Abstract
N6-methyladenosine (m6A) is one of the most prevalent epigenetic modifications of eukaryotic RNA. The m6A modification is a dynamic and reversible process, regulated by three kinds of regulator, including m6A methyltransferases, demethylases and m6A-binding proteins, and this modification plays a vital role in many diseases, especially in cancers. Accumulated evidence has proven that this modification has a significant effect on cellular biological functions and cancer progression; however, little is known about the effects of the m6A modification in non-small cell lung cancer (NSCLC). In this review, we summarized how various m6A regulators modulate m6A RNA metabolism and demonstrated the effect of m6A modification on the progression and cellular biological functions of NSCLC. We also discussed how m6A modification affects the treatment, drug resistance, diagnosis and prognosis of NSCLC patients.
Keywords: N6-methyladenosine (m6A), methyltransferases, demethylases, m6A-binding proteins, NSCLC
1. Introduction
Lung cancer is the most common type of cancer, with malignant tumors having a high incidence and mortality rate worldwide [1]. Non-small cell lung cancer (NSCLC) is the main type of lung cancer, accounting for about 80% of lung cancer cases [2]. Although targeted therapy and immunotherapy have made breakthroughs in NSCLC treatment [3], gene mutation and PD-1 expression remain obstacles in the treatment, and the five-year survival rate of NSCLC patients is still unsatisfying [4]. Therefore, it is important to explore the molecular mechanisms of applied prognostic biomarkers and therapeutic targets.
Epigenetic modifications are required for diverse biological activities in mammalians, and the role of RNA epigenetic modifications in gene expression regulation is rapidly becoming clearer [5,6]. Among these modifications, the N6-methyladenosine (m6A) modification is the most prevalent and abundant modification in eukaryotes, and has been studied extensively. m6A refers to the methylation of the sixth N of adenylated RNA/DNA, and was discovered in the early 1970s [7]. The m6A modification widely exists in the consensus sequence RRACH (where R: A or G, H: A, C or U) [8,9] and was not only enriched around stop codons, but also in the coding sequence (internal long exons) and the 3′-untranslated region (3′-UTR) [10]. In addition to the effect on messenger RNA (mRNA) translation, degradation, splicing, export and folding [11,12], m6A methylation also regulates the metabolism and functions of a variety of noncoding RNAs (ncRNAs), including long noncoding RNAs (lncRNAs), microRNAs (miRNAs) and circular RNAs (circRNAs) [12,13,14,15].
2. The Regulation of RNA m6A Modification
The m6A modification has three kinds of regulator: m6A methyltransferases (also called “writers”), demethylases (also known as “erasers”) and m6A-binding proteins (also called “readers”), which influence various m6A RNA metabolisms (Table 1).
Table 1.
Roles of m6A regulators in m6A RNA metabolism.
| m6A Regulator | Roles in m6A RNA Metabolism | Reference |
|---|---|---|
| writers | ||
| METTL3-METTL14 | reinitializes transcription during UV-induced DNA damage responses | [16] |
| WTAP | influences RNA alternative splicing | [17] |
| METTL16 | promotes mRNA splicing | [18] |
| RBM15 | mediates mRNA degradation | [19] |
| erasers | ||
| FTO | controls mRNA splicing | [20] |
| ALKBH5 | impacts longer 3′-UTR mRNA splicing and stability | [21] |
| readers | ||
| YTHDC1 | participates in transcriptional processes, mRNA splicing and mRNA nuclear export, promotes the exon inclusion of mRNA | [22,23,24] |
| YTHDC2 | participates in mRNA translation and mRNA stability | [25,26] |
| YTHDF1 | enhances translational efficiency of mRNA | [27] |
| YTHDF2 | regulates mRNA degradation, circRNA degradation and miRNA degradation | [28,29,30] |
| YTHDF3 | regulates mRNA translation, circRNA translation and mRNAs degradation | [31,32,33] |
| IGF2BPs | prevent mRNA degradation, promote mRNA stability and alter lncRNA gene expression | [34,35] |
| HNRNP particles | affects mRNA abundance, mRNA alternative splicing, mRNA gene expression, RNA maturation of mRNA and RNA export pathway | [36,37] |
| EIF3 | regulates mRNA translation | [38] |
The m6A modification is done using the “writers”, mainly including the methyltransferase complex (MTC), which is mainly composed of methyltransferase-like protein 3 (METTL3), METTL14 and Wilms tumor 1-associated protein (WTAP) [6]. METTL3 forms a stable heterodimer complex together with METTL14, in a 1:1 ratio [39]. The METTL3–METTL14 complex catalyzes the methyl transfer and promotes RNA substrate recognition [39,40,41]. WTAP is responsible for localizing the METTL3–METTL14 heterodimer to the nuclear speckles and enhancing their catalytic activity [17]. METTL16 can bind to various ncRNAs (U6 small nuclear RNA (snRNA), lncRNAs) and pre-mRNAs. RNA–binding motif protein 15 (RBM15) facilitates the recruitment of MTC to the target sites in a WTAP–dependent manner [42].
Three m6A demethylases have been reported to date, including fat mass and obesity-associated protein (FTO), alkB homolog 5 (AlKBH5) and ALKBH3 [43,44,45]. They all belong to the alkB family of non-heme iron- (II) and 2-ketoglutarate-dependent dioxygenases [46]. FTO is mainly associated with adipogenesis [47] and can bind to mRNAs, snRNAs and tRNAs as an m6A demethylase [48]. The FTO-mediated demethylation of internal m6A preferentially presents in cell nucleus [48]. In terms of the structure, the catalytic activity of FTO can be affected by the sequence and the tertiary structure of RNAs [49]. ALKBH5 is associated with infertility and normal spermatogenesis [21].
The m6A-binding proteins refer to the proteins that recognize and bind to the m6A modification on RNA and regulate translation, decay, splicing and nuclear export [23,25,28,50]. Recent studies [51,52,53] have shown that the YT521-B homology (YTH) domain, which recognizes the sites containing methylated adenines and binds to a short, degenerated, single-stranded RNA sequence, has an m6A motif within its hydrophobic pocket. In humans, YTHDC1 is a putative nucleus m6A-binding protein [23], while YTHDF1, YTHDF2, YTHDF3 and YTHDC2 are cytoplasmic m6A-binding proteins [54]. Insulin-like growth factor 2 mRNA-binding proteins (IGF2BPs), a family of m6A readers [55], bind directly to target mRNA through their hnRNP-K homology (KH) domains, which may be indispensable for recognizing and binding m6A [56]. IGF2BPs include IGF2BP1, IGF2BP2 and IGF2BP3. IGF2BP1 has the most conserved oncogenic potential among IGF2BPs [57]. The heterogeneous nuclear ribonucleoprotein (HNRNP) particles usually exist in the forms of tetramers. They include hnRNP (C1) 3C2, hnRNP (A2) 3B1 and hnRNP (A1) 3B2 [58,59,60]. The hnRNP (C1) 3C2 (hnRNP C) is a core component of hnRNP particles [61] and can recruit hnRNP particles to pre-mRNA [62]. In terms of structure, hnRNP C shows a preference for binding to uridine (U)-rich sequences [63]. A recent study [12] shows that the heterogeneous nuclear ribonucleoprotein G (HNRNPG) was also an m6A reader. Three subunits of the eukaryotic initiation factor 3 (eIF3) complex are correlated with cancer development—subunit eIF3d possesses cap-binding activity [64]; eIF3G is associated with cancer progression; eIF3h interacts with METTL3 [65,66].
m6A methyltransferases and m6A demethylases modulate the level of m6A in RNA, and RNA m6A modification significantly affects RNA splicing, transcription, nuclear export, translation, decay and stability under the action of m6A-binding proteins (Figure 1). It can be seen that m6A modification is a dynamic, reversible and continuous process. Furthermore, interaction between m6A regulators is common. For example, METTL3, METTL14 and WTAP compose a complex that promotes the localization of MTC at nuclear speckles [67] and METTL3 interacts with eIF3h to enhance mRNA translation [66]. YTHDF3 modulates the target mRNAs translation in synergy with YTHDF1 and regulates the mRNAs decay, cooperating with YTHDF2 [32]. Interestingly, the RNA-binding affinity of YTHDF1 and YTHDF3 is negatively correlated [31]. Three YTHDF proteins work together to induce the degradation of target m6A-mRNAs and one of them can fully or partially compensate for the effect of the others on target m6A-mRNAs degradation [68]. Therefore, it is believed that m6A modification regulates m6A RNA metabolism via the collective effect of m6A regulators.
Figure 1.
