Abstract
Mouse embryonic stem cells (mESCs) and mouse epiblast stem cells (mEpiSCs) are the pluripotent stem cells (PSCs), derived from the inner cell mass (ICM) of preimplantation embryos at embryonic day 3.5 (E3.5) and postimplantation embryos at E5.5-E7.5, respectively. Depending on their environment, PSCs can exist in the so-called naïve (ESCs) or primed (EpiSCs) states. Exposure to EpiSC or human ESC (hESC) culture condition can convert mESCs towards an EpiSC-like state. Here, we show that the undifferentiated epiblast state is however not stabilized in a sustained manner when exposing mESCs to hESC or EpiSC culture condition. Rather, prolonged exposure to EpiSC condition promotes a transition to a primitive streak- (PS-) like state via an unbiased epiblast-like intermediate. We show that the Brachyury-positive PS-like state is likely promoted by endogenous WNT signaling, highlighting a possible species difference between mouse epiblast-like stem cells and human Embryonic Stem Cells.
1. Introduction
Pluripotency is the intrinsic, unrestricted, flexible developmental potential of the embryonic cells in a developing embryo, to give rise to the three embryonic germ layers, ultimately forming all the cells in an adult organism. This can be captured in vitro, by deriving pluripotent stem cells (PSCs) from various developmental stages. The PSCs, derived from the epiblast of preimplantation mouse embryos (E3.75-E4.5) are called embryonic stem cells (ESCs) [1–3]. The mESCs can be brought to a so-called ground/naïve state of pluripotency, using leukaemia inhibitory factor (LIF) that sustains self-renewal [4, 5], in conjunction with the inhibition of ERK [2] and GSK3 [6] that simultaneously suppress differentiation (LIF/2i medium) [7]. The PSCs that are derived from the postimplantation embryos (E5.5-E7.5) are called the epiblast stem cells (EpiSCs), which are in a primed state of pluripotency [1, 8]. The mouse EpiSCs and human ESCs (hESCs) are conventionally cultured in Activin A and FGF2 (AA/F2). When the mESCs are exposed to the hESC/EpiSC condition (AA/F2), they transition to an EpiSC-like primed state [1, 8–11].
During gastrulation, the pluripotent epiblast cells in the developing mouse embryo undergo epithelial-mesenchymal transition (EMT) and migrate through the primitive streak (PS), forming mesendoderm cells, the common precursors of mesoderm and endoderm [12]. The epiblast cells that do not migrate through the PS form the neuroectoderm. Several signaling pathways play crucial roles in this rearrangement process, such as TGFβ/activin/nodal, WNT/β-catenin, and FGF/ERK/MAPK signaling pathways [13–15]. The collaborative interaction between various transcription factors, such as the T-box transcription factors (Tbx) T and Eomes with the key markers of mesoderm and endoderm initiate this differentiation process [16–18].
Currently, a range of pluripotent states is being discovered [19, 20], and in spite of the fact that the EpiSCs share many properties [21] that safeguard their pluripotency, they possess unique functional and molecular properties that set them apart [22, 23]. This difference probably reflects on the transformations that the epiblast cells undergo, based on their spatiotemporal positions and their environment in the developing embryo, restricting their developmental potential to certain lineages. Under the currently known extrinsic culture conditions, the EpiSCs could possible occur in heterogeneous metastable states, possessing variable differentiation potentials [24, 25].
The standard hESC culture condition has been adapted to support mEpiSCs (Activin A and FGF2 (AA/F2)) [1, 8–11], and the same can convert mESCs in the ground/naïve state of pluripotency to a primed EpiSC-like state. This study is aimed at understanding the transition from naïve to primed state of pluripotency, under the standard hESC/EpiSC condition. In this study, we show that although this condition matured the mESCs to distinct primed EpiSC-like states, these states could not be sustained, but the cells further matured to a PS/mesendoderm-like state, highlighting a possible species difference between mouse and human epiblast-like stem cells. This also highlights the requirement of additional factors for the preservation of distinct pluripotency states of the mouse EpiSCs, in contrast to the requirement of hESCs that could enhance our capability to differentiate them into any desired cell type in high proportion.
2. Materials and Methods
2.1. Mouse ESC Lines
The experiments were carried out using the mouse embryonic stem cells (mESCs) from C57BL/6N background, having fluorescent reporters for Brachyury (T) and Sox2 [26]. Mouse bacterial artificial chromosomes (BACs) containing the part of the mouse genome comprising the locus of the specific gene (BACs RP24-530D23 (T) and RP23-249O15 (Sox2) containing ∼180-230 kb of the C57BL/6 mouse genome surrounding the respective loci), by Red/ET recombineering (GeneBridges) [27]. Shortly, the start codon (ATG) of T or Sox2 was replaced with mCherry or Venus coding sequence, followed by the rabbit b-globin polyadenylation signal and an FRT-site flanked hygromycin selection cassette, driven by the Pgk promoter. 5 mg of the modified BAC was linearized with the restriction enzyme PI-SceI (NEB) and electroporated into 3 × 106 ESCs. After selection in ES cell medium containing hygromycin (150 mg/ml), clones were picked, expanded, and checked by PCR for BAC integration. For details, refer [26].
2.2. Cell Culture and Differentiation
ESCs and EpiSCs were cultured according to standard conditions [24]. For naïve conversion, the feeder-free mESCs were seeded on fibronectin- (3-5 ng/ml) (Merck-Millipore; Calbiochem) coated plates in N2B27 medium containing LIF, PD0325901 (1 μM) (Axon Medchem, Axon 1408), and CHIR99021 (GSK3β inhibitor) (3 μM) (Axon Medchem, Axon 1386) (2i medium) [7]. The next day, the cells were refed with the 2i medium. After the small dome-shaped colonies were seen, the medium was changed to N2B27 medium supplemented with Activin A (20 ng/ml) and FGF2 (10 ng/ml) (AA/F2). The AA/F2 treatment was continued for 6 days, while the medium is replaced with fresh AA/F2 medium every day.
2.3. Whole Transcriptome Data Analysis
RNA sequencing was carried out with 80 ng of total RNA. After quantification and quality assessment (details in [24]), the RNA-seq library was prepared from total RNA using the ScriptSeq Complete Kit (Human/Mouse/Rat)—Low Input (SCL24H) (Illumina), according to manufacturer's instructions. The prepared sequencing library was eventually sequenced on Illumina HiSeq 2000 for 2350 cycles (paired-end), following the standard protocol. The RNA-seq sequencing reads were mapped to the mouse genome (mm10) using TopHat (version 2.0.11) [28]. Cuffdiff was then used to calculate the normalized FPKM (fragments per kilobase per million mapped read) values for genes in all samples [29]. The results were filtered by removing genes with FPKM values lower than 1 in all samples, which were then used for further analysis (R statistical program) (http://www.r-project.org). The RNA-seq data has been deposited in the ArrayExpress database (E-MTAB-3784).
