Abstract
BACKGROUND
Diethylnitrosamine (DEN) induces hepatic neoplastic lesions over a prolonged period.
AIM
To investigate the promotive action of 2-acetylaminofluorene (2-AAF) when combined with DEN in order to develop a rat model for induction of precancerous lesion and investigate the molecular mechanism underlying the activity of 2-AAF.
METHODS
The pre-precancerous lesions were initiated by intraperitoneal injection of DEN for three weeks consecutively, followed by one intraperitoneal injection of 2-AAF at three different doses (100, 200 and 300 mg/kg). Rats were separated into naïve, DEN, DEN + 100 mg 2-AAF, DEN + 200 mg 2-AAF, and DEN + 300 mg 2-AAF groups. Rats were sacrificed after 10 wk and 16 wk. Liver functions, level of alpha-fetoprotein, glutathione S-transferase-P and proliferating cell nuclear antigen staining of liver tissues were performed. The mRNA level of RAB11A, BAX, p53, and Cyclin E and epigenetic regulation by long-noncoding RNA (lncRNA) RP11-513I15.6, miR-1262 (microRNA), and miR-1298 were assessed in the sera and liver tissues of the rats.
RESULTS
2-AAF administration significantly increased the percent area of the precancerous foci and cell proliferation along with a significant decrease in RAB11A, BAX, and p53 mRNA, and the increase in Cyclin E mRNA was associated with a marked decrease in lncRNA RP11-513I15.6 expression with a significant increase in both miR-1262 and miR-1298.
CONCLUSION
2-AFF promoted hepatic precancerous lesions initiated through DEN by decreasing autophagy, apoptosis, and tumor suppression genes, along with increased cell proliferation, in a time- and dose-dependent manner. These actions were mediated under the epigenetic regulation of lncRNA RP11-513I15.6/miR-1262/miR-1298.
Keywords: Acetylaminofluorene, Hepatic precancerous lesion, Diethylnitrosamine, Autophagy, Apoptosis, MicroRNA
Core Tip: 2-Acetylaminofluorene epigenetically regulated the expression of long-noncoding RNA RP11-513I15.6/miRNA-1262/miR-1298 (microRNA, miRNA) resulted in decrease in RAB11A, BAX, and p53 mRNA, and the increase in Cyclin E mRNA leading to increased hepatocyte proliferation and decreased apoptosis promoting hepatocellular promoted precancerous lesion in rat models.
INTRODUCTION
Hepatocellular carcinoma (HCC) is the 6th common cancer and the 2nd leading cause of cancer mortality all over the world[1]. Its incidence is elevated, which is attributed to the rising proportion of individuals infected with hepatitis C virus[2]. The molecular pathogenesis of cancer and the underlying tumor biology has been progressing. Spontaneous animal models, induced models, transplantable models, transgenic models, and viral models were used to investigate the biological mechanism of HCC with respect to the liver-targeted key pathways[3]. Rodent has a short life span due to which the cellular transformation is observed from initiation to malignancy, thereby rendering it as a preferred model system[4]. However, modeling a malignant liver disease is challenging due to the urgent need for optimal models for preclinical studies.
Several hepatotoxic agents, such as carcinogen diethylnitrosamine (DEN), have been repeatedly administered to induce general liver disease and HCC over a prolonged period. DEN produces small foci of dysplastic hepatocytes via ethylation of various nucleophilic sites in deoxyribonucleic acid[5], resulting in cirrhosis and liver cancer within 18 wk as presented by mutations in β-catenin[6] and p53[7]. HCC induced by DEN activates the H-ras proto-oncogene[8]. Interestingly, variable time intervals, tumor promoters, DEN doses, and application routes were applied by various groups to induce hepatic precancerous lesions in a dose- and time-dependent manner. A two-stage model was established using DEN as a genotoxic compound and phenobarbital to induce HCC[9]. Another two-step HCC model was established according to the Solt-Farber protocol; herein, the initiation by DEN was followed by partial hepatectomy, leading to an elevated number of initiated cells[10].
2-Acetylaminofluorene (2-AAF) serves as a model carcinogen with genotoxic and epigenetic properties[11]. The present study proposed that genotoxic 2-AAF metabolites produce G to T transversion-initiated cells along with cirrhotic alteration due to chronic toxic effect on mitochondrial respiration[12]. Also, electron drainage by 2-AAF causes an uncoupling effect on oxidative phosphorylation[13].
Malik et al[14] reported a protocol for HCC induction in the liver without hepatectomy, wherein male Wistar rats were injected with DEN intraperitoneally, and then, 2-AAF repeatedly. This model showed oxidative stress, cell damage, and advanced HCC.
The present study aimed to investigate the development of precancerous lesions by DEN injection intraperitoneally (100 mg/kg body weight), followed by a single intraperitoneal (i.p.) injection of promoter 2-AAF at three different doses (100, 200 and 300 mg/kg) at two intervals of 10 wk and 16 wk, respectively.
MATERIALS AND METHODS
Chemicals
DEN with ≥ 99% purity (CAT number 55-18-5) and 2-AAF with ≥ 98% purity (CAT number 53-96-3) were purchased from (Sigma-Aldrich, St. Louis, United States).
Experimental protocol
A total of 60 adult male Wistar rats (200-250 g) were used. The animals were maintained at 22-24 °C and twelve hours light/dark cycles and received standard rat chow and tap water. All animal experiments were carried out according to the National Institute of Health guide for dealing with laboratory animals (National Research Council (US) Institute for Laboratory Animal Research. No. 85-23, revised 1996). The study was approved by Ain Shams University, Faculty of Medicine Institutional Animal Ethics Committee (approval No. 17585). The animals were acclimatized for 1 wk and weighed before each injection for accurate determination of the drug dosage.
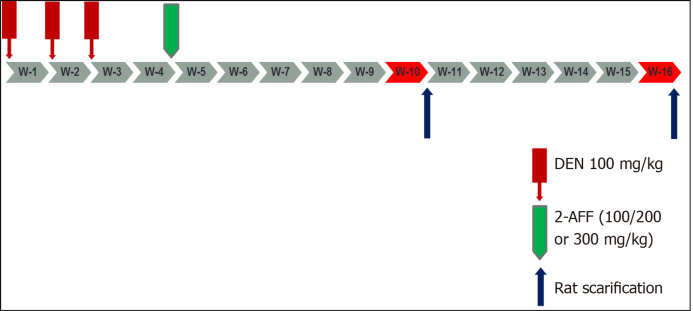
Wister rats were randomly and equally divided into naïve, DEN, DEN + 100 mg 2-AAF, DEN + 200 mg 2-AAF, and DEN + 300 mg 2-AAF groups. The four DEN groups were injected i.p. with 100 mg/kg per week for 3 wk, followed by 1 wk interval. Then, 2-AAF was injected once intraperitoneally at 3 different doses for the 2-AAF three groups (100, 200 and 300 mg/kg). The naïve group was injected with 0.9% NaCl as described above. In each group, half of the animals were sacrificed at the end of week 10 and the remaining at week 16 (Figure 1).
Figure 1.
Schematic of the study design. DEN: Diethylnitrosamine; 2-AAF: 2-Acetylaminofluorene.
Specimen collection
Rats were anesthetized before withdrawing the retro-orbital blood samples; sera were collected by centrifugation at 1200 × g for 10 min. Subsequently, the rats were sacrificed, and liver samples collected. All the samples were maintained at -80 °C for further tests of liver function, and the level of alpha-fetoprotein (AFP) and RNA extraction in the liver samples were examined.
Tissue preparation for histological and immunohistochemical examinations
The liver specimens were collected from all animals in each group, with fixation in 10% neutral formaldehyde for 24 h, followed by dehydration, then embedded in paraffin blocks. Then, 5 μm sections were subjected to hematoxylin-eosin (HE) staining to detect any histopathological changes. Images were captured using an Olympus BX50 Light microscope (Olympus, Japan).
