Abstract
Maternal nutrients are essential for proper fetal and placental development and function. However, the effects of vitamin and mineral supplementation under two rates of maternal weight gain on placental genome-wide gene expression have not been investigated so far. Furthermore, biological processes and pathways in the placenta that act in response to early maternal nutrition are yet to be elucidated. Herein, we examined the impact of maternal vitamin and mineral supplementation (from pre-breeding to day 83 post-breeding) and two rates of gain during the first 83 days of pregnancy on the gene expression of placental caruncles (CAR; maternal placenta) and cotyledons (COT; fetal placenta) of crossbred Angus beef heifers. We identified 267 unique differentially expressed genes (DEG). Among the DEGs from CAR, we identified ACAT2, SREBF2, and HMGCCS1 that underlie the cholesterol biosynthesis pathway. Furthermore, the transcription factors PAX2 and PAX8 were over-represented in biological processes related to kidney organogenesis. The DEGs from COT included SLC2A1, SLC2A3, SLC27A4, and INSIG1. Our over-representation analysis retrieved biological processes related to nutrient transport and ion homeostasis, whereas the pathways included insulin secretion, PPAR signaling, and biosynthesis of amino acids. Vitamin and mineral supplementation and rate of gain were associated with changes in gene expression, biological processes, and KEGG pathways in beef cattle placental tissues.
Keywords: caruncle, cotyledon, fetal programming, mineral, transcriptome, vitamin
1. Introduction
Maternal physiologic adaptation to pregnancy includes increased demand for nutrients to meet the maternal metabolic needs and nurture the developing fetus [1,2,3]. Furthermore, early gestation nutritional exposure affects the uterine environment and fetal development [4]. The fetomaternal interface provided by the placenta acts as a nutrient sensor to coordinate maternal nutrient supply and fetal metabolic requirements [5,6]. Thus, proper fetal development and nutrition are supported by an adequate nutrient supply through the placenta [7,8]. The placenta has many functions that include nutrient and waste product transport and hormone synthesis [6]. In ruminants, physiological exchanges between mother and fetus are supported by the caruncular-cotyledonary unit of the placenta, called the placentome [7]. While environmental stressors can trigger placental adaptations to meet fetal needs, these adjustments may affect fetal development and growth with long-lasting effects on metabolic function and performance [3,4,6,9,10].
Although growing evidence has shown the adverse effects of macronutrient (energy, protein, and fat) imbalances in fetoplacental development and function [11,12,13], less attention has been given to the role of micronutrients (vitamins and minerals) [4]. Vitamins and minerals play critical roles in animal health, growth, reproduction, and production, acting in structural, physiological, catalytic, and regulatory functions [14]. Maternal vitamin and mineral status has been suggested to affect hormonal regulatory pathways linking maternal metabolism with the fetoplacental unit [5,15]. Likewise, vitamins and minerals are required in biological processes such as energy metabolism, immune function, and gene expression [2,16,17]. Additionally, micronutrient-dependent enzymes that are involved with hormone synthesis and nutrient transport are present in the placenta [16]. While the placenta plays a pivotal role in mediating fetal needs, the changes and mechanisms associated with the placental adaptive responses to nutrient availability still need to be elucidated. Nonetheless, imprinted genes and differential gene expression are suggested as potential mechanisms that lead to placental plasticity [6].
Vitamins and minerals, as well as energy, protein, and fat are essential for proper fetal development and placental function. Lekatz et al. [18] reported hormonal and metabolic changes in pregnant ewes receiving supranutritional levels of selenium and/or nutrient restriction. Furthermore, changes in the expression of genes underlying angiogenesis and nutrient transport were identified in both caruncular and cotyledonary tissues [18]. In cows, myocardial necrosis and heart failure were identified in aborted selenium-deficient fetuses [19]. Maternal supplementation of organic trace minerals impacted offspring innate immune response and growth [20].
Despite the known effect of minerals on female reproductive traits and embryonic development [17], vitamin and mineral supplementation is still not widely adopted in cattle production [21]. Vitamins and minerals modulate the body’s energy homeostasis, and therefore, they are intricately related with the metabolism of carbohydrate, protein, and fat [14]. Furthermore, global nutrient restriction leads to reduced fetal growth and placental function [7]. Thus, a balanced diet that meets the requirements for macro and micronutrients is needed to optimize animal production. However, most studies focus on single nutrients without considering their multiple relationships on metabolic functions [22]. Similarly, the effects of vitamin and mineral supplementation on placental genome-wide gene expression have not yet been investigated.
Energy and protein availability are also essential for proper placental functions. Considering that nutrient requirements for first-calf heifers are greater than mature cows [23], it has been suggested that increasing nutrient supply through targeted supplementation strategies may optimize pregnancy rates, maternal-fetal transfer of nutrients, and increase offspring birth weights [24,25]. Herein, we tested the hypothesis that supplementing vitamin and minerals pre- and post-breeding to beef heifers grown at a low or moderate rate of maternal weight gain during the first trimester of gestation would affect placental gene expression. Therefore, we measured the gene expression profiles of maternal (caruncular; CAR) and fetal (cotyledonary; COT) portions of the placenta to identify differentially expressed genes (DEG), biological processes (BP), and pathways underlying placental development and function in response to early maternal nutrition. We identified 267 unique DEGs throughout all tissues and group contrasts. The over-represented functions are essential to fetal growth and development, and included fatty acid metabolism, hormone synthesis, nutrient transport, energy metabolism, and biosynthesis of amino acids.
2. Materials and Methods
All experiments and methods were performed following the relevant guidelines and regulations. The experimental design, animal management, and tissue collection were approved by the North Dakota State University Institutional Animal Care and Use Committee (IACUC A19012).
2.1. Animals, Experimental Design, and Tissue Collection
Angus-cross heifers (n = 35) were randomly assigned by initial body weight ( = 359.5 ± 7.1 kg) to a 2 × 2 factorial arrangement of treatments. The factors examined included vitamin and mineral supplementation (VTM or NoVTM) and rate of gain (low gain (LG) or moderate gain (MG)). The treatments were arranged as follows: (1) no vitamin and mineral supplementation and low gain (NoVTM_LG, n = 9); (2) vitamin and mineral supplementation and low gain (VTM_LG, n = 9); (3) no vitamin and mineral supplementation and moderate gain (NoVTM_MG, n = 9), and (4) vitamin and mineral supplementation and moderate gain (VTM_MG, n = 8).
Diets were delivered once daily via a total mixed ration and consisted of triticale hay, corn silage, modified distillers’ grains plus solubles, ground corn, and if indicated by treatment, mineral premix. To achieve MG (0.79 kg/d), heifers were fed the total mixed ration with the addition of the starch-based protein/energy supplement (a blend of ground corn, dried distillers’ grains plus solubles, wheat midds, fish oil, urea, and ethoxyquin). The LG heifers were maintained on the basal total mixed ration and targeted to gain 0.28 kg/d. Based on the National Research Council [26], the total mixed ration provided 105%, 158%, 215%, and 250% of the mineral requirements for NoVTM_LG, NoVTM_MG, VTM_LG, and VTM_MG treatments, respectively. Diet composition is described in Supplementary Materials, Table S1. The two rates of gain and VTM levels supplied to the heifers were chosen to represent two nutritional states (weight gain versus maintenance) as well as conditions applicable to beef production systems.
