Abstract
Chronic pancreatitis (CP) remains a diagnostic challenge as clinical symptoms are non-specific, histopathological appearances are varied and pathogenesis remains incompletely understood. Multiple classifications and grading systems have been proposed for CP, but none leverage the full capabilities of cross-sectional imaging modalities and are not widely accepted or validated. CT and MRI/MRCP are useful in identifying a wide spectrum of histopathological changes in CP and can also assess exocrine reserve of pancreas. Advanced MRI techniques such as T1 mapping and extracellular volume fraction can potentially identify early CP. Cross-sectional imaging-based severity scoring can quantify CP disease burden and may have positive implications for clinicians and researchers. In this review, we discuss the need for cross-sectional imaging-based severity scoring for CP, role of CT, and MRI/MRCP in assessment of CP and how these modalities can be used to obtain severity scoring for CP. We summarize relevant information from recently published CT and MRI/MRCP reporting standards for CP, and from international guidelines for cross-sectional imaging and severity scoring for CP.
Keywords: Chronic pancreatitis, Multidetector computed tomography, Magnetic resonance imaging, Classification
Introduction
Chronic pancreatitis (CP) is generally considered as a chronic fibroinflammatory process resulting in irreversible damage to pancreas [1]. Diagnosing CP is a challenge due to the vague and non-specific clinical symptoms, especially true of early CP [2]. The 1963 Marseille conference mentioned that CP meant “many things to many people” while Steer et al. called it an “enigmatic process of uncertain pathogenesis, unpredictable clinical course, and unclear treatment” [3, 4]. Advanced CP is characterized by various combinations of calcifications, atrophy, pancreatic duct dilation or distortion, exocrine and endocrine insufficiency, and pain syndromes, and can often be diagnosed confidently unlike early CP [5]. However, several aspects of CP including its pathogenesis, definition and diagnosis of early CP, management of CP as well as prognosis remain incompletely understood to this day [6]. Hence, it is not surprising that several classifications have been proposed for CP, and that most are not widely adopted or validated [3, 7-12]. Most of the classifications are based on clinical features and laboratory tests with varying weighting for imaging findings. Cambridge classification (CC), based on Endoscopic retrograde cholangiopancreatography (ERCP), and Rosemont classification, based on endoscopic ultrasound (EUS) are the only major classification systems that are predominantly based on imaging [7, 12]. Given its invasive nature and risk of complications, ERCP is rarely used for diagnosis of CP [13] as this role has been given to non-invasive MRCP. EUS is also not used as a first-line imaging modality to diagnose CP and is operator dependent [14]. CC for severity based on ultrasound and CT has several shortcomings and has neither been validated nor is used widely in clinical practice [11]. Similarly, M-ANNHEIM classification, which uses a slightly modified version CC for ultrasound and CT with an adaptation for MRI/MRCP, is also not widely used in clinical practice [10]. Despite advancements in imaging, no validated cross-sectional imaging-based classification or severity scoring system for CP has been developed so far [15]. Purpose of this review is to discuss the necessity for cross-sectional imaging-based scoring system for CP and to explore the ways and means to achieve this goal. We summarize relevant information from the recently published CT and MRI/MRCP reporting standards from the Consortium for the study of chronic pancreatitis, diabetes, and pancreatic cancer (CPDPC), which were based on a pilot study performed at the University of Pittsburgh, as well as from the international guidelines for cross-sectional imaging and severity scoring of CP [11, 15].
Why do we need new cross-sectional imaging-based severity scoring for CP?
Currently available classifications systems based on cross-sectional imaging features of CP such as CC and M-ANNHEIM classification have several shortcomings. For example, gland heterogeneity and irregular pancreatic head/body contour are poorly defined terms and prone to significant subjective variation in assessment. Gland enlargement is considered a feature while atrophy, an important marker for CP, is not included in the classification. Intraductal calculi are appropriately considered in the classification but parenchymal calcifications are not. Thanks to tremendous advances in imaging techniques over the last few decades, several features of CP can now be assessed by cross-sectional imaging. Specifically, recent developments in advanced MRI techniques such as T1 mapping, ECV, and diffusion-weighted imaging (DWI) have shown value, including in detecting early CP [16-18]. CT and MRI/MRCP are ubiquitous and have become first line of investigations to diagnose CP and to assess for alternative diagnoses. A newer severity scoring system specifically designed to incorporate the capabilities of CT and MRI is the need of the hour. Such a severity scoring system for CP can help in clinical management, prognosticate outcomes, and have utility for continued research in CP. The severity scoring can be used to objectively quantify the disease burden which in turn helps to facilitate communication between radiologists and with clinicians in a standardized fashion, and to assess longitudinal changes of CP to predict prognosis. The potential implications for research would be objective assessment of natural progression of disease, correlation with clinical markers, and studying effects of therapeutic interventions and lifestyle modifications on course of CP. The need for such a severity scoring system has recently been espoused by the international guidelines for cross-sectional imaging and severity scoring of CP [11] and is a focus of research studies such as The MINIMAP study (Magnetic Resonance Imaging as a Non-Invasive Method for the Assessment of Pancreatic Fibrosis) [19].
Morphological changes in CP
Advanced CP is characterized by a set of parenchymal and ductal changes which are intertwined and affect each other. The pathological changes, all or any of which may be seen in an individual patient, include chronic inflammation, progressive intralobular and interlobular fibrosis, parenchymal calcifications, acinar loss, glandular atrophy, duct wall thickening and periductal fibrosis leading to duct distortion, intraductal protein plugs/calculi, and ductal strictures [1, 20] (Fig. 1). Interlobular and periductal fibrosis can lead to ductal distortion, strictures, and altered ductal flow resulting in propensity for intraductal protein plugs and calculi (Fig. 2). On the other hand, ductal strictures/obstruction can lead to acinar atrophy and intralobular fibrosis [20].
