Abstract
Neurological symptoms occur in approximately one-third of hospitalized patients with coronavirus disease 2019 (COVID-19). Among these symptoms, hypoxic encephalopathy develops in one-fifth of severe cases, while ischemic strokes due to thrombotic complications are common in one-third of COVID-19 intensive care patients. Brain involvement of severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) is eventuated by several routes, including hematogenous spread, transsynaptic entry through infected neurons, olfactory nerve, ocular epithelium, vascular endothelium, and impaired blood–brain barrier. Besides the high angiotensin-converting enzyme-2 (ACE2) binding affinity, and FURIN preactivation, SARS-CoV-2 maintains efficient neuronal entry while evading immune surveillance by using basigin and neuropilin-1 receptors. However, the neurological manifestations and their pathogenic mechanisms are still debated in COVID-19 patients.
Keywords: COVID-19, Blood–brain barrier, ACE2, Thromboembolic complications, IL-6, Neuropilin-1
Introduction
Although severe acute respiratory syndrome-coronavirus-2 (SARS-CoV-2) mainly targets the epithelial cells of the respiratory tract, resulting in diffuse alveolar damage, among the other tissues brain cells are also potential target of viral attack [1]. In this respect, neurological abnormalities have been described in approximately one-third of patients who required hospitalization for coronavirus disease 2019 (COVID-19) [2]. Neurological symptoms appear largely with nonspecific clinical manifestation. In SARS-CoV-2 infection, these seem to be dependent on a successive series of events such as excessive neuro-inflammation due to cytokine storm, hypoxia, blood pressure imbalance because of the angiotensin II converting enzyme receptor-2 (ACE2) involvement, and severe ischemic stroke due to diffuse intravascular coagulation [2,3]. According to epidemiological studies, acute symptomatic seizures are observed in more than one-fourth of COVID-19 patients. Hypoxic encephalopathy and ischemic strokes due to thrombotic complications indicate the frequent brain involvement in COVID-19 intensive care unit patients, by one-fifth and one-third, respectively [4, 5∗, 6∗∗]. In this context, the most serious neurological signs seen in COVID-19 patients occur with widespread microthrombi and infarction patches in the brain. These histopathological findings are explicated with the increase in the susceptibility to cerebrovascular events due to hypercoagulability [7]. Recent evidences strongly suggest that clinical findings regarding the mechanisms of neurological symptoms caused by SARS-CoV-2 invasion of the central nervous system (CNS) have not yet been clarified and systematically classified. In COVID-19 patients, the role of ACE2 for the transsynaptic spread of the virus in the CNS, the effect of the hyperimmune response on the spread across the blood–brain barrier (BBB) and the persistence of SARS-CoV-2 in the brain are still debated [8].
Potential routes of brain entry
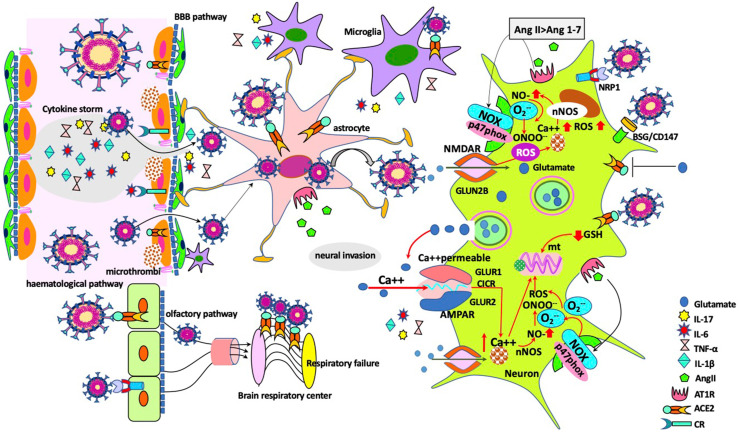
Various assumptions regarding the entry of SARS-CoV-2 into the CNS are widely discussed in recent studies. In SARS-CoV-2-infected patients, it is proposed that neuro-invasion is eventuated by several routes; hematogenous transportation of infected immune cells via the circulatory system of the brain tissue, transsynaptic entry through the infected neurons or olfactory nerve, entry from ocular epithelium, and the virus invasion from the impaired BBB [9,10] (Figure 1 ). Although ACE2 and transmembrane protease serine 2 (TMPRSS2) have been localized to epithelial cells in the nasal mucosa, whether the virus entry is limited to the olfactory epithelium or attacks the olfactory neurons is controversial [9,11]. Despite the olfactory neuron seems to be one of the major routes for the virus spreads, dissemination of the SARS-CoV-2 via hematological pathway has been reported in various studies with widely ranging frequencies between 1% and 41% [12,13]. Indeed, there have been few comprehensive studies, which identify the neuro-invasive potential of the SARS-CoV-2. In postmortem studies of COVID-19 patients, virus detection in frontal lobe neurons, presence of viral particles in brain capillary endothelial cells despite negative polymerase chain reaction testing of the cerebrospinal fluid and increase in plasma biomarkers due to astrocytic or neuronal injury have been attributed to the neural invasion. Thus, rising in glial fibrillary acidic protein (GFAp) level as a marker of astrocytic activation/injury and increasing neurofilament light chain protein (NfL) concentration as a marker of intra-axonal neuronal injury due to brain damage indicate the hematogenous spread of virus [14,15].
Figure 1.
Potential entry routes of SARS-CoV-2 to the central nervous system (CNS) and the mechanisms of neuronal damage. Taken through the olfactory pathway, virus would have access to the CNS using transneuronal/synaptic routes and reaches to brain respiratory center. In the hematogenous entry route, endothelial cells may become infected, microthrombi develop due to endothelial dysfunction. CNS infection arises with the SARS-CoV-2 entrance to the brain following blood–brain barrier disruption by cytokine storm. Astrocytes incorporate the viruses either via direct contact with infected endothelial cells, or ACE2 receptors. In response to virus infection, microglial cells trigger T cell, antigen-presenting cell activation and induce synapse loss. Following neuro-invasion and replication of SARS-CoV-2, impairment of ACE2 activity due to exploitation by SARS-CoV-2, activation of AT1R by Ang II, and glutamate mediated inhibition of ACE2 activity contribute to the development of neurotoxicity and neuroinflammation. Excessive extracellular glutamate accumulation in SARS-CoV-2 infection triggers oxidative stress and neuroinflammation via NMDA receptors, while SARS-CoV-2 infection targets glutathione (GSH) biosynthesis and makes the patients more vulnerable to the detrimental effects of virus by decreasing GSH levels (Abbreviations: ACE2: angiotensin II–converting enzyme receptor-2; AMPAR: α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor; AngII: angiotensin II; AT1R: Ang II receptor type 1; BBB: blood–brain barrier; BSG/CD147: basigin; CICR: calcium-induced calcium release; CR: cytokine receptor; GSH: reduced glutathione; mt: mitochondria; NMDAR: N-methyl-d-aspartate receptor; nNOS: neuronal nitric oxide synthase; NOX: nicotinamide adenine dinucleotide phosphate oxidase; NRP1: neuropilin 1; ROS: reactive oxygen species; SARS-CoV-2: acute respiratory syndrome-coronavirus-2).