The VTM treatment started 71 to 148 days before artificial insemination by providing 0.45 kg/heifer daily of a ground corn and vitamin and mineral premix (113 g·heifer−1·d−1 of Purina Wind & Rain Storm All-Season 7.5 Complete, Land O’Lakes, Inc., Arden Hills, MN, USA). Based on the VTM starting date, heifers were assigned to one of seven breeding groups so that the supplementation period was at least 60 days for all. At breeding, heifers were randomly assigned to either LG or MG treatments within their respective VTM treatment. Heifers were bred by artificial insemination using female-sexed semen from a single sire. Pregnancy diagnosis was performed 35 days after artificial insemination, and fetal sex was determined on day 65 using transrectal ultrasonography. Further details of animal management were described elsewhere [27].
The VTM and rate of gain treatments were carried out until day 83 ± 0.27 of gestation, when uteroplacental tissues were collected through ovariohysterectomy [28]. The largest placentome closest to the fetus was collected and maternal (CAR) and fetal (COT) portions were manually dissected [29], snap-frozen, and stored at −80 °C.
2.2. Total RNA Isolation, Library Preparation, Sequencing, and Data Analysis
Total RNA of eight female samples per treatment was isolated from the CAR and COT tissues using the RNeasy® kit (Qiagen®, Germantown, MA, USA) followed by on-column DNase treatment, according to the manufacturer’s protocol. Sample integrity and purity were evaluated using the Agilent 2100 Bioanalyzer and agarose gel electrophoresis. Strand-specific RNA libraries were prepared using the NEBNext® Ultra™ II Directional RNA Library Prep Kit for Illumina (New England BioLabs®, Ipswich, MA, USA), and sequencing was carried out on the Illumina® NovaSeq 600 platform. Library preparation and paired-end sequencing with 150-bp reads at a depth of 20 M reads/sample were carried out at Novogene Co. (Nanjing, China).
Sequencing adaptors, low-complexity reads, and reads containing low quality bases were removed in an initial data-filtering step. Reads with a PhredScore lower than 30 were filtered out. Quality control (QC) and read statistics were estimated with FastQC v0.11.8 [30] and MultiQC v1.9 [31] software. After QC, 29 and 31 samples (seven or eight samples per group) remained for further analyses from CAR and COT, respectively. Reads were mapped to the Bos taurus reference genome (ARS-UCD 1.2) [32] using the STAR aligner v. 2.7.3a [33]. Raw counts per gene were obtained using the –quantMode GeneCounts flag from STAR based on the gene annotation file (release 100, Ensembl). MultiQC, NOISeq [34], and edgeR [35] software were used to perform the post-mapping quality control.
2.3. Differential Expression and Functional Over-Representation Analyses
Genes with expression values lower than 1 count per million in 50% of the samples were filtered out. After filtering, the genes of CAR and COT tissues were analyzed using the DESeq2 v.1.22.1 R-package [36] to identify DEGs. The median of ratios method from DESeq2 was employed to normalize the data for sequencing depth and RNA composition [36]. The differential expression analysis used the negative binomial generalized linear model to fit gene expression level as a negative binomial distribution and Wald statistics to perform hypothesis testing [36]. The svaseq function of the R-package Surrogate Variable Analysis v.3.30.0 [37] was adopted to estimate unknown sources of variation in the RNA-Seq data. The DESEq2 model was used to measure the treatment effect while controlling for batch effect differences that included the surrogated variables and the heifer’s birthplace (farm of origin). To make all pair-wise comparisons between the four treatment groups, six contrasts were created as follows: (1) VTM_MG vs. NoVTM_LG, (2) VTM_MG vs. VTM_LG, (3) VTM_MG vs. NoVTM_MG, (4) VTM_LG vs. NoVTM_LG, (5) VTM_LG vs. NoVTM_MG, (6) NoVTM_MG vs. NoVTM_LG. Multiple testing adjustment of the p-values (padj) was performed using the Benjamini–Hochberg procedure for false discovery rate (FDR) [38]. Genes were identified as differentially expressed for each one of the contrasts when the false-discovery rate adjusted p-value (padj) cutoff ≤ 0.1 [11] and classified as up- or down-regulated based on the sign of the log2 fold change. The threshold (padj < 0.1) was defined a priori based on our experimental design. Furthermore, we used stringent quality control to remove lowly expressed genes and reduce the number of false-positive genes tested. As these are exploratory analyses, this combined approach allowed us to identify significant biological processes, while avoiding losing too much information.
Gene functional over-representation analysis was carried out using the ShinyGo v0.61 webtool [39] and the B. taurus annotation as background. This approach identified specific and common biological functions and KEGG pathways within and among gene lists for each tissue and contrast. Significant results after multiple testing adjustments were considered with an FDR ≤ 0.05.
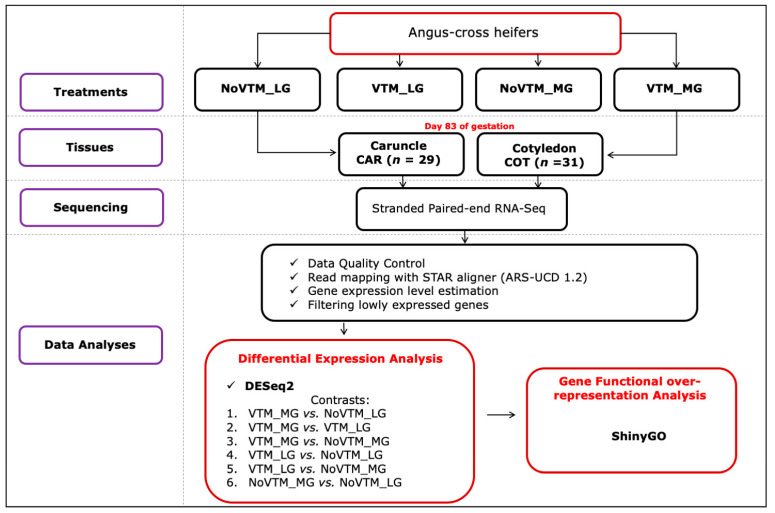
An overview of the experimental design and data analyses pipeline is presented in Figure 1.
Figure 1.
Experimental design and bioinformatics pipeline of the RNA-Seq-based differential expression analysis of bovine placental tissues—caruncle (CAR) and cotyledon (COT). The treatments were arranged as follows: NoVTM_LG—no vitamin and mineral supplementation and low gain; VTM_LG—vitamin and mineral supplementation and low gain; NoVTM_MG—no vitamin and mineral supplementation and moderate gain; and VTM_MG—vitamin and mineral supplementation and moderate gain.
3. Results
We applied an RNA-Seq-based approach to identify differentially expressed genes in maternal (CAR) and fetal (COT) placental tissues of beef heifers subjected to vitamin and mineral supplementation and two rates of gain. On average, the sequencing of the tissues generated 22.7 M reads through the 60 samples with PhredScore > 30. The sequencing throughput and mapping rates per sample and tissue are reported in Table S2. On average, 97.0% and 96.2% of the reads from CAR and COT, respectively, were uniquely mapped to genes in the bovine reference genome (Table S2). After filtering, 13,252 genes from CAR and 12,795 from COT were analyzed to identify DEGs.
