Abstract
Objective
This study aimed to compare cytokine and adipokine levels in patients with obesity with and without type 2 diabetes (T2D) at baseline and 6 months after Roux‐en‐Y gastric bypass (RYGB) with healthy controls.
Methods
A total of 34 patients (21 with T2D) with BMI of 30 to 45 kg/m2 were compared with 25 healthy controls without obesity. Cytokines, adipokines, and peptides of relevance for inflammation and metabolism were analyzed in plasma.
Results
Significant decreases in weight and glycated hemoglobin A1c were observed. At baseline, interleukin‐6 (IL‐6), IFN‐β, IL‐18, leptin, and hepatocyte growth factor were higher in all patients with obesity compared with healthy controls. In patients without T2D, TNF‐α, IL‐1α, IL‐2, IL‐15, and visfatin were also increased, whereas bone morphogenic protein‐4 was decreased. Following RYGB, IL‐6 and hepatocyte growth factor were still increased in both groups compared with controls. In T2D patients, IFN‐β, IL‐27, IL‐1α, IL‐2, regenerating islet‐derived protein 3A, visfatin, and osteopontin were found to be increased. In patients without T2D, TNF‐α, IL‐1α, IL‐2, IL‐15, leptin, and visfatin remained increased.
Conclusions
The altered cytokine profile of patients with obesity persisted after RYGB despite large weight loss and improved metabolic status, thus reflecting an inherent inflammatory state.
Study Importance Questions.
What is already known?
-
►
Various cytokines, adipokines, and chemokines are altered in patients with obesity and type 2 diabetes, and the changes after Roux‐en‐Y gastric bypass surgery have been previously described.
What does this study add?
-
►
Patients with obesity have an altered inflammatory and adipokine profile compared with healthy controls, a persisting result after Roux‐en‐Y gastric bypass surgery, despite the dramatic weight loss.
How might these results change the focus of clinical practice?
-
►
The persisting differences in the inflammatory and adipokine profile between patients who have had gastric bypass surgery and healthy controls need to be addressed clinically in order to further reduce the risk of long‐term complications.
Introduction
Obesity is a global health challenge that has become one of the leading risk factors for premature death. Patients with obesity are at risk of developing metabolic syndrome (1) and type 2 diabetes (T2D) with all of the associated cardiovascular complications. However, the state of obesity itself also has been associated with disturbances in lipid and glucose metabolism, insulin signaling, adipose tissue development, and inflammation (2). It has been proposed that low‐grade inflammation in adipose tissue contributes to insulin resistance in obesity, and there is increasing evidence of an altered immunological profile in patients with obesity. Of great interest is the fact that obesity has also been associated with the incidence and severity of infections (3). Recent data have indicated that both obesity and T2D are risk factors for severe coronavirus disease 2019 infections, with a higher prevalence of acute respiratory syndrome requiring invasive mechanical ventilation (4). Roux‐en‐Y gastric bypass (RYGB) surgery has been shown to reverse and normalize many of the metabolic changes associated with obesity. However, the underlying mechanisms remain unclear, and the importance of the inflammatory profile is so far poorly understood (5). Previous studies have shown that various cytokines, adipokines, and chemokines are altered in patients with obesity and T2D. However, the data on how these factors behave in patients with obesity without T2D compared with patients with obesity with T2D undergoing RYGB are poorly understood.
In this exploratory study, we analyzed cytokines and adipokines of importance for both metabolic regulation and inflammation. We investigated whether these circulating factors are altered differently in patients with and without T2D following RYGB and how they compare with levels in healthy controls.
Methods
Patient selection and study design
Patients (aged 18‐60 years, BMI 30‐45 kg/m2) scheduled to undergo RYGB were recruited at the Department of Endocrinology and Diabetology at Uppsala University Hospital in Sweden. Healthy controls with normal BMI were recruited through advertising. Out of the 21 patients with T2D, 13 were also part of another study (6, 7). The patients with T2D had a mean diabetes duration of 5 years (ranging from 4 to 8 years), and the majority were treated with oral antidiabetics, primarily metformin (n = 19), in combination with sulfonylureas (n = 1) and incretins (n = 3). One patient was treated with insulin.
This study was approved by the Regional Ethical Board of Uppsala County (DNR 2014/255, 2013/356) and conducted per the declaration of Helsinki as revised in 2000. Study participants were given oral and written information and they signed a consent form before their inclusion in the study.
Study procedures
The patients were assessed on the following two occasions: at baseline and 6 months following surgery. Healthy controls were assessed once. The following investigations were performed during 1‐day visits: anthropometric measurements (body weight, waist/hip circumference) and a physical examination. Blood samples were collected in the morning following an overnight fast for biochemical measurements. A subcutaneous adipose tissue (SAT) biopsy was performed in patients with T2D at baseline and 6 months after RYGB. All patients underwent a period of caloric restriction using a low‐calorie diet 2 to 4 weeks prior to surgery according to clinical routine. All treatment adjustments during follow‐up were based on clinical criteria and performed as per clinical practice.
Biochemical measurements
Glycated hemoglobin A1c (HbA1c), fasting glucose, C‐peptide, cholesterol, high‐density lipoprotein cholesterol, low‐density lipoprotein cholesterol, and triglycerides were analyzed according to clinical routine. If not analyzed immediately, plasma samples were stored at −80°C. Hormone analysis was performed using commercially available kits that tested growth hormone (Immulite 2000XPi, Siemens Healthcare Global, Erlangen, Germany) and total adiponectin (R&D Systems, Minneapolis, Minnesota). Leptin, regenerating islet‐derived protein 3A (Reg3A), tumor necrosis factor alpha (TNF‐α), interleukin‐6 (IL‐6), IL‐33, visfatin, interferon beta (IFN‐β), IL‐27, IFN‐γ, IL‐1α, IL‐2, resistin, osteopontin, IL‐15, IL‐21, hepatocyte growth factor (HGF), bone morphogenic protein‐4 (BMP4), and IL‐18 were analyzed using a magnetic bead‐based Luminex assay (R&D Systems) at the plasma profiling unit at SciLife Lab.
