Abstract
Background
To assess diagnostic performance of lung ultrasound (LUS) in identifying ARDS morphology (focal vs non-focal), compared with the gold standard computed tomography.
Methods
Mechanically ventilated ARDS patients undergoing lung computed tomography and ultrasound were enrolled. Twelve fields, were evaluated. LUS score was graded from 0 (normal) to 3 (consolidation) according to B-lines extent. Total and regional LUS score as the sum of the four ventral (LUSV), intermediate (LUSI) or dorsal (LUSD) fields, were calculated. Based on lung CT, ARDS morphology was defined as (1) focal (loss of aeration with lobar distribution); (2) non-focal (widespread loss of aeration or segmental loss of aeration distribution associated with uneven lung attenuation areas), and diagnostic accuracy of LUS in discriminating ARDS morphology was determined by AU-ROC in training and validation set of patients.
Results
Forty-seven patients with ARDS (25 training set and 22 validation set) were enrolled. LUSTOT, LUSV and LUSI but not LUSD score were significantly lower in focal than in non-focal ARDS morphologies (p < .01). The AU-ROC curve of LUSTOT, LUSV, LUSI and LUSD for identification of non-focal ARDS morphology were 0.890, 0.958, 0.884 and 0.421, respectively. LUSV value ≥ 3 had the best predictive value (sensitivity = 0.95, specificity = 1.00) in identifying non-focal ARDS morphology. In the validation set, an LUSV score ≥ 3 confirmed to be highly predictive of non-focal ARDS morphology, with a sensitivity and a specificity of 94% and 100%.
Conclusions
LUS had a valuable performance in distinguishing ARDS morphology.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13613-021-00837-1.
Keywords: Lung ultrasound, ARDS, ARDS morphology, Bedside tests, Point of care diagnostic tests, Respiratory monitoring
Background
Acute respiratory distress syndrome (ARDS) is characterized by significant loss of lung aeration and increased lung weight as a consequence of increased lung permeability leading to accumulation of protein-rich edema [1]. Computed tomography (CT) is the gold standard imaging technique for the identification, characterization of distribution, and quantification of loss of lung aeration during ARDS [2]. CT scan thus can predict the potential for alveolar recruitment, which is variable among patients with ARDS [3]. Compared to focal (lobar loss of lung aeration), non-focal (diffuse/patchy loss of lung aeration) pattern show greater alveolar recruitment and less signs of over-distension when an open lung strategy is used (i.e., use of recruitment maneuvers and higher positive end-expiratory pressure) [2]. Lung ultrasound (LUS) has been proposed as an accurate bedside and radiation-free technique for evaluation of lung consolidations [4] and for follow-up of aeration changes in response to interventions [5–7]. Scores based on detection of B-lines (the sonographic sign of increased lung density associated with interstitial syndrome) and on consolidation have been correlated with global and regional lung aeration as assessed by CT [8, 9]. However, the role of LUS in identifying ARDS morphologic pattern has not been investigated. Therefore, our study explored the feasibility and accuracy of lung ultrasound as imaging technique for identification of ARDS morphology (focal vs non-focal) as compared to CT scan. We hypothesized that LUS performed at the bedside accurately quantifies aeration loss in ARDS patients, providing useful information about ARDS morphology (focal vs non-focal).
Methods
Subjects
All consecutive mechanically ventilated patients admitted to a tertiary center intensive care unit (ICU) with a diagnosis of ARDS [10], with an expected duration of mechanical ventilation greater than 24 h and undergoing CT scan evaluation of the lung parenchyma, were included. Exclusion criteria were age below 18 years, confirmed diagnosis of pulmonary fibrosis or moribund patient. The local Ethics Committee approved the study protocol (0117126) and written consent was obtained according to Italian regulation.
Study protocol
All patients underwent to lung CT scan at study entry. Immediately after every CT scan completion, LUS was performed at bedside in the ICU, with the same level of sedation and ventilator settings as during the CT scan.
Lung ultrasound
Patients were examined in supine position, using a portable ultrasound machine (Mylab™ seven, Esaote S.p.A, Genova, Italy) equipped with a curvilinear transducer (5–3 MHz) [11]. Twelve fields, six for each hemithorax, were analyzed based on predefined anatomical landmarks to encompass ventral, intermediate and dorsal lung zones [11, 12]; a detailed description of theselandmarks is given in ESM document (Fig. 1; Additional file 2). Each area was examined for identification of four ultrasound aeration patterns [5, 7, 13–15]: 1) normal aeration (N): presence of lung sliding and/or lung pulse with A-lines or fewer than two isolated B-lines/intercostal space; 2) moderate loss of lung aeration (B1 profile): multiple spaced B-lines, ≥ 3/intercostal space; 3) severe loss of lung aeration (B2 profile): multiple coalescent B lines (± subpleural consolidations); and 4) lung consolidation (C): presence of a tissue pattern ± air bronchograms. For each field of interest, a score was assigned: N = 0, B1 = 1, B2 = 2, C = 3 [13]. A total Lung Ultrasound Score (LUSTOT), ranging between 0 and 36, was calculated as the sum of individual scores of each field [13]. Regional Lung Ultrasound Score to assess the effect of gravity on lung aeration was also calculated. LUS score in the ventral lung regions (LUSV) was calculated as the sum of the scores of the fields 1, 2, 7 and 8; in the intermediate lung regions (LUSI) was the sum of the scores of the fields 3, 4, 9 and 10; in the dorsal lung regions (LUSD) was the sum of the scores of the fields 5, 6, 11 and 12. Each regional score ranged from 0 to 12. Inter-observer agreement between operators was evaluated in validation set using Cohen κ with associated 95% confidence intervals.
