Abstract
Small double-strand RNA (dsRNA) molecules can activate endogenous genes via an RNA-based promoter targeting mechanism. RNA activation (RNAa) is an evolutionarily conserved mechanism present in diverse eukaryotic organisms ranging from nematodes to humans. Small activating RNAs (saRNAs) involved in RNAa have been successfully used to activate gene expression in cultured cells, and thereby this emergent technique might allow us to develop various biotechnological applications, without the need to synthesize hazardous construct systems harboring exogenous DNA sequences. Accordingly, this thematic issue aims to provide insights into how RNAa cellular machinery can be harnessed to activate gene expression leading to a more effective clinical treatment of various diseases.
Keywords: small interfering RNAs (siRNAs), microRNAs (miRNAs), small activating RNAs (saRNA), molecular mechanism, neuronal development, cardiac development, cancer, therapeutics
1. Introduction
Small interfering RNAs (siRNAs) and microRNAs (miRNAs), key regulators of gene expression, are recognized as small double-stranded RNA (dsRNA) molecules [1,2,3]. They are loaded onto Ago proteins generating an active Ago–RNA complex, which regulates gene expression at transcription and post-transcription levels [4,5]. Several studies have demonstrated that dsRNAs mostly inhibit gene expression either by chromatin modifications or by translation inhibition [6,7,8,9]. A new kind of small dsRNA, small activating RNAs (saRNAs), are able to induce rather than inhibit gene expression by targeting promoter sequences of some genes, which is termed “RNA activation” (RNAa) [10,11,12,13]. saRNAs are 21 nucleotides in length and act in an Ago2-dependent manner in mammals similar to RNAi [11]. Although the first reports showed that gene expression can be activated by synthetic dsRNAs targeting promoter regions, more recent studies confirmed that RNAa is an evolutionarily conserved mechanism present in diverse eukaryotic organisms ranging from nematodes to humans [14,15]. Like small double-stranded activating RNAs, it has been evidenced that some miRNAs activate gene expression through targeting both promoter sequence and/or AU-rich elements in UTRs [11,16,17]. Several independent studies have shown that a new group of activating RNAs could be generated from natural antisense transcripts (NATs) [14,15,16]. Antisense transcripts, known as long non-coding RNAs, are transcribed from the opposite DNA strand of a protein-coding gene locus, which is complementary to the corresponding coding RNA. They are functional elements, expressed in a tissue-specific manner, and generally low in abundance [17]. Recent reports demonstrate that antisense transcripts regulate their sense (protein-coding) partners through diverse transcriptional and post-transcriptional mechanisms [18,19]. While most antisense transcripts suppress corresponding sense gene expression, they can also be regarded as targets for saRNAs [13,15]. Notably, blocking the interaction of the sense (mRNA) and antisense transcripts (forming a natural duplex) and/or destruction of antisense transcripts by siRNAs or single-stranded oligonucleotides (antagoNATs) results in locus-specific transcriptional de-repression and upregulation of the gene, possibly by activation of the RNAi machinery [14,15,16]. Apart from promoter sequences, which are known as the main target sites for saRNAs, gene activation by small RNA fragments could operate in any genomic region where antisense transcripts are presented. Despite tremendous advantages offered by saRNAs, there are still critical challenges limiting the development of these cutting-edge therapeutics. The most significant challenges include low siRNA stability, degradation and opsonization in the bloodstream, and off-target effects (Table 1). Nanoparticle-mediated delivery systems can open a new way to overcome these problems.
Table 1.
Pros and cons of using saRNAs as therapeutics.
| Advantages | Disadvantages |
|---|---|
| Effective gene activation | Poor cellular uptake |
| Locus-specific activation of gene transcription, including undruggable targets | High sensitivity to RNase degradation |
| Easy to manufacture | Renal clearance |
| Cost-effectiveness | Repeated administration |
| Low toxicity | Off-target effects |
| Easy large-scale production | Activation of Toll-like receptors |
| Poor immunogenicity |
In this review, we present recent advances and challenges in the therapeutic applications of saRNAs.
2. Small Activating RNAs Are Involved in Locus-Specific Induction of Neural Genes
saRNA-mediated locus-specific gene activation has been studied in neural cells. Based on many studies, two models of locus-specific gene activation have been reported, promoter-targeted duplex RNA activating and natural antisense disruption, leading to changes in chromatin structure [8,14,15,20]. Several studies show that endogenous saRNAs selectively activate gene expression in neurons through targeting promoter sequences [21,22]. Kuwabara et al. have reported a neural-specific dsRNA of about 20 bp containing the NRSE (neuron restrictive silencer element) sequence. This sequence, which is defined as the NRSE/RE1, is localized within promoter regions of neuron-specific genes and is recognized by neuronal restricted silencing factor/RE-1 silencing transcription factor (NRSF/REST) leading to neuron-specific gene suppression in a non-neuronal cell. During an early stage of neurogenesis, the NRSE dsRNA induces the expression of genes containing the NRSE/RE1 sequence in their promoters. Indeed, the noncoding dsRNA, as an endogenous activating RNA, interacts with NRSF/REST machinery and modulates its function. Through this process, neural stem cells can be differentiated into neuronal and glial cells [21]. Cell-mediated brain repair suffers from poor survival rate of transplanted cells and the low efficiency of differentiation into neuronal cells [23]. Diodato and his colleagues have used pre-miRNAs (as activating RNAs) to increase the expression of Emx2, a human homeobox transcription factor modulating a number of developmental mechanisms such as development of cerebral cortex. Their results showed that the transactivation of Emx2 can result in delayed differentiation, self-renewal, and decreased death of neuronally committed precursors [24]. Exogenous saRNA-mediated gene activation in the brain has been reported by Fimiani and his co-workers [22]. Synthesized saRNAs induce the Foxg1 transcription factor, a key regulator of cortico-cerebral development and function. Foxg1 allele duplication and deletion in humans results in West and Rett syndromes, respectively [25]. As a prospective RNAa therapy of Rett syndrome, Foxg1 gene expression in neural cells has been induced in vitro and in vivo by intraventricular injection of saRNA to mouse neonates [22]. NATs destruction is another alternative locus-specific gene activation mechanism, which holds great therapeutic promise. For example, Modarresi et al. have studied the regulatory role of a natural antisense transcript in the brain-derived neurotrophic factor (BDNF) locus [14]. BDNF is a member of the neurotrophin growth factor family and is essential for neuronal maturation, plasticity, memory processes, and differentiation [26,27,28,29,30,31]. Interestingly, locus-specific induction of Bdnf expression is evidenced, both in vitro and in vivo, following degradation of a natural antisense transcript by single-stranded oligonucleotides (term antagoNAT) or siRNAs [14]. Induction of endogenous Bdnf expression following Bdnf-AS repression in neurospheres induced neuronal progenitor cell differentiation. Corresponding to these in vitro experiments, in vivo Bdnf mRNA and protein expression is also induced upon intracerebroventricular injection of Bdnf-antagoNAT9 in mice. It seems that in the case of Bdnf induction, a RNAa mechanism applies through regulating epigenetic modifications, namely reduction of the H3K27me3 repressive mark at the Bdnf locus. Transient induction of neurotrophin expression using the RNAa system is suggested as a pharmacological approach for several neurological disorders, as reduced neurotrophin expression has been observed in different neurodegenerative and neurodevelopmental disorders [14].
Spinal muscular atrophy (SMA) is another example of a neuromuscular disorder [32]. Insufficient expression of functional survival motor neuron protein (SMN), which is correlated with disease severity, leads to muscle weakness after birth [33,34,35]. Several therapeutic efforts have focused on increasing SMN expression [32]. d’Ydewalle et al. have identified a natural antisense transcript in SMN (SMN-AS) locus, which transcriptionally suppresses SMN expression through epigenetic modifications. Importantly, knockdown of the antisense transcript induces SMN transcriptional activity either in patient-derived cells or in the central nervous system of a SMA mouse model in vivo, improving survival of the mice and indicating a novel therapeutic target for SMA [36].
