Abstract
Leakage from blood vessels into tissues is governed by mechanisms that control endothelial barrier function and vascular leakage to maintain homeostasis. Dysregulated endothelial permeability contributes to many conditions and can influence disease morbidity and treatment. Diverse approaches used to study endothelial permeability have yielded a wealth of valuable insights. Yet, ongoing questions, technical challenges, and unresolved controversies relating to the mechanisms and relative contributions of barrier regulation, transendothelial sieving, and transport of fluid, solutes, and particulates complicate interpretations in the context of vascular physiology and pathophysiology. Here, we describe recent in vivo findings and other advances in understanding endothelial barrier function with the goal of identifying and reconciling controversies over cellular and molecular processes that regulate the vascular barrier in health and disease.
Keywords: endothelial barrier function, endothelial cell junctions, gap formation, postcapillary venules, VEGF receptors, G-protein-coupled receptors, Rho GTPases
Introduction to Vascular Permeability
Normal organ function is dependent on the regulation of the permeability of the supplying blood vessels. Vascular permeability (see Glossary) differs among organs, adapts to physiological needs, and reflects the underlying biology of each organ. Vascular permeability also contributes to the pathophysiology of many diseases. Increased permeability is a prominent feature of asthma and other inflammatory airway diseases, arthritis, chronic bowel disease, cancer, infections, trauma, ischemic stroke, and many other conditions where leakage can result in edema, impaired function, and morbidity.
This review focuses on molecular mechanisms that regulate endothelial junctions in the control of vascular permeability. Endothelial barrier function and vascular permeability are governed by intercellular junctions that create the barrier that regulate the extravasation of plasma including its macromolecular constituents. Our emphasis reflects advances in the understanding of the molecular organization and regulation of endothelial junctions in vascular permeability in health and disease, with the understanding that other pathways can contribute to the movement of substances across endothelial cells. Diaphragm-covered endothelial fenestrations, which are abundant in endocrine glands, intestinal mucosa, and certain other organs, enable transendothelial transit of fluid and small solutes [1]. Transendothelial vesicular transport provides another potential route across endothelial cells in capillaries, but the contribution to vascular permeability is still unsettled [1, 2].
Adjacent endothelial cells are connected by two types of intercellular junctions: adherens junctions and tight junctions. The number and organization of these junctions underlie permeability differences in the vasculature to accommodate organ- and tissue-specific needs. Mediators that increase permeability activate kinases, phosphatases, and other enzymatic activities that control phosphorylation of junctional proteins and focal gap formation between endothelial cells. Here, we discuss open questions, challenges, and controversies over the regulation of endothelial barrier function and the mechanism of action of agents that promote, prevent, or reverse increased endothelial permeability.
The endothelial barrier under conditions of homeostasis and in disease
Normal function and diversity of the endothelial barrier
Transendothelial fluid sieving is controlled by the vascular barrier and hydrostatic and oncotic forces that drive movement across the endothelium, as described by the Starling equation [3]. Extravasation increases, both qualitatively and quantitatively, when vascular permeability is increased in the presence of hydrostatic forces to drive flux from blood into the tissue interstitium. Vascular permeability, especially to large molecules that normally have limited extravasation, can increase with exposure to inflammatory cytokines and other factors. Key features of increased permeability are the opening of intercellular junctions and formation of gaps between endothelial cells [4]. Increased permeability and extravasation of plasma fluid and proteins are transient in healthy organs and diminish when the stimulus ends but can be sustained in chronic inflammation and cancer (Figure 1).
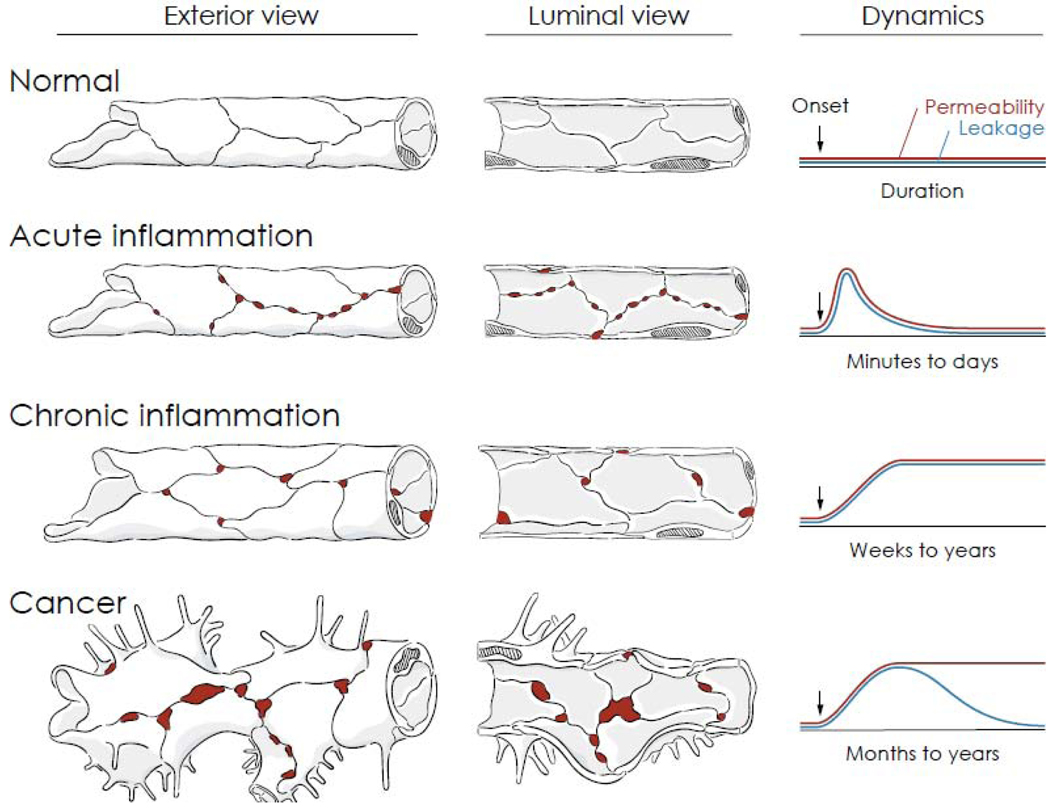
Figure 1. Vascular permeability and leakage in health and disease.
Comparison of exterior and luminal views of blood vessels of the microcirculation and corresponding dynamics of permeability and leakage over time under normal conditions, inflammation, and cancer. Durations are approximate values reflecting the vasculature of the entire organ rather than individual vessels or gaps. In normal blood vessels, junctions between endothelial cells form a uniform barrier that restricts extravasation of plasma. Gaps are not present between endothelial cells, and permeability and leakage are stable and low. In acute inflammation, leakage occurs as permeability increases rapidly through formation of focal endothelial gaps (red) in postcapillary venules. The gaps are transient, and vessels return to normal after the inflammatory stimulus ends or is inactivated. In chronic inflammation, vessels undergo structural remodeling, reflected by enlargement, proliferation (angiogenesis), increased mediator sensitivity, and sustained gap formation and leakage. The vascular changes do not resolve spontaneously but can be reversed by treatment of the chronic condition. In cancer, blood vessels that supply growing tumors undergo distinctive changes promoted by the abnormal tumor microenvironment, including sprouting angiogenesis, abnormal growth and gene expression, and defective endothelial junctions that result in leakage of plasma and even erythrocytes in some tumors. Increased endothelial permeability is sustained, but leakage decreases as interstitial pressure rises because of impaired lymphatic drainage.
Endothelial barrier function varies in different segments of the microvasculature, where permeability increases from arterioles (least) to venules (greatest). Endothelial cells in arteries and arterioles, which regulate blood flow to tissues through dilatation or constriction regulated by smooth muscle cells, provide a relatively tight barrier. Capillaries supply the expansive surface area for efficient exchange of oxygen, carbon dioxide, and metabolites between blood and tissues. Three types of capillaries have organ-specific differences in function, continuous, fenestrated, and discontinuous [5]. Although fenestrated capillaries have 60-nm openings (fenestrations), permeability is restricted to water and small hydrophilic solutes by a covering diaphragm that permits the passage of molecules as large as 5 nm [1, 6]. Transit through fenestrations of kidney glomerular capillaries and liver sinusoids that lack diaphragms is regulated by blood flow, glycocalyx, basement membrane, glomerular slit diaphragms and other barriers [7, 8]. Venules have endothelial cells that are more leaky to fluid, solutes, and proteins and are highly sensitive to mediators that increase permeability. In most organs, leukocytes extravasate from venules where endothelial cells exposed to inflammatory cytokines express adhesion molecules for leukocyte attachment. In the lung, alveolar capillaries are the main site of leukocyte extravasation. Leukocytes extravasate transcellularly or at endothelial junctions, however, passage through junctions does not require breakdown of the endothelial barrier. In contrast, barrier breakdown is required for leakage of plasma macromolecules. Details of the processes involved in leukocyte extravasation are available in excellent reviews [9, 10]. Unlike venules, large veins are less leaky and less responsive to permeability increasing mediators.
Endothelial barrier function differs greatly among organs. The endothelial barrier is tightest in the central nervous system (CNS), where the blood-brain barrier restricts transvascular flux of solutes and macromolecules across blood vessels, which is attributed to junction tightness and limited transcytosis [11]. Endothelial transporters provide the selective transfer of essential substrates from blood into the brain parenchyma [11]. Pericytes also contribute to the blood-brain barrier, as pericyte-deficient mice have abnormal leakage in the CNS due to increased transcytosis and perhaps other endothelial cell defects [2, 12]. The endothelial barrier is also tight in peripheral nerves, and is relatively tight in skeletal muscle, cardiac muscle, and lung. By building on long recognized organ-specific features of the vasculature, research is beginning to increase the understanding of the cellular and molecular mechanisms underlying permeability differences among organs [13].
Mechanisms of endothelial barrier function in disease
In the quiescent vasculature, endothelial barrier stability is actively maintained (see below: Mechanism of action of permeability modulators). Under conditions where permeability is increased (hyperpermeability), leakage is brief and reversible after a transient stimulus, as in acute inflammation after a mosquito bite, but can be sustained in chronic inflammation where abnormal conditions persist and the microvasculature undergoes remodeling into a more leaky phenotype [14] (Figure 1). Edema results when plasma leakage exceeds fluid clearance through lymphatics and other routes [15].
Endothelial barrier function changes after tissue injury by allergens, pathogens, toxins, trauma, burns, and other stimuli. Cytokine storm in COVID-19 is an example [16]. Mechanisms that regulate endothelial permeability under these conditions have multiple features in common: endothelial junction organization is altered, gaps form, and the transendothelial hydrostatic pressure gradient drives leakage [17].
Vascular remodeling is disease specific. In chronic inflammation, normally tight capillaries can change into leaky venules [14]. In cancer, blood vessels that grow as tumors enlarge lose the typical hierarchical organization of arterioles, capillaries, and venules and acquire an abnormal phenotype unique to tumors [18]. Tumor vessels undergo continuous growth, remodeling, and regression, and characteristically are leaky and have impaired blood flow [18]. Although can have barrier defects large enough for hemorrhage, plasma leakage in tumors is limited by reduced driving force due to poor vascular perfusion and high interstitial pressure resulting from impaired lymphatic drainage [19] (Figure 1).
The consequences of vascular barrier dysfunction in disease are also dependent on the afflicted organ. In the brain, disruption of the blood-brain barrier can result in cerebral edema, increased intracranial pressure, impaired cognitive or motor function, and even death [20]. In the lung, leakage can impair gas exchange and lead to hypoxia, hypercapnia, and fatal outcome.