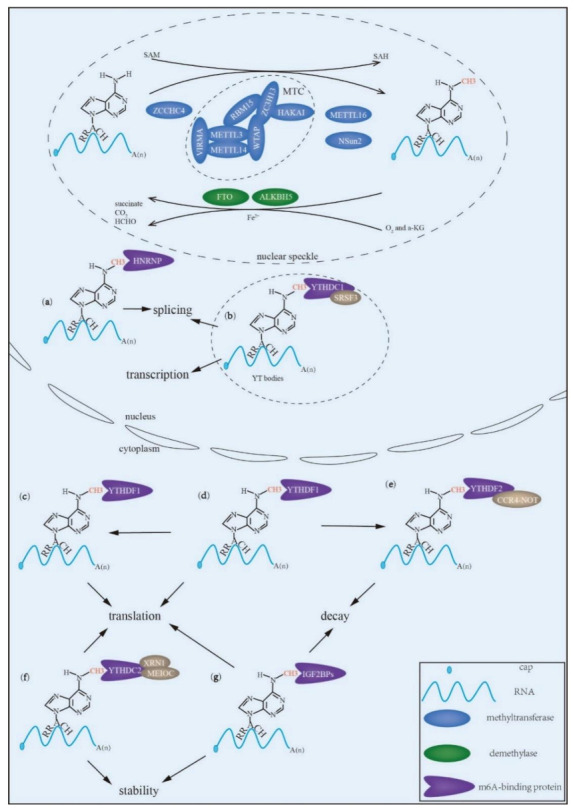
The regulation of RNA N6-methyladenosine (m6A) modification. The m6A modification of RNA is catalyzed by m6A methyltransferases including methyltransferase-like protein 3 (METTL3), METTL14, Wilms tumor 1-associated protein (WTAP), vir-like m6A methyltransferase associated (VIRMA), RNA-binding motif protein 15 (RBM15), zinc finger CCCH domain-containing protein 13 (ZC3H13), Cbl photo oncogene like 1 (CBLL-1, also termed HAKAI), NOP2/Sun domain family member 2 (NSun2) and CCHC zinc-finger-containing protein (ZCCHC4), and is removed by demethylases including fat mass and obesity-associated protein (FTO) and alkB homolog 5 (AlKBH5). In addition, the m6A modification is recognized and bound by m6A-binding proteins, which determine the fate of target RNAs—(a) Heterogeneous nuclear ribonucleoprotein (HNRNP) particles facilitate alternative splicing. (b) YT521-B homology domain-containing (YTHDC) 1 participates in alternative splicing, transcription and nuclear export. (c) YT521-B homology domain family (YTHDF) 1 enhances translation. (d) YTHDF3 regulates translation. YTHDF3 synergizes with YTHDF1 to modulate translation and cooperates with YTHDF2 to regulate decay. (e) YTHDF2 accelerates decay. (f) YTHDC2 participates in translation and increases stability. (g) Insulin-like growth factor 2 mRNA-binding proteins (IGF2BPs) augment translation, stability and decay.
3. Dual Effects of RNA with m6A Modification on Progression and Cellular Biological Functions of NSCLC
m6A modification of mRNAs and lncRNAs regulates cellular biological functions in NSCLC via the control of m6A regulators. Additionally, the m6A mRNA modification not only plays an oncogene role, but also an anti-oncogene role (Table 2 and Table 3). m6A mRNA modification plays these different roles in the progression and cellular biological function of NSCLC, likely due to different hepatic inflammatory microenvironments [69].
Table 2.
Dual effects of m6A mRNA modification on non-small cell lung cancer (NSCLC).
| m6A Regulators | Role of m6A Modification in NSCLC | Function of m6A RNA in NSCLC | Reference |
|---|---|---|---|
| writers | |||
| METTL3 | oncogene | promotes cell EMT, migration and invasion | [70,71,72] |
| METTL14 | anti-oncogene | unclear | [73] |
| KIAA1429 | oncogene | promotes cell proliferation, invasion and migration | [74] |
| erasers | |||
| ALKBH5 | oncogene | promotes cell proliferation, invasion, migration and EMT | [75,76] |
| anti-oncogene | inhibits cell under IH proliferation and invasion | [77,78] | |
| FTO | anti-oncogene | inhibits cell proliferation and invasion | [79,80] |
| readers | |||
| YTHDF3 | oncogene | promotes cell growth, migration and invasion | [70] |
| YTHDF1 | oncogene | promotes cell growth, EMT and invasion | [75] |
| YTHDF2 | oncogene | promotes cell growth and proliferation | [81] |
| anti-oncogene | inhibits cell growth, EMT and invasion | [75] | |
| YTHDC2 | anti-oncogene | unclear | [82] |
Table 3.
Effects of m6A long noncoding RNAs (lncRNAs) modification on NSCLC.
3.1. Effects of m6A Modification of mRNAs on NSCLC
3.1.1. mRNA m6A Modification Generates an Oncogene Effect on NSCLC
Messenger RNA Yes-Associated Protein (mRNA YAP)
YAP is one of the main effectors of the Hippo pathway. It plays an oncogenic role in the development, progression and prognosis of NSCLC [86]. Jin et al. [70] found that METTL3 methylated YAP pre-mRNA acts as an m6A methyltransferase to enhance YAP mRNA translation and expression in NSCLC cells. The increase in YAP m6A modification mediated by METTL3 induced resistance to cisplatin (cis-diamminedichloroplatinum, DDP) and metastasis in NSCLC. The mRNA acts as a target for m6A demethylase ALKBH5, which decreased YAP activity by regulating miR-107/LATS2 to inhibit NSCLC tumor growth and metastasis [75]. In addition, they found that YTHDF1, YTHDF2 and YTHDF3 all bound to YAP pre-mRNA, and recognized the m6A modification to regulate mRNA YAP expression. YTHDF3 recognized m6A modifications initiated by METTL3 and upregulated YAP expression to promote cellular growth, invasion and migration in NSCLC [70]. More importantly, YTHDF1 promoted mRNA translation to enhance the cellular ability of growth, invasion and epithelial–mesenchymal transition (EMT) in NSCLC [75].
Messenger RNA Enhancer of Zeste Homolog 2 (mRNA EZH2)
Histone methyltransferase EZH2 is the catalytic subunit of the polycomb repressive complex 2 (PRC2) and is known to participate in the histone deacetylase (HDAC) process [87]. METTL3-mediated m6A modification of EZH2 mRNA increased migration and invasion level in NSCLC, and Simvastatin generated anti-cancer effects by downregulating METTL3 expression [71].
mRNA JUNB
JUNB is an EMT-related transcription factor (TF) and regulates the ability of cellular EMT [88]. JUNB mRNA stability was decreased by reducing METTL3-directed m6A RNA modification in NSCLC cell lines. Elevated JUNB expression increased the TGF-β-induced migratory activity in NSCLC [72].
Messenger RNA 6-Phosphogluconate Dehydrogenase (mRNA 6PGD)
The occurrence of aerobic glycolysis is increased in cancer cells, which is known as the Warburg effect. As the third enzyme in the oxidative pentose phosphate pathway (PPP), 6PGD is vital in the process of regulating cancer cell metabolism [89,90]. Sheng et al. [81] reported that mRNA 6PGD knockdown inhibited the proliferation of NSCLC cell lines, and mRNA overexpression reversed the tumor cell growth inhibition mediated by YTHDF2 knockdown. In their work, YTHDF2 recognized that the m6A modification sites on the 6PGD mRNA 3′ UTR and YTHDF2 facilitate NSCLC cells growth and proliferation in an m6A-dependent way, via inducing 6PGD protein expression. Undoubtedly, the m6A methylation of mRNA 6PGD generates an oncogene effect via YTHDF2 in NSCLC.
mRNA MUC3A
The human intestinal mucin gene MUC3A encodes membrane-bound glycoprotein and is known to be an oncogene [91]. There is a low methylation level in MUC3A-high cell lines in colon adenocarcinoma [92]. Zhao et al. [74] found that the mRNA and m6A level of MUC3A mediated by vir-like m6A methyltransferase associated (VIRMA, also termed KIAA1429) had a positive correlation with the KIAA1429 level, which presented an opposing result to the previous study. In addition, overexpressing MUC3A induced cell proliferation, migration and invasion in lung adenocarcinoma (LUAD).
Messenger RNA Ubiquitin-Conjugating Enzyme E2C (mRNA UBE2C)
UBE2C encodes a class of ubiquitin-conjugating enzymes that are involved in ubiquitin-mediated protein degradation [93] and associated with the prognosis of stage I NSCLC patients [94]. The m6A and expression level of mRNA UBE2C was decreased by ALKBH5. Downregulation of the mRNA activated the expression of the autophagy markers ATG3 and LC3B and reduced cell proliferation, clonogenicity, and the invasive growth of NSCLC [76]. It can be seen that m6A modification is involved in deregulation of the UBE2C–autophagy repression axis.
3.1.2. m6A Modification of mRNAs Generates an Anti-Oncogene Effect on NSCLC
mRNA YAP
The m6A modification on mRNA YAP not only generates an oncogene effect, but also anti-oncogene effect in NSCLC. Research [75] has confirmed that m6A modification generated an anti-oncogene effect via YTHDF2-mediated degradation of YAP by weakening the cellular abilities of growth, invasion and EMT in NSCLC.
Messenger RNA Forkhead Box M1 (mRNA FOXM1)
A study has shown that mRNA FOXM1 is a transcription factor that enhances cellular proliferative capacity in lung cancer [95]. ALKBH5 enhances the translational efficiency of mRNA FOXM1 and increases FOXM1 protein level by decreasing the FOXM1 mRNA m6A level. Increasing FOXM1 expression strengthens the abilities of cellular proliferation and invasion in LUAD under intermittent hypoxia (IH) [77].