Gene expression analysis
Total RNA from SAT was isolated using the RNeasy Lipid Tissue Mini Kit (Qiagen, Hilden, Germany). The concentration and purity of total RNA were measured with the Nanodrop (Thermo Fisher Scientific, Waltham, Massachusetts). In total, 400 ng of RNA was reverse transcribed as per manufacturer instructions using a High‐Capacity cDNA Reverse Transcriptase Kit (Applied Biosystems, Foster City, California). The expression levels of different transcripts were measured with TaqMan gene expression assays (Thermo Fisher Scientific). Gene expression detection was performed using the QuantStudio 3 sequence detection system (Applied Biosystems). The data were calculated using a relative standard curve method or 2−delta Ct. Results were plotted as relative quantification using 18S ribosomal RNA or glucuronidase beta (GUSB) as an endogenous control. All samples were run in duplicates. This was performed only in the patients with T2D, as the adipose tissue biopsy has been performed as part of another study’s protocol.
Statistical analysis
Statistical analysis was performed using GraphPad Prism version 6.07 (GraphPad Software, San Diego, California). A nonparametric one‐way ANOVA (Kruskal‐Wallis test) for multiple comparison using a Dunn post hoc test was applied for the comparison of controls with patients with obesity and with or without T2D prior to versus following surgery, respectively. A paired two‐tailed t test or Wilcoxon matched‐pairs signed rank test was used to compare differences prior to versus following surgery within the same group for normally and not normally distributed data, respectively. Cytokines and adipokines that were significantly different between the two groups were data normalized for age and BMI, respectively, and compared using a Mann‐Whitney U test. Normality distribution was determined by a D´Agostino‐Pearson Omnibus normality test. All correlations were calculated using a nonparametric Spearman correlation. P < 0.05 was considered statistically significant. Correction for multiple testing was performed using the Bonferroni‐Dunn method. All values are given as mean (SEM).
Results
Anthropometric and metabolic parameters
In total, 34 patients with obesity (21 with T2D) and 25 healthy controls were included. Patients with obesity and with and without T2D had, as expected, higher BMI and body weight compared with controls, but BMI and body weight were comparable between the two groups. As expected, patients with obesity displayed significant weight loss following RYGB, but BMI was still increased compared with controls for both groups (Table 1). Patients with T2D were slightly older than the controls, although the difference was not significant when adjusted for multiple testing (Table 1). As expected, patients with T2D had higher HbA1c and fasting glucose levels at baseline compared with patients without T2D and controls. Also, patients without T2D had slightly increased HbA1c levels compared with controls, although the levels were within the normal range. C‐peptide levels were higher when compared with controls but not when compared with patients without T2D. Postsurgery patients with T2D, having discontinued their antidiabetic treatment (n = 14), still displayed higher HbA1c (although within the normal range), but the fasting levels of glucose were comparable to controls and patients without T2D (Table 1).
TABLE 1.
Descriptive data of patients with obesity and with and without type 2 diabetes prior to and following RYGB compared with healthy controls
| Patients with obesity and T2D (n = 21) | Patients with obesity without T2D (n = 13) | Healthy controls (n = 25) | |||
|---|---|---|---|---|---|
| Baseline | Post‐RYGB | Baseline | Post‐RYGB | ||
| Age (years) | 49 ± 2** | 42 ± 3 | 39 ± 3 | ||
| Gender (F/M) | 18/3 | 12/1 | 14/11 | ||
| BMI (kg/m2) | 38.3 ± 0.9***aa | 29.2 ± 0.6***aa ### cc | 43.1 ± 0.8***aa | 32.8 ± 1.2***aa ### cc | 24.4 ± 0.5 |
| Weight (kg) | 104.9 ± 3.2***aa | 79.1 ± 2.0### cc | 117.9 ± 4.4***aa | 91.6 ± 5.2*### cc | 76.4 ± 2.6 |
| HbA1c (mmol/mol) | 53.7 ± 2.9***aa ++ | 40.2 ± 1.4***aa +### cc | 37.4 ± 1.1* | 33.7 ± 1.3## cc | 32.5 ± 0.6 |
| Glucose (mmol/l) | 8.5 ± 0.4***aa ++ | 6.3 ± 0.3### cc | 6.2 ± 0.2 | 5.6 ± 0.1# | 5.6 ± 0.1 |
| C‐peptide (nmol/l) | 1.5 ± 0.1***aa | 0.9 ± 0.1*### cc | 1.4 ± 0.1***aa | 0.9 ± 0.04**### cc | 0.7 ± 0.06 |
| Cholesterol (mmol/l) | 5.1 ± 0.2 | 4.3 ± 0.2*## c | 5.2 ± 0.3 | 4.8 ± 0.3 | 5.6 ± 0.3 |
| HDL Cholesterol (mmol/l) | 1.0 ± 0.04***aa | 1.1 ± 0.05**a ## | 1.1 ± 0.05* | 1.1 ± 0.1 | 1.6 ± 0.1 |
| LDL Cholesterol (mmol/l) | 3.2 ± 0.2 | 2.7 ± 0.2## c | 3.5 ± 0.3 | 3.1 ± 0.3 | 3.5 ± 0.3 |
| Triglycerides (mmol/l) | 2.2 ± 0.2**a | 1.2 ± 0.1### cc | 2.1 ± 0.3* | 1.4 ± 0.2## c | 1.2 ± 0.2 |
All values are given as mean ± SEM.
Comparison with healthy controls: * denotes P < 0.05, **< 0.01, ***< 0.001; a denotes corrected P < 0.05, aa< 0.01.
Comparison between patients with T2D vs patients without T2D: + denotes. P < 0.05, ++<0.01.
