Abstract
Chronic, excessive alcohol consumption is associated with cerebrovascular hypoperfusion, which has the potential to interfere with cognitive processes. Magnetic resonance pulsed continuous arterial spin labeling (PCASL) provides a noninvasive approach for measuring regional cerebral blood flow (CBF) and was used to study 24 men and women with Alcohol Use Disorder (AUD) and 20 age- and sex-matched controls. Two analysis approaches tested group differences: a data-driven, regionally-free method to test for group differences on a voxel-by-voxel basis and a region of interest (ROI) approach, which focused quantification on atlas-determined brain structures. Whole-brain, voxel-wise quantification identified low AUD-related cerebral perfusion in large volumes of medial frontal and cingulate cortices. The ROI analysis also identified lower CBF in the AUD group relative to the control group in medial frontal, anterior/middle cingulate, insular, and hippocampal/amygdala ROIs. Further, years of AUD diagnosis negatively correlated with temporal cortical CBF, and scores on an alcohol withdrawal scale negatively correlated with posterior cingulate and occipital gray matter CBF. Regional volume deficits did not account for AUD CBF deficits. Functional relevance of attenuated regional CBF in the AUD group emerged with positive correlations between episodic working memory test scores and anterior/middle cingulum, insula, and thalamus CBF. The frontolimbic and insular cortical neuroconstellation with dampened perfusion suggests a mechanism of dysfunction associated with these brain regions in AUD.
Keywords: alcohol, working memory, perfusion, cerebral blood flow, ASL
1. Introduction
Brain function is tightly linked to regional perfusion of oxygenated blood, the basis of the brain activation detectable with functional MRI (Ogawa et al., 1990; Raichle, 1998). Cerebral blood perfusion can be measured noninvasively with arterial spin labeling (ASL), an MRI-based method for “labeling” oxygenated blood as it flows from the level of the carotid arteries into the brain (Alsop et al., 2010; Buxton, 2005; Hernandez-Garcia et al., 2019). Pulsed continuous ASL (PCASL) enables tracing blood flow volumetrically (Borogovac and Asllani, 2012; Detre et al., 2009) throughout brain tissue (Dai et al., 2008; Detre and Alsop, 1999). With PCASL, brain damage in the form of attenuated blood perfusion has been localized in stroke (Wang et al., 2020), dementias of different etiologies (Mattsson et al., 2014; Thomas et al., 2019), and neuropsychiatric disorders affecting widespread brain regions, including Alcohol Use Disorder (AUD) (Clark et al., 2007; Murray et al., 2015; Sullivan et al., 2013). Regional hypoperfusion of cerebral blood flow (CBF) has also been related to impaired performance on cognitive testing in AUD (Murray et al., 2015; Sullivan et al., 2013).
Both acute and chronic exposure to high doses of alcohol affects widespread brain regions. Rather than being diffuse, the effects involve selective, frontally-distributed neural systems (e.g.,Sullivan et al., 2018) (reviews,Oscar-Berman et al., 2014; Zahr et al., 2017). Consistent with this characterization, ASL studies of acute alcohol administration report that hyperperfusion is strongest in anterior brain regions especially frontal cortex (Gundersen et al., 2013; Kong et al., 2019; Marxen et al., 2014; Tanabe et al., 2019). Although acute alcohol administration and chronic AUD both selectively affect the frontal cortex, chronic AUD results in hypoperfusion (Clark et al., 2007; Sullivan et al., 2013), whereas acute infusion results in hyperperfusion (Durazzo et al., 2010). Further, common comorbidities of tobacco smoking and non-alcohol drug misuse modulate the role of dependent alcohol consumption on regional CBF and impede recovery in abstinent individuals with AUD (Durazzo et al., 2010; Mon et al., 2009); nonetheless, the alcohol effect appears salient (Murray et al., 2015).
AUD can also affect the insula (Sullivan et al., 2013), which is a hub of the salience network associated with attentional preparation (Seeley et al., 2007), concept and network switching (Menon and Uddin, 2010), and development of addiction (Lasek et al., 2018; Naqvi and Bechara, 2010). The salience network has an anterior distribution (Menon et al., 2020) and includes the anterior insula, anterior cingulate cortex, and ventral striatum. Nodes of this network contribute to the development and maintenance of addiction through processes involving reward, motivation (Koob and Volkow, 2016), and distorted self-awareness (Le Berre et al., 2017). That this constellation of brain structures is a target for excessive alcohol exposure causing chronic attenuation of cerebral perfusion has implications for explaining AUD as a self-perpetuating disorder (Halcomb et al., 2019; Sullivan and Pfefferbaum, 2019).
Here, we aimed to replicate and extend our earlier PCASL findings of AUD-related insular CBF deficits (Sullivan et al., 2013) with new and larger AUD and control groups and to replicate other studies reporting dampened CBF in prefrontal cortex in AUD (Clark et al., 2007; Durazzo et al., 2010; Murray et al., 2015). We used two analysis approaches to seek group differences: a traditional region of interest (ROI) analysis focusing on atlas-determined brain structures and a data-driven, region-free method to test for group differences on a voxel-by-voxel basis (Zhao et al., 2018). We expected to identify CBF deficits in the insula and prefrontal cortex of the AUD group. Further, we hypothesized that with a larger sample, CBF deficits would be detectable in the anterior cingulate and hippocampal/amygdala regions, which had previously shown only trend-level deficits (Sullivan et al., 2013). Exploratory analyses sought group differences in the division of the insula into anterior and posterior regions (Deen et al., 2011; Menon et al., 2020) and tested relations between clinical and cognitive measures and regional CBF in the AUD group.
2. Methods
2.1. Participants
The participants engaged in our earlier MRI studies on quantification of thalamic nuclei (Zahr et al., 2020), hippocampal subfields (Zahr et al., 2019), and cerebellar lobules (Sullivan et al., 2019). Descriptions of the groups appear in the previous publications and are summarized herein (Table 1). Viable PCASL data were available for age- and sex-matched 24 alcoholics (17 men, 7 women) and 20 controls (13 men, 7 women), who gave written informed consent for study. Participants received a stipend of $200 for completing the study, which was conducted with the approval of the Institutional Review Boards of Stanford University and SRI International. The work was conducted in accordance with The Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans.
Table 1.
