Abstract
The graphene revolution, which has taken place during the last 15 years, has represented a paradigm shift for science. The extraordinary properties possessed by this unique material have paved the road to a number of applications in materials science, optoelectronics, energy, and sensing. Graphene‐related materials (GRMs) are now produced in large scale and have found niche applications also in the biomedical technologies, defining new standards for drug delivery and biosensing. Such advances position GRMs as novel tools to fight against the current COVID‐19 and future pandemics. In this regard, GRMs can play a major role in sensing, as an active component in antiviral surfaces or in virucidal formulations. Herein, the most promising strategies reported in the literature on the use of GRM‐based materials against the COVID‐19 pandemic and other types of viruses are showcased, with a strong focus on the impact of functionalization, deposition techniques, and integration into devices and surface coatings.
Keywords: 2D materials, antimicrobials, carbon nanomaterials, SARS‐CoV‐2, sensing
The present pandemic has motivated researchers with different backgrounds to fight against SARS‐CoV‐2. Graphene‐related materials present numerous benefits for new antiviral solutions. The interaction of graphene‐related materials with viruses, their virucidal activity, and their possible use in antiviral coatings and sensing devices for fast and reliable detection are highlighted.
1. Introduction
The spread of COVID‐19 worldwide has dramatically changed our daily lives resulting in the most unstable and uncertain period our society is facing after the two world wars. The infections are coming as waves with increasing contagion and deaths.[ 1 ] Many governments are grappling with the competing demands of protecting the economy and protecting the healthcare systems. At the same time, researchers worldwide are devoting a significant effort in the search for strategies capable to contain the pandemic, diminish hospitalization, and develop as fast as possible a reliable vaccine.[ 2 ] So far we have learnt that our society will have to coexist at least for the next one to two years, with the virus forcing mankind to adopt new habits, use protective equipment, and take the precaution of sanitizing commonly touched surfaces. In this context, research of new antiviral solutions is highly sought after.
Over the past 15 years, graphene and graphene‐related materials (GRMs) have attracted great attention because of their unique physical and chemical properties, which render them powerful components for applications in optoelectronics, energy storage and generation, (bio)chemical sensing, reinforcement for aeronautics and constructions, membranes for purification of water, etc. GRMs are composed of various carbon 2D materials each of them with a specific structure and nomenclature.[ 3 , 4 ] Among the different GRMs, the most promising materials are graphene (G), graphene oxide (GO), and reduced graphene oxide (rGO) (Figure 1 ). G displays a typical honeycomb sp2 carbon structure that is responsible for its flexibility and strength, exceptional electrical conductivity, and high lipophilicity. Conversely, GO, the oxidized form of G, contains various oxygenated groups and some sp3 carbons that decrease the mechanical and electrical performances but render it highly hydrophilic and water dispersible. Additionally, GO can be reduced to partially restore the electrical conductivity but retaining, to a great extent, the water dispersibility, yielding rGO. GRMs are already produced at the industrial scale and their application has been included in different device prototypes.
Figure 1.
Sketch describing the preparation of antiviral GRMs. Top: Chemical structures of graphene, graphene oxide, and reduced graphene oxide. Middle: GRMs can enhance their affinity for SARS‐CoV‐2 through chemical functionalization with specific functional groups. Bottom: Following incubation, GRMs can irreversibly damage the virus, reducing their infectivity (bottom).
Herein, we will showcase and rationalize the most relevant applications based on graphene and related materials that can be foreseen to combat viral pandemics. In particular, GRM applications as antiviral material for drug delivery, smart surfaces, and (bio)sensing will be critically discussed and analyzed. Timely applications, opportunities, and future challenges will be addressed. With this contribution, we would like to stimulate scientists to think out of the box to find new efficient solutions against COVID‐19 and the next viral pandemics.
2. Medical Applications Blocking the Viral Replication Machinery
Since their discovery, GRMs have become powerful allies to fight against different diseases.[ 5 ] By taking full advantage of their tunable size, high biocompatibility, and tailored surface functionalities, GRMs have become useful components for application in different biomedical fields including drug delivery, bioimaging, and tissue engineering. Current progress of GRMs for antiviral applications is still at the early stage. Yet, the encouraging results achieved in other biomedical applications, including antimicrobial and immune activation, make G and GO potentially promising tools to fight against viral pandemics. G, GO, and rGO have been used in biomedical research against different kinds of viruses. They combine remarkable figures of merit including a high surface area offering good contact with the viral protein, a well‐established surface chemistry enabling the preparation of multifunctional platforms, and an efficient photothermal activity under near‐infrared (NIR) laser irradiation, allowing local temperature enhancement. Thus, GRMs can be ad hoc designed targeting common viral replication machinery (broad spectrum antiviral) or a specific viral antigen (specific antiviral) making them useful allies against SARS‐CoV‐2 and other viral infections. G, GO, and rGO have been already explored for their virustatic activity, and so designed to reversibly bind viral proteins or host cell proteins responsible for the attachment of the virus, thus blocking the infection at the early stage. The most popular targeting agent reported has been the mimicking cell heparan sulfate (HS), a polysaccharide present in a variety of mammalian cells and broadly used by several viruses (e.g., HIV, Herpes symplex virus, Zika, and Hepatitis B virus) to penetrate host cell membrane.[ 6 ] SARS‐CoV‐2 seems also to require HS as an assisting cofactor to enter host cells.[ 7 ] Thus, first clinical data have shown that the administration of HS in COVID‐19 patients with severe coronavirus disease is beneficial and that one possible antiviral mechanism is that HS molecules bind spike protein to block viral attachment or entry.[ 7 ] Due to the strong electrostatic interaction between the virus and HS,[ 6 ] the mimicking of this linear sulfated polysaccharide has been achieved using ligands containing sulfate or sulfonate pendent groups.