Abstract
Introduction
The Amyloid Imaging to Prevent Alzheimer's Disease (AMYPAD) Prognostic and Natural History Study (PNHS) aims at understanding the role of amyloid imaging in the earliest stages of Alzheimer's disease (AD). AMYPAD PNHS adds (semi‐)quantitative amyloid PET imaging to several European parent cohorts (PCs) to predict AD‐related progression as well as address methodological challenges in amyloid PET.
Methods
AMYPAD PNHS is an open‐label, prospective, multi‐center, cohort study recruiting from multiple PCs. Around 2000 participants will undergo baseline amyloid positron emission tomography (PET), half of whom will be invited for a follow‐up PET 12 at least 12 months later.
Results
Primary include several amyloid PET measurements (Centiloid, SUVr, BPND, R1), and secondary are their changes from baseline, relationship to other amyloid markers (cerebrospinal fluid and visual assessment), and predictive value of AD‐related decline.
Expected Impact
Determining the role of amyloid PET for the understanding of this complex disease and potentially improving secondary prevention trials.
Keywords: amyloid, positron emission tomography imaging, preclinical and prodromal Alzheimer's disease, secondary prevention
1. BACKGROUND
Recent years have seen massive effort into improving the understanding of the pathogenesis and trajectory of Alzheimer's disease (AD) in its preclinical stages. Currently, AD dementia is conceptualized as the endpoint of a cascade of events leading to cognitive impairment, with the accumulation of amyloid beta (Aβ) plaques being considered the first detectable change in the brain—a process that continues for decades prior to the onset of cognitive decline. 1 Although no causal link between Aβ plaque deposition and subsequent dementia has been established, it remains a pathological hallmark in the pathway toward AD dementia. As such, its measurement has been part of many observational studies in aging and dementia, and it has also been used in clinical trials both as inclusion criteria and as part of the outcome measures in the case of anti‐amyloid interventions.
Although no disease‐modifying therapy has been approved yet, promising signals have been observed in a number of anti‐amyloid clinical trials, in which small to modest reductions in brain amyloid levels were achieved in treatment groups, albeit not accompanied by effects on cognitive decline. 2 , 3 , 4 , 5 , 6 These trials have maintained hope in the search for a successful therapy, but also identified challenges in trial design. These include potentially starting interventions too late in the disease course, suboptimal inclusion criteria, insufficient drug dosage, and incorrect treatment target, also questioning the validity of the widely accepted amyloid cascade hypothesis. 7 Nonetheless, recent studies in cognitively normal subjects suggest that the amyloid hypothesis might still hold at stages at which Aβ is driving the cascade, that is, before widespread and irreversible neuronal damage is present. 8 , 9 , 10 , 11
Considering its placement in the cascade of events and the recent focus on the earlier stages of AD, accurately determining the presence of Aβ pathology and understanding its role in the development of dementia is relevant not only for etiological diagnosis, but also for monitoring disease progression, and in trials of secondary prevention. Currently, the presence of Aβ pathology can be determined either by cerebrospinal fluid (CSF), positron emission tomography (PET) imaging, or recently also by blood samples. 12 , 13 While prescribed for the same goal, a non‐negligible proportion of patients can present with discordant results between CSF and PET. 14 CSF is considered to indicate the presence of amyloid pathology in its earliest stages, but it plateaus relatively early in the AD disease course, while amyloid PET imaging shows continued build‐up of amyloid burden beyond the CSF plateau 15 and it provides information on the spatial distribution of the pathology in the brain.
Despite such promising characteristics, amyloid PET imaging may not have been used to its full potential previously, and there are a number of knowledge gaps that have not yet been fully addressed in the early stages of AD. For example, recent developments in AD research indicate visual assessment and/or global semi‐quantitative measures such as the Standard Uptake Value ratio (SUVR) might not be optimally suited for tracking disease progression and for some clinical trial applications. 16 , 17 More specifically, global SUVR has been shown to detect the presence of Aβ plaques at a generally later stage of pathology, indicating it might not be appropriate for capturing amyloidosis in the preclinical stages at which pathology might still be restricted to specific brain regions. 18 , 19 Visual assessment might in turn be able to capture focal signal, but it lacks the quantitative aspect necessary for accurately measuring longitudinal change and treatment effects. Further, both metrics are less specific than quantitative measurements of plaque density available from dynamic PET scans and potentially suffer from bias such as those introduced by changes in blood flow. 17 , 20 Therefore, determining the optimal methodology for amyloid PET analysis becomes increasingly relevant moving forward to understand disease progression and support both current and future AD trials. 21 , 22
The Amyloid Imaging to Prevent Alzheimer's Disease (AMYPAD) Prognostic and Natural History Study (PNHS) has been established to collect amyloid PET scans in a large‐scale population in the early stages of AD. This collection will allow the assessment of the true potential of amyloid PET imaging as a tool for visualizing and quantifying both the natural disease progression and eventual effects of anti‐amyloid interventions. To efficiently achieve its goals, this study was established within an intrinsically collaborative framework, providing European cohorts of preclinical and prodromal individuals with access to amyloid PET imaging to complement their efforts in creating well‐phenotyped cohorts. This collaborative effort allows us to complement risk prediction models and enables future disease‐modifying trials by providing relevant insight into the optimal use of amyloid PET imaging in an early population at a large scale. 23
2. METHODS
2.1. The Amyloid Imaging to Prevent Alzheimer's Disease (AMYPAD) Consortium
The AMYPAD Consortium was initiated in October 2016 as a key component of the Innovative Medicine Initiative (IMI)‐Alzheimer's Disease platform, 24 and receives the support from the European Union's Horizon 2020 research and innovation program and from the European Federation of Pharmaceutical Industries and Associations (EFPIA). Partners include academia, EFPIA, and small and medium‐sized enterprises (SMEs) such as IXICO and SYNAPSE.
