Abstract
Mood disorder is the leading intrinsic risk factor for suicidal ideation. Questioning any potency of mood-stabilizers, the monovalent cation lithium still holds the throne in medical psychiatric treatment. Furthermore, lithium`s anti-aggressive and suicide-preventive capacity in clinical practice is well established. But little is still known about trace lithium distribution and any associated metabolic effects in the human body. We applied a new technique (neutron-induced coincidence method “NIK”) utilizing the 6Li(n,α)3H reaction for the position sensitive, 3D spatially resolved detection of lithium traces in post-mortem human brain tissue in suicide versus control. NIK allowed, for the first time in lithium research, to collect a three dimensional high resolution map of the regional trace lithium content in the non lithium-medicated human brain. The results show an anisotropic distribution of lithium, thus indicating a homeostatic regulation under physiological conditions as a remarkable link to essentiality. In contrast to suicide we could empirically prove significantly higher endogenous lithium concentrations in white compared to gray matter as a general trend in non-suicidal individuals and lower lithium concentrations in emotion-modulating regions in suicide.
Subject terms: Biophysics, Neuroscience, Medical research
Introduction
Every 20–40 s a person dies due to suicide, proving a stable phenomenon with a worldwide average of nearly 1 Million suicides per year. The rate of attempted suicides and—thus possibly severe consequences—may be up to 20 times higher, which means one attempt every 1–2 s in average worldwide1,2 and is expected to increase in the current SARS-CoV-2-pandemic. As suicide is indubitable a complex multifactorial event and based on interactions in between genetic, environmental and psychosocial factors3, one of the leading risk factors for suicidal ideation is psychiatric disease4. CAVANAGH et al. showed in a systematic review of psychological autopsy studies, that 9 of 10 suicide victims were prior diagnosed with a mental disorder, mostly depression5. Mood disorders in general come with up to 10 times higher lifetime suicide risk compared to the non-psychiatric population and the risk in bipolar disorder is up to 30 times higher6.
Since numerous decades now, lithium salts represent not only the gold standard in acute and long-term maintenance management of mania and depressive disorders7–9. Lithium`s anti-aggressive and suicide-preventive capacity in clinical practice is well established10–12. Numerous ecological studies indicate similar effects of nutritional lithium13,14, with ranges up to several mg Li+/d15.
SHEARD et al. reported for the first time in the beginning of the 1970s, that lithium in pharmacological doses is accompanied by a significant reduction in aggressive behavior16–18. Furthermore lithium`s antisuicidal potency seems undeniable. A potential link between lithium-medication and antisuicidal effects was first questioned in 197219 and statistically proven in numerous studies in the following decades20–22. Lately there has been increasing evidence for the hypothesis, that naturally occurring lithium may have a modulating protective effect on suicide rates too. As early as 1970 conducted epidemiological studies already questioned a potential impact of naturally occurring lithium e.g. in tap water on mental health23,24. Two decades later SCHRAUZER proved a significant correlation between tap water lithium content and the prevalence of suicides in the catchment area25. In the last decade there has been a broader international approach reevaluating the results by conducting similar studies26–33, further confirming the postulate of trace lithium’s antisuicidal capacity.
Despite the largely banal fact, that lithium is ingested daily by food and acts as a permanent resident in the human body in analytically relevant concentrations, little is known about possible beneficial effects on human health or any physiological function. While blood serum concentrations close to toxic limits in psychiatric pharmacotherapy show lithium`s well proven remarkable efficiency in equilibration of mood cycles, although based on only sparse amounts of experimental data in literature34, any impact of subclinical or trace serum concentrations is still in doubt. Tap or mineral water, grains and vegetables are main sources for daily lithium intake. Tap water lithium concentrations can range in between few µg/l and several mg/l worldwide15. Considering usual therapeutic medical doses in bipolar disorder treatment, the lithium intake via oral administration of lithium salts in psychiatric practice like lithium carbonate or lithium orotate ranges within approx. 80 mg Li+/oral unit and 250 mg Li+/d. This dose is associated with well proven therapeutic efficacy, but contrasted by a narrow therapeutic range, a potential risk of intoxication and the necessity of close drug monitoring during lithium treatment. On the other hand, the daily dietary Li+-intake can be estimated to be in the order of up to several mg Li+-ions. This means, that depending on the environment, the trace lithium uptake ranges a factor 100 lower or even less compared to daily pharmacological doses. Therefore, a total trace lithium human body content of several mg would be in the same range as essential trace elements like cobalt or selenium. However, in spite of lithium’s comparable much lower atomic weight, under physiological conditions the number of reactive Li+-ions in the human body can thus be estimated up to 20 times higher than cobalt, up to 4 times higher than selenium and a factor 103 lower than the alkaline earth metal magnesium (Mg++), a gap which in turn is fully compensated in pharmacotherapy.
Being marked as the most ignoble of precious metals, with only 1 valence electron the Li+-cation is highly reactive. Lithium´s charge density is similar to calcium (Ca++) and in spite of it`s much lower weight lithium`s ionic radius is almost identical to Mg++35. It is well known that the ions of sodium, potassium, calcium and magnesium dominate neurotransmission and signal transduction processes. Lithium is the only monovalent alkali cation with the ability to replace sodium in stabilizing the nerve’s resting membrane potential. It also influences potassium regulated enzymatic processes36. Bringing into discussion a more fundamental sight of the microscopic neurotransmission, which goes much beyond the Hodgkin–Huxley-Model37, it sounds therefore not surprising, that lithium with its remarkable electrochemical properties might influence enzymatic processes linked to neuropsychiatric disorders as a sufficient competitor38–40. But despite being one of the most researched elements with proven essentiality in mammals15, it is still not fully clear, how lithium may change metabolic systems in favour of protective emotional stabilisation. Being the smallest and lightest solid element, its ability to carry out processes in biological organisms may only be explained at molecular levels and thus make any research into the biological profile of the “magic ion” comparably difficult35.
There is still a lack in studies on any physiological relevance of trace lithium levels in human brain. Though currently there is still no consistent detailed picture how lithium acts inside the human body. In this context of particular interest is, how endogenous lithium is distributed in the brain, a question to which, in lack of a suitable method, an answer has hitherto been reserved.
Measurement of the lithium concentration with established methods like atomic absorption spectroscopy (AAS) or inductively coupled plasma mass spectrometry (ICP-MS) need macroscopic samples disintegrated in advance and do not permit detailed anatomical information. In vivo nuclear magnetic resonance (NMR) measurements41 enable a three dimensional picture of lithium distributed in the brain in pharmacotherapy, but due to the method-associated high detection limit are not sensitive enough to measure the physiological endogenous lithium distribution. We have succeeded in developing a new technique (neutron-induced coincidence method “NIK”)42 for position sensitive, three dimensional (3D) spatially resolved detection of lithium traces in post-mortem tissue, allowing for the first time in lithium research, to collect a three dimensional high resolution map of the regional trace lithium content in the non lithium-medicated human brain.
Results
139 samples of different brain regions in bilateral setting of 3 deceased individuals I–III (Table 1) were taken at autopsy at the “Institut für Rechtsmedizin” Munich, Germany (IRM).
Table 1.
Brain sample collective, nbrain = 3, nsamples = 139.
| Brain sample collective | ||||||||
|---|---|---|---|---|---|---|---|---|
| Age/sex | Cause of death | Anamnestic data | Suicide (verified) | Lithium medication in death | Timespan death—autopsy | Samples | ||
| n | Regions | |||||||
| I | 31/m | Pulmonary embolisma (deep vein thrombosis) | No psychiatric illness | No | No | < 48 h | n = 43 |
– Frontal lobe – Temporal lobe – Occipital lobe (case II, III) – Brain stem – Basal ganglia (striatum, pallidum) – Limbic system (hippocampus, amygdala, thalamus, cingulate cortex, corpus callosum, fornix region) |
| II | 43/m | Strangulationa | Depression, aggressive impulse control disorder | Yes | No | < 48 h | n = 46 | |
| III | 46/m | Barbiturate Intoxicationa |
Amyotrophic Lateral sclerosis Barbiturate prescription |
No | Yes | < 48 h | n = 50 | |
aNo pathomorphological signs of relevant brain edema, no macroscopic signs of degradation.
