Abstract
A 10-year-old boy presented with continuous reports of pain located to the left knee. Imaging revealed a sclerotic process in the left distal femur, and biopsies were consistent with chondroblastic osteosarcoma. As part of standard treatment the patient underwent neoadjuvant chemotherapy followed by limb sparring surgery and adjuvant chemotherapy. The entire tumour was excised and femoral bone reconstruction was performed with a double barrel free vascularised fibular graft. Bone mineral density (BMD) can be decreased in childhood survivors of cancer. The patient was followed for 7 years with dual-energy X-ray absorptiometry scans in order to assess BMD and graft adaption. Despite two accidental fractures to the graft region local and global BMD underwent an overall increase. Approximately 7 years after tumour resection the patient had a global Z-score of 0.2, which is considered within normal range.
Keywords: cancer intervention, paediatric oncology, orthopaedics, osteoporosis
Background
Osteosarcoma is a relatively rare and malignant tumour of the bone. It has a predilection for the metaphyseal region of the long bones with 40% of cases found in the femoral bone.1 Osteosarcomas have a bimodal age distribution with a peak in adolescence and in the second decade of life. It can be classified based on histology, but common for almost every subtype is the treatment, which consists of a combination of neoadjuvant chemotherapy, surgical resection and adjuvant chemotherapy. The current treatment regimen is associated with an average 5-year survival of 70%.1 The aim of surgical intervention is wide tumour-excision, which often leaves a considerable bony defect.1 Previously, extremity osteosarcoma often resulted in amputation as primary treatment. Today more than 80% of cases can be treated with limb sparing surgery, for example, bone reconstruction with a free vascularised fibular graft (FVFG).2 The FVFG has arterial supply from the peroneal artery and prepossesses great versatility. The graft can be used as single or double barrel FVFG (D-FVFG). Double barrel is superior to single barrel as it strengthens the reconstruction and integration. Larger defects can only be single barrel reconstructed as the length of the donor fibular bone has limitations.3 It also has a high bony union rate, but unlike endoprosthetic reconstruction, it delays weight-bearing, and graft hypertrophy is time consuming.3
Dual-energy X-ray absorptiometry (DXA) scan may be used to quantitate bone mineral density (BMD) as an indicator of the mechanical strength of the bone graft. DXA is the preferred method for measurements of BMD in children due to both its availability as well as the low radiation exposure.4 The terms osteopenia and osteoporosis are not widely accepted as diagnostic concepts within paediatric patients, but are frequently used. Children suffering from childhood cancers such as osteosarcomas are considered at potential risk of low BMD later in life.4 Z-score is recommended in the evaluation of BMD in children as this is a reference value for normal age-matched individuals. T-score is based on healthy young adults and is not recommended for patients younger than 20 years.4 Bachrach and Gordon argue for values of bone mineral content (BMC) and BMD 2 SD below normal to be classified as low for age.4
The reason for a general finding of lower BMD in young adult survivors of childhood cancer is probably a combination of multiple factors related to the disease as well as these happening during a critical time period for the attainment of peak bone mass. Factors suspected to affect BMD could be: type of cancer and treatment received, inactivity (extremity offloading) during disease, time since cancer disease, physical activity and age at the time of DXA.5 6 The chemotherapeutic drug, methotrexate is suspected to contribute to BMD decrease because it suppresses osteoblast activity and stimulates osteoclast recruitment.7 This results in increased bone resorption. Also cisplatin and doxorubicin have been found to contribute negatively to osteogenesis in animal studies.8 9 Decreased weight-bearing has been proposed as a contributor to BMD shortfall.10
In this case, we followed a patient for 7 years after treatment for osteosarcoma of the distal femoral bone after tumour resection and reconstruction with D-FVFG. Several BMD measurements were performed as a marker of bone graft adaptation and consolidation. The continuity of measurements in the same patient in a critical growth period gives a unique insight in bone reconstruction following osteosarcoma treatment.
Case presentation
A 10-year-old boy presented with continuous reports of pain located to the left knee (figure 1). Five months prior he was hit by a car on his left side without suffering any fractures or major trauma. Diagnostic imaging revealed a sclerotic process in the distal femoral bone and mild spiculation (figure 2A). Open biopsy under general anaesthesia was consistent with chondroblastic osteosarcoma.
Figure 1.

Timeline. MAP-regimen, high-dose methotrexate, doxorubicin and cisplatin; LISS, Less invasive stabilization system; D-FVFG, double barrel free vascularised fibular graft; DXA, dual-energy X-ray absorptiometry; LCP, locking compression plate.