RESEARCH IN CONTEXT
Systematic review: Recent Alzheimer's disease (AD) secondary prevention trials highlight the need for understanding the disease pathogenesis and trajectory. Although many studies use positron emission tomography (PET) to assess amyloid pathology, the technique's potential in obtaining quantitative and topographical assessment is generally not yet fully explored in research and clinical trials. The Amyloid Imaging to Prevent Alzheimer's Disease Prognostic and Natural History Study (AMYPAD PNHS) is designed to determine amyloid PET imaging's role in assessing AD risk in pre‐dementia subjects.
Interpretation: Amyloid PET scans will be performed at a large scale in collaboration with several well‐characterized longitudinal European cohorts to determine the added value of amyloid PET imaging to determine AD risk in a pre‐dementia population, over and above widely available AD biomarkers.
Future directions: The knowledge gained within AMYPAD PNHS can be used to determine the value and best practices in amyloid PET imaging both in research and secondary prevention trials, improving inclusion criteria and measurements of treatment effects.
One of two clinical trials from the Consortium, the AMYPAD PNHS is sponsored by the UMC Amsterdam location VUmc (VUmc) and the scientific leadership is shared between the Barcelona β Brain Research Center (BBRC) and Janssen Pharmaceutica. In addition, partner sites include the University of Edinburgh, University of Geneva, Karolinska Institutet Stockholm, and the Central Hospital University of Toulouse, and several non‐partner sites are also engaged.
2.2. Parent cohorts
AMYPAD PNHS recruits from various parent cohorts (PCs) of similar characteristics. The PCs are all European, actively enrolling or performing follow‐up visits, and focused on pre‐dementia subjects. In addition, information on cognitive functioning (eg, clinical diagnosis or neuropsychological assessments), disease biomarkers (eg, CSF or magnetic resonance imaging [MRI] measures), and traditional risk factors (eg, genetic and environmental) is available within each PC. These characteristics are essential, as the study is set to efficiently complement ongoing efforts from all individual PCs by providing access to amyloid PET as an early biomarker, while being granted access to additional variables to model cross‐biomarker relationships and their effects on disease progression.
The main and original PC is European Prevention of Alzheimer's Dementia (EPAD) due to the inherent collaborative framework between the two IMI projects. 23 As of October 2019, two additional PCs have been formally included in AMYPAD PNHS. These are the PreclinAD EMIF‐AD cohort in Amsterdam 25 and a subset of the ALFA+ cohort in Barcelona. 26 As the inclusion of PCs is an ongoing and dynamic process and additional cohorts are still under consideration, an up‐to‐date overview of collaborating PCs and their details will be made available on the AMYPAD website https://amypad.eu/data.
2.3. Target population
AMYPAD PNHS aims to recruit up to 2000 subjects, who must be coming from one of the selected PCs, aged 50 years or older, and not demented. In addition, the inclusion and exclusion criteria of a particular PC indirectly apply to PNHS, and the complete information for these will also be available on the website.
Recruitment is aimed at subjects at the beginning of the amyloid cascade, where emerging pathology is traditionally undetected by established standards. More specifically, the goal is to enroll not only amyloid‐positive participants, but also those with negative amyloid PET scans, as well as those in a so‐called gray zone of amyloid burden (eg, participants with ambiguous or discordant CSF and PET amyloid status or on the verge of CSF or PET biomarker abnormality). The cohort balance will be constantly monitored and managed, dynamically adapting participant selection criteria when needed.
2.4. Participant selection and the adaptive algorithm approach
To ensure enrollment of the target population, the process of selecting eligible participants from PCs makes use of an innovative and adaptive approach that considers multiple dimensions to better identify the target population, avoiding simple single‐biomarker cut‐offs. This is made possible due to the access AMYPAD PNHS has to information on individual risk profiles available from PCs. Whenever available, previous information on amyloid status (either from an earlier amyloid PET scan or from CSF measurements) will be the main determinant for inclusion. In addition, a combination of age, family history, genetic profile, and other well‐established risk factors will also be considered.
