Abstract
Ovarian cancer is the eighth-most common cause of death among women worldwide. In the absence of distinctive symptoms in the early stages, the majority of women are diagnosed in advanced stages of the disease. Surgical debulking and systemic adjuvant chemotherapy remain the mainstays of treatment, with the development of chemoresistance in up to 75% of patients with subsequent poor treatment response and reduced survival. Therefore, there is a critical need to revisit existing, and identify potential biomarkers that could lead to the development of novel and more effective predictors for ovarian cancer diagnosis and prognosis. The capacity of these biomarkers to predict the existence, stages, and associated therapeutic efficacy of ovarian cancer would enable improvements in the early diagnosis and survival of ovarian cancer patients. This review not only highlights current evidence-based ovarian-cancer-specific prognostic and diagnostic biomarkers but also provides an update on various technologies and methods currently used to identify novel biomarkers of ovarian cancer.
Keywords: ovarian cancer, biomarkers, tumour mutation burden, DNA repair pathways, cell-cycle-related genes
1. Introduction
Ovarian cancer is the eighth-most commonly occurring cancer in women worldwide, contributing to 300,000 new cases globally in 2018. Ovarian cancer is often asymptomatic in the early stages; therefore, it is considered a silent killer. The signs and symptoms become more apparent as the cancer advances. For this reason, ovarian cancers have the highest mortality rate among the gynaecological diseases. Therapeutic intervention for a cure is possible in the early stages of the disease, and this explains the direct relation between early diagnosis and survival [1,2]. In contrast to a five-year survival rate of 95% at an earlier stage, the high case fatality ratio illustrates the reality that most cases of ovarian cancer are diagnosed in an advanced stage of the disease (stage III or IV), with a five-year survival rate of less than 30% [3]. To date, no screening approach has been recommended for early stage identification. It will also be important to establish tumour markers that could be used to diagnose cancer in the early stages in order to improve the survival of these women.
Epithelial ovarian cancer (EOC) is the commonest type, highly heterogeneous, and often linked to genetic instability [4]. Serous-type ovarian cancer comprises up to 70% of EOCs. The World Health Organization (WHO) classification system, published in 2014, reclassified serous ovarian cancer into two separate entities: high-grade and low-grade [5]. Low-grade serous ovarian cancer (LGSOC) represents 5–10% of serous ovarian cancers. LGSOC is well-differentiated and can be identified in the early stages with the overexpression of serous borderline precursor lesions, such as BRAF and KRAS mutation, and the near absence of the tumour protein p53 mutation (TP53) [6]. High-grade serous ovarian cancer (HGSOC), meanwhile, is the most common and aggressive form of EOC subtype and accounts for the majority of ovarian-cancer-related deaths [7]. Unlike LGSOC, HGSOC has no known precursor lesion in the ovary, with many studies suggesting its development from intraepithelial carcinomas in the fallopian tube [8]. Approximately 95% and 50% of HGSOC cases have TP53 mutations and homologous recombination (HR) deficiency, respectively [5,9]. Clinically, women with HGSOC tend to be older and have poor survival, while those with LGSOC are younger and tend to survive longer [10]. Although HGSOC patients have initial sensitivity to first-line chemotherapy, almost all patients develop chemoresistance. LGSOC is a slow-growing tumour and generally resistant to chemotherapy but responds well to aggressive debulking surgery. LGSOC has shown improved survival rates when treated with alternative treatments such as angiogenesis inhibitor and MEK inhibitor therapy [11]. The heterogeneity of ovarian cancer is a major obstacle in discovering novel biomarkers to aid early detection. As new therapies such as Poly ADP Ribose Polymerase (PARP) inhibitors have improved survival in women with HGSOC, specifically in those with the BRCA mutation, the discovery of molecular biomarkers is becoming important. As ovarian cancer targets various genetic and putative molecular cell signalling pathways, understanding the related proteins and genes will provide an opportunity to unravel new biomarkers. In this review, we summarise recently discovered potential biomarkers to improve the diagnosis of, and prognosis for ovarian cancer, and revisit the clinically available biomarkers and their precision of use in the management of EOC. In addition, this article is designed to play a role as a platform to search for the most effective laboratory technology and methods available to discover novel ovarian biomarkers.
Search Strategy of Review
The keywords and terms of the major concepts for this review included ovarian cancer, ovarian signalling pathway, ovarian cancer diagnosis and prognosis, and current and/or new ovarian cancer biomarkers, which were developed and combined to form the search strategy. In Section 2.1, Ovarian Cancer Cell Signalling Pathways and Their Clinical Utility, we included more specific keywords of ovarian cancer molecular pathways (e.g., tumour mutation burden, DNA repair pathways, and cell-cycle-related genes). A systematic search of Google Scholar, PubMed, Web of Science, and Sci-Hub databases was combined for relevant publications, from the time of their inception to December 2020. Results were merged using reference management software (Endnote, Thomson Reuters). The findings of relevant studies are summarised in Table 1 and Table 2.
Table 1.
Current ovarian cancer biomarkers and their clinical utility.
| Clinical Biomarker | Source | Clinical Role | Clinical Utility | Reference | ||
|---|---|---|---|---|---|---|
| Diagnosis | Prognosis | Cancer Therapy | ||||
| Carbohydrate Antigen 125 (CA125) | Serum | ✓ | ✓ | Screen for ovarian cancer; evaluate the chemotherapy response and monitor disease recurrence using ELISA. | [12,13,14,15] | |
| Osteopontin (OPN) | Plasma | ✓ | ✓ | Diagnosis of ovarian cancer and prognostic indicator of metastasis using PCR and ELISA. | [16,17] | |
| Kallikreins (KLKs) | Serum | ✓ | Diagnosis of ovarian cancer using ELISA. | [17,18,19,20,21] | ||
| Bikunin | Serum | ✓ | Predict the stage of the disease and the survival rate preoperatively using ELISA or immunoblot assay. | [22,23,24,25] | ||
| Human Epididymis Protein 4 (HE4) | Serum | ✓ | High-sensitivity diagnostic tool for detecting stage I ovarian cancer using ELISA. | [26,27,28] | ||
| Vascular Endothelial Growth Factor (VEGF) | Serum | ✓ | ✓ | ✓ | Indicator of a shorter survival time in patients with ovarian cancers using ELISA. | [29,30,31,32] |
| Prostasin (PSN) | Serum | ✓ | Identify patients with ovarian cancer through RT-PCR. | [33,34] | ||
| Creatine Kinase B (CKB) | Serum | ✓ | Predict survival rate and prognosis of ovarian cancer using microarray technology. | [35] | ||
| Mesothelin | Serum | ✓ | Detected using ELISA method and used clinically as an antimesothelin immunotoxin therapy to attack cancer cells in combination with standard chemotherapy. | [36,37,38] | ||
| Apolipoprotein A-I (apoA-I) | Plasma | ✓ | Detected clinically using ELISA method to confirm diagnosis with ovarian cancer. | [39,40,41,42] | ||
| Transthyretin (TTR) | Serum | ✓ | Diagnosis of early stage ovarian cancer patients using matrix-assisted laser desorption-ionisation (MALDI). | [43] | ||
| Transferrin | Serum | ✓ | Used in combination with CA125 as an improved and sensitive ovarian cancer detection method estimated by measurements of the serum total iron-binding capacity (TIBC). | [44,45] | ||
Table 2.
Potential newly emerging biomarkers and their clinical utility in ovarian cancer care.
| Emerging Biomarkers | Source | Potential Clinical Role | Reference |
|---|---|---|---|
| Cu isotope | Serum | Early diagnostic tool | [46] |
| Exosomes | Ascites | Cancer progression | [47] |
| lncRNA and mRNA | Tumour tissue | Early diagnostic tool and cancer therapy | [48] |
| Aldehyde Dehydrogenase 1 (ALDH1) | Blood/Cytosol | Early diagnostic and cancer progression | [49] |
| Folate Receptor Alpha (FOLR1) | Blood/Gene | Progression and cancer therapy | [50] |
| Glutathione S-Transferase Polymorphisms (GSTP) | Blood/DNA | Anticancer drug response | [51] |
2. An Overview of Ovarian Cancer Biomarkers and Cell Signalling Pathways
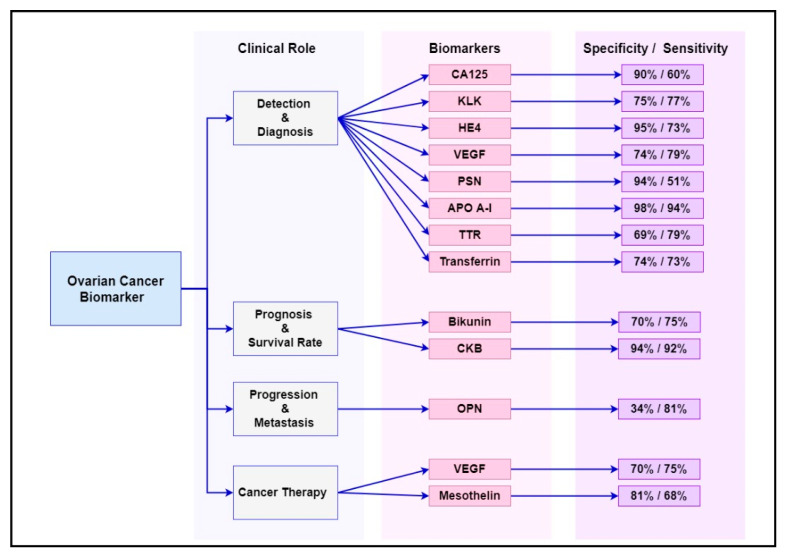
Numerous cancer genes and protein have been studied as diagnostic, prognostic and targeted therapy biomarkers in ovarian cancer management. Some of the most common biomarkers and their clinical utility are summarised in Table 1, while their sensitivity and specificity at early stages (stage I–II) of the disease are summarised in Figure 1. These biomarkers are already known to be overexpressed or deregulated in various signalling pathways in women with ovarian cancer [52]. Evidently, ovarian cancer is not a common disorder but includes a heterogeneous community of tumours that affect various molecular genetic mechanisms and putative molecular signalling pathways [53]. The main signalling pathways targeted for ovarian cancer diagnosis, progression and treatment are outlined in the following sections.
Figure 1.
Summary of the sensitivity and specificity of the most common ovarian cancer biomarkers at early stages (stage I–II) of the disease.
2.1. Ovarian Cancer Cell Signalling Pathways and Their Clinical Utility
2.1.1. BRCA1 and BRCA2 Pathway
In over 15% of women with HGSOC are associated with breast cancer 1 (BRCA1) and BRCA2 oncogenes, inherited as germline mutations. The BRCA1 and BRCA2 genes are located on chromosomes 17q21 and 13q12, respectively [54,55]. Wild-type BRCA1/2 genes are critical for DNA repair by the homologous recombination (HR) pathway; hence their deletion causes genomic instability and predisposes affected women to familial breast and ovarian cancers [56,57]. In addition, ovarian cancers with defective BRCA1/2 genes are especially susceptible to agents that induce DNA double-strand breaks (DSBs) [58] and DNA interstrand cross-links, such as platinum compounds (e.g., cisplatin and carboplatin) and polymerase (PARP) inhibitors (e.g., Olaparib, Iniparib and Veliparib) [39,59]. It is therefore conceivable that secondary or reversion mutations of the BRCA1/2 genes by multiple complex mechanisms can benefit the DNA repair of HR and increase the survival of the tumour cells, thus triggering resistance to these compounds [60]. As demonstrated in a SOLO-1 clinical trial, the use of PARP inhibitors as maintenance therapy represents a paradigm shift in ovarian cancer management, specifically in HGSOC women with BRCA mutations. There is a 70% lower risk of disease progression and a longer time before subsequent chemotherapy with PARP inhibitor maintenance therapy for 2 years [61].