Figure 2.

(A) Anterior–posterior radiograph showing a sclerotic process with periosteal spiculation in the distal lateral part of the femoral bone. (B) Diagnostic MRI showing a bone tumour measuring 55 mm in diameter with soft tissue extension but no involvement of neurovascular structures.
Treatment
The patient was started in a standard regimen for osteosarcoma treatment and received 3 months of neoadjuvant chemotherapy consisting of a combination of high-dose methotrexate, doxorubicin and cisplatin (MAP regimen). Except for a few cases of stomatitis, low haemoglobin and neutropenia, he responded well to treatment. An MRI was performed and showed the tumour to be confined to the distal metaphysis with a 2.5 cm free margin to the epiphysial plate and no involvement of neurovascular structures (figure 2B). A limb and joint sparring procedure with bone reconstruction by D-FVFG was planned and performed as a team approach of orthopaedic and plastic surgeons.
The tumour was resected en bloc with a proximal 15 mm and a distal 3 mm free margin preserving the epiphysial plate, but leaving an 85 mm large bone defect. The D-FVFG was harvested from the ipsilateral fibular bone measuring 22.5 cm before being osteotomised at the centre and positioned with the osteotomy proximal in the bone defect (figure 3). The medial genicular vessels were used as recipient vessels for the fibular bone graft and the graft insert stabilised with plate osteosynthesis (less invasive stabilization system (LISS)) bridging the distal femur epiphysis on both the medial and the lateral side of the femoral bone. Postoperative pathology report showed narrow margins of resection, but above 95% necrosis consistent with a good prognosis of survival.1 Tumour material was consistent with osteosarcoma with a chondroblastic component. Postoperative radiographs of the graft showed good alignment and reconstruction/osteosynthesis in both the anterior–posterior and lateral plane (figure 4).
Figure 3.
(A) Internal fixation of the femoral bone with a less invasive stabilization system (LISS) plate prior to excision of the tumour. (B) The free fibula graft measuring 225 mm. (C) The double-barrel free vascularised graft.
Figure 4.
Radiographs 2 days postoperative. (A) Lateral view and (B) anterior–posterior view of the left femoral bone showing correct positioning of the doublebarrel free vascularised fibular graft and preservation of the epiphysial plate.
Outcome and follow-up
Overall, the patient had an uncomplicated postoperative course after primary surgery. Chemotherapy was resumed (MAP regimen) and continued 3 months postoperatively with no signs of remaining disease. The patient continued the planned follow-up regimen.
Weight-bearing on the operated leg was not allowed for the first 3 months. The patient was fully weight-bearing 5.5 months postoperatively and had a knee motion range from 0 to 110 degrees. Radiographs showed progressive bone healing in both the distal and proximal part of the D-FVFG. Approximately half a year after tumour resection the patient suffered an epiphysiolysis of the left distal tibia as he was playing soccer against given advice.
A year after tumour resection no signs of tumour recurrence were suspected clinically or on radiographs. Fibular grafts need compression in order to prevent decalcification and to increase bone mass and strength (Wolff’s law).11 Therefore the distal screws (august 2013) and LISS plate (janaury 2014) were removed. a bone biopsy was performed (august 2013) which showed no signs of local tumour recurrence. In february 2014, a relatively mild torque load caused a femur fracture, at the osteosynthesis of the proximal end of the D-FVFG. Conservative treatment proved insufficient and the patient underwent fracture stabilising surgery with a locking compression plate (LCP) on the distal femur. The LCP was removed after a year, but only 2 months later, corresponding to almost 3 years after tumour resection, he suffered another distal femur fracture after an accidental torque while descending a small slope. This time the fracture was located to the proximal part of the D-FVFG itself with no relation to the previous fracture. The patient underwent another fracture stabilising osteosynthesis with a LISS, which was removed approximately 1 year later. The fractures could have been caused by microarchitectural and geometric fragility, however no more fractures have occurred. There has been no sign of recurrence of osteosarcoma.
Only reports are leg length discrepancy leading to a functional scoliosis (figure 5A). Leg length discrepancy was first described as 2 cm 7 months after tumour resection, increasing to approximately 6 cm 5 years after primary surgery (figure 5A). In 2019, he underwent successful bone-transport leg-lengthening surgery by use of a femoral nail with magnetic activation control. The leg correction has almost realigned the spinal scoliosis (figure 5B). Currently, there is a leg length discrepancy of 2 cm compensated for by foot posts. An additional leg-lengthening procedure with the still in place surgical magnetic nail is under consideration.
