Abstract
BACKGROUND AND PURPOSE: In this study, intensive video electroencephalogram (EEG) monitoring, high-resolution MR imaging (MR imaging), proton MR spectroscopy (1H-MR spectroscopy) and single-photon emission CT (SPECT) were compared in patients with temporal lobe epilepsy (TLE) to evaluate lateralization of affected hemisphere with regard to bilateral affection and postoperative outcome.
PATIENTS AND METHODS: Recall ratio of each technique for indicating the affected hemisphere was determined in 49 patients with TLE. Postoperative outcome was established by Engel classification.
RESULTS: Twenty-two of 25 patients with TLE with evidence for hippocampal sclerosis in MR imaging (MR imaging-positive) were graded as unilateral by EEG findings whereas 3 were classified as bilateral. Fourteen of 24 MR imaging-negative patients were graded as unilateral by EEG and 10 as bitemporal. 1H-MR spectroscopy indicated concordant lateralization to EEG in 82% of MR imaging-positive patients and 71% of MR imaging-negative patients and to SPECT in 84% of MR imaging-positive patients and 67% of MR imaging-negative patients with TLE. In unilateral TLE, the concordance rate of both modalities was 74% in MR imaging-positive patients and 67% in MR imaging-negative patients. Contralateral findings to EEG focus were found in 28% by 1H-MR spectroscopy and in 27% by SPECT. Concordant findings to the operated side of different modalities revealed a clear tendency (P = .08) for a better postoperative outcome compared with bitemporal or contralateral findings.
CONCLUSION: Our data demonstrate that multimodal imaging in patients with TLE improves lateralization of affected hemispheres, especially in patients without pathologic findings in MR imaging, and indicates bilateral effect, which is important to identify patients who will benefit from surgery.
The intention of epilepsy surgery is to achieve complete seizure control in medically intractable patients. This requires correct lateralization of the affected hemisphere with clear focus localization. Although the value of invasive tests, such as long-term intracranial electroencephalography (EEG) with surgically implanted electrodes, cannot be underestimated,1 noninvasive alternative methods and imaging tests are increasingly being implemented.2,3 Concordance of different modalities in focus localization increases evidence for correct hemispheric lateralization, which is the basis for successful treatment of patients with temporal lobe epilepsy (TLE).4–7 Particularly in the case of inconsistent findings in intensive video-EEG monitoring, proton MR spectroscopy (1H-MR spectroscopy), single-photon emission CT (SPECT), and positron-emission tomography (PET) together with high-resolution MR imaging are useful complementary tools in the diagnosis of TLE.3,7–12 For this reason, we analyzed different modalities, including high-resolution MR imaging, 1H-MR spectroscopy, and SPECT in focusing epileptogenic lesions in 49 patients diagnosed with TLE. Intensive 24-hour video EEG monitoring was used as the “gold standard” for focus localization.13,14 Postoperative outcome was compared between patients with TLE with unilateral and ipsilateral findings to EEG focus and patients with bilateral or contralateral pathologic conditions to EEG focus. The objective of the present study was to compare sensitivity of different modalities in lateralization of the affected hemisphere in patients with TLE with lesional and nonlesional MR imaging findings (MR imaging-negative and -positive subgroups). Our hypotheses were that multimodal imaging, especially in patients with TLE without pathologic findings in MR imaging (MR imaging-negative), increases sensitivity in lateralization of the affected hemisphere and additionally provides essential information concerning possible bilateral involvement. This information is important for understanding pathogenesis and planning therapeutic schedules.
With a multimodal approach, we could verify acquired data and address the most important clinical issue, which is determining patients who will benefit from surgery. If possible, in some difficult cases, intracranial EEG could be entirely avoided. Another question refers to optimizing the utilization of each technique.
Patients and Methods
Our study included 49 consecutive patients diagnosed with TLE who were scheduled for preoperative work-up in the grade IV Epilepsy Center Erlangen in the years 2002–2005. Diagnosis of TLE was based on clinical history, seizure description, and results of intensive video EEG monitoring. Surgical outcome in patients with hippocampal sclerosis (Fig 1) was classified by Engel et al15 at 3, 6, and 12 months.
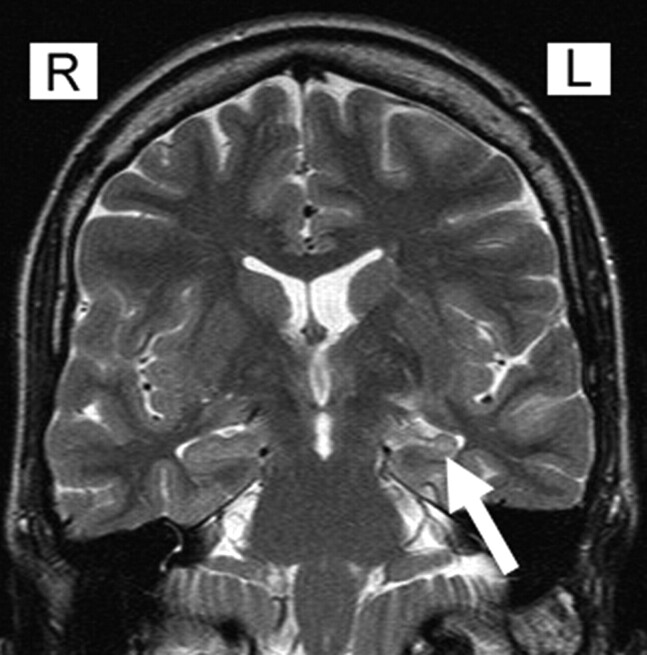
Fig 1.