Fig. 1.
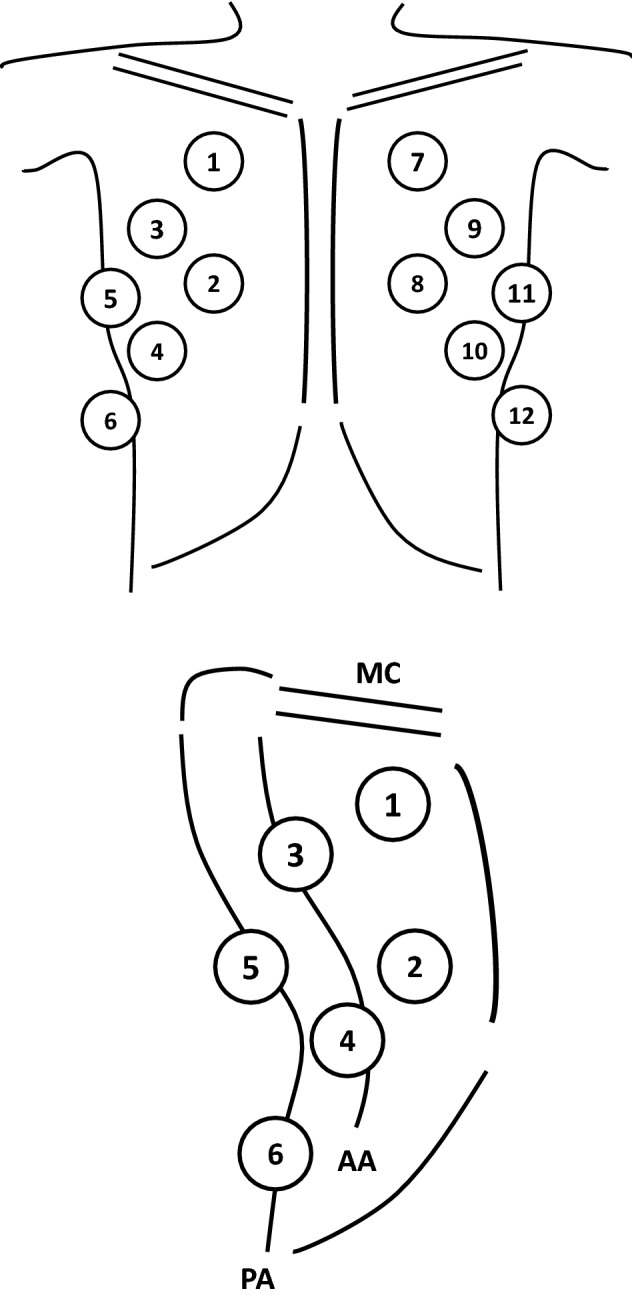
Representative image of ultrasound anatomical landmarks
Lung computed tomography
Lung CT scans were obtained at study entry. Twelve lung areas, six for each hemithorax, were identified on CT axial plane images using pre-defined anatomical landmarks matching previously described twelve LUS regions of interest. A detailed description of these landmarks is given in ESM document. Quantitative analysis of the previously acquired DICOM files was performed blindly using a dedicated software (Maluna®, University of Mannheim, Germany) [16, 17]. The total area of the selected region of interest consisted of a finite number of pixels. The X-ray attenuation of each pixel, expressed in Hounsfield units (HU), was obtained by determining the percentage of radiation adsorbed [17]. The following hyperinflated (between − 900 and − 1000 HU); normally aerated (− 900 and − 500 HU); poorly aerated (− 500 and − 100 HU); and non-aerated (− 100 and 100 HU) lung compartments were quantified [17].
Two physicians (VF and AC) blindly and independently reviewed and categorized the CT ARDS morphology, according to the CT scan ARDS Study Group criteria [18]. Two ARDS morphologies were defined: (1) focal (loss of aeration with lobar or segmental distribution) and (2) non-focal (widespread loss of aeration or segmental loss of aeration distribution associated with uneven lung attenuation areas—diffuse/patchy).