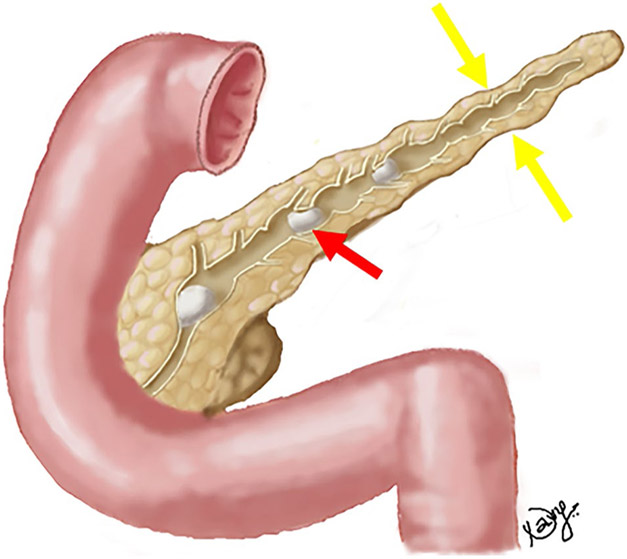
Fig. 1.
Drawing illustrating findings in chronic calcific pancreatitis: Obstructing intraductal calculus (red arrow) with irregular upstream pancreatic ductal dilation and surrounding parenchymal atrophy (yellow arrows)
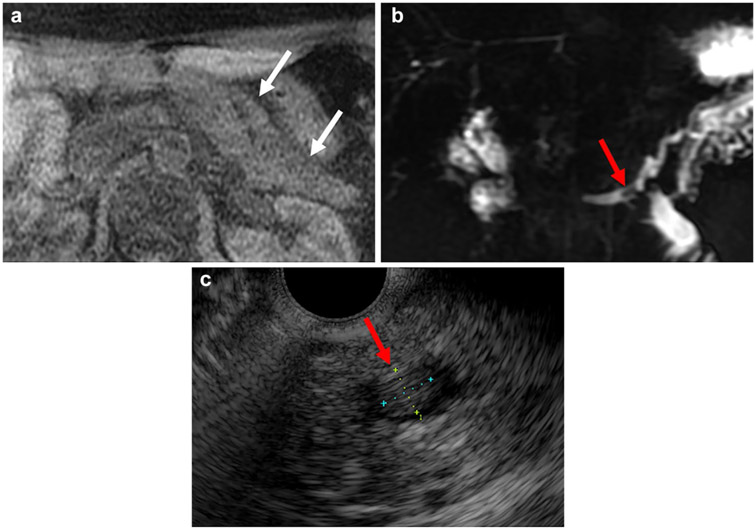
Fig. 2.
A 54-year-old woman with chronic pancreatitis of unclear etiology in the setting of prior Whipple’s procedure for IPMN in the pancreatic head with high-grade dysplasia. Unenhanced axial fat-suppressed T1-weighted sequence (a) shows low signal intensity throughout the residual pancreas (white arrows). 2D thick-slab MRCP (b) shows dilated main pancreatic duct with an intraluminal filling defect (red arrow) that corresponded to a mobile, non-shadowing isoechoic intraductal focus on endoscopic ultrasound exam (c) that on fine needle aspiration cytology demonstrated predominantly acellular debris, suggesting a protein plug
Pathogenesis of CP is yet incompletely understood but is a topic of ongoing research. Several different mechanisms have been proposed to explain the pathophysiology of CP. Activation of pancreatic stellate cells, macrophages, and cytokines have been implicated in the pathogenesis of CP eventually leading to acinar cell necrosis, inflammation, and fibrosis [21].
Role of cross-sectional imaging modalities in assessment of CP
CT scan and MRI have seen numerous technological breakthroughs in the last 2 decades which have resulted in faster scans, greater resolution, lesser radiation to the patient, and development of newer techniques and sequences [22, 23]. CT and MRI are now among the first line of investigations in diagnosing CP and its mimics, identifying its etiologies and complications, and to monitor the progression. Both modalities are also excellent for diagnosing acute pancreatitis (AP) and its complications, which can lead to CP or occur as a complication of CP. However, AP is not the focus of this review and will not be discussed further. Similarly, autoimmune pancreatitis, a distinct variant of CP with unique histopathological and imaging features and vastly different management and prognosis compared to other forms of CP, is also not discussed in the review.
Role of CT
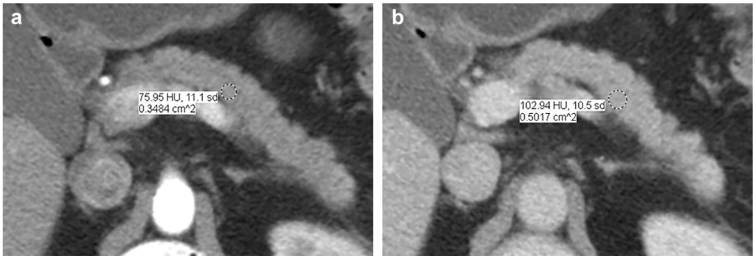
Assessment of CP is best performed using dedicated pancreatic protocol with thin slices (2.5 mm or thinner) obtained in unenhanced, pancreatic parenchymal (late arterial phase), and portal venous phases. CT scan is the most sensitive modality for detecting parenchymal and ductal calcifications, a cardinal sign of CP that was previously shown to be an independent predictor of pancreatic fibrosis (Fig. 3) [24]. Thanks to the high-resolution, CT scan can also help in distinguishing between parenchymal punctate calcifications (≤ 3 mm), which can sometimes be seen in elderly patients without CP, from macro calcifications (> 3 mm) which are virtually diagnostic of CP [25]. CT scan is also excellent in assessing pancreatic atrophy. This can be best done by calculating the volume of pancreas or alternatively, using pancreatic thickness as a surrogate for volume (Fig. 4). Calculating the volume is more accurate but may be impractical for adoption into daily use, particularly in a high-volume practice. CT scan can accurately identify ductal dilation and strictures but is less reliable in identifying ductal contour irregularity unless severe. When adapted from MRI research literature, CT scan can also potentially identify parenchymal fibrosis by demonstrating delayed peak enhancement of parenchyma in portal venous phase rather than in pancreatic parenchymal phase (Fig. 5) but this needs validation [26, 27]. CT texture analysis and DECT imaging utility in CP may potentially play a role in future but have not been validated yet.