Coronal T2 MR imaging revealing increased signal intensity and atrophy of the left hippocampus in a patient with hippocampal sclerosis (arrow).
EEG Monitoring
The simultaneous registration of continuous video EEG monitoring was performed using the Glonner system (Munich, Germany). Simultaneous documentation of behavior via a split-screen technique was used day and night over a period of 1–2 weeks. Technical details are: number of EEG/polygraphic channels, 64/128; sampling rate, 200 Hz; video EEG data were stored on videotapes and EEG data on the computer for analysis and reviewing. EEG electrodes were placed according to the international 10/20 system of EEG. In addition, a basal ring of electrodes (S01-S02; F11-F12; TP9-TP10, FT9-FT10) and sphenoidal electrodes were placed to evaluate interictal and ictal epileptic activity in temporal lobes more sufficiently. EEG analysis was performed using continuous 24-hour, day and night video EEG monitoring according to therapeutic intensive seizure analysis as described by Stefan et al.16 EEG analysis was performed with no knowledge of 1H-MR spectroscopy and SPECT results. Interictal epileptiform discharges (IEDs) and ictal EEG findings (IDs) were calculated by the number of IEDs/IDs counted on either side as a percentage of the total number of IEDs/IDs recorded on both sides. Unilateral activity was defined by IEDs/IDs, which showed ≥80% unilateral activity. In the case of <80% unilateral IEDs/IDs activity was graded as bitemporal. We have to mention here that interictal and ictal findings do not unequivocally denote the epileptogenic zone. Therefore the term “affected hemisphere,” which is used in the article, refers to the most involved hemisphere according to interictal or ictal findings of intensive 24-hour video EEG monitoring.
High-Resolution MR Imaging
MR imaging was performed at 1.5T Magnetom Sonata (Siemens Medical Solutions, Erlangen Germany) with a standard head coil and included the following sequences: T2-weighted, fluid-attenuated inversion recovery, transversal angulated (parallel to the long axis of the hippocampi; TR, 10,000 ms; TE, 109 ms; section thickness, 5 mm); T2-weighted perpendicular to the long axis of the hippocampi (TR, 7420 ms; TE, 98 ms; section thickness, 3 mm); T2*weighted gradient-echo (fast low-angle shot; TR, 858 ms; TE, 26 ms; section thickness, 5 mm); T1-weighted 3D gradient-echo (magnetization-prepared rapid acquisition of gradient echo; 1-mm 3D postprocessing; TR, 2030 ms; TE, 3.93 ms; matrix, 256 × 256); T1-weighted inversion recovery, perpendicular to the long axis of the hippocampus (TR, 9650 ms; TE, 16 ms; section thickness, 3 mm); T1-weighted gadolinium-enhanced at first admittance to exclude other pathologic alterations (eg, inflammatory disease, cavernoma). Patients with evidence for extrahippocampal pathologic conditions were excluded. Images were blinded to secure diagnoses and independently analyzed by 2 experienced neuroradiologists. The criteria for a diagnosis of hippocampal sclerosis on MR images included the presence of unilateral atrophy and high T2 signal intensity of the hippocampus (Fig 1).
Single-Voxel Spectroscopy
All experiments were carried out using a 1.5T Magnetom Sonata (Siemens Medical Solutions) clinical whole-body MR scanner using a standard head coil. Water suppression was achieved by 3 chemical shift-selective pulses before the point-resolved spectroscopy; TR, 3000 ms; TE, 30 ms; averages, 128; vector size, 1024. Determination of the absolute metabolite concentrations in single-voxel spectroscopy was carried out by water scaling, a method in which the resonance area of the unsuppressed water signal intensity was used as an internal reference. An appropriate water-unsuppressed voxel of identical voxel localization and measurement parameters was obtained for each water-suppressed voxel, differing only in the number of acquisitions that were reduced to 12. N-acetylaspartate + N-acetylaspartyl-glutamate (tNAA), choline (Cho), creatine (Cr), total glutamate plus glutamine (Glu+Gln) and myo-inositol (mIns) were quantified using LCModel (S. Provencher, Oakville, Ontario, Canada; available at http://s-provencher.com/pages/lcmodel.shtml), which is a user-independent frequency domain spectral-fitting program. Water scaling and fitting were carried out automatically. All concentrations are estimated metabolite levels with an unknown degree of T1 saturation and are referred to as institutional units (arbitrary units of our institute). Estimated uncertainties by Cramer-Rao lower bounds served as main guidelines for judging the spectra of absolute metabolite concentrations. Only metabolite spectra with LCModel estimated uncertainty of <15% SD of the evaluated concentrations and spectra with a signal-to-noise ratio (SNR) above 3 were included in this study. A constrained regularization method was used to estimate line shape and baseline, which can account for the residual water signal intensity. Spectra with a full width at half-maximum (FWHM) of more than 0.065 ppm were not included in the study. For further details, see Hammen et al17 and Provencher and Helms et al.18–20 The partial volume effect by CSF was determined by manually segmenting CSF on coronal T2-weighted images. The amount of CSF in the voxel was found by manually segmenting CSF on coronal T2-weighted images and was expressed as a percentage of voxel volume. The corrections of each metabolite for CSF dilution were carried out by the formula: Ccorr = (Cmeas × 100)/(100 [mins] CSFperc). Cmeas refers to measured concentration of metabolite of the whole voxel. CSFperc corresponds to the percentage of CSF in the voxel. In addition to absolute metabolite concentration, metabolite ratios (NAA/(Cr + Cho)) were calculated. Analysis of metabolite spectra was performed with no knowledge of EEG, SPECT, and MR imaging results. Spectra were considered pathologic in the case of hippocampal metabolites or a metabolite ratio exceeding a range of 2 SD of age-matched, healthy volunteers of 14 women and 16 men.