We believe that the next step for clinical translation of RNAa therapeutics for the treatment of various neurodegenerative and neurodevelopmental disorders is the development of novel drug delivery systems.
3. Small Activating RNAs Are Involved in Locus-Specific Induction of Cardiac Genes
RNAa-mediated locus-specific activation of gene transcription has been reported in cardiovascular cells [37,38,39]. In all reported studies, targeting of promoter regions by small dsRNA or small hairpin RNA (shRNA) leads to transcriptional activation of cardiovascular genes, which could open the way for therapeutic strategies. For instance, targeting the promoter region of vascular endothelial growth factor (VEGF) by shRNA results in transcriptional activation, suggesting a therapeutic strategy for myocardial infarction [37]. Targeting the antisense transcript has been recently demonstrated as an alternative RNA activating system in the cyclin-dependent kinase 9 (Cdk9) locus, a key player in cardiac development [15,16]. Cdk9 is associated with specific cyclins to form a heterodimer, Cyclin T/Cdk9, which is also known as the positive transcription elongation factor-b (P-TEFb) [40,41,42]. P-TEFb activates the polymerase II transcription machinery via phosphorylation of the carboxyl-terminal domain (CTD) [43,44]. Therefore, Cdk9 is mainly involved in transcriptional regulation and plays a critical role in several differentiation pathways. Furthermore, Cdk9 regulates cardiac-specific genes including Nkx2.5, Anf, and ß-Myh via interactions with the p300/GATA4 complex, particularly involved in cardiac differentiation [45]. Moreover, we have shown recently that Cdk9 regulates apoptosis in cardiomyocytes by modulating miRNA-1 expression, a critical microRNA for cardiac differentiation [46,47,48]. It is therefore possible that both synthesis and activity of Cdk9 are tightly regulated at the transcriptional and post-transcriptional levels. In this regard, at least three non-coding RNAs are involved in Cdk9 regulation [16]. In the context of normal human cardiomyocytes, Cdk9 activity is suppressed at the protein level via interaction with 7SK non-coding RNA and at the translational level through muscle-specific microRNAs, specifically miR-1 and miR-133 [46,49,50]. We have recently reported a third mode of RNA control in the Cdk9 locus [15]. Small non-coding RNA molecules of 22bp with sequences homologous to the transcript result in transcriptional activation of Cdk9. Interestingly, NATs complementary to the most 3′ and 5′ regions of the gene were identified. Indeed, hybridization of the short single-stranded cognate transcript fragments with antisense transcripts provides the signal for transcriptional activation. The requirement of Argonaute proteins and endogenous antisense transcripts for transcriptional activation indicates that the activating single-stranded small RNAs are processed by the RNAi machinery [51,52]. Similar to siRNA knockdown, antisense transcript distraction following the sense oligoribonucleotide electroporation could represent a secondary phenomenon of the activation of the RNAi machinery. This activation may then result in a change in epigenetic modifications at the locus, leading to the induction of Cdk9 transcription as described for several genes [1,53]. As a functional consequence of RNA activation in the Cdk9 locus, an increased cardiac differentiation potential is observed in ES cells when electroporated with the sense oligoribonucleotide. Interestingly, injection into wild-type blastocysts of RNA-programmed ES cells contributes specifically to heart development in vivo, indicating that a transient RNA activation system is sufficient to create a cardiac differentiation “memory” in cells and may represent a novel tool for RNA–cell reprogramming applied in regenerative medicine.
4. Small Activating RNAs: New Insights into Cancer Therapy
Recently, RNA-based therapeutics have gained more attention in cancer therapy due to their enormous potential to selectively target previously undruggable genes and gene expression modulators. Unlike RNAi, which mostly targets sense transcripts, RNAa, as an alternative and promising new therapeutic strategy, can activate gene expression in a natural manner by targeting antisense transcripts or promoter regions [13,54,55,56]. Tumor suppressor genes, which are mostly suppressed in cancers, could be targeted by saRNA to enhance transcriptional activation and restore a normal cell phenotype.
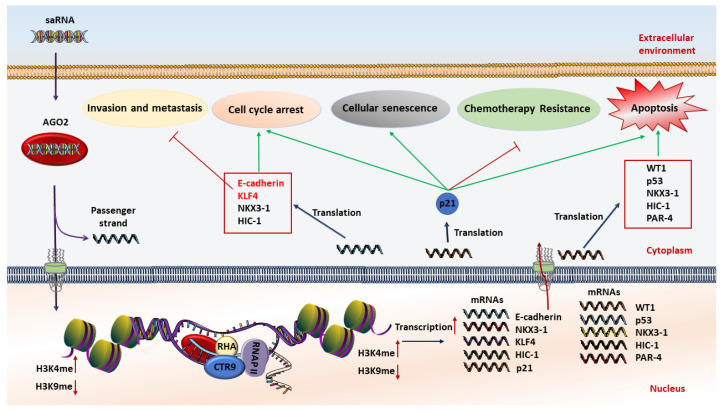
saRNAs can exert their anticancer effects through induction of cell cycle arrest, cellular senescence, proliferation inhibition, apoptosis induction, metastasis suppression, and multidrug resistance reversal (Figure 1). Although several tumor suppressor genes including E-cadherin, NKX3-1, Wt1, and P53 have been induced by this method, p21 is the most investigated tumor suppressor gene for RNAa-mediated gene activation in several tumors and cell lines [10,57,58,59,60,61,62]. P21 is a negative regulator of the cell cycle and is rarely mutated in cancers, and therefore represents a key target for small RNA activation cancer therapy. In this regard, re-activation of the p21 gene by targeting promoter regions inhibits cell viability and proliferation rates, while it induces apoptotic cell death and sensitizes lung cancer cells to chemotherapeutic agents, providing a new approach in cancer therapy [59,60,63].
Figure 1.
Schematic illustration of the anticancer mechanism of saRNA (small activating RNA)-based therapeutics. At first, saRNAs are loaded on the AGO2 protein. Then AGO2 separates the passenger strand. After that, the complex of saRNA guide strand and AGO2 cross the nuclear and interact with promoter sequences of interested genes to increase transcription by methylation of H3K4 and/or demethylation of H3K9. The expression level of tumor suppressor genes is restored, resulting in induction of apoptosis, chemoresistance reversal, inhibition of invasion and metastasis, cell cycle arrest, and cellular senescence.
5. Towards the Development of New Therapeutic Agents
In the past few years, a steadily increasing number of clinical and preclinical studies have been performed, using various saRNA-based therapies for the treatment of a multitude of different diseases (Table 2 and Supplementary Table S1).
Table 2.