Mean brain tissue concentration levels
The overall obtained concentration level (lithium area density λ, spatially resolved lithium concentration cwetLinat) of trace lithium (Case I, II) is in the range of low ppb (λ: < 0.01–1.18 pg/cm2; cwetLinat: 0.01–1.16 ng/g). The obtained concentration level of lithium in Case III (lithium medication) is in the medium range of ppb (λ: 68–231 pg/cm2, cwetLinat: < 32–189 ng/g). In case I the range of spatially resolved lithium concentration cwetLinat is based on n = 29 out of n = 43. The results are shown in Table 2.
Table 2.
Average brain lithium levels.
| Average concentration levels | ||||||
|---|---|---|---|---|---|---|
| Lithium area density λ (pg/cm2) | Normalized spatially resolved lithium cwetLinat (ppb resp. ng/g) | |||||
| Minimum | Median/Mean | Maximum | Minimum | Median/Mean | Maximum | |
| I natural death (n = 43) | ~ 0.04 | 0.38/0.46 | 1.18 | 0.02* | 0.19*/0.23* | 0.65* |
| II Suicide (n = 46) | ~ 0.03 | 0.37/0.36 | 0.99 | 0.03 | 0.32/0.31 | 1.16 |
| III Lithium medication (n = 50) | 68 | 140.3 | 231 | 32 | 73.8 | 189 |
*Based on sample analysis of n = 29 of n = 43.
Case-specific brain lithium distribution
Case I: no lithium medication; natural death, n = 43
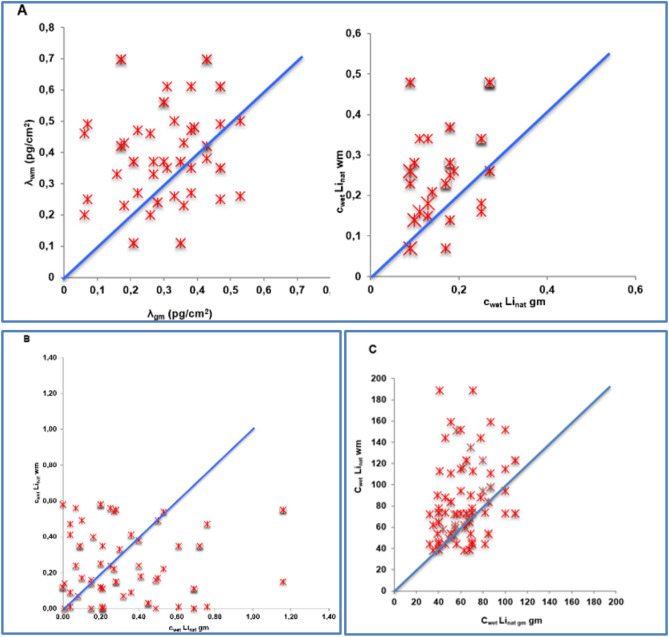
In all 43 samples the lithium area density λ was measured. In correlation with the results of the layer thickness measurement available for n = 29 samples a high significant correlation between λ and cwetLinat (r = 0.809, p < 0.001) proves, that λ is sufficient for comparative assessments (Fig. 1) and the further analysis can be based on the results for λ in addition to the evaluation of cwetLinat (n = 29).
Figure 1.
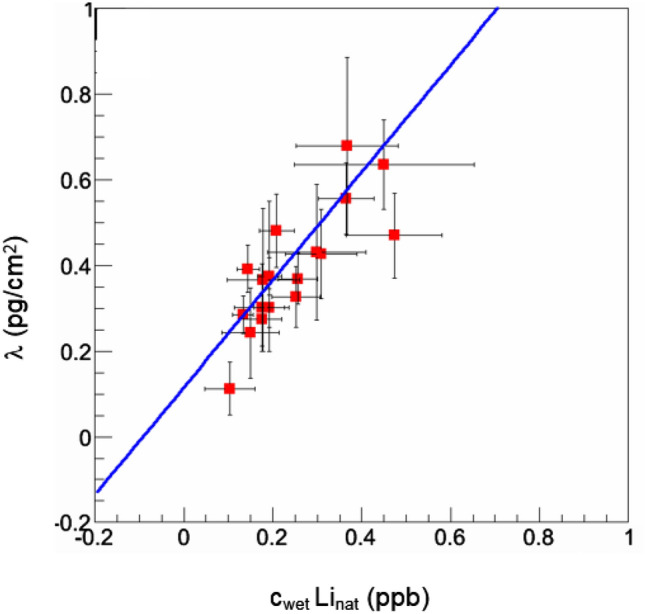
Case I: Correlation diagram cwetLinat and λ. The offset of the correlation fit indicates the level of the systematic uncertainty for the transformation from an areal density λ to a concentration in the native tissue.
The histogram of λ shows the anisotropic distribution in the examined brain regions by resembling a Gaussian distribution (Fig. 2A). Furthermore lithium concentration of λ (n = 43) proves a ratio well > 1 (1.34 ± 0.035) for white matter versus gray matter cwm/cgm. This can be further confirmed with a ratio well > 1 for cwetLinat (n = 29) correlating cwm/cgm (Fig. 3A). Compared with other brain regions the lithium area density λ in the white matter of the thalamic region is with 0.99 ± 0.19 pg/cm2 significantly higher and shows the overall highest concentration in case I. On the other hand, the gray matter proportion of the subgenual part of the cingulate gyrus, the so-called Brodmann Area 25 (BA25) shows with λ: 0.64 ± 0.15 pg/cm2 and 0.45 ± 0.27 ppb the highest lithium concentration of the gray matter measurement. Except for the proportions of gray matter in the region “Amygdala” (0.6 pg/cm2 ± 0.15) comparatively high concentrations were only found in the white matter proportion. The regional results for case I are shown in Table 3.
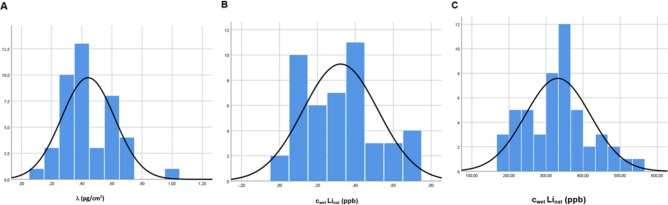
Figure 2.
Histogram of lithium distribution in the brain* (A) case I; (B) case II; (C) case III. *Case A: lithium area density λ; Case II, III: spatially resolved lithium concentration cwetLinat.
Figure 3.
Correlation of lithium distribution in white versus gray matter (A) case I; (B) case II; (C) case III. *Case A: lithium area density λ; Case I–III: spatially resolved lithium concentration cwetLinat.
Table 3.