Glutathione S transferase-placental immunohistochemistry
The sections were dewaxed using xylene, followed by hydration using ethanol gradient. The endogenous peroxidase activity was inhibited by hydrogen peroxide. Subsequently, the sections were washed with water and rinsed with phosphate-buffered saline (PBS) before probing with glutathione S-transferase-P (GST-P) primary antibody (1:250; Abcam, cat.# AB106268, San Francesco, CA, United States) at 4 °C overnight. The GST-P-positive area stained brown. The morphometric analysis was carried out using Leica Q win V.3 software after capturing the images using a Leica DM2500 microscope (Leica, Wetzlar, Germany).
Proliferating cell nuclear antigen immunohistochemistry staining
The sections were prepared for proliferating cell nuclear antigen (PCNA) staining (1:400; Santa Cruz Biotechnology, Santa Cruz, CA, United States) for 2 h as described above. Irrespective of the location within the hepatic lobule of the staining intensity, the nuclei were scored as positive or negative. The PCNA labeling indices are represented as the expression of positively stained nuclei (10 fields/slide at × 400).
AFP and liver function
The levels of alanine aminotransferase (ALT), AFP, total bilirubin, and direct bilirubin were analyzed quantitatively using a commercial ELISA kit on sera samples.
Molecular assay
Bioinformatics-based selection of molecular parameters to investigate the oncogenic mechanism of the chemicals used in the HCC model: The molecular biomarker panel was obtained in two steps: (1) A panel of key genes, such as Ras-related in brain11gene (RAB11A), p53, BAX and cell cycle-related gene Cyclin E1 according to Gene Atlas Data Base (https://www.ebi.ac.uk/gxa/home) and protein Atlas Data Base (https://www.proteinatlas.org/) that play a major role in hepatic carcinogenesis, including autophagy, apoptotic genes, and cell cycle; and (2) lncRNA-RP11-513I15.6 was selected using a database of long-noncoding RNA (lncRNA) that act as competitive endogenous RNA (ceRNAs) (http://gyanxet-beta.com/Lncedb/index.php). This lncRNA acts as a master regulator of the target mRNAs by competing with miR (microRNA, miRNA)-1262 and miR-1298 binding with the genes mentioned above. The selected lncRNA and miRNA were based on the specificity to HCC, competing endogenous RNA score, and the number of target sites of mRNA. Finally, the pathway enrichment analysis by Diana database (http://www.microrna.gr/miRPathv2) for both miR-1262 and miR-1298 revealed that these were linked to autophagy, cell cycle regulation, cell adhesion, and other pathways associated to carcinogenesis.
Total RNA extraction
Total RNA was extracted from sera samples by miRNEasy® RNA isolation kit (Qiagen, Düsseldorf, Germany). The RNA integrity and concentration were determined on an Ultraspec 1000 UV/visible spectrophotometer (Amersham Pharmacia Biotech, Cambridge, United Kingdom). The RNA purity was 1.8-2. Subsequently, the total RNA was reverse transcribed into complementary DNA by miScript II RT Kit (Qiagen, Düsseldorf, Germany) on a Hybaid thermal cycler (Thermo Electron, Waltham, MA, United States).
Real-time quantitative Polymerase Chain Reaction of the RNA panel
The expression of mRNA and lncRNA in the rat sera and liver tissues was measured by RT² SYBR Green ROX real-time quantitative polymerase chain reaction (qPCR) Mastermix and Quantitect SYBR Green Mastermix Kit (Qiagen, Düsseldorf, Germany), respectively. The specific primers were provided (Qiagen, Düsseldorf, Germany), using Step One Plus™ System (Applied Biosystems Inc., Foster City, CA, United States). B-actin (accession NM_001101) served as the endogenous control.
The miRNA expression in the sera and liver tissue was investigated according to the protocol of miScript SYBR Green kit Qiagen (Düsseldorf, Germany). RNU-6 served as the endogenous control. The specific PCR primers were synthesized by Qiagen (Düsseldorf, Germay).
The PCR program was according to the following cycles: Denaturation at 95 °C for 15 min followed by forty cycles of denaturation for 10 s at 94 °C, then annealing for 30 s at 55 °C, and finally extension for 34 s at 70 °C. Each reaction was done in duplicate.
The threshold cycle (Ct) value of each sample was calculated using the StepOnePlus™ software v2.2.2 (Applied Biosystems). Ct value > 36 was considered negative. The specificities of the amplicons were confirmed using the melting curve analysis software of Applied Biosystems. The expression of the target molecules was measured using the 2-ΔΔCt method[15]. The expression of the target gene was normalized against that of the housekeeping gene for the samples and compared to the reference sample.
Statistical analysis
The values are expressed as means ± SD. The statistical differences among all groups were assessed using one-way ANOVA, and Tukey’s test. P < 0.05 was considered to be statistically significant. The statistical analyses were done using Graphpad Prism, version 5.0. (2007: San Diego, United States).
RESULTS
The naïve groups at weeks 10 and 16 that did not show significant differences were pooled as a single group.
Histological and immunohistochemical examination
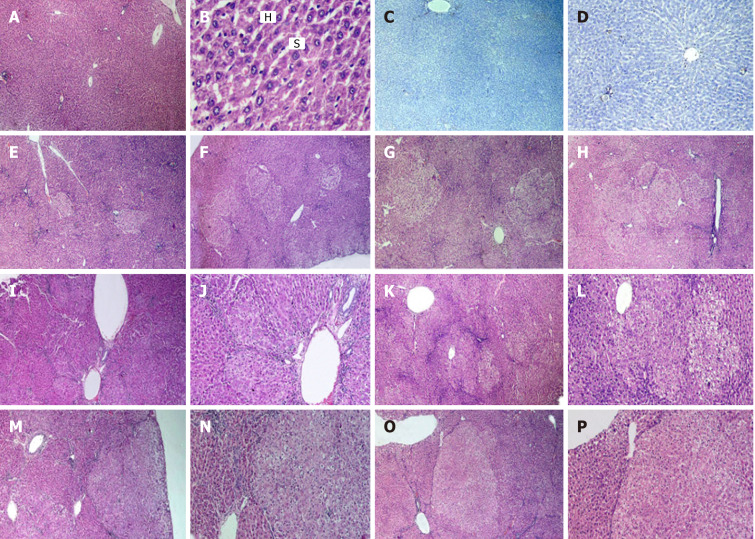
The liver sections of the naïve control group stained with HE revealed normal architecture of hepatic lobules, central veins, and portal triads. Neither localized lesion nor alternating pre precancerous foci or dysplastic nodules were observed throughout the experimental period (Figure 2A-D).
Figure 2.
Histological and immunohistochemical examination. A-D: Images of liver sections of naive group. Hematoxylin-eosin (HE) stained sections show normal hepatic architecture, portal triad, central vein and radiating cords of hepatocytes (H) with blood sinusoids (S) present in between (A and B). Immunohistochemically-stained section with anti-glutathione S transferase-P demonstrating negative reaction (C). Immunohistochemically-stained section with proliferating cell nuclear antigen antibodies (D); E-H: HE images of liver sections of rats that received diethylnitrosamine (DEN) and different doses of 2-acetylaminofluorene (2-AAF) and were sacrificed at week 10. Show multiple foci of cellular alteration of different sizes (dotted shapes), not compressing the surrounding hepatic parenchyma. DEN group (E), DEN+ 2-AAF 100 mg group (F), DEN + 2-AAF 200 mg group (G) and DEN + 2-AAF 300 mg group (H); I-P: HE liver sections of rats that received DEN and different doses of 2-AAF sacrificed at week 16, show larger, well discriminated, less differentiated dysplastic nodules compressing the surrounding liver tissue with disruption of hepatic lobular architecture were observed. DEN group (I and J), DEN + 100 mg 2AAF group (K and L), DEN + 200 mg 2AAF group (M and N), DEN + 300 mg 2AAF group (O and P). A, C, E-H × 40; D, J, L, N and P × 100; I, K, M and O × 40, B × 400.
