ABSTRACT
The occurrence of foams at oceans’ surfaces is patchy and generally short-lived, but a detailed understanding of bacterial communities inhabiting sea foams is lacking. Here, we investigated how marine foams differ from the sea-surface microlayer (SML), a <1-mm-thick layer at the air–sea interface, and underlying water from 1 m depth. Samples of sea foams, SML and underlying water collected from the North Sea and Timor Sea indicated that foams were often characterized by a high abundance of small eukaryotic phototrophic and prokaryotic cells as well as a high concentration of surface-active substances (SAS). Amplicon sequencing of 16S rRNA (gene) revealed distinctive foam bacterial communities compared with SML and underlying water, with high abundance of Gammaproteobacteria. Typical SML dwellers such as Pseudoalteromonas and Vibrio were highly abundant, active foam inhabitants and thus might enhance foam formation and stability by producing SAS. Despite a clear difference in the overall bacterial community composition between foam and SML, the presence of SML bacteria in foams supports the previous assumption that foam is strongly influenced by the SML. We conclude that active and abundant bacteria from interfacial habitats potentially contribute to foam formation and stability, carbon cycling and air–sea exchange processes in the ocean.
Keywords: air–sea interface, surfactants, particles, 16S rRNA amplicon sequencing, neuston
Floating foams at the oceans’ surfaces have a unique bacterial community signature in contrast to sea-surface microlayer and underlying water.
INTRODUCTION
Foams are patches floating on the water surface and may appear in any aquatic habitat. Foam is loosely defined as a dispersion of gas in liquid in the presence of surface-active substances (SAS) (Schilling and Zessner 2011). Convergence at zones of downwelling water and fronts, currents and breaking waves concentrate SAS and lead to foam formation at the sea surface and occasionally cause massive foam aggregates at beaches and in coastal zones (Eisenreich, Elzerman and Armstrong 1978; Bärlocher, Gordon and Ireland 1988; Thornton 1999; Kesaulya et al. 2008; Jenkinson et al. 2018). Furthermore, rising bubbles that do not burst immediately but accumulate at the surface can cause foam formation (Schilling and Zessner 2011). The nature, distribution and occurrence of foam in the marine environment are elusive, since its life span is limited to hours or days (Velimirov 1980; Pugh 1996), and the mean coverage of the ocean's surface by foams (whitecaps) is 1–6% based on satellite observations (Anguelova and Webster 2006).
One major prerequisite for foam formation is SAS, representing a complex mixture of mainly organic compounds. Due to their amphipathic nature, SAS accumulate at the sea surface (Wurl et al. 2009) and influence CO2 air–sea gas exchange (Pereira et al. 2018; Ribas-Ribas et al. 2018). In foams, SAS can originate from a variety of sources, such as marine bacteria (Satpute et al. 2010), kelp mucilage (Velimirov 1980), exudates of alive or broken phytoplankton cells (Velimirov 1980, 1982; Frew et al. 1990; Wegner and Hamburger 2002) or other organic detritus (Velimirov 1980). In addition, organic materials such as biogenic lipids and amino acids that accumulate at the sea surface during phytoplankton blooms are important substrates for the formation of foam (Eberlein et al. 1985; Riebesell 1993; Hunter et al. 2008). Even if foam is generally short-lived, its high concentration of organic matter (Eisenreich, Elzerman and Armstrong 1978; Johnson et al. 1989), especially of proteins and carbohydrates (Stefani et al. 2016), allows these nutrient-rich islands to function as microbial habitats. Despite being ephemeral feeding grounds, foams are remarkably rich and diverse in microorganisms (Tsyban 1971), including bacteria (Gobalakrishnan et al. 2014), protists and algae (Maynard 1968; Harold and Schlichting 1971). In addition, foams were shown to enclose copepods, polychaete and tunicate larvae (Armonies 1989; Castilla et al. 2007), and to trap microalgae (Roveillo et al. 2020), thus forming potentially important food sources for the higher trophic levels of the food web (Bärlocher, Gordon and Ireland 1988; Craig, Ireland and Bärlocher 1989; Scully 2009). In addition, a pharmaceutical potential of sea foam-inhabiting microbes was recently suggested (Oppong-Danquah et al. 2020).
The sea-surface microlayer (SML) is a <1-mm-thick, biofilm-like layer (Wurl and Holmes 2008; Wurl et al. 2016), located at the air–sea boundary (Figure S1, Supporting Information). It is characterized by remarkably different physicochemical and biological properties that allow its differentiation from the underlying water (ULW) (Hardy 1982; Cunliffe et al. 2013). Research throughout the last decades revealed that the accumulation of inorganic and organic substances and particles (including microorganisms) at the sea surface is a widespread phenomenon with important implications for carbon cycling processes (Engel et al. 2017; Wurl et al. 2017; Rahlff 2019). The interfacial position of the SML represents a challenging environment for inhabiting organisms also known as neuston (Naumann 1917). Differences in bacterial community composition between SML and ULW have been related to meteorological conditions (Agogué et al. 2005b; Stolle et al. 2011; Rahlff et al. 2017a); however, the specific adaptation of bacteria to the SML habitat remains an open question (Agogué et al. 2005a).
Neuston and plankton direct metabolic processing of carbon dioxide and methane (Upstill-Goddard et al. 2003; Calleja et al. 2005), and SAS occurrence and production in the SML govern gas exchange across air–water interfaces with SAS leading to a suppression of gas exchange velocity by 54% (Ribas-Ribas et al. 2018). Moreover, the impact of the neuston on carbon cycling can be high due to its higher abundance of bacteria compared with the ULW (Hardy 1982). This includes enhanced heterotrophic activity (Obernosterer et al. 2005), turnover of organic matter (Reinthaler, Sintes and Herndl 2008), colonization of buoyant and sinking particle aggregates (Bigg, Leck and Tranvik 2004; Leck and Bigg 2017) and viral lysis of prokaryotic cells (Ram et al. 2018). First approaches to quantify the contributions of neuston activity to underlying water oxygen concentrations revealed maximum neuston contributions of ≤7.1% (Rahlff et al. 2019).
Napolitano and Cicerone (1999) suggested that foams are essentially concentrated SML. Supporting this idea, enrichment of bacteria in foams compared with SML and/or ULW has been reported (Tsyban 1971; Kuznetsova and Lee 2002; Rahlff et al. 2017a). In addition, during blooms of Trichodesmium erythraeum, high concentrations of this species were also found in the corresponding foam (Maynard 1968). However, a thorough characterization of foam microbial community composition compared with the SML and ULW has never been performed. Using a microscopic approach, Druzhkov, Makarevich and Bardan (1997) found a highly identical composition of heterotrophs (nanoflagellates <5 µm and bacteria), nano- and microphytoplankton in foam and the SML. The authors further described higher abundances (one order of magnitude) of autotrophs, but not of heterotrophs in foams compared with the SML.
In this study, we investigated the bacterial community composition of marine foams in direct comparison to SML and ULW. Based on the theory that foam is an extremely concentrated form of the SML (Napolitano and Cicerone 1999), we hypothesized that the bacterial community compositions of foam and SML are more similar than those between foam and ULW. Since the SML is also considered an extreme habitat (Maki 1993) likely comprising many dead or dormant cells, we differentiated between active and total bacteria as inferred from amplicon sequencing of the complementary DNA (cDNA) from 16S ribosomes and the 16S ribosomal RNA (rRNA) genes, respectively, as previously conducted (Gentile et al. 2006; Kamke, Taylor and Schmitt 2010). Overall, we provide a detailed understanding of the bacterial community composition associated with sea foams in comparison to SML and ULW with likely implications for foam formation and stability, air–sea exchange processes and carbon cycling.
MATERIALS AND METHODS
Field sample collection
Field sampling was conducted from the bow of a small boat in the Jade Bay, North Sea, offshore Wilhelmshaven, Germany (Table S1, Supporting Information) in spring and summer 2016. Foams originated from different sources, such as phytoplankton exudates and convergence of surface water (Fig. 1A and C, respectively; Table S1, Supporting Information). Additional samples were collected during a Trichodesmium sp. bloom encountered in the Timor Sea at Stations 4, 5b and 8 (Fig. 1B; Table S1, Supporting Information) in October 2016 during R/V Falkor cruise FK161010 as described by Wurl et al. (2018). Foam and corresponding SML and ULW samples (here referred to as ‘set’) were collected from each location. From the Jade Bay, six sets were sampled in total, i.e. two from each month in April, May and July 2016. From the Timor Sea, one set was collected from each of three stations, but sequencing was only done for Station 8, i.e. one sampling set. Foam and SML were collected using the glass plate technique (Harvey and Burzell 1972) with a withdrawal rate of 5–6 cm s−1 as suggested by Carlson (1982). The glass plate was cleaned with 70% ethanol and rinsed with sample water before use. Material adhering on the glass plate was removed by wiping its surface with a squeegee into a sample-rinsed brown bottle. The procedure was repeated until the required volume of ∼100 mL was collected (∼20 dips). SML samples were collected between the foam patches and any dips contaminated with foam were rejected, and the glass plate was cleaned with ethanol again. Collected foams were not generated by the small boat whose engine was turned off. Samples from the ULW were taken at a depth of 1 m around the foams by using a syringe connected to a hose. All samples were kept on ice and immediately processed after sampling, since Velimirov (1980) showed that bacterial density in old foam was significantly higher than in fresh foam.