Furthermore, interleukin (IL)-6, IL-1β, tumor necrosis factor (TNF), and IL-17, the cytokines, which are elevated during cytokine storm associated with SARS-CoV-2 infection, lead to the disruption of the BBB and facilitate the entry of the virus to brain. Whereupon SARS-CoV-2 can infect neurons and glial cells through ACE2 receptors and launches a neuroinflammatory response with reactive astrogliosis, and microglial activation [16, 17∗∗, 18]. Nevertheless, it is claimed that certain neurological complications of COVID-19 are due to increased penetration of proinflammatory cytokines from impaired BBB rather than viral entry. Cytokine release–driven neuroinflammatory response that can activate microglial cells in the CNS cause to the release of astroglial marker, S100B protein, which reflects the increase in the BBB permeability [19,20]. As in other parts of the body, the ACE2 receptor is expressed in the brain by endothelial, neuronal, and glial cells. After the spreading of virus to CNS, it contacts with ACE2 on neurons, glia, and vascular endothelium [21,22]. However, it has been suggested that receptors such as neuropilin-1 (NRP1) play a role in viral entry in neurons lacking ACE2 expression [23].
Functions of receptors in brain invasion of SARS-CoV-2
As a rule, SARS-CoV-2 uses ACE2 as the principal binding receptor and is required for proteolytic processing of the spike (S) protein by TMPRSS2 for cell entry [24]. Although data related to ACE2, TMPRSS2, and furin distributions in the olfactory pathway are insufficient, these cells seem to express ACE2, but not TMPRSS2 or furin. But deterioration of the sense of smelling in COVID-19 patients is simultaneous with the emergence of neurological symptoms [25,26]. Actually, the ACE2 is intensely expressed in the pons and medulla oblongata of the human brainstem, which consists of the medullary respiratory centers of the brain. This finding partly elucidates the mechanism of severe respiratory distress in many COVID-19 patients [27].
Considering the above data, the expression of ACE2 receptor in the brain indicates that SARS-CoV-2 has a neuro-invasive ability [28]. Following SARS-CoV-2 entry to the CNS, ACE2 receptors mediate the development of neurotoxicity, neuroinflammation, and neurodegeneration via ensuring viral entrance and replication [29]. SARS-CoV-2 S protein binds to its receptor ACE2 via its receptor-binding domain (RBD) and is proteolytically activated by human proteases. SARS-CoV-2 is preactivated by proprotein convertase FURIN, diminishing its reliance on target cell proteases for entry. But a certain amount of TMPRSS2 is still needed. The high ACE2 binding affinity of the RBD, FURIN preactivation of the spike, and hidden RBD in the spike potentially allow SARS-CoV-2 to maintain efficient neuronal entry while evading immune surveillance [30]. This means that TMPRSS2 is simultaneously activated to allow the coronavirus S protein to enter the cell [24]. It should be added to the activation of TMPRSS2, that the FURIN/PCSK3 cleavage of the S protein facilitates the interaction of SARS-CoV-2 with the ACE2 receptor and viral invasion [31]. Another route is S protein, which also binds to alternative or the additive molecule basigin (CD147/BSG) (Figure 1). This molecule is a novel receptor glycoprotein of the immunoglobulin super family and serves as the mediating factor of the viral invasion [32]. SARS-CoV-2 can also use basigin [33] and NRP1 as well as ACE2 during entry into neurons [23]. Proteases such as TMPRSS11 A/B, cathepsin B and L and FURIN allow viral cell entry and replication via NRP1 docking receptors [30].
Unexpectedly, ACE2 expression significantly declines in the brain of individuals with Alzheimer's disease (AD). This deficit is dependent on the increased amyloid-beta (Aβ) peptide load and tau pathology and hyperactivity of the classical axis of the renin–angiotensin system [34,35]. Apart from AD, the presence of ACE2 has not been demonstrated in many neurons [36], despite increasing reports of neurological symptoms being common in COVID-19 patients [2]. However, apolipoprotein E (ApoE) is a potential link between AD and COVID-19. ApoE, as a Aβ chaperone, confers a 14-fold increase in susceptibility to AD, while constitutes a risk factor for severe SARS-CoV-2 infection and provides a twofold increase in neural entry of virus [37,38]. Paradoxically, having a decrease in ACE2 function in aged patients, coupled with the age-associated reduction in mitochondrial functions make the host more vulnerable to SARS-CoV-2 infection. Considering the more severe course of COVID-19 in elderly patient populations, these observations suggest that a new explanation is needed for the viral entry mechanism [39,40]. These findings support the claim that ACE2 is not the sole gateway for entry of SARS-CoV-2. In this context, it is suggested that the SARS-CoV-2 enters the neuron by binding with its S protein to the NRP1 receptor. Thus, NRP1, FURIN, and TMPRSS11A levels are elevated in SARS-CoV-2-infected cells of these patients. As mentioned above, the olfactory epithelial cells show high expression of NRP1 in COVID-19 patients [23,41]. In addition, vascular endothelial growth factor-A (VEGF-A) is a physiological ligand for the b1b2 pocket in NRP1 [42]. After brain invasion, primary effect of SARS-CoV-2 emerges on glial cells. Secondary effect results in neuronal damage [43].
In the brain, through the conversion of angiotensin II (Ang II) into angiotensin-(1–7) (Ang-(1–7)), ACE2 decreases oxidative stress and alleviates neuronal apoptosis, besides the maintenance of BBB function [44,45] (Figure 1). Contrarily, impairment of ACE2 activity due to exploitation by SARS-CoV-2 contributes to the development of neuroinflammation and apoptosis [46]. By activating Ang II receptor type 1 (AT1R), Ang II stimulates both internalization and ectodomain shedding of ACE2. Thus, ACE2 expression in addition to its enzymatic activity are inhibited [47,48]. Furthermore, cytotoxic levels of glutamate can significantly impair ACE2 activity in cortical neurons and induces excitotoxic damage in the nerve cells. Thereby following endothelial dysfunction, ischemic stroke, and neuronal injury develop [49].
The neurotoxicity of glutamate is launched mainly by an ample amount of Ca2+ influx emerging from overstimulation of the α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor (AMPAR) GluR1 [50]. The increase in the intracellular [Ca2+] can mainly lead to mitochondrial dysfunction and reactive oxygen species (ROS) generation [51,52]. Because SARS-CoV-2 infection targets glutathione (GSH) biosynthesis, decreased GSH, and selenoprotein levels make the patients more vulnerable to the detrimental effects of virus-induced oxidative stress and proteolysis [53]. The excitotoxicity of excessive glutamate accumulation participates in the progression of inflammatory neurodegeneration via oxidative stress and/or inducible nitric oxide synthase (iNOS)-mediated mechanisms [54,55]. N-methyl-d-aspartate (NMDA) receptor activation due to excess glutamate causes neuronal damage by increasing oxidative stress and inflammation, unlike alpha7 nicotinic acetylcholine receptor (α7 nAChR) [55,56] (Figure 1). Against excessive extracellular glutamate accumulation triggered by diverse inflammatory and oxidative processes, memantine protects cells from glutamate excitotoxicity by blocking the extrasynaptic NMDA receptors [57]. Because memantine is an antagonist of NMDA receptors, it diminishes the SARS-CoV-2-related oxidative stress and inflammation in neurons [58].