2.4. Real-Time PCR Analysis
Total RNA was isolated using RNeasy Micro and Mini Kits (Qiagen), followed by quantification using NanoDrop (Life Technologies) (unsorted cells) or Qubit fluorometer (Invitrogen) (sorted cells). The unsorted cells were directly lysed with the RLT buffer in the cell-culture wells, after rinsing once with PBS. An extra step of DNase I (Roche, Basel, Switzerland, https://www.roche-applied-science.com) treatment was carried out in order to avoid genomic DNA contamination. RNA was quantified using the spectrophotometer from either NanoDrop Technologies (unsorted cells) or Qubit (Invitrogen, Carlsbad, CA, http://www.invitrogen.com) (FACS-sorted cells). Reverse transcription was performed with the M-MLV reverse transcriptase (Promega) and Oligo-dT primers (Invitrogen) (Table S3). Quantitative reverse transcriptase PCR (qRT-PCR) was done using GoTaq qPCR Master Mix (Promega) with the help of validated gene-specific primers on StepOnePlus Real-Time PCR System (Life Technologies, Rockville, MD, https://www.lifetech.com). Data analysis was accomplished using the ΔΔCt method, with the housekeeping genes, Pmm2 and Gapdh and/or Actb for normalization.
2.5. Fluorescence-Activated Cell Sorting (FACS)
The cells were gently rinsed with phosphate-buffered saline (PBS) and detached using ice cold trypsin. In a neutralization with cold knockout DMEM-F12, the cells were quickly (30 seconds) spun down at high speed (10,000 rpm) and resuspended in cold knockout DMEM-F12 and immediately placed on ice. The cells were then sorted on FACS Aria II (Becton Dickinson, Franklin Lakes, NJ, https://www.bd.com) directly into RLT buffer containing β-mercaptoethanol, followed by total RNA isolation using the RNeasy Micro Kit (Qiagen, Hilden, Germany, http://www1.qiagen.com) and processed further.
2.6. Immunoblotting and Immunocytochemistry
Standard procedures were followed for these assays (details in [24]) with the suitable antibodies (Table S4). The cells were initially rinsed with PBS, before immunoblotting and immunocytochemistry.
For immunoblotting, after adding the lysis buffer (containing protease inhibitor and phosphatase inhibitor (PhosStop, phosphatase inhibitor cocktail Easypack, Roche)), the cells were scraped off and lysed further, after placing on ice, to preserve the phosphorylation status of the proteins. Western blot data were quantified using ImageJ, a Java-based image analysis package. H3 and Actb served as controls.
For immunocytochemistry, the cells in monolayer culture were washed with PBS and fixed with 4% paraformaldehyde. After permeabilization, primary and secondary antibody staining were carried out. Visualization was done using a confocal microscope (LSM510 Meta, Zeiss), and further analysis was performed using the software, AxionVision (Zeiss).
3. Results
3.1. Human ESC/EpiSC Culture Condition Leads Naïve ESCs to Distinct EpiSC-Like States
LIF/2i (LIF, PD0325901 (MEK inhibitor) and CHIR99021 (GSK3β inhibitor)), under defined conditions, can maintain mESCs in their naïve state of pluripotency [4, 5, 7] and AA/F2 (Activin A and FGF2) can transform them into a primed-like state [1, 8, 10]. A time course analysis for 3 days was performed to understand this transition of pluripotency states (Figure 1(a)). Round, dome-shaped colonies, characteristics of naïve ESCs, were observed when the mESCs were grown in LIF/2i. Upon AA/F2 exposure, mESCs gradually lost their dome-shaped morphology and grew as flat colonies. Expression of Rex1 is one of the hallmarks of the ground state [30]. Among the Oct4/Sox2/Nanog triumvirate, Oct4 and Sox2, defined as the core-pluripotency factors, are required for both the acquisition and maintenance of pluripotency [31], whereas Nanog is required only for the acquisition of pluripotency [32, 33]. AA/F2 treatment led to a decrease in the expression of Rex1 and Nanog right from the first day, Sox2 from second day still retaining the expression of Oct4 [31, 34, 35] till 3 days, showing the developmental advancement of the cells during the treatment (Figures 2(a), 2(b), 2(e), and 2(f), S1, Table S1). While Nanog expression is restricted exclusively to the nascent epiblast, Oct4 and Sox2 are ubiquitously expressed in the morula and blastocyst in all the cells of the inner cell mass (ICM), till the hypoblast segregates [36]. Whole transcriptome data revealed that the naïve-specific genes were downregulated, and the primed genes were induced during the treatment (Figures 2(a) and 2(b), Table S1). However, PCA and hierarchical clustering showed that the AA/F2-2D and AA/F2-3D cells were not only distinct from each other, but also from both the naïve ESCs and the EpiSCs (Figures 1(b)–1(d), S1, Table S1). This is a time when a spectrum of pluripotent states is being discovered [19, 20], and the mEpiSCs can be derived from E5.5 to E7.5 embryos that are similar in several molecular characteristics [21] and may also differ in the expression status of certain genes [22]. Taken together, our results demonstrate that the primed EpiSC-like states that were derived by the exposure of naïve ESCs to AA/F2 are distinct from each other and from the EpiSCs used in this study. The EpiSCs used here have been derived from early postimplantation E5.5 mouse embryos [9].
Figure 1.
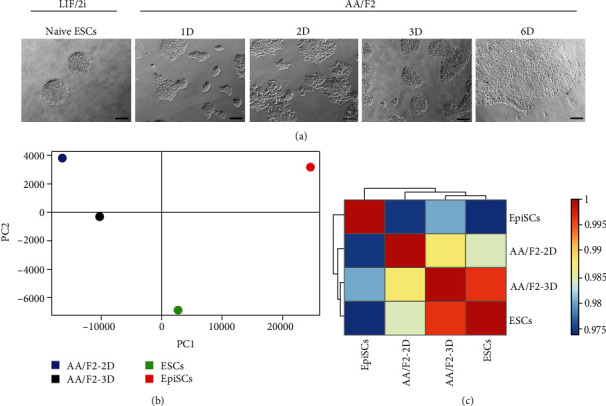
Human ESC/EpiSC culture condition leads naïve ESCs to distinct EpiSC-like states. (a) Brightfield images of the indicated samples (scale bars 100 μm). mESCs were brought to naïve state with LIF/2i (LIF+2i (PD0325901+CHIR99021)), followed by AA/F2 treatment. This treatment results in morphological changes from round dome-shaped to flat colonies. (b) Principal component analysis (PCA) of the indicated samples. (c) Heat map, based on Pearson's correlation coefficient. AA/F2 treatment for different durations led naïve ESCs to primed EpiSC states that differed from each other and the PSCs that were compared here.
Figure 2.
