Abstract
Graft cell repopulation and tendon-bone tunnel healing are important after allograft anterior cruciate ligament reconstruction (ACLR). Freshly isolated bone marrow mononuclear cells (BMMNCs) have the advantage of short isolation time during surgery and may enhance tissue regeneration. Thus, we hypothesized that the effect of intra-articular BMMNCs in post-allograft ACLR treatment is comparable to that of cultured bone marrow stromal cells (BMSCs). A rabbit model of hamstring allograft ACLR was used in this study. Animals were randomly assigned to the BMMNC, BMSC, and control groups. Fresh BMMNCs isolated from the iliac crest during surgery and cultured BMSCs at passage four were used in this study. A total of 1 × 107 BMMNCs or BMSCs in 100 µL phosphate-buffered saline were injected into the knee joint immediately after ACLR. The control group was not injected with cells. At two and six weeks post operation, we assessed graft cell repopulation with histological and cell tracking staining (PKH26), and tendon-bone healing with histological micro-computed tomography and immunohistochemical analyses for collagen I and monocyte chemoattractant protein-1 (MCP1). At two weeks post operation, there was no significant difference in the total cell population within the allograft among the three groups. However, the control group showed significantly higher cell population within the allograft than that of BM cell groups at six weeks. Histological examination of proximal tibia revealed that the intra-articular delivered cells infiltrated into the tendon-bone interface. Compared to the control group, the BM cell groups showed broader gaps with interfacial fibrocartilage healing, similar collagen I level, and higher MCP1 expression in the early stage. Micro-CT did not reveal any significant difference among the three groups. BMMNCs and BMSCs had comparable effects on cell repopulation and interfacial allograft-bone healing. Intra-articular BM cells delivery had limited benefits on graft cell repopulation and caused higher inflammation than that in the control group in the early stage, with fibrocartilage formation in the tendon-bone interface after allograft ACLR.
Keywords: ACL reconstruction, allograft, bone marrow mononuclear cells, bone marrow stromal cells, ligamentization, tendon to bone healing
1. Introduction
Knee injuries, particularly in the anterior cruciate ligament (ACL), are the most common sports-related injuries. Currently, ACL reconstruction (ACLR) with implanted graft is the standard treatment for ACL rupture. The major concerns after ACLR graft implantation are graft maturation and tendon-bone tunnel interfacial healing [1,2]. Cell repopulation is critical in all phases of tendon graft remodeling, and insufficient cell repopulation within the ACL graft may induce degeneration or micro-ruptures of the graft during the postoperative period [3]. Fast interfacial healing of grafted tendon and bone tunnel is needed to prevent graft pull-out and allow early joint rehabilitation. Therefore, accelerating cell repopulation in implanted grafts and tendon-bone tunnel interfacial healing are important for preventing graft failure, increasing graft strength, and improving clinical outcome of ACLR.
The types of grafts used in ACLR include autograft, allograft, and artificial graft. Autograft provides the benefit of faster incorporation without the risks of immune rejection and disease transmission. Nevertheless, autograft harvesting may lead to pain in anterior knee, patellar fracture, weakness of quadriceps, knee flexion weakness, and saphenous nerve injury [4,5]. The use of artificial graft in ACLR has a clinical outcome similar to that of autograft in short and mid-term follow up; however, the development of joint synovitis, high failure rate, and poor graft incorporation are some of the major concerns associated with its use [6,7]. Hence, the application of allograft in ACLR has become increasingly popular, as it provides the advantages of eliminating donor site morbidity, availability of multiple grafts, and shorter operative time [8]. However, the use of decellularized allograft tendons results in delayed healing due to insufficient population of living cells, slow migration of native cells, and inferior biomechanical strength compared to autografts [9,10]. Therefore, to enhance graft regeneration and tendon-bone interfacial healing, there is need to accelerate decellularized allograft revitalization with increased cellularity.
Cell therapy has been applied to enhance tissue regeneration, and bone marrow stromal cells (BMSCs) are the most commonly used cell source for allograft tendon regeneration both in vitro and in vivo [11,12]. Ning et al. [11] implanted rat BMSCs into decellularized tendons and showed that the implanted cells proliferated with high tenogenic gene expression. Lu et al. [12] seeded BMSCs through the lateral slit to revitalize the decellularized tendon and found that the BMSCs were redistributed in the whole decellularized tendon, which had higher collagen, tenogenic, and MMP gene expression than normal tendon. Nevertheless, there are limitations to the clinical application of BMSCs, such as the time and techniques needed for cell expansion prior to use, high cost, pathogen contamination, and risk of generic alterations [13]. Bone marrow mononuclear cells (BMMNCs), another attractive cell source for tissue regeneration, have the advantage of being easy to isolate from fresh bone marrow (BM) in a short time period (within one hour), without cell expansion. BMMNCs have been used in the treatment of myocardium infarction, limb ischemia, ischemia-reperfusion muscle injury, stroke, as well as tissue-engineered vascular grafts and bone defect reconstruction [14,15,16]. BMMNCs also possess the ability to adhere to biomaterials, promote angiogenesis, and enhance intrinsic healing through the production of cytokines [17,18,19]. To date, there are no reports in the literature that directly compare between the enhancing effects of intra-articular injection with BMMNCs and BMSCs after allograft ACLR.
For clinical application, the effect of cell therapy on enhancing cell repopulation and tendon-bone interfacial healing after allograft ACLR requires further understanding. We hypothesized that freshly isolated BMMNCs have similar or better effects on enhancing cellular repopulation and tendon-bone tunnel interfacial healing after allograft ACLR, compared to cultured BMSCs. In this study, we evaluated the effects of freshly isolated BMMNCs and cultured BMSCs on enhancement of allograft cell repopulation and interfacial healing in vivo in a rabbit model of ACLR using hamstring allograft.