Neurological manifestations of COVID-19
In the clinical studies of the hospitalized COVID-19 patients groups consisting of 214 and 841 cases, neurologic manifestations have been observed in 36.4% and 57.4% of the cases, respectively [2,59].
It is still debated whether or not ACE2 is the main route of entry of SARS-CoV-2 into neuronal cells and what strategies might block viral infection, and whether there are humoral antibody responses against SARS-CoV-2 in the CNS of infected patients. Since in some patients with COVID-19 and neurological symptoms, there is robust antibody response to the virus within the CSF, in patients with mild COVID-19, neurological manifestation is frequently emerged as nonspecific signs like dizziness by 7% and headache by 14% [60]. It is thought that the loss of taste and smell is based on the spreading of the virus to the olfactory neurons, however, due to the lack of convincing proof, it is claimed that disruption of sensory-neural mechanisms may depend on the SARS-CoV-2 encephalitis [2,60]. Nevertheless, the prevalence of hypogeusia and hyposmia was 5.6% and 5.1%, respectively [2]. In fact, it is thought that increase in the olfactory and gustatory dysfunctions are directly correlated with the increase in levels of IL-6 in COVID-19 cases [61]. Although the recovery times in most cases are accompanied by simultaneous swab negativization, and normalization of IL-6 levels, in one-fourth of cases taste and smell dysfunctions may persist a longer period after the recovery [61,62]. Actually, these manifestations have been linked to the infection of cell receptors with the virus rather than possible neurological or ischemic damage [61]. Actually, COVID-19 cases involving the CNS resembles traditional cases of acute disseminated encephalomyelitis [63] and acute necrotizing hemorrhagic encephalopathy [64]. Recently, it has been reported that Guillain–Barré syndrome, which occurs with immune attack on peripheral nerves, has been observed in COVID-19 cases [65]. ACE2 is expressed on both surface membrane and in the cytoplasm of some neurons, astrocytes, and oligodendrocytes. Expression of ACE2 is also densely localized in the substantia nigra, ventricles, middle temporal gyrus, posterior cingulate cortex, and olfactory bulb [66]. COVID-19 patients have increased Ang II. The overexpression of ACE2 and the disturbance of balance due to increased Ang II enhances the mortality of COVID-19 patients [3]. Acute ischemic stroke (AIS) has been described in approximately 1–3% of patients with COVID-19 [2,67,68]. Intracranial hemorrhage is seen in about 0.5% of the patients with COVID-19 in large population studies [2,59]. Increase in procoagulant factors in COVID-19 patients, such as serum levels of fibrinogen, platelet, IL-6 and D-dimer, contribute thromboembolic complications and higher rate of mortality and morbidity [69]. ACE2 exploited by SARS-CoV-2, and high Ang II cause endothelial dysfunction in the cerebral arteries, by increasing blood pressure (approximately one-third of all patients) and creating the risk of cerebral hemorrhage and hemorrhagic stroke [70,71]. In these cases, downregulation of the ACE2 receptors and elevated Ang II levels are proportional with viral load and mortality rate [3,72]. AIS rate associated with an inflammatory and hypercoagulable state is observed up to 5% of patients with severe COVID-19. In these patients, the risk of death due to AIS reaches up to 38% [73]. Coagulopathy underlies the occurrence of stroke. Cytokine storm-mediated by SARS-CoV-2 can produce the hypercoagulable state, causes vascular wall damage, and precipitates microthrombosis [74]. Furthermore, the overactivation of the complement cascade, and its interactions with cytokines plays important role in cerebrovascular thrombosis. However, precise data on this subject could not be yet acquired [75,76], ACE2 receptor has an important role in developing neurologic manifestations of SARS-CoV-2 infection. Cerebral hemorrhage and stroke aggravate COVID-19 outcomes [77]. Protecting patients from AIS, which is one of the most dangerous complications of COVID-19 and causes the lethality of more than a third of severe cases, requires a complex treatment program. These include immunomodulatory drugs, anti-inflammatory drugs, and prophylactic antibodies, antiviral drugs and vaccines; however, the most suitable treatment combination is still being under investigation.
Conclusion
The large number of sensitive transmembrane receptors make most of the human cells highly susceptible to the risk of SARS-CoV-2 infection and viral attack. The serious danger of SARS-CoV-2 infection is not only due to its extremely contagious behavior but also due to its attack to many vital and critical human cell and tissue types simultaneously and is deadly. Because the neurological symptoms and their mechanisms could not be comprehensively clarified, in COVID-19 patients, the spread and persistence of the virus in the brain cells are still under debate.
Credit author statement
Ayse Basak Engin: Writing – original draft preparation, Evren Doruk Engin: Reviewing and editing Atilla Engin: Figure preparation, reviewing and editing.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
This review comes from a themed issue on Mechanistic Toxicology
Edited by Monica Neagu and Ayse Basak Engin
References
- 1.Lyons-Weiler J. Pathogenic priming likely contributes to serious and critical illness and mortality in COVID-19 via autoimmunity. J Transl Autoimmun. 2020:100051. doi: 10.1016/j.jtauto.2020.100051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Mao L., Jin H., Wang M., Hu Y., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Miao X., Li Y., Hu B. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in wuhan, China. JAMA Neurol. 2020;77:683–690. doi: 10.1001/jamaneurol.2020.1127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engin A.B., Engin E.D., Engin A. Two important controversial risk factors in SARS-CoV-2 infection: obesity and smoking. Environ Toxicol Pharmacol. 2020;78:103411. doi: 10.1016/j.etap.2020.103411. [DOI] [PMC free article] [PubMed] [Google Scholar]; SARS-CoV-2 attaches the ACE2 receptor of human host cells through its receptor binding domain in the S1 region of spike protein. The expression of ACE2 and the balance of Ang II/Ang 1–7 influence the course of the disease.
- 4.Chen T., Wu D., Chen H., Yan W., Yang D., Chen G., Ma K., Xu D., Yu H., Wang H., Wang T., Guo W., Chen J., Ding C., Zhang X., Huang J., Han M., Li S., Luo X., Zhao J., Ning Q. Clinical characteristics of 113 deceased patients with coronavirus disease 2019: retrospective study. BMJ. 2020;368:m1091. doi: 10.1136/bmj.m1091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das G., Mukherjee N., Ghosh S. Neurological insights of COVID-19 pandemic. ACS Chem Neurosci. 2020;11:1206–1209. doi: 10.1021/acschemneuro.0c00201. [DOI] [PubMed] [Google Scholar]; SARS-CoV-2 infects lung but it causes severe damage in neurons, specifically those in the medulla oblongata, which regulates breathing, lung, and heart functions. COVID-19 results in severe (encephalopathy) neurological manifestations.