Elevated lung microvascular pressure can increase capillary transmural pressure and disrupt barrier function [21]. The mechanically activated ion channels Piezo1 and Piezo2 contribute to baroreceptor regulation of blood pressure and flow sensing by endothelial cells [22]. Piezo1 in endothelial cells is associated with lung microvascular leakage and diverse cardiovascular diseases [22]. Lung microvascular leakage and pulmonary edema under conditions of increased microvascular pressure is dependent on Piezo1-dependent endothelial barrier disruption through VE-cadherin endocytosis and degradation [23]. Additional clinical consequences of endothelial barrier function dysregulation are summarized in the Clinician’s Corner.
Composition and plasticity of endothelial junctions
Endothelial junction integrity is regulated by proteins that form adherens junctions and tight junctions, which connect adjacent cells in a dynamic and communicative manner (Figure 2). Gap junctions also contribute to endothelial barrier regulation. Open questions and controversies relating to the composition and plasticity of endothelial junctions are described in Box 1.
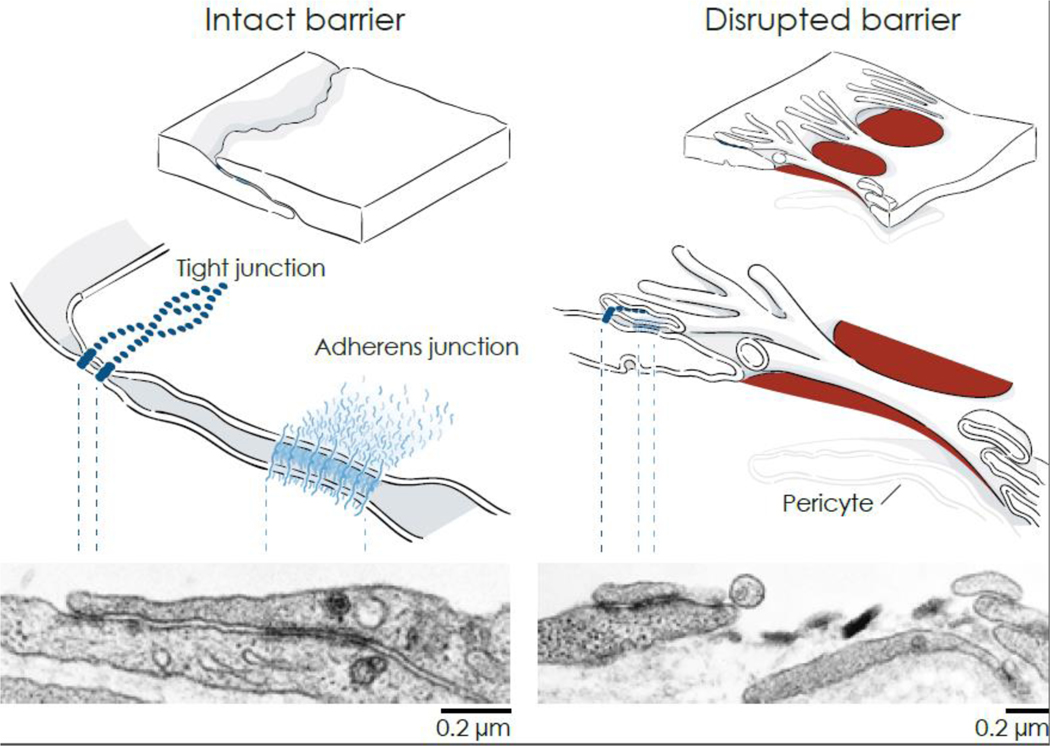
Figure 2. Comparison of endothelial junctions at intact and disrupted barriers.
Left: Drawings of the border of normal overlapping endothelial cells (upper) showing the relative locations of the tight junction and adherens junction (middle). Corresponding transmission electron micrograph (TEM) of a normal venule showing overlapping endothelial cell borders with electron-dense junctions and underlying basement membrane (lower). Locations of the tight junction near the lumen (apical) and adherens junction (basolateral) are marked by dashed lines. Right: Disrupted endothelial barrier with focal gaps (red) after exposure to a mediator that increased vascular permeability (upper). Cell connections are maintained by junctions at the tip of finger-like filopodia that extend from one endothelial cell to the adjacent cell (middle). Corresponding TEM of an endothelial gap (lower). Endothelial cells bordering the gap have filopodia on the luminal surface, some with intact intercellular junctions. Extravasated particles of the tracer Monastral blue (black crystals in the center) are trapped in basement membrane exposed by the gap. Basement membrane underlies the endothelial cells and surrounds a pericyte process on the right. TEM images from rat trachea at baseline (left) and after electrical stimulation of vagus nerve for 5 minutes to activate substance P release from sensory axons (right) [131].
BOX 1. Composition and plasticity of endothelial junctions: open questions and controversies.
The overlapping border of endothelial cells is separated by a narrow cleft bridged by two types of junctions. Tight junctions form the barrier near the luminal (apical) surface. Adherens junctions, typically located more basally, join adjacent endothelial cells. Focal changes in junctions result in endothelial gaps and leakage. Gap formation requires detachment of complexes involving the adherens junction protein VE-cadherin that joins adjacent endothelial cells through homophilic interactions. Endothelial cell tight junctions are composed of multiple transmembrane proteins that interact with membrane lipids and cytoplasmic proteins, as in epithelial cell barriers. Tight junctions differ among blood vessel types and organs, but the details are incompletely understood. Also deserving further study is how tight junctions change in response to stimuli that increase permeability, and how changes in tight junctions and adherent junctions are coordinated.
One of many challenges faced in research on endothelial junctions is the limited availability of tools to visualize junctional changes in real time and at high resolution. Diverse approaches used to examine the properties of endothelial junctions have shown that in vitro models have multiple features that differ from the permeability dynamics in blood vessels in vivo. Among the differences are: i) exposure of cultured endothelial cells to thrombin or other permeability modulator can have effects on the actin cytoskeleton that result in disconnection of junctions and gradual formation (minutes) of large intercellular separations,* unlike the discrete endothelial gaps that form rapidly (seconds) in vivo; ii) static cell cultures usually lack flow and other environmental conditions that influence the response of junctions to stimuli; iii) cultures can be mixtures of endothelial cells derived from large and small blood vessel types, and even lymphatics, that do not reflect regions of the microvasculature that leak in vivo; and iv) inflammatory genes can be upregulated in endothelial cells as cultures are established. Because of these complicating factors, mechanistic insights from in vitro studies of endothelial cells must be verified by in vivo models to test relevance to junctional regulation in the microvasculature.
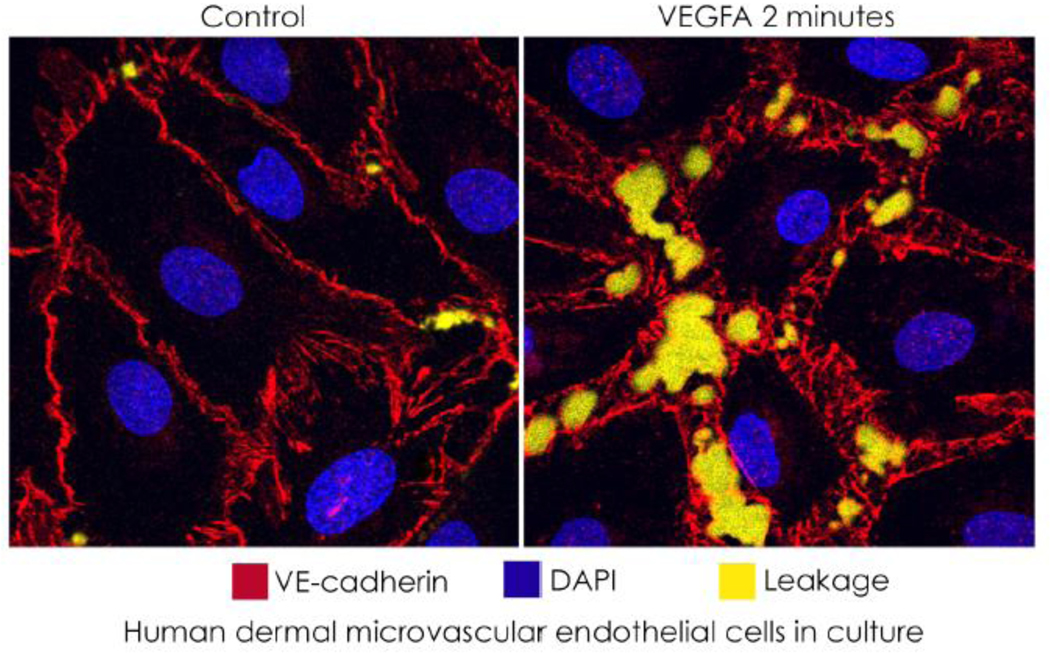
Figure I box 1.
compares control and VEGFA-stimulated microvascular endothelial cells, where leakage is detected by fluorescent streptavidin (yellow) bound to biotin in the substrate, in an in vitro vascular permeability imaging assay (Merck KGaA, Darmstadt). Separations of junctions in cultured endothelial cells exposed to VEGFA are larger and more numerous than occur in vivo.
Adherens junctions
The main component of endothelial junctions, VE-cadherin, forms Ca2+-dependent, homophilic interactions between adjacent endothelial cells that are essential for maintenance of the endothelial barrier (Figure 2). VE-cadherin is composed of extracellular cadherin motifs, a transmembrane domain and an intracellular domain that mediates interactions with β-catenin, p120-catenin, and γ-catenin, also known as plakoglobin. β-catenin and γ-catenin in turn connect to actin-binding α-catenin and other proteins. The intracellular complex of VE-cadherin and catenins is essential for junctional stability. Genetically engineered mice with stabilized VE-cadherin/α-catenin complexes are resistant to leakage [24]. Conditional deletion of the VE-cadherin gene cadherin5 (Cdh5) in mice results in elevated basal transvascular fluid flux in the heart and lung, but not brain or skin [25, 26], which is consistent with effects of inhibitory antibodies to VE-cadherin that cause endothelial gaps and leakage in the heart and lung [26, 27]. Both approaches highlight the organ-specific functional differences in VE-cadherin. VE-cadherin is complexed with VEGF receptor-2 (VEGFR2), Tie2, and other receptors that mediate the actions of permeability modulators. VE-cadherin also forms complexes with VE-protein tyrosine phosphatase (VE-PTP) and density-enhanced phosphatase 1 (DEP1) [28–31], which have a strong impact on junctional stability. Although often described as exclusively expressed in blood vascular endothelial cells, VE-cadherin is also found in lymphatic endothelial cells, certain hematopoietic cells, and in brain fibroblast-like cells identified by single cell RNA-sequencing [32].