Characteristics of the study groups: mean±SD / frequency count
| Control (n=20) | AUD (n=24) | p-value* | |
|---|---|---|---|
| N (men/women) | 13 / 7 | 17 / 7 | 0.68 |
| Age (years) | 54.1±9.3 | 53.7±8.8 | 0.90 |
| Handedness (Right/Left) | 19 / 1 | 20 / 4 | 0.23 |
| Ethnicity▯(Caucasian/African American/Asian) | 8 / 5 / 7 | 14 / 9 / 1 | 0.03 |
| Body Mass Index | 25.0±3.6 | 28.9±5.7 | 0.01 |
| Education (years) | 16.2±2.3 | 13.0±1.8 | 0.0001 |
| Socioeconomic Status# | 25.4±15.7 | 44.5±13.2 | 0.0001 |
| Global Assessment of Function (GAF) | 87.3±4.2 | 65.0±9.2 | 0.0001 |
| Beck Depression Index (BDI) | 5.2±5.2 | 14.7±10.0 | 0.001 |
| AUD onset age | - | 21.7±6.6 | n.a. |
| Days since last drink | - | 105.7±94.5 | n.a. |
| Lifetime alcohol consumption (kg) | 53.6±81.2 | 1798.3±1638.0 | <0.0001 |
| Alcohol consumption years | - | 32.0±9.6 | n.a. |
| CIWA score | - | 23.2±17.7 | n.a. |
| Smoker (never/past/current) | 18 / 0 / 2 | 4/6/14 | 0.0001 |
| Nicotine (daily) | 1.1±3.3 | 4.6±3.9 | 0.008 |
| WTAR IQ† | 102.3±11.2 | 96.3±10.2 | 0.0842 |
| Cognitive Test Composite Scores: Age-corrected Z-scores | |||
| Executive Function | −.002±0.70 | −1.18±0.75 | 0.0001 |
| Attention and Working Memory | −.001±1.00 | −0.78±1.02 | 0.0168 |
| Memory and Learning | −0.24±0.65 | −1.10±0.88 | 0.0001 |
| Visuospatial Abilities | 0.001±0.73 | −1.63±1.29 | 0.0001 |
t-tests used on continuous variables (e.g., age); |2 used on nominal variables (e.g., handedness)
self-defined
lower score = higher status
WTAR IQ = Wechsler Test of Adult Reading Intelligence Quotient
bold = statistically significant at p≤.05 (2-tailed)
Participants underwent Structured Clinical Interviews for DSM-IV revised (First, 2000) to determine diagnosis for alcohol dependence and DSM-5 diagnosis for AUD (American Psychiatric Association, 2013). Interviews, conducted by laboratory research clinicians, included structured health questionnaires, a semi-structured timeline follow-back interview to quantify lifetime alcohol consumption (Skinner, 1982; Skinner and Sheu, 1982), and the Clinical Institute Withdrawal Assessment for Alcohol-revised (CIWA-Ar; higher scores indicate more symptoms; maximum score=67) (Sullivan et al., 1989). Comorbid drug use was determined by SCID diagnosis; allowable drug use was secondary to alcohol use and typically more remote than alcohol use. Subjects were excluded for history of significant medical (e.g., HIV infection, epilepsy, stroke, multiple sclerosis, uncontrolled diabetes, or loss of consciousness > 30 minutes), psychiatric (i.e., schizophrenia, bipolar I disorder, or PTSD), or neurological (e.g., neurodegenerative disease) disorders.
AUD participants were recruited from post-detoxification, sober-living environments. Abstinence was confirmed on the days of testing with an alcohol breathalyzer, which required a 0.0 breath alcohol level (BrAL) to proceed with testing. Anyone whose BrAL exceeded 0.0 was asked to return when abstinent at a minimum of overnight. Compared with the control group, the AUD group had fewer years of education, lower socioeconomic status, lower Global Assessment of Functioning (GAF) scores, greater body mass index (BMI), more cigarette smokers, and greater lifetime alcohol consumption (Table 1). All participants were asked to refrain from smoking and from drinking caffeinated beverages for at least 1 hour before scanning.
2.2. Cognitive Tests
The AUD group achieved lower scores than the control group on the Wechsler Test of Adult Reading (WTAR) (Wechsler, 2001) for general cognitive ability. Participants also completed a battery of neuropsychological tests that were aggregated into composite scores representing four functional domains: Executive Functions comprising FAS word fluency (Borkowski et al., 1967), semantic fluency, WAIS digit symbol standard score and time to complete the full grid (Wechsler, 1981), Montreal Cognitive Assessment test (MoCA) (Nasreddine et al., 2005) abstraction score; Attention and Working Memory, comprising the Wechsler Memory Scale-Revised (WMS-R) spans (Wechsler, 1987); Memory and Learning, comprising incidental recall of symbols or digits in the Digit-Symbol (Wechsler, 1981) or Symbol-Digit coding test, short and long delay free recall scores of the California Verbal Learning Test (Delis et al., 1987), delayed recall of the MoCA, and the WMS-R logical memory stories; Visuospatial Abilities comprising the Rey-Osterrieth complex figure copy (Rey, 1942) and WMS-R visual reproduction items. Mean±SD composite scores by group are in Table 1; description of composite construction and distributions of the scores by group were published previously (Zahr et al., 2019).
2.3. MRI Acquisition Parameters
MRI structural data were acquired in the sagittal plane on a 3T General Electric (GE) Discovery MR750 (GE Healthcare Systems, Waukesha, MI) with an Array Spatial Sensitivity Encoding Technique (ASSET) for parallel and accelerated imaging with a 32-channel head coil (Nova Medical, Wilmington, MA) (Zahr et al., 2019).
T1-weighted scans were cerebral spinal fluid (CSF)-nulled Magnetization Prepared Rapid Gradient echo (MPRAGE) acquired with the following parameters: TR=8ms, TE=3.5ms, TI=1100ms, TS (i.e., time between two successive inversion pulses)=3.0s, Flip Angle=9, FOV=18cm, matrix=200×200, thick=1mm, slices= 210 slices, resolution=0.9×0.9×1.0. T2-weighted scans were 3D fast spin-echo (FSE) with variable refocusing flip angle (T2 Cube) and acquired with the following parameters: FatSat=ON, TR=3500ms, Effective TE=62ms, Echo Train Length (ETL)=84, FOV=18 cm, matrix=224×224, thick=1mm, slices=210, resolution=0.8x.08×1.0. Acquisition extended from ear to ear and from the top of the scalp to the base of the cerebellum. The T1-weighted scans yielded gray/white tissue conspicuity, and the T2 scans provided information about brain volume, especially when tissue shrinkage is replaced by CSF.
2.4. Pulsed-Continuous Arterial Spin Labelling (PCASL)
Whole brain regional CBF acquired using PCASL as previously described (Sullivan et al., 2013) but with a 32-channel head coil. Scanning parameters were as follows: TR=4844ms, TE=10.536ms, thick=4mm, skip=0mm, locations=36, field of view=240mm; 8 spiral interleaves with 512 samples per interleave; flip angle=111°, labeling duration=1.5s, post-labeling delay=2.025s.
CBF maps were derived based on a two-compartment model with finite labeling duration. CBF images were nonrigidly aligned to each subject’s T2-weighted FSE image that was in turn registered to the SRI24 atlas (Rohlfing et al., 2010) using symmetric, diffeomorphic nonrigid registration (ANTS) (Avants et al., 2008). This nonrigid alignment resampled CBF maps of all subjects to 1×1×1 mm resolution without applying any smoothing and put all subjects in to a common atlas space. Using all brain voxels (gray matter, white matter, and CSF), CBF maps were constructed by normalizing the CBF value at each voxel by the standardized z-score across all voxels. Resulting z-scores for any ROI for each subject can thus be above or below 0. In other words, CBF maps were constructed for each individual by expressing each voxel in his or her brain as a z-score based on the mean and standard deviation of all of his or her brain voxels: xz = ( x – whole brain mean)/whole brain standard deviation. For any individual, the mean value of all brain voxels would be 0. For a given ROI the mean value might be greater (have a positive z-score) or smaller (have a negative -score) than the whole brain.