The selection algorithms, customized for each PC, are determined and applied by an established selection and feasibility committee (SFC), with representatives from the project leadership independent from site activities. This ensures blinding to traceable individual participant information and limits potential bias across all studies.
2.5. Study timelines and design
The AMYPAD PNHS is a prospective, multi‐center, pan‐European, longitudinal cohort study linked to several ongoing European PCs. Participants recruited into PNHS will continue their participation in their respective PC and undergo amyloid PET imaging in addition to the procedures of the parent study. Recruitment started in late 2018 and is expected to continue until late 2021.
Participants can enter the AMYPAD PNHS at any point in their participation in their PC, and the alignment between AMYPAD and PC visits is strongly encouraged for efficiency and temporal correspondence between biomarker assessments (Figure 1). Once part of AMYPAD PNHS, participants can undergo either a standard static PET imaging session or a dual‐window quantitative dynamic PET imaging acquisition protocol. 27
FIGURE 1.
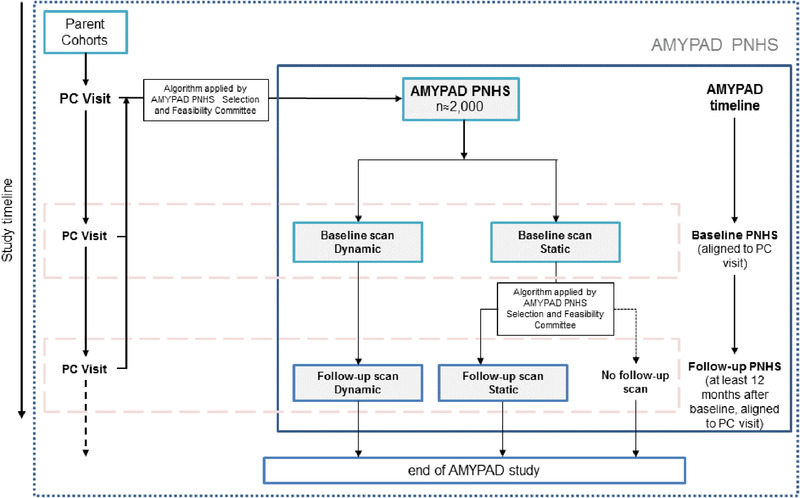
Schematic representation of Amyloid Imaging to Prevent Alzheimer's Disease (AMYPAD) Prognostic and Natural History Study (PNHS) design and its relationship with ongoing parent cohorts
After at least 12 months, ≈50% of participants will be invited for a follow‐up scan, to be performed using the same acquisition protocol as baseline. All participants undergoing dynamic baseline PET will be invited for follow‐up, while results from baseline static PET scans will be provided to the SFC so that a follow‐up algorithm can select the right participants for follow‐up scanning. As some PCs have performed amyloid PET prior to this study, these historical data sets will be made available to AMYPAD, enabling direct longitudinal assessment of amyloid accumulation.
2.6. Amyloid PET imaging in AMYPAD PNHS
Two radiotracers are available in this study, namely [18F]florbetaben (NeuraCeq®) and [18F]flutemetamol (Vizamyl®) (Figure 2), with both imaging agents aimed to be equally represented across the study and acquisition protocols. Taking advantage of the available expertise within participating centers, AMYPAD aims to perform dynamic acquisitions for as many scans as possible.
FIGURE 2.
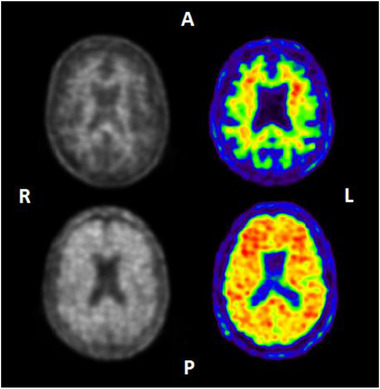
Example images of Neuraceq (left) and Vizamyl (right). Upper images: visually negative amyloid positron emission tomography (PET) scans showing only tracer retention in white matter. Lower images: visually positive amyloid PET scans showing tracer retention also in cortical areas. A, anterior; L, left hemisphere; P, posterior; R, right hemisphere
2.6.1. Image acquisition
Static scans are acquired according to the standard protocol for each tracer, starting at 90 minutes post‐injection (p.i.) and collected in four frames of 5 minutes each (90 to 110 minutes p.i.). 28 , 29 In contrast, dynamic scans are acquired in two parts; 27 first, a 30‐minute scan is started at the time of tracer injection (0 to 30 minutes p.i.), then subjects can rest outside the camera for 60 minutes, and finally, the standard static acquisition is performed for 20 minutes starting at 90 minutes p.i. (90 to 110 minutes p.i.). This protocol allows for interleaved scanning, which translates to efficient scanner use and reduced participant burden (Figure 3). For quantification purposes, the closest 3D T1‐weighed (3D T1w) MR scan available from the PCs will be used for anatomical delineation.