- Klok F.A., Kruip M.J.H.A., van der Meer N.J.M., Arbous M.S., Gommers D.a.M.P.J., Kant K.M., Kaptein F.H.J., van Paassen J., Stals M.a.M., Huisman M.V., Endeman H. Incidence of thrombotic complications in critically ill ICU patients with COVID-19. Thromb Res. 2020;191:145–147. doi: 10.1016/j.thromres.2020.04.013. [DOI] [PMC free article] [PubMed] [Google Scholar]; The 31% incidence of thrombotic complications in intensive care unit patients with COVID-19 infections is remarkably high.
- 7.Bryce C., Grimes Z., Pujadas E., Ahuja S., Beasley M.B., Albrecht R., Hernandez T., Stock A., Zhao Z., Rasheed M.A., Chen J., Li L., Wang D., Corben A., Haines K., Westra W., Umphlett M., Gordon R.E., Reidy J., Petersen B., Salem F., Fiel M., Jamal S.M.E., Tsankova N.M., Houldsworth J., Mussa Z., Liu W.-C., Veremis B., Sordillo E., Gitman M., Nowak M., Brody R., Harpaz N., Merad M., Gnjatic S., Donnelly R., Seigler P., Keys C., Cameron J., Moultrie I., Washington K.-L., Treatman J., Sebra R., Jhang J., Firpo A., Lednicky J., Paniz-Mondolfi A., Cordon-Cardo C., Fowkes M. MedRxiv; 2020. Pathophysiology of SARS-CoV-2: targeting of endothelial cells renders a complex disease with thrombotic microangiopathy and aberrant immune response. The Mount Sinai COVID-19 autopsy experience. 2020.05.18.20099960. [DOI] [Google Scholar]
- 8.Pennisi M., Lanza G., Falzone L., Fisicaro F., Ferri R., Bella R. SARS-CoV-2 and the nervous system: from clinical features to molecular mechanisms. Int J Mol Sci. 2020;21 doi: 10.3390/ijms21155475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nampoothiri S., Sauve F., Ternier G., Fernandois D., Coelho C., Imbernon M., Deligia E., Perbet R., Florent V., Baroncini M., Pasquier F., Trottein F., Maurage C.-A., Mattot V., Giacobini P., Rasika S., Prevot V. BioRxiv; 2020. The hypothalamus as a hub for putative SARS-CoV-2 brain infection. 2020.06.08.139329. [DOI] [Google Scholar]; Regardless of the entry route to the brain, the virus can infect a number of different regions through the same transsynaptic mechanism to be used in the olfactory route or by diffusion through the cerebrospinal fluid (CSF). The higher viral load or break down of endothelial barriers in peripheral organs in these patients allows the virus enters the bloodstream. Once in the bloodstream, the virus can access the brain through a number of routes.
- Zhou L., Xu Z., Castiglione G.M., Soiberman U.S., Eberhart C.G., Duh E.J. ACE2 and TMPRSS2 are expressed on the human ocular surface, suggesting susceptibility to SARS-CoV-2 infection. Ocul Surf. 2020;18:537–544. doi: 10.1016/j.jtos.2020.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]; ACE2 in the conjunctival epithelium, especially prominent in the superficial epithelium, shows focal expression in the substantia propria. Therefore, ocular surface cells including conjunctiva are susceptible to infection by SARS-CoV-2 and can therefore serve as a portal of entry as well as a reservoir for person-to-person transmission of this virus.
- Brann D.H., Tsukahara T., Weinreb C., Lipovsek M., Van den Berge K., Gong B., Chance R., Macaulay I.C., Chou H.-J., Fletcher R.B., Das D., Street K., de Bezieux H.R., Choi Y.-G., Risso D., Dudoit S., Purdom E., Mill J., Hachem R.A., Matsunami H., Logan D.W., Goldstein B.J., Grubb M.S., Ngai J., Datta S.R. Non-neuronal expression of SARS-CoV-2 entry genes in the olfactory system suggests mechanisms underlying COVID-19-associated anosmia. Sci Adv. 2020;6 doi: 10.1126/sciadv.abc5801. [DOI] [PMC free article] [PubMed] [Google Scholar]; Since the olfactory epithelium and olfactory bulb express SARS-CoV-2 cell entry molecule ACE2, SARS-CoV-2 infection of non-neuronal cell types leads to anosmia and related disturbances in odor perception in COVID-19 patients.
- 12.Engin A.B., Engin E.D., Engin A. Dual function of sialic acid in gastrointestinal SARS-CoV-2 infection. Environ Toxicol Pharmacol. 2020;79:103436. doi: 10.1016/j.etap.2020.103436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng S., Fan J., Yu F., Feng B., Lou B., Zou Q., Xie G., Lin S., Wang R., Yang X., Chen W., Wang Q., Zhang D., Liu Y., Gong R., Ma Z., Lu S., Xiao Y., Gu Y., Zhang J., Yao H., Xu K., Lu X., Wei G., Zhou J., Fang Q., Cai H., Qiu Y., Sheng J., Chen Y., Liang T. Viral load dynamics and disease severity in patients infected with SARS-CoV-2 in Zhejiang province, China, January-March 2020: retrospective cohort study. BMJ. 2020;369:m1443. doi: 10.1136/bmj.m1443. [DOI] [PMC free article] [PubMed] [Google Scholar]; In a series of 96 patients with confirmed COVID-19 by testing respiratory samples, viral nucleic acid is detected in the stool samples of 59% and serum samples of 41%. The duration of SARS-CoV-2 is significantly longer in stool samples than in respiratory and serum samples.
- 14.Kanberg N., Ashton N.J., Andersson L.-M., Yilmaz A., Lindh M., Nilsson S., Price R.W., Blennow K., Zetterberg H., Gisslén M. Neurochemical evidence of astrocytic and neuronal injury commonly found in COVID-19. Neurology. 2020;95:e1754–e1759. doi: 10.1212/WNL.0000000000010111. [DOI] [PubMed] [Google Scholar]
- 15.Paniz-Mondolfi A., Bryce C., Grimes Z., Gordon R.E., Reidy J., Lednicky J., Sordillo E.M., Fowkes M. Central nervous system involvement by severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) J Med Virol. 2020;92:699–702. doi: 10.1002/jmv.25915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Erickson M.A., Banks W.A. Neuroimmune axes of the blood-brain barriers and blood-brain interfaces: bases for physiological regulation, disease states, and pharmacological interventions. Pharmacol Rev. 2018;70:278–314. doi: 10.1124/pr.117.014647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murta V., Villarreal A., Ramos A.J. Severe acute respiratory syndrome coronavirus 2 impact on the central nervous system: are astrocytes and microglia main players or merely bystanders? ASN Neuro. 2020;12 doi: 10.1177/1759091420954960. 1759091420954960. [DOI] [PMC free article] [PubMed] [Google Scholar]; Astrocytes and microglia have a major role in neuroinflammation, responding to local CNS inflammation. Perivascular astrocytes may incorporate viral particles by direct contact with infected endothelial cells. BBB breakdown may also lead to viral infection of non-perivascular astrocytes and microglia. Astrocytes affect brain RAS system, and microglia respond to increased Ang II levels by secreting proinflammatory cytokines. Thus, downregulation in ACE2 level induced by SARS-CoV-2 could have detrimental effects both by reducing anti-inflammatory and antiapoptotic Ang (1–7) effects, exacerbating ATR1-dependent proinflammatory response and reducing brain blood flow.