Messenger RNA Tissue Inhibitor of Metalloproteinase 3 (mRNA TIMP3)
TIMP3 is an accepted tumor suppressor gene and can inhibit tumor invasion and metastasis [96,97]. ALKBH5 decreased the mRNA stability by reducing the m6A level to promote the malignant biological properties of NSCLC cells [78].
Messenger RNA Ubiquitin-Specific Protease-7 (mRNA USP7)
The deubiquitylating enzyme USP7 was previously characterized as a tumor suppressor gene, but was recently proven to promote cell proliferation in NSCLC [98,99]. The m6A level of mRNA USP7 was decreased by m6A demethylase FTO, which promoted NSCLC cell growth [79].
Messenger RNA Myeloid Zinc Finger 1 (mRNA MZF1)
MZF1, a member of the SCAN-Zinc Finger (SCAN-ZF) transcription factor family, may promote tumor progression and cause poor prognosis of the patients in LUAD [100]. The mRNA level of MZF1 was remarkably reduced, and the m6A modification of the mRNA transcripts was added by genetic silencing of the expression of FTO in lung squamous cell carcinoma (LUSC) cell lines. Forced expression of MZF1 partially reversed the suppressive effect on the viability and invasion of LUAC cell lines mediated by FTO knockdown [80]. Generally speaking, the m6A modification of mRNA MZF1 generates an anti-oncogene effect via FTO in LUSC.
Messenger RNA Solute Carrier 7A11 (mRNA SLC7A11)
Downregulating the expression of SLC7A11 inhibits cellular proliferation and colony formation in lung cancer [101]. The m6A modification destabilized SLC7A11 mRNA and accelerated mRNA decay after recognition by YTHDC2 in LUAD, which impaired cystine uptake. Significantly, this provides the theoretical basis for system X C− -targeting therapy for LUAD [82].
mRNA NOTCH1
Some studies indicated that the Notch pathway works as an oncogene by downregulating the mRNA-suppressed NSCLC cell growth and invasion [102,103]. An mRNA degradation experiment in H1299 cells demonstrated that mRNA NOTCH1 stability can be promoted by circNOTCH1 [73]. For the purpose of exploring the effect of m6A modification on mRNA NOTCH1 stability, Shen et al. [73] performed RNA interaction–precipitation (RIP), which showed that NOTCH1 mRNA level was much lower in the control group than that in the shcircNOTCH1 group. These indicated that m6A modification related to METTL14 on NOTCH1 mRNA decreases mRNA stability in NSCLC cells. As a result, there is a chance that the m6A modification of NOTCH1 mRNA may generate an anti-oncogene effect via METTL14 in NSCLC.
3.2. Effects of m6A Modification of ncRNAs on NSCLC
3.2.1. Long Non-Coding RNA Lung Adenocarcinoma Transcript 1 (lncRNA MALAT1)
lncRNA MALAT1 is up-regulated in NSCLC tissues and silencing of its expression represses cell proliferation, invasion and promotes apoptosis [104]. In addition, METTL3 increased the RNA expression level of the lncRNA in an m6A-dependent manner. In addition, YTHDF3 recognized the m6A modification sites on the lncRNA and enhanced the lncRNA stability in NSCLC cells [70].
3.2.2. Long Non-Coding RNA FEZ Family Zinc Finger 1 Antisense RNA 1 (lncRNA FEZF1-AS1)
There are seven m6A-modified sites on lncRNA FEZF1-AS1. Knocking down lncRNA FEZF1-AS1 inhibits the proliferation, migration and invasion abilities of NSCLC cell lines [85]. Meanwhile, silencing METTL3, METTL14, YTHDF1 and YTHDF2 decreased the expression level of the lncRNA [85]. Therefore, it is possible that the m6A methylation of lncRNA FEZF1-AS1 plays an “oncogene” role via METTL3, METTL14, YTHDF1 and YTHDF2 in NSCLC.
3.2.3. Long Non-Coding RNA ABHD11 Antisense RNA 1 (lncRNA ABHD11-AS1)
It has been proven that lncRNA ABHD11-AS1 contributes to many kinds of cancer tumorigenesis [105,106,107]. In addition, Xue et al. [83] found that there were modification sites on METTL3 that match the m6A modification of the lncRNA. METTL3 overexpression increased the m6A level of lncRNA ABHD11-AS1 and strengthened the stability of the lncRNA. lncRNA ABHD11-AS1 also promoted the Warburg effect of NSCLC via the EZH2/KLF4 axis.
3.2.4. miR-143-3p
miR-143-3p induces EMT of NSCLC cells and is correlated with the progression of lung cancer. m6A modification can facilitate the splicing of precursor miR-143-3p and overexpression of METTL3 can increase the expression of miR-143-3p in NSCLC cells. Then, miR-143-3p promotes the brain metastasis of NSCLC in the condition of the regulation of vasohibin-1 (VASH1)/vascular endothelial growth factor A (VEGFA) axis [84].
There are other m6A modifications of ncRNAs that influence the progression and cellular biological function of NSCLC, which have yet to be uncovered. For instance, miR-376c was carried by endothelial cells (ECs)-secreted extracellular vesicles (EVs). Previous research has concluded that miR-376c overexpression could generate an inhibitory effect on Wnt signaling, NSCLC cell growth and invasion [108]. The overexpression of YTHDF1 could attenuate this effect by targeting miR-376c [109]. Interacting with heterogeneous nuclear ribonucleoprotein A2/B1 (HNRNPA2B1), the upregulation of LINC01234 mediated by c-Myc promotes the processing of primary miR-106b-5p. miR-106b-5p enhances NSCLC cell growth by inhibiting cryptochrome 2 (CRY2) expression and upregulating c-Myc. This forms a positive-feedback loop that is participated in by the m6A reader and miR-106b-5p and promotes NSCLC cell growth [110]. In addition, derived from the NADH: ubiquinone oxidoreductase subunit B2 (NDUFB2), circNDUFB2 is significantly downregulated in NSCLC and negatively correlated with malignant features of NSCLC. It serves as a scaffold to bridge TRIM25 and IGF2BPs and forms the TRIM25/circNDUFB2/IGF2BPs ternary complex, which accelerates IGF2BP ubiquitination and degradation. Interestingly, the acceleration effect is enhanced by m6A circNDUFB2 [111]. The effect of m6A modification on miR-106b-5p, miR-376c and circNDUFB2 to NSCLC remains to be explored.
4. Roles of m6A Modification on Treatment, Drug Resistance, Diagnosis and Prognosis of NSCLC Patients
m6A modification-related treatments have received widespread attention, such as anti-inflammatory cytokine interleukin 37 (IL-37), FTO and YTHDF1. IL-37 treatment inhibits the proliferation ability of LUAD cells via regulation of RNA methylation. Meanwhile, overexpression of IL-37 decreased the expression of ALKBH5. This means that the mechanism of IL-37 used for treating NSCLC patients may be associated with m6A methylation [112]. Although the emergence of targeted therapy provides a new direction for the treatment of NSCLC, drug resistance has become a major obstacle during the treatment. Some research has indicated that m6A modification enhanced or attenuated the drug-resistance of NSCLC patients, such as resistance to gefitinib, afatinib and DDP. m6A modification decreased the gefitinib-resistance (GR) of NSCLC cells via the FTO/YTHDF2/ABCC10 axis [113]. The drug-resistance mechanism provides a theoretical basis for the theoretical application of FTO inhibitors (e.g., Rhein, meclofenamic acid and FB23-2) to the treatment of GR NSCLC patients [114,115,116]. Meng et al. analyzed how m6A methylation affected afatinib resistance of NSCLC cells. They identified the m6A-modified genes, which might affect afatinib resistance by disturbing the cell cycle [117]. When NSCLC cells are in a state of oxidative stress, depletion of YTHDF1 suppressed translation of the Kelch-like ECH-associated protein 1 (Keap1) by recognizing the m6A modification and led to DDP resistance by activating the antioxidant reactive oxygen species clearance system [118].
For diagnosis, researchers built a diagnostic score model associated with 13 m6A regulators and concluded that the diagnostic scores of the LUAD patients were much higher compared with those of the control group [119]. This indicates that m6A modification could contribute the diagnosis of NSCLC patients. IGF2BP3 was highly expressed in LUAD tissue and led to worse prognosis of LUAD patients, which suggested that m6A modification had the potential to be used for predicting the prognosis of LUAD patients [120]. Huang et al. established an effective capture system for the preparation of the circulating tumor cells’ (CTCs) lysis, nucleic acid digestion and nucleoside extraction. They then applied liquid chromatography−electrospray ionization−tandem mass spectrometry (LC–ESI–MS/MS) to further analyze the m6A level of RNA in a single cell. This strategy provided a potential basis for determining whether metastasis occurs and assessing the prognosis of NSCLC patients [121].