Comparison within groups at baseline and post‐RYGB # denotes. P < 0.05, ##< 0.01, ###< 0.001; c denotes corrected P < 0.05, cc< 0.01.
HbA1c, glycated hemoglobin A1c; HDL, high‐density lipoprotein; LDL, low‐density lipoprotein; RYGB, Roux‐en‐Y gastric bypass; T2D, type 2 diabetes.
Circulating cytokines
At baseline, IL‐6, IFN‐β, and IL‐18 were higher in patients with and without T2D compared with controls. Furthermore, patients without T2D displayed increased levels of TNF‐α, IL‐1α, IL‐2, and IL‐15 compared with controls. At follow‐up after RYGB, IL‐6 was still found to be increased in both groups (Figure 1). Also, IL‐1α and IL‐2 increased in patients with T2D and was found to have increased in both groups (Figure 1), whereas TNF‐α, IL‐15, and IL‐18 were increased only in patients without T2D (Table 2). Also, IL‐27 increased following surgery in T2D patients and was found to have increased when compared with controls.
Figure 1.
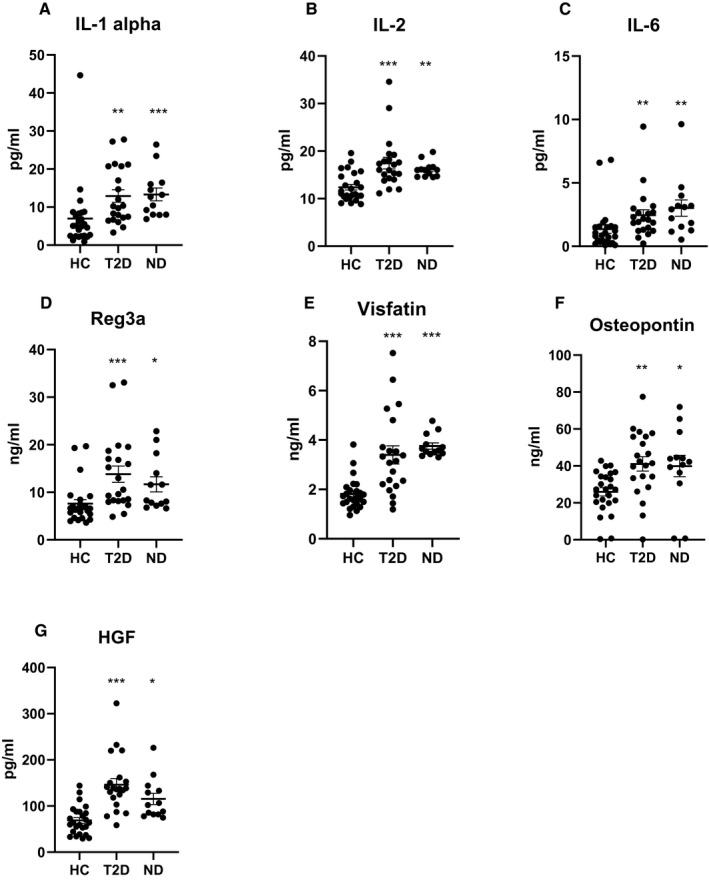
Levels of cytokines, adipokines, and hormones that were significantly different in both patients with T2D and patients without T2D (ND) 6 months after Roux‐en‐Y gastric bypass surgery compared with HC. *P < 0.05, **P < 0.01, ***P < 0.001. HC, healthy controls; T2D, type 2 diabetes.
TABLE 2.
Circulating cytokines, adipokines, and other peptides in patients with obesity and T2D vs. obesity without T2D prior to and following RYGB compared with healthy controls
| Patients with obesity and T2D (n = 21) | Patients with obesity without T2D (n = 13) | Healthy controls (n = 25) | |||
|---|---|---|---|---|---|
| Baseline | Post‐RYGB | Baseline | Post‐RYGB | ||
| Cytokines | |||||
| TNF‐α (pg/ml) | 5.8 ± 0.8+ | 5.7 ± 0.8+ | 9.6 ± 1.2***aa | 9.3 ± 1.0***aa | 3.7 ± 0.6 |
| IL‐6 (pg/ml) | 2.5 ± 0.3**a | 2.5 ± 0.4** | 3.8 ± 0.5***aa | 3.0 ± 0.6**a | 1.4 ± 0.3 |
| IL‐33 (pg/ml) | 5.9 ± 1.0 | 8.5 ± 1.8# | 10.4 ± 3.9 | 16.7 ± 5.6# | 5.6 ± 0.8 |
| IFN‐β (pg/ml) | 7.0 ± 1.4***aa | 6.1 ± 0.8* | 7.1 ± 0.9* | 7.2 ± 0.9 | 6.0 ± 0.1 |
| IL‐27 (ng/ml) | 410 ± 30 | 574 ± 38**### acc | 514 ± 68 | 597 ± 93 | 402 ± 35 |
| IFN‐γ (pg/ml) | 21.8 ± 1.9 | 34.2 ± 8.1 | 34.0 ± 11.8 | 42.2 ± 21.1 | 23.8 ± 2.2 |
| IL‐1 α (pg/ml) | 9.6 ± 1.5 | 12.9 ± 1.6**# a | 14.0 ± 2.3**a | 13.3 ± 1.7***aa | 7.0 ± 1.7 |
| IL‐2 (pg/ml) | 13.6 ± 0.6+ | 17.5 ± 1.2***### aacc | 16.9 ± 0.7***aa | 16.1 ± 0.4**a | 12.4 ± 0.6 |
| IL‐15 (pg/ml) | 5.9 ± 1.0 | 7.5 ± 1.5 | 8.7 ± 1.9* | 8.0 ± 1.7* | 4.1 ± 0.6 |
| IL‐21 (pg/ml) | 10.9 ± 1.3 | 12.4 ± 2.6 | 14.0 ± 2.8 | 19.4 ± 6.9 | 13.6 ± 1.3 |
| IL‐18 (pg/ml) | 310 ± 30* | 252 ± 19## c | 397 ± 49***aa | 285 ± 26*## | 205 ± 16 |
| Adipokines and other peptides | |||||
| Adiponectin (µg/ml) | 4.7 ± 0.5* | 7.1 ± 0.6### cc | 5.8 ± 0.9 | 7.1 ± 0.7 | 8.5 ± 1.1 |
| Leptin (ng/ml) | 74.5 ± 17.5***aa | 13.8 ± 2.0### cc | 67.0 ± 6.9***aa | 21.8 ± 3.7**### cc | 11.1 ± 2.4 |
| Reg3A (ng/ml) | 7.8 ± 0.8 | 13.8 ± 1.7***## aacc | 6.6 ± 0.9 | 11.7 ± 1.6*### cc | 7.6 ± 0.8 |
| Visfatin (ng/ml) | 2.8 ±0.4+ | 3.4 ± 0.4***##a ac | 3.7 ± 0.1***aa | 3.8 ± 0.1***aa | 1.8 ± 0.1 |
| Growth Hormone (ng/ml) | 0.8 ± 0.3 | 3.5 ± 0.8### cc | 0.9 ± 0.3 | 1.9 ± 0.5 | 2.7 ± 0.7 |
| Resistin (ng/ml) | 7.7 ± 1.1+ | 8.6 ± 1.2### cc | 9.3 ± 0.8 | 9.1 ± 1.0 | 7.6 ± 0.3 |
| Osteopontin (ng/ml) | 21.9 ± 1.9+ | 41.1 ± 3.9**### cc | 31.2 ± 2.8 | 39.9 ± 5.8* | 25.9 ± 2.3 |
| Hepatocyte Growth Factor (pg/ml) | 156 ± 12***aa | 146 ± 13***aa | 144 ± 18***aa | 115 ± 12*# | 72 ± 6 |
| BMP‐4 (pg/ml) | 18.7 ± 1.2+ | 21.0 ± 1.6# c | 14.3 ± 2.2***aa | 18.0 ± 2.1 | 23.1 ± 2.1 |
All values are given as mean ± SEM.
Comparison with healthy controls: * denotes P < 0.05, **< 0.01, ***< 0.001; a denotes corrected P < 0.05. aa< 0.01.
Comparison between patients with T2D vs patients without T2D + denotes. P < 0.05, ++< 0.01, +++< 0.001.
