Abstract
Background
The presence of SARS‐CoV‐2 RNA in plasma has been linked to disease severity and mortality. We compared RT‐qPCR to droplet digital PCR (ddPCR) to detect SARS‐CoV‐2 RNA in plasma from COVID‐19 patients (mild, moderate, and critical disease).
Methods
The presence/concentration of SARS‐CoV‐2 RNA in plasma was compared in three groups of COVID‐19 patients (30 outpatients, 30 ward patients and 30 ICU patients) using both RT‐qPCR and ddPCR. Plasma was obtained in the first 24h following admission, and RNA was extracted using eMAG. ddPCR was performed using Bio‐Rad SARS‐CoV‐2 detection kit, and RT‐qPCR was performed using GeneFinder™ COVID‐19 Plus RealAmp Kit. Statistical analysis was performed using Statistical Package for the Social Science.
Results
SARS‐CoV‐2 RNA was detected, using ddPCR and RT‐qPCR, in 91% and 87% of ICU patients, 27% and 23% of ward patients and 3% and 3% of outpatients. The concordance of the results obtained by both methods was excellent (Cohen's kappa index = 0.953). RT‐qPCR was able to detect 34/36 (94.4%) patients positive for viral RNA in plasma by ddPCR. Viral RNA load was higher in ICU patients compared with the other groups (P < .001), by both ddPCR and RT‐qPCR. AUC analysis revealed Ct values (RT‐qPCR) and viral RNA load values (ddPCR) can similarly differentiate between patients admitted to wards and to the ICU (AUC of 0.90 and 0.89, respectively).
Conclusion
Both methods yielded similar prevalence of RNAemia between groups, with ICU patients showing the highest (>85%). RT‐qPCR was as useful as ddPCR to detect and quantify SARS‐CoV‐2 RNAemia in plasma.
Keywords: COVID‐19, ddPCR, RNAemia, RT‐qPCR, SARS‐CoV‐2, viral RNA load
1. INTRODUCTION
On 31st of December 2019, China reported to World Health Organization (WHO) the first cases of pneumonia from an unknown origin and on the 11th of March 2020 WHO declared coronavirus disease 2019 (COVID‐19) as a pandemic. As of 27th of December 2020, 79.2 million cases of COVID‐19 and over 1.7 million deaths had been reported worldwide. 1 Most patients with COVID‐19 have a mild to moderate disease. Nevertheless, 10 to 20% of hospitalized patients develop severe disease and are admitted to the ICU, of which about 40% died, during the first pandemic wave. 2 , 3
The vast majority of COVID‐19 diagnosis is performed by detecting severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) RNA in nasopharyngeal swabs using real‐time reverse transcriptase PCR (RT‐qPCR). Nevertheless, some studies have demonstrated the presence of SARS‐CoV‐2 RNA in other bodily fluids, including plasma, either using RT‐qPCR 4 , 5 , 6 or droplet digital PCR (ddPCR). 7 , 8 In all of these studies, the presence of viral RNA in plasma has been associated to increased disease severity, as it has mostly been detected in patients admitted to the ICU. Furthermore, the use of ddPCR allows not only the detection but also the quantification of SARS‐CoV‐2 RNA in plasma and high viral RNA loads in plasma have also been associated with increased disease severity and also mortality. 7 , 8 These results suggest that the presence and concentration of SARS‐CoV‐2 RNA in plasma are probably an important clinical biomarker of severity, namely need to be admitted to the ICU, and its’ use in clinical practice could be an important tool to improve patients’ management.
Droplet digital PCR is a very sensitive PCR technology that is available for the absolute quantification of nucleic acids without the need for a standard curve. Even though the use of ddPCR in research laboratories has increased in the last decade, this technology is rarely used in clinical laboratories, mostly due to its’ high cost. 9
In this study, we compared a standard RT‐qPCR method normally used in clinical microbiology laboratories with a next‐generation PCR method (ddPCR) to evaluate the presence and concentration of SARS‐CoV‐2 RNA in plasma from COVID‐19 patients with mild, moderate and severe disease.
2. METHODS
2.1. Study design
The presence and concentration of SARS‐CoV‐2 RNA in plasma were compared in three groups of COVID‐19 patients (30 outpatients, 30 patients admitted to clinical wards, and 30 patients admitted to the ICU) using both RT‐qPCR and ddPCR. All the patients were adults (≥18 years old) with a positive nasopharyngeal swab RT‐qPCR for SARS‐CoV‐2 and were recruited during the first wave of SARS‐CoV‐2 pandemic in Spain (March 16th to April 5th 2020). ddPCR results came from a previous study from our group. 7 This is a sub‐study of the CIBERES‐UCI‐COVID project (registered at Clinicaltrials.gov NCT04457505). 7
2.2. Blood samples
Plasma was obtained from blood collected in EDTA tubes in the first 24 hours following admission to the emergency room, to the ward, or to the ICU, at a median collection day since disease onset of 9, 9 and 11 respectively.