3.1. Differentially Expressed Genes
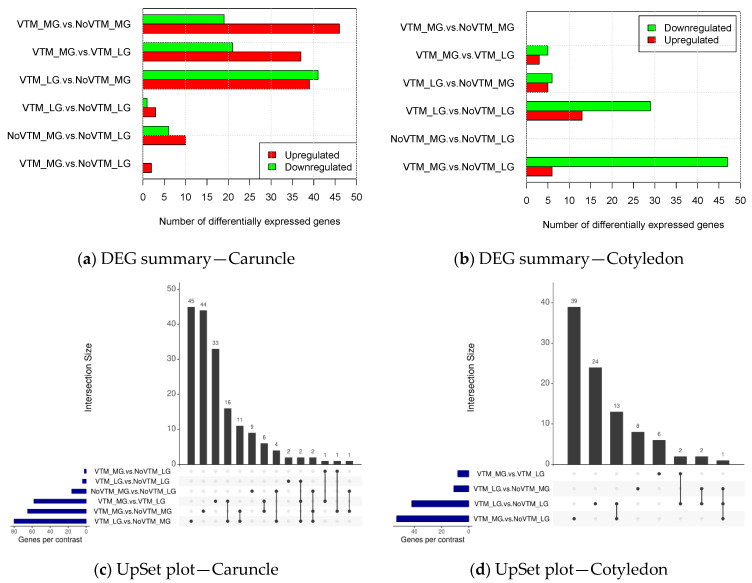
We identified 267 unique DEGs (padj ≤ 0.1) throughout all tissues and group comparisons. For the CAR tissue, gene expression analysis revealed 137 upregulated and 88 downregulated genes (Figure 2a), whereas in COT, 27 and 87 genes were upregulated or downregulated, respectively (Figure 2b). Our approach did not find significant DEGs for COT when comparing VTM_MG vs. NoVTM_MG and NoVTM_MG vs. NoVTM_LG.
Figure 2.
Differential gene expression summary of bovine placental tissues—caruncle (maternal placenta) (a) and cotyledon (fetal placenta) (b) (padj ≤ 0.1). The UpSet plot represents the intersection between the sets of differentially expressed genes (DEGs) from different contrasts of caruncle (c) and cotyledon (d). Each vertical bar shows the number of genes in the intersection. The dot plot reports the set participation in the intersection, and the horizontal bar graph reports the set sizes (total of DEGs). The treatments were arranged as follows: NoVTM_LG—no vitamin and mineral supplementation and low gain; VTM_LG—vitamin and mineral supplementation and low gain; NoVTM_MG—no vitamin and mineral supplementation and moderate gain; and VTM_MG—vitamin and mineral supplementation and moderate gain.
The overlap between the sets of DEGs identified by the different contrasts are shown in Figure 2c,d. For CAR, we observed the greatest number of shared genes (n = 18) between the contrasts VTM_MG vs. VTM_LG and VTM_LG vs. NoVTM_MG. In the COT tissue, the VTM_LG vs. NoVTM_LG and VTM_MG vs. NoVTM_LG contrasts shared 14 genes between one another. When we compared the DEGs across tissues, most of them were tissue-specific, with only five genes shared between CAR and COT. The common DEGs between CAR and COT were: DNMT3B, ESYT3, PRPFB1, FADS1, and TTC7A. The DEGs are reported for each of the significant contrasts along with the fold-change values and annotation in Table S3 (CAR) and Table S4 (COT).
3.2. Functional Over-Representation Analysis
We retrieved significant biological processes (BP) and KEGG pathways by querying the DEGs of each contrast using the ShinyGO tool. Our approach identified 15 and 20 KEGG pathways from CAR and COT, respectively, that were over-represented by the DEGs (FDR ≤ 0.05). Likewise, 76 and 49 gene ontology BP terms were identified from CAR and COT, respectively (FDR ≤ 0.05). Figure 3 shows BP and KEGG pathways that were over-represented from DEGs of the VTM_MG vs. VTM_LG and VTM_MG vs. NoVTM_LG comparisons. The BP underlying the DEGs from CAR (Figure 3a) included, for example, regulation of molecular function and catalytic activity, organ morphogenesis and development (especially kidney and ureter). For CAR, among the over-represented pathways (Figure 3b) were the fatty acid metabolism, steroid biosynthesis, and terpenoid backbone biosynthesis. Due to the reduced number of DEGs, the contrasts NoVTM_MG vs. NoVTM_LG, VTM_MG vs. NoVTM_LG, and VTM_MG vs. NoVTM_LG from CAR did not retrieve any significantly enriched BP or KEGG pathways. Regarding COT, BP underlying the DEGs (Figure 3c) included metal ion homeostasis, ion transport, and regulation of developmental processes and response to stress, whereas the over-represented pathways (Figure 3d) included PPAR signaling, thyroid hormone signaling, adipocytokine signaling, insulin resistance, HIF-1 signaling, and cysteine and methionine metabolism. Biological processes and pathways identified for all the comparisons are provided in Tables S3 and S4.
Figure 3.
Functional over-representation analysis of differentially expressed genes (DEGs) from bovine placental tissues—caruncle (maternal placenta) and cotyledon (fetal placenta). Biological processes and KEGG pathways over-represented from DEGs in the caruncle (a,b) and cotyledon (c,d). The terms are hierarchically arranged based on functional similarity. The bigger the blue dot, the more significant the term is (FDR ≤ 0.05).
4. Discussion
Nutrient demand increases throughout gestation to meet the requirements of the developing fetus. Growing evidence has shown that poor maternal nutrition, including vitamin and mineral deficiency, has adverse impacts on early placental development, with long-lasting effects on fetal programming of pre and postnatal growth and development [2,3,4,16]. In this study, we examined the impact of maternal vitamin and mineral supplementation (from pre-breeding to day 83) and two rates of gain (low or moderate) during the first 83 days of pregnancy on the gene expression of maternal (CAR) and fetal (COT) placental tissues. Our findings demonstrate that vitamin and mineral supplementation and rate of gain led to differential gene expression of CAR and COT tissues. However, the effect of rate of gain seems to be stronger on the maternal side, as for COT, few or no genes were identified as differentially expressed for the comparisons VTM_MG vs. VTM_LG and NoVTM_MG vs. NoVTM_LG. These findings suggest potential placental adaptations in response to maternal vitamin and mineral supplementation and rate of gain as indicated by the over-represented biological processes and pathways.
While our model achieved the targeted rates of gain (as designed), we did not find significant differences in fetal size or gravid uterine weight among the treatments [27]. On the other hand, fetal liver weight was greater (p-value = 0.05) from dams fed VTM than NoVTM [27]. Likewise, amino acid concentrations of maternal serum and allantoic and amniotic fluid in the same samples used in the current study were affected by vitamin and mineral supplementation and/or rate of gain [25]. Evidence suggests that biological mechanisms regulating normal growth, development, and nutrient utilization are programmed in utero for postnatal growth and adult function even during the earliest stages of development [40]. Additionally, large amounts of epidemiological data have shown that an impaired intrauterine environment has long-term consequences (reviewed in [4,41]). The key role performed by the placenta is mediated by changes in the gene expression that leads to differential programming of fetal tissues. Thus, imbalances in maternal vitamin and mineral availability during critical windows of development play a role in fetal tissue development as observed in fetal liver size.