SPECT
Technetium Tc99m ethyl cysteinate dimmer (Tc99m-ECD) SPECT was performed using the triple-headed camera Multispect 3 (Siemens, Hoffman Estates, Ill) equipped with low-energy, ultra high-resolution collimators. Data acquisition was started 30 minutes after intravenous injection of 740 MBq ECD. One hundred twenty views (3 × 40; 3.0°/step), each registered over 40 seconds, were recorded into a 128 × 128 matrix with a pixel size of 2.9 × 2.9 mm2. Each view had 32 to 52 × 103 counts. Transaxial tomograms were reconstructed without prefiltering using filtered back-projection with a Butterworth filter of fifth order and a cutoff frequency of 0.3 Nyquist. In-plane resolution of the reconstructed images was 11 mm FWHM, and section thickness was approximately 5.8 mm. Focus localization was based mainly on interictal SPECT images. Only in some difficult cases was ictal SPECT carried out (On-line Tables 1–4). Within these limits, a digital subtraction of an interictal scan from an ictal scan and then coregistration to MR anatomic images was performed (Figs 2 and 3).
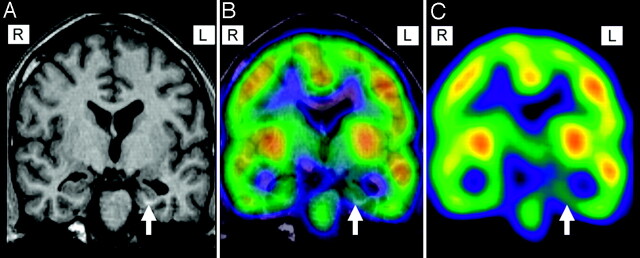
Fig 2.
Coregistration of MR imaging (magnetization-prepared rapid acquisition of gradient echo [A]) and interictal SPECT image (C) for precise anatomic focus localization in a patient with left temporal lobe epilepsy. SPECT displays hypoperfusion in the left mesial temporal lobe (arrow) ipsilateral to atrophy in MR imaging (B).
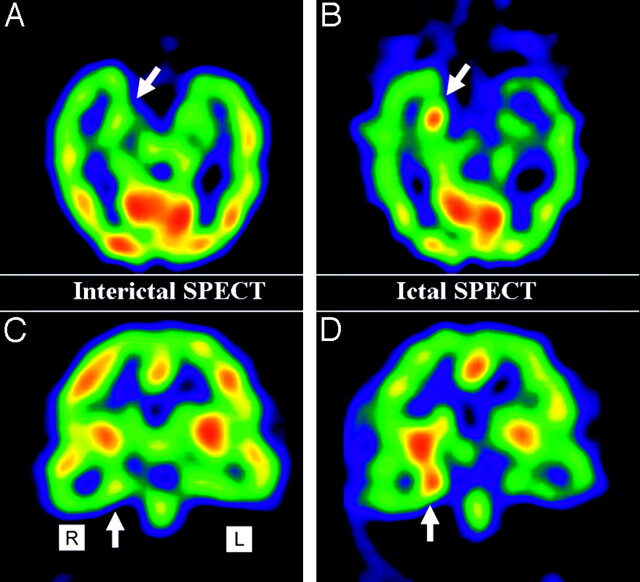
Fig 3.
Example of an interictal SPECT image with problematic changes of relative cerebral blood flow (rCBF) in the right mesial temporal lobe (arrow; A and B). Ictal SPECT elucidates hyperperfusion in the hippocampus (arrow; C and D).
Statistics
Mean and SD were calculated for each metabolite and NAA/(Cr +Cho) ratio. Spectral data were considered pathologic in the case of data exceeding 2 SD from the mean of healthy age-matched control subjects. NAA/(Cho + Cr) ratio was used in 41 of 49 patients for lateralization of the affected hemisphere. In the remaining cases, absolute metabolites (tNAA) were taken into consideration because of spectra of Cr and Cho that were unusable. Normal distribution of healthy volunteers and patients was calculated by the Kolmogorov-Smirnov index. The results of different modalities in lateralization of the affected hemisphere are displayed in 4-fold table (On-line Tables 5–7). The sensitivity was calculated as follows: sensitivity (t) = RP/RP + FN, where RP is right positive and FN is false negative. In patients who were classified as unilateral by EEG, summarized sensitivity was calculated by the number of patients who showed ipsilateral pathologic findings and bilateral findings. Bilateral findings were assigned to ipsilateral findings with additive contralateral pathologic findings. For patients with nonlesional MR imaging (negative MR imaging subgroup; On-line Table 7 [n = 10]) and bilateral EEG discharges (n = 10), the sensitivity of each technique was calculated as follows: t = total number of pathologic hemispheres in each technique/total number of all hemispheres (On-line Table 7; n = 20) × 100.