Significant preclinical and clinical studies on the features of saRNA-based therapeutics.
| Disease Condition |
Gene | Comments | Ref. |
|---|---|---|---|
| Advanced liver cancer | CEBPA | The first clinical trial for saRNA-based therapeutics (NCT ID: NCT02716012; company: Mina Alpha Limited; phase 1). MTL-CEBPA shows favorable safety and promising synergistic effects in combination with TKIs. | [64] |
| Adult solid tumors | CEBPA | A new clinical trial of MTL-CEBPA in combination with pembrolizumab (NCT ID: NCT04105335; Phase 1; recruitment status: Recruiting). | [65] |
| Prostate cancer | P21 | Proliferation inhibition and tumor shrinkage. | [58] |
| Hepatocellular carcinoma (HCC) | P21 | Cell cycle arrest and inhibition of invasion and migration. | [10] |
| Non-small-cell lung carcinomas | P21 | In vitro: Proliferation inhibition, cell cycle arrest, and apoptosis induction. In vivo: Inhibition of tumor growth. |
[59] |
| Pancreatic cancer | P21 | In vitro: Proliferation inhibition, cell cycle arrest, and apoptosis induction. In vivo: Inhibition of tumor growth; high safety. |
[66] |
| Bladder cancer | P21 | Proliferation inhibition, cell cycle arrest, and apoptosis induction. | [67] |
| HCC | WT1 | Proliferation inhibition and apoptosis induction. | [68] |
| Prostate cancer | Ecad | Inhibition of invasion and migration. | [69] |
| Bladder cancer | Ecad | Inhibition of invasion and migration. | [70] |
| Breast cancer | Ecad | In vitro: Proliferation inhibition, cell cycle arrest, apoptosis induction, and inhibition of invasion and migration. In vivo: Tumor growth inhibition |
[71] |
| Prostate cancer | KLF4 | Proliferation inhibition, cell cycle arrest, apoptosis induction, and inhibition of invasion and migration. | [72] |
| Malignant pheochromocytoma | TP53 | In vitro: Cell cycle arrest, proliferation inhibition, and apoptosis induction. In vivo: Tumor shrinkage. |
[73] |
| Breast cancer | HIC-1 | Proliferation inhibition and apoptosis induction. | [74] |
| Bladder and prostate cancer | PAWR | Proliferation inhibition and apoptosis induction. | [75] |
| Prostate cancer | NKX3-1 | In vitro: Proliferation inhibition, cell cycle arrest, apoptosis induction. In vivo: Tumor growth inhibition. |
[76] |
| Nephrolithiasis | TRPV5 | In vitro: TRPV5 expression induction. In vivo: TRPV5 expression induction and reduction in the formation of CaOx kidney stone. |
[77] |
| Renal cell carcinoma | VHL | Cell growth inhibition and apoptosis induction. | [78] |
| HCC | NIS | Apoptosis induction and viability reduction of cancer cells. | [79] |
| Bladder cancer | P21 | Tumor Shrinkage | [80] |
| HCC | CEBPA | In vitro: CEBPA overexpression. In vivo: Tumor growth inhibition and tumor shrinkage. |
[64] |
| HCC | CEBPA | In vitro: Proliferation inhibition. In vivo: Tumor burden reduction. |
[81] |
| HCC | CEBPA | In vitro: Cell migration and invasion inhibition. In vivo: Metastasis inhibition |
[82] |
| Colorectal cancer | P21 | In vitro: Apoptosis induction, proliferation inhibition, and cell migration and invasion inhibition. In vivo: Tumor growth inhibition. |
[83] |
| Pancreatic ductal adenocarcinoma | CEBPA | In vitro: Proliferation inhibition. In vivo: Tumor shrinkage. |
[84] |
| Prostate cancer | DPYSL3 | In vitro: Proliferation inhibition and cell migration and invasion inhibition. In vivo: Metastasis inhibition |
[85] |
| Diabetes-induced erectile dysfunction | Nos2 | In vitro: iNos overexpression. In vivo: iNos overexpression and enhancement of peak intracavernous pressure. |
[86] |
| Human metastatic castration-resistant prostate cancer | Notch1 | In vitro: Cell migration and invasion suppression, cell cycle arrest, and apoptosis inhibition. In vivo: Tumor growth inhibition and suppression of VEGF and AR pathways mechanisms. |
[87] |
| Non-alcoholic fatty liver disease | HNF4A | In vitro: Increase in the expression level of HNF4A, CYP450, CYP3A4, CYP3A5, and CYP3A7. In vivo: Liver triglyceride reduction, high-density lipoprotein/low-density lipoprotein (HDL/LDL) ratio enhancement, and white adipose tissue/body weight ratio reduction. |
[88] |
| Endometrial carcinoma | FHIT | Proliferation, invasion, and metastasis inhibition. | [89] |
Supplementary Table S1 provides more experimental details on the same studies mentioned in Table 2.
Due to their potential involvement in human disorders, strong efforts have been undertaken to develop new therapeutic agents applying a RNAa strategy. For instance, two companies in the USA, RNAa Therapeutics and OPKO-CURNA, are investigating therapeutic approaches based on RNA activation. Of note, mipomersen, an oligonucleotide targeting apolipoprotein B, was approved by the FDA in January 2013, but received a negative opinion from a European Medicines Agency panel [90]. RNA-activating molecules as promising drugs are under active investigation due to their high potency and specificity, locus-specific manner functions, targeting of the correct cells, small molecular size, and low toxicity [10,91]. Traditionally, an exogenous DNA construct is often required for ectopic gene expression. Apart from exogenous DNA constructs excluding regulatory elements, such systems have been problematic in the clinic because of requiring viral-based vectors for gene delivery. Several limitations and undesirable side effects of viral-based gene therapy including host genome integrity, the tedious process of construction, and various negative immunological responses which have been previously reported [92,93,94,95]. Upon delivery as mature moieties, an RNAa system offers a more natural approach and safer gene therapy method by targeting promoter regions or through natural antisense destruction, which activates endogenous targeted gene expression in a locus-specific manner in the absence of exogenous DNA. Moreover, RNAa offers an endogenous induction in a natural cellular niche leading to the correctly processed proteins with their appropriate modifications. Notably, in the case of gene activation through natural antisense destruction, short oligoribonucleotides derived from a sense transcript (mRNA) can function as an activator RNA [15], which are safer, natural, and without activating immune responses.
To improve the medicinal properties and potential in vivo applications of saRNAs, two steps must be taken. First, novel nanoparticle-based drug delivery systems must be developed to increase the drug accumulation in the targeted tissue and also address the aforementioned challenges in the therapeutic application of saRNAs. To date, different delivery systems have been developed for the delivery of saRNAs. Recently, lipid-based nanoparticles (LNPs) have attracted worldwide attention. LNP-based delivery systems face serious limitations such as toxicity, low thermodynamic stability, poor efficiency of encapsulation, and leaking challenges [96]. To overcome these limitations, various strategies have been proposed. The surface modification of delivery systems with flexible, non-ionic, and hydrophilic polymers, such as PEG, has been proposed as a robust strategy to address serum protein opsonization issues [97,98]. Furthermore, surface modification with biodegradable nano-polymers such as PEG can be used to decrease the toxicity of nanoparticles [99]. To increase the expression of P21, 2′-fluoro-modified P21 saRNA (dsP21-322-2′F) was delivered into an orthotopic bladder cancer mouse model by using a novel PEG-modified lipid nanoparticle. Results showed a significant increase in urothelium uptake and high tumor shrinkage [80]. Aptamers, nucleic acid ligands, can be used for targeted drug delivery as they can form specific three-dimensional structures based on their sequences. Yoon and his colleagues have synthesized PDAC specific 2′-Fluropyrimidine RNA-aptamers (2′F-RNA)- P19 and P1 for targeted delivery of saRNA into both PANC-1 and AsPC-1 engrafted mice. After intravenous injections of the aptamer–C/EBPα saRNA in both tumor mice models, tumor growth was significantly suppressed in comparison with mice treated with gemcitabine [84].
The second step for the development of effective saRNA therapeutics relies on tailoring site-specific chemical modifications of saRNAs. For instance, blocking the 5′-OH of the passenger strand and modifications to the 2′ backbone (i.e., 2′-OMe, 2′-Fluoro, and locked nucleic acid) has been demonstrated to reduce its off-target potential and increase endonuclease resistance and serum stability, respectively [55,58,100]. With single-stranded therapeutic oligonucleotides, designated antagoNAT, chemical modifications not only promote metabolic stability and target specificity but also minimize the length of the oligonucleotide to improve cellular uptake. In this case, 16-mer antagoNAT oligonucleotides, also known as a gapmer [101], with three locked nucleic acid (LNA) substitutions at each end and phosphorothioate-modified backbones, have been used in in vivo studies [14,102,103,104,105]. Along with chemical modifications to improve the stability and specificity, targeted and efficient in vivo delivery of oligonucleotides is also critical for RNA-based therapies. Among the currently investigated approaches [106,107,108,109], lipid-based formulations are the most promising delivery agents for systemic or localized saRNA delivery [100,110,111]. Several clinical trials that are testing RNAa-based drugs are commonly using lipid carriers [112,113]. Considering the increasing knowledge regarding RNAa-based locus-specific gene activation as a new strategy with promising therapeutic perspectives, it is clear that there is a growing scientific as well as a commercial interest to develop new therapeutic agents and clinical treatments based on this innovative approach. Lipid nanoparticle-formulated nucleoside-modified RNAs have been recently introduced as the Covid-19 vaccine, supporting the notion of efficient lipid-based delivery, safety, and efficiency of RNA therapies [114,115].