2.1.2. MAPK/ERK Pathway
In ovarian cancer, the downstream proteins of the MAPK/ERK pathway play a pertinent role in promoting the migration and invasion of the disease and may therefore contribute to metastasis and chemoresistance. Genetic mutations such as BRAF and KRAS frequently contribute to the activation of the MAPK/ERK pathway, which is usually observed in LGSOC but rarely in HGSOC [62]. The mitogen-activated protein kinase (MAPK) pathway consists of three sequentially activated protein kinases responsible for the regulation of cell proliferation, cell differentiation, and cell death in humans. In response to external stimuli, including hormones such as follicle-stimulating hormone (FSH) and luteinising hormone (LH), growth factors [63], cytokines and chemotherapeutic chemicals, the MAPK pathway is activated by interactions with a small GTPase and/or phosphorylation by protein kinases downstream from cell surface receptors, such as G-protein coupled (Gonadotropins and Gonadotropin-releasing hormones) and tyrosine kinase (RTK) receptors [63]. MAPKs often play a role in promoting ovarian cancer cell growth through membrane receptor signals for gonadotropins-FSH and LH, frequently expressed in ovarian carcinoma cells, and may therefore lead to signal transduction through MAPK [64].
Clinical trials with targeted BRAF and MEK inhibitors, such as selumetinib, have been shown to achieve long-term progression free survival (PFS) and a complete response in advanced LGSOC [65]. In Gynecologic Oncology Group study 239, a phase II trial of selumetinib showed a 15% overall response rate and a median PFS of 11 months achieved by previously treated LGSOC patients (n = 52). This a modest improvement compared to PFS of seven months with traditional chemotherapy [66].
2.1.3. EGFR/AKT Signalling Pathway
The epidermal growth factor receptor (EGFR) is expressed in 70% of ovarian cancers [67]. Various ligands, such as EGF and TGF, may be activated and play a part in promoting and inhibiting tumour survival [68,69]. EGFR is also involved in tumour infiltration, metastasis and angiogenesis [70]. AKT is a major downstream signalling element for EGFR. Upon binding to EGFR, AKT is activated by phosphorylation. AKT is regularly overexpressed in ovarian cancer and is associated with aggressive tumour activity and poor prognosis. As the EGFR/AKT pathway is implicated in different facets of cancer proliferation, such as angiogenesis and metastases, it is currently seen as an attractive option for therapeutic intervention. Cetuximab (Erbitux) was the first anti-EGFR monoclonal antibody tested in several solid tumours, such as breast, colorectal, head and neck, renal, gastrointestinal stromal tumours (GISTs) and lung cancers. Although there is evidence of the therapeutic efficacy of anti-EFGR in these solid tumours, treatment with anti-EFGR agents has induced only low treatment response in ovarian cancers. In future, more comprehensive research needs to be done to unravel the protein and gene molecules along the complex EGFR signalling pathway in ovarian cancer to identify biomarkers that can accurately gauge the sensitivity of EGFR-targeted therapeutic agents.
2.1.4. Integrin Inhibitor Pathway
Integrins are heterodimeric adhesion receptors expressed on the cell surface, and consist of two noncovalently bound subunits, namely an α subunit and a β subunit [71]. The latest research has examined the use of integrin inhibitors as possible therapeutic agents in ovarian carcinoma. Several preclinical studies on various integrin antagonists have shown their effectiveness in blocking tumour progression. Integrin antagonists inhibit tumour progression by affecting both tumour cells and tumour-associated host cells, especially the angiogenic endothelium. The initial step in ovarian carcinoma dissemination occurs with the attachment of cancer cells onto the peritoneal surface via integrins, so targeting integrins seems a rational therapy approach [16].
Although no integrin inhibitors have yet shown desirable efficacy results, integrin-targeted therapies continue to be a promising pathway for further clinical investigation. In cancer progression, as more than one integrin pathway is usually involved, it was not surprising to see that agents targeting only a single integrin, such as αvβ3 and α5β1, failed to show clinical benefits in metastatic cancer treatment. Therefore, a combination of different integrin receptor pathways is likely to be show therapeutic efficacy in clinical trials and should be further explored [16].
2.1.5. GRP78 Expression Pathway
A recent investigation has suggested GRP78 as a drug delivery system for ovarian cancer cells. GRP78 upregulation is a cellular reaction process, caused by endoplasmic reticulum stress, and usually seen in tumour cells. Since GRP78 is abundantly present on ovarian cancer cell surfaces, recent research indicates the use of GRP78 as a delivery mechanism for cytotoxic substances [17].
2.1.6. P38 Alpha Pathway
The p38alpha pathway has recently been the focus of cancer research. Small compound inhibitors of p38alpha have already been evaluated in clinical trials, showing that the pharmacological blockade of p38 reduced the growth and viability of ovarian cancer cells [18]. The pharmacological blockade of p38 in ovarian cancer has been shown to induce the formation of large autophagic vacuoles containing cytoplasmic glycoproteic material and mitochondrial debris, suggesting that the inhibition of p38 caused autophagic cell death in ovarian cancer cells. This biomarker should be further investigated as a future therapeutic opportunity for ovarian cancer [72].
2.2. Current Biomarkers Associated with Diagnosis, Progression and Treatment Response of Ovarian Cancer
2.2.1. Carbohydrate Antigen 125 (CA125)
Cancer antigen 125 or carcinoma antigen 125, also known as MUC16, is a protein encoded by the MUC16 gene [73]. Clinically, it is used as a diagnostic test to measure the amount of the protein CA125 in the serum. In most laboratories, the normal value for CA125 is 0 to 35 units/mL.
Up to 80% of women diagnosed with late-stage epithelial ovarian cancer have elevated CA125 levels in their serum [22]. Unfortunately, CA125 has limited usefulness in detecting ovarian cancer in the early stages, as only 50% of these cases had elevated CA125 levels [23]. In addition, many other conditions can also cause the elevation of CA125 levels, including endometriosis, liver cirrhosis, normal menstruation, pelvic inflammatory disease and uterine fibroid. Therefore, this antigen lacks the specificity and sensitivity to be considered a reliable biomarker for the early detection of ovarian cancer [43]. CA125 is found to be more sensitive and specific in postmenopausal women than in premenopausal women. Serum CA125 is incorporated into the Risk Malignancy Index (RMI) algorithm. The RMI is widely used as a risk assessment of ovarian malignancy in clinical practice. The RMI score is generated by the simplified serum CA125 level regression equation, the menopausal status score and the ultrasound features score (RMI = ultrasound findings × menopause status × CA125 U/mL). The use of the RMI has a higher sensitivity of 87% and a specificity of 97% for ovarian cancer detection compared to CA125 alone [74].
In a recent study using a training and confirmation cohort, four existing clinical tests available for the diagnosis of ovarian cancer (RMI score and ROMA, CA125 and HE4) and a panel of 28 immunosoluble biomarkers from 66 patients undergoing surgery for suspected ovarian cancer were assessed through a multiplex immunoassay. Using a two-step triage model for women with presumed ovarian mass, IL-6 > 3.75 pg/mL was established as the main triage, supplemented by standard testing (CA125 or RMI score) for ovarian cancer in patients greater than CA125 or RMI alone (misclassification rate 4.54–3.03 percent vs. 9.09–10.60). Therefore, in conjunction with traditional studies, IL-6 can be a beneficial therapeutic biomarker for the triage of patients with potential malignant ovarian mass [26]. The reproducibility of IL6 measurement may be a challenge in clinical practice as IL6 level can increase if infections and or inflammatory conditions occur.
2.2.2. Osteopontin (OPN)
Osteopontin is an adhesive glycophosphoprotein secreted by activated T lymphocytes, macrophages, and leukocytes, and found in the extracellular matrix, sites of inflammation and body fluids [75] Osteopontin is not only expressed in ovarian cancer but also in endometrial, cervical, breast, colorectal, nonsmall cell lung, prostate, hepatocellular and gastric cancer. OPN is associated with tumour progression, invasion and metastasis. In 2001, OPN was identified with a cDNA microarray system using RNA isolated from several ovarian cancer cell lines, with surface epithelial cells as controls [50,76]. The levels of OPN were also significantly higher (p < 0.001) in the plasma samples of 51 women with EOC (486.5 ng/mL, n = 51) compared with the healthy controls (147.1 ng/mL, n = 107), benign ovarian disease (254.4 ng/mL, n = 46), and other gynaecological cancers (260.9 ng/mL, n = 47) [77].
In addition, OPN has been utilised to predict the progression of disease in advanced EOC, as the prognosis of patients with peritoneal spread is poor. The levels of osteopontin in 32 out of 40 peritoneal metastatic biopsies were found to be significantly elevated compared to the levels found in primary ovarian tumour tissues among women with Stage III EOC [78]. In addition, the elevated OPN levels were independently correlated with extremely poor prognosis among these women (n = 32), whereas 75% of the women found with no increase in OPN levels had a 36-month survival rate (n = 8). Furthermore, the high levels of osteopontin could be measured in the urine samples of patients with high-grade ovarian cancer, so this test could potentially be used clinically as a noninvasive tool for the early diagnosis of ovarian cancer [79].
2.2.3. Kallikreins (KLKs)
Kallikreins are a subgroup of serine proteases with different physiological roles. The human kallikrein gene family has now been entirely defined to include 15 members on chromosome 19q. They are expressed in epithelial and endocrine tissues regulated by hormones in cancer and they are shed and detected in human body fluids [34]. Therefore, many studies have been carried out to find their role in cancer diagnosis and prognosis. A total of 12 out 15 KLKs are upregulated in ovarian cancer, with some KLKs correlating to poor prognosis and late-stage disease (4–7, 10 and 15), as well as chemoresistance (KLK 4 and 7) to a first line paclitaxel agent [80].
In a study by Luo et al., the preoperative serum level of human kallikrein (hk10) in 146 patients with ovarian cancer was significantly elevated compared to 97 healthy women and 141 women with benign gynaecological diseases [81].
2.2.4. Bikunin
Bikunin is a multifunctional glycoprotein, which mediates the suppression of tumour cell invasion and metastasis. The measurement of bikunin levels in the tissue of patients with malignant diseases has been introduced as a simple diagnostic tool for the evaluation of the prognosis. High preoperative bikunin levels have been reported to be a strong favourable prognostic marker for ovarian cancer [13]. Matsuzaki et al. found, in an extensive study, that bikunin protein in the plasma of women with ovarian cancer (N = 327), compared to those with benign ovarian mass (N = 200) and healthy controls (N = 200), may be useful in evaluating the prognosis of the disease.
A low bikunin level (≤11.5 ug/mL) was found to be associated with the late-stage (Stage III/IV) disease, the presence of large residual tumours (>2 cm) and poor response to chemotherapy. The median survival time was also shorter, at 26 months compared to 60 months, in those with high levels of bikunin (p = 0.002), thus corresponding to a 2.2-fold higher risk of dying (hazard ratio, 0.45; p = 0.023) [13]. Measuring levels of bikunin in plasma is easy and relatively inexpensive; therefore, it has the potential to be included as a prognostic biomarker for ovarian cancer. However, there is a significant overlap in bikunin levels across cancer, benign and healthy controls, which needs to be further investigated before it can be of clinical use.