Regional lithium concentration, case I–III.
| Regional Lithium concentration, case I–III | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Case I, n = 43 | Case II, n = 46 | Case III, n = 50 | ||||||||||
| Region | Gray matter | White matter | Gray matter | White matter | Gray matter | White matter | ||||||
| λ (pg/cm2) |
cwetLinat (ppb) |
λ (pg/cm2) |
cwetLinat (ppb) |
λ (pg/cm2) |
cwetLinat (ppb) |
λ (pg/cm2) |
cwetLinat (ppb) |
λ (pg/cm2) |
cwetLinat (ppb) |
λ (pg/cm2) |
cwetLinat (ppb) |
|
| Frontal lobe | 0.43 | 0.31 | 0.33 | −\− | 0.45 | 0.40 | 0.53 | 0.38 | 68 | 44 | 77 | 50 |
| 0.16 | −\− | 0.35 | 0.40 | 0.28 | 0.240.24 | 94 | 59 | 88 | 58 | |||
| 0.54 | 0.51 | 71 | 49 | 142 | 96 | |||||||
| 201 | 96 | |||||||||||
| Temporal lobe (Insula region) | 0.38 | 0.19 | −\− | −\− | 0.48 | 0.65 | −\− | −\− | 128 | 63 | 192 | 69 |
| 0.22 | 0.14 | 189 | 68 | |||||||||
| Occipital lobe | −\− | −\− | −\− | −\− | 0.24 | 0.20 | 0.25 | 0.25 | 78 | 54 | 111 | 62 |
| 0.05 | 0.05 | 80 | 55 | 130 | 72 | |||||||
| 94 | 56 | 185 | 151 | |||||||||
| 187 | 114 | |||||||||||
| Brain stem | 0.27 | −\− | 0.37 | −\− | 0.52 | 0.56 | −\− | −\− | −\− | −\− | −\− | −\− |
| Basal ganglia (Striatum, Pallidum) | 0.38 | 0.19 | 0.56 | 0.37 | 0.22 | 0.14 | 0.45 | 0.33 | 128 | 63 | 187 | 98 |
| 0.30 | 0.18 | 0.65 | −\− | 0.33 | 0.30 | 0.58 | 0.40 | 171 | 87 | 217 | 94 | |
| 201 | 8080 | |||||||||||
| Limbic system | ||||||||||||
| Brodman Area 25 | 0.64 | 0.45 | 0.11 | 0.10 | 0.10 | 0.09 | −\− | −\− | 96 | 64 | 105 | 59 |
| 0.20 | 0.22 | 115 | 69 | 173 | 135 | |||||||
| 0.39 | 0,38 | |||||||||||
| Cingulate cortex | 0.37 | 0.18 | −\− | −\− | Rostral: 0.22 | Rostral: 0.21 | −\− | −\− | Rostral: 201 | Rostral: 80 | −\− | −\− |
| Medial: 0.69 | Medial: 0.72 | Caudal: 200 | Caudal: 93 | |||||||||
| Dorsal: 0.60 | Dorsal: 0.45 | |||||||||||
| 0.61 | 0.66 | |||||||||||
| 0.58 | 0.60 | |||||||||||
| Caudal: 0.40 | Caudal: 0.31 | |||||||||||
| 0.48 | 0.41 | |||||||||||
| Corpus callosum | −\− | −\− | Rostral: 0.68 | Rostral: 0.37 | −\− | −\− | Rostral: 0.40 | Rostral: 0.35 | −\− | −\− | Rostral: 231 | Rostral: 123 |
| Medial: 0.43 | Medial: 0.30 | Medial: 0.34 | Medial: 0.35 | Medial: 201 | Medial: 96 | |||||||
| Dorsal: ~ 0.03 | Dorsal: ~ 0.03 | Dorsal: 142 | 96 | |||||||||
| Caudal: 0.20 | Caudal: 0.18 | |||||||||||
| Thalamus | 0.47 | 0.27 | 0.99 | −\− | Rostral: 0.10 | Rostral: 0.09 | rostral: 0.40 | Rostral: 0.35 | 122 | 62 | 211 | 116 |
| Medial: 0.16 | Medial: 0.08 | Medial: 0.15 | Medial: 0.07 | |||||||||
| Dorsal: 0.60 | Dorsal: 0.45 | Dorsal: 0.03 | Dorsal: 0.03 | |||||||||
| Fornix region | 0.30 | 0.18 |
0.56 0.65 |
0.37 | 0.69 | 0.72 | 0.34 | 0.35 | ||||
| −\− | 0.52 | 0.56 | ||||||||||
| Hippocampus | Rostral: 0.28 | Rostral: 0.13 | Rostral: 0.24 | Rostral: 0.15 | Rostral: 0.63 | Rostral: 0.37 | Medial: 0.10 | Medial: 0.07 | Rostral: 89 | Rostral: 55 | Rostral: 110 | Rostral: 59 |
| 0.27 | 0.18 | 0.33 | 0.25 | Medial: 0.43 | Medial: 0.32 | 82 | 42 | Dorsal: 192 | Medial: 52 | |||
| 0.39 | 0.14 | Dorsal: 0.38 | Dorsal: −\− | Dorsal: 0.47 | Dorsal: 0.31 | medial: 94 | medial: 54 | medial: 95 | dorsal: 69 | |||
| Dorsal: 0.43 | Dorsal: −\− | 0.48 | 0.21 | dorsal: 189 | dorsal: 68 | dorsal: 192 | ||||||
| 0.60 | −\− | |||||||||||
| 0.63 | −\− | |||||||||||
| Amygdala | Rostral: 0.28 | Rostral: 0.13 | Rostral: 0.24 | Rostral: 0.15 | 0.47 | 0.31 | −\− | −\− | Rostral: 89 | Rostral: 55 | Rostral: 110 | Rostral: 59 |
| 0.30 | 0.14 | 0.37 | 0.26 | Medial:94 | Medial: 54 | Medial: 95 | Medial:52 | |||||
| 0.39 | 0.19 | Dorsal: 0.48 | Dorsal: 0.21 | Dorsal: 189 | Dorsal: 68 | Dorsal: 192 | Dorsal: 69 | |||||
| Dorsal: 0.60 | Dorsal: −\− | |||||||||||
| 0.63 | −\− | |||||||||||
Case II: no lithium medication; suicide, n = 46
For all 46 samples λ and cwetLinat were measured. In case II the histogram of cwetLinat shows an anisotropic distribution as well, also resembling a Gaussian distribution (Fig. 2B).
Likewise in case I in case II cwm versus cgm of cwetLinat was correlated. Almost diametrically opposed to Case I the correlation diagram of cwetLinat in Case II shows a ratio well < 1 (0.71 ± 0.02) of cwm/cgm (Fig. 3B). Here significantly more entries are below the bisector, indicating that there is significantly less lithium in white matter than in gray matter. Comparably high white matter concentrations show the rostral corpus callosum and the basal ganglia. Highest concentrations in gray matter are found in the medial and dorsal cingulate cortex, the fornix region and temporal lobe. The lowest values for white matter are measured in the dorsal corpus callosum, the occipital lobe, thalamic and hippocampal regions. The lowest values measured in the gray matter were in the anterior thalamic region and BA25. Comparably low concentrations were also found in the rostral cingulate cortex. The regional results for Case II are shown in Table 3.
Case III: lithium medication; n = 50
For all 50 samples λ and cwetLinat were measured. In case III the histogram of cwetLinat confirms the anisotropic distribution in turn by also resembling a Gaussian distribution (Fig. 2C). The correlation of cwetLinat cwm versus cgm showed as in case I with 1.44 ± 0.01 a ratio well > 1 (Fig. 3C). Beside high concentrations found in the occipital lobe, the regional highest lithium concentration of the white matter section are located at BA25, the rostral section of corpus callosum and the thalamic region. The concentration in the overall gray matter area was comparably homogeneous, only basal ganglia and cingulate gyrus showed slightly higher concentrations. The regional results for case III are shown in Table 3.
Cross-case evaluation
With regard to any significance of trace lithium brain content for suicide a correlation of the results in case I and case II seems relevant. First, in case II the overall concentration is slightly lower than in case I. In average 17.5% of the case II samples less than 0,2 ppb lithium was measured, in case I only 5%. The correlation diagram (Fig. 4) of lithium area density λI to λII shows an anti-correlation with r = − 0.436 (p < 0.035). The anticorrelation string shows the fit of the measurement results.
Figure 4.