6. Conclusions and Future Perspectives
RNAa-based drugs have gained great attention in the past few years owing to their high potential in treating various disorders. However, delivery challenges slow down the clinical translation of this new class of therapeutic agents. Enzymatic digestion, low cellular uptake, poor endosomal escape, off-target effects, and fast clearance from the bloodstream have necessitated optimization of not only the RNAa molecules but also of the delivery systems. Many non-viral delivery systems have been designed and developed for the delivery of RNAa-based therapeutic systems. Nanoparticle-based delivery systems have several advantages including high functionalization and targeting capacity and ease of large-scale development, which make them a promising delivery system for medical applications. For instance, several studies demonstrated that PEGylation of different materials such as PEI can enhance their circulation time and reduce hemolysis and serum opsonization. Chemical modifications of RNA molecules herald a new era in the development of these cutting-edge RNA-based therapeutics. However, these modifications might be limited due to the occurrence of severe side effects and functionality reduction, which are topics that urgently need to be addressed.
Future applications of RNAa-based therapeutics will significantly advance due to the progress in the development of drug delivery vehicles, reduction of the off-targeting problem, and huge large-scale synthesis capacity that will be offered by pharmaceutical companies. The combinatorial strategy using RNAa with chemotherapeutic drugs also could be a new strong treatment modality for different types of diseases.
Acknowledgments
The authors acknowledge the Shahid Beheshti University of Medical Sciences and the University Cote d’Azur for support.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4409/10/3/591/s1, Table S1: Significant preclinical and clinical studies on the features of saRNA-based therapeutics—Extended characteristics.
Author Contributions
Conceptualization, H.G. and K.-D.W.; writing—original draft preparation, H.G. and M.E.; writing—review and editing, S.A., N.W. and K.-D.W.; supervision, K.-D.W., H.G., and N.W. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Aghamiri S., Mehrjardi K.F., Shabani S., Keshavarz-Fathi M., Kargar S., Rezaei N. Nanoparticle-siRNA: A potential strategy for ovarian cancer therapy? Nanomedicine. 2019;14:2083–2100. doi: 10.2217/nnm-2018-0379. [DOI] [PubMed] [Google Scholar]
- 2.Setten R.L., Rossi J.J., Han S.P. The current state and future directions of RNAi-based therapeutics. Nat. Rev. Drug Discov. 2019;18:421–446. doi: 10.1038/s41573-019-0017-4. [DOI] [PubMed] [Google Scholar]
- 3.Bajan S., Hutvagner G. RNA-Based Therapeutics: From Antisense Oligonucleotides to miRNAs. Cells. 2020;9:137. doi: 10.3390/cells9010137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Dana H., Chalbatani G.M., Mahmoodzadeh H., Karimloo R., Rezaiean O., Moradzadeh A., Mehmandoost N., Moazzen F., Mazraeh A., Marmari V., et al. Molecular Mechanisms and Biological Functions of siRNA. Int. J. Biomed Sci. 2017;13:48–57. [PMC free article] [PubMed] [Google Scholar]
- 5.Sun G., Yeh S.Y., Yuan C.W., Chiu M.J., Yung B.S., Yen Y. Molecular Properties, Functional Mechanisms, and Applications of Sliced siRNA. Mol. Ther. Nucleic Acids. 2015;4:e221. doi: 10.1038/mtna.2014.73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Holoch D., Moazed D. RNA-mediated epigenetic regulation of gene expression. Nat. Rev. Genet. 2015;16:71–84. doi: 10.1038/nrg3863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Volpe T., Martienssen R.A. RNA interference and heterochromatin assembly. Cold Spring Harb. Perspect. Biol. 2011;3:a003731. doi: 10.1101/cshperspect.a003731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Czech B., Hannon G.J. Small RNA sorting: Matchmaking for Argonautes. Nat. Rev. Genet. 2011;12:19–31. doi: 10.1038/nrg2916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Weng Y., Xiao H., Zhang J., Liang X.J., Huang Y. RNAi therapeutic and its innovative biotechnological evolution. Biotechnol. Adv. 2019;37:801–825. doi: 10.1016/j.biotechadv.2019.04.012. [DOI] [PubMed] [Google Scholar]
- 10.Kosaka M., Kang M.R., Yang G., Li L.-C. Targeted p21WAF1/CIP1 Activation by RNAa Inhibits Hepatocellular Carcinoma Cells. Nucleic Acid Ther. 2012;22:335–343. doi: 10.1089/nat.2012.0354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Meng X., Jiang Q., Chang N., Wang X., Liu C., Xiong J., Cao H., Liang Z. Small activating RNA binds to the genomic target site in a seed-region-dependent manner. Nucleic Acids Res. 2016;44:2274–2282. doi: 10.1093/nar/gkw076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang J., Place R.F., Portnoy V., Huang V., Kang M.R., Kosaka M., Ho M.K.C., Li L.C. Inducing gene expression by targeting promoter sequences using small activating RNAs. J. Biol. Methods. 2015;2 doi: 10.14440/jbm.2015.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Portnoy V., Huang V., Place R.F., Li L.C. Small RNA and transcriptional upregulation. Wiley Interdiscip. Rev. RNA. 2011;2:748–760. doi: 10.1002/wrna.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Modarresi F., Faghihi M.A., Lopez-Toledano M.A., Fatemi R.P., Magistri M., Brothers S.P., van der Brug M.P., Wahlestedt C. Inhibition of natural antisense transcripts in vivo results in gene-specific transcriptional upregulation. Nat. Biotechnol. 2012;30:453–459. doi: 10.1038/nbt.2158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ghanbarian H., Wagner N., Michiels J.F., Cuzin F., Wagner K.D., Rassoulzadegan M. Small RNA-directed epigenetic programming of embryonic stem cell cardiac differentiation. Sci. Rep. 2017;7:41799. doi: 10.1038/srep41799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ghanbarian H., Grandjean V., Cuzin F., Rassoulzadegan M. A Network of Regulations by Small Non-Coding RNAs: The P-TEFb Kinase in Development and Pathology. Front. Genet. 2011;2:95. doi: 10.3389/fgene.2011.00095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lin S., Zhang L., Luo W., Zhang X. Characteristics of Antisense Transcript Promoters and the Regulation of Their Activity. Int. J. Mol. Sci. 2015;17:9. doi: 10.3390/ijms17010009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Faghihi M., Wahlestedt C. Regulatory roles of natural antisense transcripts. Nat. Rev. Mol. Cell Biol. 2009;10:637–643. doi: 10.1038/nrm2738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Faghihi M.A., Kocerha J., Modarresi F., Engström P.G., Chalk A.M., Brothers S.P., Koesema E., St Laurent G., Wahlestedt C. RNAi screen indicates widespread biological function for human natural antisense transcripts. PLoS ONE. 2010;5:e13177. doi: 10.1371/journal.pone.0013177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim D.H., Saetrom P., Snøve O., Rossi J.J. MicroRNA-directed transcriptional gene silencing in mammalian cells. Proc. Natl. Acad. Sci. USA. 2008;105:16230–16235. doi: 10.1073/pnas.0808830105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kuwabara T., Hsieh J., Nakashima K., Taira K., Gage F.H. A small modulatory dsRNA specifies the fate of adult neural stem cells. Cell. 2004;116:779–793. doi: 10.1016/S0092-8674(04)00248-X. [DOI] [PubMed] [Google Scholar]
