Abstract
Background:
For individuals with hepatocellular carcinoma (HCC), type of insurance may be an important prognostic factor because of its impact on access to care. This study investigates the relationship between insurance type at diagnosis and stage-specific survival.
Methods:
This retrospective cohort analysis used data from 18 Surveillance, Epidemiology, and End Results Program cancer registries. Individuals ages 20 to 64 years, diagnosed with primary HCC between 2010 and 2015, with either private, Medicaid, or no insurance were eligible for cohort inclusion. Adjusted Cox proportional-hazards regression models were used to generate HRs and 95% confidence intervals (CI) for associations between insurance type at diagnosis and overall survival. All models were stratified by stage at diagnosis.
Results:
This analysis included 14,655 cases. Compared with privately insured individuals with the same stage of disease, those with Medicaid had a 43% (HR = 1.43; 95% CI, 1.13–1.32), 22% (HR = 1.22; 95% CI, 1.13–1.32), and 7% higher risk of death for localized, regional, and distant stage, respectively. Uninsured individuals had an 88% (HR = 1.88; 95% CI, 1.65–2.14), 59% (HR = 1.59; 95% CI, 1.41–1.80), and 35% (HR = 1.35; 95% CI, 1.18–1.55) higher risk of death for localized, regional, and distant stage, respectively, compared with privately insured individuals.
Conclusions:
Disparities in survival exist by the type of insurance that individuals with HCC have at the time of diagnosis.
Impact:
These findings support the need for additional research on access to and quality of cancer care for Medicaid and uninsured patients.
Introduction
Hepatocellular carcinoma (HCC) accounts for approximately 75% of all primary liver cancer diagnoses (1). Both the incidence and mortality for HCC are increasing in the United States, with the number of deaths from the disease doubling between 1999 and 2016 (2, 3). The prognosis for liver cancer is poor, with 5-year stage-specific survival probabilities of 37%, 13%, and 4% for localized, regional, and distant stage disease, respectively (4). Effective HCC treatment options depend on the extent of disease and underlying liver function and include surgery, radiotherapy, and systemic therapies. Advances in HCC treatment have led to improvements in overall survival (https://www.nccn.org/professionals/physician_gls/pdf/hepatobiliary.pdf; ref. 5).
Inequities in the timely access to HCC therapy has been proposed as a leading cause of persisting racial/ethnic and socioeconomic disparities in HCC survival. Specifically, prior studies using the population-based Surveillance, Epidemiology, and End Results (SEER) data have found that Blacks and Hispanics have substantially poorer survival than Whites and Asians, and minority patients are considerably less likely to receive HCC-directed therapy (6, 7). However, observed survival disparities are often attenuated, if not eliminated, when accounting for differences in stage and treatment (8).
The impact of insurance on the survival of individuals with HCC has not been extensively studied. Private health insurance, as compared with Medicaid or no insurance, has been associated with improved survival in other types of cancer, such as breast, colorectal, gallbladder, lung, and multiple myeloma (9–12). In HCC, disparities in receipt of surgical treatment have been observed by insurance type, with privately insured individuals consistently being more likely to receive HCC-directed surgery (13–16). However, the relationship between insurance and HCC survival is poorly understood. One study using data from the Tennessee cancer registry found that, compared with privately insured individuals, those with no insurance had a 2-fold higher risk of death while Medicaid-insured individuals had a 33% higher risk of death (15). However, adjustment for stage and treatment attenuated the relationship between insurance type and survival in their analysis. Another recent study used the SEER database to analyze and compare survival outcomes in individuals with Medicare/commercial insurance, Medicaid, and no insurance (17). The latter study reported that uninsured patients had significantly worse survival than their insured counterparts. However, the grouping of Medicare and privately insured individuals may have been problematic, as the demographic characteristics and ability to access care likely differs between the two groups (18, 19).
The objective of this study was to investigate stage-specific HCC survival by insurance type at diagnosis for a population-based cohort of working-age individuals.
Materials and Methods
Study design and source of data
We conducted a retrospective cohort study using data from SEER. We included data from 18 population-based cancer registries, covering approximately 28% of the population of the United States. SEER databases are linked to statewide and National Vital Statistics records, which allows SEER to document date of death and cause of death (COD). The study was exempt from Institutional Review Board oversight.
Study population
Males and females, aged 20–64 years, with a first primary diagnosis of HCC (defined as International Classification of Disease for Oncology, Third Edition topography code C22.0 and morphology codes 8170–8175), between 2010 and 2015 were eligible for inclusion (20). We restricted this analysis to pathologically confirmed cases with the above stated topography and morphology codes, a decision which was made to ensure our findings were comparable with existing HCC literature (21–23). In addition, individuals had to have known values for insurance type at the time of diagnosis, survival months, stage (localized, regional, distant), and race/ethnicity. Individuals, whose cancer diagnosis was solely confirmed by death certificate or autopsy report, were excluded.
Insurance type
Individuals were grouped into three categories: private insurance, any Medicaid, or no insurance, according to the insurance at diagnosis recorded in the SEER registry files. The SEER program specifically recommends against using their insurance variable for individuals older than 64 years due to the possibility of those individuals having concurrent insurance policies, (i.e. Medicare and private insurance); therefore, we did not include individuals older than 64 years of age in this study (20). We were also only interested in assessing insurance status and HCC survival in working-age individuals.
Survival time
Survival time, measured in months, was determined from SEER, using an individual’s date of diagnosis and vital status (alive, dead, or lost to follow-up) through 2015.
Survival for individuals lost to follow-up were censored at that time and those still alive at the end of 2015 were administratively censored (24).