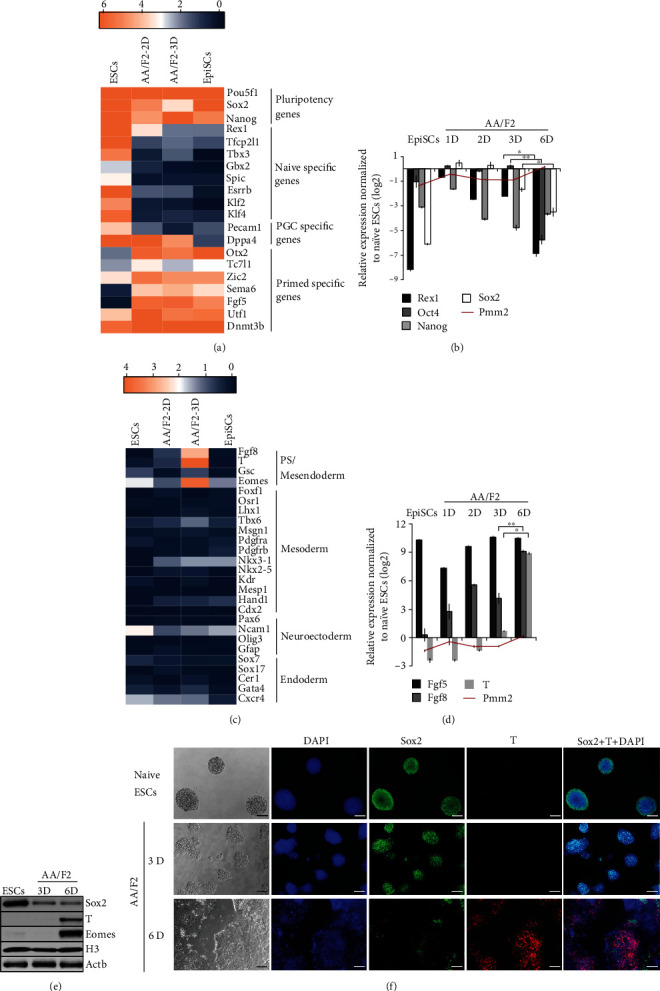
Human ESC/EpiSC culture condition matures the ESCs further to a primitive streak-like state. (a) Expression levels of the specific pluripotency-associated genes in the indicated samples. AA/F2 treatment led to the induction of primed pluripotency genes and downregulation of PGC and naïve pluripotency genes. (b) Analysis of pluripotency genes in AA/F2-treated cells in the indicated samples. Gapdh and Pmm2: housekeeping genes. Rex1, Sox2, and Nanog were downregulated, and Oct4 expression was retained. Error bars: mean ± SD (n = 2); ∗p ≤ 0.05, ∗∗p ≤ 0.01, ∗∗∗p ≤ 0.001, and ∗∗∗∗p ≤ 0.0001 (p (Rex1) = 0.0107, p(Sox2) = 0.0421, and p(Oct4) = 0.0029) (Student's t-test). (c) Heat map showing the expression status of lineage-specific markers in the indicated samples in comparison with naïve mESCs. PS markers are induced in AA/F2-3 days (3D). (d) Analysis of EpiSCs (Fgf5) and PS markers (T and Fgf8) in the indicated samples. Fgf5 and Fgf8 were induced right from 1D, and by 6D, the PS marker T was also highly induced. Error bars: mean ± SD (n = 3); ∗p ≤ 0.05, ∗∗p ≤ 0.01, ∗∗p ≤ 0.001, and ∗∗∗∗p ≤ 0.0001 (p(Fgf8) = 0.0070 and p(T) = 0.0235) (Student's t-test). (e) Western blot analysis of the indicated markers in the indicated samples. Six days of AA/F2 treatment led to high protein expression of the PS/mesendoderm markers T and Eomes and downregulation of Sox2. (f) Representative immunostainings of the indicated proteins in the indicated samples (scale bars 100 μm).
3.2. Human ESC/EpiSC Culture Condition Matures the ESCs Further to Primitive Streak-Like (PS-Like) State
The changes in pluripotency genes by three days coincided with the induction of T, Eomes, and Fgf8, and the genes were expressed in the PS (Figures 2(a)–2(e), Table S1). At protein level, the induction of T was low, which could be attributed to a probable delay between transcription and translation. At this point (AA/F2-3D), no significant induction of any of the lineage-specific markers, mesoderm (Foxf1, Gata6, Osr1, Tbx6, and Msgn1), endoderm (Sox17), neuroectoderm (Pax6 and Sox1), or primitive endoderm (Sox7) had occurred (Figure 2(c), Table S1). The EpiSCs derived from late postimplantation embryos (E7.5) have reduced neural induction potential and express T and OCT4 [22]. Therefore, AA/F2 treatment for 3 days matured ESCs from the naïve pluripotency to a more matured primed EpiSC-like state.
EpiSCs are routinely maintained using AA/F2 [1]. To know if this condition will maintain the converted EpiSC-like cells, the AA/F2 treatment was continued for 6 days. This led to the downregulation of the core pluripotency factors, Oct4, Sox2, and Nanog (Figures 2(b), 2(d), and 2(e), Table S1), and significant induction of the PS genes, Fgf8 and T (Figures 2(c)–2(e)). Taken together, although the EpiSC culture condition matures the naïve ESCs to a primed-like state, the continuous exposure of this condition for six days transforms these cells into a PS-like or mesendoderm-like state.
3.3. Epithelial-Mesenchymal Transition Occurs during Maturation to the PS-Like State
Epithelial-mesenchymal transition (EMT) and the migration of pluripotent epiblast cells through the PS facilitate the formation of mesoderm and endoderm progenitors [12]. Brachyury (T) is required for EMT and PS induction [37, 38] and is stimulated by the collaborative cross talk between FGF and WNT signaling [24, 39]. β-Catenin regulates PS induction by promoting SMAD2/3 activity [39, 40]. By 6 days of AA/F2 exposure, T, Eomes, and Fgf8 were highly induced (Figures 2(c)–2(e)), when SMAD2 was phosphorylated and β-catenin phosphorylation inhibited (Figure 3(a), S2A), reflecting on the active status of TGFβ/activin/nodal and canonical WNT signaling pathways. This coincided with a modest downregulation of the epithelial marker E-cadherin (CDH1) (Figures 3(b) and 3(c), S2D). We also found higher induction of AKT and total ERK in AA/F2-6D cells (Figure S2B-S2C). The epithelial cells in the epiblast express CDH1 which is repressed during PS formation, which in turn induces EMT [41]. Overall, the continued EpiSC condition for 6 days promotes EMT, a crucial process that occurs during PS formation.
Figure 3.
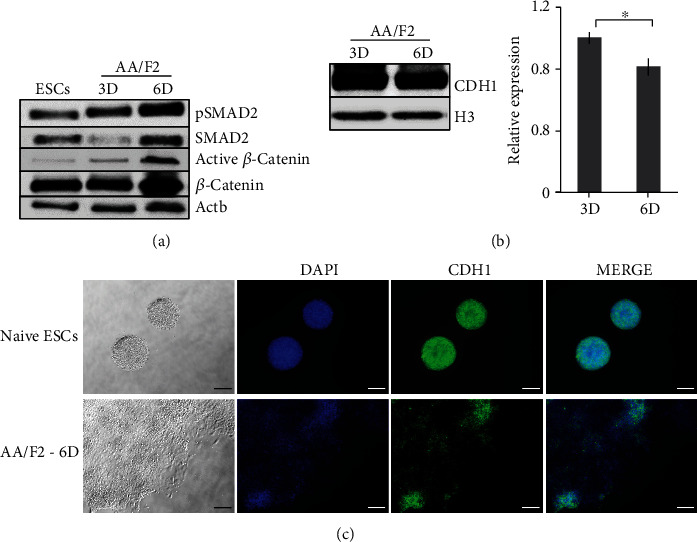
Epithelial-mesenchymal transition occurs during maturation to the PS-like state. (a) Western Blot analysis of the markers involved in WNT and TGFβ signaling pathways in the indicated samples (relative quantification graph: Figure S2A). Activin/nodal (pSMAD2) and WNT (active β-catenin) signaling were highly active by 6 days of AA/F2 treatment, compared to 3 days. Actb: control. (b) Western blot analysis of the epithelial marker CDH1 protein in the indicated samples. Histone H3 (used for normalization) and Actb (Figure S2D) served as controls. There was a slight decrease in the expression of CDH1. Error bars: mean ± SD (n = 2); ∗p ≤ 0.05; p(CDH1) = 0.0471 (Student's t-test). (c) Representative immunostainings of the indicated proteins in the cells treated with AA/F2 for 6 days (scale bars 100 μm).