In response to vascular permeability modulators and blood flow, VE-cadherin undergoes tyrosine phosphorylation through the action of Src family kinases (SFKs) [33]. Phosphotyrosine residues pY658, pY685, and pY731 of VE-cadherin have been extensively studied in this context, but other phosphotyrosine residues may also be important [34]. Both Y658 and Y685 are phosphorylated in a flow-regulated manner [35, 36]. Evidence that Y685 phosphorylation is a necessary step in the process whereby VEGFA, histamine, and bradykinin increase permeability comes not only from in vitro studies, but also from a mutant mouse with the Y685 residue of VE-cadherin replaced by phenylalanine (F) [35, 37, 38]. VEGFA and histamine induce less leakage in skin and cremaster muscle of Y685F mutant mice [38], and leakage is reduced from pathological vessels that grow in the retina of mice after exposure to hyperoxia [39]. As further evidence of the importance of VE-cadherin phosphorylation in adherens junction disassembly, Y685F mutant mice not only have less plasma leakage but also have less reverse transendothelial neutrophil migration back into the circulation [40]. Still unresolved is the contradictory finding in another Y685F mutant mouse of greater VEGFA-induced leakage in skin accompanied by uterine edema and ovarian cysts [41]. Peak VE-cadherin Y685 phosphorylation in the ovary is reported to coincide with VEGF-induced angiogenesis accompanying corpus luteum formation after ovulation in the mouse estrous cycle [42]. The very different effect of mutating Y731 of VE-cadherin is reflected in a mutant Y731F mouse that has impaired leukocyte extravasation but not altered plasma leakage [38]. Y731 phosphorylation is present in endothelial cells under baseline conditions, downregulated by leukocyte docking, and required for leukocyte extravasation [38]. Different phosphotyrosine sites in VE-cadherin thus appear to serve different purposes, but more needs to be learned about how they exert their functions and how they are regulated in conjunction with ubiquitination, intracellular trafficking, and recycling of VE-cadherin (Figure 3).
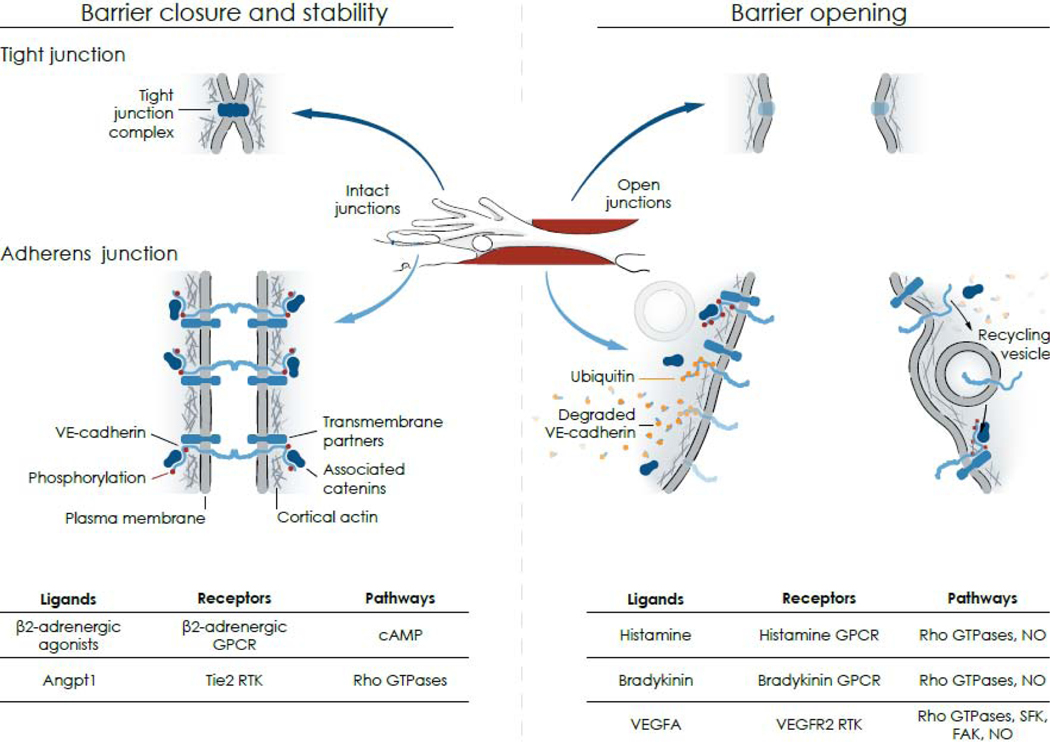
Figure 3. Mechanisms of barrier opening and closure.
Center: Drawing based on Figure 2 showing intact junctions (blue) between finger-like endothelial filopodia and an adjacent endothelial cell (left, Intact junctions) and a gap (red) between the same endothelial cells (right, Open junctions). Left arrows point to corresponding regions of intact tight junctions and adherens junctions; right arrows point to open junctions. Left: Intact endothelial cell barrier formed by homophilic interactions of tight junction complexes (upper) and adherens junction proteins (lower) at endothelial cell borders. At tight junctions, the fused outer leaflets of plasma membranes of adjacent cells create a diffusion barrier. At adherens junctions, VE-cadherin is in complex with catenins and other transmembrane proteins that provide structural attachments. Tyrosine residues of the intracellular domain of VE-cadherin have a low level of flow-dependent phosphorylation (few red dots) in endothelial cells of postcapillary venules at baseline. Right: Open barrier at endothelial gap resulting from changes in tight junctions (upper), which must still be defined, and adherens junctions (lower), where tyrosine phosphorylation of VE-cadherin increases (many red dots) followed by ubiquitination (orange dots), dismantling of complexes and degradation or internalization into vesicles and recycling back to the plasma membrane where complexes and tyrosine phosphorylation are restored. Bottom table: Ligands, receptors, and pathways implicated in barrier closure and stability (left) and barrier opening (right): Angpt1, angiopoietin 1; cAMP, cyclic adenosine monophosphate; FAK, focal adhesion kinase; GPCR, GTPase protein coupled receptor; NO, nitric oxide; RTK, receptor tyrosine kinase; SFK, Src family kinase; VEGFA, vascular endothelial growth factor.
Increases in endothelial permeability resulting from focal gaps between endothelial cells are accompanied by changes in VE-cadherin at junctions. VE-cadherin distribution changes from a continuous band along cell borders to a distinctive serrated or zig-zag pattern associated with finger-like cell processes (filopodia) that form with gaps (Figure 2) [43]. Junctions that are open at gaps remain intact where filopodia span gaps and contact adjacent endothelial cells (Figure 2). In regions where homophilic interactions of junctional proteins detach and gaps form, VE-cadherin undergoes internalization followed by recycling or degradation by ubiquitination [35]. Still unresolved is whether internalization is triggered directly by tyrosine phosphorylation of VE-cadherin or involves additional events such as phosphorylation of p120-catenin associated with VE-cadherin [44]. Also deserving further study are findings that VEGFA induces endocytosis of phosphorylated VE-cadherin, but histamine induces relocation of VE-cadherin in the plasma membrane to vinculin-associated focal adherens junctions that border gaps between cultured endothelial cells [45].
VE-cadherin is also implicated in endothelial cell processes during vascular development, including establishment of cell polarity, vascular anastomosis, lumen formation, regression, and remodeling [46]. It is unclear whether molecular mechanisms that ensure junctional stability during these processes are the same as those involved in regulation of endothelial gaps and leakage in the mature vasculature.
Tight junctions
Endothelial cell tight junctions, composed of members of the claudin and junction-associated molecule (JAM) families, occludin, endothelial cell-selective adhesion molecule (ESAM), and other adhesion molecules, share many features with tight junctions in epithelial cells [47]. Transmembrane tight junction proteins exist in complex with the intracellular scaffold proteins cingulin, paracingulin, and zona occludens (ZO) family members [48] and with membrane lipids [49, 50]. Endothelial barrier tightness is influenced by the composition of tight junction complexes [26, 51] and abundance of tight junction strands [52, 53]. Tight junction strands viewed by freeze-fracture are more abundant in endothelial cells of arterioles than venules [52, 53].
Although changes that tight junctions undergo during endothelial gap formation have been difficult to delineate and much remains to be learned, adherens junctions precede tight junctions in development and are necessary for their formation [50]. Changes in tight junctions are coordinated with changes in adherens junctions [54]. VE-cadherin can influence tight junctions and stabilize junctions by promoting claudin-5 (Cldn5) expression [54]. Reciprocally, tight junctions can influence adherens junction stability through the junctional adaptor protein ZO-1, which regulates functional coupling of VE-cadherin to the cytoskeleton [55]. These interactions of junctional proteins identified in cultured endothelial cells must still be confirmed in vivo.
Cldn5 is particularly important among cadherin family members because it is relatively specific to endothelial cells, but Cldn5 expression is not uniform throughout the vasculature. Mice lacking Cldn5 expression die postnatally from seizures due to disruption of the blood-brain barrier and leakage of small but not large tracers [56]. Endothelial cells of arteries and arterioles in the skin of Cldn5-GFP reporter mice have strong GFP fluorescence, but those of postcapillary venules that become leaky in response to VEGFA have little or no Cldn5-GFP [57]. Evidence that VEGFA induces Cldn5 downregulation and occludin phosphorylation (Ser490) and ubiquitination in cultured endothelial cells, through a protein kinase C-β (PKCβ)-dependent pathway, fits with tight junction changes in VEGFA-induced leakage [58].
ESAM is widely expressed in the vasculature, but its contribution to barrier function depends on whether Cldn5 or other tight junction proteins are also expressed. The organ-specific importance of ESAM in tight junctions is evident in the finding of modest leakage in lungs - but not brain, heart or skin - of mice with constitutive deletion of Esam, and rapid death from lung leakage and thrombosis when Esam deletion is combined with VE-cadherin ablation [26].
Further study is needed to determine whether tight junction complexes undergo focal separation, lateral movement, or dissolution during endothelial gap formation through mechanisms found in development [59] and other conditions of junctional dynamics [50, 60]. Regardless of the mechanisms underlying these changes, junctions remain intact in regions around gaps where finger-like filopodia contact adjacent endothelial cells and sites of endothelial contact where gaps are not present (Figure 2).
Gap junctions
Gap junctions communicate changes in ion currents and signals mediated by small molecules that pass between endothelial cells. Gap junctions are composed of connexins arranged in hexamers forming hemichannels that join their counterparts in neighboring endothelial cells. Connexins, 37, 40 and 43 are preferentially expressed in endothelial cells and have been implicated in modulation of basal permeability [61]. In particular, changes in connexin43 expression correlate inversely with VE-cadherin expression in the lung after vascular leakage is induced by endotoxin [62]. Gap junctions can also contribute to angiogenesis [63], associate with tight junction strands in endothelial cells [64], and play a role in regulating vascular barrier opening under some conditions [65]. Gap junctions between endothelial cells and smooth muscle cells have been implicated in vasomotor activity [61].
Modulators of endothelial barrier function
The wide range of modulators of endothelial barrier function that have been identified can promote barrier opening, restore barrier closure, or maintain barrier integrity. Some of these modulators act directly on barrier components, as discussed below, while others act indirectly. Open questions and controversies relating to modulators of endothelial barrier function are addressed in Box 2.
BOX 2. Modulators of endothelial barrier function: open questions and controversies.
Many modulators of endothelial barrier function have been studied to dissect the cellular and molecular mechanisms underlying changes in vascular permeability, to identify factors that contribute to plasma leakage and edema formation in disease, and to test the efficacy of agents that reduce leakage. VEGFA, angiopoietins (Angpt1, Angpt2), and the inflammatory cytokines histamine and bradykinin are known to have direct actions on endothelial barrier function. Factors that regulate the barrier through direct actions have rapid effects on adherens junctions, tight junctions, or other essential barrier components. In contrast, substances that act indirectly change barrier function by acting on other cells that release mediators, modulating the production of factors, or having other effects on the barrier. Examples of indirect actions are antigen-induced histamine release from mast cells and capsaicin-induced release of substance P from sensory nerves [122]. Moreover, fibroblast growth factors (FGFs) modulate the expression of protein tyrosine phosphatases, thereby potentially regulating the phosphorylation of VE-cadherin [123]. Hepatocyte growth factor (HGF) increases endothelial permeability by promoting VEGFA production [124]. A challenge in interpreting the reported effects of such factors is that few have been compared side by side with approaches that can assess alterations in endothelial barrier function independent of hemodynamic changes.