Covariates of interest
All analyses were stratified using the SEER Historic Stage variable that classifies tumors into localized, regional, or distant (25). The SEER Historic Stage variable provides a consistent measure of stage across the time period of this study, as compared with the various versions of AJCC staging that are not always strictly comparable from year to year.
Sex and age at diagnosis were ascertained from SEER. Age was classified roughly into tertiles based on distribution of cancer case ages in the population. The SEER race and Hispanic origin recode variable was used to classify individuals into the following categories: Non-Hispanic White, Black, Non-Hispanic Asian/Pacific Islander, Non-Hispanic American Indian/Alaska Native, and Hispanic (any race). The SEER marital status variable was used to further classify individuals as being in a relationship (married or in a domestic relationship), not in a relationship, or relationship status unknown. Surgical treatment was split into three broad categories: no surgery, surgery but no liver transplantation (LT), and total hepatectomy and liver transplantation (26). The “total hepatectomy and liver transplantation” group contains regional and distant stage disease patients despite transplant being contraindicated in later stages because the extent of the disease may not be known prior to surgery (e.g., staging occurs after surgery has occurred). In addition, recent transplant eligibility criteria have been expanded to include those with regional stage disease and few risk factors (27). It is also important to note that individuals who are underinsured are typically not eligible for liver transplant. Finally, individuals were classified on the basis of county of residence (rural vs. urban), using the Rural-Urban Continuum Code 2013 variable in SEER (20).
Statistical analysis
We used descriptive statistics to assess demographic characteristics of the cohort. χ2 tests were used to determine differences in covariates by insurance status. We examined overall survival by insurance type, generating separate stage-specific Kaplan–Meier plots. Log-rank tests were used to test the null hypothesis of no difference in stage-specific survival by insurance type.
Cox proportional-hazards regression models were used to calculate HRs and 95% confidence intervals (CI) for the association between each variable in the model and the risk of death from any cause (overall survival). We confirmed that all assumptions required for conducting a Cox proportional hazards regression were met using graphical representations of Schoenfeld residuals using STATA (28). Variables were included in the multivariable adjusted model if significant at the P < 0.20 level in univariate analyses. Sex and age were included as a priori confounders in all adjusted models based on the prognostic importance of both variables (29). Models stratified by race/ethnicity and rurality were also conducted; however, due to the lack of statistically or clinically meaningful differences in the data, these stratified analyses are not presented here. In an effort to adjust for socioeconomic status (SES), we used a county-level poverty indicator in one of our multivariable models. Specifically, we adjusted the multivariable model for the percentage of individuals with less than a high-school education using data from the linked county attributes data provided in SEER. We also conducted a cancer-cause-specific survival analysis as a sensitivity analysis (30). Cases were censored if they died of any cause other than liver disease or HCC in this secondary analysis intended to account for the possible influence of comorbidities. Statistical significance was determined using two-tailed tests with P < 0.05. We used SEER*Stat version 8.3.5 (seer.cancer.gov/seerstat) and Stata/IC (v15.1; ref. 31) to conduct our analyses.
Results
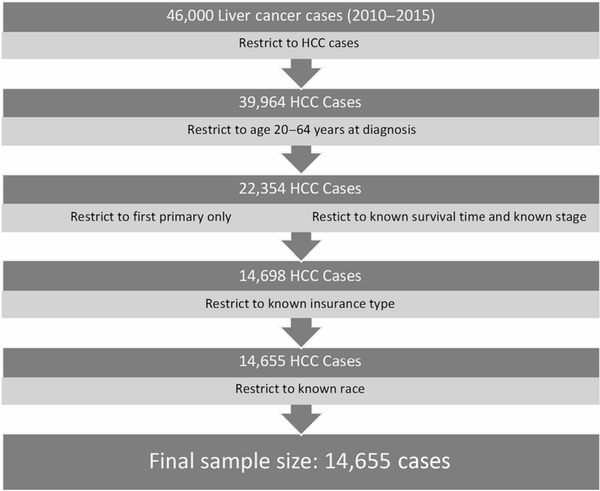
We identified 46,000 incident liver cancer cases diagnosed during the study period, of which 14,655 were determined to be eligible (Fig. 1). Of those included in our analysis, 58% (n = 8,447) were privately insured at the time of diagnosis, while 35% (n = 5,101) had Medicaid and 7% (n = 1,107) were uninsured (Table 1). Of cases included in the analysis, 83% of privately insured individuals were between the ages of 53–64 years while 76% and 74% of Medicaid and uninsured individuals were in the same age category, respectively. The majority of cases were male, with the highest proportion of males in the uninsured group (87%). Compared with privately insured individuals with HCC, those with Medicaid and those who were uninsured tended to be younger, were less likely to be Non-Hispanic White (NHW), and were less likely to be in a relationship. Uninsured individuals were also slightly more likely to live in rural counties.
Figure 1.
Summary of exclusions in the analysis. HCC, hepatocellular carcinoma.
Table 1.
Demographic characteristics of patients with HCC by insurance status.