2.3. SARS‐CoV‐2 RNA extraction
RNA was extracted from 100 µl of plasma using the automated system eMAG® (BioMérieux®, Marcy l'Etoile, France) following manufacturer instructions. The RNA extraction procedure used by eMAG is based on magnetic silica technology. 10
2.4. Droplet digital PCR
Detection and quantification of SARS‐CoV‐2 RNA were performed in 5 µl of total RNA obtained from plasma using the Bio‐Rad SARS‐CoV‐2 ddPCR kit according to manufacturer's specifications on a QX‐200 ddPCR platform (Bio‐Rad, Hercules, California, USA) as previously described. 7
2.5. Real‐time RT‐PCR
RT‐qPCR was performed with 5 µl of total RNA obtained from plasma using GeneFinder™ COVID‐19 Plus RealAmp Kit (OSANG Healthcare Co., Gyeonggi‐do, Republic of Korea) and CFX96™ thermocycler (Bio‐Rad, Hercules, California, USA). This kit detects the following SARS‐CoV‐2 genes: RdRp gene (codes for RNA‐Dependent RNA Polymerase), the E gene (codes for viral envelop), the N gene (codes for viral nucleocapside), as well as the RNAse P gene as an internal control. Samples were considered positive for SARS‐CoV‐2 if any of the genes it detects (RdRp, E or N) presented a Ct (cycle threshold) value not higher than 40, according to the manufacture instructions.
2.6. Statistical analysis
Statistical analysis was performed by using Statistical Package for the Social Science (SPSS) 20.0 (SPSS INC, Chicago, IL, USA). Gene E was the only one of the three genes analysed that yielded a result in all those samples identified as positive by RT‐qPCR. In consequence, the statistical analysis was performed using this gene. Samples with a negative result for all the three genes analysed by RT‐qPCR were given a Ct value of 41 for the Gene E for viral RNA load quantification purposes. The differences between groups were assessed using the Chi‐square test / Fisher's Exact Test where appropriated for categorical variables. Differences for continuous variables were assessed by using the Kruskal‐Wallis test. The accuracy of viral load and Cts to differentiate between severity groups was evaluated by calculating the area under the receiver curve (AUC). Spearman correlation coefficient was used to determine whether viral load or Cts was associated with time (in days) from disease onset. Cohen's Kappa Index was calculated using https://idostatistics.com/cohen‐kappa‐free‐calculator. Heatmap was built with the ‘pheatmap’ package in ‘RStudio’ using the package default conditions.
3. RESULTS
Clinical characteristics of the patients are described in Table S1 from supplementary material. By using ddPCR, SARS‐CoV‐2 RNA was detected in 91% (27/30) of the ICU patients, 27% (8/30) of the ward patients and 3% (1/30) of outpatients. Percentages of SARS‐CoV‐2 RNAemia were very similar using RT‐qPCR: 87% (26/30), 23% (7/30) and 3% (1/30), in the ICU, ward and outpatients’ groups, respectively (Figure 1). The concordance of the results obtained by both methods is excellent, as shown by the Cohen's kappa index (0.953). RT‐qPCR was able to detect 34 out of 36 of those patients positive for viral RNA in plasma using ddPCR (94.4%). Moreover, all negative results by ddPCR were also negative by RT‐qPCR (Figure 1A and 1B, Table S2 supplementary material).
FIGURE 1.