The histopathology of the liver sections of different groups with DEN either alone or when combined with 2-AAF showed the development of multistage hepatocellular pre precancerous lesions. An apparent increase in the incidence, number, and size of the lesions was observed as a result of increased dose and duration of the usage of DEN and 2-AAF. The liver specimens of rats sacrificed at week 10 showed small early and well-differentiated foci of cellular alteration after injection of DEN solely (Figure 2E), while varying numbers of multiple aggregations of small nodules were present after administration of both DEN + 2-AAF (Figure 2F-H). The simultaneous occurrence of multiple nodules reflected either the dissemination of hepatocytes with cellular atypia from a single primary lesion to form satellite nodules or the synchronous development of several other independent lesions. The localized lesions of foci of cellular alteration did not compress the surrounding hepatic parenchyma but merged with it imperceptibly. However, lack of or minimal disruption of hepatic lobular architecture was observed.
The histological analysis of these pre- precancerous lesions varied greatly from week 10-16 with respect to different stages of differentiation and growth patterns. The lesions observed by the end of week 16 were large and less differentiated (Figure 2I-P). Multiple dysplastic nodules were scattered, compressing the surrounding liver parenchyma and occupying most of the examined fields. These dysplastic nodules were uniform lesions and discriminated from the surrounding liver tissue based on their morphology, cytoplasmic staining, size of the nucleus, and presence of cellular atypia. The nodular cells did not show sinusoidal spaces and were large with clear cytoplasm.
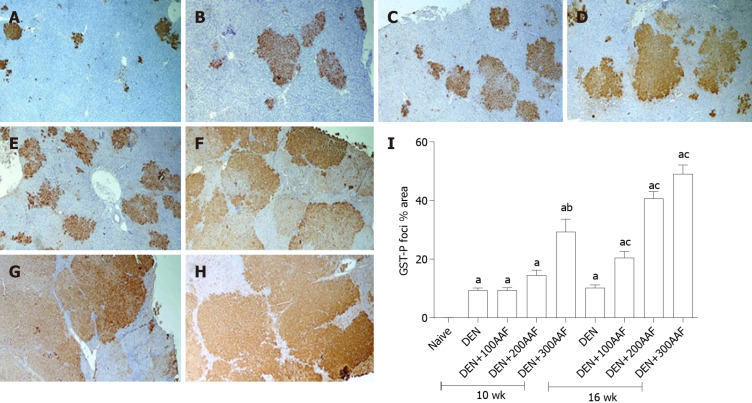
The immunohistochemically-stained liver sections with the GST-P antibody revealed the presence of multiple GST-P-positive areas in all groups after administration of DEN + 2-AAF. Moreover, small positive areas of cellular foci were noted in the group treated with DEN and sacrificed at week 10 (Figure 3A). Multiple GST-P-positive areas, variable in size, were scattered in-between negatively stained hepatocytes among groups treated with DEN + 2-AFF and sacrificed at week 10 (Figure 3B-D). The number and size of the GST-P-positive areas were markedly increased in groups that received DEN + 2-AFF and sacrificed at week 16, especially those that received high doses showed large positive hyperplastic nodules occupying most of the examined fields (Figure 3E-H). The % surface area of GST-P-positive hepatic lesions was measured among different groups and statistically analyzed (Figure 3I).
Figure 3.
Histological and immunohistochemical examination. A-H: Images of rats' liver sections immunohistochemically-stained with glutathione S transferase-P (GST-P) antibody, show multiple GST-P-positive hepatic foci and nodules (brown stained collection of cells) of different sizes scatter in-between negatively stained hepatic parenchyma. Rats sacrificed at week 10 (A-D), rats sacrificed at week 16 (E-H) [A and E: diethylnitrosamine (DEN) group; B and F: DEN + 100mg 2-acetylaminofluorene (2-AAF) group; C and G: DEN + 200 mg 2-AAF group; D and H: DEN + 300 mg 2-AAF (× 40)]; I: shows the effect of DEN and 2-AAF at different doses on GSTP foci % area in the liver. Values are mean ± SE; number of animals = 6 rats/each group. aP < 0.05 compared to naïve group; bP < 0.05 compared to DEN group at week 10, cP < 0.05 compared to DEN at week 16 group. One-way ANOVA followed by Tukey's multiple comparison test. DEN: Diethylnitrosamine; 2-AAF: 2-Acetylaminofluorene; GST-P: Glutathione S transferase-P.
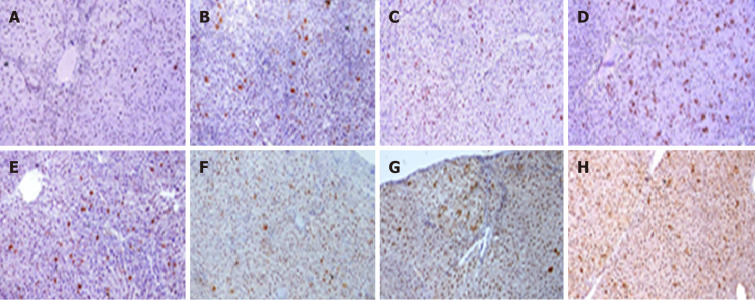
The immunohistochemical analysis showed an elevated expression of PCNA in groups that received DEN + 2-AAF as compared to those treated with DEN alone. The higher the dose of 2-AAF combined with DEN and longer the duration, higher the expression rate. Strikingly, significant differences were detected between DEN/2-AAF 200 and 300 as compared to DEN/2-AAF 100 at weeks 10 and 16, respectively (Figure 4 and Table 1).
Figure 4.
Images of rats' liver sections stained immunohistochemically with proliferating cell nuclear antigen. Positive immune-reactive nucleus (brown dots) scatter in-between negatively stained liver tissue of rats who received diethylnitrosamine (DEN) and different doses of 2-acetylaminofluorene (2-AAF). A-D: Rats sacrificed at week 10; E-H: Rats sacrificed at week 16 (A and E: DEN group; B and F: DEN + 100 mg 2AAF group; C and G: DEN + 200 mg 2AAF group; D and H: DEN + 300 mg 2AAF; magnification × 100).
Table 1.
Expression rate of hepatocytes positive for proliferating cell nuclear antigen was calculated as number of positive field expression in 10 fields per rat liver tissue
|
Group
|
10 wk duration
|
16 wk duration
|
| DEN | + | ++ |
| DEN + 100 AAF | + | ++ |
| DEN + 200 AAF | ++ | +++ |
| DEN + 300 AAF | ++ | +++ |
+: Positive expression found in 1-3 fields; ++: Positive expression found in 4-6 fields; +++: Positive expression found in 7-10 field. DEN: Diethylnitrosamine; AAF: Acetylaminofluorene.
Effect on liver function and AFP
Table 2 showed that by the end of weeks 10 and 16, liver function tests (ALT, albumin, T-bilirubin, D-bilirubin) and AFP had a significant decline after DEN and 2-AAF were administered at three doses as compared to the naïve group. 2-AAF addition to DEN significantly increased the level of AFP as compared to DEN alone with significant differences between 2-AAF doses at the two time points in a dose-dependent manner.
Table 2.