| Privately insured (n= 8,447) n (%) | Any Medicaid (n= 5,101) n (%) | Uninsured (n= 1,107) n (%) | Total n (%) | P | |
|---|---|---|---|---|---|
| Sex | <0.001 | ||||
| Male | 7,046 (83) | 4,132 (81) | 965 (87) | 12,143 (83) | |
| Female | 1,401 (17) | 969 (19) | 142 (13) | 2,512 (17) | |
| Race/Ethnicity | <0.001 | ||||
| Non-Hispanic White | 4,397 (52) | 2,074 (41) | 495 (45) | 6,966 (48) | |
| Non-Hispanic Black | 1,197 (14) | 944 (19) | 240 (22) | 2,381 (16) | |
| Non-Hispanic A/PI | 1,253 (15) | 599 (12) | 148 (13) | 2,000 (14) | |
| Non-Hispanic AI/AN | —a | —a | —a | 197 (1) | |
| Hispanic (Any Race) | 1,513 (18) | 1,378 (27) | 220 (20) | 3,111 (21) | |
| Age at diagnosis | <0.001 | ||||
| 20–40 | 202 (2) | 111 (2) | 37 (3) | 350 (2) | |
| 41–52 | 1,225 (15) | 1,093 (21) | 256 (23) | 2,574 (18) | |
| 53–64 | 7,020 (83) | 3,897 (76) | 814 (74) | 11,731 (80) | |
| Marital status | <0.001 | ||||
| In a relationship | 4,903 (58) | 1,543 (30) | 360 (33) | 6,806 (46) | |
| Not in a relationship | 3,069 (36) | 3,359 (66) | 698 (63) | 7,126 (49) | |
| Unknown | 475 (6) | 199 (4) | 49 (4) | 723 (5) | |
| County of residence | <0.001 | ||||
| Urban | 7,806 (92) | 4,659 (91) | 973 (88) | 13,438 (92) | |
| Rural | 635 (8) | 425 (8) | 134 (12) | 1,194 (8) | |
| Unknown | 6 (0) | 17 (0) | 0 (0) | 23 (0) | |
| Stage | <0.001 | ||||
| Localized | 4,752 (56) | 2,601 (51) | 420 (38) | 7,773 (53) | |
| Regional | 2,534 (30) | 1,584 (31) | 389 (35) | 4,507 (31) | |
| Distant | 1,161 (14) | 916 (18) | 298 (27) | 2,375 (16) | |
| Therapy | <0.001 | ||||
| No surgery | 5,748 (68) | 4,148 (81) | 958 (87) | 10,854 (74) | |
| Surgery and no transplant | 2,005 (24) | 763 (15) | 136 (12) | 2,904 (20) | |
| Total hepatectomy and transplant | 694 (8) | 190 (4) | 13 (1) | 897 (6) |
Abbreviations: A/PI, Asian/Pacific Islander; AI/AN, American Indian/Alaska Native.
Data not shown due to small numbers.
Medicaid-insured and uninsured individuals were more likely to be diagnosed with distant stage disease and less likely to receive surgery than privately insured individuals. In accordance with the SEER data use agreement, demographic characteristics of American Indian/Alaska Native (AI/AN) cases are not presented; however, these individuals were included in subsequent survival analyses.
Association between insurance type and overall survival: localized HCC
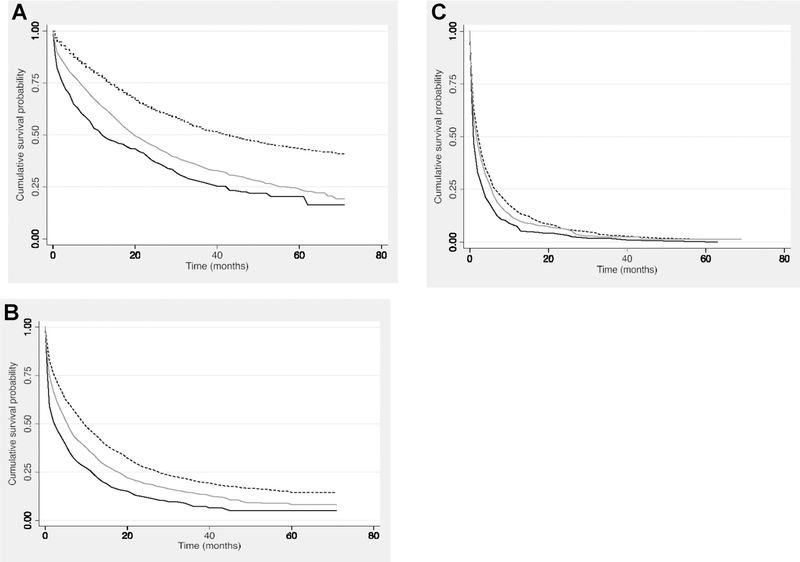
Among those diagnosed with localized stage HCC (n = 7,773), median overall survival was statistically significantly higher for privately insured individuals than for Medicaid-insured or uninsured individuals (overall survival in months: 18 vs 11 vs 8, respectively, P < 0.001; Fig. 2A). In unadjusted analyses, individuals with Medicaid had a 70% higher unadjusted risk of death than privately insured (HR = 1.70; 95% CI, 1.59–1.82) while the uninsured died at more than twice the rate of privately insured (HR = 2.25; 95% CI, 1.98–2.56; Table 2). In addition, individuals who were not in a relationship at the time of diagnosis and had localized disease had a 51% (HR = 1.51; 95% CI, 1.41–1.62) higher risk of death as compared with those in a relationship. Individuals who resided in a rural area had a 19% increased risk of death in localized cases compared with individuals living in an urban area (HR = 1.19; 95% CI, 1.05–1.33).
Figure 2.
A–C, Kaplan–Meier survival analysis of patients with HCC. Stratified by stage, privately insured patients are represented by the dotted black line, Medicaid-insured individuals are represented by the solid gray line, and uninsured individuals are represented by the solid black line. A, Localized: Uninsured individuals had the worst survival of those with localized disease (P < 0.001). B, Regional: Uninsured individuals also had the worst survival in regional stage disease (P < 0.001). C, Distant: Survival disparities by insurance status were less apparent in distant stage disease as signified by the intersection of the survival curves.
Table 2.