Figure 5.

(A) Full body dual-energy X-ray absorptiometry (DXA)-scan 60 months after tumour resection showing signs of compensatory lumbar scoliosis due to leg length discrepancy. Boxes 1 and 2 (height 32 cm, width 12.5 cm) are positioned from the knee joint and 32 cm proximal, just below trochanter minor. They are used to estimate net bone mineral density for the left and right leg as seen in table 3 and figure 10. (B) Full body DXA-scan 81 months after tumour resection showing an almost realigned spinal scoliosis after a leg-lengthening procedure.
Following tumour resection the patient underwent successive DXA-scans after the reconstructive bone graft surgery with the aim of measuring BMD changes in the bone graft area. The patient was positioned supine for the DXA scans, which were performed on a Prodigy DXA scanner (GE Healthcare) (figure 6). A total body scan was performed at 10, 13, 60 and 81 months after tumour resection for measurement of Z-score, total body bone mass and body composition (lean mass, fat mass, body mass index). The distal femoral bone of both legs was scanned separately with an orto knee scan programme. The scanned leg was fixed in a positioner keeping the first toe pointing vertically. Rice bags were placed on both sides of the thigh as tissue equivalent material. A scan area from the knee joint line to approximately 3 cm proximal to the bone graft was scanned on the left leg at each follow-up (scan window: length 21.2 cm×width 18 cm). The same scan window was used for the right leg as a control. In the enCORE software V.16 we defined three customised templates for measurements of BMD in three regions of interest (ROIs) of the left distal femoral bone (figure 7) and these were positioned on each scan follow-up together with a copy of the bone border as a position guide.12 ROI 1 defined the global area of the distal femoral bone and was also used for measurement of the BMD of the right distal femoral bone as a control. Metal artefact removal was applied in the software and was manually corrected on all scans. ROI 2 was positioned over the lateral arm of the D-FVFG and ROI 3 covered the medial femoral condyle (figure 7).
Figure 6.

(A, B) Position of patient during dual-energy X-ray absorptiometry scans of the left distal femur.
Figure 7.
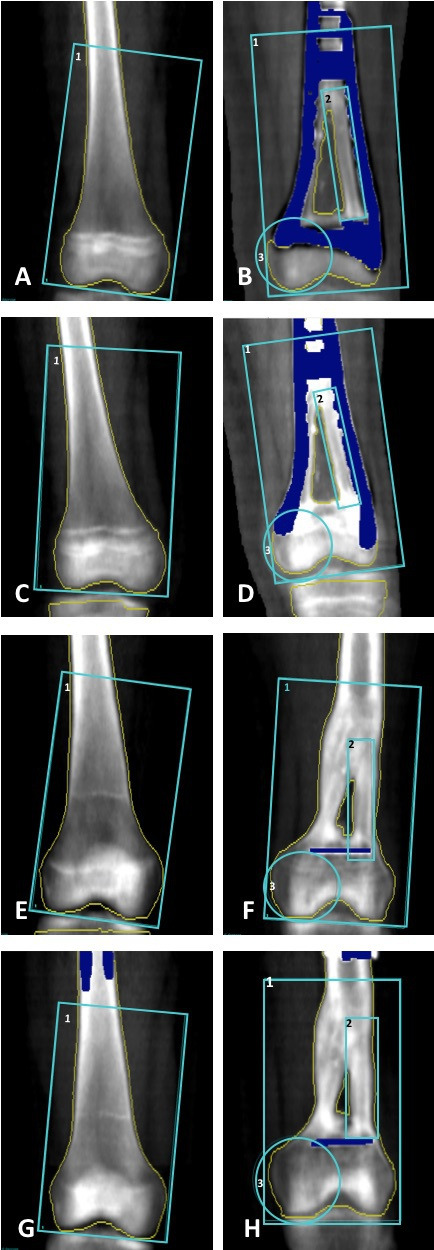
Dual-energy X-ray absorptiometry scans of the right and left distal femoral bone. For the right distal femoral bone (A, C, E and G) the blue box outlines the region of interest (ROI) 1 (height 8.73 cm, width 16.32 cm) which is positioned with the lower horizontal line along the most distal part of the femoral condyles. For the left distal femoral bone the blue boxes outline ROI 1 (height 16.35 cm, width 9.53 cm), ROI 2 (height 8.16 cm, width 1.49 cm) of the lateral half of the double barrel free vascularised fibular graft positioned with the lower horizontal line along the distal end of the graft. ROI 3 (height 5.28 cm, width 4.08 cm) is positioned as a circle including the medial femoral condyle (non-bone transplant). The yellow line marks the bone border. The dark blue colour marks metal artefacts not included in the bone mineral density estimation. (A) and (B) 10 months, (C) and (D) 13 months, (E) and (F) 60 months and (G) and (H) 81 months.