Figure 1.
Marine foam originating from (A) presumptive phytoplankton exudates (Jade Bay), (B)Trichodesmium bloom (Timor Sea) and (C) whitecaps produced by convergence of surface water (Jade Bay, North Sea).
Concentration of SAS
The concentration of SAS was measured by the voltammetry 747 VA Stand (Metrohm, Herisau, Switzerland) with a hanging mercury drop electrode as previously described (Ćosović and Vojvodić 1998; Wurl et al. 2011). The quantification is based on SAS adsorption on the Hg electrode measured by the change of capacity current (∆Ic) at an applied potential (E) of −0.6 V (Ćosović and Vojvodić 1998). Before measurement, concentrated samples such as foam samples were diluted with artificial seawater (0.55 M of NaCl solution) to achieve measurements within the linear calibration range. A standard addition technique was used with non-ionic surfactant Triton X-100 (Sigma-Aldrich, Taufkirchen, Germany) as a standard. SAS concentration in the samples was measured using two to three analytical replicates, resulting in relative standard deviations below 6% (Rickard et al. 2019). Concentration of SAS is expressed as the equivalent concentration of the additional Triton X-100 (µg Teq L–1).
Determination of microbial abundance
For determination of prokaryotic and small (<50 µm) eukaryotic phototrophic cell numbers in all Jade Bay samples and for three stations from the Timor Sea, foam and water samples were fixed with glutaraldehyde (1% final concentration), incubated at room temperature for 1 h and stored at −80°C until further analysis. Prior to staining and counting by flow cytometry, the particle-enriched foam samples were pre-filtered by gravity onto CellTrics® 50-µm filter (Sysmex Partec, Münster, Germany) to avoid clogging of the instrument by particulate matter. Autofluorescence analysis was used to count small eukaryotic phototrophic cells (Marie et al. 2000), and prokaryotic cells were stained with SYBR® Green I Nucleic Acid Gel Stain (9× in final concentration; Thermo Fisher Scientific, Darmstadt, Germany) following a protocol of Giebel et al. (2019). We did not measure biological replicates of samples because the coefficient of variation for SML and ULW flow cytometry samples is <5% (Rahlff et al. 2017a).
Calculation of enrichment factors
Enrichment factors (EFs) of SAS and cell counts were calculated for the pairings foam/SML (F/S), foam/ULW (F/U), and SML/ULW (S/U) (Table 1). Since the concentration of SAS or the abundance of cells in a foam or SML sample was divided by its SML or ULW counterpart, an EF > 1 implies enrichment of the parameter, whereas an EF < 1 indicates depletion.
Table 1.
Absolute and relative abundances given as EF of prokaryotes, small phototrophic eukaryotes and SAS in foam (F), SML (S), and ULW (U). NS = North Sea, NA = not available, Teq = Triton X-100 equivalents, TS = Timor Sea.
| Foam | SML | ULW | EF (F/S) | EF (F/U) | EF (S/U) | |
|---|---|---|---|---|---|---|
| Prokaryotes (cells mL−1) | Absolute values (106 cells mL−1) | Relative values | ||||
| NS_St1_210416 | 4.9 | NA | 2.6 | NA | 1.9 | NA |
| NS_St2_210416 | 2.6 | 2.6 | 2.5 | 1.0 | 1.1 | 1.1 |
| NS_St1_190516 | 13.7 | 3.3 | 3.2 | 4.1 | 4.2 | 1.0 |
| NS_St3_190516 | 46.2 | 4.6 | 3.1 | 10.1 | 14.8 | 1.5 |
| NS_St1_190716 | 6.6 | 3.9 | 3.7 | 1.7 | 1.8 | 1.1 |
| NS_St2_190716 | 5.0 | 3.4 | 3.5 | 1.5 | 1.4 | 1.0 |
| TS_St4_151016 | 10.0 | 1.8 | 1.1 | 5.6 | 9.3 | 1.7 |
| TS_St5b_171016 | 33.9 | NA | 1.0 | NA | 33.6 | NA |
| TS_St8_191016 | 5.8 | 1.0 | 1.2 | 5.9 | 4.9 | 0.8 |
| Small phototrophic eukaryotes (cells mL−1) | Absolute values (104 cells mL−1) | Relative values | ||||
| NS_St1_210416 | 1.9 | 1.0 | 1.6 | 1.8 | 1.1 | 0.6 |
| NS_St2_210416 | 1.4 | 1.4 | 2.2 | 1.0 | 0.6 | 0.6 |
| NS_St1_190516 | 29.6 | 0.9 | 2.3 | 34.8 | 12.9 | 0.4 |
| NS_St3_190516 | 57.1 | 1.0 | 1.9 | 56.0 | 30.4 | 0.5 |
| NS_St1_190716 | 9.1 | 4.0 | 4.2 | 2.3 | 2.2 | 1.0 |
| NS_St2_190716 | 4.2 | 2.5 | 2.8 | 1.7 | 1.5 | 0.9 |
| TS_St4_151016 | 2.1 | 0.4 | 0.1 | 5.8 | 20.2 | 3.5 |
| TS_St5b_171016 | 10.5 | NA | 0.1 | NA | 81.4 | NA |
| TS_St8_191016 | 2.7 | 0.1 | 0.3 | 23.7 | 10.4 | 0.4 |
| SAS (µg Teq L−1) | Absolute values | Relative values | ||||
| NS_St1_210416 | NA | NA | NA | NA | NA | NA |
| NS_St2_210416 | NA | NA | NA | NA | NA | NA |
| NS_St1_190516 | 77 496 | 576 | 213 | 134.4 | 364.5 | 2.7 |
| NS_St3_190516 | 148 233 | 1753 | 223 | 84.6 | 665.0 | 7.9 |
| NS_St1_190716 | 900 | 716 | 180 | 1.3 | 5.0 | 4.0 |
| NS_St2_190716 | 1397 | 270 | 133 | 5.2 | 10.5 | 2.0 |
| TS_St4_151016 | 69 370 | 240 | 268 | 288.9 | 258.5 | 0.9 |
| TS_St5b_171016 | 67 546 | 66 | 109 | 1020.5 | 618.7 | 0.6 |
| TS_St8_191016 | 28 797 | 255 | 171 | 113.1 | 168.5 | 1.5 |
Separation of particle-associated and free-living cells by filtration
A two-step filtration of foam, SML, and ULW samples was conducted as performed in previous studies (Garneau et al. 2009; Stolle et al. 2011). Sample water was filtered through 3-µm-pore-size (particle-associated cells) polycarbonate filters, after which the filtrate was filtered onto 0.2-µm-pore-size (free-living cells) polycarbonate filters (Merck Millipore, Darmstadt, Germany). Foam from the Timor Sea (Station 8) collected during a bloom of Trichodesmium sp. was additionally pre-filtered on a 100-µm mesh before subsequent filtration on the 3-µm-pore-size filter. All filters were initially stored at −80°C prior to DNA and RNA extraction.
Nucleic acid extraction and PCR
Extraction of DNA and RNA from the filters was performed by using the DNA + RNA + Protein Extraction Kit (Roboklon, Berlin, Germany) with a modified protocol (Rahlff et al. 2017a). Remaining DNA in RNA samples was digested on-column using 3 U of DNase and subsequently checked for contaminations with genomic DNA by polymerase chain reaction (PCR) (Figure S2, Supporting Information). A quantity of 10 ng RNA was reversely transcribed to cDNA using the NG dART Kit (Roboklon, Berlin, Germany), including negative controls either without reverse transcriptase or without RNA (Figure S2, Supporting Information). The reaction was incubated using the primer 1492R (5′-GGTTACCTTGTTACGACTT-3′; adapted from Lane 1991) for 60 min at 50°C followed by 5 min at 85°C. All DNAs and cDNAs were quantified using the Quant-iT PicoGreen dsDNA assay (Thermo Fisher Scientific, Darmstadt, Germany).