Univariate analysis of predictors of overall survival in patients with HCC stratified by stage.
| Local | Regional | Distant | |
|---|---|---|---|
| HR (95% CI) | HR (95% CI) | HR (95% CI) | |
| Insurance status at diagnosis | |||
| Privately insured | Ref | Ref | Ref |
| Any medicaid | 1.70 (1.59–1.82) | 1.33 (1.24–1.43) | 1.11 (1.01–1.21) |
| Uninsured | 2.25 (1.98–2.56) | 1.83 (1.62–2.06) | 1.38 (1.21–1.58) |
| Age | 1.00 (1.00–1.00) | 1.000 (0.99–1.00) | 1.000 (1.00–1.00) |
| 20–40 | Ref | Ref | Ref |
| 41–52 | 1.62 (1.20–2.20) | 1.13 (0.89–1.43) | 1.69 (1.30–2.21) |
| 53–64 | 1.60 (1.19–2.15) | 1.12 (0.89–1.41) | 1.61 (1.24–2.08) |
| Sex | |||
| Male | Ref | Ref | Ref |
| Female | 0.79 (0.79–0.87) | 0.79 (0.72–0.88) | 0.94 (0.84–1.07) |
| Race/Ethnicity | |||
| Non-Hispanic White | Ref | Ref | Ref |
| Non-Hispanic Black | 1.27 (1.16–1.40) | 1.20 (1.10–1.32) | 1.02 (0.90–1.14) |
| Non-Hispanic A/PI | 0.72 (0.64–0.80) | 0.95 (0.85–1.06) | 0.97 (0.85–1.11) |
| Non-Hispanic AI/AN | 0.92 (0.68–1.25) | 0.94 (0.71–1.24) | 1.13 (0.74–1.73) |
| Hispanic (Any Race) | 1.11 (1.03–1.21) | 0.95 (0.87–1.05) | 1.01 (0.89–1.13) |
| Relationship status | |||
| In a relationship | Ref | Ref | Ref |
| Not in a relationship | 1.51 (1.41–1.62) | 1.21 (1.13–1.30) | 1.13 (1.03–1.23) |
| Surgery | |||
| No surgery | Ref | Ref | Ref |
| Surgery and no transplant | 0.36 (0.33–0.40) | 0.37 (0.33–0.42) | 0.43 (0.33–0.55) |
| Total hepatectomy and transplant | 0.10 (0.08–0.13) | 0.09 (0.06–0.13) | 0.22 (0.05–0.88) |
| County of residence | |||
| Urban | Ref | Ref | Ref |
| Rural | 1.19 (1.05–1.33) | 1.19 (1.06–1.34) | 1.08 (0.92–1.26) |
Abbreviations: A/PI, Asian/Pacific Islander; AI/AN, American Indian/Alaska Native.
In the multivariable model restricted to individuals with localized disease (Table 3), those with Medicaid had higher risks of dying (adjusted HR = 1.42; 95% CI, 1.32–1.53) than those who were privately insured. Uninsured individuals had an elevated risk of death, as compared with privately insured individuals (adjusted HR = 1.86; 95% CI, 1.63–2.11). Black individuals with localized disease were found to have a 11% increased risk of death (adjusted HR = 1.11; 95% CI, 1.01–1.22), as compared with Non-Hispanic White individuals. Those with localized disease who were not in a relationship had an 18% greater risk of death (adjusted HR = 1.18; 95% CI, 1.09–1.27), as compared with those in a relationship.
Table 3.
Multivariate analysis: overall survival in patients with HCC stratified by stage.
| Localized | Regional | Distant | |
|---|---|---|---|
| HR (95% CI) | HR (95% CI) | HR (95% CI) | |
| Insurance status at diagnosis | |||
| Privately insured | Ref | Ref | Ref |
| Any Medicaid | 1.42 (1.32–1.53) | 1.22 (1.13–1.32) | 1.06 (0.97–1.17) |
| Uninsured | 1.86 (1.63–2.11) | 1.59 (1.40–1.80) | 1.33 (1.17–1.53) |
| Sex | |||
| Male | Ref | Ref | |
| Female | 0.78 (0.72–0.86) | 0.83 (0.75–0.92) | |
| Race/Ethnicity | |||
| Non-Hispanic White | Ref | Ref | |
| Non-Hispanic Black | 1.11 (1.01–1.22) | 1.08 (0.98–1.19) | |
| Non-Hispanic A/PI | 0.79 (0.71–0.89) | 1.01 (0.90–1.12) | |
| Non-Hispanic AI/AN | 0.83 (0.60–1.14) | 1.00 (0.75–1.35) | |
| Hispanic (Any Race) | 0.94 (0.86–1.02) | 0.91 (0.83–1.00) | |
| Relationship status | |||
| In a relationship | Ref | Ref | Ref |
| Not in a relationship | 1.18 (1.09–1.27) | 1.03 (0.96–1.11) | 1.09 (1.00–1.20) |
| Surgery | |||
| No surgery | Ref | Ref | Ref |
| Surgery and no transplant | 0.39 (0.36–0.43) | 0.39 (0.34–0.44) | 0.44 (0.35–0.56) |
| Total hepatectomy and transplant | 0.11 (0.09–0.15) | 0.10 (0.07–0.14) | 0.22 (0.05–0.90) |
| County of residence | |||
| Urban | Ref | Ref | |
| Rural | 1.09 (0.97–1.23) | 1.18 (1.05–1.34) |
Abbreviations: A/PI, Asian/Pacific Islander; AI/AN, American Indian/Alaska Native.
Association between insurance type and overall survival: regional HCC
For individuals with regional stage HCC, privately insured individuals had significantly higher median overall survival than Medicaid-insured or uninsured individuals (overall survival in months: 7 vs. 4 vs. 2, respectively, P < 0.001; Fig. 2B). In unadjusted analyses, those with Medicaid or who were uninsured had a 33% (HR = 1.33; 95% CI, 1.24–1.43) or 83% (HR = 1.83; 95% CI, 1.62–2.06) higher risk of dying than those with private insurance, respectively (Table 2). Regional stage cases residing in rural locations were 19% more likely to die than individuals living in urban areas (HR = 1.19; 95% CI, 1.06–1.34).