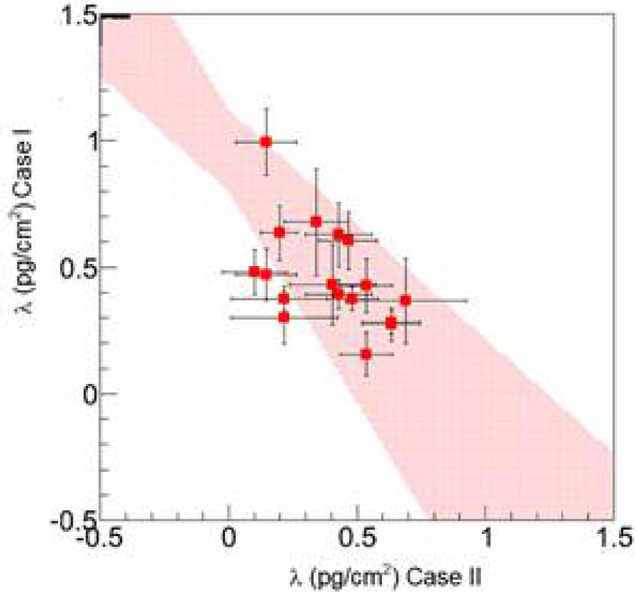
Correlation diagram of lithium area density λI versus λII; Pink colour anticorrelation string.
Discussion
The found average trace lithium concentration for case I and II was in the range of low ppb. According to MOORE et al., based on spectrometric measurements the ratio of the trace lithium content in tissue to blood plasma is in the range of ctissue/cplasma 0.50–0.9747. Our results agree very well with that, taking also in account the found tissue distribution ratio and due to hemolytic changes under postautoptic conditions the necessity for whole blood determination. On the other hand, compared with case I and II, the average lithium concentration in case III with known lithium medication was only a factor > 200 higher, thus lower than therapeutic blood levels (4–8 ppm) and so lower than the aforementioned ratio ctissue/cplasma. An inference that the lithium medication was discontinued shortly before death therefore would seem plausible. Taking into account, that abruptly stopping lithium medication without gradual titration triggers suicidal ideation48 gives also a hint, that the lethal barbiturate intoxication in case III (progredient ALS, existent barbiturate prescription, no documented suicidal ideation) may have been non accidental, but with suicidal intent. The results show an anisotropic regional lithium brain distribution in all three cases, both without and with lithium medication. They correlate well with data for trace lithium concentrations in rodent brain49 and also NMR-Studies on lithium medicated humans41.
The histogram of lithium distribution in the different brain regions of all cases approaches a Gaussian distribution, thus indicating a homeostatic regulation under endogenous and also medicated conditions. As a normal (rather than log-normal) distribution of an element’s tissue concentration is considered as criterion for essentiality50, our results accordingly support the thesis that lithium may be of essential relevance not only in animal, but also in man15.
Another essential finding from the measurements emerges when considering the differences in distribution between white and gray matter. In Case I and III there was a clear difference in the lithium concentration in favor of white matter with a ratio for cLiwm/cLigm well > 1, which allows a conclusion that the core areas contain significantly less lithium than the axonal components. Similar results with regard to an imbalance in the distribution of lithium in between white and gray matter in the lithium medicated brain have been found in animal experiments, where lithium supplemented rats showed two to three times higher lithium concentrations in white matter than in gray matter51. A possible explanation for this may be, that in the axon the density of voltage-sensitive lithium-permeable sodium channels is higher. This would enable intraneuronal lithium concentrations to vary depending on the action potential frequency52. And as the potential of lithium at equilibrium is less negative in regions with high myelin density (→ axon) than in regions with a low myelin density (→ core region) the presence of myelin would thus reduce a sodium channel-associated passive diffusion of lithium with a dampening effect on brain excitability53. However, in case II (suicide) the ratio for cLiwm/cLigm was well < 1, further proved by an anticorrelation of overall λI/λII (Fig. 10). An explanation thesis for this almost diametrical distribution imbalance would be on the basis of the above statements that based on excitatory events on the axon membrane a redistribution of lithium via sodium channels out of the myelinated area occurs in the immediate run-up to the suicidal event. Another explanation could hint to a primary, i.e. an already existing lithium deficiency or a “utilization disorder”. Although the extent to which a primarily higher endogenous concentration of lithium in the white matter of the human brain compared to gray matter could mean a mood-stabilizing and thus possibly antisuicidal effect, remains speculative to date.
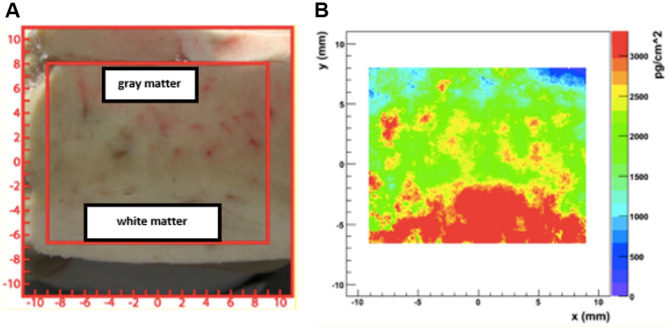
Figure 10.
NIK Measurement result: (A) cryotome sample* (B) Lithium area density λ. For the position reconstruction, including the particle straggling a typical resolution of σx,y ≈ 0.4 mm was achieved. *Comparative sample: frontal lobe (lithium intoxication).
Another interesting result is the distributional imbalance in the so called emotional regions, in particular Thalamus and BA25. Both brain regions are part of the limbic system and of exceptional relevance with regard to affect-regulating processes. In Case I the tracts of the thalamic region contained significant more lithium than any other regions with a ratio cLiThwm/cLiThgm of approximately 2. In case III also the white matter concentration of the thalamic region was comparably high with a ratio cLiThwm/cLiThgm of approximately 1.8. In Case II the rostral part showed a much higher concentration in the tracts and a very low concentration in the core section (cLiThwm/cLiThgm ca. 3.5). The medial parts of the thalamic tracts as much as the core section showed comparably low lithium concentrations with a ratio cLiThwm/cLiThgm = 1. And the dorsal part showed a high concentration in the core section, but very low concentration in the tracts (cLiThwm/cLiThgm approx.. 0.1), indicating an innerthalamic lithium distribution imbalance in suicide versus control. The thalamus is a structure consisting of numerous different core complexes as well as somatosensitive and motor fiber connections and seems to play an essential role as a switching point in mood regulation and the genesis of affective disorders54. Ischemia-related lesions of the dorsomedial thalamic nuclei can cause a transient maniform syndrome55. Furthermore, the thalamic volume appears to increase with lithium medication56 and the thalamus volume during lithium-treatment of patients with bipolar affective disorder increases compared to non-lithium-treated patients57. This indicates a structural importance of lithium in the thalamic metabolism.
The subcallous or subgenual part of the anterior cingulate gyrus BA25 consists of cortical tissue with adjacent tracts. It constitutes an important node in the limbic system network, e.g. thalamic structures and seems to play an important role in depressive disorder58. SASSI et al. showed in a study on bipolar patients, that untreated patients compared to lithium-treated patients and controls had a lower volume of the anterior cingulate gyrus59, assuming a lithium-associated structural benefit of lithium. MAYBERG et al. demonstrated, that this region, which is metabolically over reactive in therapy-resistant depression, is positively influenced by deep brain stimulation of the white matter adjacent to the area60. Our results in cases I and II show comparably high lithium levels in BA25 compared to other brain areas. In case I the cortical structures of BA25 showed the highest lithium concentration of all examined core structures, while the adjacent white matter lithium content was comparably low. In case III BA25`s overall lithium content was comparably high with an almost balanced lithium content in white vs gray matter (ratio cLiBA25wm/cLiBA25gm approx. = 1). But in case II the lithium concentration in the cortical part of the BA25 went to zero, while showing a medium range in the adjacent tracts, suggesting an almost diametral distribution imbalance in suicide versus control.