Also unclear is whether some mediators can induce the formation of smaller or larger gaps between endothelial cells, and thus permit more or less sieving and extravasation. This issue is difficult to resolve because many factors that affect the barrier also have hemodynamic effects that influence the driving force for extravasation. Underlying mechanisms can lead to concurrent changes in driving force, endothelial tight junctions and adherens junctions, or other components of the vascular barrier (glycocalyx, basement membrane, and mural cells). Additional studies are needed to distinguish changes in driving force from modulation of permeability and to determine the size range of molecules that extravasate after each mediator, the duration and rate of recovery of the leakage, whether tachyphylaxis develops, and the types of vessels and organs where leakage occur.
VEGF and VEGF receptor tyrosine kinases
VEGFA, originally named vascular permeability factor, increases plasma extravasation [66]. VEGFA also promotes endothelial cell sprouting and vascular growth and can increase leukocyte adhesion and migration, but the mechanisms are likely to differ from those involved in the regulation of endothelial barrier function [43]. VEGFA has several splice variants that differently bind to basement membrane proteins and to VEGF co-receptors neuropilin-1 and neuropilin-2 (NRP1, NRP2) [67]. Neuropilins are of interest because of their essential contribution to leakage mediated by VEGFA [68].
In addition to VEGFA, the structurally related mammalian VEGF family members include VEGFB, VEGFC, VEGFD, and placenta growth factor (PlGF). The VEGF family acts through three structurally related transmembrane tyrosine kinase receptors, VEGFR1, VEGFR2 and VEGFR3. VEGFR2 is the most abundant receptor of this family on endothelial cells and is essential for VEGFA-induced increase in vascular permeability. Binding of PlGF/VEGFB to VEGFR1 can block VEGFA binding to VEGFR1, making VEGFA available to increase permeability by binding to VEGFR2 [69]. VEGFR3 can have an effect on vascular permeability through upregulation of VEGFR2 expression [70]. VEGFR2 regulation of vascular permeability and leakage depends on signaling via SFKs and Rho family GTPases. A wide range of drugs that block VEGFA or VEGFR2 are used to suppress angiogenesis, leakage, and edema, exemplified by antibodies and recombinant receptor fragments used to treat diabetic macular edema, wet age-related macular degeneration, and other choroidal and retinal vascular diseases [71–73].
Angiopoietins and Tie receptor tyrosine kinases
The angiopoietin family is composed of three members Angpt1, Angpt2 and Angpt3 (mouse) and Angpt4 (human). Angpt1 and Angpt2, which are more closely related to each other than to Angpt3/Angpt4, have an unusual structure with about 500 amino acid residues and predicted coiled-coil and fibrinogen-like domains.
Angpt1 and Angpt2 have complex positive and negative effects on vascular permeability through actions on Tie1 and Tie2 receptor tyrosine kinases. Both Angpt1 and Angpt2 bind to Tie2 (originally named Tek) on endothelial cells of blood vessels and lymphatic vessels, as well as on some myeloid cells and perivascular cells [74]. Angpt1 activation of Tie2 promotes vascular stability and reduces leakage [75, 76]. The increased junctional stability underlying the antileakage action of Angpt1 is mediated in part by Rho family GTPase effects on the endothelial cell cytoskeleton [77].
Tie1 is an orphan receptor that regulates Angpt2 activation of Tie2. Ectodomain cleavage of Tie1 is important in determining Angpt2 effects on endothelial cells [78, 79]. In the absence of Tie1, Angpt2 inhibits Tie2, resulting in vascular destabilization and susceptibility to leakage. In the presence of Tie1, Angpt2 activates Tie2, leading to barrier stabilization. The antagonistic effects of Angpt2 are also influenced by binding to and activation of beta-1 integrins [80].
Angpt1 activation of Tie2 reduces vascular leakage induced by VEGFA, histamine, bradykinin, and other cytokines [75, 76, 81], although the underlying mechanism is complex. Angpt1 also promotes attraction of pericytes that increase vascular support and stability [75, 82].
Angpt1 mimetics BowAng1 and COMP-Ang1 reduce vascular leakage in mouse models of lung infection and cancer [79, 83–85]. Angpt2 is a prognostic biomarker of sepsis and acute respiratory distress syndrome (ARDS) [86–88]. Angpt2 inhibitors, typically in combination of VEGFA inhibitors, are in clinical trials for treatment of cancer and age-related macular degeneration and other retinal vascular diseases [72, 73, 89].
Histamine, bradykinin, thrombin, platelet-activating factor, and G-protein-coupled receptors
Histamine from mast cells binds to H1 and H2 histamine receptors, which are G-protein-coupled receptors (GPCRs) expressed on endothelial cells [90]. Histamine promotes endothelial gap formation and leakage through a direct action on VE-cadherin phosphorylation [91]. Histamine also mediates activation of endothelial nitric oxide synthase (eNOS), resulting in production of NO. NO promotes vascular smooth muscle cell relaxation, vasodilatation, increased blood flow, and greater transvascular fluid flux.
Bradykinin, generated by kallikrein proteolytic cleavage of high-molecular-weight kininogen, acts on GPCRs B1 and B2 [92]. Like histamine, bradykinin acts directly on endothelial cell mechanisms that regulate VE-cadherin in adherens junctions [35] and promotes eNOS activation, NO production, vasodilatation, and increased transendothelial fluid flux.
The serine protease thrombin strongly affects cell-cell adhesion and induces cell retraction and gap formation in cultured endothelial cells, but its action on endothelial cells in vivo is dependent on the presence of other factors and is likely to be indirect [93]. Thrombin is proteolytically cleaved from prothrombin in the clotting cascade. Binding of thrombin to its proteinase-activated receptors (PARs), which are GPCRs, increases intracellular Ca2+ and diacylglycerol, and activates Rho GTPases and cytoskeletal rearrangements in cultured endothelial cells [94].
Platelet-activating factor (PAF), which is produced by inflammatory cells, is a phospholipid that acts through its specific GPCR expressed on endothelial cells and other cell types. As indicated by its name, PAF induces platelet aggregation and degranulation. PAF is also implicated in inflammation and anaphylaxis. The mechanisms underlying the effects of PAF on barrier stability are unclear, including the vessel types affected, identity of the signaling pathways, and hemodynamic effects. PAF may also act by inducing the release of VEGFA or other endothelial modulators [95].
Additional permeability modulators, including sphingosine-1 phosphate, substance P, and macrophage migration inhibitory factor (MIF), that modulate endothelial barrier function are considered in many excellent reviews [96–98].
Mechanism of action of permeability modulators
Multiple intracellular pathways involving Src family kinases, Rho GTPases, actin cytoskeleton, focal adhesion kinase and cell-matrix adhesion, and eNOS/NO signaling are activated by mediators that promote endothelial gap formation and plasma leakage through direct actions on endothelial cells (Figure 3). Open questions and controversies relating to the mechanism of action of permeability modulators are addressed in Box 3. Dynamics and dimensions of endothelial gap formation are addressed in Box 4.
BOX 3. Mechanism of action of permeability modulators: open questions and controversies.
Many factors that increase endothelial permeability activate pathways similar to or overlapping downstream pathways activated by factors that are not implicated in barrier regulation. It is unclear how VEGFA, but not FGF2, has a potent direct effect on vascular permeability, as the two growth factors activate similar signaling pathways in endothelial cells. A potential difference is the subcellular location of activity that results in differential effects on junctional stability. A question still to be answered is whether VEGFA, but not FGF2, induces c-Src activity at endothelial junctions rather than in other subcellular domains.
Several signal transduction pathways influence the stability of VE-cadherin, and thereby adherens junctions, as well as the action of Rho family GTPases on the cytoskeleton [125]. While these downstream pathways are presumed to be activated in all endothelial cells that express the appropriate receptors for leakage-inducing mediators, permeability increases more in postcapillary venules than in other segments of the microvasculature, and is restricted to focal regions where gaps form at endothelial junctions [57, 126]. One underlying factor is the relative density of receptors on endothelial cells of different regions of the microvasculature [127], but other mechanisms yet unidentified are also likely to involved.
Signaling pathways activated by factors that lead to endothelial barrier disruption and increased permeability are balanced by pathways that protect, stabilize, or reestablish intact junctions (Figure 3). Maintenance of stable junctions depends on continuous signaling by Angpt1, S1P, and β-adrenergic receptor agonists that act through Rac1 and other pathways [128, 129]. β-adrenergic receptors and the downstream second messenger cAMP act through exchange proteins activated by cAMP (EPACs) for GTPases Rap1 and Rap2. EPACs can also stabilize the barrier by direct interaction with VE-cadherin [130]. Consistent with this balancing, permeability is increased by diminished availability of these stability factors or greater expression or activity of their antagonists.
Pericytes and the extracellular matrix also contribute to barrier regulation in the microcirculation [82, 131]. Pericytes connect to endothelial cells by N-cadherin, which has been implicated in regulation of Trio, a RhoA/Rac1 GTPase activation protein (GAP) that regulates barrier stability [132]. Focal adhesions that attach endothelial cells to the underlying matrix can impact barrier function by communicating with intercellular junctions [116]. Much remains to learned about the interplay of signal transducers with the endothelial basement membrane, pericytes, and cell-matrix adhesions.
BOX 4. Dynamics and dimensions of endothelial gaps: open questions and controversies.
Factors that increase vascular permeability lead to detachment of focal regions of endothelial adherens junctions and the formation of intercellular gaps through which fluid, solutes, and proteins can extravasate. Endothelial gaps can be readily visualized by electron microscopy [133] and also seen by light microscopy after appropriate staining [134, 135] (Figure 4). Gap dimensions and dynamics can also be assessed microscopically [136, 137] or by examining the extravasation of tracers of different sizes, such as fluorescent dextrans and microspheres or nanoparticles. Studies of in vivo models of inflammation and cancer have revealed many features of endothelial gaps that form in response to different stimuli and are accompanied by leakage [138, 139].
Electron microscopic and confocal microscopic observations have shown endothelial gap diameters ranging from 0.2 to 1.4 μm in venules after substance P or bradykinin [136, 137, 140], but whether gap dimensions and locations are predetermined is an open question. Another unresolved issue is whether different stimuli induce gaps of different size and whether gap dimensions depend on distinct downstream signaling pathways. Endothelial gaps in tumor blood vessels can be much larger and are among many abnormalities in tumor endothelial cells [18]. Despite the barrier defects, leakage in tumors is limited by impaired blood flow and high interstitial pressure [141] (Figure 1).
As a related issue, evidence that Cldn5 deletion leads to leakage of small (< 10 kDa) but not large tracers raises the possibility that loss of this tight junction protein results in barrier defects smaller than typical endothelial gaps [56]. Another interesting finding in cultured endothelial cells is that VE-cadherin at junctions undergoes endocytosis when leakage is induced by VEGFA but appears segmented at the cell border when extravasation is mediated by histamine [45]. The mechanisms underlying these differences must still be determined.
Src family kinases in VE-cadherin phosphorylation and leakage
Src family kinases (SFKs) are cytoplasmic tyrosine kinases implicated in the regulation of leakage [35, 43, 99, 100]. c-Src has been the focus of much research in this context, but the contributions of closely related Yes and Fyn in regulating endothelial junctions remain to be determined. SFKs are constitutively active in venules in a flow-dependent manner, as reflected by phosphorylation of the conserved tyrosine Y418 in postcapillary venules that are susceptible to leakage [35]. Phosphorylation of VE-cadherin by SFKs is a prerequisite but is not sufficient to trigger VE-cadherin internalization and loosening of adherens junctions [35].