In our multivariable model restricted to cases with regional stage disease, individuals with Medicaid had a 22% (adjusted HR = 1.22; 95% CI, 1.13–1.32) increased risk of death while uninsured individuals had a 59% (adjusted HR = 1.59; 95% CI, 1.40–1.80) increased risk, as compared with privately insured cases. Rural residence was associated with the risk of death in regional disease (adjusted HR = 1.18; 95% CI, 1.05–1.34).
Association between insurance type and overall survival: distant HCC
In distant stage disease, median survival was not statistically different among the three insurance categories (overall survival in months: 2 vs. 2 vs. 1, respectively, P < 0.220; Fig. 2C). In the unadjusted analysis, Medicaid-insured or uninsured individuals had an 11% (HR = 1.11; 95% CI, 1.01–1.21) or 38% (HR = 1.38; 95% CI, 1.21–1.58) higher risk of dying than those with private insurance, respectively (Table 2). Single individuals had a 13% (HR = 1.13; 95% CI, 1.03–1.23) increased risk of death as compared with individuals in relationships with late-stage disease.
In the multivariable adjusted model restricted to cases with distant stage disease, Medicaid-insured patients had a slightly higher risk of death than patients with private insurance (adjusted HR = 1.06; 95% CI, 0.97–1.17), although this difference did not achieve statistical significance. The risk of death in uninsured individuals exceeded that in patients with private insurance (adjusted HR = 1.33; 95% CI, 1.17–1.53). Finally, race/ethnicity and county of residence were not included in the distant stage model because they were not significant in the unadjusted model.
Sensitivity analyses
Cause-specific survival was investigated as a secondary outcome and compared to the results of the overall survival analyses. The results using cause-specific survival did not vary from those observed in the overall survival models. We also conducted analyses in which we adjusted our models for SES at the county level, which did not yield significantly different HRs.
Discussion
In this large, national, population-based study, we found substantial evidence of disparities in stage-specific HCC survival by insurance type at diagnosis. Among individuals diagnosed with localized or regional stage HCC, those with Medicaid at the time of diagnosis were significantly more likely to die at any given time than privately insured individuals. Uninsured individuals also had a significantly higher risk of death than privately insured individuals across all stages.
Our results align with previous studies. In Tennessee, uninsured HCC cases were more than twice as likely (HR = 2.03; 95% CI 1.24–3.31) to die as those with private insurance (15). Similarly, in a population-based cancer registry sample (18 SEER Program registries), when compared with commercially or Medicare insured individuals, localized stage HCC patients with Medicaid had a 17% higher risk of death (HR = 1.17; 95% CI, 1.13–1.22) and those who were uninsured had a 39% higher risk of death (HR = 1.39; 95% CI, 1.29–1.50; ref. 18).
In our study, disparities in HCC survival by insurance type at diagnosis were particularly pronounced in earlier stage disease, where timely access to care has the highest likelihood to improve prognosis. Surgery is the primary treatment for localized disease and several studies suggest that delays in surgical treatment may worsen survival (13, 16). Similarly, delays in, or inability to, access to chemotherapy, radiation, and targeted therapy may all lead to poorer survival for regional or distant stage disease; however, as the baseline prognosis of HCC worsens with later stage, the potential impact of clinical intervention lessens, making insurance disparities less pronounced. Although we controlled for treatment received in our multivariable analyses, the association between insurance type and survival persisted, highlighting the importance of the quality and timing of treatment.
Importantly, the type of insurance that an individual has at the time of HCC diagnosis may be an indicator for health habits and chronically mismanaged or untreated comorbidities (32–36), which may also contribute to cause-specific survival. Medicaid is an income-based insurance program (https://www.medicaid.gov/medicaid/eligibility/index.html) and having Medicaid or no insurance are important markers of socioeconomic position. Further study is warranted to disentangle the mechanisms by which insurance impacts survival. The type of insurance an individual has may also impact their treatment options and treatment decisions. For example, certain treatments may not be covered by insurance or the portion that the patient must cover is cost prohibitive (37).
Although we did not have data on the Hepatitis C (HCV) status of individuals, prior studies provide evidence of disparities in HCV incidence (38, 39). Specifically, men and non-Hispanic white individuals have an increased incidence of HCV (38–40). Recent advances in HCV treatment have led to improved health outcomes as a result of higher rates of viral clearance, but treatments are expensive and the benefits of these new antiviral regimens are not accessible to everyone (41). These treatment disparities have been previously characterized in Hispanics and Medicaid-insured populations (38, 42). This is important because chronic HCV infection is a risk factor for HCC, suggesting that unmeasured differences in chronic HCV infection likely contribute to the disparities in HCC survival observed in our study.
We also observed disparities by race/ethnicity. Our results align with findings from two other recent studies in which Black patients with HCC had approximately a 15% increased risk of death as compared with Non-Hispanic White individuals (43, 44). We also noted that in localized disease, Asian/Pacific Islanders experienced a lower risk of death than Non-Hispanic White cases. However, there are conflicting reports regarding racial disparities in HCC and not all studies have found survival disparities among different racial/ethnic groups (45).
We also found that individuals living in rural areas had an 19% higher risk of death from regional stage HCC than individuals living in urban areas. Patients residing in rural areas may be less likely to access certain treatment options such as surgery (17). The type of cancer center at which a patient receives care impacts their survival (46). Patients residing in a rural area may not be able to access care at an NCI-Designated Cancer Center due to barriers such as lack of transportation (47) and the financial burdens that arise as a result of traveling for care (48).