- 18.Teuwen L.-A., Geldhof V., Pasut A., Carmeliet P. COVID-19: the vasculature unleashed. Nat Rev Immunol. 2020;20:389–391. doi: 10.1038/s41577-020-0343-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Michetti F., D'Ambrosi N., Toesca A., Puglisi M.A., Serrano A., Marchese E., Corvino V., Geloso M.C. The S100B story: from biomarker to active factor in neural injury. J Neurochem. 2019;148:168–187. doi: 10.1111/jnc.14574. [DOI] [PubMed] [Google Scholar]
- Perrin P., Collongues N., Baloglu S., Bedo D., Bassand X., Lavaux T., Gautier-Vargas G., Keller N., Kremer S., Fafi-Kremer S., Moulin B., Benotmane I., Caillard S. Cytokine release syndrome-associated encephalopathy in patients with COVID-19. Eur J Neurol. 2021;28:248–258. doi: 10.1111/ene.14491. [DOI] [PMC free article] [PubMed] [Google Scholar]; Penetration of proinflammatory cytokines activate microglial cells in the CNS. The clinical presentation of CNS involvement occurs with confusion, agitation, tremor, impaired consciousness, dysexecutive syndrome, pyramidal syndrome, cerebellar ataxia, cranial nerve palsy, and dysautonomia
- 21.Gallagher P.E., Chappell M.C., Ferrario C.M., Tallant E.A. Distinct roles for ANG II and ANG-(1-7) in the regulation of angiotensin-converting enzyme 2 in rat astrocytes. Am J Physiol Cell Physiol. 2006;290:C420–C426. doi: 10.1152/ajpcell.00409.2004. [DOI] [PubMed] [Google Scholar]
- 22.Hamming I., Timens W., Bulthuis M.L.C., Lely A.T., Navis G.J., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cantuti-Castelvetri L., Ojha R., Pedro L.D., Djannatian M., Franz J., Kuivanen S., Kallio K., Kaya T., Anastasina M., Smura T., Levanov L., Szirovicza L., Tobi A., Kallio-Kokko H., Österlund P., Joensuu M., Meunier F.A., Butcher S., Winkler M.S., Mollenhauer B., Helenius A., Gokce O., Teesalu T., Hepojoki J., Vapalahti O., Stadelmann C., Balistreri G., Simons M. BioRxiv; 2020. Neuropilin-1 facilitates SARS-CoV-2 cell entry and provides a possible pathway into the central nervous system. 2020.06.07.137802. [DOI] [PMC free article] [PubMed] [Google Scholar]; Neuropathological analysis of human COVID-19 autopsies revealed SARS-CoV-2 infected NRP1-positive cells in the olfactory epithelium and bulb. In the olfactory bulb infection was detected particularly within NRP1-positive endothelial cells of capillaries. NRP1-mediated transport of virus-sized particles into the central nervous system occurs via respiratory system.
- 24.Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.-H., Nitsche A., Müller M.A., Drosten C., Pöhlmann S. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DosSantos M.F., Devalle S., Aran V., Capra D., Roque N.R., Coelho-Aguiar J. de M., Spohr T.C.L. de S.E., Subilhaga J.G., Pereira C.M., D'Andrea Meira I., Niemeyer Soares Filho P., Moura-Neto V. Neuromechanisms of SARS-CoV-2: a review. Front Neuroanat. 2020;14:37. doi: 10.3389/fnana.2020.00037. [DOI] [PMC free article] [PubMed] [Google Scholar]; Loss of smell is probably the most evident symptom in SARS-CoV-2 infection. However, through direct and indirect connections, the olfactory pathway may be related to neurologic symptoms. Central nervous system changes and neurological symptoms are directly or indirectly related to the olfactory pathway. Although olfactory bulb cells strongly express ACE2, they weakly express furin, and do not express TMPRSS2.
- 26.Ueha R., Kondo K., Kagoya R., Shichino S., Ueha S., Yamasoba T. BioRxiv; 2020. Background mechanisms of olfactory dysfunction in COVID-19: expression of ACE2, TMPRSS2, and Furin in the nose and olfactory bulb in human and mice. 2020.05.15.097352. [DOI] [Google Scholar]
- 27.Lukiw W.J., Pogue A., Hill J.M. SARS-CoV-2 infectivity and neurological targets in the brain, cell. Mol Neurobiol. 2020 doi: 10.1007/s10571-020-00947-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Natoli S., Oliveira V., Calabresi P., Maia L.F., Pisani A. Does SARS-Cov-2 invade the brain? Translational lessons from animal models. Eur J Neurol. 2020;27:1764–1773. doi: 10.1111/ene.14277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiappelli F. Towards neuro-CoViD-19. Bioinformation. 2020;16:288–292. doi: 10.6026/97320630016288. [DOI] [PMC free article] [PubMed] [Google Scholar]; Inactivation of ACE2 following binding of SARS-CoV-2 S protein to ACE2-receptor in situ might reduce ACE2-moderating effects on CNS neurotoxicity and neurodegeneration. Neurobiological mechanism directly involves SARS-CoV-2 binding to ACE2-R in the etiology of putative Neuro-CoViD-19.
- 30.Shang J., Wan Y., Luo C., Ye G., Geng Q., Auerbach A., Li F. Cell entry mechanisms of SARS-CoV-2. Proc Natl Acad Sci USA. 2020;117:11727–11734. doi: 10.1073/pnas.2003138117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coutard B., Valle C., de Lamballerie X., Canard B., Seidah N.G., Decroly E. The spike glycoprotein of the new coronavirus 2019-nCoV contains a furin-like cleavage site absent in CoV of the same clade. Antivir Res. 2020;176:104742. doi: 10.1016/j.antiviral.2020.104742. [DOI] [PMC free article] [PubMed] [Google Scholar]; Based on its genome sequence, 2019-nCoV is a betacoronavirus species, which contains specific furin-like protease recognition pattern present in the vicinity of one of the maturation sites of the S protein that may have significant functional implications for virus entry. S-protein has higher pathogenicity, pronounced neural symptoms, and neurotropism. As the priming event is essential for virus entry, the efficacy and extent of this activation step by the proteases of the target cells regulate cellular tropism and viral pathogenesis.