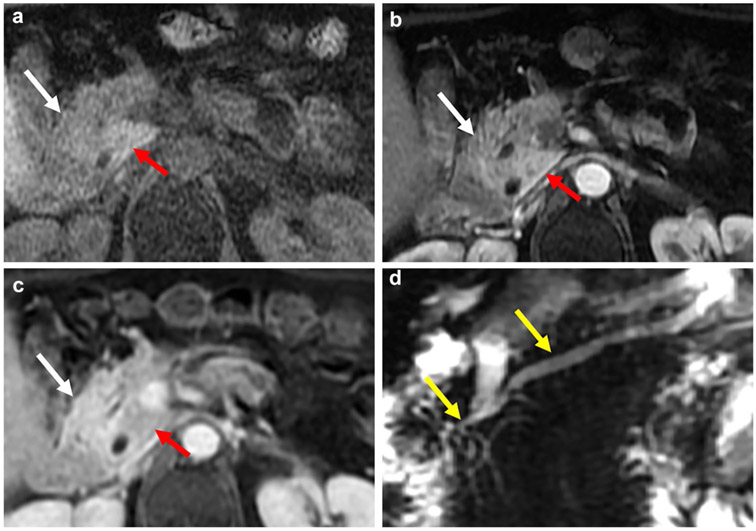
Fig. 3.
A 75-year-old woman with idiopathic chronic pancreatitis. Axial contrast-enhanced CT (a) demonstrates a large obstructing intraductal calculus in the pancreatic head causing marked upstream dilation of the main pancreatic duct (red arrows). Note the severely atrophic and barely perceptible surrounding parenchyma on the coronal reconstruction (b)
Fig. 4.
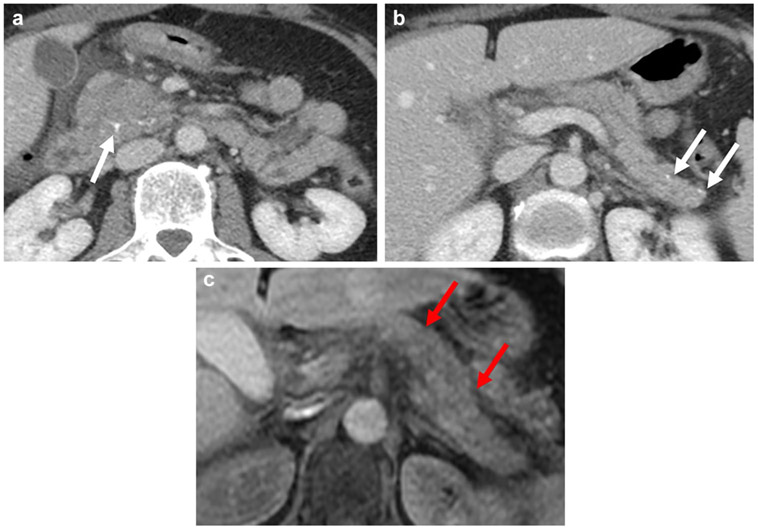
A 67-year-old man with abdominal pain. Axial contrast-enhanced CT scan (a) and axial unenhanced fat-suppressed T1-weighted sequence (b) demonstrate markedly atrophic pancreas (white arrows) as the sole manifestation of CP. Note the lack of ductal dilation and pancreatic calcifications as well as preserved parenchymal T1 signal intensity
Fig. 5.
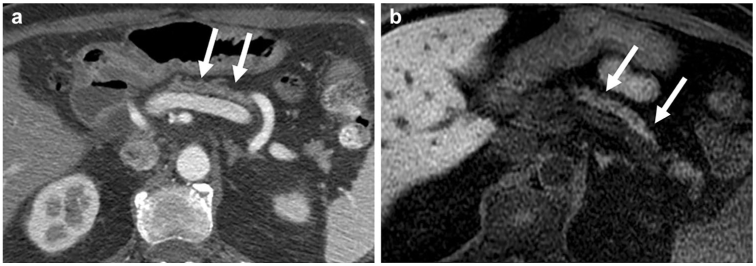
A 40-year-old man with Crohn’s disease and prior acute pancreatitis. Axial multiphasic CT scan obtained in pancreatic parenchymal (late arterial) phase (a) and portal venous phase (b) demonstrates delayed peak enhancement in the portal phase suggesting underlying pancreatic fibrosis
Role of MRI/MRCP
MRI is equivalent to CT in the assessment of pancreatic volume, thickness, and loss of lobularity [28, 29]. MRI can also identify pancreatic fibrosis on the basis of delay in the peak pancreatic enhancement which normally is seen in the pancreatic parenchymal phase (late arterial phase) but with CP, can be seen in portal venous or delayed venous phases [26, 27] (Fig. 6). Real strengths of MRI are, however, in evaluation of ductal and additional parenchymal changes and assessment of pancreatic exocrine reserve [30]. MRI, in conjunction with MRCP, is superior to CT in identifying ductal dilation, strictures, contour irregularity, presence of abnormal branch ducts, and anatomical variants. Secretin-MRCP not only allows much better assessment of the aforementioned ductal features (Fig. 7) but also helps in identifying ductal compliance (ability of the duct to distend in response to intravenous administration of Secretin), which is a surrogate for periductal fibrosis [31]. A unique advantage of MR is the ability to non-invasively assess pancreatic exocrine reserve using Secretin-enhanced MRCP. Secretin stimulates pancreatic acini to secrete bicarbonate-rich fluid which can be seen on MRCP as progressive accumulation of fluid into duodenum. The amount of the secreted fluid is believed to be proportionate to the pancreatic exocrine reserve [31] (Fig. 8). This can either be graded based on presence and extension of fluid from proximal to distal duodenum and proximal jejunum or by more objective quantification by calculating the volume of secreted fluid [32]. Care must be taken not to confuse with Secretin-induced jejunal secretion of fluid [33].