3.4. Continuous Exposure to the hESC/EpiSC Condition Draws the PSCs towards Posterior Mesoderm and Endoderm Lineages
During early development, the cardiac mesoderm and endoderm arise from the anterior PS, whereas somitic and extraembryonic mesoderm originate from the posterior PS [42, 43]. AA/F2 treatment for 6 days resulted in the upregulation of endoderm (Sox17 and Gata6) and posterior mesoderm markers (Tbx6 and Msgn1) (Figures 4(a) and 4(b), S3), reflecting on a mixed population of cells. In the epiblast, while high activin/nodal signaling supports endoderm differentiation [18, 44], WNT/β-catenin supports posterior PS genes [45]. Both these pathways were active in the AA/F2-6D cells (Figure 3(a)), and at this stage, T was highly expressed (Figures 2(c)–2(e)), with considerable population of the cells expressing T (Figure 4(c)). However, the neuronal (Sox2, Pax6, and Sox1), lateral mesoderm (Foxf1), and the intermediate mesoderm (Osr1) (Figure 4(a)) genes were not noticeable expressed then. Endogenous WNT signaling in EpiSCs induces loss of pluripotency, with increase in the proportion of T-positive cells and differentiation tendency towards definitive endoderm [46]. Analysis of the sorted T-positive cells revealed that they expressed the epiblast (Fgf5), the posterior mesoderm (Fgf8, T, Msgn1, and Tbx6), and the endoderm (Foxa2 and Sox17) markers (Figure 4(d)). The pluripotency markers, Nanog and Sox2, were downregulated. In contrast, majority of the AA/F2-3D cells that were in the EpiSC-like state expressed Sox2 and there were no T-positive cells among them. This highlights the importance of early passaging of EpiSCs that might be able to avoid their further maturation towards PS/mesendoderm-like state in order to establish stable cell lines. These observations summarize that the continuous AA/F2 treatment leads the naïve PSCs to a primed EpiSC-like state, followed by PS- or mesendoderm-like state, supporting the endoderm and posterior mesoderm lineages (Figure 4(e)).
Figure 4.
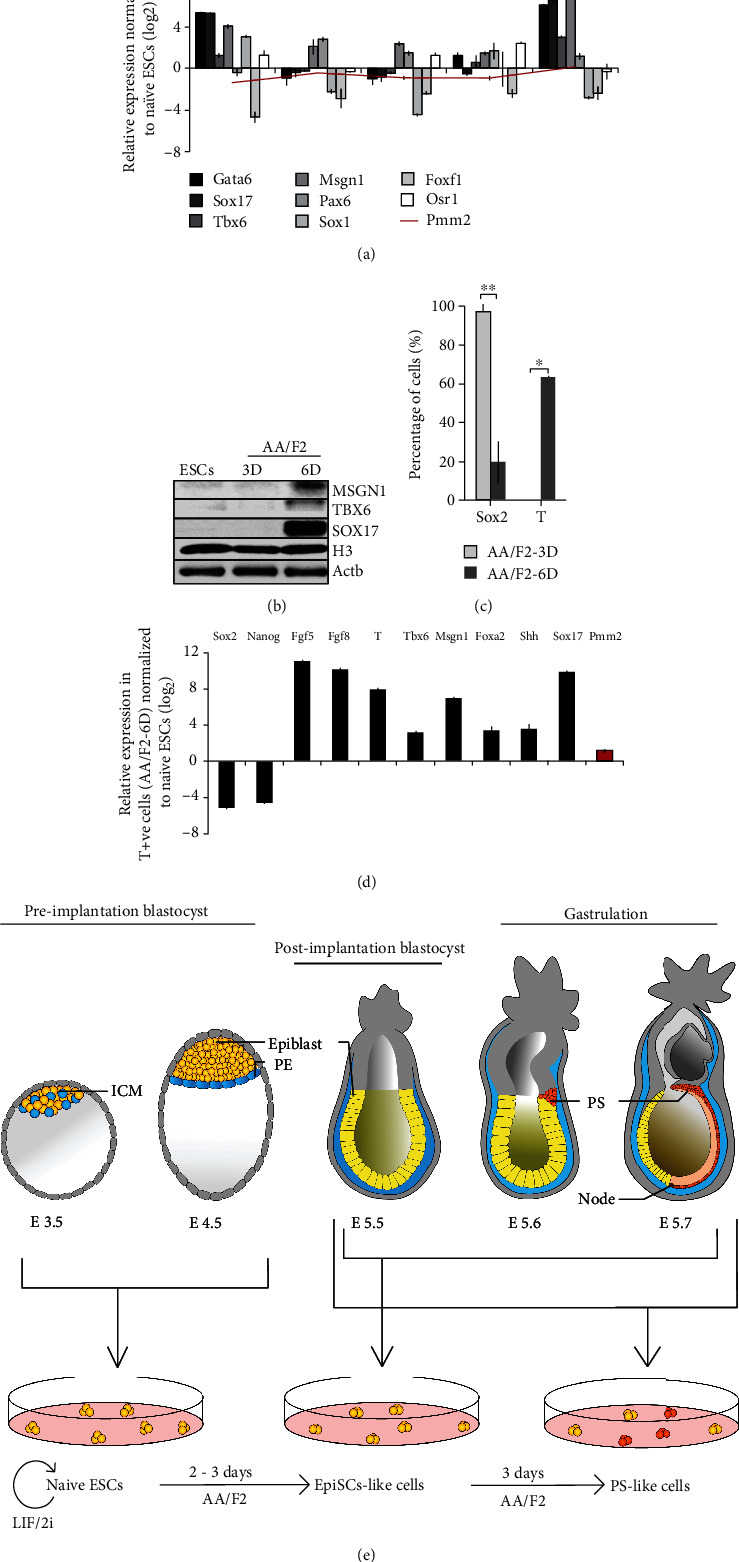
Continuous exposure to the hESC/EpiSC condition draws the PSCs towards posterior mesoderm and endoderm lineages. (a) qRT-PCR analysis of the indicated lineage-specific genes in the indicated samples. Gapdh and Pmm2: housekeeping genes. The endoderm (Gata6 and Sox17) and posterior mesoderm (Tbx6 and Msgn1) markers were induced by 6 days. The neuroectoderm (Pax6 and Sox1), lateral mesoderm (Foxf1), and intermediate mesoderm (Osr1) genes were not expressed. (b) Western blot analysis of the mesoderm (Msgn1 and Tbx6) and endoderm (Sox17) proteins in the cells treated with AA/F2 for the indicated durations. The posterior mesoderm (Msgn1 and Tbx6) and endoderm (Sox17) proteins were expressed by 6 days of the treatment. (c) Flow-cytometric analysis of the fraction of Sox2+ve and T+ve cells after 3 and 6 days of AA/F2 treatment. Error bars: mean ± SD (n = 2); ∗p ≤ 0.05 and ∗∗p ≤ 0.01 (p(Sox2) = 0.0070 and p(T) = 0.0098) (Student's t-test). By 6 days of treatment, the count of Sox2-expressing cells was very low, and almost half the proportion of the cells expressed the PS marker, T. (d) Analysis of the indicated genes in the FACS-sorted T+ve cells from the AA/F2-6D-treated cells. The T-expressing cells expressed early markers of PS, mesoderm, and endoderm. (e) A model that summarizes the major finding of this study. The mESCs were converted to naïve state of pluripotency (represent the pluripotent embryonic cells in the E3.5-E4.5 developmental stages) using the 2i medium (LIF/2i). AA/F2 treatment for 2 to 3 days led these cells to a primed state of pluripotency (may represent the pluripotent epiblast cells in the E5.5-E7.5 developmental stages). This study shows that the continued treatment of AA/F2 for 6 days led to the enrichment of cells expressing the genes of the primitive streak, mesoderm (primarily, posterior mesoderm), and endoderm (may represent the pluripotent epiblast cells and PS/mesendoderm cells in the E6.5-E7.5 developmental stages).
