of these cases (6; 50%) presented with sensorineural hearing loss, whereas only 4 (33%) presented with facial nerve neuropathy. Five (42%) cases were misdiagnosed preoperatively as an acoustic schwannoma.
Fig 1.
Axial T2-weighted MR imaging at the level of the internal auditory canals, demonstrating a large, homogeneous mass filling the right internal auditory canal and extending into the cerebellopontine angle (between white arrows). From Wiggins and Harnsberger (2001). Used with permission.
Fig 2.
Axial images demonstrating focal enlargement of the labyrinthine segment of the facial nerve on the axial bone algorithm CT (left), and a homogeneously enhancing mass filling the internal auditory canal with extension into the cerebellopontine angle and labyrinthine segment on the axial T1-weighted postcontrast-enhanced MR image from a similar level (right). These images are from the same case as in Fig 1, and the patient was misdiagnosed preoperatively because of failure to note the enhancement and enlargement along the labyrinthine segment into the geniculate fossa (arrows). From Wiggins and Harnsberger (2001). Used with permission.
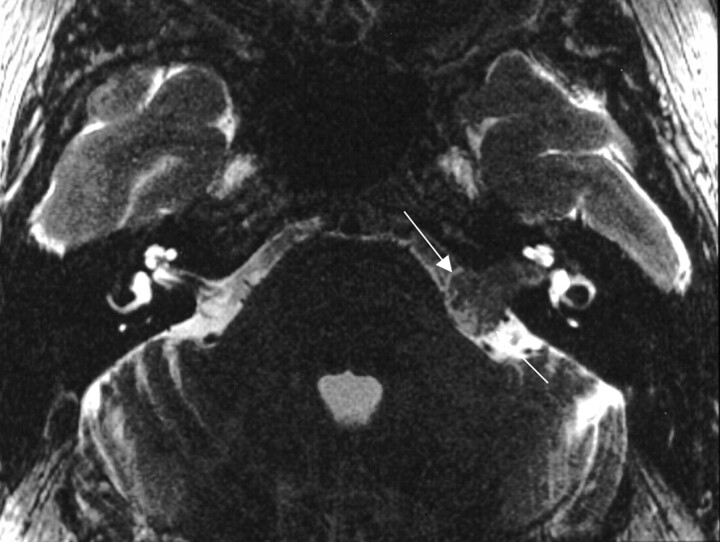
Of the 13 cases involving the labyrinthine segment, 11 (85%) of the lesions included extension into the internal auditory canal, and 12 (92%) of the lesions extended into the geniculate fossa (Fig 3). Six (46%) cases presented with a component of facial nerve neuropathy, whereas 5 (36%) presented with sensorineural hearing loss and one (8%) case described vestibular symptoms.
Fig 3.
Axial bone algorithm CT image demonstrating focal enlargement of the labyrinthine segment of the facial nerve canal from a facial nerve schwannoma (between arrows). From Wiggins and Harnsberger (2001). Used with permission.
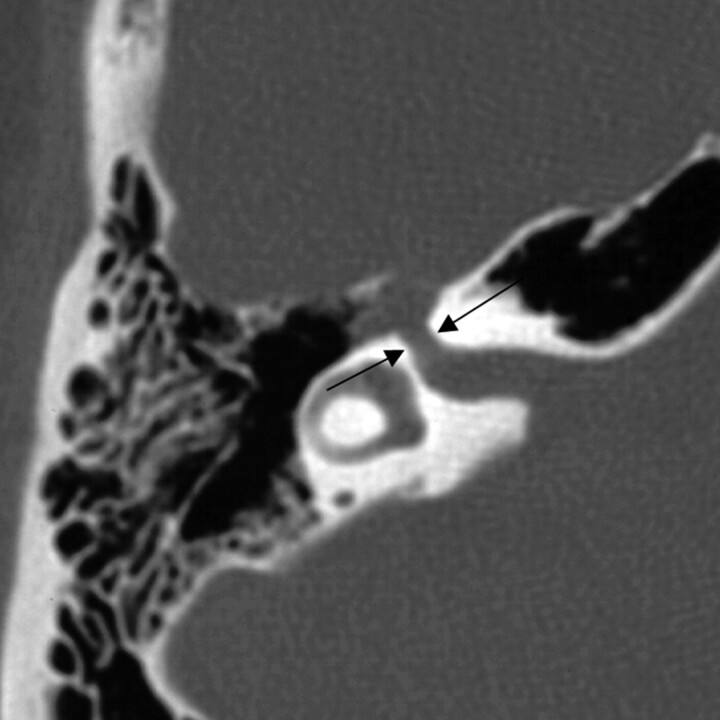
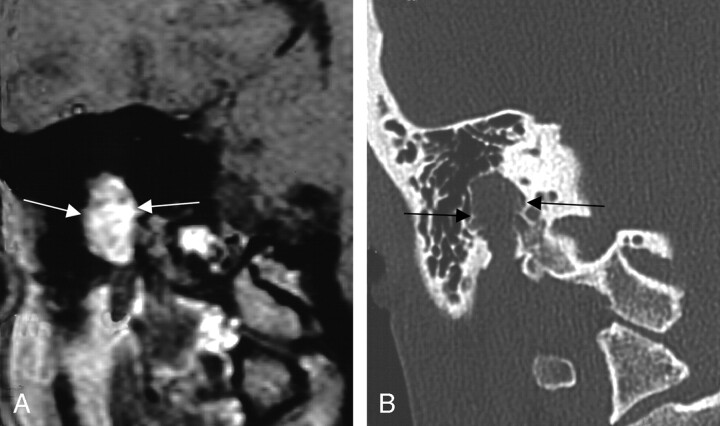