2. Results
2.1. Analysis of Cell Repopulation in the Intra-Articular Graft
2.1.1. Cell Repopulation within Allograft
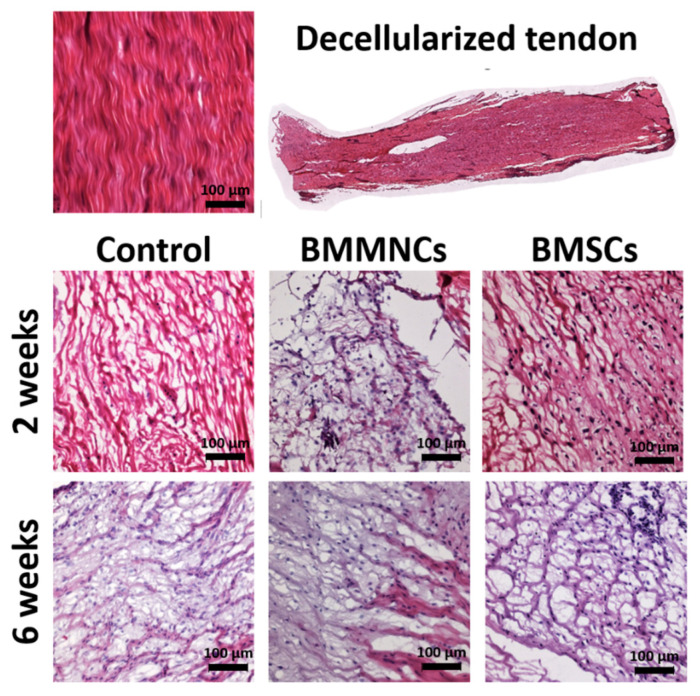
Before implantation, the decellularized tendon graft contained well-oriented fibers but no cells. Two weeks after ACLR, the grafts in the control, BMMNC, and BMSC groups had cells and disoriented collagen fibers. Six weeks after allograft implantation, all three groups showed abundant cells with improved collagen fiber alignment (Figure 1).
Figure 1.
Representative pictures of decellularized tendon graft in the control, BMMNC, and BMSC groups at 2 weeks and 6 weeks after operation. BMMNCs: Bone Marrow Mononuclear Cells; BMSCs: Bone Marrow Stromal Cells. Bar = 100 µm.
2.1.2. Higher Total Cell Population within Allograft in the Control Group Than in the Bone Marrow Cells-Seeded Groups
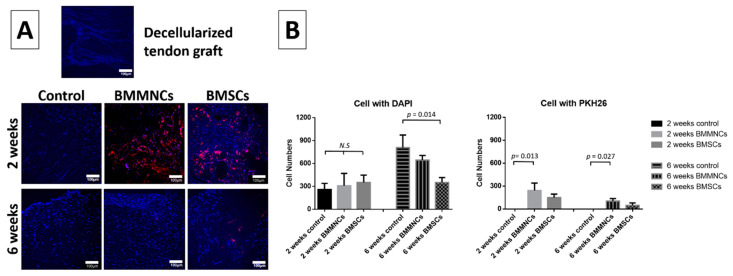
To monitor the injected cells in the allograft sites, we used the membrane-based cell tracker PKH26. The pre-implanted decellularized tendon graft showed limited cell nuclei (blue DAPI stain) and no injected cells (red PKH26 stain). The total cell population within allograft tissues increased in all three groups from two to six weeks (n = 3 per group). Although, the total cell number in the control population was lower than the BM cell-injected groups at two weeks post surgery, there was no significant difference. At six weeks post surgery, the control group had the highest total cell among the three groups. The number of injected cells (PKH26) decreased from two to six weeks (Figure 2). At two weeks, the fluorescent signals in the cells were detectable. After six weeks, cells with membrane-based trackers in the plasma membrane were diluted due to cell proliferation and the fluorescent signals were weaker than the threshold of detectable signal.
Figure 2.
(A) The histological pictures of DAPI/PKH26 staining in decellularized tendon graft, control, BMMNC, and BMSC groups. (B) The total cell number (DAPI; blue) and seeded cells (PKH26; red) in the control, BMMNC, and BMSC groups 2 and 6 weeks after operation. n = 3 per group. Blue: DAPI (cell nucleus); Red: PKH26 (seeded cells). White bar = 100 µm. N.S. indicate no statistically significant difference between groups.
2.2. Analysis of Tendon-Bone Tunnel Interfacial Healing in the Proximal Tibia
2.2.1. The Presence of Intra-Articular-Injected Bone Marrow Cells in the Interface between the Tendon-Bone Tunnel
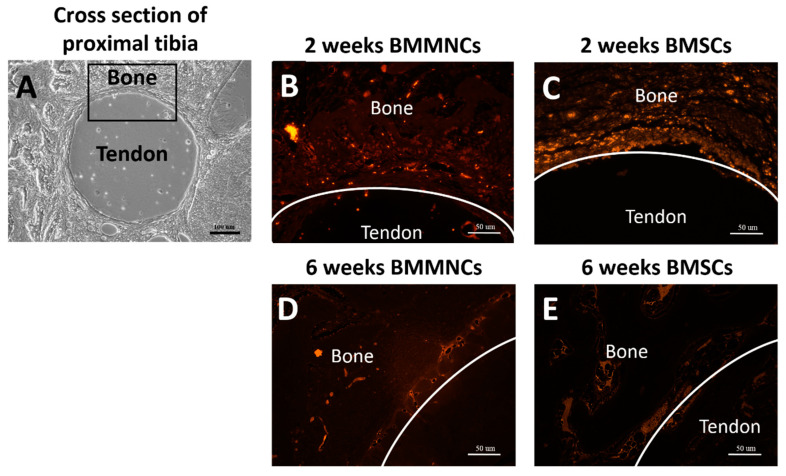
The intra-articular-injected BM cells (stained with PKH26 in red; BMMNCs (Figure 3B) and BMSCs (Figure 3C)) infiltrated into the interface between the tendon-bone tunnel after two weeks post operation. At six weeks post operation, few injected BM cells were found in the tendon-bone interface (Figure 3D,E).
Figure 3.
(A) Representative picture of the cross section of proximal tibia showed the implanted allograft tendon and surrounding bone. The black box demonstrated the observed area of tendon-bone interface. (B,C) Intra-articular-injected BMMNCs and BMSCs (PKH26 stained in red) were infiltrated in the tendon-bone interface two weeks after operation. (D,E) Few BMMNCs and BMSCs were found in the tendon-bone interface six weeks after operation. Black bar: 100 µm; White bar: 50 µm.