FIGURE 3.
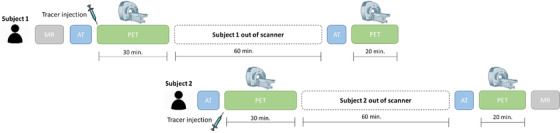
Example of interleaved dynamic dual‐time window acquisition protocol. The dual‐time window protocol allows for interleaved acquisition, in which the scan of a second subject can be initiated during the break of the first subject
2.6.2. Image analysis
(Semi‐)Quantitative analyses will be performed centrally, with the pre‐processing being performed by IXICO, together with the quantification of the static scans. The additional pharmacokinetic modeling for quantification of the dynamic scans will be performed by VUmc.
IXICO's pre‐processing pipeline uses a subject‐specific multi‐atlas structural segmentation approach (LEAP). 30 First, the four 5‐minute PET frames are co‐registered and averaged. Then, the MR is segmented using this LEAP approach and, together with the resulting anatomical regions of interest (ROIs), it is finally co‐registered to the PET. This allows flexible selection of multiple single and/or composite regions for the (semi‐)quantitative analyses. In addition, PET images will also be warped into Montreal Neurological Institute (MNI) space to enable exploratory voxel‐wise comparisons.
Once the pre‐processing is performed, IXICO's SUVr and Centiloid quantification is performed, as the pipeline has been validated for the computation of Centiloid values to allow for pooled analyses across tracers. 31 Next to IXICO's quantification and for those subjects for whom dynamic scans are available, pre‐processed images and corresponding ROIs are transferred to VUmc to undergo the quantification of non‐displaceable binding potential (BPND) values. For that purpose, both the receptor parametric mapping (RPM) method 32 and the reference model of Logan Graphical Analysis will be applied. 33
For all analyses, four commonly used reference regions (RR) will be used in this project; namely cerebellar gray matter, whole cerebellum, pons, and subcortical white matter. The whole cerebellum will be the RR used for the primary and secondary outcomes of the study because it is the standard Centiloid RR. All other regions will be explored as alternatives, and their suitability will be assessed with respect to SUV stability across individuals, clinical groups, and longitudinally. Further, comparing these RRs will allow the determination of their specific statistical power to detect cross‐sectional differences in (semi‐)quantitative metrics and/or statistically significant and plausible longitudinal changes. Therefore, whenever applicable, results with an alternative and well‐suited RR will be reported in addition to the primary outcomes.
In addition to the (semi‐)quantitative analyses, visual interpretation of PET images will be performed locally using a standardized and blinded method and following the tracer‐specific reader training provided by Life Molecular Imaging (for [18F]florbetaben/NeuraCeq®) and GE Healthcare (for [18F]flutemetamol/Vizamyl®). The standard positive/negative assessment will be complemented with regional visual classification, and the corresponding reader's confidence (1 to 5) will be recorded.
3. OBJECTIVES
The primary objective of the study is to (semi‐)quantitatively assess amyloid burden using PET to complement the extensive assessments of PCs and enable the achievement of secondary objectives. This is aligned with the fact that AMYPAD PNHS only collects amyloid PET imaging data, relying on the data available from the PCs to model any other dimension of a so‐called AD risk probability spectrum. This concept aims to assess an individual's risk of progression to AD dementia using different dimensions such as cognition, disease biomarkers, and established genetic and environmental risk factors. 23
In addition, three groups of secondary objectives are defined. The first relates to the subset of dynamic and longitudinal amyloid PET imaging data collected in the study, as well as to the main outcomes of PCs. Secondary objectives in this group include to quantify (1) relative cerebral blood flow available from dynamic PET images and (2) amyloid plaque accumulation available from longitudinal data. These will then be used alone and in combination to predict (1) decline in global measures of cognitive functioning as well as within specific cognitive domains, and (2) structural and functional brain changes.
The second group was defined to compare techniques for assessing amyloid status in their role and ability to predict cognitive decline. First, the BPND will be used to assess the validity and possible bias within the SUVR measurement in those subjects who have dynamic scans. Next, the relationship between traditional global amyloid PET measures (Centiloid, SUVR, and BPND), regional level data and available systems for staging amyloid burden 34 , 35 , 36 will be assessed. Finally, data combined from AMYPAD PNHS and PCs will be used to determine the cross‐sectional and longitudinal agreement between brain amyloid status classification methods (PET quantification and visual assessment, as well as CSF). As a result, the optimal methodology to obtain (cross‐sectional and longitudinal) (semi‐)quantitative measures of amyloid burden that robustly predict cognitive decline will be determined.
The final group will assess the relationship between amyloid burden and other AD risk factors, as well as their relation to subsequent cognitive decline, potentially informing future interventions targeting such risk factors.