[ 8 ] These findings render HS particularly interesting for viral treatment and the combination with GRMs offers two additional advantages: 1) when using surface‐functionalized GRMs, the local concentration of the targeting moieties is higher, resulting in an enhanced binding constant (multivalent effect),[ 9 ] that, as for other carbon nanoparticles, may reduce the therapeutically relevant concentration by different orders of magnitude;[ 10 ] 2) chemistry on GRMs allows multifunctionalization permitting the incorporation of different desirable groups (Figure 1). For these reasons, functionalized GRMs are now studied as antiviral drugs for medical therapies. For example, sulfonate groups have been introduced onto G, GO, and rGO through different chemical strategies. The most common one is through sp2 carbon derivatization. For instance, rGO functionalized with sulfonic acids (sulfanilic acid as precursor) have shown interesting antiviral activity against Herpes simplex Virus‐1 (HSV‐1).[ 11 ] To enhance the functionalization degree, more complex precursors have been grafted onto the GRM surface. Dendritic polyglycerol sulfate functionalized rGO has been prepared through epoxide ring opening polymerization of glycidol and sulfonation of the hydroxyl groups. This conjugate has shown similar efficacy to heparin in inhibiting Orthopoxviruses.[ 12 ] Due to similarity with the structure of HS, the inhibition effect could be enhanced by using a linear polyglycerol sulfonated rGO, instead of the dendritic polymer.[ 13 ] Another functionalization pathway well reported for the functionalization of G is the triazine derivatization proposed by the group of Haag.[ 14 ] This strategy allows an easy and elegant surface multifuctionalization of G flakes through a controlled derivatization of the triazine ring. This approach was used to introduce both sulfonated polyglycerols and long fatty chains onto G surface. In vitro results on HSV‐1 showed that a synergistic effect between electrostatic and hydrophobic interactions resulted in a high inhibition of infectivity.[ 14 ] These in vitro examples show how G, GO, and rGO can be ad hoc functionalized in order to reversibly bind viruses and boost the antiviral activity. The use sulfonic groups may be a key strategy to target in vivo different classes of viruses including SARS‐CoV‐2. However, due to the reversible nature of the binding mechanism to viral capsid, this approach can be used to slow down the infection (as for HS)[ 7 ] but not to eradicate the pathology. Indeed, following the binding thermodynamics, at low concentration the viral particles would be released from the nanomaterial surface causing the recurrence of the infection. Thus, the design of a system capable of irreversibly deactivating the viral machinery (virucidal activity) is timely and highly desirable (Figure 1). Pioneering works have demonstrated that G and GO may possess intrinsic antiviral activity resulting from their mechanical interaction with the viral capsid or the envelope.[ 15 , 16 ] However, this effect has not yet been confirmed, neither in vitro nor in vivo where the antiviral activity of non‐functionalized materials was found being negligible. This per se virucidal activity is present only when viruses are incubated with the materials prior to contact with host cells, making pristine G and GO interesting for surface disinfection or at the early stages of infection. The mechanism of virucidal effect is associated to the unique physical and chemical properties of GRMs. In particular, this virucidal activity has been attributed to the combination of three main factors: the physical insertion into the viral capsid, the negative surface charge, and the amphiphilic nature of the materials.[ 8 ] Therefore, by exploiting these characteristics it should be possible to prepare a wide spectrum antiviral agents. For instance, in silico studies have demonstrated that the exposure of Ebola virus to G leads to the poration of the protein protective shell, which induces the leakage of genomic RNA leading to virus inhibition.[ 15 ] Such virucidal activity was associated with the sharp, rigid structure together with the hydrophobic surface allowing G sheets to recognize and break VP40 protein–protein interactions. However, these studies have never been confirmed by further in vitro or in vivo assays. In this context, it is worth noting that, due to its highly hydrophobic nature, G is not colloidally stable in water and biological media, thus limiting its application. One possible solution to prepare water‐stable G dispersions is by using amphiphilic molecules during the graphite exfoliation process. Stable G water dispersions have been obtained using different classes of surfactants including biocompatible molecules such as proteins or sugars.[ 17 ] The surfactants used during the exfoliation process play an important role in the interaction of G with the viral surface, thus screening through modelling and in vitro tests would be very helpful to select the most efficient G‐based antiviral system. Conversely, GO exposes several oxygen‐containing groups onto its surface that renders it highly hydrophilic to form colloidally stable aqueous dispersions. The GO structure has been theorized as composed of oxygenated C sp3 islands surrounded by C sp2 hydrophobic areas.[ 5 ] This unique patchy amphipathic structure is associated with a high capacity to adsorb lipophilic and hydrophilic molecules onto its surface. GO is able to disrupt the cellular lipid bilayers, and interfere with protein–protein assemblies leading to unfolding.[ 18 ] For these reasons, GO is considered as being extremely promising as an antiviral material. A study demonstrated the ability of GO to denature and induce agglomeration of fragments of the viral protein R, which has a fundamental role on HIV1 host cell binding and uptake.[ 16 ] The interaction of GO with HIV1 fragments was associated with the hydrophobic interaction between the protein R and the nanomaterial that causes the conversion of the active α‐helix segments into β‐sheets promoting the formation of biologically inactive fibrillar agglomerates. GO was also used to treat enteric EV71 (e.g., pathogenic agents of hand, foot, and mouth disease) and H9N2 (e.g., gastrointestinal avian influenza A) demonstrating a strong capacity to absorb both viruses onto its surface and showing good virucidal activity.[ 19 ] In a comparative investigation using GO, rGO, graphite, and graphite oxide, it has been shown that GO and rGO display the highest virucidal activity against Pseudorabies virus, indicating that the nanosized dimension of the material plays a crucial role on their activity.[ 20 ] Additionally, covering of GO with cationic polymer causes a loss of antiviral activity, thereby highlighting the importance of negative charges present on GO surfaces.