16S rRNA library preparation, sequencing run and data analysis
The bacterial 16S rRNA gene was amplified using Bakt_341F (5′-CCTACGGGNGGCWGCAG-3′) and Bakt_805R (5′-GACTACHVGGGTATCTAATCC-3′) (Herlemann et al. 2011) with the following modifications. Genomic DNA was amplified with 35 cycles prior Index-PCR. The cDNA samples were amplified with 25 cycles prior to index PCR. Amplicon PCR, index PCR, quantity and quality control, and sequencing of the individual libraries as pool in one Illumina MiSeq run were performed by an external provider (Eurofins Genomics, Ebersberg, Germany). Raw sequencing data were deposited at the European Nucleotide Archive (ENA) under accession number PRJEB34343. For data analysis, the resulting sequences were assembled using QIIME 1.9.1 (Caporaso et al. 2010) ‘joins paired-end Illumina reads’ function with default settings to merge forward and reverse sequences with an overlap of at least 30 bp. Sequences without overlap were removed. After converting fastq to fasta using the ‘convert_fastaqual_fastq’ function, the resulting sequences were evaluated using the SILVA NGS (next-generation sequencing) pipeline. The SILVA NGS pipeline (Glöckner et al. 2017) performs additional quality checks according to the SINA-based alignments (Pruesse, Peplies and Glöckner 2012) with a curated seed database in which PCR artifacts or non-SSU reads are excluded (based on SILVA release version 128; Pruesse et al. 2007). The longest read serves as a reference for the taxonomic classification in a BLAST (version 2.2.28+) search against the SILVA SSU Ref dataset. The classification of the reference sequence of a cluster (98% sequence identity) is mapped to all members of the respective cluster and to their replicates. Best BLAST hits were only accepted if they had a (sequence identity + alignment coverage)/2 ≥ 93% or were otherwise defined as unclassified. SILVA NGS classified a total of 9 182 084 reads (2% were rejected by the quality control). Sequences assigned to chloroplasts, mitochondria, eukaryotes and archaea were removed since the primer set employed in the analysis has only a very limited coverage of these groups.
Statistical analyses
Operational taxonomic unit (OTU) counts based on genus level were rarefied to 43 500 reads per sample using the single_rarefraction.py script implemented in QIIME. Venn diagrams were calculated using the ugent webtool (http://bioinformatics.psb.ugent.be/webtools/Venn/). We visualized the differences in the bacterial community composition through non‐metric multidimensional scaling (NMDS) plots using Bray–Curtis dissimilarity indices based on a genus rank classification. A linear discriminant analysis effect size (LEfSe) analysis was performed to determine bacterial groups that are significantly different between the samples using the ‘one against all’ strategy for multi‐class analysis (Segata et al. 2011). The program LEfSe uses a non-parametric test that combines standard tests for statistical significance with additional tests encoding biological consistency and effect relevance. P< 0.05 was regarded as statistical significance.
Differences in the number of 16S rRNA (active) and 16S rRNA gene (total)-derived OTUs and Shannon–Wiener Index between habitats (foam, SML, ULW) and attachment status were statistically analyzed using a Kruskal–Wallis test, and post hoc multiple pairwise comparisons were conducted based on Dunn's z statistic approximations to the actual rank statistics within the package ‘dunn.test’ (Dinno 2017) in R version 4.0.3 (Team RC 2017). The null hypothesis was rejected if P < 0.05. For the same samples, comparisons on phylum and OTU-based differences were statistically investigated using a one-way analysis of similarities (ANOSIM) based on Bray–Curtis dissimilarity and 9999 permutations, Bonferroni correction and a significance level of 95%.
RESULTS
Foams are enriched with SAS and microorganisms
Overall, foams from both sampling areas (North Sea and Timor Sea) were enriched with prokaryotic microorganisms, small eukaryotic phototrophic cells and SAS (Table 1). Cell counts of prokaryotic microorganisms ranged 2.6–46.2 × 106, 1.0–4.6 × 106 and 1.0–3.7 × 106 cells mL−1 in foam, SML and ULW, respectively (Fig. 2A). Thus, prokaryotic microorganisms in foams were enriched with a maximum EF of 10.1 and 5.9 over SML, and with a maximum EF of 14.8 and 33.6 over ULW in North Sea and Timor Sea, respectively (Table 1). However, these numbers likely represent an underestimation of cell counts because pre-filtration of foam samples onto a 50-µm sieve before the flow cytometry measurement likely excluded some bigger aggregates of cells and colloid material. Prokaryotic cells in the SML were enriched with a maximum EF of 1.5 and 1.7 over ULW in North Sea and Timor Sea, respectively. Likewise, the total number of small eukaryotic phototrophic cells was always higher in foam (range = 1.4–57.1 × 104 cells mL−1) compared with SML (range = 1.2–39.7 × 103 cells mL−1) and ULW (range = 1.1–41.7 × 103 cells mL−1; Fig. 2B). Thus, the maximum EF was 3.5 and 81.2 for SML over ULW and foam over ULW, respectively. The absolute number of small eukaryotic phototrophic cells was two orders of magnitude lower compared with the prokaryotic cell counts (Fig. 2A and B). Interestingly, small eukaryotic phototrophic cells were sometimes depleted in the SML compared with the ULW (S/U minimum EF = 0.4), while they were enriched in foams over ULW at the same time (F/U EF = 12.9; Table 1).
Figure 2.

Absolute cell counts mL−1 for (A) prokaryotes and (B) small eukaryotic phototrophic cells and (C) concentration of SAS in µg Teq L−1 for foam, SML and ULW. Foam samples from the North Sea, NS_FO1_210416 and NS_FO2_210416, were produced by waves, whereas NS_FO1_190516, NS_FO2_190516 and NS_FO1_190716 are foams likely derived from phytoplankton exudates. Foams from the Timor Sea (TS) originated from a Trichodesmium sp. bloom.
Foams contained the highest SAS concentrations compared with the other two habitats (Fig. 2C). SAS concentrations in foams varied between 900 and 148 233 µg Teq L−1 in North Sea and Timor Sea, whereas SML SAS concentrations were in a range of 66–1753 µg Teq L−1, and ULW SAS concentrations in a range of 109–223 µg Teq L−1 (Table 1). While SAS concentrations in the SML were enriched with a maximum EF of 7.9 compared with ULW, their concentration in foams compared with ULW was typically enriched by two orders of magnitude (EF ranging from 5 to 665).
Changes in the number of OTUs among sea foam, SML and underlying water
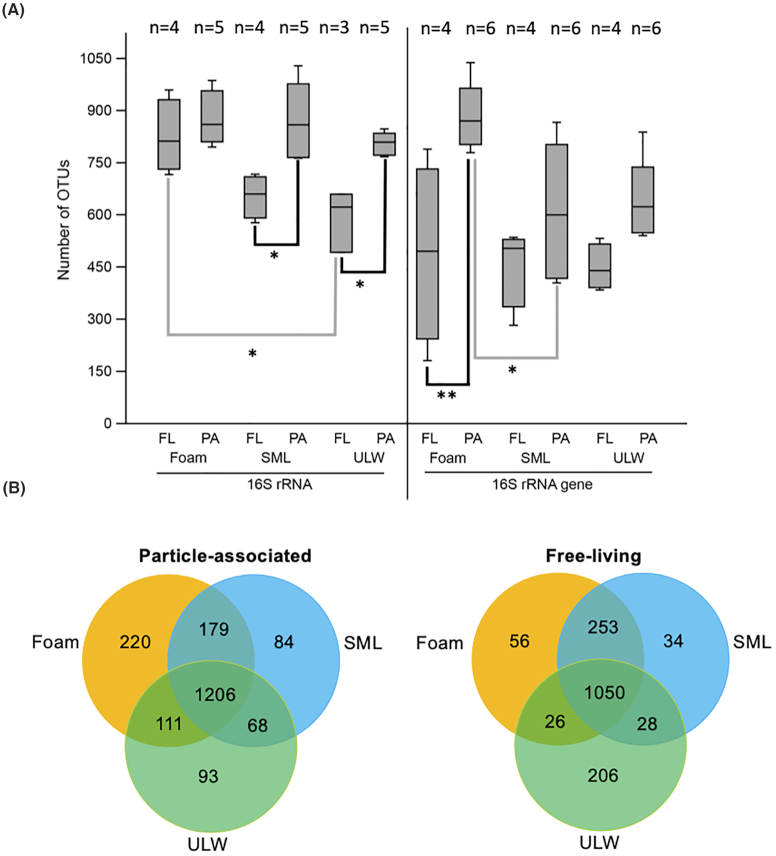
We analyzed the bacterial community composition of all North Sea samples and could not detect any obvious difference between sampling dates (Figure S3, Supporting Information). In the following, we did not differentiate between sampling dates, but by habitat (foam, SML, ULW), attachment status (particle-associated, free-living) and nucleic acid type for 16S rRNA analysis (16S rRNA gene, 16S rRNA) (Fig. 3A). Analyses revealed overall higher alpha diversity (numbers of OTUs) in active communities (median = 786.5) compared with total communities (median = 571).