Twenty of the 24 lesions involved the geniculate fossa. Twelve (60%) of these lesions extended proximally into the labyrinthine segment, and an equal number of the lesions had distal extension into the tympanic segment (Fig 4). Six (30%) cases included extension along the greater superficial petrosal nerve. This group of cases demonstrated a variety of clinical presentations, with 9 (45%) patients originally complaining of a component of facial nerve neuropathy, 5 (25%) with sensorineural hearing loss, and 3 (15%) patients presenting with either conductive hearing loss or vestibular symptoms.
Fig 4.
Axial images from the same case of a facial nerve schwannoma, demonstrating homogeneous enhancement of the labyrinthine segment and geniculate fossa on the axial T1-weighted postcontrast-enhanced MR imaging (left), and focal enlargement of the corresponding geniculate fossa and the labyrinthine segment of the facial nerve on the bone algorithm CT (right), at a similar level (between arrows). From Wiggins and Harnsberger (2001). Used with permission.
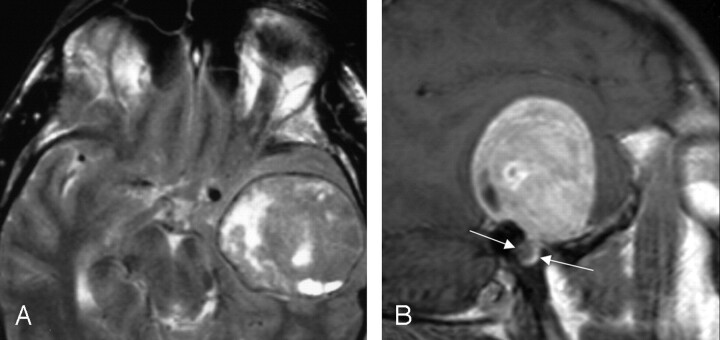
All 6 (100%) of the lesions with involvement of the greater superficial petrosal nerve included geniculate fossa involvement, but only 3 (50%) demonstrated extension along the proximal labyrinthine or distal tympanic segment. Two (33%) cases presented with the predominant imaging finding of a middle cranial fossa mass (Fig 5). There was no clinical predominant presentation in the lesions, with 2 (33%) presenting with both facial nerve neuropathy and conductive hearing loss, whereas one (17%) case presented with sensorineural hearing loss and one (17%) with vestibular findings.
Fig 5.
MR images demonstrating a large left middle cranial fossa mass. The axial T2-weighted image (left), and sagittal T1-weighted postcontrast image (right) show an extra-axial lesion, with a visible CSF/vascular cleft and associated buckling of the gray/white junction. The right image demonstrates a focal bulbous portion of the large mass extending to the geniculate fossa (between arrows), which was the origin of this FNS. From Wiggins and Harnsberger (2001). Used with permission.