Limitations
The lithium distribution patterns so far obtained with the NIK method, thus in no way contradicting given literature references, are based on post mortem tissue. Although no macroscopic signs of degradation were observed, the extent to which an element-associated post-mortem redistribution at the molecular level—also in view of the cryopreservation procedure—may have caused significant discrepancies in relation to the vital state, cannot be methodologically cross-checked. Also a potential influence by certain agonal phenomena—even if there was no higher-grade cerebral edema in particular in case II (suicide by hanging)—cannot be excluded with certainty and therefore needs to be checked on a larger collective. Another limitation arises from sample collective limitations, selections and restrictions on the basis of the tissue law and ethical considerations.
Conclusion
With the use of the NIK method, it has been possible for the first time to map the distribution pattern of the endogenous lithium content in the various regions of the human brain. The so found intra- and interindividual lithium distribution in the brain is not only anisotropic under both endogenous and medicinal conditions, but also approximates a so-called Gaussian distribution. This speaks in favor of a homeostatic regulation of both endogenous and drug-induced lithium concentrations and could serve as an indication for the essentiality of the trace element. In case of suicide the ratio of white matter versus gray matter lithium concentration was < 1 and thus almost diametral to the control collective, where the ratio was well > 1, so that an acute lithium depletion in suicide could be considered. In addition, in the so-called emotional regions thalamus and BA25 noticeable lithium distribution difference between suicide and control collective were detected. Due to the small number of cases, the quality of information is limited and should be checked on a larger collective.
Methods
The authors have further established and applied a hitherto unique measurement method to detect and spatially resolve even smallest amounts of trace lithium in organic samples for examining different brain regions in suicide versus control. Numerous samples of multiple brain regions in bilateral setting (n = 139) of 3 deceased individuals (Table 1) were taken at autopsy at the “Institut für Rechtsmedizin” Munich, Germany (IRM).
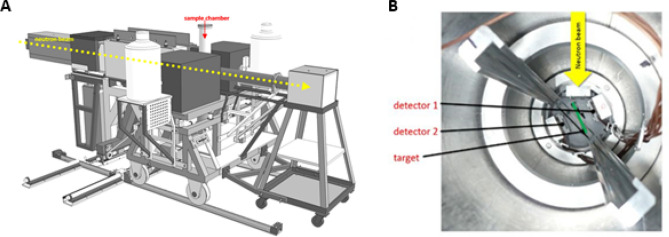
The experiments were conducted at the research reactor “Forschungs-Neutronenquelle Heinz Maier-Leibnitz” (FRM II) and the Prompt Gamma Activating Analysis (PGAA) in Garching/Munich, Bavaria, where a well focused beam of cold neutrons with a neutron flux of ɸ = 1.2 × 1010 cm−2 s−1 is available.
Methodological background
Lithium exists in nature as a mixture of two stable Isotopes 6Li and 7Li in a fixed ratio of 7.59 ± 0.04% 6Li to 92,41 ± 0.04% 7Li43. The 6Li-Isotop exhibits an exceptionally large neutron capture cross section (En = 25 meV) for thermal neutrons of σn = 940 ± 4 b, scaling for cold neutrons with En = 1.83 meV according to the optical theorem to σn = 3474 b44. After the capture of a neutron, its nucleus is highly excited so that particles fission almost instantaneously into an alpha (4He)- and a tritium (3H) particle (see Fig. 5 for illustration).
Figure 5.
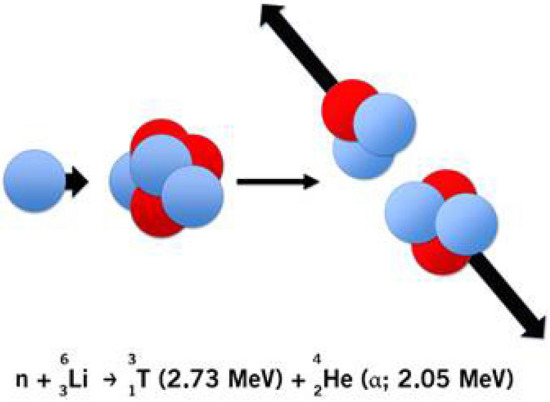
Schematic illustration of the 6Li(n,α)3H-reaction.
The dominant reaction channel 6Li(n,α)3H with the two particles in its exit channel obtains a fixed energy of E3H = 2.73 meV and Eα = 2.05 meV. With the center of mass system being nearly at rest in the laboratory frame, 4He and 3H are emitted at an angle of Θ = 180° with respect to each other. The coincident detection of the kinetic energy and the impact sites of both particles via detector measurement enables the retrograde reconstruction of the reaction site inside the sample (see Fig. 6). The associated coincident energy pattern is unique, as the total energy deposit in the detectors is far above the energy signals by the dominant background radiation (β and γ particles) or a small branch particle background from the 10B(n,α)7Li reaction with about 2 meV less kinetic energy distributed on both reaction products. But especially the time coincidence measurement of signals in both detectors with a time difference ∆t < 200 ns provides an extreme selectivity for the detection of 6Li.
Figure 6.
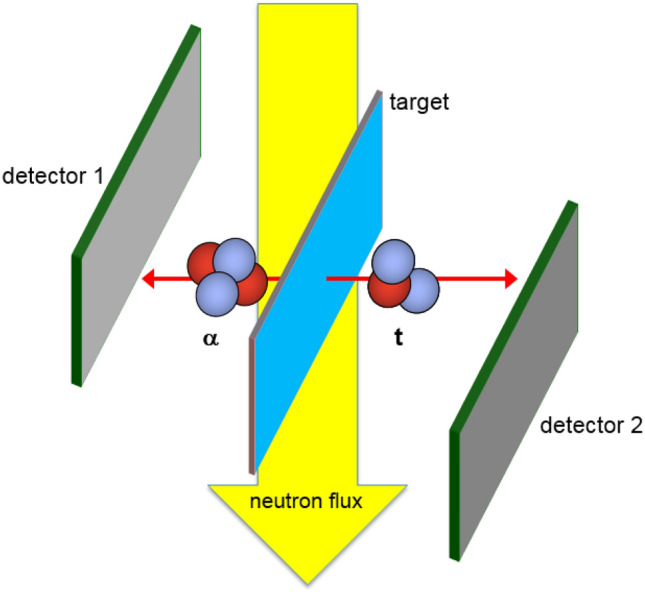
Experimental concept for the position sensitive lithium detection with the 6Li(n, α)3H-reaction. Both 20 × 20 mm2 large silicon detectors have resistive layers for charge splitting on both surfaces. Two readout strips at the edges in horizontal and vertical direction respectively allow for a position resolution of σx,y ~ 0.2 mm for a typical particle energy of 2 MeV.
The flow of cold neutrons is released from the reactor guided via neutron mirrors through vacuum pipes to the PGAA target point (Fig. 7). The sample chamber of the PGAA measurement station consists mainly of Aluminum (Al) and Polytetrafluorethylen (Fig. 8). These are materials with very small neutron capture cross-sections and daughter nuclei with very short lifespans. The measurement setup consists of a compact detector arrangement with a collimator for the neutron flux in the front, two position sensitive silicon detectors on both sides mounted diametral to each other and tilted by Θ = 25° with respect to the axis of the neutron beam to optimize the solid angle coverage and a sample holder in the middle (see Fig. 6). The sample holder with a size of 40 mm × 30 mm × 0.2 mm was designed to guarantee a maximum overlap between the collimated neutron beam and the sample, but no structural elements in the area of the neutron flux. The sample holder consists of a pure aluminum frame (cAl > 99.999%) covered with foil of lithium-free Polyethylene (PE) with a calibrated thickness of 0.9 ± 0.2 μm and is prepared with a layer of neutralizing bipolar ions (RI 65 P, Ringionisator, Haug) to avoid possible contamination by dust particles.