Understanding the interaction of GRMs with viruses may help to conceive more efficient broad‐spectrum antiviral agents. Hitherto, there are no reports proving the virucidal activity of GRMs against SARS‐CoV‐2. It is worth noting that the viricidal activity is associated with different factors including surface polarity, size, and edges. Most of the performed tests did not consider the biological media and/or the presence of protein corona. Coronation is a process that takes place between serum proteins and GRMs flakes, which may irreversibly alter the materials surface chemistry causing an attenuation or a drop of the virucidal activity. Additionally, the impact of surface functionalization has not been exhaustively explored yet. The grafting of targeting molecules (e.g., ACE2, 3CLpro, S Protein)[ 21 ] may enhance the binding capacity of the nanomaterial and hence improve the specificity and the virucidal activity against SARS‐CoV‐2. In this context, another strategy is to vehicle antiviral drugs through GRMs for the selective delivery to targeted tissues and organs. GRMs combine good biocompatibility, longer blood circulation capacity and good permeability through biological barriers and membranes thus offering outstanding drug delivery capacity. For instance, GO can efficiently release hypericin (e.g., an antiviral drug) into duck‐reovirus‐infected ducklings preventing pathological lesions and decreasing the viral load in the organs with a consequent prolonged animal survival.[ 22 ] Multifuctionalization strategies have been also developed for the preparation of virucidal GRMs. GO modified with sulfanilic acid and β‐cyclodextrin displayed high binding efficacy to respiratory syncytial virus and incorporation of curcumin, a powerful lipophilic antiviral drug capable of inducing oxidation of the capsids.[ 23 ]
Another interesting but less developed approach regards the virucidal activity of G, GO, and rGO through phototherapy. The inherent photothermal activity of G, GO, and rGO was successfully proved in cancer therapy.[ 5 ] In this context, G, GO, and rGO are able to efficiently convert NIR light into heat, generating a local and rapid temperature increase that induces cell ablation. The same strategy can be applied for virucidal purposes where the virus upon adsorption onto the nanomaterial surface can be irreversibly damaged by heat.[ 24 ] The possibility of preparing antiviral films that can be easily regenerated upon harmless NIR light might be an easy, versatile, and cost‐effective solution for the next pandemics.
3. Antiviral Surfaces and Coatings
Many studies have been devoted to evaluating the resistance of viruses under different environmental conditions. For instance, exhaled viral particles remain infective even 3 h after exhalation.[ 25 , 26 ] Beside aerosols and direct contact, one the most plausible mechanisms of transmission for SARS‐CoV‐2 and other pathogens is through fomites. Fomites are inanimate items that can transfer the pathogens to a new host through unexpected transfer by contact with virus on inanimate objects. Common examples of fomites are door handles, lift buttons, ATM machine touchscreens, shared equipment in workplaces, etc. In a recent study, Riddell et al. have analyzed the survival rates of the SARS‐CoV‐2 in materials present in our everyday life including stainless steel, polymer and paper banknotes, cotton, and vinyl.[ 27 ] Remarkably, SARS‐CoV‐2 remains active in non‐porous substrates up to 28 days when it is incubated in the dark, at 20 °C, at 50% relative humidity. The persistence in cotton (i.e., a highly porous material) was markedly shorter. Despite the variations due to the experimental conditions (e.g., viral titer, relative humidity, tested materials), these results are in line with other reports on the topic,[ 28 ] which underlined the need for constant sanitization and hand‐washing, calling for investigations to develop new functional coatings that help to shorten the persistence of pathogens. A recent perspective article highlighted the importance of the development of efficient antimicrobial surfaces, with particular emphasis on polymeric materials due to their constant presence in our everyday life.[ 29 ] Common strategies to boost the antimicrobial properties rely on the use of metal nanoparticles (e.g., CuO, Ag, or Au nanoparticles) or the exploitation of surface chemistry to tune the surface–pathogen interface. With sufficient adjustment, the strategies developed against bacteria could be translated to viruses. Chemical approaches leading to modify surfaces with pendent cationic, anionic heads or hydrophobic chains have been reported to inactivate viruses via different mechanisms. For example, polyethyleneimines (PEI) were exploited to prepare glass slides with different surface chemistry and to evaluate their antiviral activity against influenza viruses.[ 30 ] Significant differences were found related to the length of the alkyl chains and the quaternization of amines. We would like to underline that the growing chemistry of 2D materials could find application in the development of antiviral coatings with ad hoc functional groups. Recently, we reported the preparation of GO with branched PEI to study the complexation of siRNA,[ 31 ] or for water remediation.[ 32 ] These studies might inspire the use of PEI‐modified GO as antiviral surface coatings.
A key aspect for the realization of smart coatings based on GRMs concerns its integration into the actual surfaces (Figure 2 ). Extensive research effort has been dedicated to the development of GRM inks or dispersions with high processability. Application of these inks to surfaces can be done by a number of techniques such as dip‐coating, (ink‐jet) printing, screen printing, spray coating, roll‐to‐roll processing, etc.[ 33 ] The chemical nature of the surface and the ink to be deposited determine which technique suits best. For instance, spray coating might be the preferred option when transparency is required,[ 34 ] while the pad‐dry process works better for industrial‐scale coating of textiles as reported in a work describing the development of e‐textiles.[ 35 ] In this study, GO was simultaneously reduced and non‐covalently functionalized with a hydrophilic polymer to avoid the re‐stacking of the flakes. The adequate control of the chemical functionalization of 2D sheets opens the door to their use in virtually any media. This could enable their dispersion in precursor matrices of certain polymeric substrates. For example, the covalent functionalization of rGO with silsesquioxane moieties allowed their integration into electrospun fibers.[ 36 ] The electrospinning process in the preparation of textiles, filters, wound dressing materials, etc. has shown promising results in recent years.[ 37 , 38 ] Integration of GRMs into electrospun mats was beneficial in many applications as shown in the case of poly(lactide‐co‐glycolide/chitosan) fibers decorated with GO functionalized with Ag nanoparticles in a postmodification step with strong antibacterial activity against several tested microorganisms.[ 39 ]
Figure 2.
Chemical functionalization of GRMs to generate antiviral coatings on fomites. The inset box illustrate several common techniques used for GRMs deposition. Adapted with permission.[ 33 ] Copyright 2019, Wiley‐VCH.