In patients graded as unilateral by EEG, postoperative outcome was classified by Engel et al.15 In unilateral, MR imaging-positive patients with TLE, postoperative outcome was compared between ipsilateral findings to EEG in 1H-MR spectroscopy and SPECT and patients with bilateral or contralateral pathologic findings in 1H-MR spectroscopy or SPECT by Mann and Whitney U test. Statistical analysis was carried out by SPSS 12.0. (SPSS, Chicago, Ill).
Results
In 49 patients diagnosed with TLE, 25 patients gave evidence for hippocampal sclerosis in MR imaging, whereas 24 patients showed no pathologic findings in hippocampal structures. In the first patient group with evidence for hippocampal sclerosis all patients had ictal and interictal EEG discharges ipsilateral to the lesion (On-line Table 5), and patients also demonstrated contralateral EEG discharges. In the second group, including 24 patients with nonlesional MR imaging findings, 14 patients demonstrated unilateral ictal and interictal EEG focus localization (On-line Table 6), whereas 10 patients revealed bilateral EEG discharges (On-line Table 7).
In 39 patients, ictal and interictal results were available for focus determination. Ten patients had only interictal EEG findings owing to the absence of ictal activity during video-EEG monitoring. In these cases, only interictal discharges in EEG could be used for focus localization. In 5 cases, SPECT was not performed. In the MR imaging-positive subgroup showing evidence for hippocampal sclerosis, no surgical resection was done in 10 patients because of either imprecise focus determination or refusal of the patient. In the MR imaging-negative group, only a single patient underwent surgical resection. The results in hemispheric lateralization of the different imaging modalities are displayed in On-line Tables 5–7.
Unilateral EEG
Positive MR Imaging.
In patients with evidence for hippocampal sclerosis (lesional subgroup) in MR imaging and ipsilateral (unilateral) EEG focus localization, 1H-MR spectroscopy indicated concordant hemispheric lateralization to EEG findings in 82% (On-line Table 5) compared with 71% in the nonlesional subgroup with unilateral EEG findings (On-line Table 6). Summarized concordance of 82% (18/22) in the MR imaging-positive subgroup (n = 22) resulted from 13 ipsilateral to EEG focus and 5 bilaterally altered 1H-MR spectroscopy metabolite spectra. Bilateral findings were assigned to ipsilateral findings with additive contralateral pathologic findings, reflecting a widespread pathologic condition in this case. In the lesional TLE subgroup contralateral affection in 1H-MR spectroscopy was obvious in 28%, resulting from 5% contralateral and 23% bitemporal metabolite alterations. SPECT lateralized the affected hemisphere in 84% in the case of MR imaging-positive findings (On-line Table 5) versus 67% in MR imaging-negative patients with TLE (On-line Tables 5 and 6). Comparable with 1H-MR spectroscopy, SPECT demonstrated pathologic findings contralateral to EEG in 11% (+16% bitemporal = 27%). By combining both modalities (1H-MRS or SPECT concordant to EEG), a sensitivity of 100% was accomplished.
Negative MR Imaging.
In nonlesional patients (MR imaging-negative) with unilateral EEG discharges (n = 14), an overall sensitivity of 1H-MR spectroscopy of 71% was calculated, resulting from pathologic findings ipsilateral to EEG focus in 8 patients and bitemporal findings in 2 patients (Online Table 6). Hemispheric lateralization by SPECT revealed in 67% concordant lateralization compared with EEG findings, resulting from 5 ipsilateral and 3 bitemporal findings. Combination of both modalities, in which either 1H-MR spectroscopy or SPECT showed concordant findings to EEG, indicated correct lateralization in 100%. Pathologic 1H-MR spectroscopy spectra contralateral to EEG focus in MR imaging-negative patients with TLE was found in 28%, resulting from 14% contralateral and 14% bitemporal metabolite alterations. SPECT demonstrated pathologic findings contralateral to EEG in 33%.
Bilateral EEG
Negative MR Imaging.
In nonlesional patients with bilateral EEG discharges (n = 10), 1H-MR spectroscopy demonstrated a summarized sensitivity of 70%, resulting from 5 bitemporal and 2 metabolite changes, respectively, in right and left hemisphere (On-line Table 7). Pathologic findings by SPECT were revealed in 67%. The combination of both modalities increased sensitivity to 100%.
Positive MR Imaging.