Effect of diethylnitrosamine and 2-acetylaminofluorene on alpha-fetoprotein and liver function
|
|
AFP
|
ALT
|
Total bilirubin
|
Direct bilirubin
|
Albumin
|
| Naïve | 22.8 ± 1.13 | 33.3 ± 6.83 | 0.30 ± 0.18 | 0.27 ± 0.14 | 3.77 ± 0.23 |
| Week 10 | |||||
| DEN | 89.2 ± 28.8d | 63.0 ± 27.5d | 1.44 ± 0.45d | 1.03 ± 0.14d | 2.49 ± 0.15d |
| DEN + 100 AAF | 116 ± 52.1d | 78.3 ± 17.8d | 2.07 ± 0.44d,e | 1.40 ± 0.39d | 2.83 ± 0.19d |
| DEN + 200 AAF | 223 ± 124b,d | 82.7 ± 12.7d | 2.73 ± 0.23a,d,e | 1.67 ± 0.19a,d,e | 2.73 ± 0.14d |
| DEN + 300 AAF | 305 ± 126d,e | 98.0 ± 10.7d,e | 3.13 ± 0.36b,d,e | 2.13 ± 0.61d,e | 3.15 ± 0.38c,d,e |
| Week 16 | |||||
| DEN | 159 ± 32.2d | 94.1 ± 6.4d | 2.13 ± 0.55d | 1.60 ± 0.39d | 2.0 ± 0.62d |
| DEN + 100 AAF | 290 ± 241d | 104 ± 31.9d | 2.23 ± 0.36d | 2.25 ± 0.63d | 2.57 ± 0.37d |
| DEN + 200 AAF | 815 ± 143a,d,f | 128 ± 36.9d | 4.10 ± 0.39a,d,f | 2.53 ± 0.63a,d,f | 2.17 ± 0.29d |
| DEN + 300 AAF | 1059 ± 360b,d,f | 210 ± 63.2b,c,d,f | 4.47 ± 0.99b,d,f | 3.10 ± 0.39d,f | 2.13 ± 0.67b,c,d,f |
Values are mean ± SD; number of animals = 6 rats/each group.
P < 0.05 when DEN + 200 acetylaminofluorene (AAF) is compared to the DEN + 100 AAF.
P < 0.05 when DEN + 300 AAF is compared to the DEN + 100 AAF.
P < 0.05 when DEN + 300 AAF is compared to the DEN + 200 AAF.
P < 0.05 compared to the naïve group.
P < 0.05 compared to the diethylnitrosamine (DEN) group at week 10 group.
P < 0.05 compared to the DEN at week 16 group. One-way ANOVA followed by Tukey's multiple comparison test. DEN: Diethylnitrosamine; 2-AAF: 2-Acetylaminofluorene; AFP: Alpha-fetoprotein; ALT: Alanine aminotransferase.
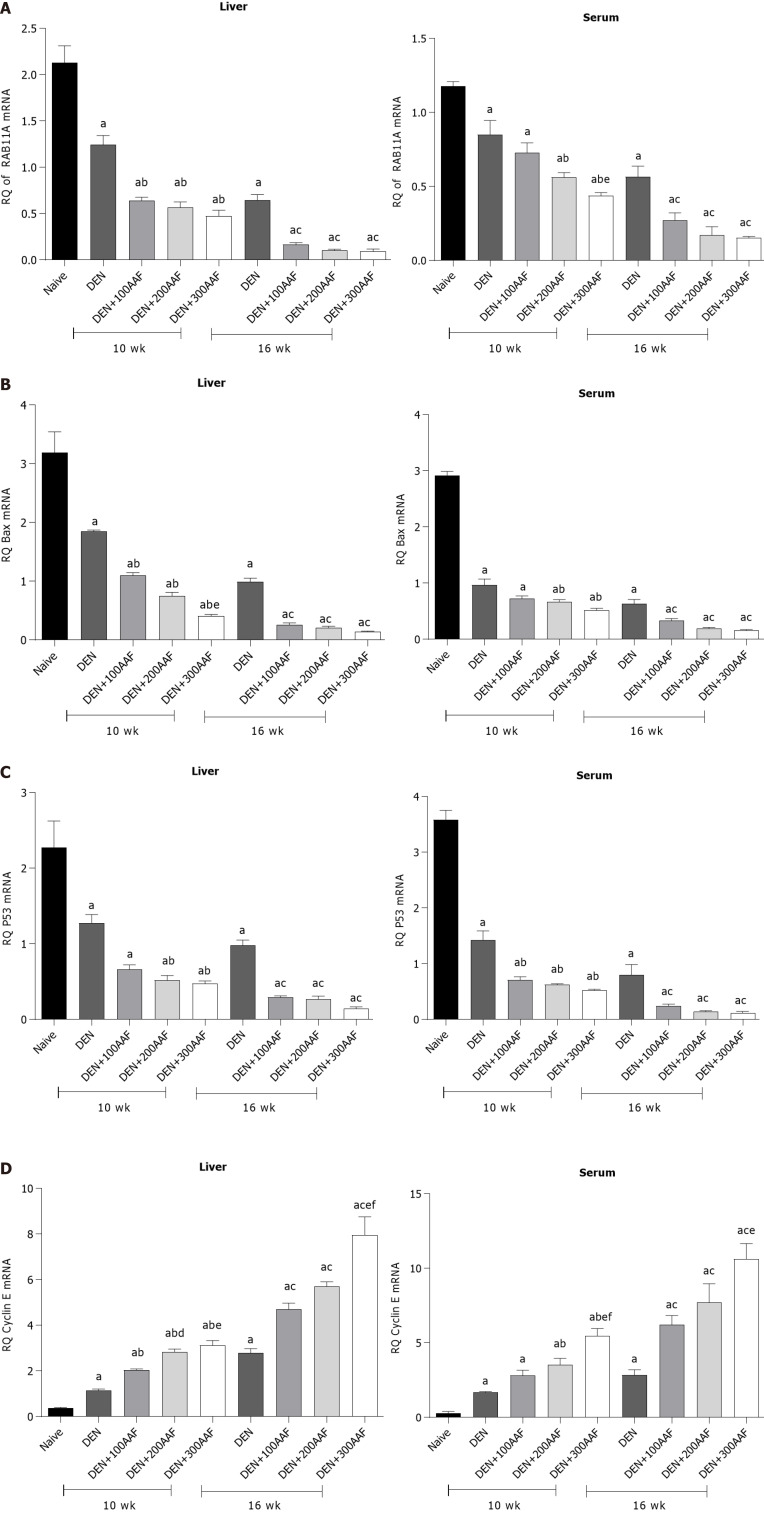
Effect of DEN/2-AAF on the expression of RAB11A, BAX, p53, Cyclin E mRNA among the rat groups
The fold-changes in the relative quantification (RQ) of RAB11A mRNA in rats' liver tissues and sera showed a significant decrease as compared to naïve rats in all groups at both weeks 10 and 16. Compared to DEN alone, a significant decrease was noted in the RQ of RAB11A mRNA in DEN/2-AAF 200 and 300 in sera and tissues at weeks 10 and 16 as compared to the significant change in DEN/2-AAF 100 in tissue at week 10. Moreover, only a significant decrease was detected in DEN/2-AAF 300 as compared to DEN/2-AAF 100 in serum at week 10 (Figure 5A).
Figure 5.
Effect of diethylnitrosamine and 2-acetylaminofluorene at different doses. A: Relative quantification (RQ) of RAB11A mRNA; B: RQ of BAX mRNA; C: RQ of p53 mRNA; D: RQ of Cyclin E mRNA in the liver and serum in rats. Values are mean ± SE; number of animals = 6 rats/each group. aP < 0.05 compared to the naïve group; bP < 0.05 compared to the diethylnitrosamine (DEN) group at week 10 group; cP < 0.05 compared to the DEN at week 16 group; dP < 0.05 when DEN 200 + acetylaminofluorene (AAF) is compared to the DEN 100 + AAF; eP < 0.05 when DEN 300 + AAF is compared to the DEN 100 + AAF; fP < 0.05 when DEN 300 + AAF is compared to the DEN 200 + AAF. One-way ANOVA followed by Tukey's multiple comparison test. DEN: Diethylnitrosamine; 2-AAF: 2-Acetylaminofluorene.