5. Conclusions and Perspectives
The m6A modification of RNA, regulated by methyltransferases, demethylases and m6A-binding proteins, affects the target RNA splicing, translation, decay and nuclear export, which determines the development and progression of NSCLC. Increasing evidence has suggested that the effect of m6A modification on NSCLC patients’ prognosis is a double-edged sword, in that m6A modification has both promoting and inhibiting impacts on NSCLC development. As a prominent topic amongst epigenetic modifications, the roles of m6A modification of RNA in the progression and cellular biological functions of NSCLC are complicated. The reason for this complexity may be that m6A regulators have intricate effects on tumor-related RNA metabolism and that the inflammatory microenvironments and immune responses influence m6A modifications differently in different stagings and histological types of NSCLC. Besides, m6A modification acts as a critical factor that affects the treatment, drug resistance, diagnosis and prognosis of NSCLC patients.
RNAs with m6A modification may interact with signaling pathways or another regulatory network, such as the ceRNAs mechanism, to affect the progression of NSCLC and drug resistance of NSCLC patients, as in the description made by Jin et al. [70]. The existence of tRNA-derived small RNAs (tsRNAs) mutations in lung cancer samples indicates that tsRNAs can be a new diagnostic marker of lung cancer. It is believed that the diagnostic efficiency of m6A modification in NSCLC can be improved by combination with tsRNAs [122].
Targeted therapy and immunotherapy are still promising in the treatment of NSCLC; however, the corresponding drug resistance caused by m6A modification hampers their development. This reflects the importance of searching for inhibitors and activators of m6A regulators. Although m6A modification is not yet widely used in the clinical diagnosis and prognosis of NSCLC patients due to the complexity of detection, the RNA m6A level, to some extent, reflects whether lung cancer occurs and whether a NSCLC patient has a good prognosis. Therefore, further exploration of simpler and more feasible detection methods of RNA m6A level will be beneficial in the application of m6A modification in the diagnosis and prognosis of NSCLC patients. Gene-transduced adipocytes therapy was used for the treatment of breast cancer and was effective [123]. The inhibitors or activators of m6A regulators and tumor-related RNAs with or without m6A modification may be applied to the treatment of NSCLC patients via combination with Gene-transduced adipocytes therapy. The epidermal growth factor (EGFR) tyrosine kinase inhibitors (TKIs) has been effectively applied to the treatment of NSCLC by the combination of gefitinib with radiotherapy or chemoradiotherapy [124]. Further research into the application of EGFR TKs combined with m6A modification-related GR to the treatment of NSCLC patients may provide a new treatment program. Therefore, it is necessary to explore m6A RNA modification in the treatment and drug-resistance of NSCLC patients.
Although research into the effect of m6A modification of RNA on NSCLC is increasing, there are a number of problems that have yet to be solved, especially regarding the combined effect of m6A regulators on NSCLC. First of all, the gene expression levels of many m6A regulators in LUAD and LUSC tissues were significantly different to the levels in normal tissues [125]. Therefore, it is important to explore whether the other m6A regulators are associated with the development and progression of NSCLC. Secondly, because the m6A regulators are diversified and have various functions, different combinations determine the levels of RNAs m6A modification in NSCLC cells under certain conditions [125]. METTL3 and METTL14 both are methyltransferases, but their expression in LUAD tissues shows opposing tendencies; while FTO is a demethylase, METTL14 and FTO showed a similar expression trend [126]. Therefore, the intricate internal mechanism must be studied. Thirdly, some prognosis-associated mutant genes can impact the expression of m6A regulators in LUAD, such as TP53, which leads us to wonder whether those regulators are only effective in certain LUAD types of genetic mutations, if the regulators have different effects on LUAD development in different types of genetic mutation, or if there are other situations [104]. Finally, it has been proven that FTO and METTL3 are potential targets of diagnosis and therapy for LUAD patients [80,127]. In addition, the results of the diagnostic score model implied that YTHDF1 was expected to be a diagnostic marker and HNRNPC has the potential to act as a therapeutic target in LUAD [119], but further validation in LUAD populations is required.
Author Contributions
Conceptualization, H.X.; writing—original draft preparation, Y.C., J.Z., M.W. and H.X.; writing—review and editing, M.W. and H.X.; literature search and review, H.D., S.W. and H.X.; funding, H.X. All authors have read and agreed to the published version of the manuscript.
Funding
This publication was supported by the National Natural Science Foundation of China (No.31600662) and Haiyan Foundation from Harbin Medical University Cancer Hospital (No.JJMS2020-02).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Data sharing not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Torre L.A., Bray F., Siegel R.L., Ferlay J., Lortet-Tieulent J., Jemal A. Global cancer statistics. CA Cancer J. Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Torre L.A., Siegel R.L., Jemal A. Lung Cancer Statistics. Adv. Exp. Med. Biol. 2016;893:1–19. doi: 10.1007/978-3-319-24223-1_1. [DOI] [PubMed] [Google Scholar]
- 3.Alexander M., Kim S.Y., Cheng H. Update 2020: Management of Non-Small Cell Lung Cancer. Lung. 2020;198:897–907. doi: 10.1007/s00408-020-00407-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lin J.J., Cardarella S., Lydon C.A., Dahlberg S.E., Jackman D.M., Jänne P.A., Johnson B.E. Five-Year Survival in EGFR-Mutant Metastatic Lung Adenocarcinoma Treated with EGFR-TKIs. J. Thorac. Oncol. 2016;11:556–565. doi: 10.1016/j.jtho.2015.12.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Reik W., Dean W., Walter J. Epigenetic Reprogramming in Mammalian Development. Science. 2001;293:1089–1093. doi: 10.1126/science.1063443. [DOI] [PubMed] [Google Scholar]
- 6.Roundtree I.A., Evans M.E., Pan T., He C. Dynamic RNA Modifications in Gene Expression Regulation. Cell. 2017;169:1187–1200. doi: 10.1016/j.cell.2017.05.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Desrosiers R., Friderici K., Rottman F. Identification of Methylated Nucleosides in Messenger RNA from Novikoff Hepatoma Cells. Proc. Natl. Acad. Sci. USA. 1974;71:3971–3975. doi: 10.1073/pnas.71.10.3971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bokar J.A., Shambaugh M.E., Polayes D., Matera A.G., Rottman F.M. Purification and cDNA cloning of the AdoMet-binding subunit of the human mRNA (N6-adenosine)-methyltransferase. RNA. 1997;3:1233–1247. [PMC free article] [PubMed] [Google Scholar]
- 9.Wei C.M., Moss B. Nucleotide sequences at the N6-methyladenosine sites of HeLa cell messenger ribonucleic acid. Biochemistry. 1977;16:1672–1676. doi: 10.1021/bi00627a023. [DOI] [PubMed] [Google Scholar]