Monomeric GTPases and the actin cytoskeleton
Signaling via phosphoinositide 3´ kinase (PI3K) leads to generation of phosphatidyl inositol 3’ phosphate (PIP3)-containing species and activation of monomeric GTPases RhoA, Rac1 and Cdc42, which collectively constitute the Rho GTPase family. Rho GTPases connect to the actin cytoskeleton and, thereby, communicate with adherens junctions and possibly tight junctions. A well-studied effect of Rho GTPases in cultured endothelial cells is the balance between actin stress fibers, which are thought to pull junctions apart, and cortical actin filaments that stabilize junctions. The current state of knowledge on the contribution of Rho family GTPases to endothelial barrier regulation includes multiple lines of evidence from endothelial cells in vitro. RhoA activation leads to formation of radial stress fibers and increased contractility and permeability of endothelial monolayers [101, 102]. Downstream Rho kinases are effectors of RhoA signaling via myosin light chain to stimulate actomyosin contraction [101, 102]. However, the downstream consequence of RhoA activation depends on the stimulus.
Rac1 is required for endothelial barrier maintenance and stability under resting conditions. Thus, Rac1 activation downstream of inflammatory cytokines and VEGFA promotes reassembly of endothelial junctions [103, 104]. VE-cadherin can signal in an outside-in fashion to activate Rac1 [105]. The effect of Rac1 is, however, context- and stimulus-dependent, as VEGFA can induce barrier breakdown via Rac1 [104].
The role of Cdc42 in barrier regulation is less well understood, but Cdc42, as for Rac1, is considered important in barrier maintenance and restoration [106].
Rap1 is implicated in stabilization of endothelial junctions in vivo via the guanine-exchange factor (GEF) Epac1, which is activated in a Rac1-dependent manner. Rap1 strengthens the barrier through formation of cortical actin bundles [107]. Moreover, Rap1 inhibits RhoA activity by Ras-interacting protein (Rasip) and promotes the recruitment of a GTPase activating protein (GAP) for RhoA [108].
Although intercellular separations and other changes in the shape of cultured endothelial cells are thought to reflect cytoskeletal stress fiber tension at junctions, the most conspicuous change in the endothelial cytoskeleton in postcapillary venules in vivo is disruption or loss of the cortical actin rim at focal sites of leakage [109]. Actin stress fibers typical of those in cultured endothelial cells are not found in the leaky vessels [109]. Together, in vivo evidence weighs against actin stress fibers mediating endothelial cell retraction, barrier breakdown, and gap formation in postcapillary venules in vivo [102, 110]. However, the cortical actin cytoskeleton is likely to contribute to the formation of finger-like processes next to gaps, maintain cell-cell contacts during gap formation, and promote gap closure, adherens junction reformation, and resolution of leakage [111].
Focal adhesion kinase (FAK) and cell-matrix adhesion
Cell-cell contacts and cell-matrix adhesions balance the stability of endothelial junctions. Junctional stability is determined by close interactions between integrins at focal adhesions and cadherins at endothelial junctions [112, 113]. Communication between these compartments could involve direct interactions of the cytoplasmic tyrosine kinase FAK and VE-cadherin. FAK participates in anchoring the actin cytoskeleton to focal adhesion sites. Although integrin-mediated FAK activation at focal adhesions is the most potent inducer of FAK activity, FAK can also be activated by SFKs at cell junctions [114]. Substrates for FAK in turn include paxillin, p130CAS, c-Src, cortactin, β-catenin and VE-cadherin [115].
FAK influences vascular permeability by participating in mechanotransduction of blood flow effects on endothelial cells [116, 117]. Blood flow, whether laminar or oscillatory, exerts forces on cell-matrix adhesions via integrins that lead to FAK and SFK activation [118]. FAK/SFK activation in turn leads to phosphorylation of actin-associated proteins and rearrangement of the cytoskeleton. VEGFA promotes FAK relocation from the cytosol to adherens junctions, the association of FAK and VE-cadherin, VE-cadherin phosphorylation on Y658, and increased endothelial permeability [116, 119]. Accordingly, VEGFA-induced vascular leakage is prevented by suppression of FAK kinase activity by genetic disruption or pharmacological inhibition [116]. Genetic inactivation of FAK in endothelial cells also prevents tumor cell transmigration in vitro and extravasation and metastasis in vivo [119].
eNOS/NO regulation of signaling
NO can influence endothelial barrier function in multiple ways through actions on blood vessels. The eNOS-NO pathway is strongly implicated in vascular leakage induced by VEGFA, as well as by inflammatory cytokines. NO diffuses from endothelial cells to surrounding vascular smooth muscle cells, causing vasodilatation, increased flow, and augmented extravasation of fluid and small molecules. Ablation of eNOS expression attenuates VEGFA-induced leakage [120]. NO can also directly regulate the phosphorylation status of VE-cadherin in vitro [91]. The underlying mechanism may involve posttranslational modifications, including nitration and nitrosylation of proteins, which can have gain- or loss-of function effects. Through this process, NO can induce S-nitrosylation of β-catenin, β-catenin dissociation from VE-cadherin, and disassembly of adherens junctions [121].
Concluding Remarks
Vascular leakage is increasingly recognized as a clinically important feature of acute respiratory distress syndrome (ARDS) occurring in COVID-19 and in asthma, arthritis, ulcerative colitis, other chronic inflammatory diseases. Altered endothelial barrier function also plays important roles in cancer, age-related macular degeneration, diabetic macular edema, psychiatric and neurodegenerative diseases, and many other pathological conditions. Because of the importance of plasma leakage in disease pathophysiology and the diversity of these conditions, further work is needed to develop a more thorough understanding of endothelial gap formation and reversal in vivo and mechanisms underlying the regulation of endothelial barrier function in different segments of the vasculature and in different organs. Additional work should also be directed to the mechanisms underlying altered vascular permeability in specific diseases and to strategies for preventing or reversing leakage and the consequences. Also deserving attention are strategies for regulated opening or traversing the endothelial barrier to improve drug delivery to organs protected by the blood-brain barrier, blood-retina barrier, and other sites where the barrier restricts access to therapeutic targets.
Supplementary Material
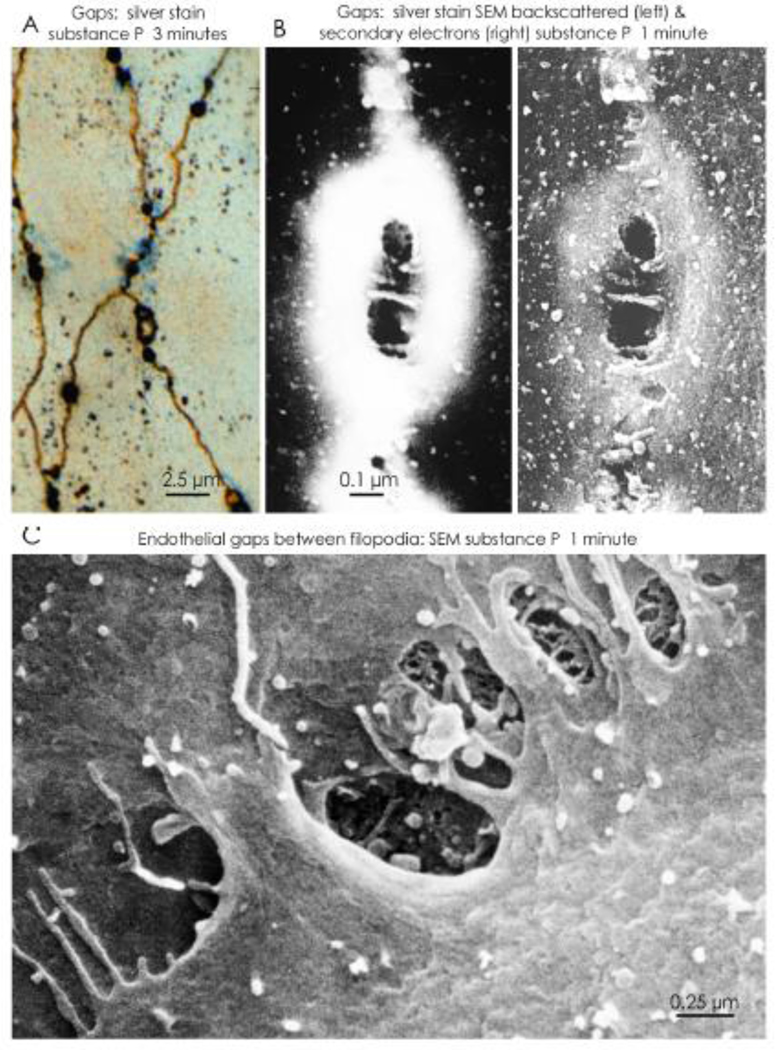
Figure 4. Endothelial gap location and structure.
Distribution and appearance of endothelial gaps in tracheal venules viewed by light microscopy and scanning electron microscopy (SEM) soon after intravenous (iv) injection of the proinflammatory peptide substance P. A. Endothelial gaps are seen as black dots along intercellular junctions (brown lines) in the endothelium of a postcapillary venule stained with silver nitrate 3 minutes after substance P. Leakage of the particulate tracer Monastral blue appears as faint blue staining (from [134]). B. SEM views of endothelial gaps stained with silver nitrate 1 minute after substance P. Silver nitrate appears as a silver annulus (white) around the central gaps (black), which are bridged by endothelial cell filopodia. The gap is visualized by detection of backscattered electrons (left) and secondary electrons (right) (from [136]). C. SEM view of gaps bordered by filopodia at the luminal rim of endothelial cells in a venule 1 minute after iv injection of substance P. Tips of filopodia, reflecting changes in the cortical actin cytoskeleton, maintain contacts between the adjacent endothelial cells. Fibrous basement membrane is visible through some gaps (from [137]).
Clinician’s Corner.
The vascular barrier is essential for normal tissue homeostasis. The barrier is formed by tight junctions and adherens junctions that join endothelial cells, with additional contributions from the endothelial cell glycocalyx on the luminal surface and basement membrane and mural cells on the abluminal surface. Barrier disruption occurs in many diseases, leading to leakage of plasma into tissues, edema formation, and tissue injury.
Transendothelial flux of plasma is governed by endothelial permeability and the balance of intravascular and interstitial hydrostatic and oncotic forces that are elements of normal circulatory physiology. Factors that cause vasoconstriction or dilatation can influence fluid flux through effects on blood pressure and flow independent of changes in permeability.
Endothelial barrier opening can be triggered by VEGFA, histamine, bradykinin, and other mediators that promote separation of endothelial tight junctions and adherens junctions. Mediators can increase permeability by acting directly on endothelial cells and causing junction opening and VE-cadherin internalization or redistribution, or by acting indirectly via factors released from mast cells or other cells. Barrier disruption leads to focal gaps between endothelial cells. Plasma leakage, which is typically greatest from postcapillary venules, can cause tissue edema if the amount exceeds clearance through lymphatics and other routes. The process can be prevented or reversed by Angpt1 activation of Tie2 receptors and by agonists of β-adrenergic receptors on endothelial cells.
Endothelial junction integrity and barrier function can also be changed by Piezo1 and other mechanosensitive ion channels that are activated by increased arterial or venous pressure accompanying left ventricular cardiac failure, head trauma, or other condition.
Insights into the mechanisms of action of factors that increase or decrease endothelial permeability are leading to new approaches for regulating vascular leakage in clinically meaningful ways. Drugs that neutralize VEGFA, inhibit VEGFR2, or activate Tie2 have proven effective in reducing vascular permeability and leakage. Serum Angpt2 is a prognostic biomarker of disease severity in sepsis. Angpt2 inhibitors can complement effects of VEGF signaling inhibitors in retinal vascular disease and cancer. Agents that increase permeability with temporal control and regional specificity could provide localized access of drugs to the CNS where the endothelial barrier normally limits drug delivery.