2.2.2. Interfacial Fibrocartilage Formation in the Tendon-Bone Tunnel in Bone Marrow Cells-Injected Groups
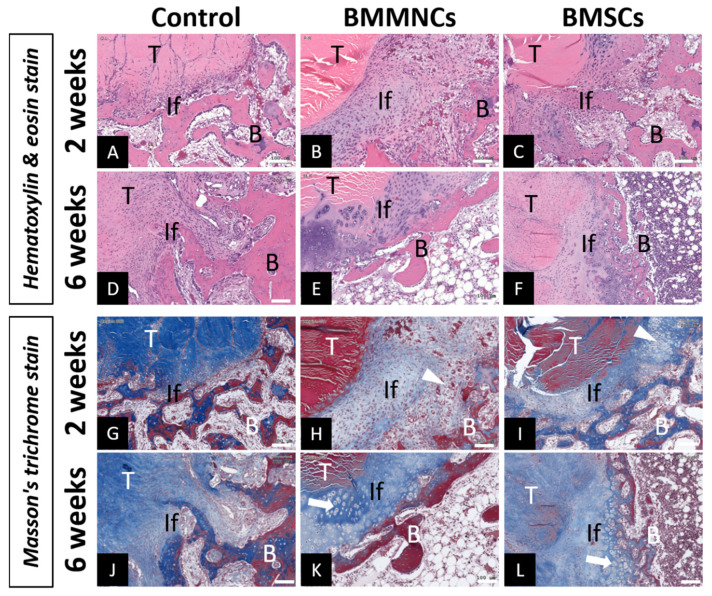
Compared to the relatively narrow interface (not quantified) in the control group, more cells infiltrated and formed localized areas of chondroid-like cells (Figure 4H (BMMNCs) and Figure 4I (BMSCs); white arrowhead) between the tendon-bone tunnel in the BM cell groups after two weeks post surgery. The dense tendon cortex was also visible at two weeks post surgery. At six weeks post surgery, the tendon cortex was unidentifiable in the control and BM cell injection (BMMNC and BMSC) groups, with different interfacial patterns. In the control group, collagen fiber infiltration into the grafted tendon was observed in the interface, indicating progressive maturation and reorganization of fibrous tissue. In contrast, broader fibrocartilage zone (Figure 4K (BMMNCs) and Figure 4L (BMSCs); white arrow) with collagen fiber infiltration was observed in the tendon-bone interface of the BMMNC and BMSC groups, indicating transitional healing. These findings were shown in Figure 4.
Figure 4.
Haematoxylin and eosin (H&E) and Masson trichrome staining of tendon-bone interface in the control, BMMNC, and BMSC groups. The control group showed smaller interface gaps at both two (A,G) and six weeks (D,J), and more maturated and reorganized collagen fibers penetrating the tendon at six weeks without obvious junction (D,J). The BM-injected groups presented broader interface with fibrocartilage healing at two weeks (B,C,H,I) and six weeks (E,F,K,L). White arrow head at two weeks (H,I) and white arrow at six weeks (K,L) indicate the fibrocartilage formation area. T: Allograft tendon; B: Bone; If: Interface; Bar = 100 µm.
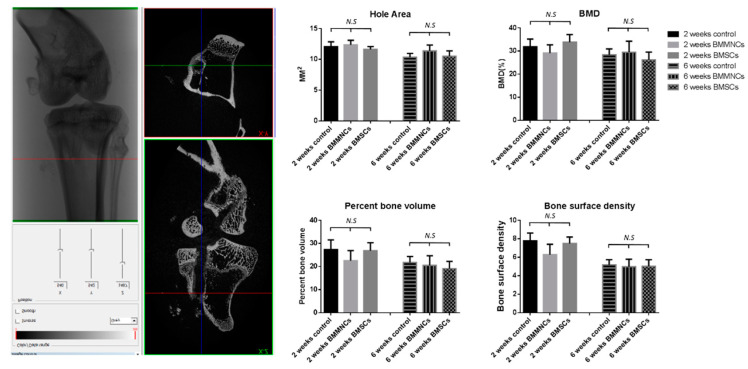
2.2.3. Micro-CT Revealed No Significant Differences between the Control and Bone Marrow Cells-Seeded Groups
Using micro-CT, we observed that the hole area decreased from two weeks to six weeks without significant difference between the control and BM cells-seeded groups. At both two and six weeks, no significant differences in BMD, percent bone volume, or bone surface density were observed among the three groups (Figure 5; n = 4 in control group, n = 5 in BM cells-injected groups at both two and six weeks).
Figure 5.
Micro-CT analysis of proximal tibia specimens. The figures show the result of bone tunnel hole area, BMD, percent bone volume, and bone surface density among the control, BMMNC, and BMSC groups at two and six weeks after operation. n = 4 in control group, n = 5 in BM cells-injected groups at both two and six weeks. N.S. indicate no statistically significant difference between groups.
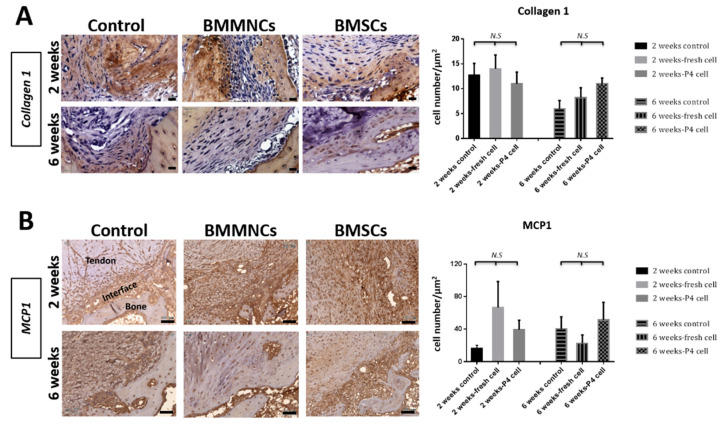
2.2.4. Upregulated MCP1 Expression in the Bone Marrow Cells-Seeded Groups
IHC revealed that the expression level of collagen I declined at six weeks compared to that at two weeks in all the three groups. There was no significant difference in collagen I expression level among the three groups at both two and six weeks (Figure 6A; n = 3 in each group at both two and six weeks). To investigate the inflammation status after injection with BM cells, we monitored the expression of MCP1, an important cytokine that modulates the infiltration and migration of macrophages/monocytes to the site of inflammation [20,21]. At two weeks, the BM cells-injected groups exhibited higher MCP1 expression levels than the control group without significant difference. At six weeks, the expression of MCP1 was variable among the three groups, but without significant difference (Figure 6B; n = 3 in each group at both two and six weeks).
Figure 6.
Representative immunohistochemistry images revealed collagen I (A) and MCP1 (B) expression in control, BMMNC, and BMSC groups at two and six weeks. Bar = 50 µm. n = 3 in each group at both two and six weeks. N.S. indicate no statistically significant difference between groups.
3. Discussion
Cell therapy has been proposed to improve graft maturation and tendon-bone tunnel healing after ACLR. However, the effects of bone marrow cells obtained from different sources in allograft ACLR are not well investigated. This study evaluated the enhancing effects of freshly isolated BMMNCs and cultured BMSCs on graft recellularization and tendon-bone interfacial healing in a rabbit allograft ACLR model. At two and six weeks post operation, BMMNC and BMSC showed similar capabilities of cell repopulation within the intra-articular allograft. However, in this study, we did not observe any benefit of intra-articular BM cell injection; indeed, the control group had a higher total cell population in the graft than the BM cells-injected groups at six weeks post operation. BMMNCs and BMSCs resulted in similar tendon-bone interfacial healing patterns with broader gap and progressive fibrocartilage formation, in contrast to fiber reorganization in the control group. IHC revealed similar collagen I levels and higher inflammation in the BM cells-injected groups than in the control group at two weeks post operation. Micro-CT showed no differences in the hole area and bone density among the three groups. These findings indicated that intra-articular injection of BM cells induced more inflammation in the early stages, with limited benefits in allograft cell repopulation, and promoted fibrocartilage transition zone development between the allograft tendon and bone tunnel.