Comparison within groups at baseline and post‐RYGB # denotes P < 0.05, ##< 0.01, ###< 0.001; c denotes corrected P < 0.05.
BMP4, bone morphogenic protein 4; Reg3A, regenerating islet‐derived protein 3A; TNF, tumor necrosis factor; IL, interleukin; IFN, inteferon; RYGB, Roux‐en‐Y gastric bypass surgery; T2D, type 2 diabetes.
Interestingly, at baseline, only TNF‐α and IL‐2 were lower in patients with T2D compared with patients without T2D. TNF‐α was also lower in T2D patients when normalized for BMI (P = 0.03) and age (P = 0.007), whereas IL‐2 was significantly lower only when normalized for age (P = 0.0006). Postsurgery TNF‐α levels were still lower in T2D patients and IFN‐β was also lower, though only marginally (Table 2). However, when correcting for multiple testing, there were no significant differences in circulating cytokines between patients with and without T2D (Table 2).
When computing correlations for all patients with obesity, baseline BMI was found to be positively correlated with the baseline levels of TNF‐α (P = 0.001, r = 0.53), IL‐6 (P = 0.01, r = 0.43), IL‐27 (P = 0.03, r = 0.38), IL‐2 (P = 0.02, r = 0.41), and IL‐18 (P = 0.04, r = 0.35). Age was not correlated with circulating cytokine levels in controls.
Adipokines and other peptides
At baseline, leptin and HGF levels of patients both with and without T2D were higher compared with controls, including when corrected for multiple testing. Patients without T2D displayed increased levels of visfatin but lower levels of BMP4 compared with controls even after correcting for multiple testing. Moreover, the T2D patients had significantly lower levels of adiponectin at baseline when compared with controls, although levels were not significant when corrected for multiple testing (Table 2). At follow‐up, HGF was still increased in both groups (Figure 1), whereas leptin was increased only in patients without T2D. Adiponectin increased in patients with T2D and it was comparable to that of controls and patients without T2D following surgery. Also, visfatin increased in patients with T2D and was increased when compared with controls at follow‐up (Figure 1). Reg3A increased in both groups and was elevated compared with controls (Figure 1). Also, the levels of osteopontin were increased in both groups at follow‐up (Figure 1).
At baseline, visfatin, resistin, and osteopontin were lower in the patients with obesity and T2D compared with those without T2D (Table 2). However, when correcting for multiple testing, there were no significant differences in circulating adipokines between the two groups with obesity at baseline. When normalizing for age and BMI, visfatin (P = 0.002 and P = 0.02) and osteopontin (P = 0.0003 and P = 0.03) remained lower in patients with T2D. Resistin was also significantly lower when normalized for age (P = 0.002), but not for BMI. BMP4 was instead higher in patients with T2D even after normalizing data for BMI (P = 0.0002), but not for age. Following surgery, the circulating levels of adipokines were similar in both groups with obesity (Table 2).
When computing correlation for all study participants with obesity, we found that the baseline BMI was positively correlated with the circulating levels of leptin (P = 0.002, r = 0.52), resistin (P = 0.003, r = 0.50), and osteopontin (P = 0.01, r = 0.44), whereas BMP4 was negatively correlated with BMI (P = 0.03, r = −0.37). When computing correlations for the two separate groups of patients with obesity, we observed no correlations between circulating adipokines and BMI among patients without T2D. Among the T2D patients, a positive correlation between BMI and leptin (P = 0.005, r = 0.59) was observed. Age was not correlated with circulating adipokine levels in controls.