- 32.Ulrich H., Pillat M.M. CD147 as a target for COVID-19 treatment: suggested effects of azithromycin and stem cell engagement. Stem Cell Rev Rep. 2020;16:434–440. doi: 10.1007/s12015-020-09976-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang K., Chen W., Zhang Z., Deng Y., Lian J.-Q., Du P., Wei D., Zhang Y., Sun X.-X., Gong L., Yang X., He L., Zhang L., Yang Z., Geng J.-J., Chen R., Zhang H., Wang B., Zhu Y.-M., Nan G., Jiang J.-L., Li L., Wu J., Lin P., Huang W., Xie L., Zheng Z.-H., Zhang K., Miao J.-L., Cui H.-Y., Huang M., Zhang J., Fu L., Yang X.-M., Zhao Z., Sun S., Gu H., Wang Z., Wang C.-F., Lu Y., Liu Y.-Y., Wang Q.-Y., Bian H., Zhu P., Chen Z.-N. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduct Target Ther. 2020;5:283. doi: 10.1038/s41392-020-00426-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ashby E.L., Kehoe P.G. Current status of renin-aldosterone angiotensin system-targeting anti-hypertensive drugs as therapeutic options for Alzheimer's disease. Expet Opin Invest Drugs. 2013;22:1229–1242. doi: 10.1517/13543784.2013.812631. [DOI] [PubMed] [Google Scholar]
- 35.Kehoe P.G., Wong S., Al Mulhim N., Palmer L.E., Miners J.S. Angiotensin-converting enzyme 2 is reduced in Alzheimer's disease in association with increasing amyloid-β and tau pathology. Alzheimer's Res Ther. 2016;8:50. doi: 10.1186/s13195-016-0217-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Shiers S., Ray P.R., Wangzhou A., Sankaranarayanan I., Tatsui C.E., Rhines L.D., Li Y., Uhelski M.L., Dougherty P.M., Price T.J. ACE2 and SCARF expression in human dorsal root ganglion nociceptors: implications for SARS-CoV-2 virus neurological effects. Pain. 2020;161:2494–2501. doi: 10.1097/j.pain.0000000000002051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kuo C.-L., Pilling L.C., Atkins J.L., Masoli J.A.H., Delgado J., Kuchel G.A., Melzer D. APOE e4 Genotype Predicts Severe COVID-19 in the UK Biobank Community Cohort. J Gerontol A Biol Sci Med Sci. 2020;75:2231–2232. doi: 10.1093/gerona/glaa131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H., Yuan Z., Pavel M.A., Hobson R., Hansen S.B. BioRxiv; 2020. The role of high cholesterol in age-related COVID19 lethality. 2020.05.09.086249. [DOI] [Google Scholar]; The cholesterol concomitantly traffics angiotensinogen-converting enzyme to the viral entry site where SARS-CoV-2 docks to properly exploit entry into the cell. Thereby, it was demonstrated that cholesterol enhances binding of SARS-CoV-2 to the cell surface which increases association with the endocytic pathway. Contrarily, rapidly dropping cholesterol in the blood may indicate severe loading of cholesterol in peripheral tissue and a dangerous situation for escalated SARS-CoV-2 infectivity.
- 39.Asselta R., Paraboschi E.M., Mantovani A., Duga S. ACE2 and TMPRSS2 variants and expression as candidates to sex and country differences in COVID-19 severity in Italy. Aging. 2020;12:10087–10098. doi: 10.18632/aging.103415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh K.K., Chaubey G., Chen J.Y., Suravajhala P. Decoding SARS-CoV-2 hijacking of host mitochondria in COVID-19 pathogenesis. Am J Physiol Cell Physiol. 2020;319:C258–C267. doi: 10.1152/ajpcell.00224.2020. [DOI] [PMC free article] [PubMed] [Google Scholar]; A decline in ACE2 function in aged individuals, coupled with the age-associated decline in mitochondrial functions result in chronic metabolic diseases. These make the host more vulnerable to infection and health complications leading to mortality. Distinct localization of viral RNA and proteins in mitochondria must play essential roles in SARS-CoV-2 pathogenesis. Understanding the mechanisms underlying virus communication with host mitochondria may provide critical insights into COVID-19 pathologies.
- Daly J.L., Simonetti B., Antón-Plágaro C., Williamson M.K., Shoemark D.K., Simón-Gracia L., Klein K., Bauer M., Hollandi R., Greber U.F., Horvath P., Sessions R.B., Helenius A., Hiscox J.A., Teesalu T., Matthews D.A., Davidson A.D., Cullen P.J., Yamauchi Y. BioRxiv; 2020. Neuropilin-1 is a host factor for SARS-CoV-2 infection. 2020.06.05.134114. [DOI] [PMC free article] [PubMed] [Google Scholar]; Cleavage of S generates a polybasic Arg-Arg-Ala-Arg carboxyl-terminal sequence on S1, which conforms to a C-end rule (CendR) motif that binds to cell surface neuropilin-1 (NRP1) and NRP2 receptors. Blocking this interaction by RNA interference or selective inhibitors reduce SARS-CoV-2 entry and infectivity. NRP1, thus, serves as a host factor for SARS-CoV-2 infection and may potentially provide a therapeutic target for COVID-19.
- 42.Moutal A., Martin L.F., Boinon L., Gomez K., Ran D., Zhou Y., Stratton H.J., Cai S., Luo S., Gonzalez K.B., Perez-Miller S., Patwardhan A., Ibrahim M.M., Khanna R. SARS-CoV-2 Spike protein co-opts VEGF-A/Neuropilin-1 receptor signaling to induce analgesia. Pain. 2020 doi: 10.1097/j.pain.0000000000002097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Vargas G., Medeiros Geraldo L.H., Gedeão Salomão N., Viana Paes M., Regina Souza Lima F., Carvalho Alcantara Gomes F. Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and glial cells: insights and perspectives. Brain Behav Immun - Health. 2020;7:100127. doi: 10.1016/j.bbih.2020.100127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jiang T., Gao L., Shi J., Lu J., Wang Y., Zhang Y. Angiotensin-(1-7) modulates renin-angiotensin system associated with reducing oxidative stress and attenuating neuronal apoptosis in the brain of hypertensive rats. Pharmacol Res. 2013;67:84–93. doi: 10.1016/j.phrs.2012.10.014. [DOI] [PubMed] [Google Scholar]
- 45.Wu J., Zhao D., Wu S., Wang D. Ang-(1-7) exerts protective role in blood-brain barrier damage by the balance of TIMP-1/MMP-9. Eur J Pharmacol. 2015;748:30–36. doi: 10.1016/j.ejphar.2014.12.007. [DOI] [PubMed] [Google Scholar]
- 46.Xu J., Sriramula S., Lazartigues E. Excessive glutamate stimulation impairs ACE2 activity through ADAM17-mediated shedding in cultured cortical neurons. Cell Mol Neurobiol. 2018;38:1235–1243. doi: 10.1007/s10571-018-0591-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Xia H., Sriramula S., Chhabra K.H., Lazartigues E. Brain angiotensin-converting enzyme type 2 shedding contributes to the development of neurogenic hypertension. Circ Res. 2013;113:1087–1096. doi: 10.1161/CIRCRESAHA.113.301811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Deshotels M.R., Xia H., Sriramula S., Lazartigues E., Filipeanu C.M. Angiotensin II mediates angiotensin converting enzyme type 2 internalization and degradation through an angiotensin II type I receptor-dependent mechanism. Hypertens. Dallas Tex. 2014;64:1368–1375. doi: 10.1161/HYPERTENSIONAHA.114.03743. 1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Doble A. The role of excitotoxicity in neurodegenerative disease: implications for therapy. Pharmacol Ther. 1999;81:163–221. doi: 10.1016/s0163-7258(98)00042-4. [DOI] [PubMed] [Google Scholar]
- 50.Szydlowska K., Tymianski M. Calcium, ischemia and excitotoxicity. Cell Calcium. 2010;47:122–129. doi: 10.1016/j.ceca.2010.01.003. [DOI] [PubMed] [Google Scholar]
- 51.Almeida A., Heales S.J., Bolaños J.P., Medina J.M. Glutamate neurotoxicity is associated with nitric oxide-mediated mitochondrial dysfunction and glutathione depletion. Brain Res. 1998;790:209–216. doi: 10.1016/s0006-8993(98)00064-x. [DOI] [PubMed] [Google Scholar]
- 52.Pereira C.F., Oliveira C.R. Oxidative glutamate toxicity involves mitochondrial dysfunction and perturbation of intracellular Ca2+ homeostasis. Neurosci Res. 2000;37:227–236. doi: 10.1016/s0168-0102(00)00124-3. [DOI] [PubMed] [Google Scholar]
- Taylor E.W., Radding W. Understanding Selenium and glutathione as antiviral factors in COVID-19: does the viral mpro protease target host selenoproteins and glutathione synthesis? Front. Nutr. 2020;7:143. doi: 10.3389/fnut.2020.00143. [DOI] [PMC free article] [PubMed] [Google Scholar]; There is a correlation between COVID-19 clinical outcomes and both host selenium and glutathione (GSH) status. SARS-CoV2 targets GSH biosynthesis as well as thioredoxin reductase-1 and glutathione peroxidases for proteolytic knockdown, in infected cells. The resulting decreases in these critical antioxidant molecules contribute to increased oxidative stress. GSH and selenoprotein levels might be low in people with suboptimal nutritional status. These patients could be particularly at risk because their nutritional deficiency makes them more vulnerable to the detrimental effects of virus-induced proteolysis.