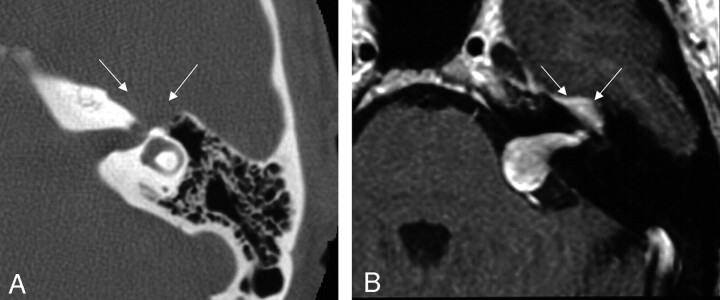
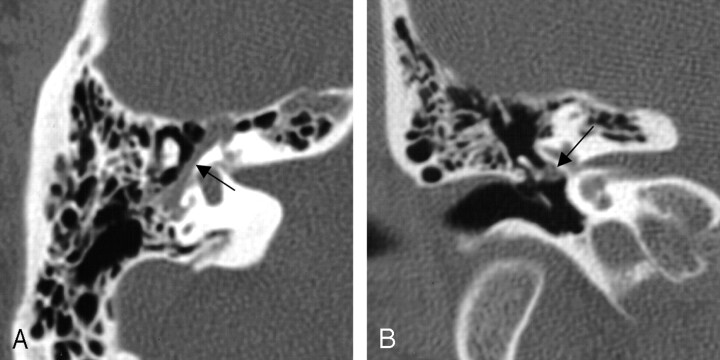
Of the 13 cases involving the tympanic segment, 12 (92%) of the lesions included proximal involvement of the geniculate fossa, but only 7 (54%) involved the mastoid segment distal to the posterior genu of the facial nerve course (Figs 6 and 7). These cases most often presented with a component of facial nerve neuropathy (6 cases; 46%), while 3 (23%) of the patients presented with a middle ear mass.
Fig 6.
Bone algorithm CT images from the same case, demonstrating focal enlargement of the right tympanic segment, in the axial (left) and coronal (right) planes (at arrows). From Wiggins and Harnsberger (2001). Used with permission.
Fig 7.
Two different cases of FNS, both coronal plane bone algorithm CT images show significant enlargement of the tympanic segment of the facial nerve with the case on the left demonstrating extension of the lesion primarily superiorly and medially, as well as extension into the cochlea, whereas the lesion on the right shows extension primarily inferiorly and laterally, displacing the ossicles, explaining this patient’s presentation with conductive hearing loss and a middle ear mass on clinical examination (at arrows). From Wiggins and Harnsberger (2001). Used with permission.
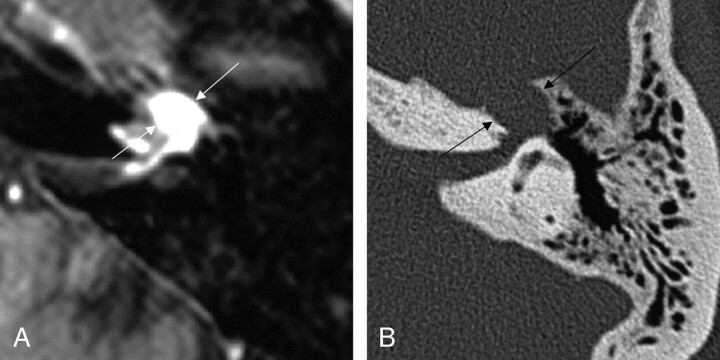
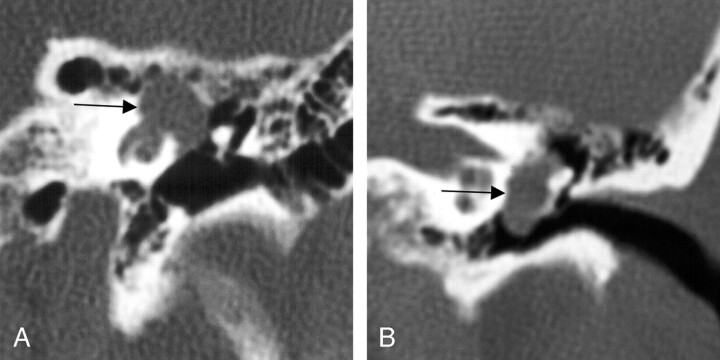
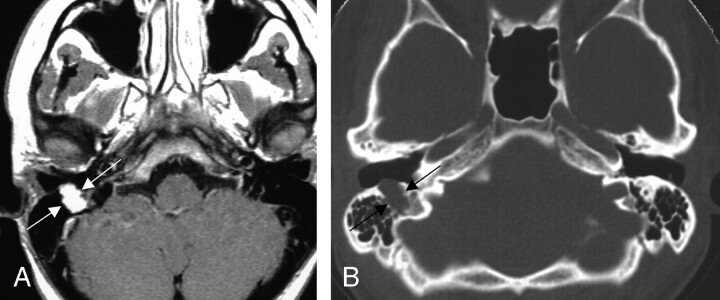
Seven (86%) of the 8 cases collected that included involvement of the mastoid, or descending, segment of the facial nerve course (Figs 8 and 9) included proximal involvement of the tympanic segment, and 2 (13%) of the lesions extended through the stylomastoid foramen to include extratemporal extension.