Although no signs of vitamin and mineral deficiency or overload were observed in these pregnant beef heifers, changes in gene expression and concentrations of amino acids in fetal fluids suggest physiological adaptations to meet the fetal and maternal metabolic needs. Furthermore, the changes in gene expression of CAR seem to be greater than in COT as more DEGs were identified and were mainly upregulated. We know that changes in maternal metabolism are sensed by the placenta to meet fetal nutrient requirements based on the maternal resources available [5,42]. Thus, the dam may insulate the fetus against short-term nutrient imbalances by using their body reserves to sustain fetal growth [5]. Alternatively, the fetal placenta (COT) has specific homeostatic mechanisms to insulate the fetus, as suggested by the differential expression of glucose transporter genes.
4.1. Pathways Underlying Caruncular Differential Gene Expression
The placenta can adapt its capacity to supply nutrients in response to insults in the maternal-fetal environment [6]. Here, we found that vitamin and mineral supplementation combined with low or moderate gain affected pathways related to energy metabolism. Additionally, several BP and KEGG pathways underlying fatty acid metabolism, hormone biosynthesis, and amino acid degradation were identified in CAR. These are processes that require vitamins and minerals as structural components or enzymatic co-factors [14].
Underlying the fatty acid metabolism pathway, we identified FADS1 and ACAT2 as DEG for CAR. The FADS1 gene codes for a rate-limiting enzyme involved with the metabolism and degradation of polyunsaturated fatty acids, such as docosahexaenoic acid and arachidonic acid [43]. Likewise, the protein encoded by ACAT2 acts in lipid biosynthesis and regulates the synthesis of cholesteryl ester [44]. Additional genes related with cholesterol metabolism include SREBF2, which was upregulated in the VTM_LG vs. NoVTM_MG and downregulated in VTM_MG vs. VTM_LG in CAR. Sterol regulatory element-binding proteins (SREBPs) are transcription factors involved in cholesterol homeostasis and fatty acid uptake [45]. Steroid biosynthesis and sterol metabolic process were over-represented in our functional analysis of DEGs in CAR. Among the differentially expressed sterol-regulated genes underpinning cholesterol biosynthesis, we identified HMGCS1, FDFT1, MSMO1, and SQLE downregulated in VTM_MG vs. VTM_LG in CAR. Cholesterol is important for fetal development as a component in the cell membranes of the growing placenta and fetus [46]. Furthermore, cholesterol is the precursor of all steroid hormones, such as progesterone, that are required for normal gestation and fetal development [46]. The production of estrogens from cholesterol is supported by the enzymes present in the bovine trophoblast. According to Schuler et al. [47], the estrogen synthesized in the trophoblast suggests a role as local regulator of caruncular growth to produce a histotroph-like cell detritus. The histotroph in turn serves as an important source of nutrients for the fetus.
From the contrast between VTM_MG vs. NoVTM_MG, we identified the genes CALM2, ATP2B4, CAMK2G, and BDKRB2 as over-represented in the calcium signaling and cyclic guanosine monophosphate (CGMP)-PKG signaling pathways. Calcium is not only essential for fetal development but also is an intracellular messenger that regulates, for example, gene transcription and cell proliferation [48]. Furthermore, calcium-mediated systems may activate steroidogenic activity of bovine placentomes [49]. The cGMP-PKG pathway plays a key role in vascular homeostasis and is mediated by nitric oxide and decreased calcium concentrations [50,51]. Previous studies have shown that maternal dietary treatments may impact placental vascularity and uterine blood flow [7,10,29]. Although we have not measured vascular development in the current study, we identified blood circulation, smooth muscle contraction, and circulatory system processes among the over-represented BP in CAR of VTM-supplemented heifers. Despite the lack of information regarding the role of vitamins and minerals in “driving” the increase in placental vascularity in bovine, Gernand et al. [16] reported that the human placenta is rich in micronutrient-dependent antioxidant enzymes that support normal maternal-fetal circulation. Moreover, vitamins E and D are suggested to enhance the expression of angiogenic factors in the placenta [52,53].
Interestingly, we identified several BP related to kidney morphogenesis, which were over-represented among the DEGs from the VTM_MG vs. VTM_LG contrast. The PAX2 and PAX8 genes were among DEGs in the BP such as kidney epithelium development and metanephros morphogenesis. These genes encode transcription factors that orchestrate kidney development, which is important for regulation of cardiovascular function, including blood pressure, later in life [54]. Additionally, the protein encoded by PAX8 plays a key role in the development of other organs and tissues by interacting with the WT1 transcription factor, which has an essential role in the normal development of the urogenital system [55]. According to Christian et al. [15], changes in micronutrient availability may lead to hormonal adaptations, and consequently, affect kidney development and function. Moreover, Mao et al. [56] reported changes in the expression of placental genes that were involved with kidney function in mice fed high-fat or low-fat diets.
4.2. Pathways Underlying Cotyledonary Differential Gene Expression
The coordinated development and function between the CAR and COT placental tissues is responsible for providing the fetus with nutrients to support its metabolic demands [57]. Nonetheless, under nutritional stress, the placenta may increase the number and the surface area of cotyledons to improve the efficiency of placental transport [58]. These adjustments are not only related to placental vascular growth and angiogenesis [7,57] but also to the regulation of genes encoding for nutrient transporters [59]. As proposed by the placental nutrient sensing or fetal demand models [59,60], different mechanisms and placental responses underlie fetal-maternal nutrient cross-talk. Based on these models, maternal downregulation of genes encoding for nutrient transporters may lead to upregulation of fetal genes, and vice-versa, to balance maternal nutrient availability and fetal nutrient demand [59]. We observed that most of the nutrient transport DEGs from COT were classified as downregulated in the supplemented groups. In light of the above-mentioned models, this may suggest that the fetuses from supplemented dams met their nutritional requirements, whereas the non-supplemented fetuses optimized nutrient transport by upregulating gene expression. Furthermore, the rate of gain (i.e., maternal dietary intake to support a moderate or low rate of gain) seems to not affect COT gene expression under the conditions tested in the current study. According to Thayer et al. [5], the dam may homeostatically regulate macronutrient availability by mobilizing the maternal body’s reserve to supply the fetus. On the other hand, the authors argue that the body’s available store of micronutrients, such as vitamins and minerals, is limited, which may compromise their supply to the fetus when the maternal diet is micronutrient restricted [5].
Placental nutrient transporters are important for delivering nutrients such as glucose, amino acids, and fatty acids to the fetus [61]. Biological processes related to nutrient transport and ion transport were over-represented in our findings. Pathways related to insulin secretion and resistance, biosynthesis of amino acids, and PPAR signaling underlie the DEGs from the VTM_MG vs. NoVTM_LG groups in COT. Insulin is a potent hormone involved with energy metabolism and is essential for regulating glucose uptake and its serum levels [62]. Due to their role as cofactors in metabolic pathways, some minerals have been suggested to enhance insulin action [2]. For example, chromium improves glucose homeostasis through increased insulin sensitivity [63]. Glucose is the primary metabolic fuel for fetal metabolism, and it is crucial for fetal development [59]. Batistel et al. [64] reported that methionine supplementation during late gestation changed the expression profile of genes related to transport of amino acids, fatty acids, glucose, and vitamins of placentomes from dairy cows. Among the DEGs encoding glucose transporters, we identified SLC2A1 (GLUT1) and SLC2A3 (GLUT3). We also identified the INSIG1 gene as differentially expressed. The protein encoded by INSIG1 regulates cholesterol metabolism, lipogenesis, and glucose homeostasis. Furthermore, INSIG1 controls cholesterol synthesis through the SREBP and HMGCS1 proteins [65].