Gene expression
Gene expression from SAT in the patients with diabetes was measured for the cytokines and adipokines that showed significant changes in the circulating plasma. The BMP4 mRNA levels remained unchanged at 6 months compared with baseline, whereas the expression of IL‐6, IL‐18, and RETN (gene encoding resistin) were reduced significantly at 24 weeks; however, no decrease was seen in the plasma levels of these cytokines. The mRNA levels of visfatin and IL‐33 did not significantly change following surgery.
Discussion
RYGB results in large weight loss, and patients with T2D can often achieve a normoglycemic state for a prolonged period without the use of glucose‐lowering drugs (8). However, many of the underlying metabolic and inflammatory mechanisms associated with weight loss and the correction of glycemic control remain elusive. Following RYGB, local effects in the intestine and adipocytes have been observed in addition to the systemic metabolic effects (9). Here, we present data on how circulating levels of cytokines and adipokines are altered following RYGB in patients with obesity with and without T2D.
Anthropometric and metabolic parameters
Patients with obesity and T2D displayed numerically lower BMI and body weight (though not statistically significant) compared with patients without T2D. This can be explained by the more active approach toward RYGB in the clinical care of patients with concomitant risk factors, such as T2D, in Sweden. The impaired metabolic control in patients with T2D before undergoing RYGB improved already 6 months following surgery with a nearly normal glycemic control even after the cessation of their antidiabetic treatment. However, HbA1c levels were still increased in patients with T2D (although within the normal range) compared with patients without T2D and controls. Also, among patients without T2D, HbA1c improved within the normal range following RYGB. Insulin resistance is a hallmark of T2D, and, despite this, we found that the fasting levels of C‐peptide were similar in both groups of patients both at baseline and following surgery. However, C‐peptide levels improved following RYGB in both groups. This sheds further light on the state of obesity itself and the metabolic changes associated with obesity even under normal glycemic control.
Circulating cytokines
At baseline, several inflammatory cytokines (IL‐6, IFN‐β, and IL‐18) were elevated in the patients with and without T2D compared with controls. In addition, patients without T2D displayed increased levels of proinflammatory cytokines TNF‐α, IL‐1α, IL‐2, and IL‐15 compared with controls. At the 6‐month follow‐up, IL‐6 was still found to be increased in both groups with obesity. The levels of IL‐18 decreased in both groups but remained increased in patients without T2D compared with controls. On the other hand, IL‐1α and IL‐2 increased in the T2D patients and, at follow‐up, were found to be significantly higher in both groups with obesity compared with controls. Taken together, these data indicate that, in patients with T2D, there was a slight reduction in IL‐18 and an increase in IL‐27 after RYGB but, overall, a more pronounced inflammatory profile compared with controls. Patients without T2D, on the other hand, displayed a more pronounced inflammatory profile already at baseline, which was maintained at follow‐up.
Adipose tissue can produce and secrete several proinflammatory cytokines and is becoming increasingly recognized for its importance in regulating immune functions. In addition to release of adipokines, adipocytes can, for instance, also secrete TNF‐α and IL‐6 (10). Traditionally, IL‐6 is considered a proinflammatory cytokine but has also been found to play a role in metabolic regulation. In some studies, IL‐6 has been found to increase insulin sensitivity through its effects on adipose tissue and skeletal muscle (11) whereas other studies have found a correlation between high levels of IL‐6 and insulin resistance (12). Therefore, whether IL‐6 exerts positive or negative effects is still the subject of controversy. Similar to our findings, previous studies have found that the levels of IL‐6 remain increased after RYGB (13). The present positive correlation of IL‐6 to BMI and the lower gene expression of IL‐6 in adipocytes following surgery implies that the levels are maintained through its secretion from immune cells. Of current interest is the notion that this could be a major contributor to the increased risk of severe coronavirus disease 2019 infections in both patients with obesity and T2D, and perhaps also an indication that even patients who have achieved normal weight following RYGB are at greater risk.
Interestingly, only two cytokines, TNF‐α and IL‐2, varied between T2D and nondiabetic patients at baseline. When normalizing the data for age and BMI, TNF‐α was still found to be significantly higher in nondiabetic patients. IL‐2 was significantly lower when normalized for age, but not for BMI. However, it should be noted that after correcting for multiple testing, there were no statistical differences in circulating cytokine levels between the two groups with obesity. Increased levels of TNF‐α could be associated with a state of chronic inflammation in obesity, as it has previously been found to be increased in patients with obesity compared with controls (14). In line with those findings, we found that levels of TNF‐α were increased in patients with obesity, but, interestingly, the levels were found to be higher in patients without T2D. We observed a positive correlation between preoperative BMI and TNF‐α levels, which further supported the contribution from adipose tissue and stromovascular fraction to the systemic levels of TNF‐α (15). Despite that, TNF‐α levels remained largely unaltered in both groups following RYGB, which indicates that it is not only the sheer volume but perhaps also the adipocyte phenotype in obesity that impacts TNF‐α production. Additionally, the levels also differed between the two groups with obesity when normalizing the data for BMI. IL‐2 was first discovered as an autocrine growth factor essential for T‐cell proliferation and effector response (16) but has later been recognized as an important regulator of the immune response to maintain self‐tolerance. Peripheral blood mononuclear cells from individuals with T2D have been shown to produce significantly less IL‐2, a surrogate marker of proliferation when stimulated with T‐cell mitogen phytohemagglutinin. The lower production of IL‐2 after phytohemagglutinin stimulation observed in individuals with obesity and T2D suggests that diabetes further impairs the ability of T cells to respond and proliferate (17). Circulating levels of IL‐2 have previously been found to be reduced by calorie restriction but unaffected by RYGB (14). In contrast, we observed an increase of IL‐2 in T2D patients after RYGB despite its positive correlation with BMI. This could indicate that the improvement of metabolic control following RYGB also affects the peripheral blood mononuclear cells’ capacity to produce IL‐2.