Fig 8.
Two axial images from the same case of a facial nerve schwannoma involving the right mastoid segment of the facial nerve canal. The axial T1-weighted postcontrast MR image (left) shows homogeneous enhancement of the mass (between arrows). The bone algorithm CT (right) at the same level shows focal enlargement of the descending segment with extension toward the external auditory canal (between arrows). From Wiggins and Harnsberger (2001). Used with permission.
Fig 9.
Two coronal views of the same case, demonstrating the aggressive appearing imaging features of mastoid segment FNSs. The left coronal T1-weighted postcontrast-enhanced image shows homogeneous enhancement. The right coronal bone algorithm CT study demonstrated an aggressive appearing lesion, as the FNS erupts into the surrounding thin-walled mastoid air cells of the same lesion (between arrows). From Wiggins and Harnsberger (2001). Used with permission.
Discussion
FNSs are classically described on T1-enhanced MR as a well-circumscribed fusiform enhancing masses along the course of the intratemporal facial nerve.2,3 Bone algorithm CT examinations are said to demonstrate sharply defined bony canal enlargement. Modern MR and CT imaging techniques with thinner sections through the temporal bones demonstrate greater detail of the imaging features of FNS and increase the number of lesions diagnosed. This refinement in the classic “one-size-fits-all” imaging description of FNSs no longer suffices to guide the radiologist’s interpretation of these lesions, as our improved imaging abilities now show the effect of the surrounding anatomic landscape on these lesions. This review of a relatively large number of these lesions demonstrates how the FNS imaging appearance is defined by the particular intratemporal region involved.
The wide variety of symptoms can signal the presence of a FNS including facial nerve paralysis, sensorineural hearing loss, and conductive hearing loss. Symptoms are highly dependent on the location of this lesion in the temporal bone. This range of symptoms makes FNSs difficult to diagnose preoperatively without radiologic examination. These lesions will usually expand along the path of least resistance and can remain asymptomatic until large without clinically presenting with facial nerve dysfunction.3 Fine-needle aspiration may not be useful for the definitive diagnosis, so the preoperative suggestion of this diagnosis can greatly affect the patient’s management.4 Early diagnoses positively affects patient outcome, because smaller lesions are more readily removed with successful facial nerve repair.