The clinical outcomes of allograft in ACLR is controversial. For example, a systemic review comparing hamstring autograft and soft-tissue allograft in ACLR revealed no significant differences in the outcomes [22]. Another long-term follow-up (>10 years) study reported significantly higher ACLR failure rate for allograft (26.5%) compared to that for autograft (8.3%) [23]. Previous studies on the efficacy of cell therapy in animal models of ACLR have shown variable results owing to different factors, including cell sources, implanted graft (autograft, allograft, xenograft), applied cell numbers, animal species, studied area (graft or tendon bone interface), delivery time (immediate, days, or weeks after ACLR), and delivery method (intra-articular injection, intra-tunnel injection, or cell sheet) [24,25,26]. Our study focused on the use of intra-articular cell injection for cell repopulation in grafts and tendon-to-bone tunnel healing after allograft ACLR. In contrast to previous studies, our study focused on the use of freshly isolated bone marrow-derived cells, which are easy to prepare for one-time surgeries. In addition, we performed intra-articular injection immediately after operation to simulate clinical situations. Interestingly, intra-articular injection of both freshly isolated BMMNCs and cultured BMSCs exhibited similar effects in our study, with limited effect on graft cell repopulation and different interfacial fibrocartilage healing patterns compared to that of the control group, which was not injected with cells.
Clinical images and previous studies have shown that both recellularization and revascularization are delayed and biomechanical strength is inferior in allografts compared to autografts after ACLR [9,10,27]. In the rabbit allograft ACLR model used in our study, we injected BM-derived cells to allow self-proliferation and differentiation, and promote the secretion of paracrine factors that attract and enhance the activity and differentiation of surrounding cells. The cells present within the allograft not only limited the proliferation of the seeded cells but also the migration of surrounding cells. We found abundant cell repopulation in the allograft at six weeks compared to two weeks post operation in all three groups. After six weeks, plentiful cells were found in the intra-articular allograft tissue in the control group, indicating that surrounding cells migrated into the allograft tissue without the injection of exogenous cells. Interestingly, the total cell population was highest in the control group compared to BM cell groups at six weeks post operation. In this study, intra-articular BM cell injection did not enhance allograft cell repopulation, indicating that it did not significantly promote and indeed, might have even impaired peripheral cell migration into the allograft.
After ACLR, the grafted tendon was initially integrated with the fibrous tissue in the bone tunnel wall. The interfacial fibrous tissue progressively matured into Sharpey’s fibers in indirect healing or developed into layered chondral formations at the tendon-bone interface in direct healing [28]. BMSCs with different delivery methods enhanced the interfacial healing in the tendon-bone tunnel after ACLR through transitional fibrocartilage zone formation [24,29]. Soon et al. [29] applied fibrin sealant to Achilles tendon allograft coated with cultured autogenous P1 BMSCs and reported improved interfacial healing in the bone tunnel with a mature fibrocartilage zone. Kanazawa et al. [24] applied autologous BMSCs, with the carrier transplanted between the autograft tendon and tibia bone pit, in an ACLR rabbit model and observed that treatment with BMSC resulted in progressive maturation of active chondroid cells layered in the interface from four to eight weeks. Nevertheless, the exact mechanism of interfacial fibrocartilage formation after BM cell therapy is unclear. In the present study, we first demonstrated that cells could migrate and infiltrate into the interface between the grafted tendon and bone tunnel after intra-articular injection. Histological analysis revealed that the injection of BM cells resulted in the formation of a broader fibrocartilage transitional zone anchored to tendons, resembling direct healing; this was obviously different from the healing pattern in the control group, which exhibited more organized fiber infiltration into the graft. In this study, higher MCP1 expression was observed at two weeks post operation, indicating early inflammation after additional BM cell delivery, which might explain the early gap and later fibrocartilage formation in the tendon-bone tunnel after allograft ACLR. This study demonstrated that intra-articular injection of both freshly isolated BMMNCs and cultured BMSCs had similar capabilities for forming interfacial fibrocartilage after ACLR [24,29].
BM-derived cells showed a biphasic role in both inflammation and tissue repair: they triggered tissue repair (non-haemopoietic cells) and promoted differentiation into inflammatory cells such as neutrophils, eosinophils, basophils, mast cells, and monocytes. After tendon repair and reconstruction, moderate inflammation is critical for tendon/ligament healing, especially for macrophage polarization to help tissue remodeling, scar resolution, and immunosuppression [30]. In contrast, prolonged inflammation induces excessive remodeling and scar healing. In a comparison study, Lu et al. [31] injected autologous BM and BMSCs directly into the extra-articular tendon-bone tunnel in a rat model and found that increased M2 macrophage polarization in the autologous BM group resulted in more interfacial organization of fibers with increasing stiffness and failure load compared to the BMSCs group. In our study, BM cells caused higher MCP1 expression at two weeks post operation, indicating that both BMMNCs and BMSCs induced more inflammation factors in the early stage, which might reduce surrounding cell migration and proliferation in the graft with interfacial broader fibrocartilage formation after allograft ACLR.
Growth factors and cytokines are important in tissue regeneration after ACL injury. Platelet-rich plasma (PRP) was shown to improve the quality of graft tissue, resulting in enhanced biomechanical properties and increased collagen expression in ACLR animal models [32,33]. Furthermore, PRP promotes tissue regeneration owing to its anti-inflammatory effects through different signaling pathways, release of multiple anti-inflammatory biomolecules, and modulation of macrophage subtypes [34,35]. Extracellular vesicles (EVs), paracrine factors from stem cells, have been applied as novel biomaterials in tissue regeneration. Shi et al. [36] demonstrated that EVs regulate BMSCs toward tenogenic differentiation, attenuate inflammation by increasing the expression of anti-inflammatory mediators, and enhance tendon healing in a patellar tendon injury model. Furthermore, EV delivery could increase M2 macrophage polarization and decrease M1 macrophage population as well as proinflammatory factors in the early phase of mouse Achilles tendon-bone reconstruction, improving tendon-bone healing [37]. The upregulated expression of MCP1 in our study indicated higher inflammation within the tendon-bone interface in the early stage after BM cell injection. Further studies are required to elucidate whether the combination of intra-articular BM cells and PRP or EVs could regulate early inflammation and enhance graft maturation and tendon-bone healing in allograft ACLR.