4. OUTCOME MEASURES
The primary outcome measures in AMYPAD PNHS are (semi‐)quantitative measurements of amyloid burden, namely the SUVR, the BPND when applicable (for dynamic acquisitions only), and corresponding Centiloid values 37 due to the use of different tracers. In addition, SUV and R1 (a proxy for cerebral blood flow available from dynamic PET imaging 38 ) will also be estimated for all regions.
Secondary outcome measures include non‐amyloid PET variables already available from the PCs, including measurements of cognitive status, basic and advanced MR sequences, CSF, and risk factors. Because the measures for global cognition may differ per PC, multiple established and exploratory measures will be considered, such as the Mini Mental State Examination (MMSE), the Clinical Dementia Rating (CDR) scale, or domain‐specific and global functioning scores determined from individual tests. This exploratory harmonization strategy will leverage the extensive number of distinct neuropsychological tests available from PCs, which always encompass at least all five commonly defined cognitive domains.
In addition, MR‐based secondary outcome measures will include traditional measures of gray‐matter volume and atrophy rates. Whenever available, additional measurements from advanced MR sequences will be assessed. At least from EPAD Longitudinal Cohort Study, measures of white matter integrity, functional and structural connectivity, as well as cerebral perfusion will also be available for a subset of the MR imaging data. Details on the complete set of advanced MR sequences per PC will be available on the AMYPAD website (https://amypad.eu/).
Several CSF measurements will be available for a large subset of the data, including at least Aβ42, Aβ40, t‐tau, and p‐tau. Finally, traditional risk factors such as apolipoprotein E (APOE) ε4 status, family history, hypertension, and diabetes are also collected in most PCs and will be available for AMYPAD PNHS analyses.
5. DATA HARMONIZATION
As a multi‐center study, AMYPAD PNHS must ensure optimal data harmonization in many levels of the data collection process. With respect to imaging, both the PET acquisition protocols and the core MR sequence (3D T1w) are the same across centers, and the scanners are qualified by IXICO prior to study commencement to ensure harmonized data quality. Exceptions to this are scans collected from PCs that entered AMYPAD PNHS with historical scans, in which case the historical protocol is maintained for longitudinal consistency and a posteriori harmonization is performed.
With respect to the remaining measurements, these will be more heterogeneous due to the inclusion of multiple PCs. Especially with respect to cognitive measures, PCs may differ due to distinct goals or experience with specific assessments. Therefore, the harmonization of non‐imaging data will be a major endeavor in AMYPAD PNHS, which will leverage the expertise of external partners and collaborators with previous experience. 39 The final strategy for harmonization of each variable will be made openly available on AMYPAD's website, providing clarity to users and potential support for future initiatives aiming to perform similar analyses. As an initial step, cognitive assessments will be analyzed per cognitive domain and z‐scored to improve comparison across tests. A list of original variables available from PCs is available on the website and will be updated as new PCs join the project.
6. EXPECTED IMPACT
Although amyloid PET imaging holds great promises for better understanding and phenotyping AD at the pre‐dementia phase, the current use of the technique has not led to an effective use of its full potential in AD research and clinical trials. AMYPAD PNHS will collect amyloid PET scans at large scale in a pre‐dementia population, creating the opportunity to address current knowledge gaps and previous challenges. The study is set in a collaborative framework with established European cohorts of well‐phenotyped individuals, which will allow for a detailed characterization of the natural history of AD and its early stages. In addition, AMYPAD PNHS will perform both dynamic and static scans and (semi‐)quantitatively assessing the images beyond current dichotomized methodologies that disregard the topography of the pathology. Therefore, by addressing both previously identified methodological issues and the complex multi‐modal aspects of AD, this study aims to ultimately identify the role of amyloid PET in AD risk assessment.
The large‐scale (semi‐)quantitative amyloid PET data collected in AMYPAD PNHS may be essential for studying amyloid accumulation in early populations, in which the pathological signal is often subtle 40 and large sample sizes may be needed. In particular, the study will obtain results from a head‐to‐head comparison between BPND and SUVR across the complete range of amyloid burden, which will help determine whether and when dynamic scans are required in the context of AD research and preventive trials. 17 This question will be answered not only in terms of power to detect longitudinal changes and robustness against physiological confounders, but also with respect to the additional information available from dynamic PET scans, namely a surrogate of cerebral blood flow R1. 38 Although hypoperfusion is a marker of AD and previous studies have shown strong correlations between R1, [18F]FDG and water‐PET, 41 the value of such information in a pre‐dementia population remains largely unexplored. Such an assessment can help distinguishing different treatment arms in an anti‐amyloid trial, but it might also serve other therapeutic initiatives, as amyloid burden can affect several other processes such as synaptic plasticity, mitochondrial function, localized neuro‐inflammation, and vascular health. 42
Recent efforts also indicate that the use of amyloid PET can be further optimized by assessing the available topographical information present in PET scans, as well as determining longitudinal rates of change. 22 In fact, regional information from amyloid PET has been suggested to improve the detection of amyloid pathology in an asymptomatic population. 11 , 34 Expanding on the concept, a number of amyloid staging models have been recently developed, 34 , 35 , 36 which could represent powerful tools both for tracking natural disease progress and supporting trials. These methods identify pathological signs earlier than conventional dichotomized measures, represent simple tools for monitoring changes in amyloid burden, and have the potential to guide trial arm stratification, effectively reducing the risks of detecting false effects that can be introduced when amyloid burden is not balanced across treatment arms. 16 In this line, AMYPAD PNHS's collection of regional visual assessments, reader confidence, and established and advanced quantification approaches will help determine the true value of such methodologies.