Schwannomas in any location in the human body characteristically demonstrate homogeneously or heterogeneously enhancing soft tissue attenuation that is isoattenuated to gray matter on contrast-enhanced CT and may contain cystic foci.5 In the temporal bone, bone algorithm CT examination of a FNS will show benign-appearing bony scalloping and remodeling of surrounding bony boundaries. FNSs do not show true “erosion” of the bony boundaries as was described in many early reports,6–9 but they do demonstrate a more benign osseous expansion of the surrounding anatomic landscape. MR imaging can show heterogeneity within large FNSs and hyperintensity on T2-weighted series, with iso- to hypointensity relative to gray matter on T1-weighted images.5,6 Postcontrast-enhanced MR images usually demonstrate homogeneous enhancement. Larger FNSs can undergo a cystic degeneration seen as focal intramural low signal intensity on contrast-enhanced T1 images.5
Three other differential considerations for the radiologist evaluating intratemporal facial nerve lesions are cholesteatoma, facial nerve hemangioma, and perineural parotid malignancy.3 In all 3, CT shows enlargement of the intratemporal facial nerve canal. Congenital or invasive acquired cholesteatoma that involve the facial nerve canal do not enhance on postcontrast-enhanced T1 MR, easily distinguishing themselves from FNS. Facial nerve hemangioma involve both the facial nerve canal and adjacent bone, with more aggressive bony changes seen as irregular margins and/or a “moth-eaten” appearance. When an internal calcified honeycomb matrix is present (50%), ossifying hemangioma is quite distinctive.6 Parotid malignancy with perineural spread can be seen extending cephalad from an intraparotid invasive mass from distal to proximal along an enlarged facial nerve canal.6
FNSs are often discovered in patients presenting with only minor facial nerve symptoms, frequently dismissed at the clinical level as an idiopathic Bell’s palsy.10,11 T1-enhanced MR imaging commonly shows intratemporal facial nerve enhancement of all or part of the facial nerve along with a “tuft” of enhancement in the fundus of the internal auditory canal in patients with Bell’s palsy. CT, however, does not show enlargement of the facial nerve canal in Bell’s palsy.6 The normal size of the bony facial nerve canal is also used to distinguish normal facial nerve enhancement from a FNS. The prominent circumneural arteriovenous plexus surrounding the facial nerve causes segmental facial nerve enhancement distal to the labyrinthine segment in 75% of thin-section, focused T1-enhanced MR images.12,13
FNSs involving the CPA-IAC facial nerve segments can be indistinguishable from acoustic schwannomas if no extension into the labyrinthine segment of the facial nerve is present.7,14 Consequently, it is imperative that all cases of newly diagnosed acoustic schwannoma be inspected for a “labyrinthine tail” that identifies them as a FNS. It is important to remember that the presenting symptom will not always suggest these lesions. Half of the cases in this series presented with sensorineural hearing loss, whereas only a third presented with a component of facial nerve paralysis. It is not surprising that half of these cases were misdiagnosed preoperatively.
When extensive, a CPA-IAC FNS can present with an unusual but distinctive imaging appearance, a “dumbbell” shape due to extension from the IAC fundus through the labyrinthine segment and into the geniculate fossa.15 This configuration along with T1 MR enhancement and sharply scalloped, or fusiform, enlargement of the facial nerve canal is diagnostic of a FNS. This dumbbell FNS must be distinguished from the transmodiolar acoustic schwannoma. This is done by identifying extension into the cochlea in a transmodiolar schwannoma.15
Lesions involving the geniculate fossa primarily, or extending along the greater superficial petrosal nerve, can present with a middle cranial fossa mass. When lesions of the middle cranial fossa are determined to be extra-axial, inspection of the skull base and temporal bone to rule out origination from the geniculate ganglion or greater superficial petrosal nerve is recommended. Lesions arising from the geniculate ganglion will have “bulbous” enlargement at the geniculate fossa itself. FNSs emanating from the greater superficial petrosal nerve scallop the anterior margin of the geniculate fossa and the adjacent bony petrous apex.16–18
The tympanic segment of the facial nerve lacks the thick surrounding bony architecture of the labyrinthine segment of the facial nerve canal. FNSs involving this segment often do not have fusiform morphology but instead are multilobular. When they lobulate superiorly or medially, a fistula due to erosion into the lateral semicircular canal may be seen. Inferolateral lobulation into the middle ear cavity causes lateral displacement of the ossicles, possibly leading to a presentation of conductive hearing loss. A retrotympanic avascular mass associated with conductive hearing loss may result.
The anatomic landscape explains the variation in imaging appearance of FNS of the mastoid segment of the facial nerve. The mastoid segment of the facial nerve is surrounded by fragile, thin-walled septations separating the mastoid air cells. When the mastoid segment of the facial nerve is involved, irregular, “invasive” tumor margins may be seen on MR. CT explains these margins by showing the FNS breaking into surrounding mastoid air cells. When viewed without this anatomic context, misdiagnosis as a facial nerve hemangioma or other locally invasive mass results.