The present study has several limitations to it, including the lack of mechanical tests to investigate the ACL allograft-bone tunnel interfacial strength. In this study, we found progressive fibrocartilage maturation in the tendon-bone interface in both BMMNCs and BMSCs groups, in contrast to fibrous healing in the control group. Previous studies on the use of BM cells in the tendon-bone interface revealed progressive fibrocartilage maturation, leading to increased failure load in biomechanical tests [29,38]. In this study, we delivered only BM-derived cells through intra-articular injection. Different cell sources and delivery methods may help clarify the best approach to augment graft cell repopulation and interfacial healing after allograft ACLR. Small sample size and a short follow up period were some other limitations to our study. Therefore, further studies with larger sample sizes and longer follow up time are needed.
In conclusion, the regeneration capacity of freshly isolated BMMNCs is comparable to that of cultured BMSCs in rabbit allograft ACLR model. However, intra-articular BM cell injection had limited benefits on graft cell repopulation, led to higher inflammation in the early stage, with later progressive interfacial fibrocartilage maturation.
4. Materials and Methods
4.1. Animal Model
In this study, 14 skeletally mature New Zealand male rabbits weighing 2.5–3.0 kg were used. Allograft ACLR were performed in both knees of these rabbits. All rabbits with two operated knees were randomly divided into the experimental groups (fresh isolated BMMNCs or cultured BMSCs injection after ACLR; n = 5 knees per group, per time-point) and control group (without cell seeding after ACLR; n = 4 knees per time-point). Rabbits were euthanized at two different time-points: two and six weeks post surgery. All animal experiments were approved on 13 June 2017 by the Institutional Animal Care and Use Committee (IACUC 105263).
4.2. Harvesting Bone Marrow Stromal Cells
Two weeks before the ACLR procedure, BMSCs were harvested from rabbit iliac crests. Bone marrow (8 mL) was aspirated from the iliac crest with a 20-mL syringe containing 2 mL heparin and gently mixed. The heparin and bone marrow solution was transferred into a 50-mL centrifuge tube and washed with 10 mL phosphate buffered saline (PBS). After centrifugation at 1500 rpm for 5 min at room temperature, 15 mL supernatant was removed and the cell pellet was resuspended in 25 mL cell growth medium containing minimal essential medium with Earle’s salts (GIBCO, Grand Island, NY, USA), 10% foetal calf serum (FCS), and 1% antibiotics (penicillin/streptomycin, GIBCO). Three equal aliquots of the resuspended bone marrow cells were seeded in 100-mm cell culture dishes and incubated at 37 °C in a humidified incubator with 5% CO2. BMSCs were grown as adherent cells and the culture medium was changed every three days. At 80–90% confluence, the cells were detached with 0.25% trypsin and sub-cultured. BMSCs at passage four were used for experiments in this study.
4.3. Preparation of Decellularized Tendon Graft
Hamstring tendons were harvested from rabbits euthanized in other IACUC-approved studies. The hamstring tendons were decellularized by freeze-thawing. Tendons were immersed in liquid nitrogen for 1 min and then thawed for 5 min in saline solution at 37 °C; this process was repeated five times. Next, the tendons were incubated in nuclease solution (RNase 100 μg/mL and DNase 150 IU/mL; Roche Diagnostic, Indianapolis, IN, USA) for 12 h at 37 °C, as described previously, to obtain an average DNA concentration of 11.5 ± 10.1 ng/mg decellularized tendon [39,40]. Finally, the tendons were rinsed three times with PBS for 30 min each at room temperature, with gentle agitation. The decellularized tendons were lyophilized for 24 h and gas sterilized using ethylene oxide. The lyophilized decellularized tendon grafts were stored at ambient room temperature. Prior to use in rabbit ACL surgery, the lyophilized decellularized tendon grafts were immersed in PBS for 24 h.
4.4. Harvesting Fresh Bone Marrow Cells
After anaesthetizing the rabbits, bone marrow (8 mL) was aspirated from the iliac with a 20-mL syringe containing 2 mL heparin. BMMNCs were isolated from the aspirated bone marrow by density gradient centrifugation. The bone marrow was diluted 1:1 in PBS and carefully overlaid on 15 mL Ficoll-Paque Plus (Amersham Biosciences, Uppsala, Sweden) in a 50-mL tube. The tubes were centrifuged at 1500 rpm for 30 min at 20 °C. After centrifugation, the top plasma layer was removed first and then the buffy coat was carefully transferred to a fresh 15-mL tube. The buffy coat layer containing BMMNCs was washed with PBS and centrifuged at 1000 rpm for 10 min at 20 °C; this was repeated twice. The suspension was discarded, the pellet was resuspended in culture medium, and cell number was counted.
4.5. PKH26 Labelled Cells
In order to monitor the seeded cells, freshly isolated BMMNCs and cultured BMSCs were labelled with PKH26 Red Fluorescent Cell Linker Kit (Sigma-Aldrich, St. Louis, MO, USA); 5 µL of the dye solution was added per mL of cell suspension (1 × 106 cells/mL). After incubation at 37 °C for 20 min, the labelled cells were centrifuged at 1500 rpm for 5 min and re-suspended in culture medium. The labeled cells were prepared at a concentration of 1 × 107 cells/100 µL PBS before injection.
4.6. ACL Allograft Reconstruction
Rabbits were anesthetized with intravenously administered sodium pentobarbital (30 mg/kg). The knee joint was approached through a lateral parapatellar incision using aseptic techniques and the ACL was reconstructed with decellularized hamstring allograft. The allograft was trimmed to 2.0 mm diameter and the two free tendon ends were prepared with Bunnell suture using a 4-0 nylon suture. After retracting the anterior tibialis muscle laterally, a 2.0-mm bone tunnel was created at a 45° angle to the tibial axis and at 30° angle to the sagittal plane through the proximal tibial metaphysis to the footprint of the ACL stump with a 2.0-mm drill bit. The femoral tunnel was created through the femoral footprint with a 2.0-mm drill bit at 70° angle knee flexion. The prepared allograft was passed through the bone tunnel and fixed by suturing to the periosteum and surrounding soft tissue (Figure 7). The joint capsule, fascia, subcutaneous tissue, and skin were closed with 4-0 nylon suture. In the experimental groups, wither PKH26-labelled freshly isolated BMMNCs (1 × 107 cells in 100 µL PBS) or cultured BMSCs (1 × 107 cells in 100 µL PBS) were injected into the knee joints (n = 5 per time-point: two and six weeks after surgery in both BMMNC and BMSC groups). In the control group, the ACL was reconstructed with decellularized hamstring allograft without cell injection (n = 4 per time-point: two and six weeks post surgery). After surgery, rabbits were kept in individual cages without activity restriction. After two and six weeks, rabbits were euthanized via carbon dioxide inhalation. Knee joints were obtained and prepared for intra-articular graft and proximal tibia analyses. Intra-articular analysis investigated cellular repopulation in the allograft, while proximal tibia analysis evaluated tendon-bone tunnel interfacial healing.