The BMD measurements (g/cm2) from the left distal femur, right distal femur and full body scans are shown in tables 1–3 as well as figures 8 and 9, respectively.
Table 1.
Net BMD (g/cm2) and BMC (g) of each ROI and BMD% and BMC % increase since baseline (10 months after tumour resection). Left distal femoral bone
| Months after tumour resection | Net BMD of ROI 1 (BMC) |
BMD (BMC) % increase | Net BMD of ROI 2 (BMC) | BMD (BMC) % increase | Net BMD of ROI 3 (BMC) | BMD (BMC) % increase |
| 10 | 0.566 (17.50) | 0 (0) | 0.587 (5.70) | 0 (0) | 0.540 (5.16) | 0 (0) |
| 13 | 0.743 (27.05) | 31.3 (54.6) | 0.775 (6.76) | 32.0 (18.6) | 0.701 (8.28) | 29.8 (60.5) |
| 60 | 1.189 (88.95) | 110.1 (408.3) | 1.385 (14.85) | 135.9 (160.5) | 0.975 (15.39) | 80.6 (198.3) |
| 81 | 1.208 (93.41) | 113.4 (433.8) | 1.383 (20.46) | 135.6 (258.9) | 0.912 (18.33) | 68.9 (255.2) |
BMC, bone mineral content; BMD, bone mineral density; ROI, Regions of interest.
Table 2.
Net BMD (g/cm2) and BMC (g) of each ROI and BMD % and BMC % increase since baseline (10 months after tumour resection). Right distal femoral bone
| Months after tumour resection | Net BMD of ROI 1 (BMC) | BMD (BMC) % increase |
| 10 | 0.635 (41.90) | 0 (0) |
| 13 | 0.743 (50.42) | 17.0 (20.3) |
| 60 | 0.993 (81.72) | 56.4 (95.0) |
| 81 | 0.927 (75.47) | 46.0 (80.1) |
BMC, bone mineral content; BMD, bone mineral density; ROI, regions of interest.
Table 3.
Total body dual-energy X-ray absorptiometry scan: Net BMD (g/cm2) and BMC (g) of the right leg, net BMD and BMC of the left leg and net BMD and BMC of the total body and % increase since baseline (10 months after tumour resection)
| Months after tumour resection | Net BMD right leg (BMC) | BMD (BMC) % increase | Net BMD left leg (BMC) | BMD (BMC)% increase | Net BMD total body (BMC) | BMD (BMC) % increase |
| 10 | 0.723 (213.8) | 0 (0) | 0.554 (120.8) | 0 (0) | 0.736 (1259.1) | 0 (0) |
| 13 | 0.801 (260.8) | 10.8 (22.0) | 0.634 (144.5) | 14.4 (19.6) | 0.774 (1341.4) | 5.2 (6.5) |
| 60 | 1.109 (427.2) | 53.4 (99.8) | 0.999 (320.1) | 80.3 (165.0) | 1.008 (2117.4) | 37.0 (68.2) |
| 81 | 1.222 (434.8) | 69.0(103.4) | 1.324 (399.6) | 139.0 (230.8) | 1.191 (2316.3) | 61.8 (84.0) |
BMC, bone mineral content; BMD, bone mineral density.
Figure 8.

BMD % change of the left distal femoral bone in ROI 1 (blue), ROI 2 (orange) and ROI 3 (green) in months after tumour resection. BMD, bone mineral density; ROI, regions of interest.
Figure 9.
BMD % change in ROI 1 (blue) of the unaffected right distal femoral bone. BMD % change measured on total body scans: right leg (orange), left leg (grey) and the full body (yellow) in months after tumour resection. BMD, bone mineral density; ROI, regions of interest.
A BMD increase was seen in all investigated regions of the D-FVFG areas with a relative increase in ROI 2 of 135.6% at 81 months after index bone graft surgery compared with baseline (10 months after tumour resection) (table 1). Figure 10 shows increasing overall Z-score with a value of 0.2 at 81 months after tumour resection.
Figure 10.