While waiting for an effective medical treatment or the herd immunity achieved with the new and upcoming vaccines against SARS‐CoV‐2, the use of face‐masks is among the most effective precautions to be exploited in order to avoid the spread of viral particles when social distancing is not possible. As a consequence, millions of surgical masks are daily produced, used and disposed around the globe causing a strong environmental impact.[ 40 ] Research activities are being directed toward the reutilization of masks after decontamination by microwave irradiation or dry heating.[ 41 ] The multifaceted GRMs can play an important role to mitigate this massive use of resources. For instance, the superhydrophobicity of laser‐induced graphene (LIG) was exploited to develop self‐cleaning recyclable masks.[ 42 ] LIG is produced after the irradiation of commercial polymers (i.e., polyimides) usually with a CO2 laser. This is a photothermal process driven by the high pressure and temperatures around the laser spot.[ 43 , 44 ] In this case, LIG synthesis was adapted to preserve the integrity of the masks. The process provided superhydrophobic masks and can be easily brought to large‐scale manufacturing. Besides, the strong light absorption and superhydrophobicity of LIG would permit the deactivation of most pathogens by only exposing it to the sun light. The strong light response would allow the recycling of the masks in solar steam generators. In another work, the health protection capacity of commercial melt‐blown fabrics (MBF) and commercial masks with activated carbon fiber (ACF) was compared to LIG.[ 45 ] Escherichia coli was used as a model microorganism with an inactivation temperature over 60 °C. Interestingly, LIG showed 81.57% intrinsic antibacterial activity in contrast to 9.13% and 2.0% of MBF and ACF, respectively. After 10 min of light irradiation, LIG films reached 62 °C, which resulted in an enhanced antibacterial activity of 99.998%, while ACF reached only 52 °C and 67.24% activity. On the other hand, MBF fibers reached only 36 °C, yet its activity increased to 85.3%, suggesting that temperature is not the only important factor.
The use of graphene in masks was not limited to LIG. For instance, a prototype filter was designed as a disposable part of reusable respiratory masks.[ 46 ] The structural part of the filter was composed of multilayers of electrospun poly(lactic acid) and cellulose acetate. By adding CuO nanoparticles and GO in a postprocessing step using ultrasonication, the mechanical, antiviral, and antibacterial properties were enhanced. Photothermal activity of LIG is expected also for other GRMs. Remarkably, LIG grows firmly on substrates, which envisions certain stability. The stability of GRM masks thereof will depend on the kind of GRMs and the deposition technique, being key factors in view of the incorporation of GRM‐containing masks into the market.
4. (Bio)Sensing
A recent mathematical study has suggested the benefits of proper testing, tracing, and isolation and quarantine of the population to control the spread of viral infections.[ 47 ] Unfortunately, scenarios with confirmed cases around 20 000 per day would require quarantining of over 500 000 people ideally for two weeks. Such circumstances impose the need to carry out a vast number of tests every single day. Alongside, the silent state of the infection leads to fast propagation of the virus, which is in contrast with the slow available testing methodologies. Current testing relies mainly on the use of reverse transcription polymerase chain reaction (RT‐PCR). Testing by means of this method requires a few hours and the intervention of trained personnel at every step of the analysis including sampling, sample treatment, and data interpretation. Consequently, fast and reliable methods without the use of sophisticated equipment or trained staff at a reasonable cost are needed for proper triage of SARS‐CoV‐2 in the current pandemic.
The electronic properties, high surface area‐to‐volume ratio, and the ultrahigh response to changes in the environment make GRMs ideal candidates for the development of novel and highly sensitive sensors associated with SARS‐CoV‐2 and other viruses. Sensing using GRMs and other 2D materials is a growing field that has provided very promising results in recent years.[ 48 , 49 ] The available know‐how is serving as a solid starting point for the development of new sensors. The process for the development of GRM‐based sensors is illustrated in Figure 3 . To the best of our knowledge, Seo et al. reported for the first time the use of G for the detection of SARS‐CoV‐2 in nasopharyngeal swab specimens.[ 50 ] In this work, CVD G was transferred onto SiO2/Si substrates and patterned into linear shapes with photolithography before growing top Au/Cr electrodes. SARS‐CoV‐2 spike antibody was then immobilized onto G using a bifunctional linker containing pyrene that binds to G via π–π stacking interaction, and an activated carboxylic acid to form a stable amide bond with the antibody. The use of the antibody enabled the specific detection of the spike protein, a transmembrane protein that differs from one coronavirus to another. This device architecture permitted the evaluation of the field‐effect transistor biosensing response to three different samples including the SARS‐CoV‐2 antigen protein, the cultured SARS‐CoV‐2, and real clinical samples. Interestingly, the authors reported a limit of detection (LOD) of 1 or 100 fg mL−1 of protein in PBS or universal transport medium, respectively, 1.6 × 101 pfu mL−1 of the cultured virus with a linear response up to 1.6 × 104 pfu mL−1. Besides, Mers‐CoV spike protein gave no response, suggesting high specificity of the biosensor. Although the high complexity of the clinical samples provided a signal with high noise background, it was possible to distinguish between positive and negative patients. The LOD in nasopharyngeal swabs was 242 copies per mL, which is close to the ≈100 copies per mL of the most performing RT‐PCR.[ 51 ]
Figure 3.
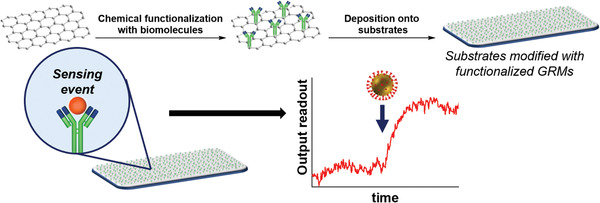
Functionalization of GRMs with specific biomolecules for the sensing of SARS‐CoV‐2. GRMs derivatization can be done either before or after the deposition onto the substrates. Most of the sensing events described so far include the interaction of a specific biomolecule with functionalized GRMs. The first attempts mainly included sensors with electrical readout, yet other outputs (e.g., spectroscopic signal) are foreseen.
Alternatively, G‐based electrochemical biosensors are attracting a lot of attention for SARS‐CoV 2 detection. The integration of these biosensors into electrochemical devices has evolved rapidly in recent years allowing the fabrication of point‐of‐care testing of relevant biomolecules.[ 52 , 53 ] A supersandwich‐type electrochemical device able to detect RNA of SARS‐CoV‐2 without nucleic acid amplification or reverse transcription was recently developed.[ 54 ] In this type of sensors, the target oligonucleotide sequence hybridizes with the capture probe and the signal probe (i.e., sandwich), which hybridizes in turn with another extreme of the target strand forming long concatamers (i.e., supersandwich) that amplify the electrochemical signal. A sophisticated synthetic approach was applied to prepare the active material, which is composed of Au@Fe3O4 nanoparticles decorated with a short oligonucleotide capture probe for the viral RNA, and rGO functionalized with Au nanoparticles binding the labelled signal probe. In addition, the basal plane of rGO was functionalized with p‐sulfocalix[8]arene hosting toluidine B to improve the electrochemical signal. The electrochemical response to the SARS‐CoV‐2 RNA was measured by differential pulse voltammetry using an electrochemical workstation operated by a smartphone. Remarkably, a LOD of 200 copies per mL was achieved with this sensor, which outperforms the LOD for viral RNA reported in the literature, and was suitable for different clinical samples including throat swabs, saliva, urine, sputum, etc. Moreover, the sensor displayed no significant response when it was exposed to samples from other viruses (e.g., Mers‐CoV, influenza A, rhinoviruses, etc.), which confirmed its high specificity and selectivity.