In addition to glucose, fatty acids are an important source of energy for placental function and fetal growth [66]. According to Lewis et al. [67], fatty acids are the precursors for PPAR transcription factors. PPARs are nuclear hormone receptors that are active in embryonic development and tissue differentiation through regulation of gene expression [66]. In the PPAR signaling pathway, we identified ACSL3, SLC27A4, and PLIN2 as over-represented DEGs for COT from the contrast VTM_MG vs. NoVTM_LG. The ACSL3 and SLC27A4 genes encode proteins that are able to activate long chain fatty acids [68]. According to Nakahara et al. [69], the ACSL3 protein is involved with fatty acids uptake for synthesis of cellular lipids and degradation via beta-oxidation, while SLC27A4 acts in fatty acids transport [59]. The gene PLIN2 is important for trophoblastic lipid droplet accumulation [70]. We identified BP related to lipid biosynthesis and metabolism among the DEGs from the VTM_MG vs. VTM_LG comparison as well. The DEGs play roles in sphingolipid biosynthesis (DEGS2) [71] and phospholipid biosynthesis and remodeling (LPCAT1) of the lipid droplets [72].
Among the DEGs, we found that AARS1, IARS1, GARS1, and NARS2 were upregulated in the VTM_LG vs. NoVTM_LG comparison in COT. These genes were over-represented in the aminoacyl-tRNA biosynthesis pathway, and they encode key enzymes required for protein biosynthesis [73]. Furthermore, the ARG2 and MTR genes were downregulated in the VTM_MG vs. NoVTM_LG comparison, and these genes are involved in the biosynthesis of amino acids. The ARG2 protein is involved in the conversion of L-arginine into L-ornithine, which is a precursor to polyamines that support cell proliferation [74], whereas MTR catalyzes the final step in methionine biosynthesis [75]. Menezes et al. [25] reported increased concentrations of methionine and arginine in allantoic fluid in response to vitamin supplementation and a moderate rate of gain when evaluating the pregnant heifers used in the current study, which further supports the current findings.
5. Conclusions
By applying a genome-wide transcriptomic analysis, we identified genes differentially expressed from caruncular and cotyledonary placental tissues of pregnant heifers in response to maternal nutrition. Vitamin and mineral supplementation and low or moderate rate of gain are associated with changes in gene expression in placental tissues. Functional analysis of DEGs pointed out the pathways underlying energy metabolism, hormone synthesis, and nutrient transport. These findings shed light on the mechanisms via which maternal nutrition may regulate placental function and, potentially, fetal growth and development. Furthermore, our findings, for the first time, unravel the putative placental adaptations in response to maternal vitamin and mineral supplementation from pre-breeding through to the first trimester (until day 83) of gestation.
Acknowledgments
The authors would like to thank Purina Animal Nutrition LLC (Land O’Lakes, Inc., Arden Hills, MN, USA) for providing financial support for this research. Authors would also like to thank the North Dakota State Board of Agricultural Research and Education, Graduate Research Assistantship Program, and the North Dakota Agricultural Experiment Station for their support of this effort, and additional product support from Zoetis Animal Health (Parsnippany, NJ, USA) and ST Genetics (Navasota, TX, USA). Appreciation is expressed to personnel at the Central Grasslands Research Extension Center and the Animal Nutrition and Physiology Center for assistance with animal handling and feeding, and the NDSU Animal Science Nutrition Laboratory. This work used resources of the Center for Computationally Assisted Science and Technology (CCAST) at North Dakota State University.
Supplementary Materials
The following are available online at https://www.mdpi.com/2073-4425/12/3/385/s1, Table S1: Nutrient composition of total mixed ration and supplements provided to beef heifers; Table S2: Caruncular and cotyledonary RNA sequencing summary and mapping statistics; Table S3: Supplementary information related to the differentially expressed genes, biological processes, and pathways from caruncular tissue; Table S4: Supplementary information related to the differentially expressed genes, biological processes, and pathways from cotyledonary tissue.
Author Contributions
Conceptualization, C.R.D., K.L.M., J.S.C., J.C.F. and R.R.S.; Animal management, K.L.M., C.J.K., F.B., J.D.K. and K.K.S.; sample collection; L.P.R., P.P.B., A.K.W., T.L.N. and S.T.D.; RNA isolation, S.T.D.; bioinformatic analysis, W.J.S.D., writing—original draft preparation, W.J.S.D.; writing—review and editing, all authors; supervision, A.K.W. and C.R.D.; funding acquisition, L.P.R., C.R.D., K.L.M. and J.S.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the North Dakota State Board of Agricultural Research and Education, grant number 19-23-0155, and by Purina Animal Nutrition LLC, Gray Summit, MO, USA.
Institutional Review Board Statement
All experiments and methods were performed following the relevant guidelines and regulations. The experimental design, animal management, and tissue collection were approved by the North Dakota State University Institutional Animal Care and Use Committee (IACUC A19012).
Informed Consent Statement
Not applicable.
Data Availability Statement
All relevant data are within the paper and its Supplementary Information files. All sequencing data is publicly available on NCBI’s Gene Expression Omnibus (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE165378).