4. Discussion
During embryo development, cells in the epiblast undergo several systematic changes in pluripotency and part of it can be mimicked in vitro. Mouse ESCs can be brought to a ground/naive state of pluripotency using LIF, along with the inhibitors of ERK and GSK3 [7] (2i medium). The standard hESC culture condition has been adapted to support mEpiSCs (Activin A and FGF2 (AA/F2)), and the same can convert mESCs in the ground/naïve state of pluripotency to a primed EpiSC-like state [1, 8–11]. The current study demonstrates that the exposure of mESCs in the ground state of pluripotency to the AA/F2 transitions them through distinct primed-like EpiSC states, and prolonged exposure of this condition leads them to a PS-like state that had active WNT and activin/nodal signaling and expressed T, Eomes, posterior mesoderm, and endoderm markers (Figure 4(e)). They did not express neuronal markers.
T and Eomes together promote the upregulation of posterior mesoderm and endoderm markers [18, 47] and are induced by FGF, WNT/β-catenin, and activin/nodal/TGFβ signaling, with a negative impact on pluripotency and neural markers [9, 18]. Moreover, the endogenous WNT signaling in EpiSCs under hESC culture condition which causes loss of pluripotency and WNT inhibition could prevent this, reflecting on possible species-specific difference in the culture conditions of mouse and human [46, 48–50]. Although the hESC/EpiSC condition (AA/F2) matured the mESCs to primed states, we show that these states could not be sustained, but the cells further matured to a PS/mesendoderm-like state that had higher WNT signaling active, compared to the intermediate EpiSC-like states (AA/F2-2D and AA/F2-3D). This highlights the requirement of additional factors such as WNT inhibitors, for the preservation of primed pluripotent states of the mouse EpiSCs, in contrast to the requirement of human ESCs [46, 48–50].
Although the AA/F2-treated cells showed primed-like features by both 2 and 3 days of induction, they were different from each other and also from the EpiSCs derived from early postimplantation stage (E5.5) [9]. The 3-day AA/F2-matured EpiSC-like cells, in addition to the pluripotency genes (Oct4, Sox2, and Nanog), also expressed T, Eomes, and Gsc. These characteristics resemble the features of the EpiSCs derived from late postimplantation embryos that express T [22]. The EpiSCs can be derived from mouse postimplantation embryos at various embryonic stages (E5.5–E7.5) that in spite of being similar in several primed-state characteristics [21], also differ among each other [22]. The emerging theory from recent studies is that among pluripotent or progenitor cells in the epiblast, there could be a wide range of molecular states having specific underlying molecular signatures. This heterogeneous population of epiblast cells having diverse differentiation capacities could depend on slightly different extrinsic culture conditions for maintenance that confer their pluripotent or multipotent properties [51], and a majority of this information still remains undefined. For instance, the different durations of AA/F2 led ESCs to distinct pluripotent states that yielded different proportions of presomitic mesoderm cells [24]. At present, the naïve and primed states of pluripotency are the only well-defined states in human and mouse that are interconvertible and can be maintained under defined in vitro conditions. The in vitro conditions used to maintain ESCs could influence the gene-expression state and cell populations within the culture largely [52, 53]. Different populations of EpiSCs, distinct from ESCs, that are interconvertible in vitro, such as Brachyury or Oct4 positive or negative, can coexist, whereby the Oct4+ve cells still can retain the chimera-forming ability [54, 55], which shows that they are totipotent. Although there have been attempts to capture totipotency in vitro [56, 57], the exact conditions that maintain such a state are still unknown [58]. Under the currently known extrinsic culture conditions, possibly, the EpiSCs could occur in heterogeneous metastable states, possessing variable differentiation potentials [24, 25], even to extraembryonic lineages, such as trophoblast [1, 8, 59–62]; [63–66]. The different states could be preserved or interconvertible with ease, by mere manipulation of signaling pathways. The discovery of the 3i: (PD184353, PD173074/SU5402, and CHIR99021, respectively) and later 2i (CHIR99021 to inhibit GSK3β and PD0325901 to inhibit MEK1/2) media by Austin Smith and colleagues (Ying et al. 2008) increased the ESC derivation efficiency in nonpermissive mouse and rat strains, such as CBA, NOD, and DBA from low or nonexistent to 50–70% [67–73]. Therefore, it is very crucial to explore the possibilities of maintaining specific states of totipotency or pluripotency in culture, not only to further increase the efficiency of ESC derivation from any strain and chimera-forming ability, but also to facilitate studies on placental and early embryonic development. More investigation into the specific factors required for maintaining a particular totipotency or pluripotency state could enhance our capability to differentiate them into any desired cell type more efficiently, in high proportion, and our understanding of mammalian embryo development. This and further research will be vital for understanding the molecular mechanism of early patterning in embryogenesis and besides, will ensure that knowledge will be gained about totipotency, pluripotency, and differentiation into progenitor cells for scientific purpose as well as regenerative medicine and thereby associated stem cell-based therapies.
Acknowledgments
We thank Prof. Bernhard. G. Herrmann for his critical comments and support. We are grateful to Dr. Boris Greber for his critical comments and the generous gift of the EpiSCs. SM.S. acknowledges the financial support from the Max Planck Society, Germany, Department of Science and Technology (DST), India (ECR/2017/001216), Department of Biotechnology (DBT), India (BT/PR29717/PFN/20/1361/2018), Indian Council of Medical Research (ICMR), India (2019-3008/SCR/ADHOC-BMS), and Kerala State Council for Science, Technology and Environment (KSCSTE), India.
Data Availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
D.A.S., A.M., and SM.S. contributed to the investigation and validation. D.A.S., A.M., S.S., R.B., and SM.S. participated in the formal analysis. SM.S. and F.K. (generated the reporter cell line) contributed to the resources. D.A.S. and SM.S. did the writing—original draft. A.M., F.K., R.B., and SM.S. did the writing—review and editing. SM.S. contributed to the conceptualization, methodology, supervision, project administration, and funding acquisition. Devika AS and Anna Montebaur contributed equally to this work.
Supplementary Materials
Figure S1: human ESC/EpiSC culture condition leads naïve ESCs to distinct EpiSC-like states. (a) Hierarchical clustering of the indicated samples. The hierarchical clustering indicates that the AA/F2-treated cells were distinct from both EpiSCs (from early postimplantation E5.5 mouse embryos) and ESCs. Figure S2: epithelial-mesenchymal transition occurs during maturation to the PS-like state. (a) Western blot analysis (quantification of the Western blot image in Figure 3(a)) of the indicated proteins in the indicated samples. The graph indicates the relative expression of the indicated proteins in AA/F2-6D, with respect to their expression in AA/F2-3D. Activin/nodal (pSMAD2) and WNT (active β-catenin) signaling were highly active by 6 days of AA/F2 treatment, compared to 3 days. (b–d). Western blot analysis of the indicated proteins in the cells treated with AA/F2 for the indicated durations. Actb served as control. The expression of total AKT and ERK increased by 6 days. There was a slight decrease in the expression of CDH1. Actb and H3 (Figure 3(b)) served as controls. Figure S3: Continuous exposure to the hESC/EpiSC condition draws the PSCs towards of posterior mesoderm. Analysis of the indicated genes in the cells treated with AA/F2 for the indicated durations. The posterior mesoderm marker Msgn1 was induced by 6 days of treatment, and its expressions were negligible at earlier states. Table S1: AA/F2 treatment for three days results in the downregulation of pluripotency genes and the expression of primitive streak genes. FPKM (log2) values of pluripotency and lineage-specific genes (Figures 1(b) and 2(a)). Table S2: Pearson's correlation coefficients between the samples (Figure 1(d)). Table S3: list of primers and their sequences (5′-3′) used for qRT-PCR. Table S4: list of primary and secondary antibodies used for Western blots and immunocytochemistry.