- 22.Fimiani C., Goina E., Su Q., Gao G., Mallamaci A. RNA activation of haploinsufficient Foxg1 gene in murine neocortex. Sci. Rep. 2016;6:39311. doi: 10.1038/srep39311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Burns T.C., Verfaillie C.M., Low W.C. Stem cells for ischemic brain injury: A critical review. J. Comp. Neurol. 2009;515:125–144. doi: 10.1002/cne.22038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Diodato A., Pinzan M., Granzotto M., Mallamaci A. Promotion of cortico-cerebral precursors expansion by artificial pri-miRNAs targeted against the Emx2 locus. Curr. Gene Ther. 2013;13:152–161. doi: 10.2174/1566523211313020009. [DOI] [PubMed] [Google Scholar]
- 25.Guerrini R., Parrini E. Epilepsy in Rett syndrome, and CDKL5- and FOXG1-gene-related encephalopathies. Epilepsia. 2012;53:2067–2078. doi: 10.1111/j.1528-1167.2012.03656.x. [DOI] [PubMed] [Google Scholar]
- 26.Ortega J.A., Alcántara S. BDNF/MAPK/ERK-induced BMP7 expression in the developing cerebral cortex induces premature radial glia differentiation and impairs neuronal migration. Cereb. Cortex. 2010;20:2132–2144. doi: 10.1093/cercor/bhp275. [DOI] [PubMed] [Google Scholar]
- 27.Antal A., Chaieb L., Moliadze V., Monte-Silva K., Poreisz C., Thirugnanasambandam N., Nitsche M.A., Shoukier M., Ludwig H., Paulus W. Brain-derived neurotrophic factor (BDNF) gene polymorphisms shape cortical plasticity in humans. Brain Stimul. 2010;3:230–237. doi: 10.1016/j.brs.2009.12.003. [DOI] [PubMed] [Google Scholar]
- 28.Castillo D.V., Escobar M.L. A role for MAPK and PI-3K signaling pathways in brain-derived neurotrophic factor modification of conditioned taste aversion retention. Behav. Brain Res. 2011;217:248–252. doi: 10.1016/j.bbr.2010.10.013. [DOI] [PubMed] [Google Scholar]
- 29.Nikolakopoulou A.M., Meynard M.M., Marshak S., Cohen-Cory S. Synaptic maturation of the Xenopus retinotectal system: Effects of brain-derived neurotrophic factor on synapse ultrastructure. J. Comp. Neurol. 2010;518:972–989. doi: 10.1002/cne.22258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Choi D.C., Maguschak K.A., Ye K., Jang S.W., Myers K.M., Ressler K.J. Prelimbic cortical BDNF is required for memory of learned fear but not extinction or innate fear. Proc. Natl. Acad. Sci. USA. 2010;107:2675–2680. doi: 10.1073/pnas.0909359107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Chapleau C.A., Larimore J.L., Theibert A., Pozzo-Miller L. Modulation of dendritic spine development and plasticity by BDNF and vesicular trafficking: Fundamental roles in neurodevelopmental disorders associated with mental retardation and autism. J. Neurodev. Disord. 2009;1:185–196. doi: 10.1007/s11689-009-9027-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.d’Ydewalle C., Sumner C.J. Spinal Muscular Atrophy Therapeutics: Where do we Stand? Neurotherapeutics. 2015;12:303–316. doi: 10.1007/s13311-015-0337-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lefebvre S., Bürglen L., Reboullet S., Clermont O., Burlet P., Viollet L., Benichou B., Cruaud C., Millasseau P., Zeviani M. Identification and characterization of a spinal muscular atrophy-determining gene. Cell. 1995;80:155–165. doi: 10.1016/0092-8674(95)90460-3. [DOI] [PubMed] [Google Scholar]
- 34.Lorson C.L., Hahnen E., Androphy E.J., Wirth B. A single nucleotide in the SMN gene regulates splicing and is responsible for spinal muscular atrophy. Proc. Natl. Acad. Sci. USA. 1999;96:6307–6311. doi: 10.1073/pnas.96.11.6307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lefebvre S., Burlet P., Liu Q., Bertrandy S., Clermont O., Munnich A., Dreyfuss G., Melki J. Correlation between severity and SMN protein level in spinal muscular atrophy. Nat. Genet. 1997;16:265–269. doi: 10.1038/ng0797-265. [DOI] [PubMed] [Google Scholar]
- 36.d’Ydewalle C., Ramos D.M., Pyles N.J., Ng S.Y., Gorz M., Pilato C.M., Ling K., Kong L., Ward A.J., Rubin L.L., et al. The Antisense Transcript SMN-AS1 Regulates SMN Expression and Is a Novel Therapeutic Target for Spinal Muscular Atrophy. Neuron. 2017;93:66–79. doi: 10.1016/j.neuron.2016.11.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Turunen M.P., Husso T., Musthafa H., Laidinen S., Dragneva G., Laham-Karam N., Honkanen S., Paakinaho A., Laakkonen J.P., Gao E., et al. Epigenetic upregulation of endogenous VEGF-A reduces myocardial infarct size in mice. PLoS ONE. 2014;9:e89979. doi: 10.1371/journal.pone.0089979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen R., Wang T., Rao K., Yang J., Zhang S., Wang S., Liu J., Ye Z. Up-regulation of VEGF by small activator RNA in human corpus cavernosum smooth muscle cells. J. Sex Med. 2011;8:2773–2780. doi: 10.1111/j.1743-6109.2011.02412.x. [DOI] [PubMed] [Google Scholar]
- 39.Husso T., Ylä-Herttuala S., Turunen M.P. A New Gene Therapy Approach for Cardiovascular Disease by Non-coding RNAs Acting in the Nucleus. Mol. Ther. Nucleic Acids. 2014;3:e197. doi: 10.1038/mtna.2014.48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Lin X., Taube R., Fujinaga K., Peterlin B.M. P-TEFb containing cyclin K and Cdk9 can activate transcription via RNA. J. Biol. Chem. 2002;277:16873–16878. doi: 10.1074/jbc.M200117200. [DOI] [PubMed] [Google Scholar]
- 41.Marshall N.F., Price D.H. Purification of P-TEFb, a transcription factor required for the transition into productive elongation. J. Biol. Chem. 1995;270:12335–12338. doi: 10.1074/jbc.270.21.12335. [DOI] [PubMed] [Google Scholar]
- 42.Price D.H. P-TEFb, a cyclin-dependent kinase controlling elongation by RNA polymerase II. Mol. Cell Biol. 2000;20:2629–2634. doi: 10.1128/MCB.20.8.2629-2634.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Leucci E., De Falco G., Onnis A., Cerino G., Cocco M., Luzzi A., Crupi D., Tigli C., Bellan C., Tosi P., et al. The role of the Cdk9/Cyclin T1 complex in T cell differentiation. J. Cell Physiol. 2007;212:411–415. doi: 10.1002/jcp.21032. [DOI] [PubMed] [Google Scholar]