Conflicts of Interest
Authors J. C. Forcherio and R. Scott are employees of Purina Animal Nutrition LLC (Land O’Lakes, Inc., Arden Hills, MN, USA), which sponsored the sample analysis for this experiment. Purina Animal Nutrition LLC manufactured the Purina® Wind & Rain® Storm® All-Season 7.5 Complete mineral, the VTM and NoVTM pellets, and the protein/energy supplement used in this study. The funders had no role in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results that were done entirely independently by North Dakota State University personnel. The first author led the writing of the paper with inputs from the co-authors, who declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.King J.C. Physiology of pregnancy and nutrient metabolism. Am. J. Clin. Nutrit. 2000;71:1218S–1225S. doi: 10.1093/ajcn/71.5.1218s. [DOI] [PubMed] [Google Scholar]
- 2.Khayat S., Fanaei H., Ghanbarzehi A. Minerals in pregnancy and lactation: A review article. J. Clin. Diagn. Res. 2017;11:QE01–QE05. doi: 10.7860/JCDR/2017/28485.10626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Caton J.S., Crouse M.S., Reynolds L.P., Neville T.L., Dahlen C.R., Ward A.K., Swanson K.C. Maternal nutrition and programming of offspring energy requirements. Transl. Anim. Sci. 2019;3:976–990. doi: 10.1093/tas/txy127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Reynolds L.P., Ward A.K., Caton J.S. Epigenetics and developmental programming in ruminants: Long-term impacts on growth and development. In: Scanes C.G., Hill R.A., editors. Biology of Domestic Animals. CRC Press; Boca Raton, FL, USA: 2017. pp. 85–120. A science publisher’s book. [Google Scholar]
- 5.Thayer Z.M., Rutherford J., Kuzawa C.W. The maternal nutritional buffering model: An evolutionary framework for pregnancy nutritional intervention. Evol. Med. Public Health. 2020;2020:14–27. doi: 10.1093/emph/eoz037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sandovici I., Hoelle K., Angiolini E., Constância M. Placental adaptations to the maternal-fetal environment: Implications for fetal growth and developmental programming. Reprod. Biomed. Online. 2012;25:68–89. doi: 10.1016/j.rbmo.2012.03.017. [DOI] [PubMed] [Google Scholar]
- 7.Vonnahme K.A., Lemley C.O., Caton J.S., Meyer A.M. Impacts of maternal nutrition on vascularity of nutrient transferring tissues during gestation and lactation. Nutrients. 2015;7:3497–3523. doi: 10.3390/nu7053497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Lemley C.O., Hart C.G., Lemire R.L., Heath King E., Hopper R.M., Park S.B., Rude B.J., Burnett D.D. Maternal nutrient restriction alters uterine artery hemodynamics and placentome vascular density in Bos indicus and Bos taurus. J. Anim. Sci. 2018;96:4823–4834. doi: 10.1093/jas/sky329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Grazul-Bilska A.T., Gaton J.S., Arndt W., Burchill K., Thorson C., Borowczyk E., Bilski J.J., Redmer D.A., Reynolds L.P., Vonnahme K.A. Cellular proliferation and vascularization in ovine fetal ovaries: Effects of undernutrition and selenium in maternal diet. Reproduction. 2009;137:699–707. doi: 10.1530/REP-08-0375. [DOI] [PubMed] [Google Scholar]
- 10.Grazul-Bilska A.T., Borowicz P.P., Johnson M.L., Minten M.A., Bilski J.J., Wroblewski R., Redmer D.A., Reynolds L.P. Placental development during early pregnancy in sheep: Vascular growth and expression of angiogenic factors in maternal placenta. Reproduction. 2010;140:165–174. doi: 10.1530/REP-09-0548. [DOI] [PubMed] [Google Scholar]
- 11.Crouse M.S., Caton J.S., Cushman R.A., McLean K.J., Dahlen C.R., Borowicz P.P., Reynolds L.P., Ward A.K. Moderate nutrient restriction of beef heifers alters expression of genes associated with tissue metabolism, accretion, and function in fetal liver, muscle, and cerebrum by day 50 of gestation. Transl. Anim. Sci. 2019;3:855–866. doi: 10.1093/tas/txz026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Long N.M., Vonnahme K.A., Hess B.W., Nathanielsz P.W., Ford S.P. Effects of early gestational undernutrition on fetal growth, organ development, and placentomal composition in the bovine. J. Anim. Sci. 2009;87:1950–1959. doi: 10.2527/jas.2008-1672. [DOI] [PubMed] [Google Scholar]
- 13.Paradis F., Wood K.M., Swanson K.C., Miller S.P., McBride B.W., Fitzsimmons C. Maternal nutrient restriction in mid-to-late gestation influences fetal mRNA expression in muscle tissues in beef cattle. BMC Genom. 2017;18:1–14. doi: 10.1186/s12864-017-4051-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Suttle N. In: Mineral Nutrition of Livestock. 4th ed. Suttle N., editor. CABI; Wallingford, UK: 2010. [Google Scholar]
- 15.Christian P., Stewart C.P. Maternal micronutrient deficiency, fetal development, and the risk of chronic disease. J. Nutr. 2010;140:437–445. doi: 10.3945/jn.109.116327. [DOI] [PubMed] [Google Scholar]
- 16.Gernand A.D., Schulze K.J., Stewart C.P., West K.P., Christian P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention HHS Public Access. Nat. Rev. Endocrinol. 2016;12:274–289. doi: 10.1038/nrendo.2016.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Keshri A., Bashir Z., Kumari V., Prasad K., Joysowal M., Singh M., Singh D., Tarun A., Shukla S. Role of micronutrients during peri-parturient period of dairy animals–A review. Biol. Rhythm Res. 2019 doi: 10.1080/09291016.2019.1613793. [DOI] [Google Scholar]
- 18.Lekatz L.A., Caton J.S., Taylor J.B., Reynolds L.P., Redmer D.A., Vonnahme K.A. Maternal selenium supplementation and timing of nutrient restriction in pregnant sheep: Effects on maternal endocrine status and placental characteristics. J. Anim. Sci. 2010;88:955–971. doi: 10.2527/jas.2009-2152. [DOI] [PubMed] [Google Scholar]
- 19.Orr J.P., Blakley B.R. Investigation of the selenium status of aborted calves with cardiac failure and myocardial necrosis. J. Vet. Diagn. Investig. 1997;9:172–179. doi: 10.1177/104063879700900211. [DOI] [PubMed] [Google Scholar]
- 20.Jacometo C.B., Osorio J.S., Socha M., Corrêa M.N., Piccioli-Cappelli F., Trevisi E., Loor J.J. Maternal consumption of organic trace minerals alters calf systemic and neutrophil mRNA and microRNA indicators of inflammation and oxidative stress. J. Dairy Sci. 2015;98:7717–7729. doi: 10.3168/jds.2015-9359. [DOI] [PubMed] [Google Scholar]
- 21.Davy J.S., Forero L.C., Shapero M.W.K., Rao D.R., Becchetti T.A., Koopman Rivers C., Stackhouse J.W., Deatley K.L., McNabb B.R. Mineral status of California beef cattle. Transl. Anim. Sci. 2019;3:1–8. doi: 10.1093/tas/txy114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Diniz W.J.S., Banerjee P., Regitano L.C.A. Cross talk between mineral metabolism and meat quality: A systems biology overview. Physiol. Genom. 2019;51:529–538. doi: 10.1152/physiolgenomics.00072.2019. [DOI] [PubMed] [Google Scholar]
- 23.Lalman D., Richards C. Nutrient Requirements of Beef Cattle. [(accessed on 4 November 2020)]; Available online: http://pods.dasnr.okstate.edu/docushare/dsweb/Get/Document-1921/E-974web.pdf.
- 24.Burns B.M., Fordyce G., Holroyd R.G. A review of factors that impact on the capacity of beef cattle females to conceive, maintain a pregnancy and wean a calf—Implications for reproductive efficiency in northern Australia. Anim. Reprod. Sci. 2010;122:1–22. doi: 10.1016/j.anireprosci.2010.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Menezes A.C.B., McCarthy K.L., Kassetas C.J., Baumgaertner F., Kirsch J.D., Dorsam S., Neville T.L., Ward A.K., Borowicz P.P., Reynolds L.P., et al. Vitamin and mineral supplementation and rate of gain during the first trimester of gestation affect concentrations of amino acids in maternal serum and allantoic fluid of beef heifers. J. Anim. Sci. 2021 doi: 10.1093/jas/skab024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Galyean M.L., Beauchemin K.A., Caton J., Cole N.A., Eisemann J.H., Engle T., Erickson G.E., Krehbiel C.R., Lemenager R.P., Tedeschi L.O. Nutrient Requirements of Beef Cattle. 8th ed. National Academies Press; Washington, DC, USA: 2016. National Academies of Sciences and Engineering and Medicine. [Google Scholar]
- 27.McCarthy K.L., Nestande J., Kassetas C.J., Menezes A.C.B., Baumgaertner F., Kirsch J.D., Dorsam S.T., Neville T.L., Ward A.K., Borowicz P.P., et al. Effects of Feeding a Vitamin and Mineral Supplement and/or an Energy Supplement to Beef Heifers During the First 84 Days of Pregnancy on Heifer Performance, Concentrations of Progesterone, and Corpus Luteum Size and Fetal Body Measurements. [(accessed on 2 November 2020)];2020 Available online: https://www.ag.ndsu.edu/publications/livestock/2020-north-dakota-beef-and-sheep-report#section-10.