References
- 1.Brons I., Smithers L. E., Trotter M. W. B., et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature. 2007;448(7150):191–195. doi: 10.1038/nature05950. [DOI] [PubMed] [Google Scholar]
- 2.Burdon T., Stracey C., Chambers I., Nichols J., Smith A. Suppression of SHP-2 and ERK signalling promotes self-renewal of mouse embryonic stem cells. Developmental Biology. 1999;210(1):30–43. doi: 10.1006/dbio.1999.9265. [DOI] [PubMed] [Google Scholar]
- 3.Evans M. J., Kaufman M. H. Establishment in culture of pluripotential cells from mouse embryos. Nature Publishing Group; 1981. [DOI] [PubMed] [Google Scholar]
- 4.Smith A. G., Heath J. K., Donaldson D. D., et al. Inhibition of pluripotential embryonic stem cell differentiation by purified polypeptides. Nature. 1988;336(6200):688–690. doi: 10.1038/336688a0. [DOI] [PubMed] [Google Scholar]
- 5.Williams R. L., Hilton D. J., Pease S., et al. Myeloid leukaemia inhibitory factor maintains the developmental potential of embryonic stem cells. Nature. 1988;336(6200):684–687. doi: 10.1038/336684a0. [DOI] [PubMed] [Google Scholar]
- 6.Wray J., Kalkan T., Gomez-lopez S., et al. Inhibition of glycogen synthase kinase-3 alleviates Tcf3 repression of the pluripotency network and increases embryonic stem cell resistance to differentiation. Nature Cell Biology. 2011;13(7):838–845. doi: 10.1038/ncb2267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ying Q.-L., Wray J., Nichols J., et al. The ground state of embryonic stem cell self-renewal. Nature. 2008;453(7194):519–523. doi: 10.1038/nature06968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Tesar P. J., Chenoweth J. G., Brook F. A., et al. New cell lines from mouse epiblast share defining features with human embryonic stem cells. Nature. 2007;448(7150):196–199. doi: 10.1038/nature05972. [DOI] [PubMed] [Google Scholar]
- 9.Greber B., Wu G., Bernemann C., et al. Conserved and divergent roles of FGF signaling in mouse epiblast stem cells and human embryonic stem cells. Cell Stem Cell. 2010;6(3):215–226. doi: 10.1016/j.stem.2010.01.003. [DOI] [PubMed] [Google Scholar]
- 10.Guo G., Yang J., Nichols J., et al. Klf4 reverts developmentally programmed restriction of ground state pluripotency. Development. 2009;136(7):1063–1069. doi: 10.1242/dev.030957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kunath T., Saba-el-leil M. K., Almousailleakh M., Wray J., Meloche S., Smith A. FGF stimulation of the Erk1 / 2 signalling cascade triggers transition of pluripotent embryonic stem cells from self-renewal to lineage commitment. Development. 2007;134(16):2895–2902. doi: 10.1242/dev.02880. [DOI] [PubMed] [Google Scholar]
- 12.Tam P. P. L., Loebel D. A. F. Gene function in mouse embryogenesis: get set for gastrulation. Nature. 2007;8:368–381. doi: 10.1038/nrg2084. [DOI] [PubMed] [Google Scholar]
- 13.Ciruna B., Rossant J. FGF signaling regulates mesoderm cell fate specification and morphogenetic movement at the primitive streak. Developmental Cell. 2001;1(1):37–49. doi: 10.1016/S1534-5807(01)00017-X. [DOI] [PubMed] [Google Scholar]
- 14.Perea-Gomez A., Vella F. D. J., Shawlot W., et al. Nodal Antagonists in the Anterior Visceral Endoderm Prevent the Formation of Multiple Primitive Streaks. Developmental Cell. 2002;3(5):745–756. doi: 10.1016/S1534-5807(02)00321-0. [DOI] [PubMed] [Google Scholar]
- 15.Skromne I., Stern C. D. Interactions between Wnt and Vg1 signalling pathways initiate primitive streak formation in the chick embryo. Development. 2001;128(15):2915–2927. doi: 10.1242/dev.128.15.2915. [DOI] [PubMed] [Google Scholar]
- 16.Arnold S. J., Hofmann U. K., Bikoff E. K., Robertson E. J. Pivotal roles for eomesodermin during axis formation, epithelium-to-mesenchyme transition and endoderm specification in the mouse. Development. 2008;135(3):501–511. doi: 10.1242/dev.014357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Herrmann B. G. Expression pattern of the Brachyury gene in whole-mount T(Wis)/T(Wis) mutant embryos. Development. 1991;113(3):913–917. doi: 10.1242/dev.113.3.913. [DOI] [PubMed] [Google Scholar]
- 18.Tosic J., Kim G.-j., Pavlovic M., Schröder C. M. Eomes and Brachyury control pluripotency exit and germ-layer segregation by changing the chromatin state. Nature Cell Biology. 2019;21(12):1518–1531. doi: 10.1038/s41556-019-0423-1. [DOI] [PubMed] [Google Scholar]
- 19.Bao S., Tang W. W. C., Wu B., et al. Derivation of hypermethylated pluripotent embryonic stem cells with high potency. Cell Research. 2018;28:22–34. doi: 10.1038/cr.2017.134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Smith A. Formative pluripotency : the executive phase in a developmental continuum. Development. 2017;144(3):365–373. doi: 10.1242/dev.142679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kojima Y., Kaufman-Francis K., Studdert J. B., et al. The transcriptional and functional properties of mouse epiblast stem cells resemble the anterior primitive streak. Cell Stem Cell. 2014;14(1):107–120. doi: 10.1016/j.stem.2013.09.014. [DOI] [PubMed] [Google Scholar]
- 22.Bernemann C., Greber B., Ko K., et al. Distinct developmental ground states of epiblast stem cell lines determine different pluripotency features. Stem Cells. 2011;29(10):1496–1503. doi: 10.1002/stem.709. [DOI] [PubMed] [Google Scholar]
- 23.Wu J., Belmonte J., Izpisua C. Dynamic pluripotent stem cell states and their applications. Cell Stem Cell. 2015;17(5):509–525. doi: 10.1016/j.stem.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 24.Sudheer S., Jinhua L., Frederic K. M. M., et al. Different concentrations of FGF Ligands, FGF2 or FGF8 determine distinct states of WNT-induced Presomitic Mesoderm. Stem Cells. 2016;34(7):1790–1800. doi: 10.1002/stem.2371. [DOI] [PubMed] [Google Scholar]
- 25.Tsakiridis A., Huang Y., Blin G., et al. Distinct Wnt-driven primitive streak-like populations reflect in vivo lineage precursors. Development (Cambridge) 2014;141(6):1209–1221. doi: 10.1242/dev.101014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Koch F., Scholze M., Wittler L., et al. Antagonistic Activities of Sox2 and Brachyury Control the Fate Choice of Neuro-Mesodermal Progenitors. Developmental Cell. 2017;42(5):514–526.e7. doi: 10.1016/j.devcel.2017.07.021. [DOI] [PubMed] [Google Scholar]