2.2.5. Human Epididymis Protein 4 (HE4)
Also known as WAP 4-disulphide core domain 2 (WFDC2), HE4 was first introduced as an ovarian cancer biomarker in 1999 [36]. The expression of HE4 is associated with cancer cell adhesion, migration and tumour growth, which can be related to its effects on the EGFR-MAPK signalling pathway [82]. Many studies suggested that HE4 is absent in normal ovarian surface epithelium but is expressed specifically in 100% of human endometrioid epithelial ovarian cancers (n = 16) and 93% of serous ovarian carcinomas stained for HE4 (n = 60) [57]. An ELISA analysis of serum HE4 levels in 37 patients with ovarian cancer, compared with 65 healthy controls, showed that HE4 had the same specificity and sensitivity as CA125 and detected fewer false positives in patients without a malignant disease [74].
The marker HE4 is significantly increased in ovarian and endometrial cancer, but not in endometriosis. HE4 can be increased, although it is less frequently elevated than CA125 in patients with benign disease, especially in premenopausal patients. The alternate probability of a malignancy algorithm (ROMA) blends the values of CA125 and HE4 with menopausal status in the predictive index and has been shown to stratify patients into high and low risk categories, with differing outcomes across many trials [83].
2.2.6. Vascular Endothelial Growth Factor (VEGF)
VEGF is a vascular permeability factor that is a key regulator of physiological and pathological angiogenesis, and makes a major contribution to tumorigenesis [84]. VEGF levels are known to be elevated in patients with ovarian cancer and contribute to the accumulation of ascites [85]. An analysis associated with VEGF levels in the preoperative sera of 314 patients with ovarian cancer recorded that higher VEGF levels were separately correlated with shorter survival periods [86]. In addition, tumour samples from 18 patients with advanced stage serous epithelial ovarian cancer were evaluated for VEGF expression by a reverse-transcriptase polymerase chain reaction (RT-PCR) [87]. It was demonstrated that 12 samples were found to be strongly positive, whereas six samples had low/negative VEGF expression. The median survival was longer, at 60 months in the VEGF-low/negative group compared to 28 months in the VEGF-positive group (p = 0.058).
Bevacizumab, the first and most studied anti-VEGF agent, when used as maintenance therapy following surgical debulking and first-line chemotherapy, led to significant improvements in the progression-free survival of patients with ovarian cancer but did not have an impact on the survival. Bevacizumab, in addition to PARP inhibitors, is currently being studied in Phase III PAOLO-1/ ENGOT-ov25 trials and has shown promising results, with reduced risk of disease progression by 41% overall, and by 69% in the subset of women with BRCA-mutated disease [88].
2.2.7. Human Prostasin (PSN)
PSN is a trypsin-like proteinase (40 KDa) found on chromosome 16p11.2. It plays a major role in the activation of epithelial sodium channels and in the reduction of invasive prostate and breast cancers in vitro [89]. Similarly, the epidermal tight junction forming and terminal differentiation are related to the matriptase-prostasin proteolytic pathway [90].
The potential use of prostasin as a novel biomarker for ovarian carcinoma was proposed by Mok et al. using microarray technologies to classify upregulated genes for secretive proteins [91]. The findings revealed an overexpression of PSN in malignant epithelial ovarian cells and stroma, relative to standard ovary tissue, with a sensitivity and specificity of 51.4% and 94%, respectively [92]. Gene expression analysis indicated that PSN was expressed in ovarian cancer at levels more than 100 times greater than those found in normal or benign ovarian lesions. This overexpression signature was found in the early stages of ovarian cancer and maintained in the higher stages and grades [93]. Costa et al., on the other hand, reported a slightly higher overexpression of mRNA prostasin in freshly frozen ovarian cancer tissues than in usual controls. Thus, it has the ability to be used clinically as a differential diagnostic marker for ovarian cancer [93]. In another study by Mok et al., the combination of CA125 and prostasin gave a sensitivity of 92% and a specificity of 94% for detecting ovarian cancer [91].
2.2.8. Creatine Kinase B (CKB)
Creatine kinase plays a crucial function in the energy homeostasis of vertebral cells. CKB is a cytosolic isoform of creatine kinase that displays upregulated expression in a number of cancers. It has been reported that certain ovarian cancer tissues have improved protein CKB expression [94]. In addition, CKB decreased the intake of glucose and lactate, and improved the ROS output and consumption of oxygen. As a result, it was indicated that the suppression of CKB induced G2 arrest in the cell cycle through the PI3K/AKT and AMPK pathways. Clinically, this mechanism has helped clinicians to use this biomarker in cancer cell survival and tumour progression. CKB activity measured in preoperative serum samples was higher in women with ovarian cancer (N = 45), compared to those with benign ovarian mass (9.6 U/L, N = 49) and healthy controls (8.5 U/L, N = 37), p = 0.0096 [27]. CKB is highly expressed in early stage ovarian tumour tissues and is, therefore, a potential biomarker for the early detection of ovarian cancer; it should be further investigated [27].
2.2.9. Mesothelin
Mesothelin, a tumour differentiation antigen found in mesothelial pleura, peritoneum and pericardium, was discovered in 1996 at the National Cancer Institute [95]. Mesothelin is widely expressed in many tumours, including 70% of ovarian cancers. Several mesothelin-directed treatments have been studied in clinical trials, including antimesothelin immunotoxins and antibody-drug conjugates (ADC) [96]. Quanz et al. showed the activity of anetumabravtansine in conjunction with conventional chemotherapy in ovarian cancer models. Anetumabravtansine is an ADC that produces a human antimesothelin antibody conjugated by a reducible disulphide linker to the DM4maytansinoid tubulin inhibitor. Both in vitro and in vivo experiments have indicated the selective activity of anetumabravtansine in injecting new expression cells and tumours, including low-sensitivity (68.2%) and high-specificity (80.5%) ovarian cancer [97]. In animal models with ovarian cancer, treatment with anetumabravtansine exhibits improved potency in combination with carboplatin, compared to either drug alone [97]. Similarly, Anetumabravtansine also demonstrates enhanced antitumour efficacy when combined with Bevacizumab, an anti-VEGF agent. A phase 1b study (NCT02751918) using anetumabravtansine in combination with pegylated liposomal doxorubicin in ovarian cancer patients is ongoing.
2.2.10. Apolipoprotein A-I (apoA-I)
ApoA-I is a high-density lipoprotein (HDL) and apolipoprotein A-I in plasma. Apo A-I levels have been reported to decrease in the sera of patients with ovarian cancer [44]. A multiplexed magnetic nanoparticle-antibody conjugates (MNPs-Abs) based fluorescence spectroscopic system analysis combining CA125, β2-M and ApoA1 for the early detection of ovarian cancer performed by Pal et al. found that while CA125 detection only identifies 50–60% of early stage ovarian cancer, the combination of the three biomarkers achieved high sensitivity (94%) and high specificity (98%) in distinguishing early stage ovarian cancer patients from healthy individuals [98]. This proposed multiplexed panel assay is also cost-effective, and further clinical investigation should be conducted to develop a clinically beneficial test kit.
2.2.11. Transthyretin (TTR)
TTR is a natural serum protein synthesised mostly in the liver [99]. It attaches and transports the thyroid hormones and retinol protein binding to the retinal complex [100]. Low TTR serum levels were found in ovarian cancer and used with other biomarkers to detect ovarian cancer [29,101]. Using liquid chromatography with tandem mass spectrometry, Kozak et al. found that TTR, in combination with beta-haemoglobin, apolipoprotein AI, transferrin and CA125, significantly improved the detection of early stage ovarian cancer [101]. TTR was found to be an important marker for the detection of stage I–II ovarian cancer, with a sensitivity and specificity of 78.6% and 68.8%, respectively.
2.2.12. Transferrin
Transferrin is essentially synthesised in hepatocytes and responsible for delivering plasma iron to the cell. It plays a major role in cell division and proliferation [43]. Ahmed et al. documented the downregulation of transferrin in the sera of patients with ovarian cancer [102]. In another case-control study, the level of transferrin was measured using an immunological turbidimetric assay in the sera of 37 women with ovarian cancer and compared to those with benign ovarian diseases (N = 31) and age–matched healthy controls (N = 31). It was found that the use of the biomarker transferrin as a detection tool for ovarian cancer has only low sensitivity and specificity, at 72.9% and 74.1%, respectively [40]. Therefore, transferrin needs to be used in combination with other biomarkers to achieve clinical significance.
2.3. Highlighting the Most Common Biomarker Combinations for the Management of Ovarian Cancer
It has been shown that a combination of certain potential biomarkers could significantly improve the detection and management of ovarian cancer. One of the most common combinations is the use of CA125 and PSN together, which resulted in an improved sensitivity (92%) and specificity (94%), compared with CA125 alone (sensitivity of 64.9% at a specificity of 94%) and PSN (sensitivity of 51.4% at a specificity of 94%) [92]. In addition, a combination of Apo-A1, TT, Connective tissue activating peptide III (CTAPIII) and CA125 achieved a sensitivity of up to 84% and a specificity of 98% in distinguishing women with early stage ovarian cancer from healthy individuals [40]. When ApoA1 was combined with CA125 and TTR, not only was a significant improvement observed in the overall sensitivity and specificity, but the panel was also sufficient for maximum discrimination between noncancer, stage I–II and all stages (I–IV) of ovarian cancer [103].
Similarly, Kozak et al. revealed that the TTR, Hb, ApoAI and TF biomarkers, when integrated with CA125, should significantly improve the detection of early stage ovarian cancer [104]. In addition, Kim et al. suggested the benefit of the combination of TTR, apolipoprotein A1 and CA125 in the diagnosis of ovarian cancer. The combination of CA125, transferrin, TTR and ApoA1, using a proteomic analysis, yielded a sensitivity of 89% at a specificity of 92% for the early detection of ovarian cancer [105].
3. Emerging Predictive Ovarian Cancer Biomarkers
3.1. An Overview of the Molecular Approaches to the Discovery of New Ovarian Cancer Biomarkers
To identify new biomarkers requires intensive, specific technologies and methods for the detection of molecules, genes and proteins in human body fluids and tissues. This section addresses some of the most common technologies recently used to discover new biomarkers of ovarian cancer.
3.1.1. Whole Genome Analysis
Comparative genomic hybridisation (CGH) is a full genome assay that detects gene copy gains or losses. This assay found multiple chromosome regions with an abnormal gene copy number for ovarian cancer [106]. Genomic expression profiling studies on epithelial ovarian cancer of different histology have elucidated not only global gene expression profiles and signalling pathways to distinguish and characterise each subtype of cancer, but also potential prognostic indicators [103].
3.1.2. Transcription Profiling
Transcription profiling, also known as ‘expression profiling’, is one of the most common types of analysis. It includes the quantification of the gene expression of several genes in the transcription (RNA) of cells or tissue samples. Quantification can be achieved by collecting biological samples and extracting RNA after a treatment or at fixed time-points in a time-series, thus generating “snapshots” of the expression patterns. Various histological subtypes of ovarian cancer are associated with different prognoses, and several transcription profiling studies have focused on the discovery of markers that can differentiate between subtypes. A number of transcription profiling studies have shown that a certain correlation between the subtypes of gene expression signature still exist, indicating some of the shared mechanisms underlying ovarian carcinogenesis [107]. Transcription profiling studies have established markers that could predict patient survival [108].
3.1.3. MicroRNA Profiling
MicroRNA was first observed in Caenorhabditis elegans in 1993 [108]. These RNAs contain 19 to 24 nucleotides and do not encode proteins. They interact with the three untranslated target mRNAs’ region, leading them to target mRNA degradation and inhibition [109].