- 44.Malumbres M., Pevarello P., Barbacid M., Bischoff J.R. CDK inhibitors in cancer therapy: What is next? Trends Pharmacol. Sci. 2008;29:16–21. doi: 10.1016/j.tips.2007.10.012. [DOI] [PubMed] [Google Scholar]
- 45.Kaichi S., Takaya T., Morimoto T., Sunagawa Y., Kawamura T., Ono K., Shimatsu A., Baba S., Heike T., Nakahata T., et al. Cyclin-dependent kinase 9 forms a complex with GATA4 and is involved in the differentiation of mouse ES cells into cardiomyocytes. J. Cell Physiol. 2011;226:248–254. doi: 10.1002/jcp.22336. [DOI] [PubMed] [Google Scholar]
- 46.Tarhriz V., Wagner K.D., Masoumi Z., Molavi O., Hejazi M.S., Ghanbarian H. CDK9 Regulates Apoptosis of Myoblast Cells by Modulation of microRNA-1 Expression. J. Cell Biochem. 2018;119:547–554. doi: 10.1002/jcb.26213. [DOI] [PubMed] [Google Scholar]
- 47.Wagner K.D., Wagner N., Ghanbarian H., Grandjean V., Gounon P., Cuzin F., Rassoulzadegan M. RNA induction and inheritance of epigenetic cardiac hypertrophy in the mouse. Dev. Cell. 2008;14:962–969. doi: 10.1016/j.devcel.2008.03.009. [DOI] [PubMed] [Google Scholar]
- 48.Xin M., Olson E.N., Bassel-Duby R. Mending broken hearts: Cardiac development as a basis for adult heart regeneration and repair. Nat. Rev. Mol. Cell Biol. 2013;14:529–541. doi: 10.1038/nrm3619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sayed D., Hong C., Chen I.Y., Lypowy J., Abdellatif M. MicroRNAs play an essential role in the development of cardiac hypertrophy. Circ. Res. 2007;100:416–424. doi: 10.1161/01.RES.0000257913.42552.23. [DOI] [PubMed] [Google Scholar]
- 50.Peterlin B.M., Brogie J.E., Price D.H. 7SK snRNA: A noncoding RNA that plays a major role in regulating eukaryotic transcription. Wiley Interdiscip. Rev. RNA. 2012;3:92–103. doi: 10.1002/wrna.106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Mattick J.S. The State of Long Non-Coding RNA Biology. Noncoding RNA. 2018;4:17. doi: 10.3390/ncrna4030017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mattick J.S., Rinn J.L. Discovery and annotation of long noncoding RNAs. Nat. Struct. Mol. Biol. 2015;22:5–7. doi: 10.1038/nsmb.2942. [DOI] [PubMed] [Google Scholar]
- 53.Zhang X., Li H., Burnett J.C., Rossi J.J. The role of antisense long noncoding RNA in small RNA-triggered gene activation. RNA. 2014;20:1916–1928. doi: 10.1261/rna.043968.113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Li L.C., Okino S.T., Zhao H., Pookot D., Place R.F., Urakami S., Enokida H., Dahiya R. Small dsRNAs induce transcriptional activation in human cells. Proc. Natl. Acad. Sci. USA. 2006;103:17337–17342. doi: 10.1073/pnas.0607015103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Matsui M., Sakurai F., Elbashir S., Foster D.J., Manoharan M., Corey D.R. Activation of LDL receptor expression by small RNAs complementary to a noncoding transcript that overlaps the LDLR promoter. Chem. Biol. 2010;17:1344–1355. doi: 10.1016/j.chembiol.2010.10.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Werner A. Biological functions of natural antisense transcripts. BMC Biol. 2013;11:31. doi: 10.1186/1741-7007-11-31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chen Z., Place R.F., Jia Z.J., Pookot D., Dahiya R., Li L.C. Antitumor effect of dsRNA-induced p21(WAF1/CIP1) gene activation in human bladder cancer cells. Mol. Cancer Ther. 2008;7:698–703. doi: 10.1158/1535-7163.MCT-07-2312. [DOI] [PubMed] [Google Scholar]
- 58.Place R.F., Wang J., Noonan E.J., Meyers R., Manoharan M., Charisse K., Duncan R., Huang V., Wang X., Li L.C. Formulation of Small Activating RNA Into Lipidoid Nanoparticles Inhibits Xenograft Prostate Tumor Growth by Inducing p21 Expression. Mol. Ther. Nucleic Acids. 2012;1:e15. doi: 10.1038/mtna.2012.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Wei J., Zhao J., Long M., Han Y., Wang X., Lin F., Ren J., He T., Zhang H. p21WAF1/CIP1 gene transcriptional activation exerts cell growth inhibition and enhances chemosensitivity to cisplatin in lung carcinoma cell. BMC Cancer. 2010;10:632. doi: 10.1186/1471-2407-10-632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wu Z.M., Dai C., Huang Y., Zheng C.F., Dong Q.Z., Wang G., Li X.W., Zhang X.F., Li B., Chen G. Anti-cancer effects of p21WAF1/CIP1 transcriptional activation induced by dsRNAs in human hepatocellular carcinoma cell lines. Acta Pharmacol. Sin. 2011;32:939–946. doi: 10.1038/aps.2011.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Frixen U.H., Behrens J., Sachs M., Eberle G., Voss B., Warda A., Löchner D., Birchmeier W. E-cadherin-mediated cell-cell adhesion prevents invasiveness of human carcinoma cells. J. Cell Biol. 1991;113:173–185. doi: 10.1083/jcb.113.1.173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Richards F.M., McKee S.A., Rajpar M.H., Cole T.R., Evans D.G., Jankowski J.A., McKeown C., Sanders D.S., Maher E.R. Germline E-cadherin gene (CDH1) mutations predispose to familial gastric cancer and colorectal cancer. Hum. Mol. Genet. 1999;8:607–610. doi: 10.1093/hmg/8.4.607. [DOI] [PubMed] [Google Scholar]
- 63.Zandsalimi F., Talaei S., Noormohammad Ahari M., Aghamiri S., Raee P., Roshanzamiri S., Yarian F., Bandehpour M., Zohrab Zadeh Z. Antimicrobial peptides: A promising strategy for lung cancer drug discovery? Expert Opin. Drug Discov. 2020;15:1343–1354. doi: 10.1080/17460441.2020.1791080. [DOI] [PubMed] [Google Scholar]
- 64.Sarker D., Plummer R., Meyer T., Sodergren M.H., Basu B., Chee C.E., Huang K.W., Palmer D.H., Ma Y.T., Evans T.R.J., et al. MTL-CEBPA, a Small Activating RNA Therapeutic Upregulating C/EBP-α, in Patients with Advanced Liver Cancer: A First-in-Human, Multicenter, Open-Label, Phase I Trial. Clin. Cancer Res. 2020;26:3936–3946. doi: 10.1158/1078-0432.CCR-20-0414. [DOI] [PubMed] [Google Scholar]
- 65.Kramer E.D., Abrams S.I. Granulocytic Myeloid-Derived Suppressor Cells as Negative Regulators of Anticancer Immunity. Front. Immunol. 2020;11:1963. doi: 10.3389/fimmu.2020.01963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Zhang Z., Wang Z., Liu X., Wang J., Li F., Li C., Shan B. Up-regulation of p21WAF1/CIP1 by small activating RNA inhibits the in vitro and in vivo growth of pancreatic cancer cells. Tumori. 2012;98:804–811. doi: 10.1177/030089161209800620. [DOI] [PubMed] [Google Scholar]
- 67.Yang K., Zheng X.Y., Qin J., Wang Y.B., Bai Y., Mao Q.Q., Wan Q., Wu Z.M., Xie L.P. Up-regulation of p21WAF1/Cip1 by saRNA induces G1-phase arrest and apoptosis in T24 human bladder cancer cells. Cancer Lett. 2008;265:206–214. doi: 10.1016/j.canlet.2008.02.014. [DOI] [PubMed] [Google Scholar]