Importantly, survival disparities persisted despite controlling for insurance type at diagnosis and receipt of treatment. This suggests that insurance alone is not enough to eliminate disparities and unequal socioeconomic factors impacting access to care, such as lack of time or adequate transportation to attend appointments, may still need to be addressed. Also, biologic differences in the aggressiveness of the tumor and/or in response to therapy may also not be addressed by improving access to care.
In our analysis, marital status was associated with an increased risk of death. Those who had localized disease and were not in a relationship had an 18% increased risk of death as compared with those in a relationship. In another study, being single was associated with a 23% increase in death for individuals diagnosed with the top ten most deadly cancers, of which liver cancer is one (49). Relationships may improve social support and reduce isolation, both of which may lead to better health outcomes (50). Our finding suggests the need to further investigate the role of social support networks in reducing insurance related cancer outcome disparities.
The results of this analysis need to be interpreted in the context of several limitations. While SEER provides a large, nationally representative database for cancer surveillance, the SEER program does not collect data on individual-level socioeconomic factors, health behaviors, comorbidities, or adherence to treatment. These factors likely differ between individuals with private, Medicaid, or no insurance. For example, the Healthy People 2020 initiative reported that people with private health insurance had an age-adjusted smoking rate of 12.6% (https://www.healthypeople.gov/2020/leading-health-indicators/2020-lhi-topics/Tobacco/data) Individuals with public health insurance had an age-adjusted smoking rate of 26.1%, which is notably higher than the prevalence in the population with private health insurance (https://www.healthypeople.gov/2020/leading-health-indicators/2020-lhi-topics/Tobacco/data). If we were able to control for the aforementioned factors, we would expect the HRs for the uninsured and Medicaid populations to decrease because some of the increased risk is likely explained by systematic differences in areas that affect survival among the different insurance categories. Similarly, residual confounding may exist despite our attempts to control for socioeconomic status and rural residence, as both of these factors were measured at the county level (51). Future studies using more granular socioeconomic information, such as census tract, or individual measurement will need to be conducted to better understand the associations that we document in this article.
In addition, the reliability and validity of the insurance type at diagnosis variable in SEER has not been well-studied. Misclassification in insurance type at diagnosis may occur because of inconsistencies across medical records, differences in the timing of abstraction of this variable, and retroactive coverage. In the United States, 46 states offer retroactive Medicaid coverage, which means that healthcare costs incurred in the 3 months prior to Medicaid enrollment are covered, as long as the individual was eligible for Medicaid during that time period (https://www.medicaid.gov/medicaid/eligibility/index.html). In addition, individuals may have more than one type of insurance and insurance status may change after a cancer diagnosis (19). The uninsured category likely includes a small minority who elect to be uninsured due to their ability to cover healthcare costs out-of-pocket when necessary.
In an analysis of insurance status in the Greater Bay Area Cancer Registry, Ellis and colleagues found agreement between 79% and 89% when examining categories based on type of insurance, and 96% to 99% agreement when using binary categories of any insurance or none (52). We conducted a sensitivity analysis, combining our Medicaid and privately insured group and comparing them to the uninsured. However, doing so attenuated the association between being uninsured and having either private or Medicaid insurance, likely because the Medicaid group was more similar in both their sociodemographic characteristics and survival to the uninsured. Thus, despite possible misclassification, we think that the most important information comes from presenting the survival probabilities separately for privately insured, Medicaid and uninsured individuals. Finally, we were unable to assess potential differences in the quality of private insurance and the access to care that the insurance affords an individual.
Despite these limitations, the magnitude of the disparities in HCC survival observed in our analyses warrant further study. Our findings illuminate the need to explore the potential impacts of insurance, race/ethnicity, and socioeconomic factors on HCC survival. As the incidence of HCC continues to rise (53), and Medicaid expansion debates continue, understanding the role of health insurance on cancer survival may help inform future policies surrounding Medicaid eligibility and coverage. Future efforts in this area should include a focus on assessing access to care and quality of care for Medicaid and uninsured individuals to potentially reduce cancer survival disparities.
Acknowledgments
This work was supported by internal funding from the University of New Mexico Comprehensive Cancer Center (to J.A. McDougall) and NCI contract HHSN261201800014I, task order HHSN26100001 (to C.L. Wiggins and A.L.W. Meisner).
Footnotes
Disclosure of Potential Conflicts of Interest
No potential conflicts of interest were disclosed.