MicroRNAs have been shown to be differentially expressed in tumours versus normal tissues in a number of solid and hematopoietic tumours. In certain cases, distinct microRNA signatures can reliably differentiate tumours from normal tissues and are associated with disease outcomes. In addition, a study examining microRNA signatures in a variety of tumour types indicated that the expression pattern of a relatively small number of microRNAs (approximately 200) was more reliable than cDNA arrays in the classification of human cancers [109]. These studies strongly suggest that microRNA profiling may have significant potential for cancer diagnosis and prognosis.
3.1.4. Proteomic Profiling
One major drawback of transcription profiling studies is that the changes in the mRNA level do not always result in changes in protein levels. Proteomic profiling has also recently been recognised as the most direct approach in the search for diagnostic and prognostic ovarian cancer biomarkers, hence mass spectrometry is one of the key methods used in proteomic profiling. The proteomics of ovarian cancer can be done using two techniques. One is the detection of distinct proteomic peptide patterns in cancer samples [1]. The second is to classify individual peptides that can distinguish between cancer and normal samples.
In addition to the proteomic profiling of serum and plasma samples, profiling can also be performed on other body fluids. These include glycosylated eosinophilic-derived neurotoxins and COOH-terminal osteopontin fragments, and the most established is ovarian cancer ascites proteome, where approximately 80 biomarkers have been discovered and identified as potential markers for early stage ovarian cancer detection [110].
3.2. Emerging Biomarkers Associated with Ovarian Cancer Diagnosis and Prognosis
The diagnosis of ovarian cancer is currently focused on restricted imaging techniques and the concentration of certain biomarkers circulating with established levels of sensitivity and specificity. New biomarkers are required to complement and improve the efficacy of the existing clinical tests. New biomarkers, including circulating DNA tumours, serum tumour proteins, circulating cancer cells or serum metals such as Cu and zinc, are emerging to complement the clinically available diagnostic methods [19,111]. A summary of these new biomarkers is shown in Table 2.
3.2.1. Cu Isotope
The concentration of Copper (Cu) in the bloodstream is regulated by two major organs: it is absorbed by the intestine and transported to the liver [112]. Changes in the concentration of Cu influenced by modified metabolic processes can affect health and disease [46]. In recent study looking at copper composition 65Cu/63Cu ratios (Δ65Cu), in blood samples from 44 ovarian cancer patients, and 13 ovarian biopsies using multicollector inductively coupled plasma mass spectrometry, a connection has been demonstrated to cancer progression [113]. The copper isotope ratio δ65Cu in the plasma of ovarian cancer patients (n = 44) was shown to be lower, in comparison to the levels among healthy donors (n = 48), suggesting that the serum was enriched with 63Cu.
3.2.2. Exosomes
Exosomes are endocytic and heterogeneous membrane-derived vesicles that are actively secreted by various forms of cell, and they can be visualised by electron microscopy [114]. Recent studies have reported the role of exosomes in immune regulation [115], intercellular communication [116,117] and biological events such as the coagulation [118] and microenvironmental regulation of tissues [119], as well as their role in the development of cancer, metastases and drug resistance [30,120].
In addition, a recent clinical trial found that the levels of exosomes were three to four times higher in the circulation of ovarian cancer patients compared to normal individuals [121]. As a result, a growing interest in determining the therapeutic importance of these nanoparticles in cancers has contributed to the discovery of either tissue or disease-specific exosome material, such as nucleic acids, proteins and lipids, as a source of new biomarkers [47]. While exosomes are known to be ideal biomarkers in the diagnosis of cancer due to their unique characteristics, there is still a long way to go in developing exosome-based assays.
3.2.3. lncRNA and mRNA Biomarkers
Recent research has suggested that certain transcriptomes contain noncoding RNAs, such as long noncoding (lncRNAs) and microRNAs (miRNAs), as well as coding RNAs (mRNAs) as modern multiomics for the production of predictive, preventive and personalised medicine (PPPM) for ovarian cancers [48,122]. PPPM has been found to be an effective and affordable strategy for ovarian cancer care [123].
The first comprehensive study, conducted in 2019, investigated the lncRNA–miRNA–mRNA networks and lncRNA–RNA binding protein-mRNA networks in ovarian cancer, and confirmed the presence of some lncRNAs and mRNAs in ovarian cancer cell models [48].
These may be important sources for developing new biomarkers and anticancer targets for early stage detection and the successful treatment of ovarian cancer patients. However, further research and clinical practice is needed to improve the understanding and application of transcriptomes (lnRNAs, miRNAs, and mRNAs) in the management of ovarian cancer.
In EOC, some lncRNAs were found to be differentially expressed compared with benign and normal tissues, which demonstrated the up- or downregulation of 663 lncRNAs [124]. Based on a systematic analysis of the profiles of lncRNA and mRNA expression from the Cancer Genome Atlas (TCGA), a platinum resistance-specific lncRNA-mRNA network was discovered involving a total of 124 significant lncRNA-mRNA coexpression relationships that primarily regulate metabolic pathways, indicating the prognostic and therapeutic potential of lncRNAs in high-grade serous ovarian cancer [125].
3.2.4. Aldehyde Dehydrogenase 1 (ALDH1)
A member of the aldehyde dehydrogenase protein family, ALDH1A1 expressed in a subpopulation of tumour-initiating cells and is therefore a potential candidate biomarker for cancer therapy. In a recent study by Chang et al. utilising an immunohistochemistry staining microarray study, the possible functions of ALDH1 in ovarian cancer consisted of the diagnosis of tumour type and disease staging, as well as therapeutic responses and overall survival rate. The data have shown that ALDH1 expression has been linked with longer average patient survival and that elevated ALDH1 expression is a positive prognosis factor in patients with ovarian cancer [126].
Comparably, a more recent analysis assessing the expression of ALDH1 in EOC stem cells found a higher expression of ALDH1 in CD44+ stem cell clones [49]. Hence, ALDH1 may be a useful biomarker for the identification of tumorigenic stem cells. Similarly, high ALDH1 expression in tumour cells was significantly associated with histological subtypes, the early FIGO stage, a well-differentiated grade and better survival probability (p < 0.05). The expression of ALDH1 in stromal cells had no clinicopathological associations in the present study (p > 0.05) [127].
3.2.5. Folate Receptor Alpha (FOLR1)
FOLR1 is a membrane-bound receptor protein that is active in the movement of folate to cells and other cellular processes. The overexpression of FOLR1 was found in 69% of uterine serous carcinoma [128] and rapidly dividing cells. The expression of FOLR1 is regulated by the loss of extracellular folate levels, the accumulation of homocysteine, steroid hormone levels and genetic mutations. An initial study suggested a significant correlation between folate levels and tumour aetiology, and folate levels and progression, with suggestions for future research in FOLR1 gene expression and regulation [129].
In addition, the overexpression of FOLR1 has been documented in various epithelial nonmucinous tumours, including ovarian carcinoma; however, its assessment as a novel biomarker for early detection has not yet been verified. In either case, the overexpression of FOLR1 was reported in serous ovarian carcinoma describing clinicopathological characteristics and outcomes, as well as the relationship between FOLR1 and chemoresistance [50].
3.2.6. Glutathione S-Transferase Polymorphisms
Members of the Glutathione S-transferase family (GSTM1, GSTT1, and GSTP1) are the result of major structural gene deletions, which in turn control the metabolism of drugs and impair chemotherapy in cancer patients. The GST polymorphisms are highly expressed in human ovaries [130]. Earlier epidemiologic studies did not confirm the association of GST polymorphisms with epithelial ovarian cancer [131], while it was proposed that in persons with homozygous deletions of GSTM, where GSTT had decreased or where there was no GST involvement, the removal of electrophilic carcinogens was challenging.
In addition, a recent analysis utilising DNA extracts from epithelial ovarian cancer tissue in which the GSTT1, GSTM1 and GSTP1 genotypes were identified using multiplex PCR and PCR-RFLP indicated that the combination of no GSTM1 and low GSTP1 resulted in over 60% improvement in progression-free survival and almost 40% improvement in overall survival [132]. Similarly, a meta-analysis investigating the relationship of GST polymorphisms with ovarian cancer risk suggested that the role of GSTs is highly significant in drug-resistant tumours where the higher expression of GSTs could alter the control of the kinase cascade during drug therapy [133].
4. Summary and Discussion
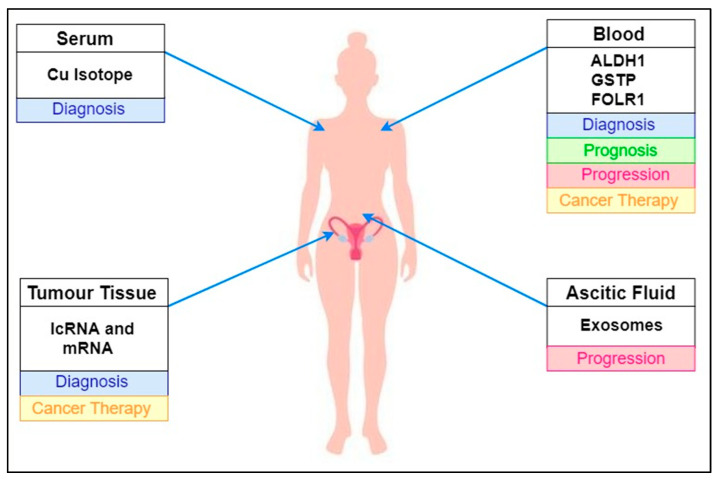
Despite both the conventional and new methods used to detect the development of ovarian cancer, such as radiographic imaging, invasive biopsies, tumour markers, and a combination of transvaginal ultrasound and tumour markers, ovarian cancer remains the most common gynaecological malignancy and has the highest mortality rate. The identification and confirmation of early warning biomarkers that are particularly specific to ovarian cancer involve the creation of minimally invasive screening approaches to identify the early onset of ovarian cancer. A summary of newly emerging ovarian cancer biomarkers highlighting their primary location in human body fluids is illustrated in Figure 2. The evaluation of promising early detection biomarkers opens up broad horizons for the detection and treatment of ovarian cancer.
Figure 2.
Summary of newly emerging ovarian cancer biomarkers highlighting their primary location in human body fluids.
Human serum proteome research has offered improved biomarker candidates for early detection, which is a crucial advancement as early diagnosis increases the five-year survival rate by more than 90%. CA125 is a well-studied high-discriminative tumour marker, especially among postmenopausal women, and it rises well before the emergence of clinical symptoms. However, it is recognised that the spike in CA125 levels in other types of cancer, benign ovarian disease, endometriosis, inflammatory conditions and ovulation, as well as its low early stage sensitivity, limit its ability as a single biomarker for ovarian cancer screening. As a consequence, a multibiomarker panel is recommended to strengthen the sensitivity and specificity of CA125, in which CA125 is used with HE4, mesothelin or a number of combinations (See Section 2.3. Highlight of the Most Common Biomarkers Combination for Ovarian Cancer Management) through which greater sensitivity and specificity has been achieved. For example, HE4 and mesothelin have been the most promising candidates to date.
Häusler et al. demonstrated higher expressions of miR-21, miR-141, miR-200a, miR-200c, miR-203, miR-205, and miR-214, as well as similarities in miRNA profiling in exosomal microRNA in ovarian cancer patients, suggesting that miRNA profiling could be a possible new technology for the early detection of ovarian cancer, biopsy profiling, and screening in asymptomatic populations [134]. On the other hand, studies have shown that ALDH1-positive ovarian cancer cells enhance tumorigenicity and chemoresistance; thus, the early detection of ovarian carcinomas along with other markers of gene mutations in BRCA1/BRCA2, Prostasin (PRSS8), GSTT1, FOLR1, KLK6, KLK7, and ALDH1 (Table 2) can also be predicted, requiring further investigation and clinical trials.