Two analysis methods compared CBF of the AUD and control group CBF: a data-driven, voxel-wise permutation cluster analysis and an hypothesis-driven, ROI-based, parcellation analysis. For the data-driven cluster analysis, the left hemisphere of the registered CBF maps was flipped and added to the right hemisphere. With the assumption that the AUD group would have lower CBF than the control group, CBF of each participant was tested on each voxel in the right hemisphere by a general linear model (GLM) analysis implemented in FSL’s Permutation Analysis of Linear Models (PALM) (Winkler et al., 2014) with 5,000 permutations. Covariates of GLM were age, sex, and group. The resulting one-tailed, voxel-wise p-values associated with the AUD group were corrected for spatial coherence by FSL Threshold-Free Cluster Enhancement (TFCE) (Smith and Nichols, 2009) and for family-wise error at the 5% level.
The ROI-based, parcellation analysis measured CBF in 12 bilateral cortical and subcortical ROIs. CBF calculation was based on gray matter voxels for cortical, cingulate, and insular regions and total tissue (gray+white matter) for subcortical regions (caudate, putamen, globus pallidum, thalamus, hippocampus, and cerebellum).
2.5. Statistical analysis
Statistical analysis was performed using R 3.5.1 (R Core Team, 2019) [htpp://www.r-project.org/]. The parcellation analysis used separate linear models (lm) predicting group differences of CBF in each region from age, sex, and the gray matter or tissue volume comprising that region. Group differences were determined by t-tests, which were considered significant at p=0.05 (2-tailed). Exploratory analyses seeking relations between clinical or performance measures and regional CBF used Pearson product-moment correlations or nonparametric Spearman tests for measures that were non-normally distributed; correlations with nominal p-values of 0.05 (uncorrected) are reported.
3. Results
3.1. Group differences in regional CBF: Voxel-wise permutation cluster analysis
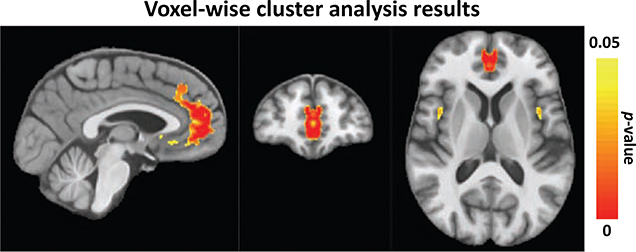
The AUD participants had significantly lower CBF than the control participants (one-tailed corrected p ≤ 0.05) in 6,124 voxels of the medial frontal region. Overlaying this region with the SRI24 brain atlas revealed that it comprised 2,588 voxels of 35,214 voxels (7.4%) in the superior medial frontal cortex, 2,108 of 10564 (20.0%) voxels in the medial orbital frontal cortex, and 1,428 of 15,756 voxels (9.1%) in the anterior cingulum. Additional CBF deficits were detected in 126 voxels of the operculum, which composed 64 of 15,676 voxels (0.4%) in inferior frontal and 62 of 22,228 voxels (0.3%) in Rolandic regions; 76 of 4,918 voxels (1.5%) in the olfactory bulb were also CBF-deficient in the AUD group. In the insula, 292 of 22,516 voxels (1.3%) had lower CBF in the AUD than control participants (Fig. 1).
Figure 1.
Sagittal, coronal, and axial views of the T1-weighted averaged MRI with regions showing voxels with significantly lower CBF in the AUD than control.
3.2. Group differences in regional CBF: Cortical parcellation approach
The mean±SD for each of the 12 ROIs by group and the t-test results appear in Table 2, and scatterplots are in Fig. 2. The parcellation GLM controlling for age and sex revealed lower CBF in the AUD than control group in the medial frontal (t=2.10, p=.0417), insular (t=3.21, p=.0025), and anterior-middle cingulate (t=2.34, p=.0243) cortices and hippocampus/amygdala (t=2.07, p=.0465). AUD deficits were present in both the anterior (t=2.93, p=.0055) and posterior (t=3.075, p=.0037) insula. Bonferroni correction for directional hypotheses (AUD<control) required p≤0.008, which was met for the total and anterior/posterior divisional insular measures. Although the differences in the medial frontal, anterior-middle cingulate, and hippocampus/amygdala in favor of controls were p<0.05, they were not significant after Bonferroni correction.
Table 2.
Mean±SD CBF Z-score for each ROI by group and lm results
| Control (N=20) | AUD (N=24) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| ROI | Mean | SD | Mean | SD | df | t | p | ||
| Lateral frontal | 0.368 | 0.123 | 0.375 | 0.143 | 41.933 | −0.168 | 0.8673 | ||
| Medial frontal | 0.493 | 0.221 | 0.348 | 0.237 | 41.416 | 2.101 | 0.0417 | ||
| Temporal | 0.390 | 0.118 | 0.359 | 0.139 | 41.969 | 0.785 | 0.4370 | ||
| Parietal | 0.128 | 0.150 | 0.237 | 0.205 | 41.387 | −2.017 | 0.0502 | ||
| Occipital | 0.458 | 0.363 | 0.470 | 0.320 | 38.267 | −0.120 | 0.9054 | ||
| Anterior-middle cingulate | 0.983 | 0.132 | 0.868 | 0.192 | 40.721 | 2.340 | 0.0243 | ||
| Posterior cingulate | 1.346 | 0.490 | 1.280 | 0.370 | 34.851 | 0.498 | 0.6219 | ||
| Hippocampus/amygdala | 0.010 | 0.096 | −0.087 | 0.202 | 34.056 | 2.066 | 0.0465 | ||
| Insula | 0.637 | 0.187 | 0.441 | 0.217 | 41.928 | 3.213 | 0.0025 | ||
| Anterior | 0.651 | 0.208 | 0.447 | 0.254 | 41.993 | 2.929 | 0.0055 | ||
| Posterior | 0.619 | 0.178 | 0.434 | 0.221 | 41.959 | 3.075 | 0.0037 | ||
| Caudate/putamen | −0.082 | 0.234 | −0.175 | 0.348 | 40.346 | 1.056 | 0.2975 | ||
| Globus pallidus | −0.332 | 0.277 | −0.428 | 0.321 | 41.941 | 1.060 | 0.2951 | ||
| Thalamus | 0.069 | 0.206 | 0.106 | 0.219 | 41.339 | −0.579 | 0.5656 | ||
bold = control > AUD at p ≤ 0.05 level
Bonferroni correction for directional hypothesis (AUD < controls), p≤0.008.
Figure 2.
Top: Axial images with regions of interest (ROIs) showing significant AUD<control group differences outlined in color: red=medial frontal; blue=anterior-middle cingulate; green=insula; black=hippocampal/amygdala. Bottom: Violin plots of control (gray) and AUD (black) regional CBF values.