Beside the virus itself, the detection of other biomarkers associated with the disease is of vital importance to control a pandemic. The identification and early detection of biomarkers can anticipate dramatically the diagnosis with the consequent increase of survival rate in many diseases. In this regard, high levels of C‐reactive protein have been found in patients with severe symptoms of COVID‐19.[ 55 ] Moreover, the presence or the absence of the specific immunoglobulins IgM and IgG provides relevant information on the immune response of the tested individuals. A smart‐solution to detect the spike protein of SARS‐CoV‐2, the associated immunoglobulins, and the C‐reactive protein in either blood or saliva using a multiplexed device based on functionalized G was reported.[ 56 ] To construct the device, a four‐channel LIG electrode was engraved onto polyimide with a CO2 laser. Pyrenebutyric acid was used as a linker for the modification of each working electrode with the specific reagent for the corresponding biomarker. Once assembled, the performance of the multiplexed device was measured by amperometry against Ag/AgCl reference electrode fabricated by electrodeposition. Incubation time as short as 10 min was enough to obtain a linear response within a physiologically relevant range. Furthermore, the sensor was integrated in a device powered by a small lithium‐ion polymer battery. The device was able to measure and send the data simultaneously via Bluetooth technology. The applicability of the device in real serum and saliva samples was proven both for positive and negative cases. The potential use of non‐invasive sampling (i.e., saliva), together with the fast response and good performance without sophisticated laboratory equipment or trained personnel at a reasonable price make this pioneer device a strong candidate for fast patient triage to block the spread of the virus.
In most of the described works, a strong focus was given to achieve high specificity and selectivity for SARS‐CoV‐2, which was assessed by exposing the sensors to different viruses. A completely different approach was proposed by Hashemi et al.[ 57 ] The authors put the focus on the electrochemical sensing of glycoproteins by differential pulse voltammetry. The active material consisted of functionalized GO decorated with Au nanostars deposited on glassy carbon electrodes. This biosensor provided low LOD for SARS‐Cov‐2, infectious bronchitis virus, avian influenza, and two strains of Newcastle disease virus. The high performance was attributed to the high adsorption capacity of functionalized GO and the electron transfer mediated by the Au nanostars. The test could be done quickly using saliva. Interestingly, the differential pulse voltammetry displayed a different pattern for all the tested viruses. This is of high importance since distinguishing between the SARS‐CoV‐2 and other viruses is frequently a challenging task because of the similarity of the symptoms. The concept developed here could be of extreme importance to discriminate people infected with concomitant viruses (e.g., SARS‐CoV‐2 and influenza in winter) and stop the spread of dangerous pathogens.
Yet, novel sensors should not be limited to biomarkers. For instance, a high detection rate of SARS‐CoV‐2 was achieved through the analysis of forced cough recording with artificial intelligence.[ 58 ] GRM‐based architectures responsive to sound[ 59 ] or pressure[ 60 ] could find application here. In addition, integration of 2D‐material‐based thermometers into wearables could constantly measure the temperature of an individual. Sensors based on 2D materials could find application to assess the state of personal protective equipment like masks. Playing with the experimental conditions for the synthesis of LIG, hygroelectric devices were integrated into surgical masks.[ 45 ] The devices consisted of films of LIG where the hydrophilicity (i.e., the level of oxidation) changed gradually along the device. This generates a gradient of protons in the presence of moisture that induces a measurable electric field. The performance of this device decreases significantly with the accumulation of bacteria, and therefore it could be used to evaluate the masks conditions.
5. Conclusion and Outlooks
Unexpected problems require unexpected solutions. The outbreak of the current pandemic is urging scientists to be creative in the development of novel strategies to fight against the spread of SARS‐CoV‐2. Such circumstances impose (more than ever) a strong collaboration between laboratories with different background. Here, we have provided some examples demonstrating that thanks to their chemical and physical properties, GRMs can be key components offering innovative solutions in antiviral research. When suitably functionalized GRMs are able to interfere with viral replication machinery, reduce viral survival on surfaces, and selectively and sensibly detect capsid proteins making them powerful allies against this and future pandemics.
On the short‐term, by taking advantage of the industrial‐scale production of 2D materials, antiviral surfaces and coatings as well as sensors based on GRMs can be realized by making use of the tools described above. Alongside, appropriate chemical modifications should be exploited to enhance the functional control over these materials. The use of cost‐effective coating techniques may allow large‐area GRM deposition on surfaces of different nature. Fine control of the chemistry of GRMs, preferably through stable covalent bonding, should be directed to generate robust coatings to expand their utility life (e.g., resistance to washing or to ambient exposure). The ideal coatings should present broad‐spectrum antimicrobial activities both for harmful viruses and bacteria. A thorough evaluation against real pathogens would therefore be required. However, access to these assays is generally limited and they require a lot of resources. In addition, while the design of new GRMs can be developed in different research groups, we must keep in mind that only few labs are equipped with access to the pathogenic viruses and so can study the efficacy of GRMs as antiviral materials. Therefore, calls for new collaborations, establishment of excellence specialized scientific clusters and financial support to innovative research initiatives are mandatory for an efficient fight against new viral pandemics.
The remarkable results on the sensing of SARS‐CoV‐2 using GRMs point to a promising future. Importantly, the approaches developed for graphene may be exploited with other 2D materials (e.g., MoS2) offering a broader choice of physical and chemical properties, toward the design of new sensors beyond GRMs. Most of the proposed strategies involved the use of non‐covalent chemistry to immobilize large biomolecules on the flat surface of 2D materials. Consequently, key aspects such as the reproducibility during the large‐scale production and the stability of the devices over time has to be assessed before bringing these sensors to the market. Besides, the sensing platforms should be also versatile enough to be adapted to the possible mutations of the virus.