5. Future Directions
Numerous genomic and proteomic profiling experiments have provided valuable mechanistic knowledge on the development and evolution of new ovarian cancer biomarkers. However, several profiling experiments have been conducted on a small number of samples underdefined parameters, while some other studies display minimal overlap and are not necessarily reproducible. In order to verify the discriminatory influence of these emerging biomarkers, a large number of samples with multiple and separate sample sets would be needed. Promising biomarkers proposed in this study include the Cu isotope, exosomes, GSTT1, FOLR1, ALDH1, and mRNAs, along with multibiomarker panels in conjunction with CA125, as the most developed and efficient biomarkers currently available for clinical use.
6. Conclusions and Recommendations
Ovarian cancer is a high-mortality gynaecological condition affecting women all over the world. While substantial improvement has been made in the detection and overall five-year survival rate of ovarian cancer patients, both rates remain very low. This is because successful early stage diagnostic biomarkers and therapeutic goals remain inadequate. It is also important to identify novel early detection molecules or therapeutic targets, including pathways with a minimally invasive approach and a high sensitivity and specificity, which can greatly increase the overall survival rate and quality of life of ovarian cancer patients.
Author Contributions
G.A.A. and N.C.K. finalised the primary literature search and screening within all available databases for relevant articles and clinical studies. N.H.A.A., M.N.S. and C.K.T. were major contributors in writing and editing the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This review study received funding from the Fundamental Research Grant Scheme by the Ministry of Higher Education Malaysia, FRGS/1/2018/SKK08/UKM/02/6.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of Universiti Kebangsaan Malaysia, FRGS/1/2018/SKK08/UKM/02/6 (29th October 2018).
Informed Consent Statement
Not applicable.
Data Availability Statement
No new data were created or analysed in this study. Data sharing is not applicable to this article.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Jayson G.C., Kohn E.C., Kitchener H.C., Ledermann J.A. Ovarian cancer. Lancet. 2014;384:1376–1388. doi: 10.1016/S0140-6736(13)62146-7. [DOI] [PubMed] [Google Scholar]
- 2.Ries L.A.G., Kosary C.L., Hankey B.F., Miller B.A., Edwards B.K. SEER Cancer Statistics Review, 1973–1995. National Cancer Institute; Bethesda, MD, USA: 1998. [Google Scholar]
- 3.Prat J. Pathology of borderline and invasive cancers. Best Pr. Res. Clin. Obstet. Gynaecol. 2017;41:15–30. doi: 10.1016/j.bpobgyn.2016.08.007. [DOI] [PubMed] [Google Scholar]
- 4.Carcangiu M.L., Kurman R.J., Carcangiu M.L., Herrington C.S. WHO Classification of Tumours of Female Reproductive Organs, International Agency for Research on Cancer. Wiley Online Library; Hoboken, NJ, USA: 2014. [Google Scholar]
- 5.Grisham R.N. Low-Grade Serous Carcinoma of the Ovary. Oncology. 2016;30:650–652. [PubMed] [Google Scholar]
- 6.Ferlay J., Steliarova-Foucher E., Lortet-Tieulent J., Rosso S., Coebergh J., Comber H., Forman D., Bray F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries in 2012. Eur. J. Cancer. 2013;49:1374–1403. doi: 10.1016/j.ejca.2012.12.027. [DOI] [PubMed] [Google Scholar]
- 7.Klotz D.M., Wimberger P. Cells of origin of ovarian cancer: Ovarian surface epithelium or fallopian tube? Arch. Gynecol. Obstet. 2017;296:1055–1062. doi: 10.1007/s00404-017-4529-z. [DOI] [PubMed] [Google Scholar]
- 8.Kurman R.J., Shih I.-M. The Origin and Pathogenesis of Epithelial Ovarian Cancer: A Proposed Unifying Theory. Am. J. Surg. Pathol. 2010;34:433–443. doi: 10.1097/PAS.0b013e3181cf3d79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ledermann J.A., Raja F.A., Fotopoulou C., Gonzalez-Martin A., Colombo N., Sessa C. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2013;24:vi24–vi32. doi: 10.1093/annonc/mdt333. [DOI] [PubMed] [Google Scholar]
- 10.A Doherty J., Peres L.C., Wang C., Way G.P., Greene C.S., Schildkraut J.M. Challenges and Opportunities in Studying the Epidemiology of Ovarian Cancer Subtypes. Curr. Epidemiol. Rep. 2017;4:211–220. doi: 10.1007/s40471-017-0115-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Smolle E., Taucher V., Pichler M., Petru E., Lax S., Haybaeck J. Targeting Signaling Pathways in Epithelial Ovarian Cancer. Int. J. Mol. Sci. 2013;14:9536–9555. doi: 10.3390/ijms14059536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Norquist B.M., Garcia R.L., Allison K.H. The molecular pathogenesis of hereditary ovarian carcinoma: Alterations in the tubal epithelium of women with BRCA1 and BRCA2 mutations. Cancer. 2010;116:5261–5271. doi: 10.1002/cncr.25439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Press J.Z., Wurz K., Norquist B.M., Lee M.K., Pennil C., Garcia R., Welcsh P., Goff B.A., Swisher E.M. Identification of a Preneoplastic Gene Expression Profile in Tubal Epithelium of BRCA1 Mutation Carriers. Neoplasia. 2010;12:993–1002. doi: 10.1593/neo.101044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Press J.Z., De Luca A., Boyd N., Young S., Troussard A., Ridge Y., Kaurah P., Kalloger S., A Blood K., Smith M., et al. Ovarian carcinomas with genetic and epigenetic BRCA1 loss have distinct molecular abnormalities. BMC Cancer. 2008;8:17. doi: 10.1186/1471-2407-8-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tinelli A., Malvasi A., Leo G., Vergara D., Pisanò M., Ciccarese M., Chiuri V.E., Lorusso V. Hereditary ovarian cancers: From BRCA mutations to clinical management. A modern appraisal. Cancer Metastasis Rev. 2010;29:339–350. doi: 10.1007/s10555-010-9218-3. [DOI] [PubMed] [Google Scholar]
- 16.Scully R., Livingston D.M. In search of the tumour-suppressor functions of BRCA1 and BRCA2. Nat. Cell Biol. 2000;408:429–432. doi: 10.1038/35044000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liang H., Tan A.R. PARP Inhibitors. Curr. Breast Cancer Rep. 2011;3:44–54. doi: 10.1007/s12609-010-0036-y. [DOI] [Google Scholar]
- 18.Mangerich A., Bürkle A. How to kill tumor cells with inhibitors of poly(ADP-ribosyl)ation. Int. J. Cancer. 2010;128:251–265. doi: 10.1002/ijc.25683. [DOI] [PubMed] [Google Scholar]
- 19.I Szabo C., King M.C. Inherited breast and ovarian cancer. Hum. Mol. Genet. 1995;4:1811–1817. doi: 10.1093/hmg/4.suppl_1.1811. [DOI] [PubMed] [Google Scholar]
- 20.Moore K., Colombo N., Scambia G., Kim B.-G., Oaknin A., Friedlander M., Lisyanskaya A., Floquet A., Leary A., Sonke G.S., et al. Maintenance Olaparib in Patients with Newly Diagnosed Advanced Ovarian Cancer. N. Engl. J. Med. 2018;379:2495–2505. doi: 10.1056/NEJMoa1810858. [DOI] [PubMed] [Google Scholar]
- 21.Nakayama K., Nakayama N., Kurman R.J., Cope L.M., Pohl G., Samuels Y., Velculescu V.E., Wang T.-L., Shih I.-M. Sequence mutations and amplification of PIK3CA and AKT2 genes in purified ovarian serous neoplasms. Cancer Biol. Ther. 2006;5:779–785. doi: 10.4161/cbt.5.7.2751. [DOI] [PubMed] [Google Scholar]
- 22.Singer G., Oldt R., Cohen Y., Wang B.G., Sidransky D., Kurman R.J., Shih I.-M. Mutations in BRAF and KRAS Characterize the Development of Low-Grade Ovarian Serous Carcinoma. J. Natl. Cancer Inst. 2003;95:484–486. doi: 10.1093/jnci/95.6.484. [DOI] [PubMed] [Google Scholar]
- 23.Peyssonnaux C., Eychène A. The Raf/MEK/ERK pathway: New concepts of activation. Biol. Cell. 2001;93:53–62. doi: 10.1016/S0248-4900(01)01125-X. [DOI] [PubMed] [Google Scholar]
- 24.Allen L.F., Sebolt-Leopold J., Meyer M.B. Seminars in Oncology. Elsevier; Amsterdam, The Netherlands: 2003. CI-1040 (PD184352), a targeted signal transduction inhibitor of MEK (MAPKK) [DOI] [PubMed] [Google Scholar]
- 25.Choi K.-C., Kang S.K., Tai C.-J. Follicle-stimulating hormone activates mitogen-activated protein kinase in preneoplastic and neoplastic ovarian surface epithelial cells. J. Clin. Endocrinol. Metab. 2002;87:2245–2253. doi: 10.1210/jcem.87.5.8506. [DOI] [PubMed] [Google Scholar]
- 26.Gershenson D. Gynecologic Oncology. Academic Press Inc.; Cambridge, MA, USA: Elsevier Science; San Diego, CA, USA: 2020. A Randomized Phase II/III Study to Assess the Efficacy of Trametinib in Patientswith Recurrent or Progressive Low-Grade Serous Ovarian or Peritoneal Cancer; p. 92101. 525 B ST, STE 1900. [Google Scholar]
- 27.Farley J.H., Brady W.E., Vathipadiekal V., Lankes H.A., Coleman R.L., Morgan M.A., Mannel R.S., Yamada S.D., Mutch D.G., Rodgers W.H., et al. Selumetinib in women with recurrent low-grade serous carcinoma of the ovary or peritoneum: An open-label, single-arm, phase 2 study. Lancet Oncol. 2013;14:134–140. doi: 10.1016/S1470-2045(12)70572-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Zeineldin R., Muller C.Y., Stack M.S., Hudson L.G. Targeting the EGF Receptor for Ovarian Cancer Therapy. J. Oncol. 2009;2010:1–11. doi: 10.1155/2010/414676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Fontanini G., Vignati S., Bigini D., Mussi A., Lucchi H., Angeletti C., Pingitore R., Pepe S., Basolo F., Bevilacqua G. Epidermal growth factor receptor (EGFr) expression in non-small cell lung carcinomas correlates with metastatic involvement of hilar and mediastinal lymph nodes in the squamous subtype. Eur. J. Cancer. 1995;31:178–183. doi: 10.1016/0959-8049(93)00421-M. [DOI] [PubMed] [Google Scholar]
- 30.Jorissen R.N. Epidermal growth factor receptor: Mechanisms of activation and signalling. Exp. Cell Res. 2003;284:31–53. doi: 10.1016/S0014-4827(02)00098-8. [DOI] [PubMed] [Google Scholar]