In MR imaging-positive patients (lesional group) with bilateral EEG discharges, sensitivity was not calculated because of a reduced number of patients (n = 3). Our data indicate that in MR imaging-positive unilateral patients with TLE, SPECT has a higher recall factor (68%) compared with 1H-MR spectroscopy (59%) in lateralizing the affected hemisphere. In unilateral patients who remained without pathologic findings in imaging, 1H-MR spectroscopy correctly classified the affected hemisphere in 57%, whereas SPECT demonstrated a recall factor of 42%. Concordance rate of both modalities in detecting the affected hemisphere in unilateral TLE was comparable in MR imaging-positive (74%) and MR imaging-negative patients with TLE (67%). Patients who demonstrated unilateral and concordant findings of EEG, SPECT, 1H-MR spectroscopy, and MR imaging to the operated hemisphere had a clear tendency for a better postoperative outcome after 6 months than patients demonstrating contralateral or bitemporal findings in SPECT or 1H-MR spectroscopy to the operated side [U test by Mann and Whitney (P = 0.08)]. Five of 7 patients classified as unilateral were graded class 1 outcome, whereas 2 remaining patients were classified 2 in Engel outcome classification. In 3 patients with unilateral EEG and bitemporal or contralateral findings in SPECT or 1H-MR spectroscopy to the operated hemisphere, only one patient was seizure free, whereas 2 patients were classified 2. Further details of individual results of different modalities of each patient are displayed in On-line Tables 1–4.
Discussion
Clear focus localization, especially in temporomesial structures of patients with TLE, is still a difficult task in which extensive video EEG monitoring is successful in 60%–90% of cases.13,14,21,22 Hippocampal sclerosis is the most common finding and can be verified with high reliability (up to 90% sensitivity and 85% specificity).8,23,24 However, even using state-of-the-art imaging protocols, MR imaging remains without pathologic findings in up to 30%.25 Although intensive 24-hour video EEG monitoring is the “gold standard” for epileptic focus localization, multimodal approaches are coming to the fore because hemispheric lateralization is verified in case of concordance of different imaging modalities.1,26–28 So far, no single technique appears clearly superior overall to any of the others. Combination of localizing tests supporting clinical and scalp EEG findings should allow more patients to skip, for example, invasive intracranial EEG.
Our results correspond with published data in which correct proton spectroscopic focus lateralization was carried out in 60%–86% of patients with TLE.2,29,30 We found evidence for involvement of contralateral hemispheres in MR imaging-negative unilateral patients with TLE in 28%. These results correspond to the findings of Woermann et al,31 who found contralateral reduced NAA levels in 3 of 15 (20%) patients with evidence for hippocampal sclerosis in MR imaging. In MR imaging-negative patients, he reported 4 of 15 with decreases in hippocampal NAA levels ipsilateral to the seizure focus and 1 with low NAA bilaterally. Cendes et al,29 who investigated 100 consecutive patients with intractable TLE, including MR imaging-positive and -negative patients, reported a correct lateralization in 86% using 1H-MR spectroscopy alone, whereas bilateral affection was reported in 54%.
Most previous in vivo 1H-MR spectroscopy studies were performed at 1.5T, whereas only some used higher field strength.28,32 The increased SNR at a high-field of 3T or above allows shorter imaging times, higher resolution, and therefore promises more precise metabolite quantification. Thus, it might be possible to find more specific metabolic profiles (glutamate, mIns, and γ-aminobutyric acid) to localize or lateralize the epileptogenic zone.28 The overall sensitivity of the structural imaging may be increased, possibly rendering positive some previous MR imaging-negative patients. Quantitative comparison of hippocampal metabolite concentrations such as tNAA, Cre, Cho, Glu, mIns, at 1.5T and 3T, obtained from the same patient, should be addressed to verify existing data. Further studies are required to understand whether possible differences may be accounted for by either repositioning effects or circadian physiologic effects.22
In our study, either ictal or interictal findings in SPECT were successful in 84% regarding correct lateralization of the affected hemisphere in unilateral MR imaging-positive patients with TLE (versus 82% in 1H-MR spectroscopy). In nonlesional MR imaging patients with unilateral EEG discharges correct SPECT focus lateralization was achieved in 67%. In the same group, focus lateralization was achieved in 71% by 1H-MR spectroscopy.
The role of SPECT in diagnosis of TLE is more complex than in other imaging modalities. Data of published studies vary concerning reliability and sensitivity in lateralizing the affected hemisphere from 11%–80%, probably based on different sensitivity of interictal and ictal SPECT and different reliability in mesial and neocortical lateral patients with TLE.1,3,8 A detailed overview is given by Duncan,3 who reports that interictal SPECT is inferior to ictal SPECT in visualization of the affected hemisphere in both mesial and lateral TLE. A blinded comparative study mentioned in the survey demonstrated that interictal SPECT was less effective in lateralizing the affected hemisphere in patients with TLE (45% of cases) compared with MR imaging (86% of cases).33 In patients with complex partial seizures, ictal SPECT studies identified the epileptic focus in 69%–97% of cases.24,34–36 Because of the complexity of rapidly evolving seizures, reliable interpretation of rCBF changes depicted in SPECT scans can be difficult, may appear subtle, and can be subject to reviewer bias. A significant improvement regarding SPECT imaging is the image digital subtraction of an interictal scan from an ictal scan and then coregistration to MR anatomic images. With this technique, it is possible to compare the patient's individual baseline patterns of rCBF and demonstrate anatomic details for precise localization.1,37,38 In our study, this technique was used within the limits of available ictal SPECT data (Fig 3). Interictal SPECT coregistration with MR imaging for precise focus localization was carried out (Fig 2).