- 10.Meyer K.D., Saletore Y., Zumbo P., Elemento O., Mason C.E., Jaffrey S.R. Comprehensive Analysis of mRNA Methylation Reveals Enrichment in 3′ UTRs and near Stop Codons. Cell. 2012;149:1635–1646. doi: 10.1016/j.cell.2012.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu Q., Gregory R.I. RNAmod: An integrated system for the annotation of mRNA modifications. Nucleic Acids Res. 2019;47:W548–W555. doi: 10.1093/nar/gkz479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Liu N., Zhou K.I., Parisien M., Dai Q., Diatchenko L., Pan T. N 6-methyladenosine alters RNA structure to regulate binding of a low-complexity protein. Nucleic Acids Res. 2017;45:6051–6063. doi: 10.1093/nar/gkx141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Linder B., Grozhik A.V., Olarerin-George A.O., Meydan C., Mason C.E., Jaffrey S.R. Single-nucleotide-resolution mapping of m6A and m6Am throughout the transcriptome. Nat. Methods. 2015;12:767–772. doi: 10.1038/nmeth.3453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Molinie B., Wang J., Lim K.S., Hillebrand R., Lu Z.X., Van Wittenberghe N., Howard B.D., Daneshvar K., Mullen A.C., Dedon P., et al. m6A-LAIC-seq reveals the census and complexity of the m6A epitranscriptome. Nat. Methods. 2016;13:692–698. doi: 10.1038/nmeth.3898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Alarcón C.R., Lee H., Goodarzi H., Halberg N., Tavazoie S.F. N6-methyladenosine marks primary microRNAs for processing. Nature. 2015;519:482–485. doi: 10.1038/nature14281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xiang Y., Laurent B., Hsu C.H., Nachtergaele S., Lu Z., Sheng W., Xu C., Chen H., Ouyang J., Wang S., et al. RNA m(6)A methylation regulates the ultraviolet-induced DNA damage response. Nature. 2017;543:573–576. doi: 10.1038/nature21671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ping X.L., Sun B.F., Wang L., Xiao W., Yang X., Wang W.J., Adhikari S., Shi Y., Lv Y., Chen Y.-S., et al. Mammalian WTAP is a regulatory subunit of the RNA N6-methyladenosine methyltransferase. Cell Res. 2014;24:177–189. doi: 10.1038/cr.2014.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pendleton K.E., Chen B., Liu K., Hunter O.V., Xie Y., Tu B.P., Conrad N.K. The U6 snRNA m 6 A Methyltransferase METTL16 Regulates SAM Synthetase Intron Retention. Cell. 2017;169:824–835. doi: 10.1016/j.cell.2017.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Xie Y., Castro-Hernández R., Sokpor G., Pham L., Narayanan R., Rosenbusch J., Staiger J.F., Tuoc T. RBM15 Modulates the Function of Chromatin Remodeling Factor BAF155 Through RNA Methylation in Developing Cortex. Mol. Neurobiol. 2019;56:7305–7320. doi: 10.1007/s12035-019-1595-1. [DOI] [PubMed] [Google Scholar]
- 20.Zhao X., Yang Y., Sun B.F., Shi Y., Yang X., Xiao W., Hao Y.J., Ping X.L., Chen Y.S., Wang W.J., et al. FTO-dependent demethylation of N6-methyladenosine regulates mRNA splicing and is required for adipogenesis. Cell Res. 2014;24:1403–1419. doi: 10.1038/cr.2014.151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tang C., Klukovich R., Peng H., Wang Z., Yu T., Zhang Y., Zheng H., Klungland A., Yan W. ALKBH5-dependent m6A demethylation controls splicing and stability of long 3′-UTR mRNAs in male germ cells. Proc. Natl. Acad. Sci. USA. 2018;115:E325–E333. doi: 10.1073/pnas.1717794115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nayler O., Hartmann A.M., Stamm S. The ER Repeat Protein Yt521-B Localizes to a Novel Subnuclear Compartment. J. Cell Biol. 2000;150:949–962. doi: 10.1083/jcb.150.5.949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Xiao W., Adhikari S., Dahal U., Chen Y.S., Hao Y.J., Sun B.F., Sun H.Y., Li A., Ping X.L., Lai W.Y., et al. Nuclear m 6 A Reader YTHDC1 Regulates mRNA Splicing. Mol. Cell. 2016;61:507–519. doi: 10.1016/j.molcel.2016.01.012. [DOI] [PubMed] [Google Scholar]
- 24.Luxton H.J., Simpson B.S., Mills I.G., Brindle N.R., Ahmed Z., Stavrinides V., Heavey S., Stamm S., Whitaker H.C. The Oncogene Metadherin Interacts with the Known Splicing Proteins YTHDC1, Sam68 and T-STAR and Plays a Novel Role in Alternative mRNA Splicing. Cancers. 2019;11:1233. doi: 10.3390/cancers11091233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kretschmer J., Rao H., Hackert P., Sloan K.E., Hobartner C., Bohnsack M.T. The m(6)A reader protein YTHDC2 interacts with the small ribosomal subunit and the 5-3 exoribonuclease XRN1. RNA. 2018;24:1339–1350. doi: 10.1261/rna.064238.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mao Y., Dong L., Liu X.M., Guo J., Ma H., Shen B., Qian S.B. m6A in mRNA coding regions promotes translation via the RNA helicase-containing YTHDC. Nat. Commun. 2019;10:1–11. doi: 10.1038/s41467-019-13317-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wang X., Zhao B.S., Roundtree I.A., Lu Z., Han D., Ma H., Weng X., Chen K., Shi H., He C. N6-methyladenosine Modulates Messenger RNA Translation Efficiency. Cell. 2015;161:1388–1399. doi: 10.1016/j.cell.2015.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wang X., Lu Z., Gomez A.M., Hon G.C., Yue Y., Han D., Fu Y., Parisien M., Dai Q., Jia G., et al. N6-methyladenosine-dependent regulation of messenger RNA stability. Nature. 2014;505:117–120. doi: 10.1038/nature12730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Park O.H., Ha H., Lee Y., Boo S.H., Kwon D.H., Song H.K., Kim Y.K. Endoribonucleolytic Cleavage of m6A-Containing RNAs by RNase P/MRP Complex. Mol. Cell. 2019;74:494–507. doi: 10.1016/j.molcel.2019.02.034. [DOI] [PubMed] [Google Scholar]
- 30.Li J., Meng S., Xu M., Wang S., He L., Xu X., Wang X., Xie L. Downregulation of N6-methyladenosine binding YTHDF2 protein mediated by miR-493–3p suppresses prostate cancer by elevating N6-methyladenosine levels. Oncotarget. 2017;9:3752–3764. doi: 10.18632/oncotarget.23365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Li A., Chen Y.S., Ping X.L., Yang X., Xiao W., Yang Y., Sun H.-Y., Zhu Q., Baidya P., Wang X., et al. Cytoplasmic m6A reader YTHDF3 promotes mRNA translation. Cell Res. 2017;27:444–447. doi: 10.1038/cr.2017.10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Shi H., Wang X., Lu Z., Zhao B.S., Ma H., Hsu P.J., Liu C., He C. YTHDF3 facilitates translation and decay of N6-methyladenosine-modified RNA. Cell Res. 2017;27:315–328. doi: 10.1038/cr.2017.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang Y., Fan X., Mao M., Song X., Wu P., Zhang Y., Jin Y., Yang Y., Chen L.-L., Wang Y., et al. Extensive translation of circular RNAs driven by N6-methyladenosine. Cell Res. 2017;27:626–641. doi: 10.1038/cr.2017.31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Weidensdorfer D., Stöhr N., Baude A., Lederer M., Köhn M., Schierhorn A., Buchmeier S., Wahle E., Hüttelmaier S. Control of c-myc mRNA stability by IGF2BP1-associated cytoplasmic RNPs. RNA. 2008;15:104–115. doi: 10.1261/rna.1175909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Shang Y. LncRNA THOR acts as a retinoblastoma promoter through enhancing the combination of c-myc mRNA and IGF2BP1 protein. Biomed. Pharmacother. 2018;106:1243–1249. doi: 10.1016/j.biopha.2018.07.052. [DOI] [PubMed] [Google Scholar]
- 36.Liu N., Dai Q., Zheng G., He C., Parisien M., Pan T. N6-methyladenosine-dependent RNA structural switches regulate RNA–protein interactions. Nature. 2015;518:560–564. doi: 10.1038/nature14234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.McCloskey A., Taniguchi I., Shinmyozu K., Ohno M. hnRNP C Tetramer Measures RNA Length to Classify RNA Polymerase II Transcripts for Export. Science. 2012;335:1643–1646. doi: 10.1126/science.1218469. [DOI] [PubMed] [Google Scholar]
- 38.Meyer K.D., Patil D.P., Zhou J., Zinoviev A., Skabkin M.A., Elemento O., Pestova T.V., Qian S.-B., Jaffrey S.R. 5′ UTR m6A Promotes Cap-Independent Translation. Cell. 2015;163:999–1010. doi: 10.1016/j.cell.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Wang P., Doxtader K.A., Nam Y. Structural Basis for Cooperative Function of Mettl3 and Mettl14 Methyltransferases. Mol. Cell. 2016;63:306–317. doi: 10.1016/j.molcel.2016.05.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wang X., Feng J., Xue Y., Guan Z., Zhang D., Liu Z., Gong Z., Huang J., Tang Z.G.C., Zou T., et al. Structural basis of N6-adenosine methylation by the METTL3–METTL14 complex. Nature. 2016;534:575–578. doi: 10.1038/nature18298. [DOI] [PubMed] [Google Scholar]