Compared to the naïve group, rats that received DEN solely or when combined to 2-AAF for 10 wk or 16 wk showed a significant decrease in the level of BAX mRNA in both liver tissues and sera. 2-AAF addition to DEN significantly decreased the expression of BAX mRNA as compared to DEN alone, except for 2-AAF at a dose of 100mg, in the serum at week 10. Only DEN/2-AAF 300 showed a significant decrease as compared to DEN/2-AAF 100 at week 10 in the liver tissues. The serum BAX mRNA level exhibited insignificant differences among the three DEN/2-AAF groups at both weeks 10 and 16 (Figure 5B).
Furthermore, compared to the naïve group, all groups that received DEN alone or combined with 2-AAF, a significant decrease was detected in the rat liver tissue and sera p53 mRNA. All 2-AAF groups showed a significant decrease over DEN alone except for 2-AAF 100 in the liver tissues at week 10. However, insignificant differences were noted among the three groups DEN/2-AAF 100, 200 and 300 at both weeks 10 and 16 in both liver tissues and sera (Figure 5C).
The Cyclin E mRNA in the rat liver tissues showed a significant increase between DEN/2-AAF 200 and 300 as compared to DEN/2-AAF 100 at week 10. In addition, a significant increase was noted between DEN/2-AAF 300 and DEN/2-AAF100, 200. Furthermore, rats that received DEN either alone or combined with 2-AAF showed a significant increase in the serum Cyclin E mRNA level as compared to the naïve group. All rats that received 2-AAF exhibited a significant increase in Cyclin E mRNA over DEN alone, except for 2-AAF 100, in the rat sera at week 10. Also, a significant increase was observed in DEN/2-AAF 200 and 300 over DEN/2-AAF 100 in the liver tissues at week 10. In addition, a significant increase occurred in 2-AAF 300 over 2-AAF 100 and 200 in the tissues at week 16. A significant increase was noted in 2-AAF 300 over both 2-AAF 100 and 200 at week 10 and in 2-AAF 300 over 2-AAF 100 at week 16 in rat sera (Figure 5D).
Finally, 2-AAF administration resulted in a significant increase in the level of Cyclin E mRNA with a concomitant decrease in RAB11A, p53, and BAX mRNA expression in the liver tissues and sera as compared to DEN alone. Also, significant differences were reported for 2-AAF 300 as compared to the other 2 doses, especially in the level of Cyclin E mRNA.
Effect of DEN/2-AAF on the expression of lncRNA-RP11-513I15.6, miR-1262, and miR-1298 among the rat groups
The levels of lncRNA-RP11-513I15.6, miRNA-1262, and miR-1298 were assessed in the liver tissues and sera of all groups at the end of weeks 10 and 16. One-way ANOVA and Tukey’s multiple comparison test showed significant differential expression in RQ among the studied groups.
Compared to the naïve group, the RQ of lncRNA-RP11-513I15.6 in rat liver tissues and sera in DEN and DEN/2-AAF groups showed a significant decrease at both weeks 10 and 16. A significant decrease was noted in the 2-AAF groups as compared to DEN alone, except for 2-AAF 100 mg, in the liver in week 10 and 2-AAF 100 mg in the sera at week 16. At week 10, a significant difference was observed between DEN/2-AAF 200 and DEN/2-AAF 300 than DEN/2-AAF100 mg in liver tissues. At week 16, a significant difference was noted in DEN/2-AAF 300 over DEN/2-AAF 100 mg in liver tissues, while the differences between the three groups either on week 10 or 16 were insignificant (Table 3).
Table 3.
Effect of diethylnitrosamine and 2-acetylaminofluorene on relative quantification of lncRNA-RP11-513I15.6 (long-noncoding RNA), relative quantification of miR-1262 and relative quantification of miR-1298 (microRNA)
|
RQ of lncRNA-RP11-513I15.6
|
RQ of miR-1262
|
RQ of miR-1298
|
||||
|
Liver
|
Serum
|
Liver
|
Serum
|
Liver
|
Serum
|
|
| Naïve | 2.33 ± 0.31 | 1.86 ± 0.41 | 0.38 ± 0.09 | 0.26 ± 0.34 | 0.77 ± 0.26 | 0.1 ± 0.04 |
| Week 10 | ||||||
| DEN | 1.31 ± 0.36d | 0.99 ± 0.15d | 1.63 ± 0.28d | 1.40 ± 0.13d | 1.55 ± 0.37d | 1.08 ± 0.11d |
| DEN + 100 AAF | 1.03 ± 0.2d | 0.63 ± 0.13d,e | 2.26 ± 0.54d | 2.23 ± 0.19d | 1.85 ± 0.12d | 1.48 ± 0.56d |
| DEN + 200 AAF | 0.58 ± 0.12a,d,e | 0.44 ± 0.05d,e | 2.52 ± 0.44d,e | 2.81 ± 0.49d,e | 2.46 ± 0.37d,e | 1.82 ± 0.47b,c,d,e |
| DEN + 300 AAF | 0.47 ± 0.037b,d,e | 0.39 ± 0.005d,e | 3.9 ± 0.36d,e | 3.59 ± 1.10b,d,e | 2.88 ± 0.11d,e | 3.30 ± 0.18d,e |
| Week 16 | ||||||
| DEN | 0.76 ± 0.1d | 0.52 ± 0.13d | 3.12 ± 0.62d | 2.15 ± 0.08d | 2.52 ± 0.56d | 2.23 ± 0.26d |
| DEN + 100 AAF | 0.46 ± 0.04d,f | 0.22 ± 0.04d | 4.48 ± 0.63d,f | 4.08 ± 0.32d | 3.92 ± 0.61d,f | 4.56 ± 0.61d,f |
| DEN + 200 AAF | 0.26 ± 0.07d,f | 0.14 ± 0.07d,f | 5.71 ± 0.76a,d,f | 7.38 ± 2.24d,f | 4.58 ± 0.56d,f | 5.78 ± 1.72d,f |
| DEN + 300 AAF | 0.14 ± 0.04b,d,f | 0.12 ± 0.06d,f | 6.45 ± 1.04b,d,f | 9.78 ± 4.32b,d,f | 5.89 ± 1.27d,f | 7.38 ± 2.05b,d,f |
Values are mean ± SD; number of animals = 6 rats/each group.
P < 0.05 when DEN + 200 acetylaminofluorene (AAF) is compared to the DEN + 100 AAF.
P < 0.05 when DEN + 300 AAF is compared to the DEN + 100 AAF.
P < 0.05 when DEN + 300 AAF is compared to the DEN + 200 AAF.
P < 0.05 compared to the naïve group.
P < 0.05 compared to the diethylnitrosamine (DEN) group at week 10 group.
P < 0.05 compared to the DEN at week 16 group. One-way ANOVA followed by Tukey's multiple comparison test. DEN: Diethylnitrosamine; 2-AAF: 2-Acetylaminofluorene; RQ: Relative quantification; lncRNA: Long-noncoding RNA; miR: MicroRNA.
miR-1262 exhibited a significant increase in the rats who received either DEN alone or in combination with 2-AFF as compared to the naïve group. Compared to DEN alone, all 2-AAF groups showed a significant increase except for 2-AAF 100 at week 10 in both liver tissues and sera. At week 10, 2-AAF 300 mg showed a significant difference over 2-AAF 100 mg and 200 mg in liver tissues. Moreover, at week 16, a considerable difference was observed between 2-AAF 200 mg and 300 mg over DEN/2-AAF 100 mg. At the serum level, significant differences were detected in DEN/2-AAF 300 mg over DEN/2-AAF 100 mg in both weeks 10 or 16 (Table 3).