References
- 1.Bosetti C, Turati F, La Vecchia C. Hepatocellular carcinoma epidemiology. Best Pract Res Clin Gastroenterol 2014;28:753–70. [DOI] [PubMed] [Google Scholar]
- 2.Ryerson AB, Eheman CR, Altekruse SF, Ward JW, Jemal A, Sherman RL, et al. Annual report to the nation on the status of cancer, 1975–2012, featuring the increasing incidence of liver cancer. Cancer 2016;122:1312–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tapper EB, Parikh ND. Mortality due to cirrhosis and liver cancer in the United States, 1999–2016: observational study. BMJ 2018;362:k2817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Islami F, Miller KD, Siegel RL, Fedewa SA, Ward EM, Jemal A. Disparities in liver cancer occurrence in the United States by race/ethnicity and state. CA Cancer J Clin 2017;67:273–89. [DOI] [PubMed] [Google Scholar]
- 5.Shah C, Mramba LK, Bishnoi R, Bejjanki H, Chhatrala HS, Chandana SR. Survival differences among patients with hepatocellular carcinoma based on the stage of disease and therapy received: pre and post sorafenib era. J Gastroint Oncol 2017;8:789–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Xu L, Kim Y, Spolverato G, Gani F, Pawlik TM. Racial disparities in treatment and survival of patients with hepatocellular carcinoma in the United States. Hepatobiliary Surg Nutr 2016;5:43–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Stewart SL, Kwong SL, Bowlus CL, Nguyen TT, Maxwell AE, Bastani R, et al. Racial/ethnic disparities in hepatocellular carcinoma treatment and survival in California, 1988–2012. World J Gastroenterol 2016;22:8584–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mathur AK, Osborne NH, Lynch RJ, Ghaferi AA, Dimick JB, Sonnenday CJ. Racial/ethnic disparities in access to care and survival for patients with early-stage hepatocellular carcinoma. Arch Surg 2010;145:1158–63. [DOI] [PubMed] [Google Scholar]
- 9.Niu X, Roche LM, Pawlish KS, Henry KA. Cancer survival disparities by health insurance status. Cancer Med 2013;2:403–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chen Z, Gao W, Pu L, Zhang L, Han G, Zhu Q, et al. Impact of insurance status on the survival of gallbladder cancer patients. Oncotarget 2017;8;51663–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gorey KM, Luginaah IN, Schwartz KL, Fung KY, Balagurusamy M, Bartfay E, et al. Increased racial differences on breast cancer care and survival in America: historical evidence consistent with a health insurance hypothesis, 1975–2001. Breast Cancer Res Treat 2009;113:595–600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Costa LJ, Brill IK, Brown EE. Impact of marital status, insurance status, income, and race/ethnicity on the survival of younger patients diagnosed with multiple myeloma in the United States. Cancer 2016;122:3183–90. [DOI] [PubMed] [Google Scholar]
- 13.Hoehn RS, Hanseman DJ, Jernigan PL, Wima K, Ertel AE, Abbott DE, et al. Disparities in care for patients with curable hepatocellular carcinoma. HPB (Oxford) 2015;17:747–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sarpel U, Suprun M, Sofianou A, Berger Y, Tedjasukmana A, Sekendiz Z, et al. Disentangling the effects of race and socioeconomic factors on liver transplantation rates for hepatocellular carcinoma. Clin Transplant 2016;30:714–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zaydfudim V, Whiteside MA, Griffin MR, Feurer ID, Wright JK, Pinson CW. Health insurance status affects staging and influences treatment strategies in patients with hepatocellular carcinoma. Ann Surg Oncol 2010;17:3104–11. [DOI] [PubMed] [Google Scholar]
- 16.Tan D, Yopp A, Beg MS, Gopal P, Singal AG. Meta-analysis: underutilisation and disparities of treatment among patients with hepatocellular carcinoma in the United States. Aliment Pharmacol Ther 2013;38:703–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wang J, Ha J, Lopez A, Bhuket T, Liu B, Wong RJ. Medicaid and uninsured hepatocellular carcinoma patients have more advanced tumor stage and are less likely to receive treatment. J Clin Gastroenterol 2018;52:437–43. [DOI] [PubMed] [Google Scholar]
- 18.Davis K, Schoen C, Doty M, Tenney K. Medicare versus private insurance: rhetoric and reality. Health Aff 2002;21:Suppl 1 Web Exclusives:W311–24. [DOI] [PubMed] [Google Scholar]
- 19.Edward RB, Hood E, Barnett CJ. Current population reports, health insurance coverage in the United States; 2017. Washington, DC: U.S. Government Printing Office; 2018. [Google Scholar]
- 20.Surveillance Epidemiology and End Results (SEER) Program. SEER 18 Regs Research Data, Nov 2017 Sub (1973–2015) þ Hurricane Katrina Impacted Louisiana Cases, Linked To County Attributes - Total U.S., 1969–2016 Counties, National Cancer Institute, DCCPS, Surveillance Research Program, released April 2018 based on the November 2017 submission. Available from: https://seer.cancer.gov/. [Google Scholar]
- 21.Yang D, Hanna DL, Usher J, LoCoco J, Chaudhari P, Lenz HJ, et al. Impact of sex on the survival of patients with hepatocellular carcinoma: a surveillance, epidemiology, and end results analysis. Cancer 2014;120:3707–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Franco RA, Fan Y, Jarosek S, Bae S, Galbraith J. Racial and geographic disparities in hepatocellular carcinoma outcomes. Am J Prev Med 2018;55:S40–S8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Brar G, McNeel T, McGlynn K, Graubard B, Floudas CS, Morelli MP, et al. Hepatocellular carcinoma (HCC) survival by etiology: a SEER-Medicare database analysis. J Clin Oncol 2019;37:201. [Google Scholar]
- 24.Mariotto AB, Noone AM, Howlader N, Cho H, Keel GE, Garshell J, et al. Cancer survival: an overview of measures, uses, and interpretation. J Natl Cancer Inst Monogr 2014;2014:145–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Young JL Jr, Roffers SD, Ries LAG, Fritz AG, Hurlbut AA, editors. SEER summary staging manual-2000: codes and coding instructions. Bethesda, MD: National Cancer Institute; 2001. [Google Scholar]