Outstanding Questions.
Mechanisms of regulation of normal endothelial barrier function
What are the respective contributions of tight junctions and adherens junctions to endothelial barrier function in vivo and how are they coordinated?
What is the mechanism of endothelial gap resealing and how can this be promoted?
How does the cytoskeleton contribute to the formation and closing of endothelial gaps in vivo?
What are the relative contributions to barrier function of the endothelial cell glycocalyx, basement membrane, pericytes, integrins, and focal adhesions to the extracellular matrix?
How does vascular permeability regulation differ among organs?
Do VEGFA, inflammatory cytokines, and other mediators promote leakage in different regions of the vasculature, through different mechanisms, or in other functionally different ways?
Mechanisms underlying dysregulation of endothelial barrier function in disease
How do mechanisms underlying changes in endothelial barrier function differ among inflammatory diseases, cancer, and other pathological conditions?
For leakage occurring in these pathological conditions, what are the relative contributions of increased permeability by endothelial gap formation and changes in transmural hydrostatic and osmotic driving forces?
What ligands, receptors, and signaling mechanisms underlie vascular leakage in inflammatory disease, cancer, and other pathological conditions?
How can altered endothelial barrier function be reversed in these pathological conditions? What are the clinical consequences of suppressing leakage under these conditions?
What strategies can be used to increase regional delivery of therapeutics by regulating endothelial barrier function in the CNS or other sites where endothelial barrier function limits drug access?
Highlights.
In the microcirculation, endothelial permeability varies from least in arterioles to greatest in venules and is regulated in an organ-specific manner to control the extravasation of fluid, solutes, and large molecules.
Inflammatory factors increase vascular permeability by inducing the formation of focal endothelial gaps that can be transient in acute inflammation or sustained in chronic conditions.
Gap formation requires changes in the organization of tight junctions and adherens junctions that join endothelial cells and create a barrier.
Adherens junction opening requires phosphorylation, loss of homophilic interactions, and internalization of VE-cadherin accompanied by changes in the cortical cytoskeleton.
Sustained hyperpermeability in pathologic conditions can lead to edema, reduced vascular perfusion, impaired drug delivery, and exaggerated disease severity.
Acknowledgements and Funding Sources
We thank Drs. Tim Padera of Harvard Medical School, Radu V. Stan of Dartmouth Medical School, and Stefan Liebner of Edinger Institute of Neurology, Goethe University Frankfurt for their valuable insights, and Dr. Dominic Love of Uppsala University for contributions to the figures. We also thank Philipp Jordan (www.JordanGraphics.eu) for the artwork. This work was supported in part by grants from the Swedish Cancer foundation (CAN2016/578), the Swedish Research Council (2015-02375), Knut and Alice Wallenberg Foundation (KAW 2015.0030, 2015.0275), and Fondation Leducq Transatlantic Network of Excellence Grant in Neurovascular Disease (17 CVD 03) to LCW, grants from the Swedish Research Council (2013-9279), Knut and Alice Wallenberg Foundation, and European Research Council (project EC-ERC-VEPC, 742922) to ED, and grants from the National Heart, Lung, and Blood Institute of the US National Institutes of Health (R01 HL143896, R01 HL127402, R01 HL059157) to DMcD.
Glossary
- Adherens junctions
specialized endothelial cell junctions composed of vascular endothelial (VE)-cadherin, a transmembrane protein that joins adjacent endothelial cells through homophilic interactions. These interactions are interrupted at focal regions of endothelial cell contacts where gaps form and serve as routes for plasma leakage.
- Capillary types
capillaries are generally classified as one of three types. Continuous capillaries (brain, lung, muscle) are lined by a continuous layer of endothelial cells joined by intercellular junctions that create a barrier to diffusion. Fenestrated capillaries have greater permeability due to arrays of transendothelial pores that are covered by a diaphragm (endocrine glands, intestinal mucosa, choroid plexus) or not (liver sinusoids, kidney glomerulus). Discontinuous capillaries have endothelial cell barrier properties that enable cells to cross (spleen, bone marrow).
- Edema
accumulations of fluid leaked from blood vessels into the interstitium that exceed clearance by lymphatics and other routes.
- Endothelial barrier
endothelial cell properties, consisting of intercellular junctions, glycocalyx, and basement membrane, that regulate the movement of fluid, solutes, and proteins across the endothelium driven by the transendothelial hydrostatic pressure gradient. Plasma extravasation is regulated separately from cell migration across the endothelial barrier.
- Endothelial fenestrations
arrays of transendothelial pores. In many organs, 60-nm endothelial fenestrations are covered by a diaphragm of spoke-like filaments of endothelial cell-specific plasmalemma vesicle-associated protein (PLVAP) that retains macromolecules and allows rapid transit of water, small peptide hormones (thyroid, pancreatic islets), and steroids (adrenal cortex, ovary). Fenestrations without diaphragms are found in kidney glomeruli (60 nm), where other structures provide macromolecular sieving, and in liver sinusoids (>100 nm).
- Glycocalyx:
layer of membrane-bound glycoproteins, proteoglycans, and glycolipids covering the luminal surface of endothelial cells that serves as a molecular filter at the blood-tissue interface.
- Tight junctions
impermeable strands of transmembrane proteins and lipids that join adjacent endothelial cells and form the barrier to plasma extravasation. Tight junctions are composed of multiple different proteins, vary in composition among vessel types and organs, and are also present in epithelial cells. The number and completeness of tight junction strands determine the relative permeability of the barrier. Formation of transient intercellular gaps leading to plasma extravasation requires changes in tight junctions coordinated with adherens junctions.
- Transendothelial sieving
filtration of molecules of different size and charge across the endothelium driven by hemodynamic forces and governed by the endothelial barrier and other components of the vessel wall. Sieving of plasma by these barriers influences the composition of tissue fluid and lymph.
- Vascular barrier
barriers of the vessel wall that restrict passage into or out of blood vessels, including the endothelial cell barrier and pericytes and smooth muscle cells (mural cells) and their basement membrane.
- Vascular leakage
extravasation of plasma constituents (fluid flux) driven by the transendothelial hydrostatic pressure gradient and governed by vascular permeability.
- Vascular permeability
property of blood vessels that reflects the tightness of the vascular barrier. Vascular permeability is an intrinsic feature of blood vessels that can increase or decrease. Changes in vascular permeability occur in normal physiology and are clinically important features of many diseases. Vascular hyperpermeability refers to increased permeability that can be brief and reversible, as in acute inflammation, or sustained, as in chronic inflammation, cancer, and other prolonged pathological conditions.
Footnotes
Conflicts of interest: The authors have no conflicting financial interests.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Stan RV (2006) Channels across endothelial cells. In Cell-Cell Channels (Baluska F. et al. eds), pp. 271–278, Landes Bioscience, Georgetown, TX, USA. [Google Scholar]
- 2.Ayloo S. and Gu C. (2019) Transcytosis at the blood-brain barrier. Curr Opin Neurobiol 57, 32–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Michel CC (1997) Starling: the formulation of his hypothesis of microvascular fluid exchange and its significance after 100 years. Exp Physiol 82 (1), 1–30. [DOI] [PubMed] [Google Scholar]
- 4.Palade GE et al. (1979) Structural aspects of the permeability of the microvascular endothelium. Acta Physiol Scand Suppl 463, 11–32. [PubMed] [Google Scholar]
- 5.Simionescu M. and Simionescu N. (1984) Ultrastructure of the microvascular wall: functional correlations. Chapter 3. In Handbook of Physiology (Renkin EM and Michel CC eds), pp. 41–101, American Physiological Society. [Google Scholar]
- 6.Bearer EL and Orci L. (1985) Endothelial fenestral diaphragms: a quick-freeze, deep-etch study. J Cell Biol 100 (2), 418–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Sorensen KK et al. (2015) Liver Sinusoidal Endothelial Cells. Compr Physiol 5 (4), 1751–74. [DOI] [PubMed] [Google Scholar]
- 8.Jourde-Chiche N. et al. (2019) Endothelium structure and function in kidney health and disease. Nat Rev Nephrol 15 (2), 87–108. [DOI] [PubMed] [Google Scholar]
- 9.Vestweber D. (2015) How leukocytes cross the vascular endothelium. Nat Rev Immunol 15 (11), 692–704. [DOI] [PubMed] [Google Scholar]
- 10.Wettschureck N. et al. (2019) Passing the Vascular Barrier: Endothelial Signaling Processes Controlling Extravasation. Physiol Rev 99 (3), 1467–1525. [DOI] [PubMed] [Google Scholar]
- 11.Langen UH et al. (2019) Development and Cell Biology of the Blood-Brain Barrier. Annu Rev Cell Dev Biol 35, 591–613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Armulik A. et al. (2010) Pericytes regulate the blood-brain barrier. Nature 468 (7323), 557–61. [DOI] [PubMed] [Google Scholar]
- 13.Augustin HG and Koh GY (2017) Organotypic vasculature: From descriptive heterogeneity to functional pathophysiology. Science 357 (6353). [DOI] [PubMed] [Google Scholar]
- 14.McDonald DM (2001) Angiogenesis and remodeling of airway vasculature in chronic inflammation. Am J Respir Crit Care Med 164 (10 Pt 2), S39–45. [DOI] [PubMed] [Google Scholar]
- 15.Scallan J. et al. (2010) Capillary fluid exchange: regulation, functions, and pathology. In Integrated Systems Physiology: from Molecule to Function to Disease (Copyright (c) 2010 by Morgan & Claypool Life Sciences edn) (Granger DN and Granger JP eds), Morgan & Claypool Life Sciences. [PubMed] [Google Scholar]
- 16.Teuwen LA et al. (2020) COVID-19: the vasculature unleashed. Nat Rev Immunol 20 (7), 389–391. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Park-Windhol C. and D’Amore PA (2016) Disorders of Vascular Permeability. Annu Rev Pathol 11, 251–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McDonald DM and Choyke PL (2003) Imaging of angiogenesis: from microscope to clinic. Nat Med 9 (6), 713–25. [DOI] [PubMed] [Google Scholar]
- 19.Jain RK et al. (2014) The role of mechanical forces in tumor growth and therapy. Annu Rev Biomed Eng 16, 321–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Stamatovic SM et al. (2016) Junctional proteins of the blood-brain barrier: New insights into function and dysfunction. Tissue Barriers 4 (1), e1154641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.West JB (2000) Invited review: pulmonary capillary stress failure. J Appl Physiol (1985) 89 (6), 2483–9;discussion 2497. [DOI] [PubMed] [Google Scholar]