- 31.Casanova M.L., Larcher F., Casanova B., Murillas R., Fernández-Aceñero M.J., Villanueva C., Martínez-Palacio J., Ullrich A., Conti C.J., Jorcano J.L. A critical role for ras-mediated, epidermal growth factor receptor-dependent angiogenesis in mouse skin carcinogenesis. Cancer Res. 2002;62:3402–3407. [PubMed] [Google Scholar]
- 32.A Altomare D., Wang H.Q., Skele K.L., De Rienzo A., Klein-Szanto A.J., Godwin A.K., Testa J.R. AKT and mTOR phosphorylation is frequently detected in ovarian cancer and can be targeted to disrupt ovarian tumor cell growth. Oncogene. 2004;23:5853–5857. doi: 10.1038/sj.onc.1207721. [DOI] [PubMed] [Google Scholar]
- 33.Harandi A., Zaidi A.S., Stocker A.M., Laber D.A. Clinical Efficacy and Toxicity of Anti-EGFR Therapy in Common Cancers. J. Oncol. 2009;2009:1–14. doi: 10.1155/2009/567486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kobayashi M., Sawada K., Kimura T. Potential of Integrin Inhibitors for Treating Ovarian Cancer: A Literature Review. Cancers. 2017;9:83. doi: 10.3390/cancers9070083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sawada K., Ohyagi-Hara C., Kimura T., Morishige K.-I. Integrin Inhibitors as a Therapeutic Agent for Ovarian Cancer. J. Oncol. 2011;2012:1–6. doi: 10.1155/2012/915140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Delie F., Petignat P., Cohen M. GRP78 Protein Expression in Ovarian Cancer Patients and Perspectives for a Drug-Targeting Approach. J. Oncol. 2012;2012:1–5. doi: 10.1155/2012/468615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Matrone A., Grossi V., Chiacchiera F., Fina E., Cappellari M., Caringella A.M., Di Naro E., Loverro G., Simone C. p38α Is Required for Ovarian Cancer Cell Metabolism and Survival. Int. J. Gynecol. Cancer. 2010;20:203–211. doi: 10.1111/IGC.0b013e3181c8ca12. [DOI] [PubMed] [Google Scholar]
- 38.Grossi V., Simone C. Special Agents Hunting down Women Silent Killer: The Emerging Role of the p38αKinase. J. Oncol. 2012;2012:1–7. doi: 10.1155/2012/382159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Bast R.C., Jr., Siegal F.P. Elevation of serum CA 125 prior to diagnosis of an epithelial ovarian carcinoma. Gynecol. Oncol. 1985;22:115–120. doi: 10.1016/0090-8258(85)90015-0. [DOI] [PubMed] [Google Scholar]
- 40.Paramasivam S., Tripcony L., Crandon A., Quinn M., Hammond I., Marsden D., Proietto A., Davy M., Carter J., Nicklin J., et al. Prognostic Importance of Preoperative CA125 in International Federation of Gynecology and Obstetrics Stage I Epithelial Ovarian Cancer: An Australian Multicenter Study. J. Clin. Oncol. 2005;23:5938–5942. doi: 10.1200/JCO.2005.08.151. [DOI] [PubMed] [Google Scholar]
- 41.Coticchia C.M., Yang J., Moses M.A. Ovarian cancer biomarkers: Current options and future promise. J. Natl. Compr. Cancer Netw. 2008;6:795–802. doi: 10.6004/jnccn.2008.0059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bao L.H., Sakaguchi H., Fujimoto J., Tamaya T. Osteopontin in metastatic lesions as a prognostic marker in ovarian cancers. J. Biomed. Sci. 2007;14:373–381. doi: 10.1007/s11373-006-9143-1. [DOI] [PubMed] [Google Scholar]
- 43.Ye B., Skates S., Mok S.C., Horick N.K., Rosenberg H.F., Vitonis A., Edwards D., Sluss P., Han W.K., Berkowitz R.S., et al. Proteomic-Based Discovery and Characterization of Glycosylated Eosinophil-Derived Neurotoxin and COOH-Terminal Osteopontin Fragments for Ovarian Cancer in Urine. Clin. Cancer Res. 2006;12:432–441. doi: 10.1158/1078-0432.CCR-05-0461. [DOI] [PubMed] [Google Scholar]
- 44.Brakora K., Lee H., Yusuf R., Sullivan L., Harris A.L., Colella T., Seiden M.V. Utility of osteopontin as a biomarker in recurrent epithelial ovarian cancer. Gynecol. Oncol. 2004;93:361–365. doi: 10.1016/j.ygyno.2004.01.050. [DOI] [PubMed] [Google Scholar]
- 45.A Borgoño C., Grass L., Soosaipillai A., Yousef G.M., Petraki C., Howarth D.H.C., Fracchioli S., Katsaros D., Diamandis E.P. Human kallikrein 14: A new potential biomarker for ovarian and breast cancer. Cancer Res. 2003;63:9032–9041. [PubMed] [Google Scholar]
- 46.Yousef G.M., ScorilasP A., Katsaros D., Fracchioli S., Iskander L., Borgono C., De La Longrais I.A.R., Puopolo M., Massobrio M., Diamandis E.P. Prognostic Value of the Human Kallikrein Gene 15 Expression in Ovarian Cancer. J. Clin. Oncol. 2003;21:3119–3126. doi: 10.1200/JCO.2003.09.111. [DOI] [PubMed] [Google Scholar]
- 47.Tanaka Y., Kobayashi H., Suzuki M., Kanayama N., Suzuki M., Terao T. Upregulation of bikunin in tumor-infiltrating macrophages as a factor of favorable prognosis in ovarian cancer. Gynecol. Oncol. 2004;94:725–734. doi: 10.1016/j.ygyno.2004.06.012. [DOI] [PubMed] [Google Scholar]
- 48.Tanaka Y., Kobayashi H., Suzuki M., Kanayama N., Suzuki M., Yamakawa T., Morishita H., Terao T. Reduced bikunin gene expression as a factor of poor prognosis in ovarian carcinoma. Cancer. 2003;98:424–430. doi: 10.1002/cncr.11506. [DOI] [PubMed] [Google Scholar]
- 49.Matsuzaki H., Kobayashi H., Yagyu T., Wakahara K., Kondo T., Kurita N., Sekino H., Inagaki K., Suzuki M., Kanayama N., et al. Plasma Bikunin As a Favorable Prognostic Factor in Ovarian Cancer. J. Clin. Oncol. 2005;23:1463–1472. doi: 10.1200/JCO.2005.03.010. [DOI] [PubMed] [Google Scholar]
- 50.Drapkin R., Von Horsten H.H., Lin Y., Mok S.C., Crum C.P., Welch W.R., Hecht J.L. Human Epididymis Protein 4 (HE4) Is a Secreted Glycoprotein that Is Overexpressed by Serous and Endometrioid Ovarian Carcinomas. Cancer Res. 2005;65:2162–2169. doi: 10.1158/0008-5472.CAN-04-3924. [DOI] [PubMed] [Google Scholar]
- 51.Moore R.G., Miller M.C., DiSilvestro P., Landrum L.M., Gajewski W., Ball J.J., Skates S.J. Evaluation of the Diagnostic Accuracy of the Risk of Ovarian Malignancy Algorithm in Women With a Pelvic Mass. Obstet. Gynecol. 2011;118:280–288. doi: 10.1097/AOG.0b013e318224fce2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hellström I., Raycraft J., Hayden-Ledbetter M., A Ledbetter J., Schummer M., McIntosh M., Drescher C., Urban N., Hellström K.E. The HE4 (WFDC2) protein is a biomarker for ovarian carcinoma. Cancer Res. 2003;63:3695–3700. [PubMed] [Google Scholar]
- 53.Yamamoto S., Konishi I., Mandai M., Kuroda H., Komatsu T., Nanbu K., Sakahara H., Mori T. Expression of vascular endothelial growth factor (VEGF) in epithelial ovarian neoplasms: Correlation with clinicopathology and patient survival, and analysis of serum VEGF levels. Br. J. Cancer. 1997;76:1221–1227. doi: 10.1038/bjc.1997.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hefler L.A., Zeillinger R., Grimm C., Sood A.K., Cheng W.-F., Gadducci A., Tempfer C.B.B., Reinthaller A. Preoperative serum vascular endothelial growth factor as a prognostic parameter in ovarian cancer. Gynecol. Oncol. 2006;103:512–517. doi: 10.1016/j.ygyno.2006.03.058. [DOI] [PubMed] [Google Scholar]
- 55.Sriram K.B., Relan V., E Clarke B., E Duhig E., A Yang I., Bowman R.V., Lee Y.C.G., Fong K.M. Diagnostic molecular biomarkers for malignant pleural effusions. Futur. Oncol. 2011;7:737–752. doi: 10.2217/fon.11.45. [DOI] [PubMed] [Google Scholar]
- 56.Mok S.C., Chao J., Skates S., Wong K.-K., Yiu G.K., Muto M.G., Berkowitz R.S., Cramer D.W. Prostasin, a Potential Serum Marker for Ovarian Cancer: Identification Through Microarray Technology. J. Natl. Cancer Inst. 2001;93:1458–1464. doi: 10.1093/jnci/93.19.1458. [DOI] [PubMed] [Google Scholar]
- 57.Costa F.P., Rodrigues-Junior V., Zelmanowicz A., Svedman C., Devenz G., Alves S., Da Silva A.S.M., Garicochea B. Prostasin, a potential tumor marker in ovarian cancer: A pilot study. Clinics. 2009;64:641–644. doi: 10.1590/S1807-59322009000700006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Li X.-H., Chen X.-J., Ou W.-B., Zhang Q., Lv Z.-R., Zhan Y., Ma L., Huang T., Yan Y.-B., Zhou H.-M. Knockdown of creatine kinase B inhibits ovarian cancer progression by decreasing glycolysis. Int. J. Biochem. Cell Biol. 2013;45:979–986. doi: 10.1016/j.biocel.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 59.Ghafoor A., Thomas A., Hassan R. Targeting mesothelin in ovarian cancer. Oncotarget. 2018;9:36050–36051. doi: 10.18632/oncotarget.26350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kreitman R.J., Hassan R., Fitzgerald D.J., Pastan I. Phase I Trial of Continuous Infusion Anti-Mesothelin Recombinant Immunotoxin SS1P. Clin. Cancer Res. 2009;15:5274–5279. doi: 10.1158/1078-0432.CCR-09-0062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Macuks R., Baidekalna I., Gritcina J., Avdejeva A., Donina S. Apolipoprotein A1 and Transferrin as Biomarkers in Ovarian Cancer Diagnostics. Acta Chir. Latv. 2010;10:16. doi: 10.2478/v10163-011-0003-3. [DOI] [Google Scholar]
- 62.Zamanian-Daryoush M., DiDonato J.A. Apolipoprotein A-I and Cancer. Front. Pharmacol. 2015;6 doi: 10.3389/fphar.2015.00265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zhang Z., Bast R.C., Yu Y., Li J., Sokoll L.J., Rai A.J., Rosenzweig J.M., Cameron B., Wang Y.Y., Meng X.-Y., et al. Three Biomarkers Identified from Serum Proteomic Analysis for the Detection of Early Stage Ovarian Cancer. Cancer Res. 2004;64:5882–5890. doi: 10.1158/0008-5472.CAN-04-0746. [DOI] [PubMed] [Google Scholar]
- 64.Liu L., Liu J., Dai S., Wang X., Wu S., Wang J., Huang L., Xiao X., He D. Reduced transthyretin expression in sera of lung cancer. Cancer Sci. 2007;98:1617–1624. doi: 10.1111/j.1349-7006.2007.00576.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Rastogi M., Gupta S., Sachan M. Biomarkers towards Ovarian Cancer Diagnostics: Present and Future Prospects. Braz. Arch. Biol. Technol. 2016;59:59. doi: 10.1590/1678-4324-2016160070. [DOI] [Google Scholar]