The results reveal that 1H-MR spectroscopy and SPECT are more successful in lateralizing the affected hemisphere in MR imaging-positive patients than in patients with TLE who remain without pathologic findings in MR imaging. A hypothesis for this phenomenon could be based on the assumption that pathologic conditions in the initial stages do not display lesional findings in MR imaging because of subtle tissue alterations.12 In further progress, disease alterations become more pronounced, visible in MR imaging as lesions, and easier to detect in 1H-MR spectroscopy and SPECT. In this case, high-field MR imaging of 3T or above might be able to detect more subtle pathologic tissue. Another approach could be based on the assumption that different results in imaging modalities are based on different illness entities affecting hippocampal structures. Recently published results supported this approach addressing metabolite profile of glutamate in the hippocampi in MR imaging-negative patients.31,39 Petroff et al40 reported increased glutamate (Glx), an excitatory epileptogenic transmitter, in the human hippocampus ipsilateral to the EEG focus in MR imaging-negative patients with TLE. Patients with hippocampal sclerosis demonstrated relatively low Glx in hippocampal structures in this study. In patients with TLE with lesional findings (in most cases hippocampal sclerosis), NAA, which is considered a neuronal marker, was decreased ipsilateral to lesion and EEG focus.28,31,39
Several studies have addressed the value of PET in TLE.41–43 Sensitivity for detecting relative temporal lobe hypometabolism with fluorodeoxyglucose-PET in TLE ranges from 80% to 90%.1,26 Unfortunately, PET was unavailable for most patients (partial data not included).
Another interesting and, for clinical practice, important aspect is detection of bilateral pathologic conditions. In the case of bilateral affection, multiple imaging approaches seem to be more reliable in detecting widespread pathology in both hemispheres than single MR imaging, in which bilateral affection is less evident. Our study and previously published data show that 1H-MR spectroscopy and SPECT are able to demonstrate bilateral changes, not only in the patient group already classified as bilateral by EEG but also in patients with TLE classified as unilateral by EEG monitoring. Detection of widespread pathologic findings involving both hemispheres is important for predicting postoperative outcome in patients with TLE who remain refractory to conservative treatment. Pathologic findings in multimodal imaging that are located contralaterally to EEG focus or affect both hemispheres are considered predictors for a bad postoperative outcome in patients with unilateral TLE.2–5,44,45 Factors contributing to contralateral damage are still unknown and pathogeneses of bilateral damage are still unclear. Bitemporal involvement is found in both adults and children.46,47 It has been postulated that these abnormalities appear more pronounced in patients with right TLE, assuming that the right lobe seems to act more bilaterally than the left in terms of either recruitment of contralateral resources or affecting contralateral structures.47 It is noteworthy that our results give further evidence addressing this matter because 50% of patients with unilateral EEG discharges and lesional findings in MR imaging in the right temporal lobe demonstrated bitemporal metabolite alterations in 1H-MR spectroscopy.
Conclusion
The main advantage of multimodal imaging in patients with TLE is the increased clarification in verifying the affected hemisphere, especially in patients without pathologic findings in MR imaging. Even though no single technique appears clearly superior overall to any of the others, with a multimodal approach, we can reduce unnecessary invasive diagnostic steps and distinguish patients who will benefit from surgery. Considerable advantages exist in displaying bilateral affection in patients with TLE, especially for calculating the relative predictive value (with respect to surgical localization and outcome), influencing further treatment schedules. Therefore, how each technique can be optimized both economically and for individual benefit must be determined.