- 68.Qin Q., Lin Y.W., Zheng X.Y., Chen H., Mao Q.Q., Yang K., Huang S.J., Zhao Z.Y. RNAa-mediated overexpression of WT1 induces apoptosis in HepG2 cells. World J. Surg. Oncol. 2012;10:11. doi: 10.1186/1477-7819-10-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Mao Q., Zheng X., Yang K., Qin J., Bai Y., Jia X., Li Y., Xie L. Suppression of migration and invasion of PC3 prostate cancer cell line via activating E-cadherin expression by small activating RNA. Cancer Investig. 2010;28:1013–1018. doi: 10.3109/07357900802620844. [DOI] [PubMed] [Google Scholar]
- 70.Mao Q., Li Y., Zheng X., Yang K., Shen H., Qin J., Bai Y., Kong D., Jia X., Xie L. Up-regulation of E-cadherin by small activating RNA inhibits cell invasion and migration in 5637 human bladder cancer cells. Biochem. Biophys. Res. Commun. 2008;375:566–570. doi: 10.1016/j.bbrc.2008.08.059. [DOI] [PubMed] [Google Scholar]
- 71.Junxia W., Ping G., Yuan H., Lijun Z., Jihong R., Fang L., Min L., Xi W., Ting H., Ke D., et al. Double strand RNA-guided endogeneous E-cadherin up-regulation induces the apoptosis and inhibits proliferation of breast carcinoma cells in vitro and in vivo. Cancer Sci. 2010;101:1790–1796. doi: 10.1111/j.1349-7006.2010.01594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wang J., Place R.F., Huang V., Wang X., Noonan E.J., Magyar C.E., Huang J., Li L.C. Prognostic value and function of KLF4 in prostate cancer: RNAa and vector-mediated overexpression identify KLF4 as an inhibitor of tumor cell growth and migration. Cancer Res. 2010;70:10182–10191. doi: 10.1158/0008-5472.CAN-10-2414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Lin D., Meng L., Xu F., Lian J., Xu Y., Xie X., Wang X., He H., Wang C., Zhu Y. Enhanced wild-type p53 expression by small activating RNA dsP53-285 induces cell cycle arrest and apoptosis in pheochromocytoma cell line PC12. Oncol. Rep. 2017;38:3160–3166. doi: 10.3892/or.2017.5993. [DOI] [PubMed] [Google Scholar]
- 74.Zhao F., Pan S., Gu Y., Guo S., Dai Q., Yu Y., Zhang W. Small activating RNA restores the activity of the tumor suppressor HIC-1 on breast cancer. PLoS ONE. 2014;9:e86486. doi: 10.1371/journal.pone.0086486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Yang K., Shen J., Xie Y.Q., Lin Y.W., Qin J., Mao Q.Q., Zheng X.Y., Xie L.P. Promoter-targeted double-stranded small RNAs activate PAWR gene expression in human cancer cells. Int. J. Biochem. Cell Biol. 2013;45:1338–1346. doi: 10.1016/j.biocel.2013.03.022. [DOI] [PubMed] [Google Scholar]
- 76.Ren S., Kang M.R., Wang J., Huang V., Place R.F., Sun Y., Li L.C. Targeted induction of endogenous NKX3-1 by small activating RNA inhibits prostate tumor growth. Prostate. 2013;73:1591–1601. doi: 10.1002/pros.22709. [DOI] [PubMed] [Google Scholar]
- 77.Zeng T., Duan X., Zhu W., Liu Y., Wu W., Zeng G. SaRNA-mediated activation of TRPV5 reduces renal calcium oxalate deposition in rat via decreasing urinary calcium excretion. Urolithiasis. 2018;46:271–278. doi: 10.1007/s00240-017-1004-z. [DOI] [PubMed] [Google Scholar]
- 78.Kang M.R., Park K.H., Lee C.W., Lee M.Y., Han S.B., Li L.C., Kang J.S. Small activating RNA induced expression of VHL gene in renal cell carcinoma. Int. J. Biochem. Cell Biol. 2018;97:36–42. doi: 10.1016/j.biocel.2018.02.002. [DOI] [PubMed] [Google Scholar]
- 79.Xia W., Li D., Wang G., Ni J., Zhuang J., Ha M., Wang J., Ye Y. Small activating RNA upregulates NIS expression: Promising potential for hepatocellular carcinoma endoradiotherapy. Cancer Gene Ther. 2016;23:333–340. doi: 10.1038/cgt.2016.36. [DOI] [PubMed] [Google Scholar]
- 80.Kang M.R., Yang G., Place R.F., Charisse K., Epstein-Barash H., Manoharan M., Li L.C. Intravesical delivery of small activating RNA formulated into lipid nanoparticles inhibits orthotopic bladder tumor growth. Cancer Res. 2012;72:5069–5079. doi: 10.1158/0008-5472.CAN-12-1871. [DOI] [PubMed] [Google Scholar]
- 81.Reebye V., Sætrom P., Mintz P.J., Huang K.W., Swiderski P., Peng L., Liu C., Liu X., Lindkaer-Jensen S., Zacharoulis D., et al. Novel RNA oligonucleotide improves liver function and inhibits liver carcinogenesis in vivo. Hepatology. 2014;59:216–227. doi: 10.1002/hep.26669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Huan H., Wen X., Chen X., Wu L., Liu W., Habib N.A., Bie P., Xia F. C/EBPα Short-Activating RNA Suppresses Metastasis of Hepatocellular Carcinoma through Inhibiting EGFR/β-Catenin Signaling Mediated EMT. PLoS ONE. 2016;11:e0153117. doi: 10.1371/journal.pone.0153117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Wang L.L., Feng C.L., Zheng W.S., Huang S., Zhang W.X., Wu H.N., Zhan Y., Han Y.X., Wu S., Jiang J.D. Tumor-selective lipopolyplex encapsulated small active RNA hampers colorectal cancer growth in vitro and in orthotopic murine. Biomaterials. 2017;141:13–28. doi: 10.1016/j.biomaterials.2017.06.029. [DOI] [PubMed] [Google Scholar]
- 84.Yoon S., Huang K.W., Reebye V., Mintz P., Tien Y.W., Lai H.S., Sætrom P., Reccia I., Swiderski P., Armstrong B., et al. Targeted Delivery of C/EBPα -saRNA by Pancreatic Ductal Adenocarcinoma-specific RNA Aptamers Inhibits Tumor Growth In Vivo. Mol. Ther. 2016;24:1106–1116. doi: 10.1038/mt.2016.60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Li C., Jiang W., Hu Q., Li L.C., Dong L., Chen R., Zhang Y., Tang Y., Thrasher J.B., Liu C.B., et al. Enhancing DPYSL3 gene expression via a promoter-targeted small activating RNA approach suppresses cancer cell motility and metastasis. Oncotarget. 2016;7:22893–22910. doi: 10.18632/oncotarget.8290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Wang T., Li M., Yuan H., Zhan Y., Xu H., Wang S., Yang W., Liu J., Ye Z., Li L.C. saRNA guided iNOS up-regulation improves erectile function of diabetic rats. J. Urol. 2013;190:790–798. doi: 10.1016/j.juro.2013.03.043. [DOI] [PubMed] [Google Scholar]
- 87.Ma L., Jiang K., Jiang P., He H., Chen K., Shao J., Deng G. Mechanism of Notch1-saRNA-1480 reversing androgen sensitivity in human metastatic castration-resistant prostate cancer. Int. J. Mol. Med. 2020;46:265–279. doi: 10.3892/ijmm.2020.4597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Huang K.W., Reebye V., Czysz K., Ciriello S., Dorman S., Reccia I., Lai H.S., Peng L., Kostomitsopoulos N., Nicholls J., et al. Liver Activation of Hepatocellular Nuclear Factor-4α by Small Activating RNA Rescues Dyslipidemia and Improves Metabolic Profile. Mol. Ther. Nucleic Acids. 2020;19:361–370. doi: 10.1016/j.omtn.2019.10.044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhu Q., Wu X., Huang Y., Tang M., Wu L. Upregulation of FHIT gene expression in endometrial carcinoma by RNA activation. Int. J. Clin. Exp. Pathol. 2020;13:1372–1380. [PMC free article] [PubMed] [Google Scholar]