Compared to the naïve group, all groups that received DEN or DEN in combination with 2-AAF showed a remarkable increase in the level of miR-1298. Compared to DEN alone, all groups that received 2-AFF showed a significant increase in the level of miR-1298, except for 2-AFF 100 mg, at week 10 in both liver tissues and sera. At week 10 in liver tissues, DEN/2-AAF 200 mg and 300 mg showed a significant increase over DEN/2-AAF 100 mg, while at week 16, a significant difference was detected in DEN/2-AAF 300 mg over both DEN/2-AAF 100 mg and 200 mg. At week 10, significant differences were noted in DEN/2-AAF 300 over DEN/2-AAF 100 mg and 200 mg in the sera, and at week 16, a significant increase was observed in DEN/2-AAF 300 mg over DEN/2-AAF 100 mg (Table 3).
Finally, 2-AAF administration exhibited a significant increase in miR-1298 and miR-1262 with a concomitant decrease in lncRNA-RP11-513I15.6 expression in the liver tissues and sera over DEN alone; also, significant differences were observed in 2-AFF 300 mg over the other two doses.
DISCUSSION
The nodules and cancer progression has been analyzed using animal models of carcinogenesis[16]. The present study aimed to develop a model of chemically-induced pre precancerous nodules in rat liver using DEN + 2-AAF and explore the putative molecular mechanism at the genetic and epigenetic levels. The conformation of premalignant epithelial tissues was disrupted by pre- and neoplastic liver nodules in experimental animals before the onset of cancer[17]. DEN is used to induce precancerous and cancerous lesions. It is metabolically activated by the liver cytochrome cytochrome P450 (CYP450) system, followed by induced DNA damage and oxidative stress in hepatocytes during cancer initiation[18]. The drawback of this model is the duration required for appropriate tumor development[19]. The initiated cells can be stimulated to proliferate and form hepatocyte foci and nodules by the administration of promotor agent, such as 2-AAF that causes toxicity, cell death, and carcinogenesis[20]. Carcinogens exert their carcinogenicity through either epigenetic effects without direct interaction with DNA or genotoxic effects[21].
GST-P immunohistochemistry served as an optimal marker of hepatic pre precancerous in rats[22]. In addition, PCNA is an essential cell cycle regulator; its expression serves as a tool for studying cell proliferation and identifying the replicating cells[23]. The nuclei of hepatocytes with positive PCNA immunostaining indicate hepatic regeneration. Also, a large number of cells circulating in GST-P-positive areas were observed. Furthermore, liver regeneration induced by massive hepatic necrosis was associated with the proliferation of hepatocytes.
Accumulating evidence suggested that oncogenic transformation is associated with resistance or impeded apoptotic pathway. The cancer therapy targets such autophagic imbalance[24]. RAB proteins are members of the Ras superfamily consisting of small monomeric GTPases that regulate the intracellular trafficking of several cell types. RAB11 GTPases are involved in the recycling of endosomes as well as controlling trafficking and autophagy process[25]. Previous studies demonstrated a significant role of RAB11A in pancreatic cancer[26] and non-small cell lung cancer[27].
A majority of the tumors present defects in the cell cycle, especially the loss of tumor suppressor p53, which prevents cell proliferation in response to DNA damage or dysregulation of oncogenes, inducing apoptosis or cellular senescence. p53 heterozygous mutant is susceptible to the occurrence of HCC[28,29]. Cyclin was overexpressed in many human cancers, including ovarian and breast cancers. AKT acts as a cytoplasmic central regulator of cell cycle signaling (Cyclin D1 and E) and cell survival (Mdm2/p53)[30,31]. Cyclin E1 is a regulatory subunit of Cyclin-dependent kinase 2 (CDK2). Cyclin E1 is upregulated in human HCCs and associated with poor prognosis[32,33]. Notably, the dysfunction of apoptosis with dysregulation of BCL-2 and BAX has been reported in many cancers, including bladder cancer[34]. BAX is a central regulator of cell death, leading to mitochondrial dysfunction. Also, it is one of the proapoptotic Bcl-2 family proteins that regulate apoptosis in normal and cancer cells[35].
Interestingly, previous studies reported the role of tumor suppressor miR-1262 in cancers. The expression of miR-1262 was dysregulated in the lung[36] and colon cancers[37]. On the other hand, hsa-miR-1298 is a microRNA gene, correlated to undefined RNA class and localized on the X chromosome (Xq23), (114715233-114715344 bp), 112 bases in length. Calvisi et al[6] demonstrated the secretion of circulating miR-21, miR-221a, miR-519d and miR-1228 in HCC patients. The high mobility group “A” family consisted of lncRNA RP11-513I15.6, which encoded the small nuclear proteins. Moreover, it play a significant role as an oncogene and is frequently overexpressed in different malignancies, such as HCC out[38], breast cancer[39], and ovarian cancer[40].
CONCLUSION
Administration of DEN to rats produced changes in hepatocytes with increased GST-P and PCNA expression and development of precancerous hepatic foci. The transformed cells proliferated when challenged with another carcinogen (2-AAF) as a promoter. These changes increased with the elevated dose of 2-AFF and duration of the experiment. DEN and 2-AAF affected the mRNA-biomarkers, including RAB11A, BAX, p53, and Cyclin E. Thus, the oncogenic properties of DEN and 2-AAF were observed in induced HCC model, which might be attributed to the suppression of p53, autophagy, and apoptosis along with the activation of the cell cycle. Moreover, it significantly increased the level of miR-1262 and miR-1298 with a concomitant decrease in the expression of lncRNA-RP11-513I15.6. This phenomenon led to the hypothesis that lncRNA-RP11-513I15.6 is a part of competing endogenous RNA, decreasing the level of miR-1262 and miR-1298, which, in turn, regulates the selected target mRNAs.
ARTICLE HIGHLIGHTS
Research background
2-Acetylaminofluorene (2-AAF) dose dependently promoted hepatic precancerous lesion. Over diethylnitrosamine (DEN), 2-AAF decreased autophagy. Over DEN, 2-AAF decreased apoptosis and tumor suppression gene. Over DEN, 2-AAF increased hepatic cell proliferation. 2-AAF epigenetically regulated long-noncoding RNA (lncRNA) RP11-513I15.6/miRNA-1262/miR-1298 (microRNA = miRNA = miR).
Research motivation
Urgent need for hepatocellular carcinoma (HCC) rat model for preclinical trials.
Research objectives
The present study aimed to develop a model of chemically-induced pre precancerous nodules in rat liver using DEN + 2-AAF and explore the putative molecular mechanism at the genetic and epigenetic levels.
Research methods
Bioinformatics-based selection of molecular parameters to investigate the oncogenic mechanism of the chemicals used in the HCC model followed by induction of animal model by. intraperitoneal injection of DEN for three weeks consecutively, followed by one intraperitoneal injection of 2-AAF at three different doses (100, 200 and 300 mg/kg. Rats were sacrificed after 10 wk and 16 wk. Liver functions, level of alpha-fetoprotein, glutathione S-transferase-P and proliferating cell nuclear antigen staining of liver tissues were performed. The mRNA level of RAB11A, BAX, p53, and Cyclin E and epigenetic regulation by lncRNA RP11-513I15.6, miR-1262, and miR-1298 were assessed in the sera and liver tissues of the rats.
Research results
2-AAF administration significantly increased the percent area of the precancerous foci and cell proliferation along with a significant decrease in RAB11A, BAX, and p53 mRNA, and the increase in Cyclin E mRNA was associated with a marked decrease in lncRNA RP11-513I15.6 expression with a significant increase in both miR-1262 and miR-1298.