- 22.Beech DJ and Kalli AC (2019) Force Sensing by Piezo Channels in Cardiovascular Health and Disease. Arterioscler Thromb Vasc Biol 39 (11), 2228–2239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Friedrich EE et al. (2019) Endothelial cell Piezo1 mediates pressure-induced lung vascular hyperpermeability via disruption of adherens junctions. Proc Natl Acad Sci U S A 116 (26), 12980–12985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schulte D. et al. (2011) Stabilizing the VE-cadherin-catenin complex blocks leukocyte extravasation and vascular permeability. EMBO J 30 (20), 4157–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Frye M. et al. (2015) Interfering with VE-PTP stabilizes endothelial junctions in vivo via Tie-2 in the absence of VE-cadherin. J Exp Med 212 (13), 2267–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Duong CN et al. (2020) Interference With ESAM (Endothelial Cell-Selective Adhesion Molecule) Plus Vascular Endothelial-Cadherin Causes Immediate Lethality and Lung-Specific Blood Coagulation. Arterioscler Thromb Vasc Biol 40 (2), 378–393. [DOI] [PubMed] [Google Scholar]
- 27.Corada M. et al. (1999) Vascular endothelial-cadherin is an important determinant of microvascular integrity in vivo. Proc Natl Acad Sci U S A 96 (17), 9815–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Rodriguez F. et al. (2008) The receptor protein-tyrosine phosphatase, Dep1, acts in arterial/venous cell fate decisions in zebrafish development. Dev Biol 324 (1), 122–30. [DOI] [PubMed] [Google Scholar]
- 29.Broermann A. et al. (2011) Dissociation of VE-PTP from VE-cadherin is required for leukocyte extravasation and for VEGF-induced vascular permeability in vivo. J Exp Med 208 (12), 2393–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Verma S. and Sharma S. (2018) Protein Tyrosine Phosphatase as Potential Therapeutic Target in various Disorders. Curr Mol Pharmacol 11 (3), 191–202. [DOI] [PubMed] [Google Scholar]
- 31.Braun LJ et al. (2019) VE-PTP inhibition stabilizes endothelial junctions by activating FGD5. EMBO Rep 20 (7), e47046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Vanlandewijck M. and Betsholtz C. (2018) Single-Cell mRNA Sequencing of the Mouse Brain Vasculature. Methods Mol Biol 1846, 309–324. [DOI] [PubMed] [Google Scholar]
- 33.Lampugnani MG et al. (1997) Cell confluence regulates tyrosine phosphorylation of adherens junction components in endothelial cells. J Cell Sci 110 ( Pt 17), 2065–77. [DOI] [PubMed] [Google Scholar]
- 34.Potter MD et al. (2005) Tyrosine phosphorylation of VE-cadherin prevents binding of p120- and beta-catenin and maintains the cellular mesenchymal state. J Biol Chem 280 (36), 31906–12. [DOI] [PubMed] [Google Scholar]
- 35.Orsenigo F. et al. (2012) Phosphorylation of VE-cadherin is modulated by haemodynamic forces and contributes to the regulation of vascular permeability in vivo. Nat Commun 3, 1208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Conway DE et al. (2017) VE-Cadherin Phosphorylation Regulates Endothelial Fluid Shear Stress Responses through the Polarity Protein LGN. Curr Biol 27 (14), 2219–2225 e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Adam AP et al. (2010) Src-induced tyrosine phosphorylation of VE-cadherin is not sufficient to decrease barrier function of endothelial monolayers. J Biol Chem 285 (10), 7045–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Wessel F. et al. (2014) Leukocyte extravasation and vascular permeability are each controlled in vivo by different tyrosine residues of VE-cadherin. Nat Immunol 15 (3), 223–30. [DOI] [PubMed] [Google Scholar]
- 39.Smith RO et al. (2020) Vascular permeability in retinopathy is regulated by VEGFR2 Y949 signaling to VE-cadherin. Elife 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Owen-Woods C. et al. (2020) Local microvascular leakage promotes trafficking of activated neutrophils to remote organs. J Clin Invest 130 (5), 2301–2318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sidibe A. et al. (2014) VE-cadherin Y685F knock-in mouse is sensitive to vascular permeability in recurrent angiogenic organs. Am J Physiol Heart Circ Physiol 307 (3), H455–63. [DOI] [PubMed] [Google Scholar]
- 42.Sidibe A. et al. (2014) Dynamic phosphorylation of VE-cadherin Y685 throughout mouse estrous cycle in ovary and uterus. Am J Physiol Heart Circ Physiol 307 (3), H448–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Li X. et al. (2016) VEGFR2 pY949 signalling regulates adherens junction integrity and metastatic spread. Nat Commun 7, 11017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Mariner DJ et al. (2001) Identification of Src phosphorylation sites in the catenin p120ctn. J Biol Chem 276 (30), 28006–13. [DOI] [PubMed] [Google Scholar]
- 45.Mikelis CM et al. (2015) RhoA and ROCK mediate histamine-induced vascular leakage and anaphylactic shock. Nat Commun 6, 6725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Szymborska A. and Gerhardt H. (2018) Hold Me, but Not Too Tight-Endothelial Cell-Cell Junctions in Angiogenesis. Cold Spring Harb Perspect Biol 10 (8). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dejana E. and Orsenigo F. (2013) Endothelial adherens junctions at a glance. J Cell Sci 126 (Pt 12), 2545–9. [DOI] [PubMed] [Google Scholar]
- 48.Zihni C. et al. (2016) Tight junctions: from simple barriers to multifunctional molecular gates. Nat Rev Mol Cell Biol 17 (9), 564–80. [DOI] [PubMed] [Google Scholar]
- 49.Lee DB et al. (2008) A lipid-protein hybrid model for tight junction. Am J Physiol Renal Physiol 295 (6), F1601–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Shigetomi K. et al. (2018) Adherens junctions influence tight junction formation via changes in membrane lipid composition. J Cell Biol 217 (7), 2373–2381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Winkler L. et al. (2020) Tight junctions in the blood-brain barrier promote edema formation and infarct size in stroke - Ambivalent effects of sealing proteins. J Cereb Blood Flow Metab, 271678X20904687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Simionescu M. et al. (1975) Segmental differentiations of cell junctions in the vascular endothelium. The microvasculature. J Cell Biol 67 (3), 863–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Schneeberger EE and Karnovsky MJ (1976) Substructure of intercellular junctions in freeze-fractured alveolar-capillary membranes of mouse lung. Circ Res 38 (5), 404–11. [DOI] [PubMed] [Google Scholar]
- 54.Taddei A. et al. (2008) Endothelial adherens junctions control tight junctions by VE-cadherin-mediated upregulation of claudin-5. Nat Cell Biol 10 (8), 923–34. [DOI] [PubMed] [Google Scholar]
- 55.Tornavaca O. et al. (2015) ZO-1 controls endothelial adherens junctions, cell-cell tension, angiogenesis, and barrier formation. J Cell Biol 208 (6), 821–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Nitta T. et al. (2003) Size-selective loosening of the blood-brain barrier in claudin-5-deficient mice. J Cell Biol 161 (3), 653–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Honkura N. et al. (2018) Intravital imaging-based analysis tools for vessel identification and assessment of concurrent dynamic vascular events. Nat Commun 9 (1), 2746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Murakami T. et al. (2012) Protein kinase cbeta phosphorylates occludin regulating tight junction trafficking in vascular endothelial growth factor-induced permeability in vivo. Diabetes 61 (6), 1573–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kniesel U. et al. (1996) Development of blood-brain barrier tight junctions in the rat cortex. Dev Brain Res 96 (1–2), 229–40. [DOI] [PubMed] [Google Scholar]
- 60.Varadarajan S. et al. (2019) Multiscale dynamics of tight junction remodeling. J Cell Sci 132 (22). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pohl U. (2020) Connexins: Key Players in the Control of Vascular Plasticity and Function. Physiol Rev 100 (2), 525–572. [DOI] [PubMed] [Google Scholar]
- 62.Kandasamy K. et al. (2015) Changes in endothelial connexin 43 expression inversely correlate with microvessel permeability and VE-cadherin expression in endotoxin-challenged lungs. Am J Physiol Lung Cell Mol Physiol 309 (6), L584–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Okamoto T. et al. (2019) The Functional Implications of Endothelial Gap Junctions and Cellular Mechanics in Vascular Angiogenesis. Cancers (Basel) 11 (2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Schneeberger EE (1982) Structure of intercellular junctions in different segments of the intrapulmonary vasculature. Ann N Y Acad Sci 384, 54–63. [DOI] [PubMed] [Google Scholar]
- 65.Yin J. et al. (2019) Connexin 40 regulates lung endothelial permeability in acute lung injury via the ROCK1-MYPT1- MLC20 pathway. Am J Physiol Lung Cell Mol Physiol 316 (1), L35–l44. [DOI] [PubMed] [Google Scholar]
- 66.Senger DR et al. (1983) Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 219 (4587), 983–5. [DOI] [PubMed] [Google Scholar]
- 67.Simons M. et al. (2016) Mechanisms and regulation of endothelial VEGF receptor signalling. Nat Rev Mol Cell Biol 17 (10), 611–25. [DOI] [PubMed] [Google Scholar]
- 68.Fantin A. et al. (2017) VEGF165-induced vascular permeability requires NRP1 for ABL-mediated SRC family kinase activation. J Exp Med 214 (4), 1049–1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang F. et al. (2018) Lacteal junction zippering protects against diet-induced obesity. Science 361 (6402), 599–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Heinolainen K. et al. (2017) VEGFR3 Modulates Vascular Permeability by Controlling VEGF/VEGFR2 Signaling. Circ Res 120 (9), 1414–1425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Ferrara N. and Adamis AP (2016) Ten years of anti-vascular endothelial growth factor therapy. Nat Rev Drug Discov 15 (6), 385–403. [DOI] [PubMed] [Google Scholar]
- 72.Papadopoulos KP et al. (2016) A Phase I First-in-Human Study of Nesvacumab (REGN910), a Fully Human Anti-Angiopoietin-2 (Ang2) Monoclonal Antibody, in Patients with Advanced Solid Tumors. Clin Cancer Res 22 (6), 1348–55. [DOI] [PubMed] [Google Scholar]
- 73.Sahni J. et al. (2020) Safety and Efficacy of Different Doses and Regimens of Faricimab vs Ranibizumab in Neovascular Age-Related Macular Degeneration: The AVENUE Phase 2 Randomized Clinical Trial. JAMA Ophthalmol 138 (9), 955–963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Teichert M. et al. (2017) Pericyte-expressed Tie2 controls angiogenesis and vessel maturation. Nat Commun 8, 16106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Thurston G. et al. (1999) Leakage-resistant blood vessels in mice transgenically overexpressing angiopoietin-1. Science 286 (5449), 2511–4. [DOI] [PubMed] [Google Scholar]
- 76.Thurston G. et al. (2000) Angiopoietin-1 protects the adult vasculature against plasma leakage. Nat Med 6 (4), 460–3. [DOI] [PubMed] [Google Scholar]
- 77.Mammoto T. et al. (2007) Angiopoietin-1 requires p190 RhoGAP to protect against vascular leakage in vivo. J Biol Chem 282 (33), 23910–8. [DOI] [PubMed] [Google Scholar]