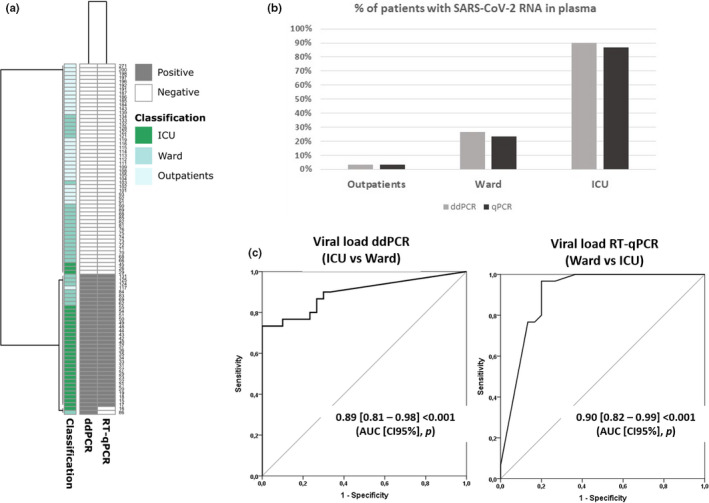
Panel (a) Heatmap showing individual results for the presence/absence of SARS‐CoV‐2 RNA in plasma using qPCR or ddPCR. (b) Percentage of SARS‐CoV‐2 RNA in plasma in each group of patients. (c) AUROC analysis to differentiate patients admitted to the ward or to the ICU based on viral load in plasma assessed by RT‐qPCR or ddPCR
Both ddPCR and RT‐qPCR evidenced that viral RNA load was higher in those patients admitted to the ICU compared with the other groups (P < .001). No differences between viral RNA load or Cts were found between ward patients and outpatients, in agreement with our previous report. 7 AUC analysis revealed that using the Ct values obtained by RT‐qPCR was as accurate as using viral RNA load values obtained by ddPCR to differentiate between patients admitted to the wards and those admitted to the ICU, yielding areas of 0.90 and 0.89, respectively (Figure 1C).
Spearman correlation coefficient analysis revealed that there was no association between the number of days from disease onset and viral load (0.106, P = .333) or Cts (−0.111, P = .307). AUC analysis showed that both viral load evaluated by ddPCR or the Cts assessed by RT‐qPCR were able to differentiate critically ill patients from non‐critically ill ones, independently whether the patients were recruited in the first nine days following symptoms onset (dso) or later: ddPCR < 10 dso (AUC = 0.85[0.66‐0.99], P = .001); ddPCR ≥ 10 dso (AUC = 0.95[0.87‐0.99], P < .001); Cts < 10 dso (AUC = 0.87[0.70‐0.99], P = .001); Cts ≥ 10 dso AUC = 0.94[0.85‐0.99], P < .001) (day nine was chosen to split the patients in two groups since this value was the median dso in our cohort).
4. DISCUSSION
The comparison of results obtained by RT‐qPCR and ddPCR supports that RT‐qPCR, using GeneFinder™ COVID‐19 Plus RealAmp Kit, could be a useful tool to detect the presence of SARS‐CoV‐2 RNA in plasma. The percentage of patients with SARS‐CoV‐2 RNA in plasma was higher than those reported in other studies using RT‐qPCR. 4 , 5 This discrepancy might be due to differences in patients` severity between studies, but could also be explained by the different probes and primers employed.
AUC results clearly support that RT‐qPCR is useful to estimate viral RNA load in plasma, in contrast to previous reports in respiratory samples. 11 The amount of virus in nasopharyngeal swabs has an intrinsic variability that depends on the operator and on the tolerance of the patients for sample collection. 12 In addition, the use of respiratory samples makes it difficult to adjust the results of viral RNA load to a reference mass or volume unit. 13 These problems are avoided using plasma, since sample collection is highly standardized and viral RNA concentration can be referenced to a fixed volume.
In our study, correlation analysis revealed that viral load was not associated with the time from symptoms onset. In turn, AUC analysis suggests that the detection of SARS‐CoV‐2 RNA in plasma could be potentially useful to detect patients deteriorating at any time point during disease course. These findings support that the amount of viral RNA in plasma depends more on severity than on the time spent from disease onset.
The main advantages of RT‐qPCR over ddPCR to detect and quantify viral RNA load of SARS‐CoV‐2 in plasma are as follows: i) RT‐qPCR is the standard method to diagnose SARS‐CoV‐2 infection in clinical microbiology laboratories, being widely available worldwide; ii) RT‐qPCR implementation is less complex and faster than ddPCR, needing less specialized training as required by the later; and iii) RT‐qPCR is cheaper to perform than ddPCR. 9
5. CONCLUSIONS
Our results evidence that standard RT‐qPCR is a useful method for the detection and quantification of SARS‐CoV‐2 RNA in plasma, which is a promising marker to early assess COVID‐19 severity. 7
CONFLICT OF INTEREST
The authors declare no conflicts of interest regarding this submission.
AUTHOR CONTRIBUTIONS
JFBM, DJK, JB, RF, FB, AT and RM designed the study. APT and JFBM wrote the manuscript and interpreted the data. RA coordinated the clinical study. MDG, MGR, DM, PRM, RM, NBL, FPG, EB and JMG recruited the patients and /or collected the clinical data. MDG, AO, RO and JME performed the assays for the detection of SARS‐CoV‐2 by RT‐qPCR. APT and CD developed the ddPCR works. AAK, ATO and RB performed the literature search. All authors read and approved the final manuscript.