Quantification of gray matter or total tissue volume in each ROI used in the CBF analysis indicated smaller volumes in the AUD than control group in all regions but the caudate/putamen, although the differences were significant at p<.05 (uncorrected for multiple comparisons) for 5 ROIs: medial frontal, temporal, parietal, hippocampus/amygdala, and thalamus (Table 3). Of the four ROIs with CBF deficits in the AUD group, volume deficits were present in two ROIs: medial frontal and hippocampus/amygdala. For all CBF group comparisons, volume was a covariate in the lm analyses and thus statistically adjusted for such differences. As a check on the effectiveness of the model to account for volume differences that could influence CBF, we sought correlations between CBF and the underlying volume for each ROI in the AUD group and found that none approached significance.
Table 3.
Mean±SD volume (cc) for each ROI by group and t-test results
| Control (N=20) | AUD (N=24) | Group Difference | ||||||
|---|---|---|---|---|---|---|---|---|
| ROI | Mean | SD | Mean | SD | t | p | ||
| Lateral frontal | 120.47 | 12.08 | 114.66 | 10.66 | 1.690 | 0.0984 | ||
| Medial frontal | 27.51 | 2.86 | 25.50 | 2.20 | 2.628 | 0.0119 | ||
| Temporal | 95.09 | 9.56 | 88.65 | 8.53 | 2.359 | 0.0231 | ||
| Parietal | 54.86 | 7.01 | 49.51 | 4.77 | 3.005 | 0.0045 | ||
| Occipital | 51.27 | 6.03 | 48.36 | 5.91 | 1.616 | 0.1135 | ||
| Anterior-middle cingulate | 18.97 | 2.35 | 17.93 | 1.93 | 1.622 | 0.1123 | ||
| Posterior cingulate | 1.49 | 0.35 | 1.43 | 0.22 | 0.761 | 0.4509 | ||
| Hippocampus/amygdala | 24.61 | 2.22 | 22.73 | 1.87 | 3.051 | 0.0039 | ||
| Insula | 12.62 | 1.32 | 12.32 | 1.33 | 0.748 | 0.4585 | ||
| Caudate/putamen | 18.55 | 1.72 | 18.57 | 2.00 | −0.039 | 0.9689 | ||
| Globus pallidus | 3.00 | 0.39 | 2.88 | 0.33 | 1.067 | 0.2920 | ||
| Thalamus | 12.05 | 1.07 | 11.39 | 0.90 | 2.241 | 0.0304 | ||
bold = control > AUD at p ≤ 0.05 level
Bonferroni correction for directional hypothesis (AUD < controls), p≤0.008.
Another factor that could influence CBF is smoking. Only 2 of the 20 control participants were smokers, whereas 20 of the 24 AUD participants were smokers, thereby precluding a direct diagnosis-by-smoking comparison. Nonetheless, we tested CBF differences between smokers and non-smokers within the AUD group and found that in 8 of the 12 regions, CBF was higher in smokers than non-smokers and that these differences were significant with Mann-Whitney nonparametric tests (p<0.02) for temporal and parietal cortices and showed a trend for the medial frontal region (p=0.0528). These AUD-smoking subgroups did not differ in age, years of education SES, amount of alcohol drunk in their lifetimes, days since last drink, or CIWA-Ar history scores.
3.3. Alcohol history and performance correlations with regional CBF
Exploratory analyses were limited to relations in the AUD group and revealed several modest correlations. For alcohol history (Fig. 3), higher CIWA-Ar scores (i.e., more withdrawal signs) correlated with lower CBF in the occipital (r=−.442, p=0.0304) and posterior cingulate (r=−.462, p=0.0232) regions. Further, the more years a participant met diagnostic criteria for AUD, the lower the temporal lobe CBF (r=−.512, p=0.0105). Although the AUD group had, on average, a higher index of depressive symptoms than the control group, that index did not correlate significantly with any rCBF measure; the highest correlation was for the medial frontal CBF but was not significant (r=−.268, p=.206).
Figure 3.
Correlations between alcohol history variables and regional CBF.
Regarding cognitive performance (Fig. 4), lower Attention and Working Memory scores correlated with lower CFB in the insula (r=.410, p=.047) and thalamus (r=.501, p=0.0126) and with the anterior/middle cingulate CFB at a trend level (r=.37, p=0.0712). A multiple regression entering insula, thalamus, and anterior/middle cingulate CBF as simultaneous predictors of Attention and Working Memory scores accounted for 33% of the adjusted variance (p=0.011). In addition, Memory and Learning scores were positively correlated with anterior/middle cingulate CBF (r=.404, p=.05).
Figure 4.
Correlations between Working Memory or Memory and Learning composite scores and regional CBF.
4. Discussion
Both the data-driven and the ROI analyses replicated and extended findings of attenuated CBF in the prefrontal (Clark et al., 2007) and insular cortices (Sullivan et al., 2013) of the AUD relative to the control group. Comparison of CBF across ROIs was enabled by using normalized perfusion values (Pfefferbaum et al., 2011; Pfefferbaum et al., 2010). The ROI analysis also identified lower CBF in the anterior/middle cingulate cortex and hippocampal/amygdala complex of the AUD group compared with the control group.
4.1. Regions and functions implicated
The normalized regional CBF observed herein was similar in pattern to that observed in our earlier studies of healthy control participants thereby demonstrating consistency and reliability (Pfefferbaum et al., 2011; Pfefferbaum et al., 2010). CBF was highest in the posterior cingulate cortex, possibly because of its proximity to major anterior cerebral artery blood supplies, and lowest in the globus pallidus, possibly related to the presence of iron (Hallgren and Sourander, 1958), which can result in T1 shortening (Bartzokis et al., 2009; Pfefferbaum et al., 2009) and low CBF (cf.,Rodrigue et al., 2020).
Regions showing low AUD-related cerebral perfusion were large volumes of medial frontal and cingulate cortices visible with voxel-wise quantification and the anterior and posterior insular cortex and medial temporal structures detected with ROI analysis. Correlational analysis provided indirect assurance of the relevance of AUD to regional CBF metrics. Specifically, more years with an AUD diagnosis correlated with lower temporal cortical CBF, and higher scores on the CIWA-Ar scale, indicating history of more withdrawal signs correlated with lower posterior cingulate and occipital CBF. Suggestion for the functional relevance of attenuated regional CBF in AUD emerged with the modest positive relations between episodic working memory test scores and CBF in anterior-middle cingulum, insula, and thalamus, which are regions known to contribute to working memory processes (Mencarelli et al., 2019). This constellation of factors—withdrawal signs, frontocingulate and insular perfusion, and executive functions—converges on findings from several recent studies of AUD using multiple imaging modalities. Specifically, compromised executive functioning occurred in the presence of low gray matter density of frontal and anterior insular cortices (Galandra et al., 2018), poor frontostriatal connectivity determined with resting-state fMRI (Galandra et al., 2019), compromised connectivity among the central executive, salience, and default mode networks (Crespi et al., 2020; Crespi et al., 2019), and mild to moderate alcohol withdrawal signs as predictors of deficits in frontal, insular, and thalamic volumes (Laniepce et al., 2020). The perfusion deficit in these brain regions noted herein and in other studies gives rise to the speculation that local blood hypoperfusion could be a mechanism contributing to the attenuated BOLD effect of fMRI and tissue degradation of the above-reviewed reports.