Statistics
The study was conducted in two phases. In Phase 1, a first group of patients (training set) was analysed to determine diagnostic accuracy of global and regional LUS and threshold values able to best discriminate patients with non-focal ARDS morphology. In Phase 2, a second group of patients (validation set) was used to prospectively assess the diagnostic performance of LUS thresholds. Descriptive data are presented as mean and standard deviation (SD) or median and interquartile range (IQR) (continuous variables), and as numbers and percentages (categorical variables), as appropriate. Comparisons were performed using paired or unpaired t-test for continuous parametric variables, the Wilcoxon test for matched non-parametric continuous variables, the Wilcoxon-Mann–Whitney or the Kruskal–Wallis test with Dunn's pairwise or Friedman comparison for unpaired or paired continuous variables, as appropriate. Categorical variables were analyzed with Pearson chi-square test or Fisher’s exact test, as appropriate.
Optimal cut-offs values of regional and total LUS scores in identifying ARDS morphologies were analyzed by non-parametric Receiver Operating Characteristic (ROC) curve analyses with Youden method for empirical cut-point estimation. Statistical analyses were performed using Stata 13.1/SE (Stata Corporation, Texas, USA).
Results
Study population
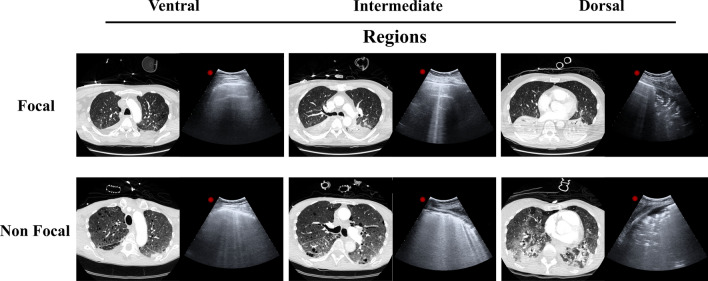
Forty-seven consecutive patients (25 in the training set and 22 in the validation set) were enrolled in the study. Baseline characteristics of patients, ventilation settings, blood gas exchange, hemodynamics are reported in Table 1. Forty-seven lung CT scans were performed (25 in the training set and 22 in the validation set); representative lung CT and LUS images of different ARDS morphologies are shown in Fig. 2.
Table 1.
Baseline characteristics of the study population patients, ventilation settings, blood gas exchange and hemodynamic
| Variables | Overall (N = 47) | Training set (N = 25) | Validation set (N = 22) |
|---|---|---|---|
| Age (years) | 52 (44–63) | 57 (50–65) | 48 (41–58) |
| PBW (kg) | 64 (57–71) | 65 (57–71) | 64 (58–70) |
| Gender (M/F) | 31/16 | 17/8 | 14/8 |
| Risk factors for ARDS—N (%) | |||
| Pneumonia | 42 (89) | 24 (85) | 21 (96) |
| Non-pulmonary sepsis | 2 (4) | 2 (7) | 0 (0) |
| Pancreatitis | 3 (6) | 2 (7) | 1 (4) |
| SAPS II score | 36 (29–45) | 34 (27–44) | 39 (29–47) |
| SOFA score | 8 (7–11) | 8 (7–11) | 9 (7–11) |
| TV/PBW (mL/kg) | 6.6 (5.9–7.8) | 6.9 (6.0–7.9) | 6.4 (5.8–7.5) |
| PEEPtot (cmH2O) | 14 (11–15) | 13 (10–15) | 14 (12–16) |
| Pplat (cmH2O) | 25 (23–27) | 25 (23–27) | 25 (20–26) |
| FiO2 | 0.6 (0.5–0.8) | 0.6 (0.5–0.8) | 0.5 (0.45–0.7) |
| Blood gas exchange | |||
| pH | 7.41 (7.35–7.46) | 7.41 (7.35–7.45) | 7.39 (7.35–7.46) |
| PaO2/FiO2 | 166 (109–232) | 156 (108–233) | 178 (114–240) |
| PaCO2 | 48 (41–52) | 48 (41–52) | 48 (42–50) |
| Lactate | 1.7 (1.3–2.2) | 1.7 (1.3–2.6) | 1.6 (1.2–1.8) |
| MAP (mmHg) | 83 (75–89) | 82 (73–88) | 84 (77–94) |
| HR (bpm) | 89 (77–98) | 91 (81–98) | 86 (77–100) |
PBW: predicted body weight; TV: tidal volume; RR: respiratory rate; PEEP: positive end-expiratory pressure; Pplat: plateau pressure; FiO2: fraction of inspired oxygen; PaO2: partial pressure of oxygen (arterial blood); PaCO2: partial pressure of carbon dioxide (arterial blood); MAP: mean arterial pressure; HR: heart rate
Fig. 2.