Research conclusions
Administration of DEN to rats produced changes in hepatocytes with increased glutathione S-transferase-P and proliferating cell nuclear antigen expression and development of precancerous hepatic foci. The transformed cells proliferated when challenged with another carcinogen (2-AAF) as a promoter. These changes increased with the elevated dose of 2-AFF and duration of the experiment. DEN and 2-AAF affected the mRNA-biomarkers, including RAB11A, BAX, p53, and Cyclin E. Thus, the oncogenic properties of DEN and 2-AAF were observed in induced HCC model, which might be attributed to the suppression of p53, autophagy, and apoptosis along with the activation of the cell cycle. Moreover, it significantly increased the level of miR-1262 and miR-1298 with a concomitant decrease in the expression of lncRNA-RP11-513I15.6. This phenomenon led to the hypothesis that lncRNA-RP11-513I15.6 is a part of competing endogenous RNA, decreasing the level of miR-1262 and miR-1298, which, in turn, regulates the selected target mRNAs.
Research perspectives
More in vitro functional studies are urgently need to explore the competing endogenur role of lncRNA in HCC pathogenesis.
Footnotes
Institutional animal care and use committee statement: All animal procedures were carried out in accordance with the National Institute of Health guide for the care and use of laboratory animals (NIH Publication No. 85-23, revised 1996) and were approved by the Institutional Animal Ethics Committee for Ain Shams University, Faculty of Medicine (approval No. 17585).
Conflict-of-interest statement: The authors declare no competing interest.
ARRIVE guidelines statement: The authors have read the ARRIVE Guidelines, and the manuscript was prepared and revised according to the ARRIVE Guidelines.
Manuscript source: Invited manuscript
Peer-review started: November 23, 2020
First decision: December 12, 2020
Article in press: March 2, 2021
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Egypt
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Koustas E S-Editor: Gao CC L-Editor: A P-Editor: Wang LL
Contributor Information
Amany Helmy Hasanin, Clinical Pharmacology Department, Faculty of Medicine, Ain Shams University, Cairo 11318, Egypt.
Eman K Habib, Anatomy and Embryology Department, Faculty of Medicine, Ain Shams University, Cairo 11318, Egypt.
Nesreen El Gayar, Clinical Pharmacology Department, Faculty of Medicine, Ain Shams University, Cairo 11318, Egypt.
Marwa Matboli, Medical Biochemistry and Molecular Biology Department, Faculty of Medicine, Ain Shams University, Cairo 11381, Egypt. marwasayed472@yahoo.com.
Data sharing statement
No additional data are available.
References
- 1.Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394–424. doi: 10.3322/caac.21492. [DOI] [PubMed] [Google Scholar]
- 2.Matboli M, Shafei AE, Ali MA, Ashry AM, Kamal KM, Agag MA, Reda I, Tash EF, Ali M. circRNAs (hsa_circ_00156, hsa_circ _000224, and hsa_circ _000520) are novel potential biomarkers in hepatocellular carcinoma. J Cell Biochem. 2018 doi: 10.1002/jcb.28045. [DOI] [PubMed] [Google Scholar]
- 3.Teng YC, Shen ZQ, Kao CH, Tsai TF. Hepatocellular carcinoma mouse models: Hepatitis B virus-associated hepatocarcinogenesis and haploinsufficient tumor suppressor genes. World J Gastroenterol. 2016;22:300–325. doi: 10.3748/wjg.v22.i1.300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stahl S, Ittrich C, Marx-Stoelting P, Köhle C, Altug-Teber O, Riess O, Bonin M, Jobst J, Kaiser S, Buchmann A, Schwarz M. Genotype-phenotype relationships in hepatocellular tumors from mice and man. Hepatology. 2005;42:353–361. doi: 10.1002/hep.20768. [DOI] [PubMed] [Google Scholar]
- 5.Newell P, Villanueva A, Friedman SL, Koike K, Llovet JM. Experimental models of hepatocellular carcinoma. J Hepatol. 2008;48:858–879. doi: 10.1016/j.jhep.2008.01.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Calvisi DF, Conner EA, Ladu S, Lemmer ER, Factor VM, Thorgeirsson SS. Activation of the canonical Wnt/beta-catenin pathway confers growth advantages in c-Myc/E2F1 transgenic mouse model of liver cancer. J Hepatol. 2005;42:842–849. doi: 10.1016/j.jhep.2005.01.029. [DOI] [PubMed] [Google Scholar]
- 7.Xue W, Zender L, Miething C, Dickins RA, Hernando E, Krizhanovsky V, Cordon-Cardo C, Lowe SW. Senescence and tumour clearance is triggered by p53 restoration in murine liver carcinomas. Nature. 2007;445:656–660. doi: 10.1038/nature05529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Omerovic J, Laude AJ, Prior IA. Ras proteins: paradigms for compartmentalised and isoform-specific signalling. Cell Mol Life Sci. 2007;64:2575–2589. doi: 10.1007/s00018-007-7133-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Heindryckx F, Colle I, Van Vlierberghe H. Experimental mouse models for hepatocellular carcinoma research. Int J Exp Pathol. 2009;90:367–386. doi: 10.1111/j.1365-2613.2009.00656.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Espandiari P, Robertson LW, Srinivasan C, Glauert HP. Comparison of different initiation protocols in the resistant hepatocyte model. Toxicology. 2005;206:373–381. doi: 10.1016/j.tox.2004.07.014. [DOI] [PubMed] [Google Scholar]
- 11.Bagnyukova TV, Tryndyak VP, Montgomery B, Churchwell MI, Karpf AR, James SR, Muskhelishvili L, Beland FA, Pogribny IP. Genetic and epigenetic changes in rat preneoplastic liver tissue induced by 2-acetylaminofluorene. Carcinogenesis. 2008;29:638–646. doi: 10.1093/carcin/bgm303. [DOI] [PubMed] [Google Scholar]
- 12.Klöhn PC, Bitsch A, Neumann HG. Mitochondrial permeability transition is altered in early stages of carcinogenesis of 2-acetylaminofluorene. Carcinogenesis. 1998;19:1185–1190. doi: 10.1093/carcin/19.7.1185. [DOI] [PubMed] [Google Scholar]
- 13.Klöhn PC, Soriano ME, Irwin W, Penzo D, Scorrano L, Bitsch A, Neumann HG, Bernardi P. Early resistance to cell death and to onset of the mitochondrial permeability transition during hepatocarcinogenesis with 2-acetylaminofluorene. Proc Natl Acad Sci USA. 2003;100:10014–10019. doi: 10.1073/pnas.1633614100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Malik S, Bhatnagar S, Chaudhary N, Katare DP, Jain SK. DEN+2-AAF-induced multistep hepatotumorigenesis in Wistar rats: supportive evidence and insights. Protoplasma. 2013;250:175–183. doi: 10.1007/s00709-012-0392-8. [DOI] [PubMed] [Google Scholar]
- 15.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 16.Xin B, Cui Y, Wang Y, Wang L, Yin J, Zhang L, Pang H, Zhang H, Wang RA. Combined use of alcohol in conventional chemical-induced mouse liver cancer model improves the simulation of clinical characteristics of human hepatocellular carcinoma. Oncol Lett. 2017;14:4722–4728. doi: 10.3892/ol.2017.6800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Obeid M, Khabbaz R, Garcia K, Schachtschneider K, Gaba R. Translational Animal Models for Liver Cancer. Am J Interv Radiol. 2018;2:1–7. [Google Scholar]