- 28.McLean K.J., Dahlen C.R., Borowicz P.P., Reynolds L.P., Crosswhite M.R., Neville B.W., Walden S.D., Caton J.S. Technical note: A new surgical technique for ovariohysterectomy during early pregnancy in beef heifers. J. Anim. Sci. 2016;94:5089–5096. doi: 10.2527/jas.2016-0761. [DOI] [PubMed] [Google Scholar]
- 29.Grazul-Bilska A.T., Johnson M.L., Borowicz P.P., Minten M., Bilski J.J., Wroblewski R., Velimirovich M., Coupe L.R., Redmer D.A., Reynolds L.P. Placental development during early pregnancy in sheep: Cell proliferation, global methylation, and angiogenesis in the fetal placenta. Reproduction. 2011;141:529–540. doi: 10.1530/REP-10-0505. [DOI] [PubMed] [Google Scholar]
- 30.Andrews S. FASTQC. A Quality Control Tool for High Throughput Sequence Data. [(accessed on 5 October 2020)]; Available online: https://www.bioinformatics.babraham.ac.uk/projects/fastqc/
- 31.Ewels P., Magnusson M., Lundin S., Käller M. MultiQC: Summarize analysis results for multiple tools and samples in a single report. Bioinformatics. 2016;32:3047–3048. doi: 10.1093/bioinformatics/btw354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rosen B.D., Bickhart D.M., Schnabel R.D., Koren S., Elsik C.G., Tseng E., Rowan T.N., Low W.Y., Zimin A., Couldrey C., et al. De novo assembly of the cattle reference genome with single-molecule sequencing. Gigascience. 2020;9:1–9. doi: 10.1093/gigascience/giaa021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Dobin A., Davis C.A., Schlesinger F., Drenkow J., Zaleski C., Jha S., Batut P., Chaisson M., Gingeras T.R. STAR: Ultrafast universal RNA-seq aligner. Bioinformatics. 2013;29:15–21. doi: 10.1093/bioinformatics/bts635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tarazona S., Furió-Tarí P., Turrà D., Di Pietro A., Nueda M.J., Ferrer A., Conesa A. Data quality aware analysis of differential expression in RNA-seq with NOISeq R/Bioc package. Nucleic Acids Res. 2015 doi: 10.1093/nar/gkv711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Robinson M.D., McCarthy D.J., Smyth G.K. edgeR: A Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics. 2010;26:139–140. doi: 10.1093/bioinformatics/btp616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Love M.I., Huber W., Anders S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014;15:550. doi: 10.1186/s13059-014-0550-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Leek J.T., Storey J.D. Capturing heterogeneity in gene expression studies by Surrogate Variable Analysis. PLoS Genet. 2007;3:e161. doi: 10.1371/journal.pgen.0030161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Benjamini Y., Hochberg Y. Controlling the False Discovery Rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B. 1995;57:289–300. doi: 10.1111/j.2517-6161.1995.tb02031.x. [DOI] [Google Scholar]
- 39.Ge S.X., Jung D., Yao R. ShinyGO: A graphical gene-set enrichment tool for animals and plants. Bioinformatics. 2020;36:2628–2629. doi: 10.1093/bioinformatics/btz931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Godfrey K.M., Barker D.J.P. Fetal nutrition and adult disease. Am. J. Clin. Nutr. 2000;71:1344–1352. doi: 10.1093/ajcn/71.5.1344s. [DOI] [PubMed] [Google Scholar]
- 41.Caton J.S., Crouse M.S., McLean K.J., Dahlen C.R., Ward A.K., Cushman R.A., Grazul-Bilska A.T., Neville B.W., Borowicz P.P., Reynolds L.P. Maternal periconceptual nutrition, early pregnancy, and developmental outcomes in beef cattle. J. Anim. Sci. 2020;98 doi: 10.1093/jas/skaa358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sibley C.P. Understanding placental nutrient transfer—Why bother? New biomarkers of fetal growth. J. Physiol. 2009;587:3431–3440. doi: 10.1113/jphysiol.2009.172403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Shrestha N., Holland O.J., Kent N.L., Perkins A.V., McAinch A.J., Cuffe J.S.M., Hryciw D.H. Maternal high linoleic acid alters placental fatty acid composition. Nutrients. 2020;12:2183. doi: 10.3390/nu12082183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cerri R., Thompson I., Kim I., Ealy A., Hansen P., Staples C., Li J., Santos J., Thatcher W. Effects of lactation and pregnancy on gene expression of endometrium of Holstein cows at day 17 of the estrous cycle or pregnancy. J. Dairy Sci. 2012;95:5657–5675. doi: 10.3168/jds.2011-5114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Madison B.B. Srebp2: A master regulator of sterol and fatty acid synthesis1. J. Lipid Res. 2016;57:333–335. doi: 10.1194/jlr.C066712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Chatuphonprasert W., Jarukamjorn K., Ellinger I. Physiology and pathophysiology of steroid biosynthesis, transport and metabolism in the human placenta. Front. Pharmacol. 2018;9:1027. doi: 10.3389/fphar.2018.01027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Schuler G., Greven H., Kowalewski M.P., Döring B., Özalp C.R., Hoffmann B. Placental steroids in cattle: Hormones, placental growth factors or by-products of trophoblast giant cell differentiation? Exp. Clin. Endocrinol. Diabetes. 2008;116:429–436. doi: 10.1055/s-2008-1042408. [DOI] [PubMed] [Google Scholar]
- 48.Baczyk D., Kingdom J.C.P., Uhlén P. Calcium signaling in placenta. Cell Calcium. 2011;49:350–356. doi: 10.1016/j.ceca.2010.12.003. [DOI] [PubMed] [Google Scholar]
- 49.Shemesh M., Strauss J.F., Hansel W., Shore L.S., Izhar M. Control of bovine placental progesterone synthesis: Roles of cholesterol availability and calcium-activated systems. J. Steroid Biochem. 1988;29:21–25. doi: 10.1016/0022-4731(88)90371-8. [DOI] [PubMed] [Google Scholar]
- 50.Sellak H., Choi C.S., Dey N.B., Lincoln T.M. Transcriptional and post-transcriptional regulation of cGMP-dependent protein kinase (PKG-I): Pathophysiological significance. Cardiovasc. Res. 2013;97:200–207. doi: 10.1093/cvr/cvs327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Dordea A.C., Sweeney M., Taggart J., Lartey J., Wessel H., Robson S.C., Taggart M.J. Differential vasodilation of human placental and myometrial arteries related to myofilament Ca2+-desensitization and the expression of Hsp20 but not MYPT1. Mol. Hum. Reprod. 2013;19:727–736. doi: 10.1093/molehr/gat045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Shin J.S., Choi M.Y., Longtine M.S., Nelson D.M. Vitamin D effects on pregnancy and the placenta. Placenta. 2010;31:1027–1034. doi: 10.1016/j.placenta.2010.08.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zingg J.-M., Meydani M., Azzi A. α-Tocopheryl phosphate-An activated form of vitamin E important for angiogenesis and vasculogenesis? BioFactors. 2012;38:24–33. doi: 10.1002/biof.198. [DOI] [PubMed] [Google Scholar]