Total Z-score in months after tumour resection. DXA, dual-energy X-ray absorptiometry.
Discussion
Several studies have found decreased BMD in survivors of childhood cancers. The results tend to be based on haematological cancers, but some studies include data from childhood survivors of solid cancers. Comparison of the studies is however challenging due to variation in type of treatment, degree of immobilisation, race, area of measurement, time since treatment as well as age at BMD measurements as some studies assess adult patients, some children and others a combination.6 13–16
In a longitudinal study Ruza et al measured BMD by DXA at clinical remission in 38 patients with osteosarcoma and 25 patients with Ewing’s sarcoma.13 Mean age at BMD measurement was 20.65 years (SD 4.42) and 19.13 (SD 4.20), respectively. Mean age at diagnosis was 13.8 and 12.1 years with a mean treatment duration of 12 and 11 months. BMD measurements were made of the lumbar spine (L2−L4) and the neck of the right femur, the latter when unaffected. Results showed that 33.9% of patients had a BMD in the range of −1 and −2 SD, and 9.7% were below −2 SD in the lumbar region. In the femoral region, 25.4% of patients were in the range of −1 and −2 SD, while 17.5% were under −2 SD.
Holzer et al studied long-term survivors of highly malignant osteosarcomas. Osteopenic values were defined as an overall Z-score between −1.0 and −2.5. Osteoporotic values were defined as an overall Z-score below −2.5. The mean age was 31 (±4.2 SD) years at the time of DXA scan and the mean follow-up was 16±2.2 years. Out of 48 patients (22 men), 31 (64.5%) patients (21 categorised as osteopenic and 10 as osteoporotic) had decreased BMD in either the lumbar spine or the proximal femur.15
Pirker-Frühauf et al retrospectively studied 18 patients with Ewing’s sarcoma and 25 patients with osteosarcoma at a mean 7–8 years after their diagnosis. BMD measurements were performed with DXA. Patient mean age at follow-up was 27±9 years and 58% had lower BMD (Z-score below −1), than normal for young adults, in at least one measuring area (lumbar region and femoral region).14
The patient in the presented case was 17 years old at the last BMD measurement, which revealed a total Z-score of 0.2. According to the criteria set out by Bachrach and Gordon the patient does therefore not fulfil the criteria for a BMD of low for age and never did during the 7 years of follow-up.4 Positively, we found an overall increase in the both global and local BMD and BMC compared with baseline indicating general bone formation during a critical period and graft integration and hypertrophy in spite of two femoral fractures with osteosynthesis in the graft area and several periods with reduced mobility and reduced weight-bearing. Between 60 and 81 months after tumour resection the patient had a leg-lengthening procedure which also changed the mechanical axis towards more normal alignment. This could have caused a change on the load balance and a net gain of BMC on the left femur and net loss/stress shielding on the right femur. Studies have found both statistically significant results as well as tendencies towards physical activity having a positive impact on BMD.17 18 Exactly how much impact immobility has had for our case patient, or whether he has a higher risk of general osteoporosis compared with age-matched individuals is unknown. Future studies evaluating BMD in patients with osteosarcoma and risk factors for low BMD with immobilisation in childhood, and possible bone anti-resorption treatment interventions, are needed. Currently the case patient is well with no signs of recurrence of disease. He has full mobility and participates in physical sports activities such as soccer training.
Learning points.
Dual-energy X-ray absorptiometry (DXA) may be used to evaluate the bone mineral density (BMD) and bone mineral content of the local bone graft and systemically in survivors of osteosarcomas.
DXA may be used to evaluate the risk of adverse effects after sarcoma treatment such as osteoporosis.
BMD shortfall in childhood cancer survivors may be correlated to time since treatment.
Acknowledgments
We would like to thank Inger Krog-Mikkelsen for performing the DXA scans.
Footnotes
Contributors: BJK and PHJ performed the surgery and collected relevant perioperative data. PHJ and MS collected the data and analysed the data. ARJ drafted the first manuscript and analysed the data. All authors revised the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient consent for publication: Obtained.
Provenance and peer review: Not commissioned; externally peer reviewed.