- 18.Singh D, Singh M, Yadav E, Falls N, Komal U, Singh Dangi D, Kumare V, Verma A. Amelioration of diethylnitrosamine (DEN)-induced hepatocellular carcinogenesis in animal models via knockdown oxidative stress and proinflammatory markers by Madhuca longifolia embedded silver nanoparticles. RSC Adv. 2018;8:6940. doi: 10.1039/c7ra12775h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gade TP, Hunt SJ, Harrison N, Nadolski GJ, Weber C, Pickup S, Furth EE, Schnall MD, Soulen MC, Celeste Simon M. Segmental Transarterial Embolization in a Translational Rat Model of Hepatocellular Carcinoma. J Vasc Interv Radiol. 2015;26:1229–1237. doi: 10.1016/j.jvir.2015.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wu K, Ding J, Chen C, Sun W, Ning BF, Wen W, Huang L, Han T, Yang W, Wang C, Li Z, Wu MC, Feng GS, Xie WF, Wang HY. Hepatic transforming growth factor beta gives rise to tumor-initiating cells and promotes liver cancer development. Hepatology. 2012;56:2255–2267. doi: 10.1002/hep.26007. [DOI] [PubMed] [Google Scholar]
- 21.Barnes JL, Zubair M, John K, Poirier MC, Martin FL. Carcinogens and DNA damage. Biochem Soc Trans. 2018;46:1213–1224. doi: 10.1042/BST20180519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Muramatsu M, Sakai M. Mechanisms of a tumor marker, glutathione transferase P, expression during hepatocarcinogenesis of the rat. Proc Jpn Acad Ser B Phys Biol Sci. 2006;82:339–352. doi: 10.2183/pjab.82.339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Strzalka W, Ziemienowicz A. Proliferating cell nuclear antigen (PCNA): a key factor in DNA replication and cell cycle regulation. Ann Bot. 2011;107:1127–1140. doi: 10.1093/aob/mcq243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kowalik MA, Perra A, Ledda-Columbano GM, Ippolito G, Piacentini M, Columbano A, Falasca L. Induction of autophagy promotes the growth of early preneoplastic rat liver nodules. Oncotarget. 2016;7:5788–5799. doi: 10.18632/oncotarget.6810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lock JG, Stow JL. Rab11 in recycling endosomes regulates the sorting and basolateral transport of E-cadherin. Mol Biol Cell. 2005;16:1744–1755. doi: 10.1091/mbc.E04-10-0867. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yu L, Li X, Li H, Chen H, Liu H. Rab11a sustains GSK3β/Wnt/β-catenin signaling to enhance cancer progression in pancreatic cancer. Tumour Biol. 2016;37:13821–13829. doi: 10.1007/s13277-016-5172-1. [DOI] [PubMed] [Google Scholar]
- 27.Dong Q, Fu L, Zhao Y, Du Y, Li Q, Qiu X, Wang E. Rab11a promotes proliferation and invasion through regulation of YAP in non-small cell lung cancer. Oncotarget. 2017;8:27800–27811. doi: 10.18632/oncotarget.15359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Farazi PA, Glickman J, Horner J, Depinho RA. Cooperative interactions of p53 mutation, telomere dysfunction, and chronic liver damage in hepatocellular carcinoma progression. Cancer Res. 2006;66:4766–4773. doi: 10.1158/0008-5472.CAN-05-4608. [DOI] [PubMed] [Google Scholar]
- 29.Lewis BC, Klimstra DS, Socci ND, Xu S, Koutcher JA, Varmus HE. The absence of p53 promotes metastasis in a novel somatic mouse model for hepatocellular carcinoma. Mol Cell Biol. 2005;25:1228–1237. doi: 10.1128/MCB.25.4.1228-1237.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Testa JR, Bellacosa A. AKT plays a central role in tumorigenesis. Proc Natl Acad Sci USA. 2001;98:10983–10985. doi: 10.1073/pnas.211430998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Stanger BZ, Stiles B, Lauwers GY, Bardeesy N, Mendoza M, Wang Y, Greenwood A, Cheng KH, McLaughlin M, Brown D, Depinho RA, Wu H, Melton DA, Dor Y. Pten constrains centroacinar cell expansion and malignant transformation in the pancreas. Cancer Cell. 2005;8:185–195. doi: 10.1016/j.ccr.2005.07.015. [DOI] [PubMed] [Google Scholar]
- 32.Sonntag R, Giebeler N, Nevzorova YA, Bangen JM, Fahrenkamp D, Lambertz D, Haas U, Hu W, Gassler N, Cubero FJ, Müller-Newen G, Abdallah AT, Weiskirchen R, Ticconi F, Costa IG, Barbacid M, Trautwein C, Liedtke C. Cyclin E1 and cyclin-dependent kinase 2 are critical for initiation, but not for progression of hepatocellular carcinoma. Proc Natl Acad Sci USA. 2018;115:9282–9287. doi: 10.1073/pnas.1807155115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Geng Y, Michowski W, Chick JM, Wang YE, Jecrois ME, Sweeney KE, Liu L, Han RC, Ke N, Zagozdzon A, Sicinska E, Bronson RT, Gygi SP, Sicinski P. Kinase-independent function of E-type cyclins in liver cancer. Proc Natl Acad Sci USA. 2018;115:1015–1020. doi: 10.1073/pnas.1711477115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ajili F, Kaabi B, Darouiche A, Tounsi H, Kourda N, Chebil M, Manai M, Boubaker S. Prognostic value of Bcl-2 and Bax tumor cell expression in patients with non muscle-invasive bladder cancer receiving bacillus Calmette-Guerin immunotherapy. Ultrastruct Pathol. 2012;36:31–39. doi: 10.3109/01913123.2011.620221. [DOI] [PubMed] [Google Scholar]
- 35.Cheng EH, Wei MC, Weiler S, Flavell RA, Mak TW, Lindsten T, Korsmeyer SJ. BCL-2, BCL-X(L) sequester BH3 domain-only molecules preventing BAX- and BAK-mediated mitochondrial apoptosis. Mol Cell. 2001;8:705–711. doi: 10.1016/s1097-2765(01)00320-3. [DOI] [PubMed] [Google Scholar]
- 36.Xie K, Chen M, Zhu M, Wang C, Qin N, Liang C, Song C, Dai J, Jin G, Shen H, Lin D, Ma H, Hu Z. A polymorphism in miR-1262 regulatory region confers the risk of lung cancer in Chinese population. Int J Cancer. 2017;141:958–966. doi: 10.1002/ijc.30788. [DOI] [PubMed] [Google Scholar]
- 37.Bobowicz M, Skrzypski M, Czapiewski P, Marczyk M, Maciejewska A, Jankowski M, Szulgo-Paczkowska A, Zegarski W, Pawłowski R, Polańska J, Biernat W, Jaśkiewicz J, Jassem J. Prognostic value of 5-microRNA based signature in T2-T3N0 colon cancer. Clin Exp Metastasis. 2016;33:765–773. doi: 10.1007/s10585-016-9810-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen M, Liu Y, Varley P, Chang Y, He XX, Huang H, Tang D, Lotze MT, Lin J, Tsung A. High-Mobility Group Box 1 Promotes Hepatocellular Carcinoma Progression through miR-21-Mediated Matrix Metalloproteinase Activity. Cancer Res. 2015;75:1645–1656. doi: 10.1158/0008-5472.CAN-14-2147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Duda P, Akula SM, Abrams SL, Steelman LS, Martelli AM, Cocco L, Ratti S, Candido S, Libra M, Montalto G, Cervello M, Gizak A, Rakus D, McCubrey JA. Targeting GSK3 and Associated Signaling Pathways Involved in Cancer. Cells. 2020;9 doi: 10.3390/cells9051110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wu J, Wei JJ. HMGA2 and high-grade serous ovarian carcinoma. J Mol Med (Berl) 2013;91:1155–1165. doi: 10.1007/s00109-013-1055-8. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No additional data are available.