Footnotes
Indicates article with supplemental online tables
References
- 1.Knowlton RC. The role of FDG-PET, ictal SPECT, and MEG in the epilepsy surgery evaluation. Epilepsy Behav 2006;8:91–101 [DOI] [PubMed] [Google Scholar]
- 2.Knowlton RC, Laxer KD, Ende G, et al. Presurgical multimodality neuroimaging in electroencephalographic lateralized temporal lobe epilepsy. Ann Neurol 1997;42:829–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Duncan JS. Imaging and epilepsy. Brain 1997;120:339–77 [DOI] [PubMed] [Google Scholar]
- 4.Berkovic SF, McIntosh AM, Kalnins RM, et al. Preoperative MRI predicts outcome of temporal lobectomy: an actuarial analysis. Neurology 1995;45:1358–63 [DOI] [PubMed] [Google Scholar]
- 5.Jack CR Jr. Epilepsy: surgery and imaging. Radiology 1993;189:635–46 [DOI] [PubMed] [Google Scholar]
- 6.Stefan H, Feichtinger M, Pauli E, et al. Magnetic resonance spectroscopy and histopathological findings in temporal lobe epilepsy. Epilepsia 2001;42:41–46 [DOI] [PubMed] [Google Scholar]
- 7.Yun CH, Lee SK, Lee SY, et al. Prognostic factors in neocortical epilepsy surgery: multivariate analysis. Epilepsia 2006;47:574–79 [DOI] [PubMed] [Google Scholar]
- 8.Park SW, Chang KH, Kim HD, et al. Lateralizing ability of single-voxel proton MR spectroscopy in hippocampal sclerosis: comparison with MR imaging and positron emission tomography. AJNR Am J Neuroradiol 2001;22:625–31 [PMC free article] [PubMed] [Google Scholar]
- 9.Achten E, Santens P, Boon P, et al. Single-voxel proton MR spectroscopy and positron emission tomography for lateralization of refractory temporal lobe epilepsy. AJNR Am J Neuroradiol 1998;19:1–8 [PMC free article] [PubMed] [Google Scholar]
- 10.Gaillard WD, Bhatia S, Bookheimer SY, et al. FDG-PET and volumetric MRI in the evaluation of patients with partial epilepsy. Neurology 1995;45:123–26 [DOI] [PubMed] [Google Scholar]
- 11.Van PW, Dupont P, Van DG, et al. SPECT perfusion changes during complex partial seizures in patients with hippocampal sclerosis. Brain 2003;126:1103–11 [DOI] [PubMed] [Google Scholar]
- 12.Hammen T, Stefan H, Eberhardt KE, et al. Clinical applications of 1H-MR spectroscopy in the evaluation of epilepsies–what do pathological spectra stand for with regard to current results and what answers do they give to common clinical questions concerning the treatment of epilepsies? Acta Neurol Scand 2003;108:223–38 [DOI] [PubMed] [Google Scholar]
- 13.Cendes F, Li LM, Watson C, et al. Is ictal recording mandatory in temporal lobe epilepsy? Not when the interictal electroencephalogram and hippocampal atrophy coincide. Arch Neurol 2000;57:497–500 [DOI] [PubMed] [Google Scholar]
- 14.Stefan H, Pawlik G, Bocher-Schwarz HG, et al. Functional and morphological abnormalities in temporal lobe epilepsy: a comparison of interictal and ictal EEG, CT, MRI, SPECT and PET. J Neurol 1987;234:377–84 [DOI] [PubMed] [Google Scholar]
- 15.Engel J Jr, Van Ness PC, Rasmussen T, et al. Outcome with respect to epileptic seizures. In: Engel J Jr, ed. Surgical Treatment of the Epilepsies, 2nd ed. New York: Raven Press;1993. :609–22
- 16.Stefan H, Wang Y, Pauli E, et al. A new approach in anti-epileptic drug evaluation. Eur J Neurol 2004;11:467–73 [DOI] [PubMed] [Google Scholar]
- 17.Hammen T, Stadlbauer A, Tomandl B, et al. Short TE single-voxel 1H-MR spectroscopy of hippocampal structures in healthy adults at 1.5 Tesla–how reproducible are the results? NMR Biomed 2005;18:195–201 [DOI] [PubMed] [Google Scholar]
- 18.Provencher SW. Estimation of metabolite concentrations from localized in vivo proton NMR spectra. Magn Reson Med 1993;30:672–79 [DOI] [PubMed] [Google Scholar]
- 19.Provencher SW. Automatic quantitation of localized in vivo 1H spectra with LCModel. NMR Biomed 2001;14:260–64 [DOI] [PubMed] [Google Scholar]
- 20.Helms G. A precise and user-independent quantification technique for regional comparison of single volume proton MR spectroscopy of the human brain. NMR Biomed 2000;13:398–406 [DOI] [PubMed] [Google Scholar]
- 21.Pataraia E, Lurger S, Serles W, et al. Ictal scalp EEG in unilateral mesial temporal lobe epilepsy. Epilepsia 1998;39:608–14 [DOI] [PubMed] [Google Scholar]
- 22.Soreni N, Noseworthy MD, Cormier T, et al. Intraindividual variability of striatal 1H-MRS brain metabolite measurements at 3 T. Magn Reson Imaging 2006;24:187–94 [DOI] [PubMed] [Google Scholar]
- 23.Duc CO, Trabesinger AH, Weber OM, et al. Quantitative 1H MRS in the evaluation of mesial temporal lobe epilepsy in vivo. Magn Reson Imaging 1998;16:969–79 [DOI] [PubMed] [Google Scholar]
- 24.Lee DH, Gao FQ, Rogers JM, et al. MR in temporal lobe epilepsy: analysis with pathologic confirmation. AJNR Am J Neuroradiol 1998;19:19–27 [PMC free article] [PubMed] [Google Scholar]