Surgical Considerations
The surgical management of facial schwannoma depends on not only the size and anatomical location of the tumor, but also the patient’s hearing status. A tumor proximal to the geniculate ganglion with serviceable hearing should be approached through the middle cranial fossa provided the tumor does not extend far into the cerebellopontine angle. If the tumor is proximal to the midIAC with ≤1 cm of a CPA component, an extended middle cranial fossa approach is best, whereas a retrosigmoid approach gives the best chance of hearing conservation in lesions with a CPA component >1 cm. With nonserviceable hearing, a translabyrinthine approach is the most direct route to the tumor and is the procedure of choice.19–21 This approach also provides the best access for facial nerve grafting. Involvement of the tympanic segment can be reached by a transmastoid approach with facial recess opening. Mastoid segment tumor alone can be extirpated through the mastoid, whereas additional extratemporal involvement may require following the nerve into the parotid gland. These exposures may be used in combination for tumors involving multiple nerve segments.
Timing of surgery is controversial. Many patients with facial nerve tumors have normal or near-normal nerve function and removal with grafting cannot lead to function better than House-Brackmann grade III.21 Results from grafting, however, are influenced by the length of time of any preoperative facial dysfunction, so early surgery may offer the best hope for good facial function.20,21 Moreover, labyrinthine fistula can develop from bony erosion from the tumor and can lead to deafness and dizziness if the tumor is neglected too long.1,22 Some authors recommend following patients with small tumors and normal clinical function until they show >50% denervation on electrical testing of the nerve to tackle these difficult issues.23
A further controversy involves reconstruction of the nerve. Some authors have reported success with extirpation of the tumor and preservation of nerve integrity,24,25 but this does not guarantee good facial function. Primary anastomosis without tension is rarely possible without mastoid segment rerouting, which (when possible anatomically) may compromise neural blood supply. Interposition grafting is usually performed with the greater auricular or sural nerves, but any rehabilitative technique that relies on the native facial muscles becomes ineffective once severe atrophy has occurred. Although a cranial nerve 12 to cranial nerve 7 anastamosis often works after as much as a couple of years, long delays require other strategies such as static or dynamic slings or serratus microvascular techniques. Cranial nerve 12 to cranial nerve 7 anastomosis is usually indicated when there is no useful cranial nerve VII stump at the brain stem.
Conclusion
The imaging appearance of facial nerve schwannoma is more varied than originally described. The classic tubular enhancing mass on T1 MR associated with smooth enlargement of the facial nerve canal on CT is actually only seen in FNS localized to the CPA-IAC or labyrinthine segments. More extensive FNSs that extend from the CPA-IAC to the geniculate fossa conform to a “dumbbell” shape. Geniculate ganglion and greater superficial petrosal nerve FNSs can present as a middle cranial fossa mass that may be misdiagnosed unless inspection of the skull base and temporal bone is completed. Tympanic segment FNSs are often multilobular, dehiscing into the middle ear to present as an avascular retrotympanic mass with conductive hearing loss. Finally, mastoid segment FNSs can appear on MR imaging as locally aggressive masses when they break into the surrounding mastoid air cells.