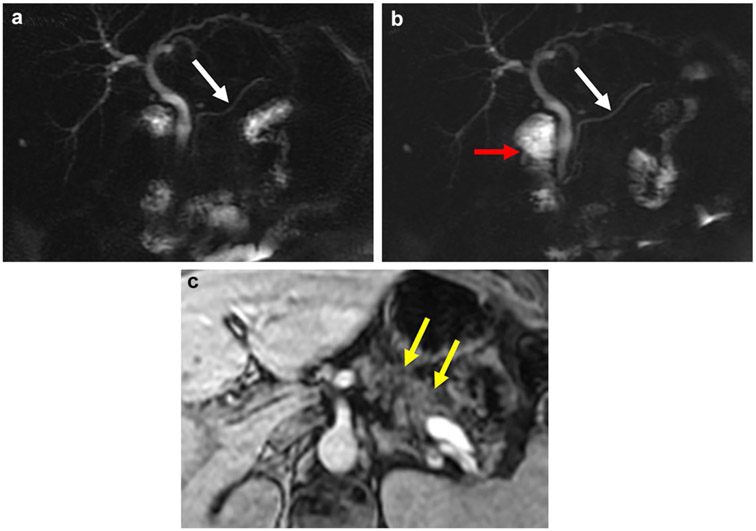
Fig. 6.
A 53-year-old woman with chronic pancreatitis involving the dorsal anlage in the setting of pancreas divisum and recurrent acute pancreatitis. The dorsal anlage (white arrows) is low in signal on pre-contrast fat-suppressed T1-weighted sequence (a), and demonstrates peak enhancement in the delayed venous phase (c) unlike the normal ventral anlage (red arrows) that shows normal T1 signal intensity and expected peak enhancement in the pancreatic parenchymal phase (late arterial phase) (b). 2D thick-slab MRCP (d) demonstrates dilated main duct (yellow arrows) and abnormal branch ducts
Fig. 7.
A 20-year-old man with recurrent acute pancreatitis secondary to elevated triglyceride levels. Axial unenhanced fat-suppressed T1-weighted sequence (a) shows normal pancreatic signal intensity and thickness (arrow heads). The main pancreatic duct is also non-dilated (white arrow) and appears unremarkable on 2D thick-slab MRCP (b) but Secretin-MRCP (c) shows a mildly irregular main duct (white arrow) with multiple faintly visualized abnormal branch ducts (red arrows) suggesting early CP
Fig. 8.
A 63-year-old woman with cystic fibrosis mutation (CFTR) with recurrent acute pancreatitis and CP. Pre- and post-secretin images show normal main pancreatic duct (white arrow) with diminished exocrine reserve with only minimal fluid accumulating in the proximal duodenum (red arrow). Note the low signal intensity of pancreas on axial fat-suppressed gradient-echo image (yellow arrows) compatible with CP
Novel quantitative MRI-based markers
High aqueous protein content in the pancreatic acini, which contributes to high signal on fat-suppressed T1-weighted gradient-echo sequences compared to other upper abdominal organs, is diminished in CP resulting in decreased pancreatic T1 signal [34-36]. Tissue T1 signal is markedly affected by the type of T1 sequence and scan acquisition parameters. Hence, assessment of pancreatic T1 signal was traditionally performed using an internal reference, such as liver, spleen, or muscle (Fig. 9). More recently, this has been achieved by calculation of absolute T1 relaxation time [18, 37]. T1 mapping (calculation of T1 relaxation time) is now feasible using rapid volumetric acquisitions. Median T1 of pancreas on 1.5T is 654 ms and on 3T is 717 ms [38]. Recent studies showed T1 relaxation time was significantly different between normal, mild CP, and moderate/severe CP [16, 18]. MRI-based assessment of extracellular volume (ECV) fraction is a relaxometry method for quantification of tissue fibrosis (based on enhancement of pancreas on delayed postcontrast images which is mainly attributed to fibrosis). In a study of 143 patients, Trikes et al. proposed ECV as a potential marker for diagnosis and staging of CP [16]. In that study, ECV of > 0.27 showed 91% sensitivity and 77% specificity for the diagnosis of CP (AUC = 0.90). Fibrosis and changes of CP also result in reduction of diffusive motion of water molecules in pancreas. Prior studies showed lower apparent diffusion coefficient (ADC) values in patients with CP compared to normal pancreas [17, 39]. MR spectroscopy may play a potential role in evaluation of CP but has not been adequately explored [40].
Fig. 9.
A 51-year-old woman with prior history of smoking, recurrent acute pancreatitis, and chronic calcific pancreatitis of unclear etiology. Axial contrast-enhanced CT images (a, b) demonstrate small calcifications in the pancreatic head and tail (white arrows). Axial unenhanced fat-suppressed T1-weighted sequence (c) shows low signal intensity diffusely throughout the pancreas (red arrows) consistent with CP
CT severity scoring of features of CP
Based on the histopathological changes of CP and abilities of CT, the following features can be graded/scored by CT:
Atrophy
Pancreatic thickness or volume can be graded as normal or mild, moderate, or severe atrophy and a score can be assigned to each—such as 0 through 3. However, given the wide range of normal volume/thickness in the general population and expected age-related changes, this can be a challenging task [41, 42]. The severity scoring system should be cautiously applied in older patients, particularly those above 70 years.