- 54.Ward R.J., Colivicchi M.A., Allen R., Schol F., Lallemand F., de Witte P., Ballini C., Corte L.D., Dexter D. Neuro-inflammation induced in the hippocampus of “binge drinking” rats may be mediated by elevated extracellular glutamate content. J Neurochem. 2009;111:1119–1128. doi: 10.1111/j.1471-4159.2009.06389.x. [DOI] [PubMed] [Google Scholar]
- 55.Chang Y.C., Kim H.-W., Rapoport S.I., Rao J.S. Chronic NMDA administration increases neuroinflammatory markers in rat frontal cortex: cross-talk between excitotoxicity and neuroinflammation. Neurochem Res. 2008;33:2318–2323. doi: 10.1007/s11064-008-9731-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Qi X.-L., Nordberg A., Xiu J., Guan Z.-Z. The consequences of reducing expression of the alpha7 nicotinic receptor by RNA interference and of stimulating its activity with an alpha7 agonist in SH-SY5Y cells indicate that this receptor plays a neuroprotective role in connection with the pathogenesis of Alzheimer's disease. Neurochem Int. 2007;51:377–383. doi: 10.1016/j.neuint.2007.04.002. [DOI] [PubMed] [Google Scholar]
- 57.McShane R., Westby M.J., Roberts E., Minakaran N., Schneider L., Farrimond L.E., Maayan N., Ware J., Debarros J. Memantine for dementia. Cochrane Database Syst Rev. 2019;3:CD003154. doi: 10.1002/14651858.CD003154.pub6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasanagic S., Serdarevic F. Potential role of memantine in the prevention and treatment of COVID-19: its antagonism of nicotinic acetylcholine receptors and beyond. Eur Respir J. 2020;56 doi: 10.1183/13993003.01610-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]; Memantine, in addition to its noncompetitive NMDA receptor antagonism, is a very potent α7-nAChR antagonist. Memantine may reduce virulence and pathogenicity of SARS-CoV-2 and potentially exerts its effects both in the lungs and the brain. However, such claims require further thorough experimental, epidemiological, and clinical confirmation.
- Romero-Sánchez C.M., Díaz-Maroto I., Fernández-Díaz E., Sánchez-Larsen Á., Layos-Romero A., García-García J., González E., Redondo-Peñas I., Perona-Moratalla A.B., Del Valle-Pérez J.A., Gracia-Gil J., Rojas-Bartolomé L., Feria-Vilar I., Monteagudo M., Palao M., Palazón-García E., Alcahut-Rodríguez C., Sopelana-Garay D., Moreno Y., Ahmad J., Segura T. Neurologic manifestations in hospitalized patients with COVID-19: the ALBACOVID registry. Neurology. 2020;95:e1060–e1070. doi: 10.1212/WNL.0000000000009937. [DOI] [PMC free article] [PubMed] [Google Scholar]; More than half of COVID-19 patients present neurologic symptom. Clinicians need to maintain close neurologic surveillance for prompt recognition of these complications. However, the mechanisms and consequences of neurologic involvement require further studies.
- Song E., Zhang C., Israelow B., Lu-Culligan A., Prado A.V., Skriabine S., Lu P., Weizman O.-E., Liu F., Dai Y., Szigeti-Buck K., Yasumoto Y., Wang G., Castaldi C., Heltke J., Ng E., Wheeler J., Alfajaro M.M., Levavasseur E., Fontes B., Ravindra N.G., Dijk D.V., Mane S., Gunel M., Ring A., Kazmi S.A.J., Zhang K., Wilen C.B., Horvath T.L., Plu I., Haik S., Thomas J.-L., Louvi A., Farhadian S.F., Huttner A., Seilhean D., Renier N., Bilguvar K., Iwasaki A. BioRxiv; 2020. Neuroinvasion of SARS-CoV-2 in human and mouse brain. 2020.06.25.169946. [DOI] [PMC free article] [PubMed] [Google Scholar]; It is not known whether or not ACE2 is the main route of entry of SARS-CoV-2 into neuronal cells and what strategies might block viral infection, and whether there are humoral antibody responses against SARS-CoV-2 in the CNS of infected patients. Furthermore, it is also not known whether antibodies present in the CNS can prevent infection of neurons. In some patients with COVID-19 and neurological symptoms, there is robust antibody response to the virus within the CSF. Thereby, nonspecific neurologic symptoms may manifest.
- Cazzolla A.P., Lovero R., Lo Muzio L., Testa N.F., Schirinzi A., Palmieri G., Pozzessere P., Procacci V., Di Comite M., Ciavarella D., Pepe M., De Ruvo C., Crincoli V., Di Serio F., Santacroce L. Taste and smell disorders in COVID-19 patients: role of interleukin-6. ACS Chem Neurosci. 2020;11:2774–2781. doi: 10.1021/acschemneuro.0c00447. [DOI] [PubMed] [Google Scholar]; Interleukin-6 levels in COVID-19 patients in relation to olfactory or gustatory disorders were correlated from the time of their admission to the time of swab negativization.