4.2. Pathophysiology of perfusion deficits
Cerebral blood is supplied by three main sources: anterior cerebral artery, which feeds frontal superior and medial cortices; posterior communicating artery, which includes the circle of Willis and other anterior-to-posterior circulation; and the middle cerebral artery, which extends to the basal ganglia, insula and operculum, anterior (Broca’s area) and parietal (Wernicke’s area) cortices, and lateral and medial temporal regions. The regions exhibiting the greatest perfusion differences between AUD and controls include targets of the anterior and middle arterial feeders. The large medial frontal and anterior/middle cingulate regions of low AUD-related perfusion visualized with the voxel-based analysis likely reflects compromised blood flow of the anterior arteries, whereas the low perfusion of the insula and medial temporal regions may arise from the compromised blood flow of the middle cerebral artery. An anterior pattern of perfusion dampening in AUD was also reported in earlier studies using ASL (Clark et al., 2007; Durazzo et al., 2010), (18)fluorodeoxyglucose Positron Emission Tomography (FDG PET) (Gilman et al., 1990; Ritz et al., 2016), and Single Photon Emission Computed Tomography (SPECT) (Demir et al., 2002; Gansler et al., 2000; Nicolas et al., 1993) especially during alcohol withdrawal (SPECT: Tutus et al., 1998). One potential metabolic mechanism for the AUD-related CBF difference may be a deficit in brain glucose delivery and metabolism (cf., Tanabe et al., 2019). This speculation is supported by studies using FDG PET, which reported deficits in glucose metabolism in similar brain regions of heavy drinkers (thalamus, frontal and temporal cortices) (Volkow et al., 2015) and those with AUD [nodes of Papez circuit (Ritz et al., 2016); frontal cortex (Gilman et al., 1990)].
A more tenuous yet intriguing speculation derives from a study of mice in a repeated binge paradigm of “drinking-in the dark” for one week or six weeks (Chen et al., 2015). That study focused on the alcohol-related development of perineuronal nets (PNNs), which are extracellular matrix structures typically embracing GABA-ergic neurons that regulate learning, memory, and addiction (Arnsten, 2013; Lasek et al., 2018) and form a basis for neuronal plasticity. The mice with the longer, more chronic alcohol exposure exhibited greater accumulation of PNNs around neurons in the insula but not in a comparison region, the motor cortex despite presence of PNNs in both structures. A function of the insula is to enable cognitive flexibility and switching between processes (Menon et al., 2020), which requires working memory to track where one was and where one is going. Chronically-drinking mice with abnormally dense PNNs developed on insular neurons have implications for a mechanism of establishing addictive behavior, which involves “locking in” addiction-related activities and disabling plasticity for change required for mitigating addiction (Lasek et al., 2018). Other studies found abnormally high PNN density in the mouse orbitofrontal cortex (Coleman et al., 2014) and the rat hippocampus (Zhang et al., 2014). In the current study, hypoperfusion in the insula and memory-based limbic structures correlated with low working memory scores, raising questions about a possible relation between a PNN-density effect on CBF or, alternatively, a CBF effect on PNN density, especially in light of PNN restructuring that can occur with experimentally-induced, mild hypoperfusion not resulting in stroke (Dzyubenko et al., 2018).
Smokers, on average, have lower CBF than non-smokers, a finding observed with each of the major invasive functional imaging methods measuring perfusion, including FDG PET (Ritz et al., 2016; Wong et al., 2003), SPECT (Tutus et al., 1998), and xenon inhalation (Kubota et al., 1983) as well as non-invasive ASL (Gazdzinski et al., 2006; Mon et al., 2009). A series of studies from Meyerhoff and colleagues found frontal and parietal gray matter perfusion deficits in 1-week abstinent AUD participants who were smokers but not in the non-smokers (Gazdzinski et al., 2006); the smokers showed less CBF recovery after 5 weeks of sobriety than did non-smokers (Mon et al., 2009). Further, a dampening effect of being a smoker was detectable in polysubstance users but not AUD participants (Murray et al., 2015). These reports led us to question whether smokers in our study were those with the lowest CBF. Despite the small sample, the 4 non-smoking AUD participants had statistically lower CBF than smokers in the medial frontal, temporal, and parietal cortices. This inverse relation between smoking and PCASL-determined CBF replicates our previous studies (Pfefferbaum et al., 2011; Sullivan et al., 2013). It echoes a pattern of frontal and temporal CBF increases observed in smokers who were abstinent for 4 hours compared with participants who smoked as usual before doing a perfusion scan (Franklin et al., 2018). That study indicated that CBF correlated positively with craving indices, suggesting that smoking abstinence induced craving and heightened CBF, which was detected in orbitofrontal cortex, anterior and posterior ventral insula, posterior cingulum, and superior temporal gyrus. The similarity of affected regions between the Franklin et al. study and the current study raises the possibility that our participants were experiencing cigarette craving, given that they were required to refrain from smoking for at least 1 hour before scanning.
4.3. Limitations
One study limitation is that we did not administer a craving questionnaire on smoking before or after the scanning session, thereby precluding any possibility of testing cigarette craving as contributing to or explaining CBF group differences. Further, despite the larger sample and the inclusion of women and men in the present study, the number of women in the sample was inadequate to test for reliable sex differences, as was also the case in other studies (Durazzo et al., 2010; Mon et al., 2009; Murray et al., 2015). That said, we found no evidence for sex differences when sex was entered as a variable in the statistical models. Another limitation is the image resolution, a problem we attempted to address using a 32-channel head coil and a voxel-based, whole-brain analysis search for group differences. The analysis was successful, nonetheless, in identifying the medial frontal, anterior cingulate, and insular cortices as showing lower CBF in AUD than control individuals.
Whether the AUD deficit precedes the onset of drinking remains to be established, but a longitudinal study of youth who had a low response to alcohol and later developed AUD showed a greater regional CBF difference between alcohol and placebo than did high responders at the earlier observation time (Courtney et al., 2019). As a cross-sectional study, we were unable to address questions about potential improvement in regional CBF with prolonged sobriety or further decline with continued hazardous drinking.
4.4. Conclusion
To the extent that MR PCASL can validly track and quantify cerebral blood perfusion (Dai et al., 2008; Detre and Alsop, 1999), the attenuated cerebrovascular perfusion observed in AUD has implications for dampening cognitive abilities over and above that detectable with structure. That the PCASL signal correlated with several alcohol history factors strengthens the assumption that the perfusion deficit was related to chronic alcoholic drinking. Among the avenues of discovery laying ahead are investigations exploring relations between cerebral blood flow and cardiovascular health and cerebrovascular liability for white matter hyperintensities and stroke, which are known to occur with higher than normal incidence in AUD and hazardous levels of alcohol consumption (Fein et al., 2009; Larsson et al., 2020; Piano, 2017). On a practical level, fundamental differences in CBF between AUD and healthy controls has the potential of confounding fMRI activation patterns, whether experiments involve task-activated paradigms or resting state (Raichle and Snyder, 2007). Finally, the frontolimbic and insular cortical neuroconstellation with AUD-related dampened perfusion provides a lead for revealing a mechanism of dysfunction in attention and working memory associated with these brain regions in AUD.
Highlights.