Figure 3.
(A) 16S rRNA and 16S rRNA gene-derived numbers of OTUs for foam, SML and ULW habitat of pooled North Sea stations. The total number of OTUs of the three habitats is further distinguished between free-living (FL) and particle-associated (PA) bacterial communities. Grey and black lines indicate inter- and intrahabitat comparisons, respectively. The box plot shows the 25–75% quartiles; the median is indicated by the horizontal line inside the box. Error bars show minimal and maximal values. Asterisks indicate the level of significant differences: *P ≤ 0.05, **P ≤ 0.01; for reasons of the different number of observations (n), see Table S1 (Supporting Information) and for all statistical results, see Table S2 (Supporting Information). (B) Venn diagram showing overlapping and unique OTUs for foam, SML and ULW separated by free-living and particle-associated OTUs.
In 16S rRNA gene-based samples, the mean number of foam OTUs was significantly increased for particle-associated over free-living communities (Dunn's test, P = 0.0090), and also significantly higher compared with the SML particle-associated fraction with P = 0.035 (Fig. 3A). OTU numbers derived from active bacterial communities were significantly different between the particle-associated and the free-living fractions of SML (Dunn's test, P = 0.015) and ULW (P = 0.040) as well as between the free-living communities of foam and ULW (P = 0.032, Fig. 3A). Further significant differences existing between different biomes and different attachment states are shown in Table S2 (Supporting Information). The Shannon–Wiener index (Figure S4, Supporting Information), which accounts for both abundance and evenness of OTUs, confirmed many of the abovementioned trends, e.g. significant differences between particle-associated and free-living fractions of total and active communities within SML and ULW (all P< 0.05).
When comparing the total and active as well as the particle-attached and free-living communities, foam, SML and ULW generally shared a high number of similar OTUs (953–1206). Interestingly, foams and SML always had more OTUs in common compared with foam-ULW and ULW-SML (Fig. 3B; Figure S5, Supporting Information). In the particle-attached fraction, foam had the highest fraction of specific OTUs (220) and also shared many with the SML (179). In addition, SML and foam shared many OTUs in the free-living fraction (253) with less specific OTUs in foam (56) and the SML (34) compared with the particle-associated fraction (Fig. 3B).
Bacterial community composition of sea foam from the Jade Bay, North Sea
NMDS plots comparing the bacterial community composition based on the abundance of OTUs revealed that the foam bacterial community composition was distinct from SML and ULW communities, irrespective of differentiating active and total, or free-living and particle-associated bacterial communities (Fig. 4; Figure S6, Supporting Information). This was supported by the pairwise ANOSIM test, which revealed that the active and total communities differed significantly between foam and SML (both P = 0.01) as well as between foam and ULW (P = 0.01 and 0.04, respectively). In contrast, SML and ULW bacterial community compositions were more similar to each other as shown by the clustering (Fig. 4; Figure S6, Supporting Information).
Figure 4.
NMDS plot shows distinct clustering of foam (red), SML (blue) and underlying water (green) based on total (squares) and active (circles) bacterial communities. Further separation of communities into (A) 16S rRNA-based with free-living (open symbols) and particle-associated (filled symbols) stress = 0.14; (B) 16S rRNA gene-based with free-living (open symbols) and particle-associated (filled symbols) stress = 0.11.
On a phylum level, analyses of the total bacterial community showed that for all three habitats, Gammaproteobacteria, Verrucomicrobia and Cyanobacteria formed a higher portion of particle-associated than free-living communities with the exception of Cyanobacteria in foam (Fig. 5). In contrast, Alphaproteobacteria and Actinobacteria were more abundant in the free-living fraction (Fig. 5; Table S3, Supporting Information).
Figure 5.

Composition of phylum level of foam, SML and ULW samples of 16S rRNA and 16S rRNA gene-based relative abundance of OTUs of pooled North Sea stations. Each habitat is further separated into free-living (FL) and particle-associated (PA) bacterial communities. Dashed lines separate foam from SML and ULW.
Most phyla were similarly distributed in active and total bacterial communities, suggesting that many of the phyla were also active. Gammaproteobacteria, as one exception, tended to have higher relative abundance in the active bacterial community composition in foam (37.4% and 35.0% of free-living and particle-associated OTUs, respectively) compared with the total bacterial community composition (22.7% and 26.0% of free-living and particle-associated OTUs, respectively). In contrast, Verrucomicrobia had a higher portion in all habitats and fractions based on the total bacterial community compared with the active community. This was particularly striking for particle-associated communities in foams, where Verrucomicrobia were quite abundant among the total community (24.9%) but little active (3.1%). The active communities of foam contained less Alphaproteobacteria but more Gammaproteobacteria compared with SML and ULW communities (Fig. 5).
MB11C04 marine group (Verrucomicrobia), SAR11 clade (Alphaproteobacteria) and Oceanospirillales (Gammaproteobacteria) had a higher relative abundance in ULW and SML compared with foam in total and active communities and when comparing the respective attachment status, i.e. free-living and particle-associated fractions. (Figures S7–S9, Supporting Information). A higher relative abundance of active OTUs (based on 16S rRNA) in foam compared with SML and ULW was found among the Puniceicoccales (Verrucomicrobia), Sphingomonadales (Alphaproteobacteria), Alteromonadales and Vibrionales (both Gammaproteobacteria) (Figures S7–S9, Supporting Information). Active, free-living OTUs belonging to the order Flavobacteriales and Oceanospirillales were more, whereas free-living Sphingobacteriales were less numerous than their particle-associated counterparts in all three habitats (Figures S7 and S10, Supporting Information).
Except from the order Rhodobacterales (Figure S9, Supporting Information), foam generally had less alphaproteobacterial 16S rRNA gene-based OTUs compared with SML and ULW. However, foam contained a higher 16S rRNA gene-based relative abundance of Verrucomicrobia and Gammaproteobacteria compared with SML and ULW (Fig. 5). Among the Gammaproteobacteria, especially more OTUs of the orders Cellvibrionales,Vibrionales,Legionellales andAlteromonadales were increasingly detected in foam compared with the SML and ULW, whereas the order Oceanospirilliales was more depleted in foam (Figure S7, Supporting Information).
Gammaproteobacteria are typical sea-foam colonizers
Using the LEfSe method, we could identify OTUs that were significantly more abundant in foam compared with SML and ULW (Fig. 6). The analysis does not refer to the most abundant OTUs in terms of absolute numbers, but points out the largest differences between foam and the other two habitats. Members of the Gammaproteobacteria were typically abundant, active and total foam colonizers (Fig. 6). Taxa including Winogradskyella, Vibrio, Halioglobus and Pseudoalteromonas were particularly abundant in both 16S rRNA and 16S rRNA gene-derived foam samples as well as when compared with SML and ULW habitats. Persicirhabdus and other Verrucomicrobiaceae were typical foam dwellers with 11% and 7% relative abundance according to their presence in 16S rRNA gene-based samples, but with low activity according to 16S rRNA samples.
Figure 6.
Heat map showing OTUs with significant different relative abundance in foam, SML and ULW based on LEfSe analysis from pooled North Sea samples; OTUs are derived from sequencing amplicons derived from 16S rRNA and its gene. The vertical axis indicates the key species for these three biomes, respectively, while the horizontal axis shows how abundant these key OTUs were in the other two habitats. Dashed lines separate foam from SML and ULW.
Typical SML-populating bacteria belonged to taxa, which were phylogenetically related to Alphaproteobacteria, Gammaproteobacteria and Flavobacteria. Strikingly, abundance of total and active foam-specific OTUs often indicated a decreasing gradient from foam to SML to ULW (Figure S1, Supporting Information). High relative abundances (>5%) of Planktomarina, SAR116 and SAR86 were found for 16S rRNA and 16S rRNA gene-based SML samples. SAR11 and Candidatus Actinomarina typically occurred in high abundances in the ULW.
Trichodesmium sp.-produced foam—a case study
Due to technical restrictions, we could only obtain a single sample from the Timor Sea (Station 8) but found this valuable to analyze because the immediate source (Trichodesmium sp.-produced foam) was clear. Among the total community in foam, we found most particle-associated OTUs assigned to Trichodesmium (relative abundance = 33.4%), Alteromonas (26.4%) and Rhodobium (5.4%), whereas free-living OTUs were mostly assigned to Alteromonas (18.0%) and Rhodobium (10.2%) (Table S4, Supporting Information). Particle-associated OTUs were mainly assigned to Trichodesmium (68%) and Rhodobium (10.9%) in the SML, and to Trichodesmium (23.8%) and Oscillatoria (26.7%) in the ULW. Bacteria of the genus Saprospira were also detected in foam and SML, mainly in the particle-associated fractions. Most free-living OTUs from SML and ULW were assigned to Synechococcus with 15.7% and 21.6% relative abundance, respectively. In all 16S rRNA samples (active communities), Trichodesmium was also the most abundant OTU in foam and SML; only in the ULW Oscillatoria (48.2%) had higher relative abundance compared with Trichodesmium (29.1%). The relative abundance of 16S rRNA-based OTUs assigned to Alteromonas in foams (particle-associated: 17.8%, free-living: 12.6%) was comparatively enhanced to the SML (particle-associated: 0.2%, free-living: 1.2%) (Table S4, Supporting Information).