Supporting information
Supplementary Material
ACKNOWLEDGMENTS
We thank the nurse teams of the participant Clinical Services for their collaboration in sample collection.
Tedim AP, Almansa R, Domínguez‐Gil M, et al. Comparison of real‐time and droplet digital PCR to detect and quantify SARS‐CoV‐2 RNA in plasma. Eur J Clin Invest. 2021;51:e13501. 10.1111/eci.13501
Funding
This work was supported by awards from the Canadian Institutes of Health Research, the Canadian 2019 Novel Coronavirus (COVID‐19) Rapid Research Funding initiative (CIHR OV2 – 170357), Research Nova Scotia (DJK), Atlantic Genome/Genome Canada (DJK), Li‐Ka Shing Foundation (DJK), Dalhousie Medical Research Foundation (DJK), the ‘Subvenciones de concesión directa para proyectos y programas de investigación del virus SARS‐CoV2, causante del COVID‐19’, FONDO ‐ COVID19, Instituto de Salud Carlos III (COV20/00110, CIBERES, 06/06/0028), (AT). DJK is a recipient of the Canada Research Chair in Translational Vaccinology and Inflammation. The funding sources did not play any role neither in the design of the study and collection, not in the analysis, in the interpretation of data or in writing the manuscript. APT was supported by the Sara Borrell Research Grant CD018/0123 funded by Instituto de Salud Carlos III and co‐financed by the European Development Regional Fund (A Way to Achieve Europe program). This study has also been funded by a Research Grant 2020 from the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) APT.
REFERENCES
- 1. World Health Organization (WHO) . (2020). Weekly epidemiological update‐29. 2020 [cited 2021 Jan 5]. p. 21. Retrieved from: https://www.who.int/publications/m/item/weekly‐epidemiological‐update–‐29‐december‐2020
- 2. Rodriguez‐Gonzalez C, Chamorro‐de‐Vega E, Valerio M, et al. COVID‐19 in hospitalized patients in Spain: a cohort study in Madrid. Int J Antimicrob Agents. 2020;57:106249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Armstrong RA, Kane AD, Cook TM. Outcomes from intensive care in patients with COVID‐19: a systematic review and meta‐analysis of observational studies. Anaesthesia. 2020;75:1340‐1349. [DOI] [PubMed] [Google Scholar]
- 4. Chen X, Zhao B, Qu Y, et al. Detectable serum severe acute respiratory syndrome coronavirus 2 viral load (RNAemia) Is closely correlated with drastically elevated interleukin 6 level in critically Ill patients with coronavirus disease 2019. Clin Infect Dis. 2020;71:1937‐1942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Chen W, Lan Y, Yuan X, et al. Detectable 2019‐nCoV viral RNA in blood is a strong indicator for the further clinical severity. Emerging Microbes & Infections. 2020;9:469‐473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Gutmann C, Takov K, Burnap S, Singh B, Theofilatos K, Reed E, et al. SARS‐CoV‐2 RNAemia and proteomic biomarker trajectory inform prognostication in COVID‐19 patients admitted to intensive care. Reseach Sq. 2020. [DOI] [PMC free article] [PubMed]
- 7. Bermejo‐Martin JF, González‐Rivera M, Almansa R, et al. Viral RNA load in plasma is associated with critical illness and a dysregulated host response in COVID‐19. Crit Care. 2020;24:691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Veyer D, Kernéis S, Poulet G, et al. Highly sensitive quantification of plasma SARS‐CoV‐2 RNA shelds light on its potential clinical value. Clin Infect Dis. 2020;ciaa1196. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hindson CM, Chevillet JR, Briggs HA, et al. Absolute quantification by droplet digital PCR versus analog real‐time PCR. Nat Methods. 2013;10:1003‐1005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Boom R, Sol CJA, Salimans MMM, Jansen CL, Wertheim‐Van Dillen PME, Van Der Noordaa J. Rapid and simple method for purification of nucleic acids. J Clin Microbiol. 1990;28:495‐503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Binnicker MJ. Challenges and controversies to testing for COVID‐19. J Clin Microbiol. 2020;58:11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Ye G, Li Y, Lu M, et al. Experience of different upper respiratory tract sampling strategies for detection of COVID‐19. Journal of Hospital Infection. W.B. Saunders Ltd. 2020;105:1‐2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Dahdouh E, Lázaro‐Perona F, Romero‐Gómez MP, Mingorance J, García‐Rodriguez J. Ct values from SARS‐CoV‐2 diagnostic PCR assays should not be used as direct estimates of viral load. J Infect. 2020. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material