- 41.Liu J., Yue Y., Han D., Wang X., Fu Y., Zhang L., Jia G., Yu M., Lu Z., Deng X., et al. A METTL3–METTL14 complex mediates mammalian nuclear RNA N6-adenosine methylation. Nat. Chem. Biol. 2014;10:93–95. doi: 10.1038/nchembio.1432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Patil D.P., Chen C.K., Pickering B.F., Chow A., Jackson C., Guttman M., Jaffrey S. m6A RNA methylation promotes XIST-mediated transcriptional repression. Nature. 2016;537:369–373. doi: 10.1038/nature19342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Jia G., Fu Y., Zhao X., Dai Q., Zheng G., Yang Y., Yi C., Lindahl T., Pan T., Yang Y.-G., et al. N6-Methyladenosine in nuclear RNA is a major substrate of the obesity-associated FTO. Nat. Chem. Biol. 2011;7:885–887. doi: 10.1038/nchembio.687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zheng G., Dahl J.A., Niu Y., Fedorcsak P., Huang C.M., Li C.J., Vågbø C.B., Shi Y., Wang W.L., Song S.-H., et al. ALKBH5 Is a Mammalian RNA Demethylase that Impacts RNA Metabolism and Mouse Fertility. Mol. Cell. 2013;49:18–29. doi: 10.1016/j.molcel.2012.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ueda Y., Ooshio I., Fusamae Y., Kitae K., Kawaguchi M., Jingushi K., Hase H., Harada K., Hirata K., Tsujikawa K. AlkB homolog 3-mediated tRNA demethylation promotes protein synthesis in cancer cells. Sci. Rep. 2017;7:srep42271. doi: 10.1038/srep42271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sundheim O., Talstad V.A., Vågbø C.B., Slupphaug G., Krokan H.E. AlkB demethylases flip out in different ways. DNA Repair. 2008;7:1916–1923. doi: 10.1016/j.dnarep.2008.07.015. [DOI] [PubMed] [Google Scholar]
- 47.Wang X., Zhu L., Chen J., Wang Y. mRNA m6A methylation downregulates adipogenesis in porcine adipocytes. Biochem. Biophys. Res. Commun. 2015;459:201–207. doi: 10.1016/j.bbrc.2015.02.048. [DOI] [PubMed] [Google Scholar]
- 48.Wei J., Liu F., Lu Z., Fei Q., Ai Y., He P.C., Shi H., Cui X., Su R., Klungland A., et al. Differential m6A, m6Am, and m1A Demethylation Mediated by FTO in the Cell Nucleus and Cytoplasm. Mol. Cell. 2018;71:973–985. doi: 10.1016/j.molcel.2018.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Zhang X., Wei L.H., Wang Y., Xiao Y., Liu J., Zhang W., Yan N., Amu G., Tang X., Zhang L., et al. Structural insights into FTO’s catalytic mechanism for the demethylation of multiple RNA substrates. Proc. Natl. Acad. Sci. USA. 2019;116:2919–2924. doi: 10.1073/pnas.1820574116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Roundtree I.A., Luo G.Z., Zhang Z., Wang X., Zhou T., Cui Y., Sha J., Huang X., Guerrero L., Xie P., et al. YTHDC1 mediates nuclear export of N6-methyladenosine methylated mRNAs. eLife. 2017;6:e31311. doi: 10.7554/eLife.31311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Luo S., Tong L. Molecular basis for the recognition of methylated adenines in RNA by the eukaryotic YTH domain. Proc. Natl. Acad. Sci. 2014;111:13834–13839. doi: 10.1073/pnas.1412742111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Zhang Z., Theler D., Kaminska K.H., Hiller M., de la Grange P., Pudimat R., Rafalska I., Heinrich B., Bujnicki J.M., Allain F.H.-T., et al. The YTH Domain Is a Novel RNA Binding Domain. J. Biol. Chem. 2010;285:14701–14710. doi: 10.1074/jbc.M110.104711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Theler D., Dominguez C., Blatter M., Boudet J., Allain F.H.-T. Solution structure of the YTH domain in complex with N6-methyladenosine RNA: A reader of methylated RNA. Nucleic Acids Res. 2014;42:13911–13919. doi: 10.1093/nar/gku1116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Zhao Y.L., Liu Y.H., Wu R.F., Bi Z., Yao Y.X., Liu Q., Wang Y.Z., Wang X.X. Understanding m6A Function Through Uncovering the Diversity Roles of YTH Domain-Containing Proteins. Mol. Biotechnol. 2019;61:355–364. doi: 10.1007/s12033-018-00149-z. [DOI] [PubMed] [Google Scholar]
- 55.Nielsen J., Christiansen J., Lykke-Andersen J., Johnsen A.H., Wewer U.M., Nielsen F.C. A family of insulin-like growth factor II mRNA-binding proteins represses translation in late development. Mol. Cell. Biol. 1999;19:1262–1270. doi: 10.1128/MCB.19.2.1262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bell J.L., Wachter K., Muhleck B., Pazaitis N., Kohn M., Lederer M., Huttelmaier S. Insulin-like growth factor 2 mRNA-binding proteins (IGF2BPs): Post-transcriptional drivers of cancer progression? Cell Mol. Life Sci. 2013;70:2657–2675. doi: 10.1007/s00018-012-1186-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Muller S., Bley N., Glass M., Busch B., Rousseau V., Misiak D., Fuchs T., Lederer M., Huttelmaier S. IGF2BP1 enhances an aggressive tumor cell phenotype by impairing miRNA-directed downregulation of oncogenic factors. Nucleic Acids Res. 2018;46:6285–6303. doi: 10.1093/nar/gky229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Barnett S.F., Friedman D.L., LeStourgeon W.M. The C proteins of HeLa 40S nuclear ribonucleoprotein particles exist as anisotropic tetramers of (C1) 3 C Mol. Cell. Biol. 1989;9:492–498. doi: 10.1128/mcb.9.2.492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Barnett S.F.A., Theiry T., LeStourgeon W.M. The core proteins A2 and B1 exist as (A2)3B1 tetramers in 40S nuclear ribonucleoprotein particles. Mol. Cell. Biol. 1991;11:864–871. doi: 10.1128/MCB.11.2.864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Harris S., Martin T., Smith H. Reversible chemical cross-linking and ribonuclease digestion analysis of the organization of proteins in ribonucleoprotein particles. Mol. Cell. Biochem. 1988;84:17–28. doi: 10.1007/BF00235189. [DOI] [PubMed] [Google Scholar]
- 61.Beyer A.L., Christensen M.E., Walker B.W., LeStourgeon W.M. Identification and characterization of the packaging proteins of core 40S hnRNP particles. Cell. 1977;11:127–138. doi: 10.1016/0092-8674(77)90323-3. [DOI] [PubMed] [Google Scholar]
- 62.König J., Zarnack K., Rot G., Curk T., Kayikci M., Zupan B., Turner D.J., Luscombe N.M., Ule J. iCLIP reveals the function of hnRNP particles in splicing at individual nucleotide resolution. Nat. Struct. Mol. Biol. 2010;17:909–915. doi: 10.1038/nsmb.1838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Cieniková Z., Damberger F.F., Hall J., Allain F.H.T., Maris C. Structural and Mechanistic Insights into Poly(uridine) Tract Recognition by the hnRNP C RNA Recognition Motif. J. Am. Chem. Soc. 2014;136:14536–14544. doi: 10.1021/ja507690d. [DOI] [PubMed] [Google Scholar]
- 64.Lee A.S., Kranzusch P.J., Doudna J.A., Cate J.H. eIF3d is an mRNA cap-binding protein that is required for specialized translation initiation. Nature. 2016;536:96–99. doi: 10.1038/nature18954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Yang C., Zhang Y., Du W., Cheng H., Li C. Eukaryotic translation initiation factor 3 subunit G promotes human colorectal cancer. Am. J. Transl. Res. 2019;11:612–623. [PMC free article] [PubMed] [Google Scholar]
- 66.Choe J., Lin S., Zhang W., Liu Q., Wang L., Ramirez-Moya J., Du P., Kim W., Tang S., Sliz P., et al. mRNA circularization by METTL3–eIF3h enhances translation and promotes oncogenesis. Nature. 2018;561:556–560. doi: 10.1038/s41586-018-0538-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Schöller E., Weichmann F., Treiber T., Ringle S., Treiber N., Flatley A., Feederle R., Bruckmann A., Meister G. Interactions, localization, and phosphorylation of the m6A generating METTL3–METTL14–WTAP complex. RNA. 2018;24:499–512. doi: 10.1261/rna.064063.117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zaccara S., Jaffrey S.R. A Unified Model for the Function of YTHDF Proteins in Regulating m6A-Modified mRNA. Cell. 2020;181:1582–1595.e18. doi: 10.1016/j.cell.2020.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Ni X.F., Xie Q.Q., Zhao J.M., Xu Y.J., Ji M., Hu W.W., Wu J., Wu C.P. The hepatic microenvironment promotes lung adenocarcinoma cell proliferation, metastasis, and epithelial–mesenchymal transition via METTL3-mediated N6-methyladenosine modification of YAP. Aging. 2021;13:4357–4369. doi: 10.18632/aging.202397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Jin D., Guo J., Wu Y., Du J., Yang L., Wang X., Di W., Hu B., An J., Kong L., et al. m6A mRNA methylation initiated by METTL3 directly promotes YAP translation and increases YAP activity by regulating the MALAT1-miR-1914–3p-YAP axis to induce NSCLC drug resistance and metastasis. J. Hematol. Oncol. 2019;12:1–22. doi: 10.1186/s13045-019-0830-6. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 71.Chen W.W., Qi J.W., Hang Y., Wu J.X., Zhou X.X., Chen J.Z., Wang J., Wang H.-H. Simvastatin is beneficial to lung cancer progression by inducing METTL3-induced m6A modification on EZH2 mRNA. Eur. Rev. Med. Pharmacol. Sci. 2020;24:4263–4270. doi: 10.26355/eurrev_202004_21006. [DOI] [PubMed] [Google Scholar]