IL‐18 is produced by a large number of tissues, including adipose tissue, skeletal muscle, pancreas, brain, and endothelium (18). In patients with obesity, levels of IL‐18 have previously been found to be increased and correlated with insulin resistance and metabolic syndrome (19). IL‐18 is also increased in T2D patients (20). However, in experimental studies, IL‐18 has been shown to exert potent anti‐obesity effects, and mice administered with IL‐18 are protected from weight gain, whereas knockout mouse models gain excessive amounts of adipocytes and become insulin resistant (21). It has been proposed that the production of IL‐18 is a response to increasing obesity as an attempt to prevent further weight gain but that the tonic increase of IL‐18 eventually leads to a decrease in IL‐18 receptor expression (22). Similar to our findings, previous studies have reported that IL‐18 decreased following RYGB (23), and, in line with those findings, we observed a positive correlation between IL‐18 and BMI. We also found that gene expression of IL‐18 in adipose tissue was reduced following RYGB, which further supports the notion that the reduced circulating levels of IL‐18 are related to weight loss. Interestingly, an experimental study has recently shown that the gene expression of IL‐18 from the intestinal mucosa is reduced following RYGB (24). However, to the best of our knowledge, this is the first report in which the levels of IL‐18 have been directly compared in patients with obesity with and without T2D.
IL‐15 is a proinflammatory cytokine but has also been found to increase glucose uptake in skeletal muscle through the activation of glucose transporter type 4 (GLUT4) and increase insulin sensitivity (25). Accumulating evidence has suggested that IL‐15 can block high‐fat‐diet‐induced obesity, dyslipidemia, and hyperglycemia (26). In a previous report, circulating levels of IL‐15 were found to be positively correlated with fat mass (27), but we did not observe any correlation between BMI and IL‐15 levels.
IL‐27 is secreted from immune cells, primarily antigen‐presenting cells (28). Interestingly, IL‐27 signals through a receptor consisting of two intracellular chains of which one is shared with IL‐6. IL‐27 was initially found to enhance the function of type 1 helper T cells and suppress type 2 helper T cells, regulatory T cells, and type 17 helper T cells, but more recent data have also pointed toward an anti‐inflammatory role of IL‐27 (29). There has also been experimental evidence suggesting that IL‐27, in contrast to other IL‐12 family members, can be secreted from adipocytes in response to inflammatory stress (30). At baseline, the IL‐27 levels were comparable between patients with obesity and the controls, but the levels increased in T2D patients after RYGB. To the best of our knowledge, there are no previous reports on how the circulating levels of IL‐27 are affected in obesity or T2D following RYGB.
Adipokines and other peptides
Adiponectin levels are known to be reduced in patients with T2D compared with controls (31). We have previously found that the levels of adiponectin increase after RYGB in patients with T2D and that gene expression of adiponectin in adipose tissue also increases (6). The increase of adiponectin could contribute to improved insulin sensitivity and a more favorable metabolic control.
Reg3A was initially discovered in pancreatic exocrine secretion after an experimental induction of pancreatitis and has since been associated with acute and chronic inflammatory conditions of the pancreas (32). However, more recently, Reg3A has been ascribed anti‐microbial and anti‐inflammatory effects and has been found to reduce proinflammatory cytokine release in ulcerative colitis (33). The intestinal expression of regenerating proteins is influenced by gut microbiota (34). To the best of our knowledge, levels of Reg3A have not previously been studied in patients with obesity undergoing RYGB. Interestingly, we found that levels of Reg3A increased following RYGB in both groups with obesity and were elevated compared with controls. These changes could reflect alterations of gut microbiota, which is known to be permanently altered following RYGB (35). The increased levels of Reg3A could, as IL‐27 does, reflect an attempt to counteract the increase of other proinflammatory cytokines, such as IL1‐α and IL‐2.
Visfatin is secreted from both adipocytes and leukocytes in response to both metabolic and inflammatory cues. There have been a large number of reports linking visfatin to T2D and obesity, and there are some studies supporting a correlation between visfatin and BMI (36), but we did not observe such a correlation. In line with previously published reports, visfatin was increased in patients without T2D but did not change following RYGB, whereas the levels increased following RYGB in T2D patients. However, gene expression in adipose tissue was unaltered, which is suggestive of increased secretion from leukocytes. Also, visfatin levels were higher in patients without T2D compared with T2D patients at baseline, which was also true when normalizing the data for age and BMI. Apart from its relation to body weight and metabolic markers, previous reports have described a strong correlation between visfatin levels and inflammatory cytokines, such as IL‐6, which could explain our observed increase in visfatin levels following RYGB. In fact, when computing correlations for postsurgery visfatin levels, we found a positive correlation with both TNF‐α, IL‐1α, IL‐2, IL‐6, and IL‐21.
Osteopontin is a matrix glycoprotein secreted from various cell types in the bone and is also expressed by macrophages. Osteopontin increased significantly in patients with T2D following surgery and was increased in both groups compared with controls at 6 months. Diet‐induced weight loss lowers elevated osteopontin concentrations in patients with obesity and could be secondary to the loss of adipose tissue (37). The observed increase of osteopontin after surgery was in line with what has been previously described and appears to be related to the increased bone resorption after RYGB in relation to osteopontin’s well‐known effects on bone in this malabsorptive procedure.
Circulating levels of BMP4 are associated with adiposity, and studies have indicated that BMP4 promotes a differentiation of pre‐adipocytes into adipocytes and white adipose tissue (38). We found that the levels of BMP4 were lower in patients without T2D compared with controls, which is in agreement with previous reports in which BMP4 levels have been found to decrease after RYGB (39). In patients with T2D, the levels increased following RYGB but were still comparable to those of controls.