- 25.Van PW, Connelly A, Johnson CL, et al. The amygdala and intractable temporal lobe epilepsy: a quantitative magnetic resonance imaging study. Neurology 1996;47:1021–31 [DOI] [PubMed] [Google Scholar]
- 26.Ryvlin P, Philippon B, Cinotti L, et al. Functional neuroimaging strategy in temporal lobe epilepsy: a comparative study of 18FDG-PET and 99mTc-HMPAO-SPECT. Ann Neurol 1992;31:650–56 [DOI] [PubMed] [Google Scholar]
- 27.Swartz BE, Brown C, Mandelkern MA, et al. The use of 2-deoxy-2-[18F]fluoro-d-glucose (FDG-PET) positron emission tomography in the routine diagnosis of epilepsy. Mol Imaging Biol 2002;4:245–52 [DOI] [PubMed] [Google Scholar]
- 28.Riederer F, Bittsansky M, Schmidt C, et al. 1H magnetic resonance spectroscopy at 3 T in cryptogenic and mesial temporal lobe epilepsy. NMR Biomed 2006 [DOI] [PubMed]
- 29.Cendes F, Andermann F, Dubeau F, et al. Normalization of neuronal metabolic dysfunction after surgery for temporal lobe epilepsy. Evidence from proton MR spectroscopic imaging. Neurology 1997;49:1525–33 [DOI] [PubMed] [Google Scholar]
- 30.Kuzniecky R, Hugg JW, Hetherington H, et al. Relative utility of 1H spectroscopic imaging and hippocampal volumetry in the lateralization of mesial temporal lobe epilepsy. Neurology 1998;51:66–71 [DOI] [PubMed] [Google Scholar]
- 31.Woermann FG, McLean MA, Bartlett PA, et al. Short echo time single-voxel 1H magnetic resonance spectroscopy in magnetic resonance imaging-negative temporal lobe epilepsy: different biochemical profile compared with hippocampal sclerosis. Ann Neurol 1999;45:369–76 [DOI] [PubMed] [Google Scholar]
- 32.Petroff OA, Rothman DL, Behar KL, et al. Effects of valproate and other antiepileptic drugs on brain glutamate, glutamine, and GABA in patients with refractory complex partial seizures. Seizure 1999;8:120–27 [DOI] [PubMed] [Google Scholar]
- 33.Jack CR, Jr., Mullan BP, Sharbrough FW, et al. Intractable nonlesional epilepsy of temporal lobe origin: lateralization by interictal SPECT versus MRI. Neurology 1994;44:829–36 [DOI] [PubMed] [Google Scholar]
- 34.Stefan H, Bauer J, Feistel H, et al. Regional cerebral blood flow during focal seizures of temporal and frontocentral onset. Ann Neurol 1990;27:162–66 [DOI] [PubMed] [Google Scholar]
- 35.Shen W, Lee BI, Park HM, et al. HIPDM-SPECT brain imaging in the presurgical evaluation of patients with intractable seizures. J Nucl Med 1990;31:1280–84 [PubMed] [Google Scholar]
- 36.Duncan R, Patterson J, Roberts R, et al. Ictal/postictal SPECT in the pre-surgical localisation of complex partial seizures. J Neurol Neurosurg Psychiatry 1993;56:141–48 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zubal IG, Spencer SS, Imam K, et al. Difference images calculated from ictal and interictal technetium-99m-HMPAO SPECT scans of epilepsy. J Nucl Med 1995;36:684–89 [PubMed] [Google Scholar]
- 38.O’Brien TJ, So EL, Mullan BP, et al. Subtraction ictal SPECT co-registered to MRI improves clinical usefulness of SPECT in localizing the surgical seizure focus. Neurology 1998;50:445–54 [DOI] [PubMed] [Google Scholar]
- 39.Savic I, Thomas AM, Ke Y, et al. In vivo measurements of glutamine + glutamate (Glx) and N-acetyl aspartate (NAA) levels in human partial epilepsy. Acta Neurol Scand 2000;102:179–88 [DOI] [PubMed] [Google Scholar]
- 40.Petroff OA, Errante LD, Rothman DL, et al. Glutamate-glutamine cycling in the epileptic human hippocampus. Epilepsia 2002;43:703–10 [DOI] [PubMed] [Google Scholar]
- 41.Benbadis SR, So NK, Antar MA, et al. The value of PET scan (and MRI and Wada test) in patients with bitemporal epileptiform abnormalities. Arch Neurol 1995;52:1062–68 [DOI] [PubMed] [Google Scholar]
- 42.Blum DE, Ehsan T, Dungan D, et al. Bilateral temporal hypometabolism in epilepsy. Epilepsia 1998;39:651–59 [DOI] [PubMed] [Google Scholar]
- 43.Wong CY, Geller EB, Chen EQ, et al. Outcome of temporal lobe epilepsy surgery predicted by statistical parametric PET imaging. J Nucl Med 1996;37:1094–100 [PubMed] [Google Scholar]
- 44.Jack CR, Jr., Sharbrough FW, Cascino GD, et al. Magnetic resonance image-based hippocampal volumetry: correlation with outcome after temporal lobectomy. Ann Neurol 1992;31:138–46 [DOI] [PubMed] [Google Scholar]
- 45.Kim MA, Heo K, Choo MK, et al. Relationship between bilateral temporal hypometabolism and EEG findings for mesial temporal lobe epilepsy: analysis of 18F-FDG PET using SPM. Seizure 2006;15:56–63 [DOI] [PubMed] [Google Scholar]
- 46.Gadian DG, Isaacs EB, Cross JH, et al. Lateralization of brain function in childhood revealed by magnetic resonance spectroscopy. Neurology 1996;46:974–77 [DOI] [PubMed] [Google Scholar]
- 47.Zubler F, Seeck M, Landis T, et al. Contralateral medial temporal lobe damage in right but not left temporal lobe epilepsy: a 1H magnetic resonance spectroscopy study. J Neurol Neurosurg Psychiatry 2003;74:1240–44 [DOI] [PMC free article] [PubMed] [Google Scholar]