Application of GRMs and in general of hard nanomaterials in drug delivery has to be considered in a long‐term perspective. Multifunctionalization strategies and antiviral efficacy must be still deepened. Additionally, there are different concerns about biocompatibility and long‐term toxicity of 2D materials. Although studies are already available,[ 61 ] the toxicological risks of GRMs are not completely cleared. In this context, we solicit the scientific community work for the standardization of the tested GRMs in order to obtain robust answers for their safe applications in any field. Different hard nanomaterials are now clinically approved for human treatment,[ 62 ] and we hope that some of the GRMs will be soon included into this list. Despite the current extraordinary results, GRMs have been only marginally explored in viral infections. In particular, their potential in blocking cell uptake, inhibiting viral replication, and alerting the immune system has not been unleashed yet. There is still a long way to go before establishing GRMs as optimal solution to combat SARS‐CoV‐2. However, by considering the huge potential held by such unique materials in view of their unique properties, which can be further modulated via controlled functionalization, one can foresee a bright future for GRMs in the fight against the current COVID‐19 and future pandemics.
Conflict of Interest
The authors declare no conflict of interest.
Acknowledgements
G.R. and D.I. contributed equally to this work. This work was supported the Agence Nationale de la Recherche (ANR) through the LabEx project Chemistry of Complex Systems (ANR‐10‐LABX‐0026_CSC), the Centre National de la Recherche Scientifique (CNRS), the International Center for Frontier Research in Chemistry (icFRC), the EC through the Graphene Flagship Core 3 project (GA‐881603), and the ERC project SUPRA2DMAT (GA‐833707) as well as the Institut Universitaire de France (IUF).
Biographies
Giacomo Reina received his Ph.D. in 2016 at the University of Roma “Tor Vergata” under the supervision of Prof. Silvia Orlanducci and Prof. Maria Letizia Terranova. Afterward, he joined the group of Dr. Alberto Bianco at the Institut de Biologie Moléculaire et Cellulaire, CNRS in Strasbourg as a postdoctoral researcher. In 2020, he was awarded with the JSPS scholarship in the group of Prof. Naoki Komatsu at the University of Kyoto. His research interests focus on preparation, functionalization and supramolecular interactions of carbon nanomaterials for imaging, therapeutic, and diagnostic applications.

Daniel Iglesias obtained his Masters degree in 2013 at the University of Castilla‐La Mancha. After that, he moved to the University of Trieste to do his Ph.D. under the supervision of Prof. Maurizio Prato and Prof. Silvia Marchesan that he defended in 2017. After a postdoctoral experience in Trieste, in 2019 he moved to the Institut de Science et d'Ingénierie Supramoléculaires at the University of Strasbourg to work in the team of Prof. Paolo Samorì, where he is currently working as postdoctoral researcher. His research interests focus on the use of organic and supramolecular chemistry to develop new functional nanomaterials with application in sensing, electronics, and catalysis.

Paolo Samorì is Distinguished Professor and Director of the Institut de Science et d'Ingénierie Supramoléculaires at the Université de Strasbourg. He is interested in unravelling the architecture versus function relationship in tailor‐made, multifunctional materials and devices based on hybrid systems for applications in (bio)sensing, optoelectronics and energy storage.

Alberto Bianco received his Ph.D. in 1996 from the University of Padova. As a visiting scientist, he worked at the University of Lausanne, the University of Tübingen (as an Alexander von Humboldt fellow), the University of Padova, and Kyoto University. He is currently Research Director at the CNRS in Strasbourg. His research interests focus on the design of multifunctional carbon‐based nanomaterials for therapy, diagnostics, and imaging.

Reina G., Iglesias D., Samorì P., Bianco A., Graphene: A Disruptive Opportunity for COVID‐19 and Future Pandemics?. Adv. Mater. 2021, 33, 2007847. 10.1002/adma.202007847
Contributor Information
Paolo Samorì, Email: samori@unistra.fr.
Alberto Bianco, Email: a.bianco@ibmc-cnrs.unistra.fr.
References
- 1. Palmieri V., Papi M., Nano Today 2020, 33, 100883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Almaghaslah D., Kandasamy G., Almanasef M., Vasudevan R., Chandramohan S., Int. J. Clin. Pract. 2020, 74, e13637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Wick P., Louw‐Gaume A. E., Kucki M., Krug H. F., Kostarelos K., Fadeel B., Dawson K. A., Salvati A., Vázquez E., Ballerini L., Tretiach M., Benfenati F., Flahaut E., Gauthier L., Prato M., Bianco A., Angew. Chem., Int. Ed. 2014, 53, 7714. [DOI] [PubMed] [Google Scholar]
- 4. Bianco A., Cheng H. M., Enoki T., Gogotsi Y., Hurt R. H., Koratkar N., Kyotani T., Monthioux M., Park C. R., Tascon J. M. D., Zhang J., Carbon 2013, 65, 1. [Google Scholar]