MR pulsed continuous arterial spin labeling quantifies cerebral blood flow (CBF)
Individuals with Alcohol Use Disorder (AUD) and controls were compared
Data- and region-driven analyses located low frontolimbic and insular CBF in AUD
Regional CBF deficits accounted for working memory deficits in AUD
Local hypoperfusion suggests a mechanism of AUD-related dysfunction
Acknowledgment
This research was supported by grants from the National Institute on Alcohol Abuse and Alcoholism (AA010723, AA005965, AA013521, AA017347, AA023582).
Footnotes
Declaration of competing interest
None of the authors has any conflict of interest with this work, which is not being considered for publication elsewhere.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Alsop DC, Dai W, Grossman M, Detre JA, 2010. Arterial spin labeling blood flow MRI: its role in the early characterization of Alzheimer’s disease. J Alzheimers Dis 20(3), 871–880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- American Psychiatric Association, 2013. Diagnostic and Statistical Manual of Mental Disorder (DSM-V). American Psychiatric Association, Washington, D.C. [Google Scholar]
- Arnsten AF, 2013. The neurobiology of thought: the groundbreaking discoveries of Patricia Goldman-Rakic 1937–2003. Cereb Cortex 23(10), 2269–2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Avants BB, Epstein CL, Grossman M, Gee JC, 2008. Symmetric diffeomorphic image registration with cross-correlation: evaluating automated labeling of elderly and neurodegenerative brain. Med Image Anal 12(1), 26–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartzokis G, Lu PH, Tingus K, Mendez MF, Richard A, Peters DG, Oluwadara B, Barrall KA, Finn JP, Villablanca P, Thompson PM, Mintz J, 2009. Lifespan trajectory of myelin integrity and maximum motor speed. Neurobiology of aging in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borkowski JG, Benton AL, Spreen O, 1967. Word fluency and brain damage. Neuropsychologia 5, 135–140. [Google Scholar]
- Borogovac A, Asllani I, 2012. Arterial Spin Labeling (ASL) fMRI: advantages, theoretical constrains, and experimental challenges in neurosciences. Int J Biomed Imaging 2012, 818456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buxton RB, 2005. Quantifying CBF with arterial spin labeling. J Magn Reson Imaging 22(6), 723–726. [DOI] [PubMed] [Google Scholar]
- Chen H, He D, Lasek AW, 2015. Repeated Binge Drinking Increases Perineuronal Nets in the Insular Cortex. Alcoholism, clinical and experimental research 39(10), 1930–1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clark CP, Brown GG, Eyler LT, Drummond SP, Braun DR, Tapert SF, 2007. Decreased perfusion in young alcohol-dependent women as compared with age-matched controls. Am J Drug Alcohol Abuse 33(1), 13–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coleman LG Jr., Liu W, Oguz I, Styner M, Crews FT, 2014. Adolescent binge ethanol treatment alters adult brain regional volumes, cortical extracellular matrix protein and behavioral flexibility. Pharmacol Biochem Behav 116, 142–151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Courtney KE, Infante MA, Brown GG, Tapert SF, Simmons AN, Smith TL, Schuckit MA, 2019. The Relationship Between Regional Cerebral Blood Flow Estimates and Alcohol Problems at 5-Year Follow-Up: The Role of Level of Response. Alcoholism, clinical and experimental research 43(5), 812–821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crespi C, Galandra C, Canessa N, Manera M, Poggi P, Basso G, 2020. Microstructural damage of white-matter tracts connecting large-scale networks is related to impaired executive profile in alcohol use disorder. Neuroimage Clin 25, 102141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crespi C, Galandra C, Manera M, Basso G, Poggi P, Canessa N, 2019. Executive Impairment in Alcohol Use Disorder Reflects Structural Changes in Large-Scale Brain Networks: A Joint Independent Component Analysis on Gray-Matter and White-Matter Features. Front Psychol 10, 2479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai W, Garcia D, de Bazelaire C, Alsop DC, 2008. Continuous flow-driven inversion for arterial spin labeling using pulsed radio frequency and gradient fields. Magn Reson Med 60(6), 1488–1497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deen B, Pitskel NB, Pelphrey KA, 2011. Three systems of insular functional connectivity identified with cluster analysis. Cereb Cortex 21(7), 1498–1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delis DC, Kramer JH, Kaplan E, Ober B, 1987. The California Verbal Learning Test-Research edition. Psychological Corporation, New York. [Google Scholar]
- Demir B, Ulug B, Lay Ergun E, Erbas B, 2002. Regional cerebral blood flow and neuropsychological functioning in early and late onset alcoholism. Psychiatry research 115(3), 115–125. [DOI] [PubMed] [Google Scholar]
- Detre JA, Alsop DC, 1999. Perfusion magnetic resonance imaging with continuous arterial spin labeling: methods and clinical applications in the central nervous system. European journal of radiology 30(2), 115–124. [DOI] [PubMed] [Google Scholar]
- Detre JA, Wang J, Wang Z, Rao H, 2009. Arterial spin-labeled perfusion MRI in basic and clinical neuroscience. Current opinion in neurology 22(4), 348–355. [DOI] [PubMed] [Google Scholar]
- Durazzo TC, Gazdzinski S, Mon A, Meyerhoff DJ, 2010. Cortical perfusion in alcohol-dependent individuals during short-term abstinence: relationships to resumption of hazardous drinking after treatment. Alcohol (Fayetteville, N.Y 44(3), 201–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dzyubenko E, Manrique-Castano D, Kleinschnitz C, Faissner A, Hermann DM, 2018. Topological remodeling of cortical perineuronal nets in focal cerebral ischemia and mild hypoperfusion. Matrix Biol 74, 121–132. [DOI] [PubMed] [Google Scholar]
- Fein G, Shimotsu R, Di Sclafani V, Barakos J, Harper C, 2009. Increased white matter signal hyperintensities in long-term abstinent alcoholics compared with nonalcoholic controls. Alcoholism, clinical and experimental research 33(1), 70–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- First M, 2000. Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, Text Revision (DSM-IV-TR). American Psychiatric Association, Washington, DC. [Google Scholar]
- Franklin TR, Jagannathan K, Hager N, Fang Z, Xu S, Wong J, Childress AR, Detre JA, Rao H, Wetherill R, 2018. Brain substrates of early (4h) cigarette abstinence: Identification of treatment targets. Drug and alcohol dependence 182, 78–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galandra C, Basso G, Manera M, Crespi C, Giorgi I, Vittadini G, Poggi P, Canessa N, 2018. Salience network structural integrity predicts executive impairment in alcohol use disorders. Sci Rep 8(1), 14481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galandra C, Basso G, Manera M, Crespi C, Giorgi I, Vittadini G, Poggi P, Canessa N, 2019. Abnormal fronto-striatal intrinsic connectivity reflects executive dysfunction in alcohol use disorders. Cortex 115, 27–42. [DOI] [PubMed] [Google Scholar]