- 26.Peters NA, Javed AA, He J, Wolfgang CL, Weiss MJ. Association of socioeconomics, surgical therapy, and survival of early stage hepatocellular carcinoma. J Surg Res 2017;210:253–60. [DOI] [PubMed] [Google Scholar]
- 27.Viveiros A, Zoller H, Finkenstedt A. Hepatocellular carcinoma: when is liver transplantation oncologically futile? Transl Gastroenterol Hepatol 2017;2:63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schoenfeld D Partial residuals for the proportional hazards regression model. Biometrika 1982;69:239–41. [Google Scholar]
- 29.Ha J, Yan M, Aguilar M, Bhuket T, Tana MM, Liu B, et al. Race/ethnicity-specific disparities in cancer incidence, burden of disease, and overall survival among patients with hepatocellular carcinoma in the United States. Cancer 2016;122: 2512–23. [DOI] [PubMed] [Google Scholar]
- 30.Howlader N, Ries LA, Mariotto AB, Reichman ME, Ruhl J, Cronin KA. Improved estimates of cancer-specific survival rates from population-based data. J Natl Cancer Inst 2010;102:1584–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.StataCorp. Stata Statistical Software: Release 15. College Station, TX: StataCorp LLC; 2017. [Google Scholar]
- 32.Zafar SY. Financial toxicity of cancer care: it’s time to intervene. J Natl Cancer Inst 2016;108:e1–4. [DOI] [PubMed] [Google Scholar]
- 33.Sinclair AH, Schymura MJ, Boscoe FP, Yung RL, Chen K, Roohan P, et al. Measuring colorectal cancer care quality for the publicly insured in New York State. Cancer Med 2012;1:363–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Parikh-Patel A, Morris CR, Kizer KW. Disparities in quality of cancer care: the role of health insurance and population demographics. Medicine 2017;96:e9125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hassett MJ, Schymura MJ, Chen K, Boscoe FP, Gesten FC, Schrag D. Variation in breast cancer care quality in New York and California based on race/ethnicity and Medicaid enrollment. Cancer 2016;122:420–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Bittoni MA, Wexler R, Spees CK, Clinton SK, Taylor CA. Lack of private health insurance is associated with higher mortality from cancer and other chronic diseases, poor diet quality, and inflammatory biomarkers in the United States. Prev Med 2015;81:420–6. [DOI] [PubMed] [Google Scholar]
- 37.Siminoff LA, Ross L. Access and equity to cancer care in the USA: a review and assessment. Postgrad Med J 2005;81:674–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thrift AP, El-Serag HB, Kanwal F. Global epidemiology and burden of HCV infection and HCV-related disease. Nat Rev Gastroenterol Hepatol 2017;14: 122–32. [DOI] [PubMed] [Google Scholar]
- 39.Cummins CA, Erlyana E, Fisher DG, Reynolds GL. Hepatitis C infection among Hispanics in California. J Addict Dis 2015;34:263–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wu T, Konyn PG, Cattaneo AW, Saab S. New face of hepatitis C. Dig Dis Sci 2019;64:1782–8. [DOI] [PubMed] [Google Scholar]
- 41.Younossi Z, Gordon SC, Ahmed A, Dieterich D, Saab S, Beckerman R. Treating Medicaid patients with hepatitis C: clinical and economic impact. Am J Manag Care 2017;23:107–12. [PubMed] [Google Scholar]
- 42.Wong RJ, Jain MK, Therapondos G, Shiffman ML, Kshirsagar O, Clark C, et al. Race/ethnicity and insurance status disparities in access to direct acting antivirals for hepatitis C virus treatment. Am J Gastroenterol 2018; 113:1329–38. [DOI] [PubMed] [Google Scholar]
- 43.Li J, Hansen BE, Peppelenbosch MP, De Man RA, Pan Q, Sprengers D. Factors associated with ethnical disparity in overall survival for patients with hepatocellular carcinoma. Oncotarget 2017;8:15193–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hoehn RS, Hanseman DJ, Wima K, Ertel AE, Paquette IM, Abbott DE, et al. Does race affect management and survival in hepatocellular carcinoma in the United States? Surgery 2015;158:1244–51. [DOI] [PubMed] [Google Scholar]
- 45.Aparo S, Goel S, Lin D, Ohri N, Schwartz JM, Lo Y, et al. Survival analysis of Hispanics in a cohort of patients with hepatocellular carcinoma. Cancer 2014; 120:3683–90. [DOI] [PubMed] [Google Scholar]
- 46.Wolfson JA, Sun CL, Wyatt LP, Hurria A, Bhatia S. Impact of care at comprehensive cancer centers on outcome: results from a population-based study. Cancer 2015;121:3885–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Onega T, Duell EJ, Shi X, Wang D, Demidenko E, Goodman D. Geographic access to cancer care in the U.S. Cancer 2008;112:909–18. [DOI] [PubMed] [Google Scholar]
- 48.Zahnd WE, Davis MM, Rotter JS, Vanderpool RC, Perry CK, Shannon J, et al. Rural-urban differences in financial burden among cancer survivors: an analysis of a nationally representative survey. Support Care Cancer 2019; 27:4779–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Walker GV, Grant SR, Guadagnolo BA, Hoffman KE, Smith BD, Koshy M, et al. Disparities in stage at diagnosis, treatment, and survival in nonelderly adult patients with cancer according to insurance status. J Clin Oncol 2014; 32:3118–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Aizer AA, Chen M-H, McCarthy EP, Mendu ML, Koo S, Wilhite TJ, et al. Marital status and survival in patients with cancer. J Clin Oncol 2013;31: 3869–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Singh GK, Williams SD, Siahpush M, Mulhollen A. Socioeconomic, rural-urban, and racial inequalities in US cancer mortality: part I-all cancers and lung cancer and part II-colorectal, prostate, breast, and cervical cancers. J Cancer Epidemiol 2011;2011:107497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ellis L CA, Spiegel D, Ladabaum U, Haile R, Gomez SL. Understanding the limitations of cancer registry insurance data—implications for policy. JAMA Oncol 2018;4:1433–34. [DOI] [PubMed] [Google Scholar]
- 53.Fitzmorris P, Shoreibah M, Anand BS, Singal AK. Management of hepatocellular carcinoma. J Cancer Res Clin Oncol 2015;141:861–76. [DOI] [PubMed] [Google Scholar]