- 66.Yin B.W., Lloyd K.O. Molecular cloning of the CA125 ovarian cancer antigen identification as a new mucin, MUC16. J. Biol. Chem. 2001;276:27371–27375. doi: 10.1074/jbc.M103554200. [DOI] [PubMed] [Google Scholar]
- 67.Kampan N.C., Madondo M.T., Reynolds J., Hallo J., McNally O.M., Jobling T.W., Stephens A.N., Quinn M.A., Plebanski M. Pre-operative sera interleukin-6 in the diagnosis of high-grade serous ovarian cancer. Sci. Rep. 2020;10:1–15. doi: 10.1038/s41598-020-59009-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Chen Y.-L., Chiang Y.-C., Huang C.-Y., Lin H.-W., Chen C.-A., Hsieh C.-Y., Cheng W.-F. Serous ovarian carcinoma patients with high alpha-folate receptor had reducing survival and cytotoxic chemo-response. Mol. Oncol. 2011;6:360–369. doi: 10.1016/j.molonc.2011.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Wong K., Cheng R.S., Mok S.C. Identification of Differentially Expressed Genes from Ovarian Cancer Cells by MICROMAX™ cDNA Microarray System. Biotechniques. 2001;30:670–675. doi: 10.2144/01303dd05. [DOI] [PubMed] [Google Scholar]
- 70.Kim J.-H., Skates S.J., Uede T., Wong K., Schorge J.O., Feltmate C.M., Berkowitz R.S., Cramer D.W., Mok S.C. Osteopontin as a potential diagnostic biomarker for ovarian cancer. JAMA. 2002;287:1671–1679. doi: 10.1001/jama.287.13.1671. [DOI] [PubMed] [Google Scholar]
- 71.Diamandis E.P., Yousef G.M. Human Tissue Kallikreins: A Family of New Cancer Biomarkers. Clin. Chem. 2002;48:1198–1205. doi: 10.1093/clinchem/48.8.1198. [DOI] [PubMed] [Google Scholar]
- 72.Wang P., Magdolen V., Seidl C., Dorn J., Drecoll E., Kotzsch M., Yang F., Schmitt M., Schilling O., Rockstroh A., et al. Kallikrein-related peptidases 4, 5, 6 and 7 regulate tumour-associated factors in serous ovarian cancer. Br. J. Cancer. 2018;119:1–9. doi: 10.1038/s41416-018-0260-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Luo L.Y., Katsaros D., Scorilas A., Fracchioli S., Piccinno R., Longrais I.A.R.D.L., Howarth D.J., Diamandis E.P. Prognostic value of human kallikrein 10 expression in epithelial ovarian carcinoma. Clin. Cancer Res. 2001;7:2372–2379. [PubMed] [Google Scholar]
- 74.Schummer M., Ng W.V., E Bumgarner R., Nelson P.S., Schummer B., Bednarski D.W., Hassell L., Baldwin R.L., Karlan B.Y., Hood L. Comparative hybridization of an array of 21 500 ovarian cDNAs for the discovery of genes overexpressed in ovarian carcinomas. Gene. 1999;238:375–385. doi: 10.1016/S0378-1119(99)00342-X. [DOI] [PubMed] [Google Scholar]
- 75.Lu R., Sun X., Xiao R., Zhou L., Gao X., Guo L. Human epididymis protein 4 (HE4) plays a key role in ovarian cancer cell adhesion and motility. Biochem. Biophys. Res. Commun. 2012;419:274–280. doi: 10.1016/j.bbrc.2012.02.008. [DOI] [PubMed] [Google Scholar]
- 76.Huhtinen K., Suvitie P., Hiissa J., Junnila J., Huvila J., Kujari H., Setala M., Härkki P., Jalkanen J., Fraser J.K., et al. Serum HE4 concentration differentiates malignant ovarian tumours from ovarian endometriotic cysts. Br. J. Cancer. 2009;100:1315–1319. doi: 10.1038/sj.bjc.6605011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chung A.S., Ferrara N. Developmental and Pathological Angiogenesis. Annu. Rev. Cell Dev. Biol. 2011;27:563–584. doi: 10.1146/annurev-cellbio-092910-154002. [DOI] [PubMed] [Google Scholar]
- 78.A Nagy J., Masse E.M., Herzberg K.T., Meyers M.S., Yeo K.T., Yeo T.K., Sioussat T.M., Dvorak H.F. Pathogenesis of ascites tumor growth: Vascular permeability factor, vascular hyperpermeability, and ascites fluid accumulation. Cancer Res. 1995;55:360–368. [PubMed] [Google Scholar]
- 79.Hartenbach E.M., A Olson T., Goswitz J.J., Mohanraj D., Twiggs L.B., Carson L.F., Ramakrishnan S. Vascular endothelial growth factor (VEGF) expression and survival in human epithelial ovarian carcinomas. Cancer Lett. 1997;121:169–175. doi: 10.1016/S0304-3835(97)00350-9. [DOI] [PubMed] [Google Scholar]
- 80.Konstantinopoulos P.A., Lheureux S., Moore K.N. PARP Inhibitors for Ovarian Cancer: Current Indications, Future Combinations, and Novel Assets in Development to Target DNA Damage Repair. Am. Soc. Clin. Oncol. Educ. Book. 2020;40:e116–e131. doi: 10.1200/EDBK_288015. [DOI] [PubMed] [Google Scholar]
- 81.Cramer D.W., Bast R.C., Berg C.D., Diamandis E.P., Godwin A.K., Hartge P., Lokshin A.E., Lu K.H., McIntosh M.W., Mor G., et al. Ovarian Cancer Biomarker Performance in Prostate, Lung, Colorectal, and Ovarian Cancer Screening Trial Specimens. Cancer Prev. Res. 2011;4:365–374. doi: 10.1158/1940-6207.CAPR-10-0195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yu J., Chao L., Chao J. Prostasin is a novel human serine proteinase from seminal fluid. Purification, tissue distribution, and localization in prostate gland. J. Biol. Chem. 1994;269:18843–18848. doi: 10.1016/S0021-9258(17)32244-5. [DOI] [PubMed] [Google Scholar]
- 83.Tamir A., Gangadharan A., Balwani S., Tanaka T., Patel U., Hassan A., Benke S., Agas A., D’Agostino J., Shin D., et al. The serine protease prostasin (PRSS8) is a potential biomarker for early detection of ovarian cancer. J. Ovarian Res. 2016;9:1–13. doi: 10.1186/s13048-016-0228-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Huddleston H.G., Wong K., Welch W.R., Berkowitz R.S., Mok S.C. Clinical applications of microarray technology: Creatine kinase B is an up-regulated gene in epithelial ovarian cancer and shows promise as a serum marker. Gynecol. Oncol. 2005;96:77–83. doi: 10.1016/j.ygyno.2004.08.047. [DOI] [PubMed] [Google Scholar]
- 85.Chang K., Pastan I. Molecular cloning of mesothelin, a differentiation antigen present on mesothelium, mesotheliomas, and ovarian cancers. Proc. Natl. Acad. Sci. USA. 1996;93:136–140. doi: 10.1073/pnas.93.1.136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Hassan R., Thomas A., Alewine C., Le D.T., Jaffee E.M., Pastan I. Mesothelin Immunotherapy for Cancer: Ready for Prime Time? J. Clin. Oncol. 2016;34:4171–4179. doi: 10.1200/JCO.2016.68.3672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Quanz M., Hagemann U.B., Zitzmann-Kolbe S., Stelte-Ludwig B., Golfier S., Elbi C., Mumberg D., Ziegelbauer K., Schatz C.A. Anetumab ravtansine inhibits tumor growth and shows additive effect in combination with targeted agents and chemotherapy in mesothelin-expressing human ovarian cancer models. Oncotarget. 2018;9:34103–34121. doi: 10.18632/oncotarget.26135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Gadomska H., Grzechocińska B., Janecki J., Nowicka G., Powolny M., Marianowski L. Serum lipids concentration in women with benign and malignant ovarian tumours. Eur. J. Obstet. Gynecol. Reprod. Biol. 2005;120:87–90. doi: 10.1016/j.ejogrb.2004.02.045. [DOI] [PubMed] [Google Scholar]
- 89.Pal M.K., Rashid M., Bisht M. Multiplexed magnetic nanoparticle-antibody conjugates (MNPs-ABS) based prognostic detection of ovarian cancer biomarkers, CA125, β-2M and ApoA1 using fluorescence spectroscopy with comparison of surface plasmon resonance (SPR) analysis. Biosens. Bioelectron. 2015;73:146–152. doi: 10.1016/j.bios.2015.05.051. [DOI] [PubMed] [Google Scholar]
- 90.Kozak K.R., Su F., Whitelegge J.P., Faull K., Reddy S., Farias-Eisner R. Characterization of serum biomarkers for detection of early stage ovarian cancer. Proteom. 2005;5:4589–4596. doi: 10.1002/pmic.200500093. [DOI] [PubMed] [Google Scholar]
- 91.Kim Y.-W., Bee S.M. Development of multiplexed bead-based immunoassays for the detection of early stage ovarian cancer using a combination of serum biomarkers. PLoS ONE. 2012;7:e44960. doi: 10.1371/journal.pone.0044960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Fassl S., Leisser C., Maier S. Transferrin ensures survival of ovarian carcinoma cells when apoptosis is induced by TNF α, FasL, TRAIL, or Myc. Oncogene. 2003;22:8343–8355. doi: 10.1038/sj.onc.1207047. [DOI] [PubMed] [Google Scholar]
- 93.Ahmed N., Oliva K.T., Barker G., Hoffmann P., Reeve S., Smith I.A., Quinn M.A., Rice G.E. Proteomic tracking of serum protein isoforms as screening biomarkers of ovarian cancer. Proteomics. 2005;5:4625–4636. doi: 10.1002/pmic.200401321. [DOI] [PubMed] [Google Scholar]
- 94.Clarke C.H., Yip C., Badgwell D., Fung E.T., Coombes K.R., Zhang Z., Lu K.H., Bast R.C. Proteomic biomarkers apolipoprotein A1, truncated transthyretin and connective tissue activating protein III enhance the sensitivity of CA125 for detecting early stage epithelial ovarian cancer. Gynecol. Oncol. 2011;122:548–553. doi: 10.1016/j.ygyno.2011.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zheng X., Chen S., Li L., Liu X., Liu X., Dai S., Zhang P., Lu H., Lin Z., Yu Y., et al. Evaluation of HE4 and TTR for diagnosis of ovarian cancer: Comparison with CA125. J. Gynecol. Obstet. Hum. Reprod. 2018;47:227–230. doi: 10.1016/j.jogoh.2018.03.010. [DOI] [PubMed] [Google Scholar]
- 96.Su F., Lang J., Kumar A., Ng C., Hsieh B., Suchard M.A., Reddy S.T., Farias-Eisner R. Validation of Candidate Serum Ovarian Cancer Biomarkers for Early Detection. Biomark. Insights. 2007;2:369–375. doi: 10.1177/117727190700200011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Suzuki S., Moore D.H., Ginzinger D.G., E Godfrey T., Barclay J., Powell B., Pinkel D., Zaloudek C., Lu K., Mills G., et al. An approach to analysis of large-scale correlations between genome changes and clinical endpoints in ovarian cancer. Cancer Res. 2000;60:5382–5385. [PubMed] [Google Scholar]