- 72.Wanna-Udom S., Terashima M., Lyu H., Ishimura A., Takino T., Sakari M., Tsukahara T., Suzuki T. The m6A methyltransferase METTL3 contributes to Transforming Growth Factor-beta-induced epithelial-mesenchymal transition of lung cancer cells through the regulation of JUNB. Biochem. Biophys. Res. Commun. 2020;524:150–155. doi: 10.1016/j.bbrc.2020.01.042. [DOI] [PubMed] [Google Scholar]
- 73.Shen Y., Li C., Zhou L., Huang J. G protein-coupled oestrogen receptor promotes cell growth of non-small cell lung cancer cells via YAP1/QKI/circNOTCH1/m6A methylated NOTCH1 signalling. J. Cell. Mol. Med. 2020;25:284–296. doi: 10.1111/jcmm.15997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhao W., Xie Y. KIAA1429 promotes the progression of lung adenocarcinoma by regulating the m6A level of MUC3A. Pathol. Res. Pract. 2021;217:153284. doi: 10.1016/j.prp.2020.153284. [DOI] [PubMed] [Google Scholar]
- 75.Jin D., Guo J., Wu Y., Yang L., Wang X., Du J., Dai J., Chen W., Gong K., Miao S., et al. m6A demethylase ALKBH5 inhibits tumor growth and metastasis by reducing YTHDFs-mediated YAP expression and inhibiting miR-107/LATS2–mediated YAP activity in NSCLC. Mol. Cancer. 2020;19:1–24. doi: 10.1186/s12943-020-01161-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Guo J., Wu Y., Du J., Yang L., Chen W., Gong K., Dai J., Miao S., Jin D., Xi S. Deregulation of UBE2C-mediated autophagy repression aggravates NSCLC progression. Oncogenesis. 2018;7:1–16. doi: 10.1038/s41389-018-0054-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chao Y., Shang J., Ji W. ALKBH5-m(6)A-FOXM1 signaling axis promotes proliferation and invasion of lung adenocarcinoma cells under intermittent hypoxia. Biochem. Biophys. Res. Commun. 2020;521:499–506. doi: 10.1016/j.bbrc.2019.10.145. [DOI] [PubMed] [Google Scholar]
- 78.Zhu Z., Qian Q., Zhao X., Ma L., Chen P. N6-methyladenosine ALKBH5 promotes non-small cell lung cancer progress by regulating TIMP3 stability. Gene. 2020;731:144348. doi: 10.1016/j.gene.2020.144348. [DOI] [PubMed] [Google Scholar]
- 79.Li J., Han Y., Zhang H., Qian Z., Jia W., Gao Y., Zheng H., Li B. The m6A demethylase FTO promotes the growth of lung cancer cells by regulating the m6A level of USP7 mRNA. Biochem. Biophys. Res. Commun. 2019;512:479–485. doi: 10.1016/j.bbrc.2019.03.093. [DOI] [PubMed] [Google Scholar]
- 80.Liu J., Ren D., Du Z., Wang H., Zhang H., Jin Y. m 6 A demethylase FTO facilitates tumor progression in lung squamous cell carcinoma by regulating MZF1 expression. Biochem. Biophys. Res. Commun. 2018;502:456–464. doi: 10.1016/j.bbrc.2018.05.175. [DOI] [PubMed] [Google Scholar]
- 81.Sheng H., Li Z., Su S., Sun W., Zhang X., Li L., Li J., Liu S., Lu B., Zhang S., et al. YTH domain family 2 promotes lung cancer cell growth by facilitating 6-phosphogluconate dehydrogenase mRNA translation. Carcinogenesis. 2020;41:541–550. doi: 10.1093/carcin/bgz152. [DOI] [PubMed] [Google Scholar]
- 82.Ma L., Chen T., Zhang X., Miao Y., Tian X., Yu K., Xu X., Niu Y., Guo S., Zhang C., et al. The m6A reader YTHDC2 inhibits lung adenocarcinoma tumorigenesis by suppressing SLC7A11-dependent antioxidant function. Redox Biol. 2021;38:101801. doi: 10.1016/j.redox.2020.101801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Xue L., Li J., Lin Y., Liu D., Yang Q., Jian J., Peng J. m(6) A transferase METTL3-induced lncRNA ABHD11-AS1 promotes the Warburg effect of non-small-cell lung cancer. J. Cell. Physiol. 2020;236:2649–2658. doi: 10.1002/jcp.30023. [DOI] [PubMed] [Google Scholar]
- 84.Wang H., Deng Q., Lv Z., Ling Y., Hou X., Chen Z., Dinglin X., Ma S., Li D., Wu Y., et al. N6-methyladenosine induced miR-143–3p promotes the brain metastasis of lung cancer via regulation of VASHMol. Cancer. 2019;18:1–17. doi: 10.1186/s12943019-1108-x. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 85.Song H., Li H., Ding X., Li M., Shen H., Li Y., Zhang X., Xing L. Long noncoding RNA FEZF1AS1 facilitates nonsmall cell lung cancer progression via the ITGA11/miR516b5p axis. Int. J. Oncol. 2020;57:1333–1347. doi: 10.3892/ijo.2020.5142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sardo F.L., Strano S., Blandino G. YAP and TAZ in Lung Cancer: Oncogenic Role and Clinical Targeting. Cancers. 2018;10:137. doi: 10.3390/cancers10050137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cao R., Zhang Y. The functions of E(Z)/EZH2-mediated methylation of lysine 27 in histone H3. Curr. Opin. Genet. Dev. 2004;14:155–164. doi: 10.1016/j.gde.2004.02.001. [DOI] [PubMed] [Google Scholar]
- 88.Chang H., Liu Y., Xue M., Liu H., Du S., Zhang L., Wang P. Synergistic action of master transcription factors controls epithelial-to-mesenchymal transition. Nucleic Acids Res. 2016;44:2514–2527. doi: 10.1093/nar/gkw126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Cairns R.A., Harris I.S., Mak T.W. Regulation of cancer cell metabolism. Nat. Rev. Cancer. 2011;11:85–95. doi: 10.1038/nrc2981. [DOI] [PubMed] [Google Scholar]
- 90.Lin R., Elf S., Shan C., Kang H.B., Ji Q., Zhou L., Hitosugi T., Zhang L., Zhang S., Seo J.H., et al. 6-Phosphogluconate dehydrogenase links oxidative PPP, lipogenesis and tumour growth by inhibiting LKB1–AMPK signalling. Nat. Cell Biol. 2015;17:1484–1496. doi: 10.1038/ncb3255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Niu T., Liu Y., Zhang Y., Fu Q., Liu Z., Wang Z., Fu H., Xu J., Liu K. Increased expression of MUC3A is associated with poor prognosis in localized clear-cell renal cell carcinoma. Oncotarget. 2016;7:50017–50026. doi: 10.18632/oncotarget.10312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Kitamoto S., Yamada N., Yokoyama S., Houjou I., Higashi M., Yonezawa S. Promoter hypomethylation contributes to the expression of MUC3A in cancer cells. Biochem. Biophys. Res. Commun. 2010;397:333–339. doi: 10.1016/j.bbrc.2010.05.124. [DOI] [PubMed] [Google Scholar]
- 93.Burger A.M., Seth A.K. The ubiquitin-mediated protein degradation pathway in cancer: Therapeutic implications. Eur. J. Cancer. 2004;40:2217–2229. doi: 10.1016/j.ejca.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 94.Kadara H., Lacroix L., Behrens C., Solis L., Gu X., Lee J.J., Tahara E., Lotan D., Hong W.K., Wistuba I.I., et al. Identification of Gene Signatures and Molecular Markers for Human Lung Cancer Prognosis using an In vitro Lung Carcinogenesis System. Cancer Prev. Res. 2009;2:702–711. doi: 10.1158/1940-6207.CAPR-09-0084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Cheng Z., Yu C., Cui S., Wang H., Jin H., Wang C., Li B., Qin M., Yang C., He J., et al. circTP63 functions as a ceRNA to promote lung squamous cell carcinoma progression by upregulating FOXM Nat. Commun. 2019;10:3200. doi: 10.1038/s41467-019-11162-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Chetty C., Lakka S.S., Bhoopathi P., Kunigal S., Geiss R., Rao J.S. Tissue Inhibitor of Metalloproteinase 3 Suppresses Tumor Angiogenesis in Matrix Metalloproteinase 2–Down-regulated Lung Cancer. Cancer Res. 2008;68:4736–4745. doi: 10.1158/0008-5472.CAN-07-6612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Visse R., Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: Structure, function, and biochemistry. Circ. Res. 2003;92:827–839. doi: 10.1161/01.RES.0000070112.80711.3D. [DOI] [PubMed] [Google Scholar]
- 98.Nicholson B., Kumar K.G.S. The Multifaceted Roles of USP7: New Therapeutic Opportunities. Cell Biophys. 2011;60:61–68. doi: 10.1007/s12013-011-9185-5. [DOI] [PubMed] [Google Scholar]
- 99.Zhang C., Lu J., Zhang Q.W., Zhao W., Guo J.H., Liu S.L., Wu Y.L., Jiang B., Gao F.H. USP7 promotes cell proliferation through the stabilization of Ki-67 protein in non-small cell lung cancer cells. Int. J. Biochem. Cell Biol. 2016;79:209–221. doi: 10.1016/j.biocel.2016.08.025. [DOI] [PubMed] [Google Scholar]