- 62.Niklassen A.S., Draf J., Huart C., Hintschich C., Bocksberger S., Trecca E.M.C., Klimek L., Le Bon S.D., Altundag A., Hummel T. COVID-19: recovery from chemosensory dysfunction. A multicentre study on smell and taste. Laryngoscope. 2021 doi: 10.1002/lary.29383. [DOI] [PubMed] [Google Scholar]
- 63.Parsons T., Banks S., Bae C., Gelber J., Alahmadi H., Tichauer M. COVID-19-associated acute disseminated encephalomyelitis (ADEM) J Neurol. 2020;267:2799–2802. doi: 10.1007/s00415-020-09951-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Poyiadji N., Shahin G., Noujaim D., Stone M., Patel S., Griffith B. COVID-19-associated acute hemorrhagic necrotizing encephalopathy: imaging features. Radiology. 2020;296:E119–E120. doi: 10.1148/radiol.2020201187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Manji H.K., George U., Mkopi N.P., Manji K.P. Guillain-Barré syndrome associated with COVID-19 infection. Pan Afr. Med. J. 2020;35:118. doi: 10.11604/pamj.supp.2020.35.2.25003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou Z., Kang H., Li S., Zhao X. Understanding the neurotropic characteristics of SARS-CoV-2: from neurological manifestations of COVID-19 to potential neurotropic mechanisms. J Neurol. 2020;267:2179–2184. doi: 10.1007/s00415-020-09929-7. [DOI] [PMC free article] [PubMed] [Google Scholar]; It has also been reported recently that some cases of COVID-19 have presented with concurrent acute ischemic stroke, cerebral venous sinus thrombosis, cerebral hemorrhage, subarachnoid hemorrhage. Although detection of SARS-CoV-2 RNA in cerebrospinal fluid specimens of a patient with COVID-19 have provided direct evidence to support the theory of neurotropic involvement of SARS-CoV-2, the underlying neurotropic mechanisms are not clear.
- Lodigiani C., Iapichino G., Carenzo L., Cecconi M., Ferrazzi P., Sebastian T., Kucher N., Studt J.-D., Sacco C., Bertuzzi A., Sandri M.T., Barco S. Humanitas COVID-19 Task Force, Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thromb Res. 2020;191:9–14. doi: 10.1016/j.thromres.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]; The rate of ischemic stroke and acute coronary syndrome/myocardial infarction is 2.5% and 1.1%, respectively. Overt disseminated intravascular coagulation is present in eight (2.2%) patients.
- Yaghi S., Ishida K., Torres J., Mac Grory B., Raz E., Humbert K., Henninger N., Trivedi T., Lillemoe K., Alam S., Sanger M., Kim S., Scher E., Dehkharghani S., Wachs M., Tanweer O., Volpicelli F., Bosworth B., Lord A., Frontera J. SARS-CoV-2 and stroke in a New York healthcare system. Stroke. 2020;51:2002–2011. doi: 10.1161/STROKEAHA.120.030335. [DOI] [PMC free article] [PubMed] [Google Scholar]; Out of 3556 hospitalized patients with diagnosis of COVID-19 infection, 32 patients (0.9%) had imaging proven ischemic stroke. Patients with COVID-19 and stroke had significantly higher mortality than controls. Most strokes are related to an acquired hypercoagulability. Studies are needed to determine the utility of therapeutic anticoagulation for stroke and other thrombotic event prevention in patients with COVID-19
- 69.Ranucci M., Ballotta A., Di Dedda U., Bayshnikova E., Dei Poli M., Resta M., Falco M., Albano G., Menicanti L. The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome. J Thromb Haemost JTH. 2020;18:1747–1751. doi: 10.1111/jth.14854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fan H., Tang X., Song Y., Liu P., Chen Y. Influence of COVID-19 on cerebrovascular disease and its possible mechanism. Neuropsychiatric Dis Treat. 2020;16:1359–1367. doi: 10.2147/NDT.S251173. [DOI] [PMC free article] [PubMed] [Google Scholar]; Cerebrovascular disease has high incidence, disability rate, and fatality rate in COVID-19 patients.
- 71.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet Lond Engl. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Liu Y., Yang Y., Zhang C., Huang F., Wang F., Yuan J., Wang Z., Li J., Li J., Feng C., Zhang Z., Wang L., Peng L., Chen L., Qin Y., Zhao D., Tan S., Yin L., Xu J., Zhou C., Jiang C., Liu L. Clinical and biochemical indexes from 2019-nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63:364–374. doi: 10.1007/s11427-020-1643-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Li M., Wang M., Zhou Y., Chang J., Xian Y., Wang D., Mao L., Jin H., Hu B. Acute cerebrovascular disease following COVID-19: a single center, retrospective, observational study. Stroke Vasc Neurol. 2020;5:279–284. doi: 10.1136/svn-2020-000431. [DOI] [PMC free article] [PubMed] [Google Scholar]; Of 219 patients with COVID-19, 10 (4.6%) have developed acute ischemic stroke and 1 (0.5%) had intracerebral hemorrhage. Older patients with cardiovascular risk factors are more likely to develop acute cerebrovascular disease. These patients have increased inflammatory response and hypercoagulable state as reflected in C reactive protein and D-dimer levels.
- Mohammadi S., Moosaie F., Aarabi M.H. Understanding the immunologic characteristics of neurologic manifestations of SARS-CoV-2 and potential immunological mechanisms. Mol Neurobiol. 2020;57:5263–5275. doi: 10.1007/s12035-020-02094-y. [DOI] [PMC free article] [PubMed] [Google Scholar]; Besides direct viral invasion to the central nervous system (CNS), aberrant immune pathways such as extreme release of cytokines (cytokine storm), autoimmunity mediated by cross-reactivity between CNS components and viral particles, and microglial activation increase the CNS damage in these patients.
- Chauhan A.J., Wiffen L.J., Brown T.P. COVID-19: a collision of complement, coagulation and inflammatory pathways. J Thromb Haemost JTH. 2020;18:2110–2117. doi: 10.1111/jth.14981. [DOI] [PMC free article] [PubMed] [Google Scholar]; Disruption of the homeostatic interactions between complement and extrinsic and intrinsic coagulation pathways contributes to a net procoagulant state in the microvasculature of critical organs. Fatal COVID-19 has been associated with a systemic inflammatory response accompanied by a procoagulant state and organ damage, particularly microvascular thrombi.
- Fletcher-Sandersjöö A., Bellander B.-M. Is COVID-19 associated thrombosis caused by overactivation of the complement cascade? A literature review. Thromb Res. 2020;194:36–41. doi: 10.1016/j.thromres.2020.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]; Increased complement activity leads to increased coagulation cascade activity and platelet aggregation. Inappropriate activation of the complement pathways in COVID-19 result in thrombotic complications. Although similarities exist with known complementopathies, there are yet no studies that have examined if there is an interaction between the complement and coagulation system in COVID-19.
- Soltani Zangbar H., Gorji A., Ghadiri T. A review on the neurological manifestations of COVID-19 infection: a mechanistic view. Mol Neurobiol. 2020 doi: 10.1007/s12035-020-02149-0. [DOI] [PMC free article] [PubMed] [Google Scholar]; ACE2 plays an important role in developing neurologic manifestations of SARS-CoV-2 infection, through their regulatory effects on blood circulation. Stroke aggravates COVID-19 outcomes.