- 78.Korhonen EA et al. (2016) Tie1 controls angiopoietin function in vascular remodeling and inflammation. J Clin Invest 126 (9), 3495–510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kim M. et al. (2016) Opposing actions of angiopoietin-2 on Tie2 signaling and FOXO1 activation. J Clin Invest 126 (9), 3511–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Hakanpaa L. et al. (2015) Endothelial destabilization by angiopoietin-2 via integrin beta1 activation. Nat Commun 6, 5962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Ngok SP et al. (2012) VEGF and Angiopoietin-1 exert opposing effects on cell junctions by regulating the Rho GEF Syx. J Cell Biol 199 (7), 1103–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Fuxe J. et al. (2011) Pericyte requirement for anti-leak action of angiopoietin-1 and vascular remodeling in sustained inflammation. Am J Pathol 178 (6), 2897–909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cho CH et al. (2004) COMP-Ang1: a designed angiopoietin-1 variant with nonleaky angiogenic activity. Proc Natl Acad Sci U S A 101 (15), 5547–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wu FT et al. (2016) Aflibercept and Ang1 supplementation improve neoadjuvant or adjuvant chemotherapy in a preclinical model of resectable breast cancer. Sci Rep 6, 36694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Van Skike CE et al. (2018) Inhibition of mTOR protects the blood-brain barrier in models of Alzheimer’s disease and vascular cognitive impairment. Am J Physiol Heart Circ Physiol 314 (4), H693-h703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Leligdowicz A. et al. (2018) Endothelial Activation: The Ang/Tie Axis in Sepsis. Front Immunol 9, 838. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Reilly JP et al. (2018) Plasma angiopoietin-2 as a potential causal marker in sepsis-associated ARDS development: evidence from Mendelian randomization and mediation analysis. Intensive Care Med 44 (11), 1849–1858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sack KD et al. (2020) The Angiopoietin-Tie2 Pathway in Critical Illness. Crit Care Clin 36 (2), 201–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gillen J. et al. (2019) Angiopoietin-1 and Angiopoietin-2 Inhibitors: Clinical Development. Curr Oncol Rep 21 (3), 22. [DOI] [PubMed] [Google Scholar]
- 90.Marshall I. (1984) Characterization and distribution of histamine H1- and H2-receptors in precapillary vessels. J Cardiovasc Pharmacol 6 Suppl 4, S587–97. [DOI] [PubMed] [Google Scholar]
- 91.Di Lorenzo A. et al. (2009) Akt1 is critical for acute inflammation and histamine-mediated vascular leakage. Proc Natl Acad Sci U S A 106 (34), 14552–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Sharma JN and Al-Dhalmawi GS (2003) Bradykinin receptor antagonists: therapeutic implications. IDrugs 6 (6), 581–6. [PubMed] [Google Scholar]
- 93.Curry FR and Adamson RH (2010) Vascular permeability modulation at the cell, microvessel, or whole organ level: towards closing gaps in our knowledge. Cardiovasc Res 87 (2), 218–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bogatcheva NV et al. (2002) Molecular mechanisms of thrombin-induced endothelial cell permeability. Biochemistry (Mosc) 67 (1), 75–84. [DOI] [PubMed] [Google Scholar]
- 95.Stafforini DM et al. (2003) Platelet-activating factor, a pleiotrophic mediator of physiological and pathological processes. Crit Rev Clin Lab Sci 40 (6), 643–72. [DOI] [PubMed] [Google Scholar]
- 96.Bignami F. et al. (2016) Substance P and its Inhibition in Ocular Inflammation. Curr Drug Targets 17 (11), 1265–74. [DOI] [PubMed] [Google Scholar]
- 97.Chuang YC et al. (2015) Pathogenic roles of macrophage migration inhibitory factor during dengue virus infection. Mediators Inflamm 2015, 547094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Lucke S. and Levkau B. (2010) Endothelial functions of sphingosine-1-phosphate. Cell Physiol Biochem 26 (1), 87–96. [DOI] [PubMed] [Google Scholar]
- 99.Eliceiri BP et al. (2002) Src-mediated coupling of focal adhesion kinase to integrin alpha(v)beta5 in vascular endothelial growth factor signaling. J Cell Biol 157 (1), 149–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Wallez Y. et al. (2007) Src kinase phosphorylates vascular endothelial-cadherin in response to vascular endothelial growth factor: identification of tyrosine 685 as the unique target site. Oncogene 26 (7), 1067–77. [DOI] [PubMed] [Google Scholar]
- 101.Waschke J. et al. (2004) Requirement of Rac activity for maintenance of capillary endothelial barrier properties. Am J Physiol Heart Circ Physiol 286 (1), H394–401. [DOI] [PubMed] [Google Scholar]
- 102.Waschke J. et al. (2004) Role of adhesion and contraction in Rac 1-regulated endothelial barrier function in vivo and in vitro. Am J Physiol Heart Circ Physiol 287 (2), H704–11. [DOI] [PubMed] [Google Scholar]
- 103.Marinkovic G. et al. (2015) The Ins and Outs of Small GTPase Rac1 in the Vasculature. J Pharmacol Exp Ther 354 (2), 91–102. [DOI] [PubMed] [Google Scholar]
- 104.van Buul JD and Timmerman I. (2016) Small Rho GTPase-mediated actin dynamics at endothelial adherens junctions. Small GTPases 7 (1), 21–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Timmerman I. et al. (2015) A local VE-cadherin and Trio-based signaling complex stabilizes endothelial junctions through Rac1. J Cell Sci 128 (18), 3514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Waschke J. et al. (2006) Activation of Rac-1 and Cdc42 stabilizes the microvascular endothelial barrier. Histochem Cell Biol 125 (4), 397–406. [DOI] [PubMed] [Google Scholar]
- 107.Noda K. et al. (2010) Vascular endothelial-cadherin stabilizes at cell-cell junctions by anchoring to circumferential actin bundles through alpha- and beta-catenins in cyclic AMP-Epac-Rap1 signal-activated endothelial cells. Mol Biol Cell 21 (4), 584–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Kooistra MR et al. (2005) Epac1 regulates integrity of endothelial cell junctions through VE-cadherin. FEBS Lett 579 (22), 4966–72. [DOI] [PubMed] [Google Scholar]
- 109.Thurston G. et al. (1995) Changes in endothelial actin cytoskeleton at leakage sites in the rat mesenteric microvasculature. Am J Physiol 268 (1 Pt 2), H316–29. [DOI] [PubMed] [Google Scholar]
- 110.Adamson RH et al. (2003) PAF- and bradykinin-induced hyperpermeability of rat venules is independent of actin-myosin contraction. Am J Physiol Heart Circ Physiol 285 (1), H406–17. [DOI] [PubMed] [Google Scholar]
- 111.Shigetomi K. and Ikenouchi J. (2019) Cell Adhesion Structures in Epithelial Cells Are Formed in Dynamic and Cooperative Ways. Bioessays 41 (7), e1800227. [DOI] [PubMed] [Google Scholar]
- 112.Yamamoto H. et al. (2015) Integrin beta1 controls VE-cadherin localization and blood vessel stability. Nat Commun 6, 6429. [DOI] [PubMed] [Google Scholar]
- 113.Pulous FE et al. (2019) Talin-Dependent Integrin Activation Regulates VE-Cadherin Localization and Endothelial Cell Barrier Function. Circ Res 124 (6), 891–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Kleinschmidt EG and Schlaepfer DD (2017) Focal adhesion kinase signaling in unexpected places. Curr Opin Cell Biol 45, 24–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Sulzmaier FJ et al. (2014) FAK in cancer: mechanistic findings and clinical applications. Nat Rev Cancer 14 (9), 598–610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Chen XL et al. (2012) VEGF-induced vascular permeability is mediated by FAK. Dev Cell 22 (1), 146–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Arnold KM et al. (2013) Loss of focal adhesion kinase enhances endothelial barrier function and increases focal adhesions. Microcirculation 20 (7), 637–49. [DOI] [PubMed] [Google Scholar]
- 118.Schwartz MA (2010) Integrins and extracellular matrix in mechanotransduction. Cold Spring Harb Perspect Biol 2 (12), a005066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Jean C. et al. (2014) Inhibition of endothelial FAK activity prevents tumor metastasis by enhancing barrier function. J Cell Biol 204 (2), 247–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Fukumura D. et al. (2001) Predominant role of endothelial nitric oxide synthase in vascular endothelial growth factor-induced angiogenesis and vascular permeability. Proc Natl Acad Sci U S A 98 (5), 2604–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Thibeault S. et al. (2010) S-nitrosylation of beta-catenin by eNOS-derived NO promotes VEGF-induced endothelial cell permeability. Mol Cell 39 (3), 468–76. [DOI] [PubMed] [Google Scholar]
- 122.Saria A. et al. (1983) Vascular protein linkage in various tissue induced by substance P, capsaicin, bradykinin, serotonin, histamine and by antigen challenge. Naunyn Schmiedebergs Arch Pharmacol 324 (3), 212–8. [DOI] [PubMed] [Google Scholar]
- 123.Hatanaka K. et al. (2012) Fibroblast growth factor signaling potentiates VE-cadherin stability at adherens junctions by regulating SHP2. PLoS One 7 (5), e37600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Lin YM et al. (2012) Hepatocyte growth factor increases vascular endothelial growth factor-A production in human synovial fibroblasts through c-Met receptor pathway. PLoS One 7 (11), e50924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Cavallaro U. and Dejana E. (2011) Adhesion molecule signalling: not always a sticky business. Nat Rev Mol Cell Biol 12 (3), 189–97. [DOI] [PubMed] [Google Scholar]
- 126.Gawlowski DM et al. (1982) Reproducibility of microvascular permeability responses to successive topical applications of bradykinin in the hamster cheek pouch. Microvasc Res 24 (3), 354–63. [DOI] [PubMed] [Google Scholar]
- 127.Heltianu C. et al. (1982) Histamine receptors of the microvascular endothelium revealed in situ with a histamine-ferritin conjugate: characteristic high-affinity binding sites in venules. J Cell Biol 93 (2), 357–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Curry FR and Adamson RH (2013) Tonic regulation of vascular permeability. Acta Physiol (Oxf) 207 (4), 628–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Radeva MY and Waschke J. (2018) Mind the gap: mechanisms regulating the endothelial barrier. Acta Physiol (Oxf) 222 (1). [DOI] [PubMed] [Google Scholar]
- 130.Roberts OL and Dart C. (2014) cAMP signalling in the vasculature: the role of Epac (exchange protein directly activated by cAMP). Biochem Soc Trans 42 (1), 89–97. [DOI] [PubMed] [Google Scholar]
- 131.McDonald DM (1988) Neurogenic inflammation in the rat trachea. I. Changes in venules, leucocytes and epithelial cells. J Neurocytol 17 (5), 583–603. [DOI] [PubMed] [Google Scholar]
- 132.Kruse K. et al. (2019) N-cadherin signaling via Trio assembles adherens junctions to restrict endothelial permeability. J Cell Biol 218 (1), 299–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Majno G. and Palade GE (1961) Studies on inflammation. 1. The effect of histamine and serotonin on vascular permeability: an electron microscopic study. J Biophys Biochem Cytol 11, 571–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.McDonald DM (1994) Endothelial gaps and permeability of venules in rat tracheas exposed to inflammatory stimuli. Am J Physiol 266 (1 Pt 1), L61–83. [DOI] [PubMed] [Google Scholar]
- 135.Thurston G. et al. (1996) Permeability-related changes revealed at endothelial cell borders in inflamed venules by lectin binding. Am J Physiol 271 (6 Pt 2), H2547–62. [DOI] [PubMed] [Google Scholar]
- 136.Hirata A. et al. (1995) Location of focal silver staining at endothelial gaps in inflamed venules examined by scanning electron microscopy. Am J Physiol 269 (3 Pt 1), L403–18. [DOI] [PubMed] [Google Scholar]
- 137.Baluk P. et al. (1997) Endothelial gaps: time course of formation and closure in inflamed venules of rats. Am J Physiol 272 (1 Pt 1), L155–70. [DOI] [PubMed] [Google Scholar]
- 138.McDonald DM et al. (1999) Endothelial gaps as sites for plasma leakage in inflammation. Microcirculation 6 (1), 7–22. [PubMed] [Google Scholar]
- 139.Carmeliet P. and Jain RK (2011) Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases. Nat Rev Drug Discov 10 (6), 417–27. [DOI] [PubMed] [Google Scholar]
- 140.Baffert F. et al. (2006) Angiopoietin-1 decreases plasma leakage by reducing number and size of endothelial gaps in venules. Am J Physiol Heart Circ Physiol 290 (1), H107–18. [DOI] [PubMed] [Google Scholar]
- 141.Fukumura D. and Jain RK (2007) Tumor microenvironment abnormalities: causes, consequences, and strategies to normalize. J Cell Biochem 101 (4), 937–49. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.