- 98.Kiechle M., Jacobson A. Comparative genomic hybridization detects genetic imbalances in primary ovarian carcinomas as correlated with grade of differentiation. Cancer. 2001;91:534–540. doi: 10.1002/1097-0142(20010201)91:3<534::AID-CNCR1031>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 99.Schwartz D.R., Kardia S.L.R., A Shedden K., Kuick R., Michailidis G., Taylor J.M.G., Misek D.E., Wu R., Zhai Y., Darrah D.M., et al. Gene expression in ovarian cancer reflects both morphology and biological behavior, distinguishing clear cell from other poor-prognosis ovarian carcinomas. Cancer Res. 2002;62:4722–4729. [PubMed] [Google Scholar]
- 100.Spentzos D., Levine D.A., Ramoni M.F., Joseph M., Gu X., Boyd J., Libermann T.A., Cannistra S.A. Gene Expression Signature With Independent Prognostic Significance in Epithelial Ovarian Cancer. J. Clin. Oncol. 2004;22:4700–4710. doi: 10.1200/JCO.2004.04.070. [DOI] [PubMed] [Google Scholar]
- 101.Garzon R., Fabbri M., Cimmino A., Calin G.A., Croce C.M. MicroRNA expression and function in cancer. Trends Mol. Med. 2006;12:580–587. doi: 10.1016/j.molmed.2006.10.006. [DOI] [PubMed] [Google Scholar]
- 102.Petricoin E.F., Ardekani A.M., Hitt B.A., Levine P.J., Fusaro V.A., Steinberg S.M., Mills G.B., Simone C., Fishman D.A., Kohn E.C., et al. Use of Proteomic Patterns in Serum to Identify Ovarian Cancer. Obstet. Gynecol. Surv. 2002;57:352–353. doi: 10.1097/00006254-200206000-00015. [DOI] [PubMed] [Google Scholar]
- 103.Gortzak-Uzan L., Ignatchenko A., Evangelou A.I., Agochiya M., Brown K.A., St.Onge P., Kireeva I., Schmitt-Ulms G., Brown T.J., Murphy J., et al. A Proteome Resource of Ovarian Cancer Ascites: Integrated Proteomic and Bioinformatic Analyses To Identify Putative Biomarkers. J. Proteome Res. 2008;7:339–351. doi: 10.1021/pr0703223. [DOI] [PubMed] [Google Scholar]
- 104.Zowczak M., Iskra M., Torlinski L., Cofta S. Analysis of Serum Copper and Zinc Concentrations in Cancer Patients. Biol. Trace Element Res. 2001;82:1–8. doi: 10.1385/BTER:82:1-3:001. [DOI] [PubMed] [Google Scholar]
- 105.Wang Y., Sun Z., Li A., Zhang Y. Association between serum zinc levels and lung cancer: A meta-analysis of observational studies. World J. Surg. Oncol. 2019;17:1–8. doi: 10.1186/s12957-019-1617-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Baxter E., Caddick M., Dragovic B. Garnet: A Rock-Forming Mineral Petrochronometer. Rev. Miner. Geochem. 2017;83:469–533. doi: 10.2138/rmg.2017.83.15. [DOI] [Google Scholar]
- 107.Jalalian S.H., Ramezani M., Abnous K., Taghdisi S.M. Exosomes, new biomarkers in early cancer detection. Anal. Biochem. 2019;571:1–13. doi: 10.1016/j.ab.2019.02.013. [DOI] [PubMed] [Google Scholar]
- 108.Li N., Zhan X. Identification of clinical trait–related lncRNA and mRNA biomarkers with weighted gene co-expression network analysis as useful tool for personalized medicine in ovarian cancer. EPMA J. 2019;10:273–290. doi: 10.1007/s13167-019-00175-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Steffensen K.D., Alvero A.B., Yang Y., Waldstrøm M., Hui P., Holmberg J.C., Silasi D.-A., Jakobsen A., Rutherford T., Mor G. Prevalence of Epithelial Ovarian Cancer Stem Cells Correlates with Recurrence in Early-Stage Ovarian Cancer. J. Oncol. 2011;2011:1–12. doi: 10.1155/2011/620523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Townsend D.M., Tew K.D., Tapiero H. The importance of glutathione in human disease. Biomed. Pharmacother. 2003;57:145–155. doi: 10.1016/S0753-3322(03)00043-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Olivares M., Uauy R. Limits of metabolic tolerance to copper and biological basis for present recommendations and regulations. Am. J. Clin. Nutr. 1996;63:846S–852S. doi: 10.1093/ajcn/63.5.846. [DOI] [PubMed] [Google Scholar]
- 112.Toubhans B., Gourlan A., Telouk P., Lutchman-Singh K., Francis L., Conlan R., Margarit L., Gonzalez D., Charlet L. Cu isotope ratios are meaningful in ovarian cancer diagnosis. J. Trace Elements Med. Biol. 2020;62:126611. doi: 10.1016/j.jtemb.2020.126611. [DOI] [PubMed] [Google Scholar]
- 113.Zhang X., Yuan X., Shi H., Wu L., Qian H., Xu W. Exosomes in cancer: Small particle, big player. J. Hematol. Oncol. 2015;8:1–13. doi: 10.1186/s13045-015-0181-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Greening D.W., Gopal S.K., Xu R., Simpson R.J., Chen W. Exosomes and their roles in immune regulation and cancer. Semin. Cell Dev. Biol. 2015;40:72–81. doi: 10.1016/j.semcdb.2015.02.009. [DOI] [PubMed] [Google Scholar]
- 115.Mathivanan S., Ji H., Simpson R.J. Exosomes: Extracellular organelles important in intercellular communication. J. Proteom. 2010;73:1907–1920. doi: 10.1016/j.jprot.2010.06.006. [DOI] [PubMed] [Google Scholar]
- 116.Chan Y.-K., Zhang H., Liu P., Tsao S.-W., Lung M.L., Mak N.-K., Wong R.N.-S., Yue P.Y.-K., Tsao G.S.-W. Proteomic analysis of exosomes from nasopharyngeal carcinoma cell identifies intercellular transfer of angiogenic proteins. Int. J. Cancer. 2015;137:1830–1841. doi: 10.1002/ijc.29562. [DOI] [PubMed] [Google Scholar]
- 117.del Conde I., Shrimpton C.N. Tissue-factor–bearing microvesicles arise from lipid rafts and fuse with activated platelets to initiate coagulation. Blood. 2005;106:1604–1611. doi: 10.1182/blood-2004-03-1095. [DOI] [PubMed] [Google Scholar]
- 118.Théry C. Exosomes: Secreted vesicles and intercellular communications. F1000 Biol. Rep. 2011;3:15. doi: 10.3410/B3-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Azmi A.S., Bao B., Sarkar F.H. Exosomes in cancer development, metastasis, and drug resistance: A comprehensive review. Cancer Metastasis Rev. 2013;32:623–642. doi: 10.1007/s10555-013-9441-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Paggetti J., Haderk F., Seiffert M., Janji B., Distler U., Ammerlaan W., Kim Y.J., Adam J., Lichter P., Solary E., et al. Exosomes released by chronic lymphocytic leukemia cells induce the transition of stromal cells into cancer-associated fibroblasts. Blood. 2015;126:1106–1117. doi: 10.1182/blood-2014-12-618025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Went P.T., Lugli A., Meier S., Bundi M., Mirlacher M., Sauter G., Dirnhofer S. Frequent EpCam protein expression in human carcinomas. Hum. Pathol. 2004;35:122–128. doi: 10.1016/j.humpath.2003.08.026. [DOI] [PubMed] [Google Scholar]
- 122.Golubnitschaja O., Baban B., Boniolo G., Wang W., Bubnov R., Kapalla M., Krapfenbauer K., Mozaffari M.S., Costigliola V. Medicine in the early twenty-first century: Paradigm and anticipation-EPMA position paper 2016. EPMA J. 2016;7:1–13. doi: 10.1186/s13167-016-0072-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Janssens J.P., Schuster K., Voss A. Preventive, predictive, and personalized medicine for effective and affordable cancer care. EPMA J. 2018;9:113–123. doi: 10.1007/s13167-018-0130-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Wang H., Fu Z., Dai C., Cao J., Liu X., Xu J., Lv M., Gu Y., Zhang Y.G.J., Hua X., et al. LncRNAs expression profiling in normal ovary, benign ovarian cyst and malignant epithelial ovarian cancer. Sci. Rep. 2016;6:38983. doi: 10.1038/srep38983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Anaya J. OncoLnc: Linking TCGA survival data to mRNAs, miRNAs, and lncRNAs. PeerJ Comput. Sci. 2016;2:e67. doi: 10.7717/peerj-cs.67. [DOI] [Google Scholar]
- 126.Chang M.-C., Chen C.-A. Mesothelin enhances invasion of ovarian cancer by inducing MMP-7 through MAPK/ERK and JNK pathways. Biochem. J. 2012;442:293–302. doi: 10.1042/BJ20110282. [DOI] [PubMed] [Google Scholar]
- 127.Huang R., Li X., Holm R., Trope C.G., Nesland J.M., Suo Z. The expression of aldehyde dehydrogenase 1 (ALDH1) in ovarian carcinomas and its clinicopathological associations: A retrospective study. BMC Cancer. 2015;15:502. doi: 10.1186/s12885-015-1513-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Dainty L.A., Risinger J.I., Morrison C., Chandramouli G., Bidus M.A., Zahn C., Rose G.S., Fowler J., Berchuck A., Maxwell G.L. Overexpression of folate binding protein and mesothelin are associated with uterine serous carcinoma. Gynecol. Oncol. 2007;105:563–570. doi: 10.1016/j.ygyno.2006.10.063. [DOI] [PubMed] [Google Scholar]
- 129.Kelemen L.E. The role of folate receptor α in cancer development, progression and treatment: Cause, consequence or innocent bystander? Int. J. Cancer. 2006;119:243–250. doi: 10.1002/ijc.21712. [DOI] [PubMed] [Google Scholar]
- 130.Rahilly M., Carder P.J., Al Nafussi A., Harrison D.J. Distribution of glutathione S-transferase isoenzymes in human ovary. Reproduction. 1991;93:303–311. doi: 10.1530/jrf.0.0930303. [DOI] [PubMed] [Google Scholar]
- 131.Coughlin S.S., Hall I.J. Glutathione S-transferase polymorphisms and risk of ovarian cancer: A HuGE review. Genet. Med. 2002;4:250–257. doi: 10.1097/00125817-200207000-00003. [DOI] [PubMed] [Google Scholar]
- 132.Beeghly A., Katsaros D., Chen H., Fracchioli S., Zhang Y., Massobrio M., Risch H., Jones B., Yu H. Glutathione S-transferase polymorphisms and ovarian cancer treatment and survival. Gynecol. Oncol. 2006;100:330–337. doi: 10.1016/j.ygyno.2005.08.035. [DOI] [PubMed] [Google Scholar]
- 133.Zhang T., Xu J., Deng S., Zhou F., Li J. Core signaling pathways in ovarian cancer stem cell revealed by integrative analysis of multi-marker genomics data. PLoS ONE. 2018;13:e0196351. doi: 10.1371/journal.pone.0196351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Häusler S.F.M., Keller A., A Chandran P., Ziegler K., Zipp K., Heuer S., Krockenberger M., Engel J.B., Hönig A., Scheffler M., et al. Whole blood-derived miRNA profiles as potential new tools for ovarian cancer screening. Br. J. Cancer. 2010;103:693–700. doi: 10.1038/sj.bjc.6605833. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No new data were created or analysed in this study. Data sharing is not applicable to this article.