References
- 1.O’Donoghue GM, Brackmann DE, House JW, et al. Neuromas of the facial nerve. Am J Otol 1989;10:49–54 [PubMed] [Google Scholar]
- 2.Symon L, Cheesman AD, Kawauchi M, et al. Neuromas of the facial nerve: a report of 12 cases. Br J Neurosurg 1993;7:13–22 [DOI] [PubMed] [Google Scholar]
- 3.Chung SY, Kim DI, Lee BH, et al. Facial nerve schwannomas: CT and MR findings. Yonsei Med J 1998;39:148–53 [DOI] [PubMed] [Google Scholar]
- 4.Balle VH, Greisen O. Neurilemmomas of the facial nerve presenting as parotid tumors. Ann Otol Rhinol Laryngol 1984;93:70–72 [DOI] [PubMed] [Google Scholar]
- 5.Lidov M, Som PM, Stacy C, et al. Eccentric cystic facial schwannoma: CT and MR features. J Comp Axial Tomgr 1991;15:1065–67 [DOI] [PubMed] [Google Scholar]
- 6.Martin N, Sterkers O, Mompoint D, et al. Facial nerve neuromas: MR imaging. Neuroradiology 1992;34:62–67 [DOI] [PubMed] [Google Scholar]
- 7.Inoue Y, Tabuchi T, Hakuba A, et al. Facial nerve neuromas: CT findings. J Comp Axial Tomogr 1987;11:942–47 [DOI] [PubMed] [Google Scholar]
- 8.Latack JT, Gabrielson TO, Knake JE, et al. Facial nerve neuromas: radiologic evaluation. Radiology 1983;149:731–39 [DOI] [PubMed] [Google Scholar]
- 9.McMenomey SO, Glasscock ME III, Minor LB, et al. Facial nerve neuromas presenting as acoustic tumors. Am J Otol 1994;15:307–12 [PubMed] [Google Scholar]
- 10.Pillsbury HC, Price HC, Gardiner LJ. Primary tumors of the facial nerve: diagnosis and management. Laryngoscope 1983;93:1045–48 [DOI] [PubMed] [Google Scholar]
- 11.Zhang Q, Jessurun J, Schachern PA, et al. Outgrowing schwannoma arising from tympanic segments of the facial nerve. Am J Otol 1996;17:311–15 [DOI] [PubMed] [Google Scholar]
- 12.Daniels DL, Czervionke LF, Pojunas KW, et al. Facial nerve enhancement in MR imaging. AJNR Am J Neuroradiol 1987;8:605–7 [PMC free article] [PubMed] [Google Scholar]
- 13.Gebarski SS, Telian SA, Niparko JK. Enhancement along the normal facial nerve in the facial canal: MR imaging and anatomic correlation. Radiology 1992;183:391–94 [DOI] [PubMed] [Google Scholar]
- 14.Fagan PA, Misra SN, Doust B. Facial neuroma of the cerebellopontine angle and the internal auditory canal. Laryngoscope 1993;103:442–46 [DOI] [PubMed] [Google Scholar]
- 15.Salzman KL, Davidson HC, Harnsberger HR, et al. Dumbbell schwannomas of the internal auditory canal. AJNR Am J Neuroradiol 2001;22:1368–76 [PMC free article] [PubMed] [Google Scholar]
- 16.Kienzle GD, Goldberg MH, Just NWM, et al. Facial nerve neurinoma presenting as middle cranial fossa mass: CT appearance. J Comp Axial Tomogr 1986;10:391–94 [PubMed] [Google Scholar]
- 17.Ginsberg LE, DeMonte F. Case 16: Facial nerve schwannoma with middle cranial fossa involvement. Radiology 1999;213:364–68 [DOI] [PubMed] [Google Scholar]
- 18.KumonY, Sakaki S, Ohta S, et al. Greater superficial petrosal nerve neurinoma: case report. J Neurosurg 1999;91:691–96 [DOI] [PubMed] [Google Scholar]
- 19.Browning ST, Phil M, Phillips JJ, et al. Pathology in focus. Schwannoma of the chorda tympani nerve. J Laryngol Otol 2000;114:81–82 [DOI] [PubMed] [Google Scholar]
- 20.King TT, Morrison AW. Primary facial nerve tumors within the skull. J Neurosurg 1990;72:1–8 [DOI] [PubMed] [Google Scholar]
- 21.Lipkin AF, Coker NJ, Jenkins HA, et al. Intracranial and intratemporal facial neuroma. Otolaryngol Head Neck Surg 1987;96:71–79 [DOI] [PubMed] [Google Scholar]
- 22.Sanna M, Zini C, Gamoletti R, et al. Primary intratemporal tumors of the facial nerve: Diagnosis and treatment. J Laryngol Otol 1990;104:765–71 [DOI] [PubMed] [Google Scholar]
- 23.Shelton C. Facial nerve tumors. In: Brackmann D, Shelton C, Arriaga M. eds. Otologic surgery. Philadelphia: WB Saunders;1994. :413–23
- 24.Pulec J. Symposium on ear surgery. 11. Facial nerve neuroma. Laryngoscope 1972;82:1160–76 [DOI] [PubMed] [Google Scholar]
- 25.Lundgren N. Neuroma n. facialis. Acta Oto Laryngol 1947;35:535–37 [Google Scholar]
- 26.Wiggins RH, Harnsberger HR. Used with permission from Head and Neck Digital Teaching File. Salt Lake City: Advanced Medical Imaging Reference Systems;2003