Representative lung TC images and their corresponding LUS images in ventral, intermediate and dorsal lung regions in focal and non-focal ARDS morphologies
Total and regional LUS score in different ARDS morphologies
Overall, LUSTOT was significantly lower in the focal compared to the non-focal ARDS morphology [focal 14 (IQR 10–20); non-focal 24 (IQR 18–27), p < 0.01] (Fig. 3). In both ventral and intermediate regions, LUS scores were significantly lower in focal [LUSV 1 (IQR 0–2); LUSI 4 (IQR 2–7) compared to non-focal [LUSV 6 (IQR 6–8), p < 0.01; LUSI 8 (IQR 5–9), p < 0.05] ARDS morphology (Fig. 3). Finally, in focal morphology, LUS score was significantly lower in ventral compared to dorsal [LUSV 1 (0–2) vs LUSD 10 (6–12); p < 0.01] and in intermediate vs dorsal lung regions [LUSI 4 (IQR 2–7) vs LUSD 10 (6–12); p < 0.05]. In non-focal morphology, LUS score was significantly lower in ventral compared to intermediate and dorsal lung regions [LUSV 6 (4–8) vs LUSI 8 (5–9); p < 0.01 and LUSD 9 (8–11); p < 0.01] and in intermediate compared to dorsal regions [LUSI 8 (5–9) vs LUSD 9 (8–11); p < 0.01] (Fig. 3). A detailed description of regional and global LUS in training and validation set is presented in Table 2. Inter-observer agreement showed a substantial agreement (κ = 0.87, 95% CI 0.81–0.92).
Fig. 3.
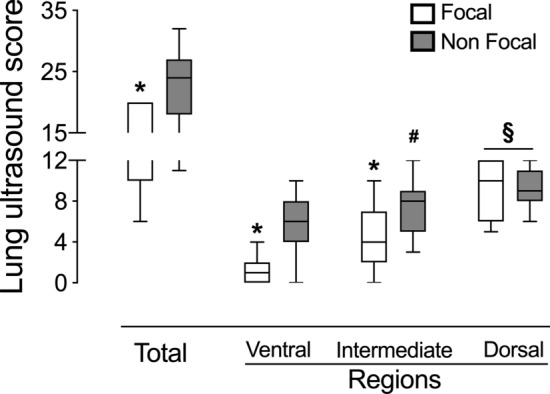
Total and regional LUS score in focal and non-focal ARDS morphologies in the overall population. *p < 0.01 focal vs non-focal ARDS morphologies; #p < 0.01 Ventral vs Intermediate lung regions in non-focal ARDS morphology; §p < 0.01 Ventral and §p < 0.05 Intermediate vs Dorsal lung regions in focal and non-focal ARDS morphology
Table 2.
Global and regional LUS scores in overall patients and in training and validation sets
| Variables | Overall (N = 47) | Training set (N = 25) | Validation set (N = 22) | |||
|---|---|---|---|---|---|---|
| Focal (N = 11) | Non-focal (N = 36) | Focal (N = 5) | Non-focal (N = 20) | Focal (N = 6) | Non-focal (N = 16) | |
| LUSTOT | 14 (10–20) | 24 (18–27)* | 14 (8–19) | 24 (19–27)* | 16 (12–20) | 22 (17–27) ** |
| LUSV | 1 (0–2) | 6 (4–8)* | 1 (0–1) | 6 (5–8)* | 2 (1–3) | 6 (4–7)* |
| LUSI | 4 (2–7) | 8 (5–9)*,o | 3 (1–7) | 9 (6–9)**,oo | 4 (3–9) | 8 (4–10) |
| LUSD | 10 (6–12)o,@@ | 9 (8–11)o,@@ | 12 (6–12)oo | 9 (8–11)o | 10 (7–11)o | 10 (7–12) o |
Abbreviations: LUSTOT: lung ultrasound score total; LUSV: lung ultrasound score ventral region; LUSI lung ultrasound score intermediate region; LUSD lung ultrasound score dorsal region
*p < 0.01, **p < 0.05 VS focal; op < 0.01, oop < 0.05 VS LUSV; @@ p < 0.05 VS LUSI
Amount of normally, poorly and not aerated lung tissue at different LUS scores
To better understand the differences between LUS and CT in assessing lung aeration, we analyzed the distribution of lung aeration on CT at different LUS score (Additional file 1: Fig. S1). In both focal and non-focal ARDS morphologies, the amount of normally aerated tissue significantly decreased from LUS 0 to 3, while the amount of not aerated tissue significantly increased (Additional file 1: Fig. S1 panel A and B). On the contrary, the amount of poorly aerated tissue did not change at different scores (Additional file 1: Fig. S1 panel A) in focal ARDS while significantly increased in non-focal ARDS (Additional file 1: Fig. S1 panel B, Additional file 2).
Accuracy of LUS in identifying ARDS morphology
In the overall population, the areas under the ROC curve of LUSTOT, LUSV, LUSI and LUSD for identification of non-focal ARDS morphology were 0.839, 0.948, 0.786 and 0.478, respectively. In the training set, the areas under the ROC curve of LUSTOT, LUSV, LUSI and LUSD for identification of non-focal ARDS morphology were 0.890, 0.958, 0.884 and 0.421, respectively. In the validation set, the areas under the ROC curve of LUSTOT, LUSV, LUSI and LUSD for identification of non-focal ARDS morphology were 0.781, 0.932, 0.703 and 0.516, respectively (Fig. 4). An LUSV score ≥ 3 (calculated on training set) had the best predictive value (sensitivity = 0.95, specificity = 1.00) for the identification of non-focal ARDS morphology (Table 3). In the validation set, an LUSV score ≥ 3 confirmed to be highly predictive of non-focal ARDS morphology, with a sensitivity and a specificity of 94% (95% CI 70–100%) and 100% (95% CI 54–100%), respectively, and a positive predictive value and a negative predictive value of 100% and 86% (95% CI 47–98%), respectively.