- 90.Wahlestedt C. Targeting long non-coding RNA to therapeutically upregulate gene expression. Nat. Rev. Drug Discov. 2013;12:433–446. doi: 10.1038/nrd4018. [DOI] [PubMed] [Google Scholar]
- 91.Poller W., Tank J., Skurk C., Gast M. Cardiovascular RNA interference therapy: The broadening tool and target spectrum. Circ. Res. 2013;113:588–602. doi: 10.1161/CIRCRESAHA.113.301056. [DOI] [PubMed] [Google Scholar]
- 92.Hedman M., Hartikainen J., Ylä-Herttuala S. Progress and prospects: Hurdles to cardiovascular gene therapy clinical trials. Gene Ther. 2011;18:743. doi: 10.1038/gt.2011.43. [DOI] [PubMed] [Google Scholar]
- 93.Cotrim A.P., Baum B.J. Gene therapy: Some history, applications, problems, and prospects. Toxicol. Pathol. 2008;36:97–103. doi: 10.1177/0192623307309925. [DOI] [PubMed] [Google Scholar]
- 94.Phillips A.J. The challenge of gene therapy and DNA delivery. J. Pharm. Pharmacol. 2001;53:1169–1174. doi: 10.1211/0022357011776603. [DOI] [PubMed] [Google Scholar]
- 95.Jafarlou M., Baradaran B., Saedi T., Jafarlou V., Shanehbandi D., Maralani M., Othman F. An overview of the history, applications, advantages, disadvantages and prospects of gene therapy. J. Biol. Regul. Homeost Agents. 2016;30:315–321. [PubMed] [Google Scholar]
- 96.Aldosari B.N., Alfagih I.M., Almurshedi A.S. Lipid Nanoparticles as Delivery Systems for RNA-Based Vaccines. Pharmaceutics. 2021;13:206. doi: 10.3390/pharmaceutics13020206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Owens D.E., Peppas N.A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 2006;307:93–102. doi: 10.1016/j.ijpharm.2005.10.010. [DOI] [PubMed] [Google Scholar]
- 98.Kaczmarek J.C., Patel A.K., Kauffman K.J., Fenton O.S., Webber M.J., Heartlein M.W., DeRosa F., Anderson D.G. Polymer-Lipid Nanoparticles for Systemic Delivery of mRNA to the Lungs. Angew. Chem. Int. Ed. Engl. 2016;55:13808–13812. doi: 10.1002/anie.201608450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Ickenstein L.M., Garidel P. Lipid-based nanoparticle formulations for small molecules and RNA drugs. Expert Opin. Drug Deliv. 2019;16:1205–1226. doi: 10.1080/17425247.2019.1669558. [DOI] [PubMed] [Google Scholar]
- 100.Watts J.K., Yu D., Charisse K., Montaillier C., Potier P., Manoharan M., Corey D.R. Effect of chemical modifications on modulation of gene expression by duplex antigene RNAs that are complementary to non-coding transcripts at gene promoters. Nucleic Acids Res. 2010;38:5242–5259. doi: 10.1093/nar/gkq258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Wahlestedt C., Salmi P., Good L., Kela J., Johnsson T., Hökfelt T., Broberger C., Porreca F., Lai J., Ren K., et al. Potent and nontoxic antisense oligonucleotides containing locked nucleic acids. Proc. Natl. Acad. Sci. USA. 2000;97:5633–5638. doi: 10.1073/pnas.97.10.5633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Veedu R.N., Wengel J. Locked nucleic acid as a novel class of therapeutic agents. RNA Biol. 2009;6:321–323. doi: 10.4161/rna.6.3.8807. [DOI] [PubMed] [Google Scholar]
- 103.Zhang Y., Qu Z., Kim S., Shi V., Liao B., Kraft P., Bandaru R., Wu Y., Greenberger L., Horak I. Down-modulation of cancer targets using locked nucleic acid (LNA)-based antisense oligonucleotides without transfection. Gene Ther. 2011;18:326. doi: 10.1038/gt.2010.133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Mook O., Vreijling J., Wengel S.L., Wengel J., Zhou C., Chattopadhyaya J., Baas F., Fluiter K. In vivo efficacy and off-target effects of locked nucleic acid (LNA) and unlocked nucleic acid (UNA) modified siRNA and small internally segmented interfering RNA (sisiRNA) in mice bearing human tumor xenografts. Artif. DNA PNA XNA. 2010;1:36–44. doi: 10.4161/adna.1.1.12204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Fluiter K., ten Asbroek A.L., de Wissel M.B., Jakobs M.E., Wissenbach M., Olsson H., Olsen O., Oerum H., Baas F. In vivo tumor growth inhibition and biodistribution studies of locked nucleic acid (LNA) antisense oligonucleotides. Nucleic Acids Res. 2003;31:953–962. doi: 10.1093/nar/gkg185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Dirin M., Winkler J. Influence of diverse chemical modifications on the ADME characteristics and toxicology of antisense oligonucleotides. Expert Opin. Biol. Ther. 2013;13:875–888. doi: 10.1517/14712598.2013.774366. [DOI] [PubMed] [Google Scholar]
- 107.Bennett C.F., Swayze E.E. RNA targeting therapeutics: Molecular mechanisms of antisense oligonucleotides as a therapeutic platform. Annu. Rev. Pharmacol. Toxicol. 2010;50:259–293. doi: 10.1146/annurev.pharmtox.010909.105654. [DOI] [PubMed] [Google Scholar]
- 108.Shim M.S., Kwon Y.J. Efficient and targeted delivery of siRNA in vivo. FEBS J. 2010;277:4814–4827. doi: 10.1111/j.1742-4658.2010.07904.x. [DOI] [PubMed] [Google Scholar]
- 109.Huang L., Liu Y. In vivo delivery of RNAi with lipid-based nanoparticles. Annu. Rev. Biomed. Eng. 2011;13:507–530. doi: 10.1146/annurev-bioeng-071910-124709. [DOI] [PubMed] [Google Scholar]
- 110.Juliano R., Carver K., Cao C., Ming X. Receptors, endocytosis, and trafficking: The biological basis of targeted delivery of antisense and siRNA oligonucleotides. J. Drug Target. 2013;21:27–43. doi: 10.3109/1061186X.2012.740674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Aghamiri S., Talaei S., Roshanzamiri S., Zandsalimi F., Fazeli E., Aliyu M., Kheiry Avarvand O., Ebrahimi Z., Keshavarz-Fathi M., Ghanbarian H. Delivery of genome editing tools: A promising strategy for HPV-related cervical malignancy therapy. Expert Opin. Drug Deliv. 2020;17:753–766. doi: 10.1080/17425247.2020.1747429. [DOI] [PubMed] [Google Scholar]
- 112.Yee F., Ericson H., Reis D.J., Wahlestedt C. Cellular uptake of intracerebroventricularly administered biotin- or digoxigenin-labeled antisense oligodeoxynucleotides in the rat. Cell Mol. Neurobiol. 1994;14:475–486. doi: 10.1007/BF02088832. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Rigo F., Hua Y., Krainer A.R., Bennett C.F. Antisense-based therapy for the treatment of spinal muscular atrophy. J. Cell Biol. 2012;199:21–25. doi: 10.1083/jcb.201207087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Vogel A.B., Kanevsky I., Che Y., Swanson K.A., Muik A., Vormehr M., Kranz L.M., Walzer K.C., Hein S., Güler A., et al. Immunogenic BNT162b vaccines protect rhesus macaques from SARS-CoV-2. Nature. 2021 doi: 10.1038/s41586-021-03275-y. [DOI] [PubMed] [Google Scholar]
- 115.Sahin U., Muik A., Derhovanessian E., Vogler I., Kranz L.M., Vormehr M., Baum A., Pascal K., Quandt J., Maurus D., et al. COVID-19 vaccine BNT162b1 elicits human antibody and T. Nature. 2020;586:594–599. doi: 10.1038/s41586-020-2814-7. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.