Several limitations must be acknowledged. First, the cytokine values were taken at 6 months, which is far from weight nadir in the majority of RYGB patients. The analyses could be repeated after the patients achieved a stable weight to further help with understanding cytokine panels. Second, since the study was exploratory in nature, the patient cohort and controls were not selected with any distinct criteria besides the planned surgery and whether they had T2D. The gender distribution in the control group was not matched, and we have therefore computed comparisons with an age‐ and gender‐matched subcohort of the control group (n = 18), which gave similar results with the main exception that IL‐15 was no longer significantly increased in the non‐T2D group at baseline or at follow‐up. Owing to the exploratory nature of the study, the sample size was not powered to detect specific changes in cytokines.
Conclusion
In conclusion, we observed that patients with and without T2D mainly retained a proinflammatory cytokine profile at 6 months following RYGB surgery. In patients with T2D, some cytokines were even further elevated. This was observed despite the dramatic weight loss and improvement in metabolic measures.
Funding agencies
This work was supported by research grants from the Swedish Diabetes Foundation (grant DIA 2017‐267), the Exodiab‐Excellence of Diabetes Research in Sweden (grant 201903), the Ernfors Foundation (grant 1905), Swedish Society for Medical Research, and Uppsala University Hospital ALF grants.
Disclosure
The authors declared no conflict of interest.
Author contributions
PK performed study design, performed data collection and analysis, and wrote the manuscript; PGK performed data collection and analysis and manuscript revision; MJP performed data collection and analysis and manuscript revision; MS performed data collection and manuscript revision; POC performed study design, data analysis, and manuscript revision; JWE performed study design, data analysis, and manuscript revision; and DE performed study design, data analysis, and manuscript revision. All authors contributed to the manuscript and have reviewed and approved the final version.
Acknowledgments
We gratefully thank the study participants for their important contributions. We are grateful for the expert assistance from the Plasma Profiling unit at SciLife Lab and acknowledge the valuable technical, administrative, and analytical contributions and expert advice from coworkers at Uppsala University and Uppsala University Hospital.
References
- 1. Eckel RH, Grundy SM, Zimmet PZ. The metabolic syndrome. Lancet 2005;365:1415‐1428. [DOI] [PubMed] [Google Scholar]
- 2. Singla P, Bardoloi A, Parkash AA. Metabolic effects of obesity: a review. World J Diabetes 2010;1:76‐88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Falagas ME, Kompoti M. Obesity and infection. Lancet Infect Dis 2006;6:438‐446. [DOI] [PubMed] [Google Scholar]
- 4. Caussy C, Wallet F, Laville M, Disse E. Obesity is associated with severe forms of COVID‐19. Obesity (Silver Spring) 2020;28;1175. doi: 10.1002/oby.22842 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Batterham RL, Cummings DE. Mechanisms of diabetes improvement following bariatric/metabolic surgery. Diabetes Care 2016;39:893‐901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Katsogiannos P, Kamble PG, Boersma GJ, et al. Early changes in adipose tissue morphology, gene expression, and metabolism after RYGB in patients with obesity and T2D. J Clin Endocrinol Metab 2019;104:2601‐2613. [DOI] [PubMed] [Google Scholar]
- 7. Katsogiannos P, Kamble PG, Wiklund U, et al. Rapid changes in neuroendocrine regulation may contribute to reversal of type 2 diabetes after gastric bypass surgery. Endocrine 2020;67:344‐353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Schauer PR, Bhatt DL, Kirwan JP, et al. Bariatric surgery versus intensive medical therapy for diabetes–3‐year outcomes. N Engl J Med 2014;370:2002‐2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Holst JJ, Madsbad S, Bojsen‐Moller KN, et al. Mechanisms in bariatric surgery: gut hormones, diabetes resolution, and weight loss. Surg Obes Relat Dis 2018;14:708‐714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Tilg H, Moschen AR. Adipocytokines: mediators linking adipose tissue, inflammation and immunity. Nat Rev Immunol 2006;6:772‐783. [DOI] [PubMed] [Google Scholar]
- 11. Mauer J, Chaurasia B, Goldau J, et al. Signaling by IL‐6 promotes alternative activation of macrophages to limit endotoxemia and obesity‐associated resistance to insulin. Nat Immunol 2014;15:423‐430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Rehman K, Akash MSH, Liaqat A, Kamal S, Qadir MI, Rasul A. Role of interleukin‐6 in development of insulin resistance and type 2 diabetes mellitus. Crit Rev Eukaryot Gene Expr 2017;27:229‐236. [DOI] [PubMed] [Google Scholar]
- 13. Miller GD, Nicklas BJ, Fernandez A. Serial changes in inflammatory biomarkers after Roux‐en‐Y gastric bypass surgery. Surg Obes Relat Dis 2011;7:618‐624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Lips MA, van Klinken JB, Pijl H, et al. Weight loss induced by very low calorie diet is associated with a more beneficial systemic inflammatory profile than by Roux‐en‐Y gastric bypass. Metabolism 2016;65:1614‐1620. [DOI] [PubMed] [Google Scholar]
- 15. Fain JN, Bahouth SW, Madan AK. TNFalpha release by the nonfat cells of human adipose tissue. Int J Obes Relat Metab Disord 2004;28:616‐622. [DOI] [PubMed] [Google Scholar]