DISCUSSION
Foams form ecological niches for selected water bacterial communities and opportunistic Gammaproteobacteria
Foams are peculiar but understudied microbial habitats at air–water interfaces, and as soon as they subside, their material becomes part of the SML (Kuznetsova and Lee 2002). As foam has been suggested to represent concentrated SML (reviewed by Schilling and Zessner 2011), we hypothesized that foams contain typical SML bacteria. Our analysis showed that a considerable number of OTUs (>1000) overlap between all three habitats (Fig. 3B), supporting that bacteria are regularly dispersed between the biomes by wind-induced mixing processes, small-scale turbulences (Wu et al. 2019) and bubble transport (Robinson et al. 2019). However, also the sampling method, being identical for foam and SML, could cause some cross-contamination between habitats. As expected for a concentrated form of SML, we found high concentrations of SAS (max. 148 233 µg Teq L−1) in foams compared with reported values of 4989 µg Teq L−1 in nearshore SML (Wurl et al. 2011). In line with previous reports, we also found enrichments of cells in foam (Kuznetsova and Lee 2002; Rahlff et al. 2017a; Robinson et al. 2019). Decreasing abundance of small eukaryotic phototrophic cells in the SML during simultaneous increase of those in the respective foam sample supports passive transport from SML to foam, and that SML concentration is a mechanism for foam formation (Schilling and Zessner 2011). In our study, typically abundant and free-living groups from the ULW, including Planktomarina, SAR116 and SAR86, were found in foams at lower relative abundance (Fig. 6; Figure S11, Supporting Information) suggesting that these groups were passively transferred to the sticky SML and further into the foam.
The foam environment is an ecological niche with unique properties that harbors a distinctive bacterial community when compared with the SML and ULW (Fig. 4), which contradicts our initial hypothesis that SML and foam communities would be more similar to each other than foam and ULW. The occurrence of specific foam bacterial communities most likely resembles the particularities of the foam habitat, e.g. its dynamics in formation and disruption, its ephemerality and high organic matter accumulation. Foam consists of 90% air (Napolitano and Cicerone 1999) and contains channels, referred to as Plateau borders (Schramm and Wassmuth 1994), tending to trap living, motile algae, but draining dead ones (Roveillo et al. 2020). If this mechanism also applies to bacteria, it may explain why most OTUs found in foams were also active (Fig. 3A). In addition, foam is heavily enriched not only in nutrients but also in pollutants (Eisenreich, Elzerman and Armstrong 1978; Pojasek and Zajicek 1978) probably affecting its bacterial community structure. The type of foam under investigation also seems to be important: Foams presumably originating from phytoplankton exudates (Table S1, Supporting Information) contained high loads of SAS (Fig. 2C) and were linked to higher abundances of microbes (Fig. 2A and B) compared with foams formed by convergence of surface water (Fig. 1C; Table S1, Supporting Information). However, the role of the foam source for shaping its microbiome requires further research.
The overall difference in the bacterial community composition of foams compared with other habitats (Fig. 4) could be due to bacteria originating from SML and ULW that were selectively enriched in foam, including Vibrio, Pseudoalteromonas and Halioglobus (Fig. 6). Foams associated with phytoplankton biomass likely contain substantial amounts of labile organic matter and inorganic nutrients. Labile organic matter particularly selects for fast-growing, opportunistic and active bacteria in foams such as Gammaproteobacteria (Teeling et al. 2012; Landa et al. 2013), which are known to be dominant in the SML (Sun et al. 2020) and on transparent exopolymer particles (Zäncker, Engel and Cunliffe 2019). For instance, Alteromonas spp., of which we found an OTU among the free-living bacteria present in Trichodesmium foam (Table S4, Supporting Information), were previously shown to be highly abundant and active degraders of alginate, a cell wall component from marine macroalgae (Mitulla et al. 2016), and of labile dissolved organic carbon (Pedler, Aluwihare and Azam 2014).
The role of particles for foam-populating bacteria and biogenic foam formation
Foams in aquatic habitats contain large numbers of benthic or symbiotic rather than free-living bacteria (Maynard 1968). Since particulate organic matter is frequently enriched in both the SML (Aller et al. 2005) and foams (Johnson et al. 1989) compared with the ULW, foams are ideal substrates for particle-attached colonization. Particle-associated and free-living bacteria form separate communities in many aquatic environments (Crump, Armbrust and Baross 1999; Crespo et al. 2013), including all habitats from this study, especially foams (Fig. 4). SML bacteria are rather attached to substrates than occurring in the free-living state (Cunliffe et al. 2009), and particle-associated bacteria of the SML are generally more prone to changes in community composition than free-living ones (Stolle et al. 2010). In agreement with that and former studies (Parveen et al. 2013; Rieck et al. 2015), we found higher OTU numbers present on particles independent of the habitat under investigation (Fig. 3A). However, it should be noted that the size-fractionating filtration will not prevent free-living bacteria to also adhere to the 3-µm filter membrane, especially if the sample is highly enriched in particulate matter.
Although attachment to particles might have some drawbacks for bacteria regarding grazing (Albright et al. 1987), disadvantages might be easily outweighed by benefits of particles providing organic material and shelter from extreme levels of ultraviolet and solar radiation experienced at the air–sea boundary. The LEfSe analysis revealed that Winogradskyella (Flavobacteriaceae), often being associated with brown algae or sponges (Yoon and Lee 2012; Park and Yoon 2013; Schellenberg et al. 2017), was abundant among the metabolically active OTUs in foams (Fig. 6). As broken algal cells and detritus are major parts of foams, high relative abundance of Winogradskyella in the foam particle-associated fraction (Figure S11, Supporting Information) might be due to its attachment to algal-derived substrates. Persicirhabdus, known for particle adherence in sediments (Freitas et al. 2012) or on plastic debris (Oberbeckmann, Osborn and Duhaime 2016), was abundant in particle-attached total communities in foam but not very active (Fig. 6). Both Persicirhabdus and Winogradskyella are well known for their polysaccharide-degrading capacities (Yoon and Lee 2012; Cardman et al. 2014) and, hence, might prefer to stick to organic materials feeding or sheltering them. Microbes with a free-living lifestyle, such as Planktomarina, represent a very prominent and active group of the Rhodobacteraceae in marine temperate and (sub)polar areas of the oceans and coastal waters (Selje, Simon and Brinkhoff 2004; Giebel et al. 2009, 2011; Voget et al. 2015). Planktomarina is not a typical biofilm colonizer, but due to its high abundances in surface waters, we assume it was ‘glued’ in the SML and foam fraction and can benefit from enriched dissolved organic material, from aerobic anoxygenic photosynthesis, and oxidation of carbon monoxide as complementary energy sources (Giebel et al. 2019).
Foam events were observed in association with blooms of the haptophyte Phaeocystis globosa or Phaeocystispouchetii, or Cyanobacteria, or linked to certain plants (Velimirov 1980; Eberlein et al. 1985; Wegner and Hamburger 2002; Seuront, Vincent and Mitchell 2006; Wu et al. 2019). Experiments by Velimirov (1980) demonstrated foam formation in the presence of Ecklonia maxima, while most bacterial growth was antibiotically inhibited. The author showed that an algal component was crucial for the production of stable foam. Nevertheless, foam bacteria might contribute to the foam formation process (Heard et al. 2008), because, like phytoplankton, they produce SAS and exopolysaccharides (Satpute et al. 2010). Foam samples from our study contained bacterial OTUs likely capable of producing SAS, as previously demonstrated for Vibrio and Pseudoalteromonas (Hu, Wang and Wang 2015; Dang, Landfald and Willassen 2016). Moreover, Saprospira found in Trichodesmium-associated foam can produce sticky substances such as acidic polysaccharides, which enhance aggregate formation (Furusawa, Hartzell and Navaratnam 2015). We further speculate that in the Jade Bay, foam formation could be additionally supported by blooming Phaeocystis sp. (OSPAR_Assessments_HASEC17/D503 2017). Surface scum formation by cyanobacteria using intracellular gas vacuole formation as described for Trichodesmium sp. (Walsby 1992) could be an ecological strategy to reach atmospheric carbon dioxide supplies at the air–sea interface (Paerl and Ustach 1982). Trichodesmium sp. was active in our Timor Sea samples (Table S4, Supporting Information). During research in the Timor Sea, we incubated surface water with 1-mL Trichodesmium foam and found complete oxygen depletion after <14 h. Since samples without foam showed incomplete O2 consumption (Rahlff, Stolle and Wurl 2017b), we assume that complete O2 depletion was attributable to highly active, heterotrophic bacteria that accompanied Trichodesmium and its associated foam.