- 5. Reina G., González‐Domínguez J. M., Criado A., Vázquez E., Bianco A., Prato M., Chem. Soc. Rev. 2017, 46, 4400. [DOI] [PubMed] [Google Scholar]
- 6. Cagno V., Tseligka E. D., Jones S. T., Tapparel C., Viruses 2019, 11, 596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Liu J., Li J., Arnold K., Pawlinski R., Key N. S., Res. Pract. Thromb. Haemostasis 2020, 4, 518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Reina G., Peng S., Jacquemin L., Andrade A. F., Bianco A., ACS Nano 2020, 14, 9364. [DOI] [PubMed] [Google Scholar]
- 9. Qi Z., Bharate P., Lai C. H., Ziem B., Böttcher C., Schulz A., Beckert F., Hatting B., Mülhaupt R., Seeberger P. H., Haag R., Nano Lett. 2015, 15, 6051. [DOI] [PubMed] [Google Scholar]
- 10. Luczkowiak J., Muñoz A., Sánchez‐Navarro M., Ribeiro‐Viana R., Ginieis A., Illescas B. M., Martín N., Delgado R., Rojo J., Biomacromolecules 2013, 14, 431. [DOI] [PubMed] [Google Scholar]
- 11. Sametband M., Kalt I., Gedanken A., Sarid R., ACS Appl. Mater. Interfaces 2014, 6, 1228. [DOI] [PubMed] [Google Scholar]
- 12. Ziem B., Thien H., Achazi K., Yue C., Stern D., Silberreis K., Gholami M. F., Beckert F., Gröger D., Mülhaupt R., Rabe J. P., Nitsche A., Haag R., Adv. Healthcare Mater. 2016, 5, 2922. [DOI] [PubMed] [Google Scholar]
- 13. Ziem B., Rahn J., Donskyi I., Silberreis K., Cuellar L., Dernedde J., Keil G., Mettenleiter T. C., Haag R., Macromol. Biosci. 2017, 17, 1600499. [DOI] [PubMed] [Google Scholar]
- 14. Donskyi I. S., Azab W., Cuellar‐Camacho J. L., Guday G., Lippitz A., Unger W. E. S., Osterrieder K., Adeli M., Haag R., Nanoscale 2019, 11, 15804. [DOI] [PubMed] [Google Scholar]
- 15. GC J. B., Pokhrel R., Bhattarai N., Johnson K. A., Gerstman B. S., Stahelin R. V., Chapagain P. P., Biochem. Biophys. Res. Commun. 2017, 493, 176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Zhang M., Mao X., Wang C., Zeng W., Zhang C., Li Z., Fang Y., Yang Y., Liang W., Wang C., Biomaterials 2013, 34, 1383. [DOI] [PubMed] [Google Scholar]
- 17. Paredes J. I., Villar‐Rodil S., Nanoscale 2016, 8, 15389. [DOI] [PubMed] [Google Scholar]
- 18. Reina G., Ruiz A., Murera D., Nishina Y., Bianco A., ACS Appl. Mater. Interfaces 2019, 11, 7695. [DOI] [PubMed] [Google Scholar]
- 19. Song Z., Wang X., Zhu G., Nian Q., Zhou H., Yang D., Qin C., Tang R., Small 2015, 11, 1171. [DOI] [PubMed] [Google Scholar]
- 20. Ye S., Shao K., Li Z., Guo N., Zuo Y., Li Q., Lu Z., Chen L., He Q., Han H., ACS Appl. Mater. Interfaces 2015, 7, 21571. [DOI] [PubMed] [Google Scholar]
- 21. Liu C., Zhou Q., Li Y., Garner L. V., Watkins S. P., Carter L. J., Smoot J., Gregg A. C., Daniels A. D., Jervey S., Albaiu D., ACS Cent. Sci. 2020, 6, 315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Du X., Xiao R., Fu H., Yuan Z., Zhang W., Yin L., He C., Li C., Zhou J., Liu G., Shu G., Chen Z., Mater. Sci. Eng., C 2019, 105, 110052. [DOI] [PubMed] [Google Scholar]
- 23. Yang X. X., Li C. M., Li Y. F., Wang J., Huang C. Z., Nanoscale 2017, 9, 16086. [DOI] [PubMed] [Google Scholar]
- 24. Deokar A. R., Nagvenkar A. P., Kalt I., Shani L., Yeshurun Y., Gedanken A., Sarid R., Bioconjugate Chem. 2017, 28, 1115. [DOI] [PubMed] [Google Scholar]
- 25. van Doremalen N., Bushmaker T., Morris D. H., Holbrook M. G., Gamble A., Williamson B. N., Tamin A., Harcourt J. L., Thornburg N. J., Gerber S. I., Lloyd‐Smith J. O., de Wit E., Munster V. J., N. Engl. J. Med. 2020, 382, 1564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Smither S. J., Eastaugh L. S., Findlay J. S., Lever M. S., Smither S. J., Eastaugh L. S., Findlay J. S., Lever M. S., Emerging Microbes Infect. 2020, 9, 1415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Riddell S., Goldie S., Hill A., Eagles D., Drew T. W., Virol. J. 2020, 17, 145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Fernández‐Raga M., Díaz‐Marugán L., García Escolano M., Bort C., Fanjul V., Environ. Res. 2021, 192, 110293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Xue X., Ball J. K., Alexander C., Alexander M. R., Matter 2020, 3, 1433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Liu H., Elkin I., Chen J., Klibanov A. M., Biomacromolecules 2015, 16, 351. [DOI] [PubMed] [Google Scholar]
- 31. Chau N. D. Q., Reina G., Raya J., Vacchi I. A., Ménard‐Moyon C., Nishina Y., Bianco A., Carbon 2017, 122, 643. [Google Scholar]
- 32. Pakulski D., Czepa W., Witomska S., Aliprandi A., Patroniak V., Ciesielski A., Samorì P., J. Mater. Chem. A 2018, 6, 9384. [Google Scholar]
- 33. Witomska S., Leydecker T., Ciesielski A., Samorì P., Adv. Funct. Mater. 2019, 29, 1901126. [Google Scholar]
- 34. Aliprandi A., Moreira T., Anichini C., Stoeckel M. A., Eredia M., Sassi U., Bruna M., Pinheiro C., Laia C. A. T., Bonacchi S., Samorì P., Adv. Mater. 2017, 29, 1703225. [DOI] [PubMed] [Google Scholar]
- 35. Karim N., Afroj S., Tan S., He P., Fernando A., Carr C., Novoselov K. S., ACS Nano 2017, 11, 12266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Guo Y., Xu G., Yang X., Ruan K., Ma T., Zhang Q., Gu J., Wu Y., Liu H., Guo Z., J. Mater. Chem. C 2018, 6, 3004. [Google Scholar]