References
- 1.Moore DD, Luu HH. Osteosarcoma. Cancer Treat Res 2014;162:65–92. 10.1007/978-3-319-07323-1_4 [DOI] [PubMed] [Google Scholar]
- 2.Bacci G, Ferrari S, Bertoni F, et al. Long-Term outcome for patients with nonmetastatic osteosarcoma of the extremity treated at the Istituto ortopedico rizzoli according to the Istituto ortopedico rizzoli/osteosarcoma-2 protocol: an updated report. J Clin Oncol 2000;18:4016–27. 10.1200/JCO.2000.18.24.4016 [DOI] [PubMed] [Google Scholar]
- 3.Bumbasirevic M, Stevanovic M, Bumbasirevic V, et al. Free vascularised fibular grafts in orthopaedics. Int Orthop 2014;38:1277–82. 10.1007/s00264-014-2281-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bachrach LK, Gordon CM. Bone densitometry in children and adolescents. Pediatrics 2016;138:e20162398. 10.1542/peds.2016-2398 [DOI] [PubMed] [Google Scholar]
- 5.Kaste SC, Ahn H, Liu T, et al. Bone mineral density deficits in pediatric patients treated for sarcoma. Pediatr Blood Cancer 2008;50:1032–8. 10.1002/pbc.21281 [DOI] [PubMed] [Google Scholar]
- 6.Kelly J, Damron T, Grant W, et al. Cross-sectional study of bone mineral density in adult survivors of solid pediatric cancers. J Pediatr Hematol Oncol 2005;27:248–53. 10.1097/01.mph.0000162526.77400.78 [DOI] [PubMed] [Google Scholar]
- 7.Ahn JH, Cho WH, Lee JA, et al. Bone mineral density change during adjuvant chemotherapy in pediatric osteosarcoma. Ann Pediatr Endocrinol Metab 2015;20:150–4. 10.6065/apem.2015.20.3.150 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rana T, Chakrabarti A, Freeman M, et al. Doxorubicin-Mediated bone loss in breast cancer bone metastases is driven by an interplay between oxidative stress and induction of TGFβ. PLoS One 2013;8:e78043. 10.1371/journal.pone.0078043 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stine KC, Wahl EC, Liu L, et al. Cisplatin inhibits bone healing during distraction osteogenesis. J Orthop Res 2014;32:464–70. 10.1002/jor.22527 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wasilewski-Masker K, Kaste SC, Hudson MM, et al. Bone mineral density deficits in survivors of childhood cancer: long-term follow-up guidelines and review of the literature. Pediatrics 2008;121:e705–13. 10.1542/peds.2007-1396 [DOI] [PubMed] [Google Scholar]
- 11.de Boer HH, Wood MB. Bone changes in the vascularised fibular graft. J Bone Joint Surg Br 1989;71:374–8. 10.1302/0301-620X.71B3.2722923 [DOI] [PubMed] [Google Scholar]
- 12.Veng C, Jørgensen PH, Krog-Mikkelsen I, et al. Measurement of bone mineral density as an efficacy marker in denosumab treatment of giant cell tumour of bone. BMJ Case Rep 2017;2017. 10.1136/bcr-2017-220369. [Epub ahead of print: 23 Oct 2017]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ruza E, Sierrasesúmaga L, Azcona C, et al. Bone mineral density and bone metabolism in children treated for bone sarcomas. Pediatr Res 2006;59:866–71. 10.1203/01.pdr.0000219129.12960.c2 [DOI] [PubMed] [Google Scholar]
- 14.Pirker-Frühauf UM, Friesenbichler J, Urban E-C, et al. Osteoporosis in children and young adults: a late effect after chemotherapy for bone sarcoma. Clin Orthop Relat Res 2012;470:2874–85. 10.1007/s11999-012-2448-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Holzer G, Krepler P, Koschat MA, et al. Bone mineral density in long-term survivors of highly malignant osteosarcoma. J Bone Joint Surg Br 2003;85:231–7. 10.1302/0301-620X.85B2.13257 [DOI] [PubMed] [Google Scholar]
- 16.Henderson RC, Madsen CD, Davis C, et al. Bone density in survivors of childhood malignancies. J Pediatr Hematol Oncol 1996;18:367–71. 10.1097/00043426-199611000-00006 [DOI] [PubMed] [Google Scholar]
- 17.Hind K, Burrows M. Weight-bearing exercise and bone mineral accrual in children and adolescents: a review of controlled trials. Bone 2007;40:14–27. 10.1016/j.bone.2006.07.006 [DOI] [PubMed] [Google Scholar]
- 18.Tlacuilo-Parra A, Morales-Zambrano R, Tostado-Rabago N, et al. Inactivity is a risk factor for low bone mineral density among haemophilic children. Br J Haematol 2008;140:562–7. 10.1111/j.1365-2141.2007.06972.x [DOI] [PubMed] [Google Scholar]