Fig. 4.
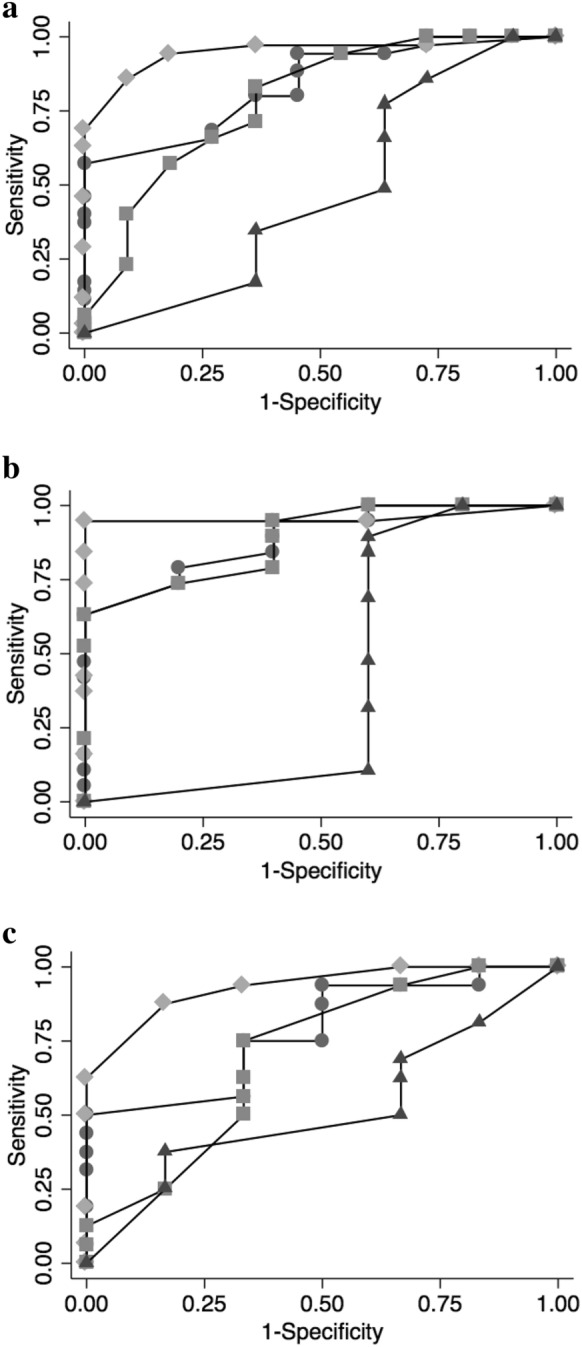
Combined Receiver Operating Characteristic (ROC) curves of overall (panel A), training set (panel B) and validation set (panel C) for total (circles) and regional ventral (rhombus), intermediate (squares) and dorsal (triangles) LUS score in identifying non-focal ARDS morphologies. AUCROC (95% CI) of LUSV for non-focal ARDS was 0.948 (0.888–1.000), 0.958 (0.881–1.000) and 0.932 (0.832–1.000) in overall, training set and validation set, respectively
Table 3.
Accuracy of prediction of regional LUSV score for non-focal ARDS morphology, by non-parametric receiver operating characteristic (ROC) analysis in the training set
| LUSV | Sensitivity (%) | Specificity (%) | Correctly classified (%) | LR+ | LR− |
|---|---|---|---|---|---|
| ≥ 0 | 100.00 | 0.00 | 80.00 | 1.0000 | |
| ≥ 1 | 95.00 | 40.00 | 84.00 | 1.5833 | 0.1250 |
| ≥ 3 | 95.00 | 100.00 | 96.00 | 0.0500 | |
| ≥ 4 | 85.00 | 100.00 | 88.00 | 0.1500 | |
| ≥ 6 | 75.00 | 100.00 | 80.00 | 0.2500 | |
| ≥ 7 | 40.00 | 100.00 | 52.00 | 0.6000 | |
| ≥ 8 | 35.00 | 100.00 | 48.00 | 0.6500 | |
| ≥ 9 | 15.00 | 100.00 | 32.00 | 0.8500 | |
| > 9 | 0.00 | 100.00 | 20.00 | 1.0000 | |
| Observations (n) | 25, training set | ||||
| ROC area (SE; 95% CI) | 0.9600 (0.0405; 0.88069–1.0000) |
LUS: lung ultrasound score; LR: likelihood ratio; ROC: receiver-operating characteristics curve
Discussion
The main finding of this study is that LUS score ≥ 3 in the ventral lung regions accurately excludes focal ARDS morphology.