Calcifications
Pancreatic calcification burden can be graded/scored by taking into account the number of punctate and macro calcifications. As punctate calcifications can be senescent, macro calcifications can be given more weightage [25].
Parenchymal fibrosis
Based on parenchymal enhancement pattern, as a ratio of increased density from unenhanced phase to pancreatic and portal phases, fibrosis can be graded as not present, equivocal, and present. Grading into mild moderate and severe may not be feasible given the lack of sensitivity to detect fibrosis.
Ductal stricture or intraductal obstructing calculus
Strictures in head can result in upstream dilation and associated parenchymal atrophy affecting a larger portion of pancreas than a stricture in tail. Hence, strictures in head should be scored higher. For example, scores 1, 2, and 3 for strictures in tail, body, and head, respectively, and 0 for none. If multiple, the dominant site of obstruction closest to the ampulla should be scored.
Ductal dilation
Ductal dilation can be graded as normal, mild, moderate, or severe. However, like atrophy, grading ducts dilation is also challenging given lack of widely accepted value for normal and in view of expected age-related changes [43]. 3 or 3.5 mm has been considered as the threshold by some authors in the past [1, 44]. Irrespective of the diameter (i.e., even if less than 3 mm), increase in caliber upstream to a stricture or intraductal calculus must be considered dilated.
Ductal contour irregularity
Though a subjective parameter and inadequately evaluated by CT unless severe, this feature cannot be ignored as it implies the presence of periductal fibrosis, an important sign of CP. Contour irregularity can be graded as normal, mild/possible, and “moderate to severe”/definite.
MRI/MRCP severity scoring of features of CP
Grading of features of CP with MRI is similar to CT for atrophy, ductal dilation, and ductal obstruction (strictures/intraductal obstructing calculi). Parenchymal calcifications cannot be assessed by MRI.
Ductal contour irregularity
Given subjective nature of this parameter, grading of main duct contour can be normal, mild/possible, and “moderate to severe’/definite. Due to better visualization of branch ducts with MRCP, presence of any abnormal branch ducts should also be included in this category as these changes are part of the spectrum of same pathology [7]. Essentially, ERCP-based CC for severity grading of CP can adapted for MRCP. ERCP has superior resolution and can better delineate the abnormal branch ducts by distending them with contrast but is invasive and associated with risk of complications [45]. MRCP, on the contrary, is non-invasive and provides a more accurate estimation of PD caliber in an undistended physiological state [46]. Similar to contour irregularity, loss of ductal compliance on Secretin-MRCP also implies periductal fibrosis and should be included in this category [31].
Parenchymal fibrosis
This can be graded based on comparison of pancreatic T1 signal intensity to other internal structures such as liver, spleen, and paraspinal muscles. Alternatively, though still experimental, more objective grading can potentially be performed using newer techniques such as T1 mapping, ECV fraction, and DWI.
Exocrine reserve
Based on the extent of fluid accumulation in duodenum and proximal jejunum with Secretin-MRCP, this can be graded as normal, mildly diminished, moderately diminished, or severely diminished [47].
Additional considerations and tips for an effective cross-sectional imaging-based severity scoring of CP
Attempt should be made to keep the scoring system simple and avoid cluttering with too many variables. Simplicity and ease of use are key to acceptance by the clinical and research community. Scoring should be assigned to imaging surrogates of major histopathologic changes of CP and not to the spectrum of imaging findings of CP. For example, fibrosis can be assessed based on T1-weighted signal intensity ratio, enhancement pattern, and DWI. Rather than assigning separate scoring for each of these variables, which can inflate the table and overall score, it would be best to condense the findings into the single category of fibrosis. Minor features that have questionable or weak association with CP should be avoided. For example, grading of pancreatic lobularity has not shown to be a strong predictor of CP and is best to be avoided [42]. The scoring system should ideally include parameters that can be easily assessed without special skills/software/equipment, etc. For example, atrophy assessment by volume calculation in the current scenario is time consuming and requires dedicated software application. A representative measurement or mean of measurements that can be obtained on an axial plane that can act as surrogate for volume without significant loss of accuracy are simpler and more likely to be accepted.
It may be prudent to give weighted scores to features that either have strong diagnostic correlation with CP or may have strong implications for disease burden or prognosis. For example, focal changes of CP in pancreatic tail may result in minimal or no functional pancreatic deficiency but a global involvement by severe atrophy may correlate with profound exocrine and endocrine deficiencies, and hence can be given weighted scores such as 0, 2, and 4 unlike scoring for other features that may go from 0 to 3.
The cross-sectional imaging-based scoring system for CP should be developed in such a way that it allows for seamless future revisions to incorporate both technological advances and advances in clinicopathological research. This can be best achieved by focusing on creating a scoring system anchored on basic tenets of pathogenesis of CP. Finally, the scoring system should be validated prospectively, on large cohort of subjects with clinically well-phenotyped patients with early or suspected CP, advanced CP as well as controls. Interobserver and intraobserver variability of blinded radiologists’ assessment should also be evaluated.
Proposal for CT and MRI/MRCP severity scoring of CP
We propose a framework for CT scan (Table 1) and MRI/MRCP (Table 2) severity scoring system for features of CP based on the recently published international guidelines for cross-sectional imaging and severity scoring of CP as well as the CT and MRI/MRCP reporting standards from the CPDPC [11, 15]. The scoring system, as discussed earlier, takes into account the pathogenesis and major histopathological changes of CP. The aggregate score based on CT and MRI is similar but not identical. An additional scoring is obtained for exocrine reserve of pancreas assessed with Secretin, that can be labeled as S-0 (normal), S-1 (mildly diminished exocrine reserve), S-2 (moderately diminished exocrine reserve), and S-3 (severely diminished or no exocrine reserve). An imaginary aggregate CT score in a patient with advanced CP could be 16 out of a maximum score of 20, while a similar aggregate MRI/MRCP score could be 18 out of maximum score of 20, or if performed in conjunction with Secretin-MRCP, it could be 18 S-3.