- 54.Narlis M., Grote D., Gaitan Y., Boualia S.K., Bouchard M. Pax2 and Pax8 regulate branching morphogenesis and nephron differentiation in the developing kidney. J. Am. Soc. Nephrol. 2007;18:1121–1129. doi: 10.1681/ASN.2006070739. [DOI] [PubMed] [Google Scholar]
- 55.Ferretti E., Arturi F., Mattei T., Scipioni A., Tell G., Tosi E., Presta I., Morisi R., Lacroix L., Gulino A., et al. Expression, regulation, and function of PAX8 in the human placenta and placental cancer cell lines. Endocrinology. 2005;146:4009–4015. doi: 10.1210/en.2005-0084. [DOI] [PubMed] [Google Scholar]
- 56.Mao J., Zhang X., Sieli P.T., Falduto M.T., Torres K.E., Rosenfeld C.S. Contrasting effects of different maternal diets on sexually dimorphic gene expression in the murine placenta. Proc. Natl. Acad. Sci. USA. 2010;107:5557–5562. doi: 10.1073/pnas.1000440107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Reynolds L.P., Biondini M.E., Borowicz P.P., Vonnahme K.A., Caton J.S., Grazul-Bilska A.T., Redmer D.A. Functional significance of developmental changes in placental microvascular architecture: The sheep as a model. Endothel. J. Endothel. Cell Res. 2005;12:11–19. doi: 10.1080/10623320590933734. [DOI] [PubMed] [Google Scholar]
- 58.Van Eetvelde M., Kamal M.M., Hostens M., Vandaele L., Fiems L.O., Opsomer G. Evidence for placental compensation in cattle. Animal. 2016;10:1342–1350. doi: 10.1017/S1751731116000318. [DOI] [PubMed] [Google Scholar]
- 59.Huang X., Anderle P., Hostettler L., Baumann M.U., Surbek D.V., Ontsouka E.C., Albrecht C. Identification of placental nutrient transporters associated with intrauterine growth restriction and pre-eclampsia. BMC Genom. 2018;19:1–17. doi: 10.1186/s12864-018-4518-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gaccioli F., Lager S., Powell T.L., Jansson T. Placental transport in response to altered maternal nutrition. J. Dev. Orig. Health Dis. 2013;4:101–115. doi: 10.1017/S2040174412000529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Dunlap K.A., Brown J.D., Keith A.B., Satterfield M.C. Factors controlling nutrient availability to the developing fetus in ruminants. J. Anim. Sci. Biotechnol. 2015;6:16. doi: 10.1186/s40104-015-0012-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Ruiz-Palacios M., Ruiz-Alcaraz A.J., Sanchez-Campillo M., Larqué E. Role of Insulin in placental transport of nutrients in gestational diabetes mellitus. Ann. Nutr. Metab. 2017;70:16–25. doi: 10.1159/000455904. [DOI] [PubMed] [Google Scholar]
- 63.Hoffman N.J., Penque B.A., Habegger K.M., Sealls W., Tackett L., Elmendorf J.S. Chromium Enhances Insulin Responsiveness via AMPK. J. Nutr. Biochem. 2015;25:565–572. doi: 10.1016/j.jnutbio.2014.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Batistel F., Alharthi A.S., Wang L., Parys C., Pan Y.X., Cardoso F.C., Loor J.J. Placentome nutrient transporters and mammalian target of rapamycin signaling proteins are altered by the methionine supply during late gestation in dairy cows and are associated with newborn birth weight. J. Nutr. 2017;147:1640–1647. doi: 10.3945/jn.117.251876. [DOI] [PubMed] [Google Scholar]
- 65.Yang T., Espenshade P.J., Wright M.E., Yabe D., Gong Y., Aebersold R., Goldstein J.L., Brown M.S. Crucial step in cholesterol homeostasis: Sterols promote binding of SCAP to INSIG-1, a membrane protein that facilitates retention of SREBPs in ER. Cell. 2002;110:489–500. doi: 10.1016/S0092-8674(02)00872-3. [DOI] [PubMed] [Google Scholar]
- 66.Matsuda S., Kobayashi M., Kitagishi Y. Expression and Function of PPARs in Placenta. PPAR Res. 2013;2013:1–7. doi: 10.1155/2013/256508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lewis R.M., Wadsack C., Desoye G. Placental fatty acid transfer. Curr. Opin. Clin. Nutr. Metab. Care. 2018;21:78–82. doi: 10.1097/MCO.0000000000000443. [DOI] [PubMed] [Google Scholar]
- 68.Weedon-Fekjaer M.S., Dalen K.T., Solaas K., Staff A.C., Duttaroy A.K., Nebb H.I. Activation of LXR increases acyl-CoA synthetase activity through direct regulation of ACSL3 in human placental trophoblast cells. J. Lipid Res. 2010;51:1886–1896. doi: 10.1194/jlr.M004978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Nakahara K., Ohkuni A., Kitamura T., Abe K., Naganuma T., Ohno Y., Zoeller R.A., Kihara A. The Sjögren-Larsson syndrome gene encodes a hexadecenal dehydrogenase of the sphingosine 1-phosphate degradation pathway. Mol. Cell. 2012;46:461–471. doi: 10.1016/j.molcel.2012.04.033. [DOI] [PubMed] [Google Scholar]
- 70.Bildirici I., Schaiff W.T., Chen B., Morizane M., Oh S.Y., O’Brien M., Sonnenberg-Hirche C., Chu T., Barak Y., Nelson D.M., et al. PLIN2 is essential for trophoblastic lipid droplet accumulation and cell survival during hypoxia. Endocrinology. 2018;159:3937–3949. doi: 10.1210/en.2018-00752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ohi K., Ursini G., Li M., Shin J.H., Ye T., Chen Q., Tao R., Kleinman J.E., Hyde T.M., Hashimoto R., et al. DEGS2 polymorphism associated with cognition in schizophrenia is associated with gene expression in brain. Transl. Psychiatry. 2015;5:550. doi: 10.1038/tp.2015.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Lemaitre R.N., Johnson C.O., Hesselson S., Sotoodhenia N., McKnight B., Sitlani C.M., Rea T.D., King I.B., Kwok P.Y., Mak A., et al. Common variation in fatty acid metabolic genes and risk of incident sudden cardiac arrest. Hear. Rhythm. 2014;11:471–477. doi: 10.1016/j.hrthm.2014.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Putzer H., Laalami S. Regulation of the Expression of Aminoacyl-tRNA Synthetases and Translation Factors. [(accessed on 2 November 2020)]; Available online: https://www.ncbi.nlm.nih.gov/books/NBK6026/
- 74.Robles M., Couturier-Tarrade A., Derisoud E., Geeverding A., Dubois C., Dahirel M., Aioun J., Prezelin A., Calvez J., Richard C., et al. Effects of dietary arginine supplementation in pregnant mares on maternal metabolism, placental structure and function and foal growth. Sci. Rep. 2019;9:1–19. doi: 10.1038/s41598-019-42941-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bassila C., Ghemrawi R., Flayac J., Froese D.S., Baumgartner M.R., Guéant J.L., Coelho D. Methionine synthase and methionine synthase reductase interact with MMACHC and with MMADHC. Biochim. Biophys. Acta Mol. Basis Dis. 2017;1863:103–112. doi: 10.1016/j.bbadis.2016.10.016. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All relevant data are within the paper and its Supplementary Information files. All sequencing data is publicly available on NCBI’s Gene Expression Omnibus (https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE165378).