Recently, it has been shown that LUS identifies patients with pulmonary edema [4] at risk of developing ARDS [8, 19, 20], especially if mechanically ventilated [21], and it notably correlates with changes in lung tissue aeration [9]. Our data showed that LUS reliably identifies ARDS morphology. In fact, exploring only four fields of non-dependent (ventral) lung regions, LUS score equal or higher than 3 accurately excludes lobar ARDS. This LUS application is clinical relevant, because patients with focal lung morphology at ZEEP are at risk of significant hyperinflation of their baby lung during recruitment maneuvers and the extent of lung recruitment is quite limited [2]. On the contrary, patients with diffuse aeration loss may benefit from recruitment maneuvers and higher PEEP levels especially in presence of life-threatening hypoxemia [22]. Bouhemad and colleagues showed a good correlation between pressure–volume curves and LUS assessment of PEEP induced recruitment. However, this association may imply over-inflation of well aerated regions [7]. In fact, mechanical ventilation with open lung strategy may not result in a homogeneous lung parenchyma, as recruited lung does not always reassume the elastic characteristics of normally aerated lung [23], thereby increasing the risk of hyperinflation of normally aerated alveoli. Moreover, Chiumello and colleague demonstrated that changes in LUS have not been associated with alveolar recruitment as demonstrated by lung CT analysis when the level of PEEP was increased from 5 to 15 cmH2O [9]. In fact, PEEP related changes of global LUS weakly correlated with lung CT decrease of not aerated tissue [9]. Our data may in part explain why changes in LUS score were not able to assess positive end expiratory pressure induced lung recruitment [9]. In fact, in focal ARDS morphology increasing positive end expiratory pressure from 5 to 15 may induce over-inflation of already open alveolar units without recruitment. Our findings allow us to speculate that LUS evaluation, confined to only four ventral lung regions, can help the clinician to evaluate the ARDS morphology at the bedside and be part of pre- and post-test probability to predict response to PEEP and lung recruitment maneuvers [24]. In fact, balancing risk and benefit of higher levels of PEEP in individualized cases is warranted [25]: Recently, the Lung Imaging for Ventilator Setting in ARDS (LIVE) study failed to demonstrate 90-day improvement mortality of ARDS patients who underwent personalized mechanical ventilation strategy based on radiographic phenotype (focal vs non-focal). However, 21% of the radiographic phenotypes were misclassified and only 34% of patients were actually classified using CT scans, with the remainder classified using chest X-ray [26]. In light of these results, the role of LUS in phenotyping ARDS patients need to be addressed in future clinical trials [27].
Some limitations of the current study should be addressed. First, different spatial resolution of CT and LUS may affect the evaluation of lung aeration. In fact, different from LUS, CT scan analysis encompasses the total area of interest along the pleural side of the chest wall and the inner boundary along the mediastinal organs. Second, the current study was performed at single center, then further external validation of identified cutoff to distinguish between lobar from diffuse/patchy ARDS morphology is warranted. Third, the current study design was observational; future studies are needed to evaluate response to PEEP based on ARDS morphology as defined by LUS. Finally, this study was performed before the Covid-19 pandemic and our cohort did not include patients with Covid-19 associated ARDS (CARDS). We speculate that our findings may apply also to CARDS patients; however, further studies are needed to address this issue.
Conclusions
In this cohort of patients with ARDS, LUS was a reliable bedside tool able to distinguish focal from non-focal morphologies. Using LUS in terms of pre-test probability to set ventilation strategy in individualized cases should be investigated further.
Supplementary Information
Additional file 1: Figure S1. Percentage of normally (blue boxes), poorly (red boxes) and not aerated (grey boxes) lung tissue at different LUS scores in Focal (panels A), and Non Focal (panel B) ARDS morphologies. *p<.05 Normal aerated tissue at LUS 0 vs 1, 2 and 3. #p<0.05 Poorly aerated tissue at LUS 0 vs 1, 2, and 3. °p<0.05 Not aerated tissue at LUS 0 vs 1, 2 and 3. Blu, red and grey box plots indicate normally, poorly and not aerated lung tissue.
Additional file 2. Supplementary material and methods and results.
Acknowledgements
The authors thank Prof. Michel Quintel who made available MALUNA software for lung CT images analysis.
Abbreviations
- ARDS
Acute respiratory distress syndrome
- CT
Computed tomography
- LUS
Lung ultrasound
- ICU
Intensive care unit
- LUSTOT
Total Lung Ultrasound Score
- LUSV
LUS score in the ventral lung regions
- LUSI
LUS score in the intermediate lung regions
- LUSD
LUS score in the dorsal lung regions
- HU
Hounsfield units
- Pair
Percentage of aerated tissue in regions of interest
- ROC
Receiver Operating Characteristic
Authors' contributions
AC: study design, data interpretation and analysis and manuscript writing and revision. EP: study design, data analysis and manuscript revision. AG, ATM, LS, LB: data interpretation and manuscript revision. IS, PA: data collection and analysis, manuscript revision. SV: study design, data collection and manuscript revision. OD: study design, manuscript revision. Ranieri VM: study design, data interpretation and manuscript revision. VF: study design, data analysis and interpretation, manuscript writing and revision. All the authors approved the final version of the manuscript to be published and agreed to be accountable for all aspects of the work. VF is the guarantor of the paper, taking responsibility for the integrity of the work as a whole, from inception to published article. All authors read and approved the final manuscript.