Figure 7.
(A) The rabbit knee joint was exposed and the anterior cruciate ligament (ACL) was identified. (B) The allograft was passed through the tibial and femoral bone tunnel.
4.7. Analysis of Cell Repopulation in the Intra-Articular Graft
4.7.1. Histological Study
Intra-articular graft samples were prepared for histological analysis at two and six weeks post surgery. After fixation, the samples were embedded in paraffin and cut parallelly to the longitudinal axis of the graft. These samples were stained with haematoxylin and eosin (H&E) to examine collagen fiber and cell distribution in the graft.
4.7.2. Total Cell Count
The slices were counterstained with DAPI (Vector Laboratories Inc, Burlingame, CA, USA) for nuclear staining and observed with confocal microscopy (Z2; Carl Zeiss, Oberkochen, Germany). Total cells (DAPI stained in blue) and seeded cells (PKH26 stained in red) in the allografts of the control, BMMNC, and BMSC groups were counted in three randomly selected fields (300 × 300 μm) of three samples per study group using ImageJ (64-bit Java v. 1.6.0_24; NIH, Bethesda, MD, USA) software and expressed as cell number per mm2.
4.8. Analysis of Tendon-Bone Tunnel Interfacial Healing in the Proximal Tibia
4.8.1. Histological Study
Proximal tibia samples were prepared at two and six weeks post surgery. After fixation and decalcification, the samples were embedded in paraffin and cut cross-sectionally and parallelly to the tibial plateau surface for observing the tendon-bone tunnel interface. The samples were stained with H&E and Masson’s trichrome to examine the healing pattern within the interface of the tendon-bone tunnel. Samples from the BMMNC and BMSC groups (stained with PKH26) were collected at two and six weeks after injection with the respective bone marrow cells and observed under microscope (Leica CTR6000) to confirm cell infiltration in the tendon-bone interface.
4.8.2. Micro–Computed Tomography (Micro-CT)
Micro-CT measurements (Skyscan 1272; Bruker. Billerica, MA, USA) quantified the mineralized tissue ingrowth around the bone tunnel and the status of tunnel widening. Each specimen (n = 4 in the control group, n = 5 in BM cells-injected groups at both two and six weeks) was scanned perpendicular to the long bone axis covering the entrance and exit of the tibial tunnel. We evaluated the cross section hole area, bone marrow density (BMD), percentage bone volume (Bone Volume (BV)/Total Volume (TV)), and bone surface density (BS/TV) with a CT Analyzer (version 1.16.1.0+ (64-bit)) software.
4.8.3. Immunohistochemical Analysis
To observe collagen formation and inflammatory cell infiltration, the proximal tibia samples were cut into 5-µm thick sections; IHC was performed with collagen I (1:200; Arigo Biolaboratories, Hsinchu, Taiwan) and monocyte chemoattractant protein-1 (MCP1) (1:200; Proteintech, Rosemont, IL, USA) antibodies. After antigen retrieval, the slides were washed twice with Tris-buffered saline (TBS) (Sigma-Aldrich, St. Louis, MO, USA) containing 0.025% Triton X-100 (TBST) for 5 min. The slides were blocked with IHC detecting kit (ab64264; Abcam, Cambridge, UK) for 1 h at room temperature according to manufacturer’s instructions. The primary antibodies were diluted in TBS with 1% BSA, incubated overnight at 4 °C, and then rinsed with TBST five times. The enzyme-conjugated secondary antibody was diluted in TBS with 1% BSA and incubated on the slides for 1 h at room temperature. The slides were counterstained with DAPI and observed under a light microscope (CTR6000; Leica Microsystems CMS GmbH, Wetzlar, Germany). The IHC results of collagen I and MCP1 were collected from three randomized areas per sample (n = 3 in each group at both two and six weeks) and analyzed with Image-Pro Plus 6.0 software (Media Cybernetics. Rockville, MD, USA).
4.9. Statistical Analysis
The results of cell number, Micro-CT, and IHC are shown as mean ± standard error of the mean (SE). Statistical analyses of the control, BMMNC, and BMSC groups were performed using one-way analysis of variance (ANOVA) with Tukey’s post hoc analysis and p < 0.05 was considered statistically significant.
Acknowledgments
We appreciate the support from members of orthopedic department of Kaohsiung Municipal Siaogang Hospital and Regeneration Medicine and Cell Therapy Research Center, Graduate Institute of Medicine, College of Medicine of Kaohsiung Medical University, Kaohsiung City, Taiwan.
Author Contributions
Conceptualization, S.-H.C.; data curation, C.-C.L., C.-J.H., and S.-Y.L.; formal analysis, C.-C.L., S.-Y.L., S.-H.C., and P.-H.C.; funding acquisition, C.-C.L. and Y.-C.T.; investigation, H.-T.H.; methodology, C.-C.L., C.-J.H., P.-H.C., and M.-L.H.; resources, S.-Y.L., H.-T.H., and Y.-C.T.; software, M.-L.H.; supervision, H.-T.H. and Y.-C.T.; validation, C.-J.H., S.-H.C., and M.-L.H.; visualization, S.-H.C.; writing—original draft, C.-C.L.; writing—review and editing, P.-H.C., M.-L.H., and Y.-C.T. All authors have read and agreed to the published version of the manuscript.
Funding
This study was supported by grants from Kaohsiung Municipal Siaogang Hospital (H-108-004), the Ministry of Science and Technology, Taiwan (MOST 109-2314-B-037-016-MY2), and Regenerative Medicine and Cell Therapy Research Center, Kaohsiung Medial University, Taiwan (KMU-TC108A02-6 and -7).
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Animal Care and Use Committee of Kaohsiung Medical University on 13 June 2017 (IACUC 105263).
Informed Consent Statement
Not applicable.