Overall, our cell count and SAS data support the previous assumption that foams are strongly influenced by the SML and, as an ephemeral and discrete habitat, select for specific bacteria, including the typical SML inhabitants Vibrio and Pseudoalteromonas. Moreover, ultramicrobacteria in foams can probably be easily aerosolized to the atmosphere and dispersed to land (Rahlff et al. 2020). Future studies should elucidate how wind-stirred bulk water and bubbles transfer colonies to SML and foams, and whether bacteria use these habitats as nutrient-rich ‘rest stop’ before being transferred to sea-spray aerosols, dispersed to beaches or returned to bulk water. The SML spans 71% of the Earth's surface and much remains to be learned about patchy surface phenomena such as foams and their ecological implications for the functioning of the marine food web and carbon turnover.
ACKNOWLEDGMENTS
We thank the captain and crew of the R/V Falkor during cruise FK161010. We highly appreciate the help of Julius Schmidt and the Institute for Chemistry and Biology of the Marine Environment (ICBM) workshop team for arranging small boat operations in the Jade Bay, North Sea. We are also grateful to our colleagues Isabel Stahmer, Tiera-Brandy Robinson, Kimberley Bird and Mariana Ribas-Ribas for their assistance during water sampling. Further, we appreciate the expert technical assistance of Lisa Maria Engl and Mathias Wolterink.
Supplementary Material
Contributor Information
Janina Rahlff, Institute for Chemistry and Biology of the Marine Environment (ICBM), Carl von Ossietzky University of Oldenburg, Schleusenstraße 1, 26382 Wilhelmshaven, Germany.
Christian Stolle, Institute for Chemistry and Biology of the Marine Environment (ICBM), Carl von Ossietzky University of Oldenburg, Schleusenstraße 1, 26382 Wilhelmshaven, Germany; Leibniz Institute for Baltic Sea Research (IOW), Seestraße 15, 18119 Rostock, Germany.
Helge-Ansgar Giebel, Institute for Chemistry and Biology of the Marine Environment (ICBM), Carl von Ossietzky University of Oldenburg, Carl-von-Ossietzky-Straße 9–11, 26129 Oldenburg, Germany.
Nur Ili Hamizah Mustaffa, Institute for Chemistry and Biology of the Marine Environment (ICBM), Carl von Ossietzky University of Oldenburg, Schleusenstraße 1, 26382 Wilhelmshaven, Germany.
Oliver Wurl, Institute for Chemistry and Biology of the Marine Environment (ICBM), Carl von Ossietzky University of Oldenburg, Schleusenstraße 1, 26382 Wilhelmshaven, Germany.
Daniel P. R. Herlemann, Leibniz Institute for Baltic Sea Research (IOW), Seestraße 15, 18119 Rostock, Germany; Institute of Agricultural and Environmental Sciences, Estonian University of Life Sciences, Kreutzwaldi 5, Tartu 51006, Estonia.
FUNDING
This work was supported by the European Research Council (ERC) project "Parameterization of the Sea-Surface Microlayer Effect" (PASSME, grant number GA336408), and the Leibniz Association project "Marine biogenic production, organic aerosols and maritime clouds: a process chain" (MarParCloud, grant number SAW-2016-TROPOS-2). DPRH was supported by the European Regional Development Fund/Estonian Research Council-funded Mobilitas Pluss Top Researcher (grant numbers MOBTT24 and P200028PKKH).
Conflict of Interest
None declared.
REFERENCES
- Agogué H, Casamayor EO, Bourrain Met al. A survey on bacteria inhabiting the sea surface microlayer of coastal ecosystems. FEMS Microbiol Ecol. 2005a;54:269–80. [DOI] [PubMed] [Google Scholar]
- Agogué H, Joux F, Obernosterer Iet al. Resistance of marine bacterioneuston to solar radiation. Appl Environ Microbiol. 2005b;71:5282–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albright LJ, Sherr EB, Sherr BFet al. Grazing of ciliated protozoa on free and particle-attached bacteria. Mar Ecol Prog Ser. 1987;38:125–9. [Google Scholar]
- Aller JY, Kuznetsova MR, Jahns CJet al. The sea surface microlayer as a source of viral and bacterial enrichment in marine aerosols. J Aerosol Sci. 2005;36:801–12. [Google Scholar]
- Anguelova MD, Webster F.. Whitecap coverage from satellite measurements: a first step toward modeling the variability of oceanic whitecaps. J Geophys Res. 2006;111:C03017. [Google Scholar]
- Armonies W. Occurrence of meiofauna in Phaeocystis seafoam. Mar Ecol Prog Ser. 1989;53:305–9. [Google Scholar]
- Bigg EK, Leck C, Tranvik L.. Particulates of the surface microlayer of open water in the central Arctic Ocean in summer. Mar Chem. 2004;91:131–41. [Google Scholar]
- Bärlocher F, Gordon J, Ireland RJ.. Organic composition of seafoam and its digestion by Corophium volutator (Pallas). J Exp Mar Biol Ecol. 1988;115:179–86. [Google Scholar]
- Calleja ML, Duarte CM, Navarro Net al. Control of air–sea CO2 disequilibria in the subtropical NE Atlantic by planktonic metabolism under the ocean skin. Geophys Res Lett. 2005;32:L08606. [Google Scholar]
- Caporaso JG, Kuczynski J, Stombaugh Jet al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7:335–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cardman Z, Arnosti C, Durbin Aet al. Verrucomicrobia are candidates for polysaccharide-degrading bacterioplankton in an Arctic fjord of Svalbard. Appl Environ Microbiol. 2014;80:3749–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carlson DJ. A field evaluation of plate and screen microlayer sampling techniques. Mar Chem. 1982;11:189–208. [Google Scholar]
- Castilla JC, Manriquez PH, Delgado APet al. Bio-foam enhances larval retention in a free-spawning marine tunicate. Proc Natl Acad Sci USA. 2007;104:18120–2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Craig D, Ireland RJ, Bärlocher F.. Seasonal variation in the organic composition of seafoam. J Exp Mar Biol Ecol. 1989;130:71–80. [Google Scholar]
- Crespo BG, Pommier T, Fernández-Gómez Bet al. Taxonomic composition of the particle-attached and free-living bacterial assemblages in the Northwest Mediterranean Sea analyzed by pyrosequencing of the 16S rRNA. Microbiologyopen. 2013;2:541–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crump BC, Armbrust EV, Baross JA. Phylogenetic analysis of particle-attached and free-living bacterial communities in the Columbia river, its estuary, and the adjacent coastal ocean. Appl Environ Microbiol. 1999;65:3192–204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cunliffe M, Engel A, Frka Set al. Sea surface microlayers: a unified physicochemical and biological perspective of the air–ocean interface. Progr Oceanogr. 2013;109:104–16. [Google Scholar]
- Cunliffe M, Whiteley AS, Newbold Let al. Comparison of bacterioneuston and bacterioplankton dynamics during a phytoplankton bloom in a fjord mesocosm. Appl Environ Microbiol. 2009;75:7173–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ćosović B, Vojvodić V.. Voltammetric analysis of surface active substances in natural seawater. Electroanalysis. 1998;10:429–34. [Google Scholar]
- Dang NP, Landfald B, Willassen NP.. Biological surface-active compounds from marine bacteria. Environ Technol. 2016;37:1151–8. [DOI] [PubMed] [Google Scholar]
- Dinno A. Package ‘dunn.test’. 2017, https://cran.r-project.org/web/packages/dunn.test/dunn.test.pdf (4 March 2021, date last accessed). [Google Scholar]
- Druzhkov NV, Makarevich PR, Bardan SI.. Sea foam as an object of sea-surface film studies. Polar Res. 1997;16:117–21. [Google Scholar]
- Eberlein K, Leal MT, Hammer KDet al. Dissolved organic substances during a Phaeocystis pouchetii bloom in the German Bight (North-Sea). Mar Biol. 1985;89:311–6. [Google Scholar]
- Eisenreich SJ, Elzerman AW, Armstrong DE. Enrichment of micronutrients, heavy metals, and chlorinated hydrocarbons in wind-generated lake foam. Environ Sci Technol. 1978;12:413–7. [Google Scholar]
- Engel A, Bange HW, Cunliffe Met al. The ocean's vital skin: toward an integrated understanding of the sea surface microlayer. Front Mar Sci. 2017;4:165. [Google Scholar]