Table 1.
CT severity scoring for CP. This table has been adapted from Tirkes et al. [15] with permission from Radiology
| Feature | Score |
|||
|---|---|---|---|---|
| Low (0) | High (up to 3 or 4) | |||
| Pancreatic duct caliber | Normal | Mildly dilated | Moderately dilated | Markedly dilated |
| Pancreatic duct contour | Smooth | Markedly Irregular | ||
| Pancreatic duct strictures/intraductal obstructing filling defect a | None | In the tail | In the body | In the head |
| Pancreatic parenchymal calcifications (pancreatic fibrosis, based on parenchymal enhancement pattern—optional)b | None | Few large/punctate calcifications | Multiple large calcifications | |
| Pancreatic parenchymal atrophy | None | Mild | Moderate | Severe |
| Distribution of findings (atrophy or parenchymal changes) | Focal | Segmental | Diffuse | |
Assign score only for the dominant site of obstruction closest to ampulla
Pancreatic parenchymal fibrosis scoring by CT is optional given lack of validation, but could potentially be included in future
Table 2.
MRI-MRCP severity scoring for CP. This table has been adapted from Tirkes et al. [15] with from Radiology
| Feature | Score |
||||
|---|---|---|---|---|---|
| Low (0) | High (up to 3 or 4) | ||||
| Pancreatic duct caliber | Normal | Mildly dilated | Moderately dilated | Markedly dilated | |
| Pancreatic duct contour | Smooth | Abnormal branch ducts | Markedly Irregular | ||
| Pancreatic duct strictures/intraductal obstructing filling defect a | None | In the tail | In the body | In the head | |
| Pancreatic parenchymal fibrosis (based on relative T1 Signal, T1 mapping, DWI, or parenchymal enhancement pattern)b | None | Minimal/equivocal | Severe | ||
| Pancreatic parenchymal atrophy | None | Mild | Moderate | Severe | |
| Distribution of findings (atrophy or parenchymal changes) | Focal | Segmental | Diffuse | ||
| Exocrine response on secretin-MRCP | Normal | Mildly diminished | Severely diminished/No response | ||
Assign score only for the dominant site of obstruction closest to ampulla
It is preferable to have a single score for pancreatic fibrosis based on overall assessment of relative T1 signal, T1 mapping, DWI, and parenchymal enhancement pattern, rather than inflate the aggregate by assigning individual scores to the different methods by which pancreatic fibrosis is assessed
Future directions
As progressive research elucidates components of early CP, the same should be incorporated into the scoring system. Perhaps this can be done by focusing on functional imaging aimed at identifying the biomarkers of pancreatic inflammation preceding the onset of fibrosis when the disease process can potentially be stalled or reversed. Positron Emission Tomography (PET) imaging and single-photon emission computerized tomography (SPECT) imaging with novel tracers such as 125 I-iodo-DPA-713 are some such examples [48, 49]. Role of texture analysis, deep learning, and artificial intelligence in medical imaging is exponentially growing and should be leveraged in future scoring systems [50].
Conclusion
CP continues to be a disease of uncertain pathogenesis and unpredictable prognosis. Ongoing technological advancements in CT and MRI have enabled better assessment of CP. Cross-sectional imaging-based scoring system for CP will help in quantifying the disease burden, improve communication, and can in turn aid clinicians and researchers. An ideal scoring system should be simple, easy to use, and based on basic histopathological and functional hallmarks of CP.
Acknowledgements
The authors would like to thank Dr. Dhiraj Yadav, Professor of Medicine, Division of Gastroenterology, Hepatology & Nutrition, University of Pittsburgh School of Medicine, for reviewing the manuscript.
Footnotes
Compliance with ethical standards
Conflict of interest The authors declare that they have no conflict of interest.
Publisher’s Note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Conwell DL, Lee LS, Yadav D, Longnecker DS, Miller FH, Mortele KJ, et al. American Pancreatic Association Practice Guidelines in Chronic Pancreatitis: evidence-based report on diagnostic guidelines. Pancreas. 2014;43(8):1143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Duggan SN, Chonchubhair HMN, Lawal O, O’Connor DB, Conlon KCJWjog. Chronic pancreatitis: A diagnostic dilemma. 2016;22(7):2304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Perrier C Symposium of the etiology and pathological anatomi of chronic pancreatics. Marseilles, 1963. Am J Dig Dis. 1964;9:371–6 [Google Scholar]
- 4.Steer ML, Waxman I, Freedman S. Chronic pancreatitis. New England Journal of Medicine. 1995;332(22):1482–90 [DOI] [PubMed] [Google Scholar]
- 5.Whitcomb DC. Peering Into the “black box” of the complex chronic pancreatitis syndrome. Pancreas. 2016;45(10):1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Whitcomb DC, Shimosegawa T, Chari ST, Forsmark CE, Frulloni L, Garg P, et al. International consensus statements on early chronic Pancreatitis. Recommendations from the working group for the international consensus guidelines for chronic pancreatitis in collaboration with The International Association of Pancreatology, American Pancreatic Association, Japan Pancreas Society, PancreasFest Working Group and European Pancreatic Club. Pancreatology. 2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sarner M, Cotton P. Classification of pancreatitis. Gut. 1984;25(7):756–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shimosegawa T, Kataoka K, Kamisawa T, Miyakawa H, Ohara H, Ito T, et al. The revised Japanese clinical diagnostic criteria for chronic pancreatitis. Journal of gastroenterology. 2010;45(6):584–91 [DOI] [PubMed] [Google Scholar]