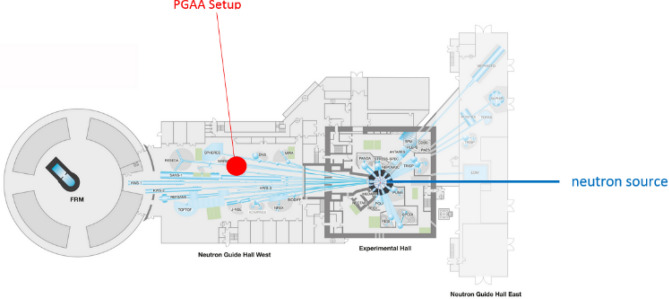
Figure 7.
Reactor assessment FRM II, schematic overview. mod. n. MLZ Overview: Lageplan_2013_All_engl; http://www.mlz-garching.de/instrumente/bildgebende-verfahren-und-analyse.html. Accessed 01 October 2020.
Figure 8.
Prompt Gamma Activation Analysis (PGAA): (A) schematic overview; (B) sample chamber.
Sample preparation
After the extraction of approximately 2.5 × 2.5 × 1.5 cm3 large brain samples and storage in polyethylene bags at a temperature of − 20 °C they were sliced with a cryotome at the IRM without interrupting the cold chain. Because of limitations by the energy loss of particles passing through matter with an maximum range of 1.3 mg/cm2 for the alpha-particles, a sample thickness of d = 10 μm was chosen. The slices were then centered on the substrate foil of the sample frame (see Fig. 9A,B). To minimize dust contaminations on the support foils of the sample frame fabrication was done in a clean room. An accessory slice was put on a microscopic slide for further neuropathological assessment. After each slice section the next (n ≤ 10) slices made were discarded to minimize destructive temperature influences on the sample surface. For further reduction of the sample mass layer and guarantee a long term stability the cryotome sample then is lyophilized for 8 h in vacuum and conserved in a lithium-free PE container before PGAA measurement. To control contamination of the samples, empty sample frames were treated in parallel with identical procedures and stored in each box for reference.
Figure 9.
Cryotome brain sample: (A) sample on cryotome holder; (B) 10 µm slice attached to the sample frames made from a self-supporting d = 0.9 µm polyethylene cling film, fixed to an outer 300 μm thick aluminum target frame.
Neuropathological assessment
The additionally obtained microscopic samples were stained with hematoxylin-and-eosin technique and examined with focus on brain region and tissue texture resp. discriminating regional white matter and gray matter structures.
Layer thickness measurement
Lithium coverage of a cryosection sample depends on the respective sample thickness and varies significantly because of artifacts caused by cutting e.g. resulting in micro-cracks. Therefore a layer thickness measurement of every sample was applied. Every PGAA sample was bombarded with monoenergetic alpha-particles of an 241Americium (Am)-Source at a distance of 10 cm and the energy loss was measured with the same type of position sensitive silicon detectors as used in the NIK setup, which recorded the energy of the alpha-particles right after the sample. Taking into account of the “stopping power” amount defined for brain tissue45 the effective spatially resolved mass occupancy of the sample could be calculated.
Calibration and background measurement
Reference samples with a known homogeneous lithium thickness and empty target holders were used to calibrate the detector setup and to evaluate the position resolution.
Analytical controls
For all three cases additional measurement of brain tissue via atomic absorption spectroscopy (AAS) were conducted, analytically confirming the concentration range.
Data analysis
Statistic data analysis was carried out with IBM SPSS Statistics Software, Microsoft Excel Mac OS 2011, Vers. 23 and “ROOT” (Extention of C++/LHC Cern).
Using two hit points in the detector planes and the assumption of a planar target plane the emission points were reconstructed according to the theorem on intersecting lines. Hitting the detector, the particle’s impact energy is converted into an electrical charge collected in preamplifiers, converted into a voltage equivalent signals, digitized and stored on a hard drive. After data calibration and subtraction of background effects the lithium area density λ (pg/cm2) was obtained (Fig. 10). After normalization of λ with the local layer thickness of the sample the Lithium concentration cdry6Li could be calculated and by scaling with the average water content of brain structures46 and a three dimensional 6Li concentration in tissue cwet6Li was obtained. In the last step, with consideration of the natural lithium isotope ratio, the conversion in Linat resulted in the substrate morphometric documentation of the spatially resolved lithium concentration cwetLinat (ppb resp. ng/g).
Detection and quantification limit
The currently possible quantification limit for natLi with NIK is 60 fg/cm2 resp. 60 pg/gwet. The detection limit could be fixed with 8 fg/cm2 resp. 8 pg/gwet.
Ethical approval
The study is approved by the ethics committee of the Ludwig-Maximilian-University Munich LMU (Nr. 300-10). The study was conducted in concordance with the human tissue act 2004/23/EG and the 59th WMA General Assembly Seoul 2008. The informed consent of the Legally Authorized Representative (LAR) and/or the next of kin of all deceased individuals whose brain tissue was studied was given prior to examination and data anonymization in accordance with the votum of the ethics committee of the Ludwig-Maximilian-University Munich LMU (Nr. 300-10). The study was performed in accordance with the ethical standards of the Declaration of Helsinki (1964) and its subsequent amendments.
Acknowledgements
We thank the FRMII staff who ensured a stable and high intensity neutron beam. We would like to acknowledge Ralf Lang and Michael Klöckner for the precise production of all mechanical parts of this detection system. We also thank Petra Kudejova for the instrument support at the PGAA station, Reiner Krücken who initiated our collaboration, Karl Zeitelhack for the electronics and detector support and Dominik Seiler for the preparation of a long list of reference samples. This work is supported by DFG Contract No. GE2296/1.