- Freitas S, Hatosy S, Fuhrman JAet al. Global distribution and diversity of marine Verrucomicrobia. ISME J. 2012;6:1499–505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frew NM, Goldman JC, Dennett MRet al. Impact of phytoplankton-generated surfactants on air–sea gas exchange. J Geophys Res. 1990;95:3337–52. [Google Scholar]
- Furusawa G, Hartzell PL, Navaratnam V.. Calcium is required for ixotrophy of Aureispira sp. CCB-QB1. Microbiology. 2015;161:1933–41. [DOI] [PubMed] [Google Scholar]
- Garneau M-È, Vincent WF, Terrado Ret al. Importance of particle-associated bacterial heterotrophy in a coastal Arctic ecosystem. J Mar Syst. 2009;75:185–97. [Google Scholar]
- Gentile G, Giuliano L, D'Auria Get al. Study of bacterial communities in Antarctic coastal waters by a combination of 16S rRNA and 16S rDNA sequencing. Environ Microbiol. 2006;8:2150–61. [DOI] [PubMed] [Google Scholar]
- Giebel HA, Brinkhoff T, Zwisler Wet al. Distribution of Roseobacter RCA and SAR11 lineages and distinct bacterial communities from the subtropics to the Southern Ocean. Environ Microbiol. 2009;11:2164–78. [DOI] [PubMed] [Google Scholar]
- Giebel HA, Kalhoefer D, Lemke Aet al. Distribution of Roseobacter RCA and SAR11 lineages in the North Sea and characteristics of an abundant RCA isolate. ISME J. 2011;5:8–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giebel HA, Wolterink M, Brinkhoff Tet al. Complementary energy acquisition via aerobic anoxygenic photosynthesis and carbon monoxide oxidation by Planktomarina temperata of the Roseobacter group. FEMS Microbiol Ecol. 2019;95:fiz050. [DOI] [PubMed] [Google Scholar]
- Glöckner FO, Yilmaz P, Quast Cet al. 25 years of serving the community with ribosomal RNA gene reference databases and tools. J Biotechnol. 2017;261:169–76. [DOI] [PubMed] [Google Scholar]
- Gobalakrishnan R, Sivakumar K, Sethubathi GVet al. Seafoam bacteria from the Havelock island of the Andamans, India. Indian J Geo-Mar Sci. 2014;43:1562–5. [Google Scholar]
- Hardy JT. The sea surface microlayer: biology, chemistry and anthropogenic enrichment. Progr Oceanogr. 1982;11:307–28. [Google Scholar]
- Harold E, Schlichting JR.. A preliminary study of the algae and protozoa in seafoam. Bot Mar. 1971;14:24–8. [Google Scholar]
- Harvey GW, Burzell LA.. A simple microlayer method for small samples. Limnol Oceanogr. 1972;17:156–7. [Google Scholar]
- Heard J, Harvey E, Johnson BBet al. The effect of filamentous bacteria on foam production and stability. Colloids Surf B Biointerfaces. 2008;63:21–6. [DOI] [PubMed] [Google Scholar]
- Herlemann DPR, Labrenz M, Jurgens Ket al. Transitions in bacterial communities along the 2000 km salinity gradient of the Baltic Sea. ISME J. 2011;5:1571–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter TN, Pugh RJ, Franks GVet al. The role of particles in stabilising foams and emulsions. Adv Colloid Interface Sci. 2008;137:57–81. [DOI] [PubMed] [Google Scholar]
- Hu X, Wang C, Wang P.. Optimization and characterization of biosurfactant production from marine Vibrio sp. strain 3B-2. Front Microbiol. 2015;6:976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenkinson IR, Laurent S, Ding Het al. Biological modification of mechanical properties of the sea surface microlayer, influencing waves, ripples, foam and air–sea fluxes. Elem Sci Anth. 2018;6:26. [Google Scholar]
- Johnson BD, Zhou X, Parrish CCet al. Fractionation of particulate matter, the trace metals Cu, Cd, and Zn, and lipids in foam and water below Niagara Falls. J Great Lakes Res. 1989;15:189–96. [Google Scholar]
- Kamke J, Taylor MW, Schmitt S.. Activity profiles for marine sponge-associated bacteria obtained by 16S rRNA vs 16S rRNA gene comparisons. ISME J. 2010;4:498–508. [DOI] [PubMed] [Google Scholar]
- Kesaulya I, Leterme SC, Mitchell JGet al. The impact of turbulence and phytoplankton dynamics on foam formation, seawater viscosity and chlorophyll concentration in the eastern English Channel. Oceanologia. 2008;50:167–82. [Google Scholar]
- Kuznetsova M, Lee C.. Dissolved free and combined amino acids in nearshore seawater, sea surface microlayers and foams: influence of extracellular hydrolysis. Aquat Sci. 2002;64:252–68. [Google Scholar]
- Landa M, Cottrell MT, Kirchman DLet al. Changes in bacterial diversity in response to dissolved organic matter supply in a continuous culture experiment. Aquat Microb Ecol. 2013;69:157–68. [Google Scholar]
- Lane D. 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds). Nucleic Acid Techniques in Bacterial Systematics. Chichester, UK: John Wiley & Sons, 1991, 115–75. [Google Scholar]
- Leck C, Bigg EK.. Biogenic particles in the surface microlayer and overlaying atmosphere in the central Arctic Ocean during summer. Tellus B Chem Phys Meteorol. 2017;57:305–16. [Google Scholar]
- Maki J. The air–water interface as an extreme environment. In: Ford T (ed). Aquatic Microbiology: An Ecological Approach. Boston, MA: Blackwell Scientific Publication, 1993, 409–39. [Google Scholar]
- Marie D, Simon N, Guillou Let al. Flow cytometry analysis of marine picoplankton. In: Diamond RA, Demaggio S (eds). Living Color Springer Lab Manuals. Berlin, Heidelberg: Springer, 2000, 421–54. [Google Scholar]
- Maynard NG. Aquatic foams as an ecological habitat. Zeitschrift für allgemeine Mikrobiologie. 1968;8:119–26. [DOI] [PubMed] [Google Scholar]
- Mitulla M, Dinasquet J, Guillemette Ret al. Response of bacterial communities from California coastal waters to alginate particles and an alginolytic Alteromonas macleodii strain. Environ Microbiol. 2016;18:4369–77. [DOI] [PubMed] [Google Scholar]
- Napolitano GE, Cicerone DS., Lipids in water-surface microlayers and foams. In: Lipids in Freshwater Ecosystems. New York, NY: Springer, 1999, 235–62. [Google Scholar]
- Naumann E. Beiträge zur Kenntnis des Teichnannoplanktons. II. Über das Neuston des Süsswassers. Biol Centralblatt. 1917;37:98–106. [Google Scholar]
- Oberbeckmann S, Osborn AM, Duhaime MB.. Microbes on a bottle: substrate, season and geography influence community composition of microbes colonizing marine plastic debris. PLoS One. 2016;11:e0159289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Obernosterer I, Catala P, Reinthaler Tet al. Enhanced heterotrophic activity in the surface microlayer of the Mediterranean Sea. Aquat Microb Ecol. 2005;39:293–302. [Google Scholar]
- Oppong-Danquah E, Passaretti C, Chianese Oet al. Mining the metabolome and the agricultural and pharmaceutical potential of sea foam-derived fungi. Mar Drugs. 2020;18:128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- OSPAR_Assessments_HASEC17/D503 . Trends in Blooms of the Nuisance Phytoplankton Species Phaeocystis in Belgian, Dutch and German Waters. OSPAR Agreement 2016-01 OSPAR Coordinated Environmental Monitoring Programme (CEMP) Revised in 2017 volume 2020. D5 - Eutrophication HASEC17/D503: OSPAR, 2017. [Google Scholar]
- Paerl HW, Ustach JF.. Blue-green algal scums: an explanation for their occurrence during freshwater blooms. Limnol Oceanogr. 1982;27:212–7. [Google Scholar]
- Park S, Yoon JH.. Winogradskyella undariae sp. nov., a member of the family Flavobacteriaceae isolated from a brown algae reservoir. Antonie Van Leeuwenhoek. 2013;104:619–26. [DOI] [PubMed] [Google Scholar]
- Parveen B, Mary I, Vellet Aet al. Temporal dynamics and phylogenetic diversity of free-living and particle-associated Verrucomicrobia communities in relation to environmental variables in a mesotrophic lake. FEMS Microbiol Ecol. 2013;83:189–201. [DOI] [PubMed] [Google Scholar]
- Pedler BE, Aluwihare LI, Azam F.. Single bacterial strain capable of significant contribution to carbon cycling in the surface ocean. Proc Natl Acad Sci USA. 2014;111:7202–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pereira R, Ashton I, Sabbaghzadeh Bet al. Reduced air–sea CO2 exchange in the Atlantic Ocean due to biological surfactants. Nat Geosci. 2018;11:492–6. [Google Scholar]