- 27.Muyrers J. P. P., Zhang Y., Testa G., Francis Stewart A. Rapid modification of bacterial artificial chromosomes by ET-recombination. Nucleic Acids Research. 1999;27(6):1555–1557. doi: 10.1093/nar/27.6.1555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trapnell C., Pachter L., Salzberg S. L. TopHat: discovering splice junctions with RNA-Seq. Bioinformatics. 2009;25(9):1105–1111. doi: 10.1093/bioinformatics/btp120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Trapnell C., Williams B. A., Pertea G., et al. Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nature Biotechnology. 2010;28(5):511–515. doi: 10.1038/nbt.1621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Marks H., Kalkan T., Menafra R., et al. The transcriptional and epigenomic foundations of ground state pluripotency. Cell. 2012;149(3):590–604. doi: 10.1016/j.cell.2012.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nichols J., Zevnik B., Anastassiadis K., et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell. 1998;95(3):379–391. doi: 10.1016/S0092-8674(00)81769-9. [DOI] [PubMed] [Google Scholar]
- 32.Carter A. C., Davis-dusenbery B. N., Koszka K., Ichida J. K., Eggan K. Nanog -Independent Reprogramming to iPSCs with Canonical Factors. Stem Cell Reports. 2014;2(2):119–126. doi: 10.1016/j.stemcr.2013.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schwarz B. A., Ori B.-n., Silva C. R., Hochedlinger K. Nanog Is Dispensable for the Generation of Induced Pluripotent Stem Cells. Current Biology. 2014;24(3):347–350. doi: 10.1016/j.cub.2013.12.050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Joo J. Y., Choi H. W., Kim M. J., et al. Establishment of a primed pluripotent epiblast stem cell in FGF4-based conditions. Scientific Reports. 2014;4(1):p. 7477. doi: 10.1038/srep07477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mulas C., Chia G., Jones K. A., Hodgson A. C., Stirparo G. G., Nichols J. Oct4 regulates the embryonic axis and coordinates exit from pluripotency and germ layer specification in the mouse embryo. Development. 2018;145(12, article dev159103) doi: 10.1242/dev.159103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Chazaud C., Yamanaka Y., Pawson T., Rossant J. Early lineage segregation between epiblast and primitive endoderm in mouse blastocysts through the Grb2-MAPK pathway. Developmental Cell. 2006;10(5):615–624. doi: 10.1016/j.devcel.2006.02.020. [DOI] [PubMed] [Google Scholar]
- 37.Herrmann B. G., Labeit S., Poustka A., King T. R., Lehrach H. Cloning of the _T_ gene required in mesoderm formation in the mouse. Nature. 1990;343(6259):617–622. doi: 10.1038/343617a0. [DOI] [PubMed] [Google Scholar]
- 38.Turner D. A., Rué P., Mackenzie J. P., Davies E., Arias A. M. Brachyury cooperates with Wnt/β-catenin signalling to elicit primitive-streak-like behaviour in differentiating mouse embryonic stem cells. BMC Biology. 2014;12(1):p. 63. doi: 10.1186/s12915-014-0063-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Arnold S. J., Stappert J., Bauer A., Kispert A., Herrmann B. G., Kemler R. Brachyury is a target gene of the Wnt/ β-catenin signaling pathway. Mechanisms of Development. 2000;91(1–2):249–258. doi: 10.1016/S0925-4773(99)00309-3. [DOI] [PubMed] [Google Scholar]
- 40.Funa N. S., Schachter K. A., Lerdrup M., et al. Β-Catenin regulates primitive streak induction through collaborative interactions with SMAD2/SMAD3 and OCT4. Cell Stem Cell. 2015;16(6):639–652. doi: 10.1016/j.stem.2015.03.008. [DOI] [PubMed] [Google Scholar]
- 41.Xu J., Lamouille S., Derynck R. TGF-Β-induced epithelial to mesenchymal transition. Cell Research. 2009;19(2):156–172. doi: 10.1038/cr.2009.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Murry C. E., Keller G. Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell. 2008;132(4):661–680. doi: 10.1016/j.cell.2008.02.008. [DOI] [PubMed] [Google Scholar]
- 43.Wang L. Signaling control of differentiation of embryonic stem cells toward mesendoderm. Journal of Molecular Biology. 2016;428(7):1409–1422. doi: 10.1016/J.JMB.2015.06.013. [DOI] [PubMed] [Google Scholar]
- 44.D’Amour K. A., Agulnick A. D., Eliazer S., Kelly O. G., Kroon E., Baetge E. E. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nature Biotechnology. 2005;23(12):1534–1541. doi: 10.1038/nbt1163. [DOI] [PubMed] [Google Scholar]
- 45.Yamaguchi T. P., Takada S., Yoshikawa Y., Wu N., McMahon A. P. T (Brachyury) is a direct target of Wnt3a during paraxial mesoderm specification. Genes and Development. 1999;13(24):3185–3190. doi: 10.1101/gad.13.24.3185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kurek D., Neagu A., Tastemel M., et al. Endogenous WNT signals mediate BMP-induced and spontaneous differentiation of epiblast stem cells and human embryonic stem cells. Stem Cell Reports. 2015;4(1):114–128. doi: 10.1016/j.stemcr.2014.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Wittler L., Shin E. H., Grote P., et al. Expression of Msgn1 in the presomitic mesoderm is controlled by synergism of WNT signalling and Tbx6. EMBO Reports. 2007;8(8):784–789. doi: 10.1038/sj.embor.7401030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Kim H., Wu J., Ye S., et al. Modulation of β-catenin function maintains mouse epiblast stem cell and human embryonic stem cell self-renewal. Nature Communications. 2013;4(1):p. 2403. doi: 10.1038/ncomms3403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sugimoto M., Kondo M., Koga Y., et al. A simple and robust method for establishing homogeneous mouse epiblast stem cell lines by Wnt inhibition. Stem Cell Reports. 2015;4(4):744–757. doi: 10.1016/j.stemcr.2015.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Sumi T., Oki S., Kitajima K., Meno C. Epiblast ground state is controlled by canonical Wnt/β-catenin signaling in the postimplantation mouse embryo and epiblast stem cells. PLoS One. 2013;8(5, article e63378) doi: 10.1371/journal.pone.0063378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Illich D. J., Zhang M., Ursu A., et al. Distinct signaling requirements for the establishment of ESC pluripotency in late-stage EpiSCs. Cell Reports. 2016;15(4):787–800. doi: 10.1016/j.celrep.2016.03.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Czechanski A., Byers C., Greenstein I., et al. Derivation and characterization of mouse embryonic stem cells from permissive and nonpermissive strains. Nature Protocols. 2014;9(3):559–574. doi: 10.1038/nprot.2014.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Mulas C., Kalkan T., von Meyenn F., Leitch H. G., Nichols J., Smith A. Correction: defined conditions for propagation and manipulation of mouse embryonic stem cells (Doi:10.1242/Dev.173146) Development (Cambridge, England) 2019;146(7):p. dev178970. doi: 10.1242/dev.178970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Han D. W., Tapia N., Joo J. Y., et al. Epiblast stem cell subpopulations represent mouse embryos of distinct pregastrulation stages. Cell. 2010;143(4):617–627. doi: 10.1016/j.cell.2010.10.015. [DOI] [PubMed] [Google Scholar]