- 37. Arrieta M. P., Peponi L., Rodríguez‐Hernández J., in Materials for Biomedical Engineering: Bioactive Materials for Antimicrobial, Anticancer, and Gene Therapy (Eds: Holban A.‐M., Grumezescu A. M.), Elsevier, Amsterdam, The Netherlands: 2019, pp. 53–76. [Google Scholar]
- 38. Gao Y., Truong Y. B., Zhu Y., Kyratzis I. L., J. Appl. Polym. Sci. 2014, 131, 40797. [Google Scholar]
- 39. De Faria A. F., Shaulsky E., Chavez L. H. A., Elimelech M., ACS Appl. Mater. Interfaces 2015, 7, 12751. [DOI] [PubMed] [Google Scholar]
- 40. Klemeš J. J., Van Fan Y., Jiang P., Energy 2020, 211, 118701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Pascoe M. J., Robertson A., Crayford A., Durand E., Steer J., Castelli A., Wesgate R., Evans S. L., Porch A., Maillard J. Y., J. Hosp. Infect. 2020, 106, 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Zhong H., Zhu Z., Lin J., Cheung C. F., Lu V. L., Yan F., Chan C. Y., Li G., ACS Nano 2020, 14, 6213. [DOI] [PubMed] [Google Scholar]
- 43. Lin J., Peng Z., Liu Y., Ruiz‐Zepeda F., Ye R., Samuel E. L. G., Yacaman M. J., Yakobson B. I., Tour J. M., Nat. Commun. 2014, 5, 5714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Ye R., James D. K., Tour J. M., Acc. Chem. Res. 2018, 51, 1609. [DOI] [PubMed] [Google Scholar]
- 45. Huang L., Xu S., Wang Z., Xue K., Su J., Song Y., Chen S., Zhu C., Tang B. Z., Ye R., ACS Nano 2020, 14, 12045. [DOI] [PubMed] [Google Scholar]
- 46. Ahmed M. K., Afifi M., Uskoković V., J. Infect. Public Health 2020, 13, 1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Kucharski A. J., Klepac P., Conlan A. J. K., Kissler S. M., Tang M. L., Fry H., Gog J. R., Edmunds W. J., Emery J. C., Medley G., Munday J. D., Russell T. W., Leclerc Q. J., Diamond C., Procter S. R., Gimma A., Sun F. Y., Gibbs H. P., Rosello A., van Zandvoort K., Hué S., Meakin S. R., Deol A. K., Knight G., Jombart T., Foss A. M., Bosse N. I., Atkins K. E., Quilty B. J., Lowe R., Prem K., Flasche S., Pearson C. A. B., Houben R. M. G. J., Nightingale E. S., Endo A., Tully D. C., Liu Y., Villabona‐Arenas J., O'Reilly K., Funk S., Eggo R. M., Jit M., Rees E. M., Hellewell J., Clifford S., Jarvis C. I., Abbott S., Auzenbergs M., Davies N. G., Simons D., Lancet Infect. Dis. 2020, 20, 1151.32559451 [Google Scholar]
- 48. Anichini C., Czepa W., Pakulski D., Aliprandi A., Ciesielski A., Samorì P., Chem. Soc. Rev. 2018, 47, 4860. [DOI] [PubMed] [Google Scholar]
- 49. Menard‐Moyon C., Bianco A., Kalantar‐Zadeh K., ACS Sens. 2020, 5, 3739. [DOI] [PubMed] [Google Scholar]
- 50. Seo G., Lee G., Kim M. J., Baek S. H., Choi M., Ku K. B., Lee C. S., Jun S., Park D., Kim H. G., Kim S. J., Lee J. O., Kim B. T., Park E. C., Il Kim S., ACS Nano 2020, 14, 5135. [DOI] [PubMed] [Google Scholar]
- 51. Arnaout R., Lee R., Lee G. R., Callahan C., Yen C., Smith K., Arora R., Kirby J., bioRxiv 2020, 10.1101/2020.06.02.131144. [DOI] [Google Scholar]
- 52. Lawal A. T., Biosens. Bioelectron. 2018, 106, 149. [DOI] [PubMed] [Google Scholar]
- 53. da Silva E. T. S. G., Souto D. E. P., Barragan J. T. C., de F. Giarola J., de Moraes A. C. M., Kubota L. T., ChemElectroChem 2017, 4, 778. [Google Scholar]
- 54. Zhao H., Liu F., Xie W., Zhou T. C., OuYang J., Jin L., Li H., Zhao C. Y., Zhang L., Wei J., Zhang Y. P., Li C. P., Sens. Actuators, B 2021, 327, 128899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Wu C., Chen X., Cai Y., Xia J., Zhou X., Xu S., Huang H., Zhang L., Zhou X., Du C., Zhang Y., Song J., Wang S., Chao Y., Yang Z., Xu J., Zhou X., Chen D., Xiong W., Xu L., Zhou F., Jiang J., Bai C., Zheng J., Song Y., JAMA Intern. Med. 2020, 180, 934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Torrente‐rodrı R. M., Lukas H., Tu J., Xu C., Rossiter H. B., Gao W., Lukas H., Tu J., Min J., Yang Y., Matter 2020, 3, 1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Hashemi S. A., Behbahan N. G. G., Bahrani S., Mousavi S. M., Gholami A., Ramakrishna S., Firoozsani M., Moghadami M., Lankarani K. B., Omidifar N., Biosens. Bioelectron. 2021, 171, 112731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Laguarta J., Hueto F., Subirana B., IEEE Open J. Eng. Med. Biol. 2020, 1, 275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Tao L., Tian H., Liu Y., Ju Z., Pang Y., Chen Y., Wang D., Tian X., Yan J., Deng N., Yang Y., Ren T., Nat. Chem. 2017, 8, 14579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Huang C. B., Witomska S., Aliprandi A., Stoeckel M. A., Bonini M., Ciesielski A., Samorì P., Adv. Mater. 2019, 31, 1804600. [DOI] [PubMed] [Google Scholar]
- 61. Fadeel B., Bussy C., Merino S., Vázquez E., Flahaut E., Mouchet F., Evariste L., Gauthier L., Koivisto A. J., Vogel U., Martín C., Delogu L. G., Buerki‐Thurnherr T., Wick P., Beloin‐Saint‐Pierre D., Hischier R., Pelin M., Candotto Carniel F., Tretiach M., Cesca F., Benfenati F., Scaini D., Ballerini L., Kostarelos K., Prato M., Bianco A., ACS Nano 2018, 12, 10582. [DOI] [PubMed] [Google Scholar]
- 62. Nardecchia S., Sánchez‐Moreno P., de Vicente J., Marchal J. A., Boulaiz H., Nanomaterials 2019, 9, 191. [DOI] [PMC free article] [PubMed] [Google Scholar]