Abbreviations
- AAS
Atomic absorption spectroscopy
- Al
Aluminium
- Am
Americium
- BA25
Brodmann area 25
- Ca++
Calcium cation
- cwetLinat
Normalized spatially resolved lithium
- cwm/cgm
Lithium concentration in white versus gray matter
- FRM II
Forschungs-Neutronenquelle Heinz Maier–Leibnitz
- ICP-MS
Inductively coupled plasma mass spectrometry
- IRM
Institut für Rechtsmedizin München
- lambda λ
Lithium area density
- Li+
Lithium cation
- Li+/d
Daily uptake of lithium cations
- Li+/oral unit
Lithium cation uptake per oral unit of pharmaceutical products
- Mg++
Magnesium cation
- n
Number of samples
- NIK
Neutron-induced coincidence method
- NMR
Nuclear magnetic resonance
- PE
Polyethylene
- PGAA
Prompt gamma activating analysis
- ppb
Parts per billion
- 3D
Three dimensional
Author contributions
J.S. and R.G. wrote the main manuscript text and had the idea. J.L. and R.G. developed the method with the help of all authors. J.L. did the measurements with assistence of A.S., M.B., S.W. and T.W., who performed the layer thickness measurements. Data acquisition, data analysis and interpretation: J.L., R.G. and J.S. All authors except M.G. took part in sample preparation and the experiments. C.D. and J.S. did the neuropathological examinations. M.G. reviewed the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kumar U, editor. Handbook of Suicidal Behaviour. Springer; 2017. [Google Scholar]
- 2.WHO. who.int/mental_health/prevention/suicide/suicideprevent/en/. (2020). Accessed 01 Dec 2020
- 3.Hawton K. Suicide. Lancet. 2009;373(9672):1372–1381. doi: 10.1016/S0140-6736(09)60372-X. [DOI] [PubMed] [Google Scholar]
- 4.Baldessarini RJ. Epidemiology of suicide: Recent developments. Epidemiol. Psychiatr. Sci. 2020;29:1–5. doi: 10.1017/S2045796019000672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cavanagh JT, Carson AJ, Sharpe M, Lawrie SM. Psychological autopsy studies of suicide: A systematic review. Psychol. Med. 2003;33(3):395–405. doi: 10.1017/S0033291702006943. [DOI] [PubMed] [Google Scholar]
- 6.Smith KA, Cipriani A. Lithium and suicide in mood disorders: Updated meta-review of the scientific literature. Bipolar Disord. 2017;19(7):575–586. doi: 10.1111/bdi.12543. [DOI] [PubMed] [Google Scholar]
- 7.Severus E, Bauer M, Geddes J. Efficacy and effectiveness of lithium in the long-term treatment of bipolar disorders: An update 2018. Pharmacopsychiatry. 2018;51:173–176. doi: 10.1055/a-0627-7489. [DOI] [PubMed] [Google Scholar]
- 8.Volkmann C, Bschor T, Köhler S. Lithium treatment over the lifespan in bipolar disorders. Front. Psych. 2020;11:377. doi: 10.3389/fpsyt.2020.00377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pinna M, Manchia M, Visioli C, Tondo L. Clinical response and metabolic effects of lithium in 323 mood disorder patients. J. Affect. Disord. 2020;270:9–14. doi: 10.1016/j.jad.2020.03.066. [DOI] [PubMed] [Google Scholar]
- 10.Lewitzka U, Severus E, Bauer R, Ritter P, Müller-Oerlinghausen B, Bauer M. The suicide prevention effect of lithium: More than 20 years of evidence: A narrative review. Int. J. Bipolar Disord. 2015;3(1):15. doi: 10.1186/s40345-015-0032-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Baldessarini RJ, Tondo L, Hennen J. Treating the suicidal patient with bipolar disorder: Reducing suicide risk with lithium. Ann. N. Y. Acad. Sci. 2001;932(1):24–43. doi: 10.1111/j.1749-6632.2001.tb05796.x. [DOI] [PubMed] [Google Scholar]
- 12.Del Matto L, Muscas M, Murru A, Verdolini N, Anmella G, Fico G, Corponi F, Carvalho AF, Samalin L, Carpiniello B, Fagiolini A, Vieta E, Pacchiarotti I. Lithium and suicide prevention in mood disorders and in the general population: A systematic review. Neurosci. Biobehav. Rev. 2020;116:142–153. doi: 10.1016/j.neubiorev.2020.06.017. [DOI] [PubMed] [Google Scholar]
- 13.Memon A, Rogers I, Fitzsimmons SM, Carter B, Strawbridge R, Hidalgo-Mazzei D, Young AH. Association between naturally occurring lithium in drinking water and suicide rates: Systematic review and meta-analysis of ecological studies. Br. J. Psychiatry. 2020;217:667–678. doi: 10.1192/bjp.2020.128. [DOI] [PubMed] [Google Scholar]
- 14.Barjasteh-Askari F, Davoudi M, Amini H, Ghorbani M, Yaseri M, Yunesian M, Mahvi HM, Lester D. Relationship between suicide mortality and lithium in drinking water: A systematic review and meta-analysis. J. Affect. Disord. 2020;264:234–241. doi: 10.1016/j.jad.2019.12.027. [DOI] [PubMed] [Google Scholar]
- 15.Schrauzer GN. Lithium: Occurrence, dietary intakes, nutritional essentiality. J. Am. Coll. Nutr. 2002;21(1):14–21. doi: 10.1080/07315724.2002.10719188. [DOI] [PubMed] [Google Scholar]
- 16.Sheard MH. Effects of lithium on human aggression. Nature. 1971;230:113–114. doi: 10.1038/230113a0. [DOI] [PubMed] [Google Scholar]
- 17.Sheard MH. Lithium in the treatment of aggression. J. Nerv. Ment. Dis. 1975;160:108–118. doi: 10.1097/00005053-197502000-00005. [DOI] [PubMed] [Google Scholar]
- 18.Sheard MH, Marini JL, Bridges CI, Wagner E. The effect of lithium on impulsive aggressive behavior in man. Am. J. Psychiatry. 1976;133:1409–1413. doi: 10.1176/ajp.133.12.1409. [DOI] [PubMed] [Google Scholar]
- 19.Barraclough B. Suicide prevention, recurrent affective disorder and lithium. Br. J. Psychiatry. 1972;121(563):391–392. doi: 10.1192/bjp.121.4.391. [DOI] [PubMed] [Google Scholar]
- 20.Tondo L, Jamison KR, Baldessarini RJ. Effect of lithium maintenance on suicidal behavior in major mood disorders. Ann. N. Y. Acad. Sci. 1997;836(1):339–351. doi: 10.1111/j.1749-6632.1997.tb52369.x. [DOI] [PubMed] [Google Scholar]
- 21.Baldessarini RJ, Tondo L, Davis P, Pompili M, Goodwin FK, Hennen J. Decreased risk of suicides and attempts during long-term lithium treatment: A meta-analytic review. Bipolar Disord. 2006;8(52):625–639. doi: 10.1111/j.1399-5618.2006.00344.x. [DOI] [PubMed] [Google Scholar]
- 22.Cipriani A, Smith KA, Burgess SS, Carney SM, Goodwin G, Geddes J. Lithium versus antidepressants in the long-term treatment of unipolar affective disorder. Cochrane Datab. Syst. Rev. 2006;4:3492. doi: 10.1002/14651858.CD003492.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Dawson EB, Moore TD, McGanity WJ. The mathematical relationship of drinking water lithium and rainfall to mental hospital admission. Dis. Nerv. Syst. 1970;31:811–820. [PubMed] [Google Scholar]
- 24.Dawson EB, et al. Relationship of lithium metabolism to mental hospital admission and homicide. Dis. Nerv. Syst. 1972;33(8):546–556. [PubMed] [Google Scholar]
- 25.Schrauzer GN, Shrestha KP. Lithium in drinking water and the incidences of crimes, suicides, and arrests related to drug addictions. Biol. Trace Elem. Res. 1990;25(2):105–113. doi: 10.1007/BF02990271. [DOI] [PubMed] [Google Scholar]
- 26.Ohgami H, Terao T, Shiotsuki I, Ishii N, Iwata N. Lithium levels in drinking water and risk of suicide. Br. J. Psychiatry. 2009;194(5):464–465. doi: 10.1192/bjp.bp.108.055798. [DOI] [PubMed] [Google Scholar]
- 27.Schöpfer J, Schrauzer GN. Lithium and other elements in scalp hair of residents of Tokyo Prefecture as investigational predictors of suicide risk. Biol. Trace Elem. Res. 2011;144(1–3):418–425. doi: 10.1007/s12011-011-9114-x. [DOI] [PubMed] [Google Scholar]