Despite these advances, the large number of failures from anti‐amyloid therapeutic trials have challenged the hypothesis of a causal role for misfolded amyloid proteins in the disease pathogenesis, and as a consequence, the number of trials investigating alternative disease mechanisms and pathways are increasing. 43 , 44 AMYPAD understands that the accumulation of amyloid plaques is a necessary but not sufficient step in the development of Alzheimer's dementia. Thus, the study purposely aligns itself with cohorts that collect extensive assessments in a pre‐dementia population to enable the assessment of a variety of risk factors and biomarker interactions and contribute to the understanding of this complex disease, also beyond amyloid pathology. Nonetheless, the increased availability of larger longitudinal datasets of cognitively unimpaired subjects has already resulted in the compilation of an initial but growing evidence on the role of amyloid burden as a predictor of early cognitive decline, also independently from or corrected for other factors. 9 , 10 , 45 In fact, early accumulation of amyloid plaques in specific regions of initially amyloid‐negative healthy adults has also been linked to subclinical amyloid‐related decline in episodic memory. 11 Although limited, these findings support the notion that the seemingly elusive relationship between amyloid pathology and cognitive impairment could be more pronounced in stages of the disease during which amyloid is driving the cascade, that is, before clinically relevant cognitive impairment is present. AMYPAD PNHS might be particularly equipped to shed light into such a hypothesis, as the heterogeneity in the cognitive assessments available from different cohorts create unique opportunities to explore cognitive functioning in detail, despite the resulting analytical challenges.
Finally, the intrinsic collaboration between AMYPAD PNHS and several parent cohorts results in an efficient recruitment strategy, as the operational teams are able to leverage the available historical information on subject characteristics to ensure a targeted inclusion process. Further, AMYPAD PNHS does not burden de novo participants because it builds on established cohorts and establishes a framework that benefits all parties. While the PCs provide demographic and extensive cognitive and biomarker data, the amyloid PET data collected under AMYPAD's umbrella is returned to each PC, assisting them in reaching their own original objectives.
7. CONCLUSION
AMYPAD PNHS is a large‐scale amyloid PET study established to complement the phenotyping of longitudinal cohorts across Europe to better understand the value of the technique in assessing an individual's risk of AD‐related progression. The project focuses on providing high‐quality (semi‐)quantitative information on amyloid burden while leveraging extensive additional information collected in a number of parent cohorts. The study is set to run until late 2021, after which the data will be made openly available to the scientific community.
FUNDING INFORMATION
The project leading to this paper has received funding from the Innovative Medicines Initiative 2 Joint Undertaking under grant agreement no. 115952. This Joint Undertaking receives the support from the European Union's Horizon 2020 research and innovation programme and EFPIA. FB is supported by the NIHR UCLH biomedical research center.
Lopes Alves I, Collij LE, Altomare D, et al. Quantitative amyloid PET in Alzheimer's disease: the AMYPAD prognostic and natural history study. Alzheimer's Dement. 2020;16:750–758. 10.1002/alz.12069
Isadora Lopes Alves and Lyduine Collij contributed equally to this study.