Funding
The authors received no funding for the submitted work.
Availability of data and materials
The datasets generated and/or analysed during the current study are not publicly available due to ethic and privacy statements but are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
The local Ethics Committee approved the study protocol (0117126) and written consent was obtained according to Italian regulation.
Consent for publication
Not applicable.
Competing interests
The authors report the absence of conflicts of interest related to the submitted work.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Thompson BT, Chambers RC, Liu KD. Acute respiratory distress syndrome. N Engl J Med. 2017;377(6):562–572. doi: 10.1056/NEJMra1608077. [DOI] [PubMed] [Google Scholar]
- 2.Constantin JM, Grasso S, Chanques G, Aufort S, Futier E, Sebbane M, et al. Lung morphology predicts response to recruitment maneuver in patients with acute respiratory distress syndrome. Crit Care Med. 2010;38(4):1108–1117. doi: 10.1097/CCM.0b013e3181d451ec. [DOI] [PubMed] [Google Scholar]
- 3.Gattinoni L, Caironi P, Cressoni M, Chiumello D, Ranieri VM, Quintel M, et al. Lung recruitment in patients with the acute respiratory distress syndrome. N Engl J Med. 2006;354(17):1775–1786. doi: 10.1056/NEJMoa052052. [DOI] [PubMed] [Google Scholar]
- 4.Lichtenstein DA, Meziere GA. Relevance of lung ultrasound in the diagnosis of acute respiratory failure: the BLUE protocol. Chest. 2008;134(1):117–125. doi: 10.1378/chest.07-2800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bouhemad B, Liu ZH, Arbelot C, Zhang M, Ferarri F, Le-Guen M, et al. Ultrasound assessment of antibiotic-induced pulmonary reaeration in ventilator-associated pneumonia. Crit Care Med. 2010;38(1):84–92. doi: 10.1097/CCM.0b013e3181b08cdb. [DOI] [PubMed] [Google Scholar]
- 6.Via G, Lichtenstein D, Mojoli F, Rodi G, Neri L, Storti E, et al. Whole lung lavage: a unique model for ultrasound assessment of lung aeration changes. Intensive Care Med. 2010;36(6):999–1007. doi: 10.1007/s00134-010-1834-4. [DOI] [PubMed] [Google Scholar]
- 7.Bouhemad B, Brisson H, Le-Guen M, Arbelot C, Lu Q, Rouby JJ. Bedside ultrasound assessment of positive end-expiratory pressure-induced lung recruitment. Am J Respir Crit Care Med. 2011;183(3):341–347. doi: 10.1164/rccm.201003-0369OC. [DOI] [PubMed] [Google Scholar]
- 8.Baldi G, Gargani L, Abramo A, D'Errico L, Caramella D, Picano E, et al. Lung water assessment by lung ultrasonography in intensive care: a pilot study. Intensive Care Med. 2013;39(1):74–84. doi: 10.1007/s00134-012-2694-x. [DOI] [PubMed] [Google Scholar]
- 9.Chiumello D, Mongodi S, Algieri I, Vergani GL, Orlando A, Via G, et al. Assessment of Lung Aeration and Recruitment by CT Scan and Ultrasound in Acute Respiratory Distress Syndrome Patients. Crit Care Med. 2018. [DOI] [PubMed]
- 10.Force ADT, Ranieri VM, Rubenfeld GD, Thompson BT, Ferguson ND, Caldwell E, et al. Acute respiratory distress syndrome: the Berlin definition. JAMA. 2012;307(23):2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 11.Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, et al. International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med. 2012;38(4):577–591. doi: 10.1007/s00134-012-2513-4. [DOI] [PubMed] [Google Scholar]
- 12.Mongodi S, Bouhemad B, Orlando A, Stella A, Tavazzi G, Via G, et al. Modified lung ultrasound score for assessing and monitoring pulmonary aeration. Ultraschall Med. 2017;38(5):530–537. doi: 10.1055/s-0042-120260. [DOI] [PubMed] [Google Scholar]
- 13.Soummer A, Perbet S, Brisson H, Arbelot C, Constantin JM, Lu Q, et al. Ultrasound assessment of lung aeration loss during a successful weaning trial predicts postextubation distress*. Crit Care Med. 2012;40(7):2064–2072. doi: 10.1097/CCM.0b013e31824e68ae. [DOI] [PubMed] [Google Scholar]
- 14.Lichtenstein DA, Meziere GA, Lagoueyte JF, Biderman P, Goldstein I, Gepner A. A-lines and B-lines: lung ultrasound as a bedside tool for predicting pulmonary artery occlusion pressure in the critically ill. Chest. 2009;136(4):1014–1020. doi: 10.1378/chest.09-0001. [DOI] [PubMed] [Google Scholar]
- 15.Tierney DM, Huelster JS, Overgaard JD, Plunkett MB, Boland LL, St Hill CA, et al. Comparative performance of pulmonary ultrasound, chest radiograph, and CT among patients with acute respiratory failure. Crit Care Med. 2020;48(2):151–157. doi: 10.1097/CCM.0000000000004124. [DOI] [PubMed] [Google Scholar]