- Gansler DA, Harris GJ, Oscar-Berman M, Streeter C, Lewis RF, Ahmed I, Achong D, 2000. Hypoperfusion of inferior frontal brain regions in abstinent alcoholics: a pilot SPECT study. Journal of studies on alcohol 61(1), 32–37. [DOI] [PubMed] [Google Scholar]
- Gazdzinski S, Durazzo T, Jahng GH, Ezekiel F, Banys P, Meyerhoff D, 2006. Effects of chronic alcohol dependence and chronic cigarette smoking on cerebral perfusion: a preliminary magnetic resonance study. Alcoholism, clinical and experimental research 30(6), 947–958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilman S, Adams K, Koeppe RA, Berent S, Kluin KJ, Modell JG, Kroll P, Brunberg JA, 1990. Cerebellar and frontal hypometabolism in alcoholic cerebellar degeneration studied with positron emission tomography. Annals of neurology 28(6), 775–785. [DOI] [PubMed] [Google Scholar]
- Gundersen H, van Wageningen H, Gruner R, 2013. Alcohol-induced changes in cerebral blood flow and cerebral blood volume in social drinkers. Alcohol and alcoholism (Oxford, Oxfordshire) 48(2), 160–165. [DOI] [PubMed] [Google Scholar]
- Halcomb ME, Chumin EJ, Goni J, Dzemidzic M, Yoder KK, 2019. Aberrations of anterior insular cortex functional connectivity in nontreatment-seeking alcoholics. Psychiatry Res Neuroimaging 284, 21–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hallgren B, Sourander P, 1958. The effect of age on the non-haemin iron in the human brain. Journal of neurochemistry 3(1), 41–51. [DOI] [PubMed] [Google Scholar]
- Hernandez-Garcia L, Lahiri A, Schollenberger J, 2019. Recent progress in ASL. NeuroImage 187, 3–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kong LM, Zeng JY, Zheng WB, Shen ZW, Wu RH, 2019. Effects of Acute Alcohol Consumption on the Human Brain: Diffusional Kurtosis Imaging and Arterial Spin-Labeling Study. Ajnr 40(4), 641–647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koob GF, Volkow ND, 2016. Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3(8), 760–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kubota K, Yamaguchi T, Abe Y, Fujiwara T, Hatazawa J, Matsuzawa T, 1983. Effects of smoking on regional cerebral blood flow in neurologically normal subjects. Stroke; a journal of cerebral circulation 14(5), 720–724. [DOI] [PubMed] [Google Scholar]
- Laniepce A, Cabé N, André C, Bertran F, Boudehent C, Lahbairi N, Maillard A, Mary A, Segobin S, Vabret F, Rauchs G, Pitel AL, 2020. The effect of alcohol withdrawal syndrome severity on sleep, brain and cognition. Brain Communications 2, 1–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larsson SC, Burgess S, Mason AM, Michaelsson K, 2020. Alcohol Consumption and Cardiovascular Disease: A Mendelian Randomization Study. Circ Genom Precis Med 13(3), e002814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lasek AW, Chen H, Chen WY, 2018. Releasing Addiction Memories Trapped in Perineuronal Nets. Trends Genet 34(3), 197–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Berre AP, Müller-Oehring EM, Schulte T, Serventi MR, Pfefferbaum A, Sullivan EV, 2017. Deviant functional activation and connectivity of the right insula contribute to lack of awareness of episodic memory impairment. Cortex, in revision. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marxen M, Gan G, Schwarz D, Mennigen E, Pilhatsch M, Zimmermann US, Guenther M, Smolka MN, 2014. Acute effects of alcohol on brain perfusion monitored with arterial spin labeling magnetic resonance imaging in young adults. J Cereb Blood Flow Metab 34(3), 472–479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattsson N, Tosun D, Insel PS, Simonson A, Jack CR Jr., Beckett LA, Donohue M, Jagust W, Schuff N, Weiner MW, Alzheimer’s Disease Neuroimaging I, 2014. Association of brain amyloid-beta with cerebral perfusion and structure in Alzheimer’s disease and mild cognitive impairment. Brain 137(Pt 5), 1550–1561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mencarelli L, Neri F, Momi D, Menardi A, Rossi S, Rossi A, Santarnecchi E, 2019. Stimuli, presentation modality, and load-specific brain activity patterns during n-back task. Human brain mapping 40(13), 3810–3831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menon V, Gallardo G, Pinsk MA, Nguyen VD, Li JR, Cai W, Wassermann D, 2020. Microstructural organization of human insula is linked to its macrofunctional circuitry and predicts cognitive control. Elife 9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menon V, Uddin LQ, 2010. Saliency, switching, attention and control: a network model of insula function. Brain structure & function 214(5–6), 655–667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mon A, Durazzo TC, Gazdzinski S, Meyerhoff DJ, 2009. The impact of chronic cigarette smoking on recovery from cortical gray matter perfusion deficits in alcohol dependence: longitudinal arterial spin labeling MRI. Alcoholism, clinical and experimental research 33(8), 1314–1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray DE, Durazzo TC, Mon A, Schmidt TP, Meyerhoff DJ, 2015. Brain perfusion in polysubstance users: relationship to substance and tobacco use, cognition, and self-regulation. Drug and alcohol dependence 150, 120–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naqvi NH, Bechara A, 2010. The insula and drug addiction: an interoceptive view of pleasure, urges, and decision-making. Brain structure & function 214(5–6), 435–450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H, 2005. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. Journal of the American Geriatrics Society 53(4), 695–699. [DOI] [PubMed] [Google Scholar]
- Nicolas JM, Catafau AM, Estruch R, Lomena FJ, Salamero M, Herranz R, Monforte R, Cardenal C, Urbano-Marquez A, 1993. Regional cerebral blood flow-SPECT in chronic alcoholism: relation to neuropsychological testing. J Nucl Med 34(9), 1452–1459. [PubMed] [Google Scholar]
- Ogawa S, Lee TM, Nayak AS, Glynn P, 1990. Oxygenation-sensitive contrast in magnetic resonance image of rodent brain at high magnetic fields. Magn Reson Med 14(1), 68–78. [DOI] [PubMed] [Google Scholar]
- Oscar-Berman M, Valmas MM, Sawyer KS, Ruiz SM, Luhar RB, Gravitz ZR, 2014. Profiles of impaired, spared, and recovered neuropsychologic processes in alcoholism. Handb Clin Neurol 125, 183–210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfefferbaum A, Adalsteinsson E, Rohlfing T, Sullivan EV, 2009. MRI estimates of brain iron concentration in normal aging: Comparison of field-dependent (FDRI) and phase (SWI) methods. NeuroImage. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfefferbaum A, Chanraud S, Pitel AL, Muller-Oehring E, Shankaranarayanan A, Alsop DC, Rohlfing T, Sullivan EV, 2011. Cerebral blood flow in posterior cortical nodes of the default mode network decreases with task engagement but remains higher than in most brain regions. Cereb Cortex 21(1), 233–244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfefferbaum A, Chanraud S, Pitel AL, Shankaranarayanan A, Alsop DC, Rohlfing T, Sullivan EV, 2010. Volumetric cerebral perfusion imaging in healthy adults: regional distribution, laterality, and repeatability of pulsed continuous arterial spin labeling (PCASL). Psychiatry research 182(3), 266–273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piano MR, 2017. Alcohol’s Effects on the Cardiovascular System. Alcohol Res 38(2), 219–241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raichle ME, 1998. Behind the scenes of functional brain imaging: a historical and physiological perspective. Proceedings of the National Academy of Sciences of the United States of America 95(3), 765–772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raichle ME, Snyder AZ, 2007. A default mode of brain function: a brief history of an evolving idea. NeuroImage 37(4), 1083–1090; discussion 1097–1089. [DOI] [PubMed] [Google Scholar]