Data Availability Statement
MDPI Research Data Policies.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Janssen R.P., Scheffler S.U. Intra-articular remodeling of hamstring tendon grafts after anterior cruciate ligament reconstruction. Knee Surg. Sports Traumatol. Arthrosc. 2014;22:2102–2108. doi: 10.1007/s00167-013-2634-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sato Y., Akagi R., Akatsu Y., Matsuura Y., Takahashi S., Yamaguchi S. The effect of femoral bone tunnel configuration on tendon-bone healing in an anterior cruciate ligament reconstruction: An animal study. Bone Joint Res. 2018;7:327–335. doi: 10.1302/2046-3758.75.BJR-2017-0238.R2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tohyama H., Yoshikawa T., Ju Y.J., Yasuda K. Revascularization in the tendon graft following anterior cruciate ligament reconstruction of the knee: Its mechanisms and regulation. Chang. Gung. Med. J. 2009;32:133–139. doi: 10.1016/S0021-9290(06)83128-3. [DOI] [PubMed] [Google Scholar]
- 4.Hu J., Qu J., Xu D., Zhou J., Lu H. Allograft versus autograft for anterior cruciate ligament reconstruction: An up-to-date meta-analysis of prospective studies. Int. Orthop. 2013;37:311–320. doi: 10.1007/s00264-012-1720-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lamblin C.J., Waterman B.R., Lubowitz J.H. Anterior cruciate ligament reconstruction with autografts compared with non-irradiated, non-chemically treated allografts. Arthroscopy. 2013;29:1113–1122. doi: 10.1016/j.arthro.2013.01.022. [DOI] [PubMed] [Google Scholar]
- 6.Tulloch S.J., Devitt B.M., Porter T., Hartwig T., Klemm H., Hookway S., Norsworthy C.J. Primary ACL reconstruction using the LARS device is associated with a high failure rate at minimum of 6-year follow-up. Knee Surg. Sports Traumatol. Arthrosc. 2019;27:3626–3632. doi: 10.1007/s00167-019-05478-3. [DOI] [PubMed] [Google Scholar]
- 7.Tiefenboeck T.M., Thurmaier E., Tiefenboeck M.M., Ostermann R.C., Joestl J., Winnisch M. Clinical and functional outcome after anterior cruciate ligament reconstruction using the LARS™ system at a minimum follow-up of 10 years. Knee. 2015;22:565–568. doi: 10.1016/j.knee.2015.06.003. [DOI] [PubMed] [Google Scholar]
- 8.Granan L.P., Inacio M.C., Maletis G.B., Funahashi T.T., Engebretsen L. Intraoperative findings and procedures in culturally and geographically different patient and surgeon populations: An anterior cruciate ligament reconstruction registry comparison between Norway and the USA. Acta Orthop. 2012;83:577–582. doi: 10.3109/17453674.2012.741451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Scheffler S.U., Schmidt T., Gangey I., Dustmann M., Unterhauser F., Weiler A. Fresh-frozen free-tendon allografts versus autografts in anterior cruciate ligament reconstruction: Delayed remodeling and inferior mechanical function during long-term healing in sheep. Arthroscopy. 2008;24:448–458. doi: 10.1016/j.arthro.2007.10.011. [DOI] [PubMed] [Google Scholar]
- 10.Li H., Tao H., Cho S., Chen S., Yao Z., Chen S. Difference in graft maturity of the reconstructed anterior cruciate ligament 2 years postoperatively: A comparison between autografts and allografts in young men using clinical and 3.0-T magnetic resonance imaging evaluation. Am. J. Sports Med. 2012;40:1519–1526. doi: 10.1177/0363546512443050. [DOI] [PubMed] [Google Scholar]
- 11.Ning L.J., Zhang Y.J., Zhang Y., Qing Q., Jiang Y.L., Yang J.L., Luo J.-C., Qin T.-W. The utilization of decellularized tendon slices to provide an inductive microenvironment for the proliferation and tenogenic differentiation of stem cells. Biomaterials. 2015;52:539–550. doi: 10.1016/j.biomaterials.2015.02.061. [DOI] [PubMed] [Google Scholar]
- 12.Lu C.C., Zhang T., Amadio P.C., An K.N., Moran S.L., Gingery A. Lateral slit delivery of bone marrow stromal cells enhances regeneration in the decellularized allograft flexor tendon. J. Orthop. Translat. 2019;19:58–67. doi: 10.1016/j.jot.2019.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang Y., Han Z.B., Song Y.P., Han Z.C. Safety of mesenchymal stem cells for clinical application. Stem Cells Int. 2012;16:652034. doi: 10.1155/2012/652034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Blatt A., Minha S., Moravsky G., Vered Z., Krakover R. Intracoronary administration of autologous bone marrow mononuclear cells in patients with chronic ischemic symptomatic cardiomyopathy: 5 years follow-up. Isr. Med. Assoc. J. 2010;12:738–741. [PubMed] [Google Scholar]
- 15.Hisatome T., Yasunaga Y., Yanada S., Tabata Y., Ikada Y., Ochi M. Neovascularization and bone regeneration by implantation of autologous bone marrow mononuclear cells. Biomaterials. 2005;26:4550–4556. doi: 10.1016/j.biomaterials.2004.11.032. [DOI] [PubMed] [Google Scholar]
- 16.Seebach C., Henrich D., Schaible A., Relja B., Jugold M., Bonig H., Marzi I. Cell-based therapy by implanted human bone marrow-derived mononuclear cells improved bone healing of large bone defects in rats. Tissue Eng. Part A. 2015;21:1565–1578. doi: 10.1089/ten.tea.2014.0410. [DOI] [PubMed] [Google Scholar]
- 17.Henrich D., Verboket R., Schaible A., Kontradowitz K., Oppermann E., Brune J.C. Characterization of bone marrow mononuclear cells on biomaterials for bone tissue engineering in vitro. Biomed. Res. Int. 2015;23:762407. doi: 10.1155/2015/762407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hibino N., Nalbandian A., Devine L., Martinez R.S., McGillicuddy E., Yi T., Karandish S. Comparison of human bone marrow mononuclear cell isolation methods for creating tissue-engineered vascular grafts: Novel filter system versus traditional density centrifugation method. Tissue Eng. Part C Methods. 2011;17:993–998. doi: 10.1089/ten.tec.2011.0110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kamihata H., Matsubara H., Nishiue T., Fujiyama S., Tsutsumi Y., Ozono R., Masaki H., Mori Y. Implantation of bone marrow mononuclear cells into ischemic myocardium enhances collateral perfusion and regional function via side supply of angioblasts, angiogenic ligands, and cytokines. Circulation. 2001;104:1046–1052. doi: 10.1161/hc3501.093817. [DOI] [PubMed] [Google Scholar]