- 16.Terragni PP, Rosboch G, Tealdi A, Corno E, Menaldo E, Davini O, et al. Tidal hyperinflation during low tidal volume ventilation in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2007;175(2):160–166. doi: 10.1164/rccm.200607-915OC. [DOI] [PubMed] [Google Scholar]
- 17.Grasso S, Terragni P, Mascia L, Fanelli V, Quintel M, Herrmann P, et al. Airway pressure-time curve profile (stress index) detects tidal recruitment/hyperinflation in experimental acute lung injury. Crit Care Med. 2004;32(4):1018–1027. doi: 10.1097/01.CCM.0000120059.94009.AD. [DOI] [PubMed] [Google Scholar]
- 18.Puybasset L, Cluzel P, Gusman P, Grenier P, Preteux F, Rouby JJ. Regional distribution of gas and tissue in acute respiratory distress syndrome. I. Consequences for lung morphology. CT Scan ARDS Study Group. Intensive Care Med. 2000;26(7):857–69. doi: 10.1007/s001340051274. [DOI] [PubMed] [Google Scholar]
- 19.Leblanc D, Bouvet C, Degiovanni F, Nedelcu C, Bouhours G, Rineau E, et al. Early lung ultrasonography predicts the occurrence of acute respiratory distress syndrome in blunt trauma patients. Intensive Care Med. 2014;40(10):1468–1474. doi: 10.1007/s00134-014-3382-9. [DOI] [PubMed] [Google Scholar]
- 20.Copetti R, Soldati G, Copetti P. Chest sonography: a useful tool to differentiate acute cardiogenic pulmonary edema from acute respiratory distress syndrome. Cardiovasc Ultrasound. 2008;6:16. doi: 10.1186/1476-7120-6-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Pisani L, Vercesi V, van Tongeren PSI, Lagrand WK, Leopold SJ, Huson MAM, et al. The diagnostic accuracy for ARDS of global versus regional lung ultrasound scores - a post hoc analysis of an observational study in invasively ventilated ICU patients. Intensive Care Med Exp. 2019;7(Suppl 1):44. doi: 10.1186/s40635-019-0241-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Briel M, Meade M, Mercat A, Brower RG, Talmor D, Walter SD, et al. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA. 2010;303(9):865–73. doi: 10.1001/jama.2010.218. [DOI] [PubMed] [Google Scholar]
- 23.Grasso S, Stripoli T, Sacchi M, Trerotoli P, Staffieri F, Franchini D, et al. Inhomogeneity of lung parenchyma during the open lung strategy: a computed tomography scan study. Am J Respir Crit Care Med. 2009;180(5):415–423. doi: 10.1164/rccm.200901-0156OC. [DOI] [PubMed] [Google Scholar]
- 24.Rouby JJ, Lu Q, Goldstein I. Selecting the right level of positive end-expiratory pressure in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2002;165(8):1182–1186. doi: 10.1164/ajrccm.165.8.2105122. [DOI] [PubMed] [Google Scholar]
- 25.Xirouchaki N, Kondili E, Prinianakis G, Malliotakis P, Georgopoulos D. Impact of lung ultrasound on clinical decision making in critically ill patients. Intensive Care Med. 2014;40(1):57–65. doi: 10.1007/s00134-013-3133-3. [DOI] [PubMed] [Google Scholar]
- 26.Constantin JM, Jabaudon M, Lefrant JY, Jaber S, Quenot JP, Langeron O, et al. Personalised mechanical ventilation tailored to lung morphology versus low positive end-expiratory pressure for patients with acute respiratory distress syndrome in France (the LIVE study): a multicentre, single-blind, randomised controlled trial. Lancet Respir Med. 2019;7(10):870–880. doi: 10.1016/S2213-2600(19)30138-9. [DOI] [PubMed] [Google Scholar]
- 27.Mongodi S, Santangelo E, Bouhemad B, Vaschetto R, Mojoli F. Personalised mechanical ventilation in acute respiratory distress syndrome: the right idea with the wrong tools? Lancet Respir Med. 2019;7(12):e38. doi: 10.1016/S2213-2600(19)30353-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Figure S1. Percentage of normally (blue boxes), poorly (red boxes) and not aerated (grey boxes) lung tissue at different LUS scores in Focal (panels A), and Non Focal (panel B) ARDS morphologies. *p<.05 Normal aerated tissue at LUS 0 vs 1, 2 and 3. #p<0.05 Poorly aerated tissue at LUS 0 vs 1, 2, and 3. °p<0.05 Not aerated tissue at LUS 0 vs 1, 2 and 3. Blu, red and grey box plots indicate normally, poorly and not aerated lung tissue.
Additional file 2. Supplementary material and methods and results.
Data Availability Statement
The datasets generated and/or analysed during the current study are not publicly available due to ethic and privacy statements but are available from the corresponding author on reasonable request.