- Rey A, 1942. L’examen psychologique dans les cas d’encephalopathie traumatique. Archives de Psychologie 28, 286–340. [Google Scholar]
- Ritz L, Segobin S, Lannuzel C, Boudehent C, Vabret F, Eustache F, Beaunieux H, Pitel AL, 2016. Direct voxel-based comparisons between grey matter shrinkage and glucose hypometabolism in chronic alcoholism. J Cereb Blood Flow Metab 36(9), 1625–1640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodrigue KM, Daugherty AM, Foster CM, Kennedy KM, 2020. Striatal iron content is linked to reduced fronto-striatal brain function under working memory load. NeuroImage 210, 116544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rohlfing T, Zahr NM, Sullivan EV, Pfefferbaum A, 2010. The SRI24 multichannel atlas of normal adult human brain structure. Human brain mapping 31(5), 798–819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seeley WW, Menon V, Schatzberg AF, Keller J, Glover GH, Kenna H, Reiss AL, Greicius MD, 2007. Dissociable intrinsic connectivity networks for salience processing and executive control. J Neurosci 27(9), 2349–2356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skinner HA, 1982. Development and Validation of a Lifetime Alcohol Consumption Assessment Procedure. Addiction Research Foundation, Toronto, Canada. [Google Scholar]
- Skinner HA, Sheu WJ, 1982. Reliability of alcohol use indices: The lifetime drinking history and the MAST. Journal of studies on alcohol 43, 1157–1170. [DOI] [PubMed] [Google Scholar]
- Smith SM, Nichols TE, 2009. Threshold-free cluster enhancement: addressing problems of smoothing, threshold dependence and localisation in cluster inference. NeuroImage 44(1), 83–98. [DOI] [PubMed] [Google Scholar]
- Sullivan EV, Muller-Oehring EM, Pitel AL, Chanraud S, Shankaranarayanan A, Alsop DC, Rohlfing T, Pfefferbaum A, 2013. A selective insular perfusion deficit contributes to compromised salience network connectivity in recovering alcoholic men. Biological psychiatry 74(7), 547–555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan EV, Pfefferbaum A, 2019. Brain-behavior relations and effects of aging and common comorbidities in alcohol use disorder: A review. Neuropsychology 33(6), 760–780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan EV, Zahr NM, Saranathan M, Pohl KM, Pfefferbaum A, 2019. Convergence of three parcellation approaches demonstrating cerebellar lobule volume deficits in Alcohol Use Disorder. Neuroimage Clin 24, 101974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan EV, Zahr NM, Sassoon SA, Thompson WK, Kwon D, Pohl KM, Pfefferbaum A, 2018. The Role of Aging, Drug Dependence, and Hepatitis C Comorbidity in Alcoholism Cortical Compromise. JAMA Psychiatry 75(5), 474–483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan JT, Sykora K, Schneiderman J, Naranjo CA, Sellers EM, 1989. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict 84(11), 1353–1357. [DOI] [PubMed] [Google Scholar]
- Tanabe J, Yamamoto DJ, Sutton B, Brown MS, Hoffman PL, Burnham EL, Glueck DH, Tabakoff B, 2019. Effects of Alcohol and Acetate on Cerebral Blood Flow: A Pilot Study. Alcoholism, clinical and experimental research 43(10), 2070–2078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas B, Sheelakumari R, Kannath S, Sarma S, Menon RN, 2019. Regional Cerebral Blood Flow in the Posterior Cingulate and Precuneus and the Entorhinal Cortical Atrophy Score Differentiate Mild Cognitive Impairment and Dementia Due to Alzheimer Disease. Ajnr 40(10), 1658–1664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tutus A, Kugu N, Sofuoglu S, Nardali M, Simsek A, Karaaslan F, Gonul AS, 1998. Transient frontal hypoperfusion in Tc-99m hexamethylpropyleneamineoxime single photon emission computed tomography imaging during alcohol withdrawal. Biological psychiatry 43(12), 923–928. [DOI] [PubMed] [Google Scholar]
- Volkow ND, Wang GJ, Shokri Kojori E, Fowler JS, Benveniste H, Tomasi D, 2015. Alcohol decreases baseline brain glucose metabolism more in heavy drinkers than controls but has no effect on stimulation-induced metabolic increases. J Neurosci 35(7), 3248–3255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang K, Shou Q, Ma SJ, Liebeskind D, Qiao XJ, Saver J, Salamon N, Kim H, Yu Y, Xie Y, Zaharchuk G, Scalzo F, Wang DJJ, 2020. Deep Learning Detection of Penumbral Tissue on Arterial Spin Labeling in Stroke. Stroke; a journal of cerebral circulation 51(2), 489–497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wechsler D, 1981. Wechsler Adult Intelligence Scale-Revised. Psychological Corporation, New York. [Google Scholar]
- Wechsler D, 1987. Wechsler Memory Scale - Revised. The Psychological Corporation, San Antonio, TX. [Google Scholar]
- Wechsler D, 2001. Wechsler Test of Adult Reading (WTAR). Pearson Education, Inc, San Antonio, TX. [Google Scholar]
- Winkler AM, Ridgway GR, Webster MA, Smith SM, Nichols TE, 2014. Permutation inference for the general linear model. NeuroImage 92, 381–397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong DF, Maini A, Rousset OG, Brasic JR, 2003. Positron emission tomography--a tool for identifying the effects of alcohol dependence on the brain. Alcohol Res Health 27(2), 161–173. [PMC free article] [PubMed] [Google Scholar]
- Zahr NM, Pfefferbaum A, Sullivan EV, 2017. Perspectives on fronto-fugal circuitry from human imaging of alcohol use disorders. Neuropharmacology 122, 189–200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahr NM, Pohl KM, Saranathan M, Sullivan EV, Pfefferbaum A, 2019. Hippocampal subfield CA2+3 exhibits accelerated aging in Alcohol Use Disorder: A preliminary study. Neuroimage: Clinical. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zahr NM, Sullivan EV, Pohl KM, Pfefferbaum A, Saranathan M, 2020. Sensitivity of ventrolateral posterior thalamic nucleus to back pain in alcoholism and CD4 nadir in HIV. Human brain mapping 41(5), 1351–1361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Bhattacharyya S, Kusumo H, Goodlett CR, Tobacman JK, Guizzetti M, 2014. Arylsulfatase B modulates neurite outgrowth via astrocyte chondroitin-4-sulfate: dysregulation by ethanol. Glia 62(2), 259–271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao Q, Fritz M, Pfefferbaum A, Sullivan EV, Pohl KM, Zahr NM, 2018. Jacobian Maps Reveal Under-reported Brain Regions Sensitive to Extreme Binge Ethanol Intoxication in the Rat. Front Neuroanat 12, 108. [DOI] [PMC free article] [PubMed] [Google Scholar]