REFERENCES
- 1. Jack CR, Jr. , Knopman DS, Jagust WJ, et al. Tracking pathophysiological processes in Alzheimer's disease: an updated hypothetical model of dynamic biomarkers. Lancet Neurol. 2013;12(2):207‐216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Vandenberghe R, Rinne JO, Boada M, et al. Bapineuzumab for mild to moderate Alzheimer's disease in two global, randomized, phase 3 trials. Alzheimers Res Ther. 2016;8(1):18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Honig LS, Vellas B, Woodward M, et al. Trial of solanezumab for mild dementia due to Alzheimer's disease. N Engl J Med. 2018;378(4):321‐330. [DOI] [PubMed] [Google Scholar]
- 4. Salloway S, Sperling R, Fox NC, et al. Two phase 3 trials of bapineuzumab in mild‐to‐moderate Alzheimer's disease. N Engl J Med. 2014;370(4):322‐333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Egan MF, Kost J, Tariot PN, et al. Randomized trial of verubecestat for mild‐to‐moderate Alzheimer's disease. N Engl J Med. 2018;378(18):1691‐1703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sevigny J, Chiao P, Bussiere T, et al. The antibody aducanumab reduces Abeta plaques in Alzheimer's disease. Nature. 2016;537(7618):50‐56. [DOI] [PubMed] [Google Scholar]
- 7. Jack CR, Jr. , Holtzman DM. Biomarker modeling of Alzheimer's disease. Neuron 2013;80(6):1347‐1358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Donohue MC, Aisen PS. Elevated brain amyloid in cognitively normal individuals‐reply. JAMA. 2017;318(14):1393‐1394. [DOI] [PubMed] [Google Scholar]
- 9. Insel PS, Hansson O, Mackin RS, et al. Amyloid pathology in the progression to mild cognitive impairment. Neurobiol Aging. 2018;64:76‐84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Roberts RO, Aakre JA, Kremers WK, et al. Prevalence and outcomes of amyloid positivity among persons without dementia in a longitudinal, population‐based setting. JAMA Neurol. 2018;75(8):970‐979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Farrell ME, Chen X, Rundle MM, et al. Regional amyloid accumulation and cognitive decline in initially amyloid‐negative adults. Neurology. 2018;91(19). 10.1212/WNL.0000000000006469 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Stocker H, Nabers A, Perna L, et al. Prediction of Alzheimer's disease diagnosis within 14 years through Abeta misfolding in blood plasma compared to APOE4 status, and other risk factors. Alzheimers Dement. 2020;16:283‐291. [DOI] [PubMed] [Google Scholar]
- 13. Veerabhadrappa B, Delaby C, Hirtz C, et al. Detection of amyloid beta peptides in body fluids for the diagnosis of alzheimer's disease: where do we stand? Crit Rev Clin Lab Sci. 2020;57:99‐113. [DOI] [PubMed] [Google Scholar]
- 14. de Wilde A, Reimand J, Teunissen CE, et al. Discordant amyloid‐beta PET and CSF biomarkers and its clinical consequences. Alzheimers Res Ther. 2019;11(1):78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Palmqvist S, Mattsson N, Hansson O, et al. Cerebrospinal fluid analysis detects cerebral amyloid‐beta accumulation earlier than positron emission tomography. Brain. 2016;139(Pt 4):1226‐1236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Guo T, Dukart J, Brendel M, et al. Rate of β‐amyloid accumulation varies with baseline amyloid burden: implications for anti‐amyloid drug trials. Alzheimers Dement. 2018;14(11):1387‐1396. [DOI] [PubMed] [Google Scholar]
- 17. Lammertsma AA. Forward to the past: the case for quantitative PET imaging. J Nucl Med. 2017;58(7):1019‐1024. [DOI] [PubMed] [Google Scholar]
- 18. Thal DR, Beach TG, Zanette M, et al. [(18)F]flutemetamol amyloid positron emission tomography in preclinical and symptomatic Alzheimer's disease: specific detection of advanced phases of amyloid‐beta pathology. Alzheimers Dement. 2015;11(8):975‐985. [DOI] [PubMed] [Google Scholar]
- 19. Villeneuve S, Rabinovici GD, Cohn‐Sheehy BI, et al. Existing Pittsburgh Compound‐B positron emission tomography thresholds are too high: statistical and pathological evaluation. Brain. 2015;138(Pt 7):2020‐33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. van Berckel BN, Ossenkoppele R, Tolboom N, et al. Longitudinal amyloid imaging using 11C‐PiB: methodologic considerations. J Nucl Med. 2013;54(9):1570‐1576. [DOI] [PubMed] [Google Scholar]
- 21. Schmidt ME, Chiao P, Klein G, et al. The influence of biological and technical factors on quantitative analysis of amyloid PET: points to consider and recommendations for controlling variability in longitudinal data. Alzheimers Dement. 2015;11(9):1050‐1068. [DOI] [PubMed] [Google Scholar]
- 22. Bischof GN, Jacobs HIL. Subthreshold amyloid and its biological and clinical meaning: long way ahead. Neurology. 2019;93(2):72‐79. [DOI] [PubMed] [Google Scholar]
- 23. Ritchie CW, Molinuevo JL, Truyen L, et al. Development of interventions for the secondary prevention of Alzheimer's dementia: the European Prevention of Alzheimer's Dementia (EPAD) project. Lancet Psychiatry. 2016;3(2):179‐186. [DOI] [PubMed] [Google Scholar]