- 20.Deshmane S.L., Kremlev S., Amini S., Sawaya B.E. Monocyte chemoattractant protein-1 (MCP-1): An overview. J. Interferon. Cytokine Res. 2009;29:313–326. doi: 10.1089/jir.2008.0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Xu L.L., Warren M.K., Rose W.L., Gong W., Wang J.M. Human recombinant monocyte chemotactic protein and other C-C chemokines bind and induce directional migration of dendritic cells in vitro. J. Leukoc. Biol. 1996;60:365–371. doi: 10.1002/jlb.60.3.365. [DOI] [PubMed] [Google Scholar]
- 22.Cvetanovich G.L., Mascarenhas R., Saccomanno M.F., Verma N.N., Cole B.J., Bush-Joseph C.A., Bach B.R. Hamstring autograft versus soft-tissue allograft in anterior cruciate ligament reconstruction: A systematic review and meta-analysis of randomized controlled trials. Arthroscopy. 2014;30:1616–1624. doi: 10.1016/j.arthro.2014.05.040. [DOI] [PubMed] [Google Scholar]
- 23.Bottoni C.R., Smith E.L., Shaha J., Shaha S.S., Raybin S.G., Tokish J.M., Rowles D.J. Autograft Versus Allograft Anterior Cruciate Ligament Reconstruction: A Prospective, Randomized Clinical Study with a Minimum 10-Year Follow-up. Am. J. Sports Med. 2015;43:2501–2509. doi: 10.1177/0363546515596406. [DOI] [PubMed] [Google Scholar]
- 24.Kanazawa T., Soejima T., Noguchi K., Tabuchi K., Noyama M., Nakamura K., Tabuchi K. Tendon-to-bone healing using autologous bone marrow-derived mesenchymal stem cells in ACL reconstruction without a tibial bone tunnel-A histological study. Muscles Ligaments Tendons J. 2014;4:201–206. doi: 10.32098/mltj.02.2014.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Proffen B.L., Vavken P., Haslauer C.M., Fleming B.C., Harris C.E., Machan J.T., Murray M.M. Addition of autologous mesenchymal stem cells to whole blood for bioenhanced ACL repair has no benefit in the porcine model. Am. J. Sports Med. 2015;43:320–330. doi: 10.1177/0363546514559826. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guo R., Gao L., Xu B. Current Evidence of Adult Stem Cells to Enhance Anterior Cruciate Ligament Treatment: A Systematic Review of Animal Trials. Arthroscopy. 2018;34:331–340.e2. doi: 10.1016/j.arthro.2017.07.010. [DOI] [PubMed] [Google Scholar]
- 27.Dustmann M., Schmidt T., Gangey I., Unterhauser F.N., Weiler A., Scheffler S.U. The extracellular remodeling of free-soft-tissue autografts and allografts for reconstruction of the anterior cruciate ligament: A comparison study in a sheep model. Knee Surg. Sports Traumatol. Arthrosc. 2008;16:360–369. doi: 10.1007/s00167-007-0471-0. [DOI] [PubMed] [Google Scholar]
- 28.Park M.J., Lee M.C., Seong S.C. A comparative study of the healing of tendon autograft and tendon-bone autograft using patellar tendon in rabbits. Int. Orthop. 2001;25:35–39. doi: 10.1007/s002640000199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Soon M.Y., Hassan A., Hui J.H., Goh J.C., Lee E.H. An analysis of soft tissue allograft anterior cruciate ligament reconstruction in a rabbit model: A short-term study of the use of mesenchymal stem cells to enhance tendon osteointegration. Am. J. Sports Med. 2007;35:962–971. doi: 10.1177/0363546507300057. [DOI] [PubMed] [Google Scholar]
- 30.Thomopoulos S., Parks W.C., Rifkin D.B., Derwin K.A. Mechanisms of tendon injury and repair. J. Orthop. Res. 2015;33:832–839. doi: 10.1002/jor.22806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lu J., Chamberlain C.S., Ji M.L., Saether E.E., Leiferman E.M., Li W.J., Vanderby R. Tendon-to-Bone Healing in a Rat Extra-articular Bone Tunnel Model: A Comparison of Fresh Autologous Bone Marrow and Bone Marrow-Derived Mesenchymal Stem Cells. Am. J. Sports Med. 2019;47:2729–2736. doi: 10.1177/0363546519862284. [DOI] [PubMed] [Google Scholar]
- 32.Fallouh L., Nakagawa K., Sasho T., Arai M., Kitahara S., Wada Y., Moriya H., Takahashi K. Effects of autologous platelet-rich plasma on cell viability and collagen synthesis in injured human anterior cruciate ligament. J. Bone Joint Surg. Am. 2010;92:2909–2916. doi: 10.2106/JBJS.I.01158. [DOI] [PubMed] [Google Scholar]
- 33.Xie X., Zhao S., Wu H., Xie G., Huangfu X., He Y., Zhao Z. Platelet-rich plasma enhances autograft revascularization and reinnervation in a dog model of anterior cruciate ligament reconstruction. J. Surg. Res. 2013;183:214–222. doi: 10.1016/j.jss.2013.01.020. [DOI] [PubMed] [Google Scholar]
- 34.Khatab S., van Buul G.M., Kops N., Bastiaansen-Jenniskens Y.M., Bos P.K., Verhaar J.A., Van Osch G.J. Intra-articular Injections of Platelet-Rich Plasma Releasate Reduce Pain and Synovial Inflammation in a Mouse Model of Osteoarthritis. Am. J. Sports Med. 2018;46:977–986. doi: 10.1177/0363546517750635. [DOI] [PubMed] [Google Scholar]
- 35.Mariani E., Roffi A., Cattini L., Pulsatelli L., Assirelli E., Krishnakumar G.S., Kon E. Release kinetic of pro- and anti-inflammatory biomolecules from platelet-rich plasma and functional study on osteoarthritis synovial fibroblasts. Cytotherapy. 2020;22:344–353. doi: 10.1016/j.jcyt.2020.02.006. [DOI] [PubMed] [Google Scholar]
- 36.Shi Z., Wang Q., Jiang D. Extracellular vesicles from bone marrow-derived multipotent mesenchymal stromal cells regulate inflammation and enhance tendon healing. J. Transl. Med. 2019;17:211. doi: 10.1186/s12967-019-1960-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Shi Y., Kang X., Wang Y., Bian X., He G., Zhou M., Tang K. Exosomes Derived from Bone Marrow Stromal Cells (BMSCs) Enhance Tendon-Bone Healing by Regulating Macrophage Polarization. Med. Sci. Monit. 2020;26:e923328. doi: 10.12659/MSM.923328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen B., Li B., Qi Y.J., Ni Q.B., Pan Z.Q., Wang H., Chen L.-B. Enhancement of tendon-to-bone healing after anterior cruciate ligament reconstruction using bone marrow-derived mesenchymal stem cells genetically modified with bFGF/BMP2. Sci. Rep. 2016;6:25940. doi: 10.1038/srep25940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ning L.J., Zhang Y., Chen X.H., Luo J.C., Li X.Q., Yang Z.M., Qin T.-W. Preparation and characterization of decellularized tendon slices for tendon tissue engineering. J. Biomed. Mater. Res. A. 2012;100:1448–1456. doi: 10.1002/jbm.a.34083. [DOI] [PubMed] [Google Scholar]
- 40.Ozasa Y., Amadio P.C., Thoreson A.R., An K.N., Zhao C. Repopulation of intrasynovial flexor tendon allograft with bone marrow stromal cells: An ex vivo model. Tissue Eng. Part A. 2014;20:566–574. doi: 10.1089/ten.tea.2013.0284. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
MDPI Research Data Policies.