- 16. Taniguchi T, Matsui H, Fujita T, et al. Structure and expression of a cloned cDNA for human interleukin‐2. Nature 1983;302:305‐310. [DOI] [PubMed] [Google Scholar]
- 17. Richard C, Wadowski M, Goruk S, Cameron L, Sharma AM, Field CJ. Individuals with obesity and type 2 diabetes have additional immune dysfunction compared with obese individuals who are metabolically healthy. BMJ Open Diabetes Res Care 2017;5:e000379. doi: 10.1136/bmjdrc-2016-000379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Alboni S, Cervia D, Sugama S, Conti B. Interleukin 18 in the CNS. J Neuroinflammation 2010;7:9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hung J, McQuillan BM, Chapman CM, Thompson PL, Beilby JP. Elevated interleukin‐18 levels are associated with the metabolic syndrome independent of obesity and insulin resistance. Arterioscler Thromb Vasc Biol 2005;25:1268‐1273. [DOI] [PubMed] [Google Scholar]
- 20. Zhuang H, Han J, Cheng L, Liu SL. A positive causal influence of IL‐18 levels on the risk of T2DM: a Mendelian randomization study. Front Genet 2019;10:295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Netea MG, Joosten LA, Lewis E, et al. Deficiency of interleukin‐18 in mice leads to hyperphagia, obesity and insulin resistance. Nat Med 2006;12:650‐656. [DOI] [PubMed] [Google Scholar]
- 22. Zilverschoon GR, Tack CJ, Joosten LA, Kullberg BJ, van der Meer JW, Netea MG. Interleukin‐18 resistance in patients with obesity and type 2 diabetes mellitus. Int J Obes (Lond) 2008;32:1407‐1414. [DOI] [PubMed] [Google Scholar]
- 23. Schernthaner GH, Kopp HP, Kriwanek S, et al. Effect of massive weight loss induced by bariatric surgery on serum levels of interleukin‐18 and monocyte‐chemoattractant‐protein‐1 in morbid obesity. Obes Surg 2006;16:709‐715. [DOI] [PubMed] [Google Scholar]
- 24. Subramaniam R, Aliakbarian H, Bhutta HY, Harris DA, Tavakkoli A, Sheu EG. Sleeve gastrectomy and Roux‐en‐Y gastric bypass attenuate pro‐inflammatory small intestinal cytokine signatures. Obes Surg 2019;29:3824‐3832. [DOI] [PubMed] [Google Scholar]
- 25. Krolopp JE, Thornton SM, Abbott MJ. IL‐15 activates the Jak3/STAT3 signaling pathway to mediate glucose uptake in skeletal muscle cells. Front Physiol 2016;7:626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Duan Y, Li F, Wang W, et al. Interleukin‐15 in obesity and metabolic dysfunction: current understanding and future perspectives. Obes Rev 2017;18:1147‐1158. [DOI] [PubMed] [Google Scholar]
- 27. Perez‐Lopez A, Valades D, Vazquez Martinez C, de Cos Blanco AI, Bujan J, Garcia‐Honduvilla N. Serum IL‐15 and IL‐15Rα levels are decreased in lean and obese physically active humans. Scand J Med Sci Sports 2018;28:1113‐1120. [DOI] [PubMed] [Google Scholar]
- 28. Shahi A, Afzali S, Salehi S, et al. IL‐27 and autoimmune rheumatologic diseases: the good, the bad, and the ugly. Int Immunopharmacol 2020;84:106538. doi: 10.1016/j.intimp.2020.106538 [DOI] [PubMed] [Google Scholar]
- 29. Li MS, Liu Z, Liu JQ, Zhu X, Liu Z, Bai XF. The Yin and Yang aspects of IL‐27 in induction of cancer‐specific T‐cell responses and immunotherapy. Immunotherapy 2015;7:191‐200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Nam H, Ferguson BS, Stephens JM, Morrison RF. Modulation of IL‐27 in adipocytes during inflammatory stress. Obesity (Silver Spring) 2016;24:157‐166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Li S, Shin HJ, Ding EL, van Dam RM. Adiponectin levels and risk of type 2 diabetes: a systematic review and meta‐analysis. JAMA 2009;302:179‐188. [DOI] [PubMed] [Google Scholar]
- 32. Closa D, Motoo Y, Iovanna JL. Pancreatitis‐associated protein: from a lectin to an anti‐inflammatory cytokine. World J Gastroenterol 2007;13:170‐174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Lv Y, Yang X, Huo Y, et al. Adenovirus‐mediated hepatocarcinoma‐intestine‐pancreas/pancreatitis‐associated protein suppresses dextran sulfate sodium‐induced acute ulcerative colitis in rats. Inflamm Bowel Dis 2012;18:1950‐1960. [DOI] [PubMed] [Google Scholar]
- 34. Natividad JM, Hayes CL, Motta JP, et al. Differential induction of antimicrobial REGIII by the intestinal microbiota and Bifidobacterium breve NCC2950. Appl Environ Microbiol 2013;79:7745‐7754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Paganelli FL, Luyer M, Hazelbag CM, et al. Roux‐Y gastric bypass and sleeve gastrectomy directly change gut microbiota composition independent of surgery type. Sci Rep 2019;9:10979. doi: 10.1038/s41598-019-47332-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Carbone F, Liberale L, Bonaventura A, et al. Regulation and function of extracellular nicotinamide phosphoribosyltransferase/visfatin. Compr Physiol 2017;7:603‐621. [DOI] [PubMed] [Google Scholar]
- 37. Gomez‐Ambrosi J, Catalan V, Ramirez B, et al. Plasma osteopontin levels and expression in adipose tissue are increased in obesity. J Clin Endocrinol Metab 2007;92:3719‐3727. [DOI] [PubMed] [Google Scholar]
- 38. Bowers RR, Lane MD. A role for bone morphogenetic protein‐4 in adipocyte development. Cell Cycle 2007;6:385‐389. [DOI] [PubMed] [Google Scholar]
- 39. Kim MK, Jang EH, Hong OK, et al. Changes in serum levels of bone morphogenic protein 4 and inflammatory cytokines after bariatric surgery in severely obese Korean patients with type 2 diabetes. Int J Endocrinol 2013;2013:681205. doi: 10.1155/2013/681205 [DOI] [PMC free article] [PubMed] [Google Scholar]