- 55.Song L., Chen J., Peng G., Tang K., Jing N. Dynamic Heterogeneity of Brachyury in Mouse Epiblast Stem Cells Mediates Distinct Response to Extrinsic Bone Morphogenetic Protein (BMP) Signaling. Journal of Biological Chemistry. 2016;291(29):15212–15225. doi: 10.1074/jbc.M115.705418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Yang J., Ryan D. J., Wang W., Tsang J. C.-h., Lan G. Establishment of mouse expanded potential stem cells. Nature. 2017;550(7676):393–397. doi: 10.1038/nature24052.Establishment. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Yang Y., Liu B., Xu J., et al. Derivation of pluripotent stem cells with in vivo embryonic and extraembryonic potency. Cell. 2017;169(2):243–257.e25. doi: 10.1016/j.cell.2017.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Geng T., Zhang D., Jiang W. Epigenetic regulation of transition among different pluripotent states: concise review. Stem Cells. 2019;37(11):1372–1380. doi: 10.1002/stem.3064. [DOI] [PubMed] [Google Scholar]
- 59.Beddington R. S., Robertson E. J. An assessment of the developmental potential of embryonic stem cells in the midgestation mouse embryo. Development (Cambridge, England) 1989;105(4):733–737. doi: 10.1242/dev.105.4.733. https://www.ncbi.nlm.nih.gov/pubmed/2598811. [DOI] [PubMed] [Google Scholar]
- 60.Morgani S. M., Canham M. A., Nichols J., et al. Totipotent Embryonic Stem Cells Arise in Ground-State Culture Conditions. Cell Reports. 2013;3(6):1945–1957. doi: 10.1016/j.celrep.2013.04.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Niakan K. K., Ji H., Maehr R., et al. Sox17 promotes differentiation in mouse embryonic stem cells by directly regulating extraembryonic gene expression and indirectly antagonizing self-renewal. Genes and Development. 2010;24(3):312–326. doi: 10.1101/gad.1833510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Sudheer S., Bhushan R., Fauler B., Lehrach H., Adjaye J. FGF inhibition directs BMP4-mediated differentiation of human embryonic stem cells to syncytiotrophoblast. Stem Cells and Development. 2012;21(16):2987–3000. doi: 10.1089/scd.2012.0099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Pera M. F., Andrade J., Houssami S., et al. Regulation of human embryonic stem cell differentiation by BMP-2 and its antagonist noggin. Journal of Cell Science. 2004;117(7):1269–1280. doi: 10.1242/jcs.00970. [DOI] [PubMed] [Google Scholar]
- 64.Xu R. H., Chen X., Li D. S., et al. BMP4 initiates human embryonic stem cell differentiation to trophoblast. Nature Biotechnology. 2002;20(12):1261–1264. doi: 10.1038/nbt761. [DOI] [PubMed] [Google Scholar]
- 65.Das P., Ezashi T., Schulz L. C., Westfall S. D., Livingston K. A., Roberts R. M. Effects of Fgf2 and oxygen in the Bmp4-driven differentiation of trophoblast from human embryonic stem cells. Stem Cell Research. 2007;1(1):61–74. doi: 10.1016/j.scr.2007.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kojima J., Fukuda A., Taira H., et al. Efficient production of trophoblast lineage cells from human induced pluripotent stem cells. Laboratory Investigation. 2017;97(10):1188–1200. doi: 10.1038/labinvest.2016.159. [DOI] [PubMed] [Google Scholar]
- 67.Buehr M., Meek S., Blair K., et al. Capture of authentic embryonic stem cells from rat blastocysts. Cell. 2008;135(7):1287–1298. doi: 10.1016/j.cell.2008.12.007. [DOI] [PubMed] [Google Scholar]
- 68.Hanna J., Markoulaki S., Mitalipova M., et al. Metastable Pluripotent States in NOD-Mouse-Derived ESCs. Cell Stem Cell. 2009;4(6):513–524. doi: 10.1016/j.stem.2009.04.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Kawase E., Suemori H., Takahashi N., Okazaki K., Hashimoto K., Nakatsuji N. Strain difference in establishment of mouse embryonic stem (ES) cell lines. The International Journal of Developmental Biology. 1994;38(2):385–390. [PubMed] [Google Scholar]
- 70.Kiyonari H., Kaneko M., Abe S. I., Aizawa S. Three inhibitors of FGF receptor, ERK, and GSK3 establishes germline-competent embryonic stem cells of C57BL/6N mouse strain with high efficiency and stability. Genesis. 2010;48(5):NA–27. doi: 10.1002/dvg.20614. [DOI] [PubMed] [Google Scholar]
- 71.Li P., Tong C., Mehrian-Shai R., et al. Germline competent embryonic stem cells derived from rat blastocysts. Cell. 2008;135(7):1299–1310. doi: 10.1016/j.cell.2008.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Nichols J., Jones K., Phillips J. M., et al. Validated germline-competent embryonic stem cell lines from nonobese diabetic mice. Nature Medicine. 2009;15(7):814–818. doi: 10.1038/nm.1996. [DOI] [PubMed] [Google Scholar]
- 73.Silva J., Barrandon O., Nichols J., Kawaguchi J., Theunissen T. W., Smith A. Promotion of reprogramming to ground state pluripotency by signal inhibition. PLoS Biology. 2008;6(10):p. e253. doi: 10.1371/journal.pbio.0060253. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1: human ESC/EpiSC culture condition leads naïve ESCs to distinct EpiSC-like states. (a) Hierarchical clustering of the indicated samples. The hierarchical clustering indicates that the AA/F2-treated cells were distinct from both EpiSCs (from early postimplantation E5.5 mouse embryos) and ESCs. Figure S2: epithelial-mesenchymal transition occurs during maturation to the PS-like state. (a) Western blot analysis (quantification of the Western blot image in Figure 3(a)) of the indicated proteins in the indicated samples. The graph indicates the relative expression of the indicated proteins in AA/F2-6D, with respect to their expression in AA/F2-3D. Activin/nodal (pSMAD2) and WNT (active β-catenin) signaling were highly active by 6 days of AA/F2 treatment, compared to 3 days. (b–d). Western blot analysis of the indicated proteins in the cells treated with AA/F2 for the indicated durations. Actb served as control. The expression of total AKT and ERK increased by 6 days. There was a slight decrease in the expression of CDH1. Actb and H3 (Figure 3(b)) served as controls. Figure S3: Continuous exposure to the hESC/EpiSC condition draws the PSCs towards of posterior mesoderm. Analysis of the indicated genes in the cells treated with AA/F2 for the indicated durations. The posterior mesoderm marker Msgn1 was induced by 6 days of treatment, and its expressions were negligible at earlier states. Table S1: AA/F2 treatment for three days results in the downregulation of pluripotency genes and the expression of primitive streak genes. FPKM (log2) values of pluripotency and lineage-specific genes (Figures 1(b) and 2(a)). Table S2: Pearson's correlation coefficients between the samples (Figure 1(d)). Table S3: list of primers and their sequences (5′-3′) used for qRT-PCR. Table S4: list of primary and secondary antibodies used for Western blots and immunocytochemistry.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author, upon reasonable request.