- 24. Vaudano E, Vannieuwenhuyse B, Van Der Geyten S, et al. Boosting translational research on Alzheimer's disease in Europe: the Innovative Medicine Initiative AD research platform. Alzheimers Dement. 2015;11(9):1121‐1122. [DOI] [PubMed] [Google Scholar]
- 25. Konijnenberg E, Carter SF, Ten Kate M, et al. The EMIF‐AD PreclinAD study: study design and baseline cohort overview. Alzheimers Res Ther. 2018;10(1):75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Molinuevo JL, Gramunt N, Gispert JD, et al. The ALFA project: a research platform to identify early pathophysiological features of Alzheimer's disease. Alzheimers Dement (N Y). 2016;2(2):82‐92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Heeman F, Yaqub M, Lopes Alves I, et al. Optimized dual‐time‐window protocols for quantitative [(18)F]flutemetamol and [(18)F]florbetaben PET studies. EJNMMI Res. 2019;9(1):32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Nelissen N, Van Laere K, Thurfjell L, et al. Phase 1 study of the Pittsburgh compound B derivative 18F‐flutemetamol in healthy volunteers and patients with probable Alzheimer disease. J Nucl Med. 2009;50(8):1251‐1259. [DOI] [PubMed] [Google Scholar]
- 29. Barthel H, Gertz HJ, Dresel S, et al. Cerebral amyloid‐beta PET with florbetaben (18F) in patients with Alzheimer's disease and healthy controls: a multicentre phase 2 diagnostic study. Lancet Neurol. 2011;10(5):424‐435. [DOI] [PubMed] [Google Scholar]
- 30. Wolz R, Aljabar P, Hajnal JV, et al. LEAP: learning embeddings for atlas propagation. Neuroimage. 2010;49(2):1316‐1325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Grecchi E, Foley C, Gispert JD, Wolz R. Centiloid PET SUVR analysis using the supratentorial white matter as reference region. Alzheimers Dement. 2018;14:P1278. [Google Scholar]
- 32. Gunn RN, Lammertsma AA, Hume SP, Hume SP, Cunningham VJ. Parametric imaging of ligand‐receptor binding in PET using a simplified reference region model. Neuroimage. 1997;6(4):279‐287. [DOI] [PubMed] [Google Scholar]
- 33. Logan J, Fowler JS, Volkow ND, et al. Graphical analysis of reversible radioligand binding from time‐activity measurements applied to [N‐11C‐methyl]‐(‐)‐cocaine PET studies in human subjects. J Cereb Blood Flow Metab. 1990;10(5):740‐747. [DOI] [PubMed] [Google Scholar]
- 34. Grothe MJ, Barthel H, Sepulcre J, et al. In vivo staging of regional amyloid deposition. Neurology. 2017;89(20):2031‐2038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Hanseeuw BJ, Betensky RA, Mormino EC, et al. PET staging of amyloidosis using striatum. Alzheimers Dement. 2018;14(10):1281‐1292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Mattsson N, Palmqvist S, Stomrud E, Vogel J, Hansson O. Staging β‐amyloid pathology with amyloid positron emission tomography. JAMA Neurol. 2019;76:1319‐1329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Klunk WE, Koeppe RA, Price JC, et al. The Centiloid Project: standardizing quantitative amyloid plaque estimation by PET. Alzheimers Dement. 2015;11(1):1‐15 e1‐4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Chen YJ, Rosario BL, Mowrey W, et al. Relative 11C‐PiB delivery as a proxy of relative CBF: quantitative evaluation using single‐session 15O‐water and 11C‐PiB PET. J Nucl Med. 2015;56(8):1199‐1205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Fortier I, Raina P, Van den Heuvel ER, et al. Maelstrom research guidelines for rigorous retrospective data harmonization. Int J Epidemiol. 2016;46(1):103‐105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Collij L, Konijnenberg E, Reimand J, et al. Assessing amyloid pathology in cognitively normal subjects using [(18)F]Flutemetamol PET: comparing visual reads and quantitative methods. J Nucl Med. 2019;60(4):541‐547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Ottoy J, Verhaeghe J, Niemantsverdriet E, et al. 18F‐FDG PET, the early phases and the delivery rate of 18F‐AV45 PET as proxies of cerebral blood flow in Alzheimer's disease: validation against 15O‐H2O PET. Alzheimer's & Dementia. 2019;15(9):1172‐1182. [DOI] [PubMed] [Google Scholar]
- 42. Loera‐Valencia R, Cedazo‐Minguez A, Kenigsberg PA, et al. Current and emerging avenues for Alzheimer's disease drug targets. J Intern Med. 2019;286(4):398‐437. [DOI] [PubMed] [Google Scholar]
- 43. Cummings J, Lee G, Ritter A, et al. Alzheimer's disease drug development pipeline: 2018. Alzheimers Dement 2018;4:195‐214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Cummings J, Ritter A, Zhong K. Clinical trials for disease‐modifying therapies in Alzheimer's disease: a primer, lessons learned, and a blueprint for the future. J Alzheimers Dis. 2018;64(s1):S3‐S22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Donohue MC, Sperling RA, Petersen R, et al. Association between elevated brain amyloid and subsequent cognitive decline among cognitively normal persons. JAMA. 2017;317(22):2305‐2316. [DOI] [PMC free article] [PubMed] [Google Scholar]