- 28.Kapusta ND, Mossaheb N, Etzersdorfer E, Hlavin G, Thau K, Willeit M, Leithner-Dziubas K. Lithium in drinking water and suicide mortality. Br. J. Psychiatry. 2011;198(5):346–350. doi: 10.1192/bjp.bp.110.091041. [DOI] [PubMed] [Google Scholar]
- 29.Giotakos O, Nisianakis P, Tsouvelas G, Giakalou VV. Lithium in the public water supply and suicide mortality in Greece. Biol. Trace Elem. Res. 2013;156(1–3):376–379. doi: 10.1007/s12011-013-9815-4. [DOI] [PubMed] [Google Scholar]
- 30.Sugawara N, Yasui-Furukori N, Ishii N, Iwata N, Terao T. Lithium in tap water and suicide mortality in Japan. Int. J. Environ. Res. Public Health. 2013;10(11):6044–6048. doi: 10.3390/ijerph10116044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ishii N, Terao T, Araki Y, Kohno K, Mizokami Y, Shiotsuki I, Iwata N. Low risk of male suicide and lithium in drinking water. J. Clin. Psychiatry. 2015;76:319–326. doi: 10.4088/JCP.14m09218. [DOI] [PubMed] [Google Scholar]
- 32.Vita A, De Peri L, Sacchetti E. Lithium in drinking water and suicide prevention: A review of the evidence. Int. Clin. Psychopharmacol. 2015;30(1):1–5. doi: 10.1097/YIC.0000000000000048. [DOI] [PubMed] [Google Scholar]
- 33.Kozaka N, Takeuchi S, Ishii N, Terao T, Kuroda Y. Association between lithium in tap water and suicide mortality rates in Miyazaki Prefecture. Environ. Health Prev. Med. 2020;25(1):1–7. doi: 10.1186/s12199-020-00865-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Lehmann K. Endogenous lithium levels. Pharmacopsychiatry. 1994;27(03):130–132. doi: 10.1055/s-2007-1014293. [DOI] [PubMed] [Google Scholar]
- 35.Birch N. Lithium and the Cell. Academic Press; 1991. [Google Scholar]
- 36.Sheard MH. The biological effects of lithium. Trends Neurosci. 1980;3(4):85–86. doi: 10.1016/0166-2236(80)90033-8. [DOI] [Google Scholar]
- 37.Lautrup B, Appali R, Jackson AD, Heimburg T. The stability of solitons in biomembranes and nerves. Eur. Phys. J. E. 2011;34(6):57. doi: 10.1140/epje/i2011-11057-0. [DOI] [PubMed] [Google Scholar]
- 38.Lenox RH, Hahn CG. Overview of the mechanism of action of lithium in the brain: Fifty-year update. J. Clin. Psychiatry. 2000;61:5–15. [PubMed] [Google Scholar]
- 39.Malhi GS, Outhred T. Therapeutic mechanisms of lithium in bipolar disorder: Recent advances and current understanding. CNS Drugs. 2016;30(10):931–949. doi: 10.1007/s40263-016-0380-1. [DOI] [PubMed] [Google Scholar]
- 40.Qaswal AB. Lithium stabilizes the mood of bipolar patients by depolarizing the neuronal membrane via quantum tunneling through the sodium channels. Clin. Psychopharmacol. Neurosci. 2020;18(2):214. doi: 10.9758/cpn.2020.18.2.214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Renshaw PF, Wicklund S. In vivo measurement of lithium in humans by nuclear magnetic resonce spectroscopy. Biol. Psychiat. 1988;23(5):465–475. doi: 10.1016/0006-3223(88)90018-2. [DOI] [PubMed] [Google Scholar]
- 42.Lichtinger J, Gernhäuser R, Bauer A, Bendel M, Canella L, Graw M, Krücken R, Kudejova P, Muetzel E, Ring S, Seiler D, Winkler S, Zeitelhack K, Schöpfer J. Position sensitive measurement of lithium traces in brain tissue with neutrons. Med. Phys. 2013;40(2):023501. doi: 10.1118/1.4774053. [DOI] [PubMed] [Google Scholar]
- 43.Berglund M, Wieser ME. Isotopic compositions of the elements 2009 (IUPAC technical report) Pure Appl. Chem. 2011;83(2):197–410. doi: 10.1351/PAC-REP-10-06-02. [DOI] [Google Scholar]
- 44.Bellac ML. Quantum Physics. Cambridge University Press; 2011. p. 2011. [Google Scholar]
- 45.Ziegler, J. F., Biersack, J. P., & Ziegler, M. D. (2010). Stopping and Range of ions in matter. www.srim.org, http://dtic.mil/dtic/tr/fulltext/u2/a515302.pdf.
- 46.Ernst T, Kreis R, Ross B. Absolute quantitation of water and metabolites in the human brain. I. Compartments and water. J. Magn. Reson. B. 1993;102(1):1–8. doi: 10.1006/jmrb.1993.1055. [DOI] [Google Scholar]
- 47.Moore CM, Demopulos CM, Henry ME, Steingard RJ, Zamvil L, Katic A, Renshaw PF. Brain-to-serum lithium ratio and age: An in vivo magnetic resonance spectroscopy study. Am. J. Psychiatry. 2002;159(7):1240–1242. doi: 10.1176/appi.ajp.159.7.1240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Grof P. Old treatment and new curiosity: Lithium in drinking water. Bipolar Disord. 2017;19(7):597–598. doi: 10.1111/bdi.12545. [DOI] [PubMed] [Google Scholar]
- 49.Eichner D, Opitz K. Über den natürlichen Lithiumgehalt tierischer Gewebe. Histochemistry. 1974;42(3):295–300. doi: 10.1007/BF00492662. [DOI] [PubMed] [Google Scholar]
- 50.Mertz W. Review of the scientific basis for establishing the essentiality of trace elements. Biol. Trace Elem. Res. 1998;66(1–3):185–191. doi: 10.1007/BF02783137. [DOI] [PubMed] [Google Scholar]
- 51.Edelfors S. Distribution of sodium, potassium and lithium in the brain of lithium-treated rats. Acta Pharmacol. Toxicol. 1975;37(5):387–392. doi: 10.1111/j.1600-0773.1975.tb00857.x. [DOI] [PubMed] [Google Scholar]
- 52.Gonzalez RG, Guimaraes AR, Sachs GS, Rosenbaum JF, Garwood M, Renshaw PF. Measurement of human brain lithium in vivo by MR spectroscopy. Am. J. Neuro-Radiol. 1993;14(5):1027–1037. [PMC free article] [PubMed] [Google Scholar]
- 53.Heurteaux C, Baumann N, Lachapelle F, Wissocq JC, Thellier M. Lithium distribution in the brain of normal mice and of “quaking” dysmyelinating mutants. J. Neurochem. 1986;46(4):1317–1321. doi: 10.1111/j.1471-4159.1986.tb00657.x. [DOI] [PubMed] [Google Scholar]
- 54.Soares JC. Contributions from brain imaging to the elucidation of pathophysiology of bipolar disorder. Int. J. Neuropsychopharmacol. 2003;6(2):171–180. doi: 10.1017/S1461145703003390. [DOI] [PubMed] [Google Scholar]
- 55.Benke T, Kurzthaler I, Schmidauer C, Moncayo R, Donnemiller E. Mania caused by a diencephalic lesion. Neuropsychologia. 2002;40(3):245–252. doi: 10.1016/S0028-3932(01)00108-7. [DOI] [PubMed] [Google Scholar]
- 56.Chen RW, Chuang DM. Long term lithium treatment suppresses p53 and Bax expression but increases Bcl-2 expression. A prominent role in neuroprotection against excitotoxicity. J. Biol. Chem. 1999;274(10):6039–6042. doi: 10.1074/jbc.274.10.6039. [DOI] [PubMed] [Google Scholar]
- 57.Radenbach K, Flaig V, Schneider-Axmann T, Usher J, Reith W, Falkai P, Gruber O, Scherk H. Thalamic volumes in patients with bipolar disorder. Eur. Arch. Psychiatry Clin. Neurosci. 2010;260(8):601–607. doi: 10.1007/s00406-010-0100-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Hamani C, Mayberg H, Stone S, Laxton A, Haber S, Lozano AM. The subcallosal cingulate gyrus in the context of major depression. Biol. Psychiat. 2011;69(4):301–308. doi: 10.1016/j.biopsych.2010.09.034. [DOI] [PubMed] [Google Scholar]
- 59.Sassi RB, Brambilla P, Hatch JP, Nicoletti MA, Mallinger AG, Frank E, Kupfer DJ, Keshavan MS, Soares JC. Reduced left anterior cingulate volumes in untreated bipolar patients. Biol. Psychiat. 2004;56(7):467–475. doi: 10.1016/j.biopsych.2004.07.005. [DOI] [PubMed] [Google Scholar]
- 60.Mayberg HS, Lozano AM, Voon V, McNeely HE, Seminowicz D, Hamani C, Schwalb JN, Kennedy SH. Deep brain stimulation for treatment-resistant depression. Neuron. 2005;45(5):651–660. doi: 10.1016/j.neuron.2005.02.014. [DOI] [PubMed] [Google Scholar]