- 9.Ammann RW. A clinically based classification system for alcoholic chronic pancreatitis: summary of an international workshop on chronic pancreatitis. Pancreas. 1997;14(3):215–21 [DOI] [PubMed] [Google Scholar]
- 10.Schneider A, Löhr JM, Singer MV. The M-ANNHEIM classification of chronic pancreatitis: introduction of a unifying classification system based on a review of previous classifications of the disease. Journal of Gastroenterology. 2007;42(2):101–19 [DOI] [PubMed] [Google Scholar]
- 11.Frøkjær JB, Akisik F, Farooq A, Akpinar B, Dasyam A, Drewes AM, et al. Guidelines for the Diagnostic Cross Sectional Imaging and Severity Scoring of Chronic Pancreatitis. Pancreatology. 2018;18(7):764–73 [DOI] [PubMed] [Google Scholar]
- 12.Catalano MF, Sahai A, Levy M, Romagnuolo J, Wiersema M, Brugge W, et al. EUS-based criteria for the diagnosis of chronic pancreatitis: the Rosemont classification. Gastrointestinal endoscopy. 2009;69(7):1251–61 [DOI] [PubMed] [Google Scholar]
- 13.Andriulli A, Loperfido S, Napolitano G, Niro G, Valvano MR, Spirito F, et al. Incidence rates of post-ERCP complications: a systematic survey of prospective studies. The American journal of gastroenterology. 2007;102(8):1781. [DOI] [PubMed] [Google Scholar]
- 14.Wallace MB, Hawes RH, Durkalski V, Chak A, Mallery S, Catalano MF, et al. The reliability of EUS for the diagnosis of chronic pancreatitis: interobserver agreement among experienced endosonographers. Gastrointestinal endoscopy. 2001;53(3):294–9 [DOI] [PubMed] [Google Scholar]
- 15.Tirkes T, Shah ZK, Takahashi N, Grajo JR, Chang ST, Venkatesh SK, et al. Reporting Standards for Chronic Pancreatitis by Using CT, MRI, and MR Cholangiopancreatography: The Consortium for the Study of Chronic Pancreatitis, Diabetes, and Pancreatic Cancer. Radiology. 2019;290(1):207–15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tirkes T, Lin C, Cui E, Deng Y, Territo PR, Sandrasegaran K, et al. Quantitative MR evaluation of chronic pancreatitis: extracellular volume fraction and MR relaxometry. American Journal of Roentgenology. 2018;210(3):533–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bieliuniene E, Frøkjær JB, Pockevicius A, Kemesiene J, Lukosevicius S, Basevicius A, et al. Magnetic Resonance Imaging as a Valid Noninvasive Tool for the Assessment of Pancreatic Fibrosis. Pancreas. 2019;48(1):85–93 [DOI] [PubMed] [Google Scholar]
- 18.Tirkes T, Lin C, Fogel EL, Sherman SS, Wang Q, Sandrasegaran K. T1 mapping for diagnosis of mild chronic pancreatitis. J Magn Reson Imaging. 2017;45(4): 1171–6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tirkes T, Yadav D, Conwell DL, Territo PR, Zhao X, Venkatesh SK, et al. Magnetic resonance imaging as a non-invasive method for the assessment of pancreatic fibrosis (MINIMAP): a comprehensive study design from the consortium for the study of chronic pancreatitis, diabetes, and pancreatic cancer. Abdom Radiol (NY). 2019;44(8):2809–21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kloppel G, Detlefsen S, Feyerabend B. Fibrosis of the pancreas: the initial tissue damage and the resulting pattern. Virchows Archiv. 2004;445(1):1–8 [DOI] [PubMed] [Google Scholar]
- 21.Kleeff J, Whitcomb DC, Shimosegawa T, Esposito I, Lerch MM, Gress T, et al. Chronic pancreatitis. Nature Reviews Disease Primers. 2017;3:nrdp201760. [DOI] [PubMed] [Google Scholar]
- 22.Ulzheimer S, Bongers M, Flohr T. Multi-slice CT: Current Technology and Future Developments. 2018. [Google Scholar]
- 23.Siddiqui N, Vendrami CL, Chatterjee A, Miller FH. Advanced MR imaging techniques for pancreas imaging. Magnetic Resonance Imaging Clinics. 2018;26(3):323–44 [DOI] [PubMed] [Google Scholar]
- 24.Sinha A, Singh VK, Cruise M, Afghani E, Matsukuma K, Ali S, et al. Abdominal CT predictors of fibrosis in patients with chronic pancreatitis undergoing surgery. European radiology. 2015;25(5):1339–46 [DOI] [PubMed] [Google Scholar]
- 25.Lesniak RJ, Hohenwalter MD, Taylor AJ. Spectrum of causes of pancreatic calcifications. American Journal of Roentgenology. 2002;178(1):79–86 [DOI] [PubMed] [Google Scholar]
- 26.Zhang XM, Shi H, Parker L, Dohke M, Holland GA, Mitchell DG. Suspected early or mild chronic pancreatitis: enhancement patterns on gadolinium chelate dynamic MRI. Journal of Magnetic Resonance Imaging: An Official Journal of the International Society for Magnetic Resonance in Medicine. 2003;17(1):86–94 [DOI] [PubMed] [Google Scholar]
- 27.Balci NC, Alkaade S, Magas L, Momtahen AJ, Burton FR. Suspected chronic pancreatitis with normal MRCP: findings on MRI in correlation with secretin MRCP. Journal of Magnetic Resonance Imaging: An Official Journal of the International Society for Magnetic Resonance in Medicine. 2008;27(1):125–31 [DOI] [PubMed] [Google Scholar]