- 100.Tsai L.H., Wu J.Y., Cheng Y.W., Chen C.Y., Sheu G.T., Wu T.C., Lee H. The MZF1/c-MYC axis mediates lung adenocarcinoma progression caused by wild-type lkb1 loss. Oncogene. 2014;34:1641–1649. doi: 10.1038/onc.2014.118. [DOI] [PubMed] [Google Scholar]
- 101.Yuan B., Liao F., Shi Z.Z., Ren Y., Deng X.L., Yang T.T., Li D.Y., Li R.F., Pu D.D., Wang Y.J., et al. Dihydroartemisinin Inhibits the Proliferation, Colony Formation and Induces Ferroptosis of Lung Cancer Cells by Inhibiting PRIM2/SLC7A11 Axis. OncoTargets Ther. 2020;13:10829–10840. doi: 10.2147/OTT.S248492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Gan R.H., Wei H., Xie J., Zheng D.P., Luo E.L., Huang X.Y., Xie J., Zhao Y., Ding L.C., Su B.H., et al. Notch1 regulates tongue cancer cells proliferation, apoptosis and invasion. Cell Cycle. 2017;17:216–224. doi: 10.1080/15384101.2017.1395534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Liu J., Liu S., Deng X., Rao J., Huang K., Xu G., Wang X. MicroRNA-582–5p suppresses non-small cell lung cancer cells growth and invasion via downregulating NOTCH1. PLoS ONE. 2019;14:e0217652. doi: 10.1371/journal.pone.0217652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Song J., Su Z.Z., Shen Q.-M. Long non-coding RNA MALAT1 regulates proliferation, apoptosis, migration and invasion via miR-374b-5p/SRSF7 axis in non-small cell lung cancer. Eur. Rev. Med. Pharmacol. Sci. 2020;24:1853–1862. doi: 10.26355/eurrev_202002_20363. [DOI] [PubMed] [Google Scholar]
- 105.Zeng X., Jiang X., Yong J., Xie H., Yuan J., Zeng D., Dou Y., Xiao S. lncRNA ABHD11-AS1, regulated by the EGFR pathway, contributes to the ovarian cancer tumorigenesis by epigenetically suppressing TIMP2. Cancer Med. 2019;8:7074–7085. doi: 10.1002/cam4.2586. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 106.Zhuang X., Tong H., Ding Y., Wu L., Cai J., Si Y., Zhang H., Shen M. Long noncoding RNA ABHD11-AS1 functions as a competing endogenous RNA to regulate papillary thyroid cancer progression by miR-199a-5p/SLC1A5 axis. Cell Death Dis. 2019;10:1–18. doi: 10.1038/s41419-019-1850-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.He D., Yue Z., Liu L., Fang X., Chen L., Han H. Long noncoding RNA ABHD11-AS1 promote cells proliferation and invasion of colorectal cancer via regulating the miR-1254-WNT11 pathway. J. Cell. Physiol. 2019;234:12070–12079. doi: 10.1002/jcp.27877. [DOI] [PubMed] [Google Scholar]
- 108.Jiang W., Tian Y., Jiang S., Liu S., Zhao X., Tian D. MicroRNA-376c suppresses non-small-cell lung cancer cell growth and invasion by targeting LRH-1-mediated Wnt signaling pathway. Biochem. Biophys. Res. Commun. 2016;473:980–986. doi: 10.1016/j.bbrc.2016.04.002. [DOI] [PubMed] [Google Scholar]
- 109.Zhou J., Xiao D., Qiu T., Li J., Liu Z. Loading MicroRNA-376c in Extracellular Vesicles Inhibits Properties of Non-Small Cell Lung Cancer Cells by Targeting YTHDF1. Technol. Cancer Res. Treat. 2020;19 doi: 10.1177/1533033820977525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Chen Z., Chen X., Lei T., Gu Y., Gu J., Huang J., Lu B., Yuan L., Sun M., Wang Z. Integrative Analysis of NSCLC Identifies LINC01234 as an Oncogenic lncRNA that Interacts with HNRNPA2B1 and Regulates miR-106b Biogenesis. Mol. Ther. 2020;28:1479–1493. doi: 10.1016/j.ymthe.2020.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Li B., Zhu L., Lu C., Wang C., Wang H., Jin H., Ma X., Cheng Z., Yu C., Wang S., et al. circNDUFB2 inhibits non-small cell lung cancer progression via destabilizing IGF2BPs and activating anti-tumor immunity. Nat. Commun. 2021;12:295. doi: 10.1038/s41467-020-20527-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Mu X., Zhao Q., Chen W., Zhao Y., Yan Q., Peng R., Zhu J., Yang C., Lan K., Gu X., et al. IL-37 Confers Anti-Tumor Activity by Regulation of m6A Methylation. Front. Oncol. 2021;10 doi: 10.3389/fonc.2020.526866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Xiao P., Liu Y.K., Han W., Hu Y., Zhang B.Y., Liu W.L. Exosomal Delivery of FTO Confers Gefitinib Resistance to Recipient Cells through ABCC10 Regulation in an m6A-dependent Manner. Mol. Cancer Res. 2021 doi: 10.1158/1541-7786.MCR-20-0541. [DOI] [PubMed] [Google Scholar]
- 114.Yang L., Li J., Xu L., Lin S., Xiang Y., Dai X., Liang G., Huang X., Zhu J., Zhao C. Rhein shows potent efficacy against non-small-cell lung cancer through inhibiting the STAT3 pathway. Cancer Manag. Res. 2019;11:1167–1176. doi: 10.2147/CMAR.S171517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Huang Y., Yan J., Li Q., Li J., Gong S., Zhou H., Gan J., Jiang H., Jia G.F., Luo C., et al. Meclofenamic acid selectively inhibits FTO demethylation of m6A over ALKBH. Nucleic Acids Res. 2015;43:373–384. doi: 10.1093/nar/gku1276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Huang Y., Su R., Sheng Y., Dong L., Dong Z., Xu H., Ni T., Zhang Z.S., Zhang T., Li C., et al. Small-Molecule Targeting of Oncogenic FTO Demethylase in Acute Myeloid Leukemia. Cancer Cell. 2019;35:677–691. doi: 10.1016/j.ccell.2019.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Meng Q., Wang S., Zhou S., Liu H., Ma X., Zhou X., Liu H., Xu C., Jiang W. Dissecting the m6A methylation affection on afatinib resistance in non-small cell lung cancer. Pharm. J. 2020;20:227–234. doi: 10.1038/s41397-019-0110-4. [DOI] [PubMed] [Google Scholar]
- 118.Shi Y., Fan S., Wu M., Zuo Z., Li X., Jiang L., Shen Q., Xu P., Zeng L., Zhou Y., et al. YTHDF1 links hypoxia adaptation and non-small cell lung cancer progression. Nat. Commun. 2019;10:1–14. doi: 10.1038/s41467-019-12801-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zhuang Z., Chen L., Mao Y., Zheng Q., Li H., Huang Y., Hu Z., Jin Y. Diagnostic, progressive and prognostic performance of m6A methylation RNA regulators in lung adenocarcinoma. Int. J. Biol. Sci. 2020;16:1785–1797. doi: 10.7150/ijbs.39046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Guo W., Huai Q., Wan H., Guo L., Song P., Gao S., He J. Prognostic Impact of IGF2BP3 Expression in Patients with Surgically Resected Lung Adenocarcinoma. DNA Cell Biol. 2021;40:316–331. doi: 10.1089/dna.2020.6136. [DOI] [PubMed] [Google Scholar]
- 121.Huang W., Qi C.B., Lv S.W., Xie M., Feng Y.Q., Huang W.H., Yuan B.F. Determination of DNA and RNA Methylation in Circulating Tumor Cells by Mass Spectrometry. Anal. Chem. 2016;88:1378–1384. doi: 10.1021/acs.analchem.5b03962. [DOI] [PubMed] [Google Scholar]
- 122.Li S., Xu Z., Sheng J. tRNA-Derived Small RNA: A Novel Regulatory Small Non-Coding RNA. Genes. 2018;9:246. doi: 10.3390/genes9050246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Masuda T., Fujimoto H., Teranaka R., Kuroda M., Aoyagi Y., Nagashima T., Sangai T., Takada M., Nakagawa A., Kubota Y., et al. Anti-HER2 antibody therapy using gene-transduced adipocytes for HER2-positive breast cancer. Breast Cancer Res. Treat. 2020;180:625–634. doi: 10.1007/s10549-020-05581-x. [DOI] [PubMed] [Google Scholar]
- 124.Toulany M. Targeting DNA Double-Strand Break Repair Pathways to Improve Radiotherapy Response. Genes. 2019;10:25. doi: 10.3390/genes10010025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Liu Y., Guo X., Zhao M., Ao H., Leng X., Liu M., Wu C., Ma J., Zhu J. Contributions and prognostic values of m 6 A RNA methylation regulators in non-small-cell lung cancer. J. Cell. Physiol. 2020;235:6043–6057. doi: 10.1002/jcp.29531. [DOI] [PubMed] [Google Scholar]
- 126.Zhu J., Wang M., Hu D. Deciphering N6-Methyladenosine-Related Genes Signature to Predict Survival in Lung Adenocarcinoma. BioMed Res. Int. 2020;2020:2514230. doi: 10.1155/2020/2514230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Du M., Zhang Y., Mao Y., Mou J., Zhao J., Xue Q., Wang D., Huang J., Gao S., Gao Y. MiR-33a suppresses proliferation of NSCLC cells via targeting METTL3 mRNA. Biochem. Biophys. Res. Commun. 2017;482:582–589. doi: 10.1016/j.bbrc.2016.11.077. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data sharing not applicable.