- 28.Szczepaniak EW, Malliaras K, Nelson MD, Szczepaniak LS. Measurement of pancreatic volume by abdominal MRI: a validation study. PloS one. 2013;8(2):e55991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Burute N, Nisenbaum R, Jenkins DJ, Mirrahimi A, Anthwal S, Colak E, et al. Pancreas volume measurement in patients with Type 2 diabetes using magnetic resonance imaging-based planimetry. Pancreatology. 2014;14(4):268–74 [DOI] [PubMed] [Google Scholar]
- 30.Sandrasegaran K, Lin C, Akisik FM, Tann M. State-of-the-art pancreatic MRI. American Journal of Roentgenology. 2010;195(1):42–53 [DOI] [PubMed] [Google Scholar]
- 31.Balci NC, Smith A, Momtahen AJ, Alkaade S, Fattahi R, Tariq S, et al. MRI and S-MRCP findings in patients with suspected chronic pancreatitis: Correlation with endoscopic pancreatic function testing (ePFT). Journal of Magnetic Resonance Imaging. 2010;31(3):601–6 [DOI] [PubMed] [Google Scholar]
- 32.Gillams A, Punwani S, Smart S, Lees W, editors. Quantification of Pancreatic Exocrine Function Using Secretln Stimulated MRCP. [DOI] [PubMed] [Google Scholar]
- 33.Hafezi-Nejad N, Singh VK, Faghih M, Kamel IR, Zaheer A. Jejunal response to secretin is independent of the pancreatic response in secretin-enhanced magnetic resonance cholangiopancreatography. European journal of radiology. 2019;112:7–13 [DOI] [PubMed] [Google Scholar]
- 34.Semelka RC, Patrick Shoenut J, Kroeker MA, Micflikier AB. Chronic pancreatitis: MR imaging features before and after administration of gadopentetate dimeglumine. Journal of Magnetic Resonance Imaging. 1993;3(1):79–82 [DOI] [PubMed] [Google Scholar]
- 35.Winston CB, Mitchell DG, Cutwater EK, Ehrlich SM. Pancreatic signal intensity on Tl-weighted fat saturation MR images: Clinical correlation. Journal of Magnetic Resonance Imaging. 1995;5(3):267–71 [DOI] [PubMed] [Google Scholar]
- 36.Manikkavasakar S, AlObaidy M, Busireddy KK, Ramalho M, Nilmini V, Alagiyawanna M, et al. Magnetic resonance imaging of pancreatitis: an update. World Journal of Gastroenterology: WJG. 2014;20(40):14760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tirkes T, Lin C, Cui E, Deng Y, Territo PR, Sandrasegaran K, et al. Quantitative MR Evaluation of Chronic Pancreatitis: Extracellular Volume Fraction and MR Relaxometry. AJR Am J Roentgenol. 2018;210(3):533–42 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tirkes T, Mitchell JR, Li L, Zhao X, Lin C. Normal T1 relaxometry and extracellular volume of the pancreas in subjects with no pancreas disease: correlation with age and gender. Abdom Radiol (NY). 2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Akisik MF, Sandrasegaran K, Jennings SG, Aisen AM, Lin C, Sherman S, et al. Diagnosis of chronic pancreatitis by using apparent diffusion coefficient measurements at 3.0-T MR following secretin stimulation. Radiology. 2009;252(2):418–25. [DOI] [PubMed] [Google Scholar]
- 40.Cho SG, Lee DH, Lee KY, Ji H, Lee KH, Ros PR, et al. Differentiation of chronic focal pancreatitis from pancreatic carcinoma by in vivo proton magnetic resonance spectroscopy. 2005;29(2):163–9 [DOI] [PubMed] [Google Scholar]
- 41.Heuck A, Maubach PA, Reiser M, Feuerbach S, Allgayer B, Lukas P, et al. Age-related morphology of the normal pancreas on computed tomography. Gastrointestinal radiology. 1987; 12(1): 18–22 [DOI] [PubMed] [Google Scholar]
- 42.Sato T, Ito K, Tamada T, Sone T, Noda Y, Higaki A, et al. Age-related changes in normal adult pancreas: MR imaging evaluation. European journal of radiology. 2012;81(9):2093–8 [DOI] [PubMed] [Google Scholar]
- 43.Hastier P, Buckley MJ, Dumas R, Kuhdorf H, Staccini P, Demarquay J-F, et al. A study of the effect of age on pancreatic duct morphology. Gastrointestinal endoscopy. 1998;48(1):53–7 [DOI] [PubMed] [Google Scholar]
- 44.Edge MD, Hoteit M, Patel AP, Wang X, Baumgarten DA, Cai Q. Clinical significance of main pancreatic duct dilation on computed tomography: single and double duct dilation. World Journal of Gastroenterology: WJG. 2007; 13(11):1701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Sai JK, Suyama M, Kubokawa Y, Watanabe S. Diagnosis of mild chronic pancreatitis (Cambridge classification): comparative study using secretin injection-magnetic resonance cholangiopancreatography and endoscopic retrograde pancreatography. World journal of gastroenterology: WJG. 2008;14(8):1218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Tamura R, Ishibashi T, Takahashi S. Chronic pancreatitis: MRCP versus ERCP for quantitative caliber measurement and qualitative evaluation. Radiology. 2006;238(3):920–8 [DOI] [PubMed] [Google Scholar]
- 47.Cappeliez O, Delhaye M, Deviere J, Le Moine O, Metens T, Nicaise N, et al. Chronic pancreatitis: evaluation of pancreatic exocrine function with MR pancreatography after secretin stimulation. Radiology. 2000;215(2):358–64 [DOI] [PubMed] [Google Scholar]
- 48.Wu C, Li F, Niu G, Chen X. PET imaging of inflammation biomarkers. Theranostics. 2013;3(7):448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Foss CA, Liu L, Mease RC, Wang H, Pasricha P, Pomper MG. Imaging macrophage accumulation in a murine model of chronic pancreatitis with 125I-Iodo-DPA-713 SPECT/CT. Journal of Nuclear Medicine. 2017;58(10):1685–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yasaka K, Abe O. Deep learning and artificial intelligence in radiology: Current applications and future directions. PLoS medicine. 2018;15(11):e1002707. [DOI] [PMC free article] [PubMed] [Google Scholar]