- Pojasek R, Zajicek O.. Surface microlayers and foams: source and metal transport in aquatic systems. Water Res. 1978;12:7–10. [Google Scholar]
- Pruesse E, Peplies J, Glöckner FO.. SINA: accurate high-throughput multiple sequence alignment of ribosomal RNA genes. Bioinformatics. 2012;28:1823–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruesse E, Quast C, Knittel Ket al. SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res. 2007;35:7188–96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pugh RJ. Foaming, foam films, antifoaming and defoaming. Adv Colloid Interface Sci. 1996;64:67–142. [Google Scholar]
- Rahlff J, Giebel H-A, Stolle Cet al. Overlooked diversity of ultramicrobacterial minorities at the air–sea interface. Atmosphere. 2020;11:1214. [Google Scholar]
- Rahlff J, Stolle C, Giebel HAet al. High wind speeds prevent formation of a distinct bacterioneuston community in the sea-surface microlayer. FEMS Microbiol Ecol. 2017a;93:fix041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahlff J, Stolle C, Giebel HAet al. Oxygen profiles across the sea-surface microlayer: effects of diffusion and biological activity. Front Mar Sci. 2019;6:11. [Google Scholar]
- Rahlff J, Stolle C, Wurl O.. SISI: a new device for in situ incubations at the ocean surface. J Mar Sci Eng. 2017b;5:46. [Google Scholar]
- Rahlff J. The virioneuston: a review on viral-bacterial associations at air–water interfaces. Viruses. 2019;11:191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ram ASP, Mari X, Brune Jet al. Bacterial–viral interactions in the sea surface microlayer of a black carbon-dominated tropical coastal ecosystem (Halong Bay, Vietnam). Elem Sci Anth. 2018;6:13. [Google Scholar]
- Reinthaler T, Sintes E, Herndl GJ.. Dissolved organic matter and bacterial production and respiration in the sea-surface microlayer of the open Atlantic and the western Mediterranean Sea. Limnol Oceanogr. 2008;53:122–36. [Google Scholar]
- Ribas-Ribas M, Helleis F, Rahlff Jet al. Air–sea CO2-exchange in a large annular wind-wave tank and the effects of surfactants. Front Mar Sci. 2018;5:457. [Google Scholar]
- Rickard PC, Uher G, Upstill-Goddard RCet al. Reconsideration of seawater surfactant activity analysis based on an inter-laboratory comparison study. Mar Chem. 2019;208:103–11. [Google Scholar]
- Riebesell U. Aggregation of Phaeocystis during phytoplankton spring blooms in the southern North Sea. Mar Ecol Prog Ser. 1993;96:281–9. [Google Scholar]
- Rieck A, Herlemann DP, Jürgens Ket al. Particle-associated differ from free-living bacteria in surface waters of the Baltic Sea. Front Microbiol. 2015;6:1297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson TB, Wurl O, Bahlmann Eet al. Rising bubbles enhance the gelatinous nature of the air–sea interface. Limnol Oceanogr. 2019;64:2358–72. [Google Scholar]
- Roveillo Q, Dervaux J, Wang Yet al. Trapping of swimming microalgae in foam. J R Soc Interface. 2020;17:20200077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satpute SK, Banat IM, Dhakephalkar PKet al. Biosurfactants, bioemulsifiers and exopolysaccharides from marine microorganisms. Biotechnol Adv. 2010;28:436–50. [DOI] [PubMed] [Google Scholar]
- Schellenberg J, Busse HJ, Hardt Met al. Winogradskyella haliclonae sp. nov., isolated from a marine sponge of the genus Haliclona. Int J Syst Evol Microbiol. 2017;67:4902–10. [DOI] [PubMed] [Google Scholar]
- Schilling K, Zessner M.. Foam in the aquatic environment. Water Res. 2011;45:4355–66. [DOI] [PubMed] [Google Scholar]
- Schramm LL, Wassmuth F., Foams: basic principles. Foams: Fundamentals and Applications in the Petroleum Industry. Washington, DC: ACS Publications, 1994, 3–45. [Google Scholar]
- Scully EP. Utilization of surface foam as a food source by the Hermit crab, Pagurus longicarpus Say, 1817. Mar Behav Physiol. 2009;5:159–62. [Google Scholar]
- Segata N, Izard J, Waldron Let al. Metagenomic biomarker discovery and explanation. Genome Biol. 2011;12:R60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selje N, Simon M, Brinkhoff T.. A newly discovered Roseobacter cluster in temperate and polar oceans. Nature. 2004;427:445–8. [DOI] [PubMed] [Google Scholar]
- Seuront L, Vincent D, Mitchell JG.. Biologically induced modification of seawater viscosity in the Eastern English Channel during a Phaeocystis globosa spring bloom. J Mar Syst. 2006;61:118–33. [Google Scholar]
- Stefani F, Salerno F, Copetti Det al. Endogenous origin of foams in lakes: a long-term analysis for Lake Maggiore (northern Italy). Hydrobiologia. 2016;767:249–65. [Google Scholar]
- Stolle C, Labrenz M, Meeske Cet al. Bacterioneuston community structure in the southern Baltic sea and its dependence on meteorological conditions. Appl Environ Microbiol. 2011;77:3726–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stolle C, Nagel K, Labrenz Met al. Succession of the sea-surface microlayer in the coastal Baltic Sea under natural and experimentally induced low-wind conditions. Biogeosciences. 2010;7:2975–88. [Google Scholar]
- Sun H, Zhang Y, Tan Set al. DMSP-producing bacteria are more abundant in the surface microlayer than subsurface seawater of the East China Sea. Microb Ecol. 2020;80:350–65. [DOI] [PubMed] [Google Scholar]
- Team RC. R: a language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing, 2017. [Google Scholar]
- Teeling H, Fuchs BM, Becher Det al. Substrate-controlled succession of marine bacterioplankton populations induced by a phytoplankton bloom. Science. 2012;336:608–11. [DOI] [PubMed] [Google Scholar]
- Thornton DCO. Phytoplankton mucilage production in coastal waters: a dispersal mechanism in a front dominated system?. Ethol Ecol Evol. 1999;11:179–85. [Google Scholar]
- Tsyban A. Marine bacterioneuston. J Oceanogr Soc Jpn. 1971;27:56–66. [Google Scholar]
- Upstill-Goddard RC, Frost T, Henry GRet al. Bacterioneuston control of air-water methane exchange determined with a laboratory gas exchange tank. Global Biogeochem Cy. 2003;17:19.1–19.15. [Google Scholar]
- Velimirov B. Formation and potential trophic significance of marine foam near kelp beds in the Benguela upwelling system. Mar Biol. 1980;58:311–8. [Google Scholar]
- Velimirov B. Sugar and lipid components in sea foam near kelp beds. Mar Ecol. 1982;3:97–107. [Google Scholar]
- Voget S, Wemheuer B, Brinkhoff Tet al. Adaptation of an abundant Roseobacter RCA organism to pelagic systems revealed by genomic and transcriptomic analyses. ISME J. 2015;9:371–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walsby AE., The gas vesicles and buoyancy of Trichodesmium. In: Marine Pelagic Cyanobacteria: Trichodesmium and Other Diazotrophs. Dordrecht: Springer, 1992, 141–61. [Google Scholar]
- Wegner C, Hamburger M.. Occurrence of stable foam in the upper Rhine River caused by plant-derived surfactants. Environ Sci Technol. 2002;36:3250–6. [DOI] [PubMed] [Google Scholar]
- Wurl O, Bird K, Cunliffe Met al. Warming and inhibition of salinization at the ocean's surface by cyanobacteria. Geophys Res Lett. 2018;45:4230–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wurl O, Ekau W, Landing WMet al. Sea surface microlayer in a changing ocean: a perspective. Elem Sci Anth. 2017;5:31. [Google Scholar]
- Wurl O, Holmes M.. The gelatinous nature of the sea-surface microlayer. Mar Chem. 2008;110:89–97. [Google Scholar]
- Wurl O, Miller L, Ruttgers Ret al. The distribution and fate of surface-active substances in the sea-surface microlayer and water column. Mar Chem. 2009;115:1–9. [Google Scholar]
- Wurl O, Stolle C, Van Thuoc Cet al. Biofilm-like properties of the sea surface and predicted effects on air–sea CO2 exchange. Progr Oceanogr. 2016;144:15–24. [Google Scholar]
- Wurl O, Wurl E, Miller Let al. Formation and global distribution of sea-surface microlayers. Biogeosciences. 2011;8:121–35. [Google Scholar]
- Wu X, Noss C, Liu Let al. Effects of small-scale turbulence at the air–water interface on microcystis surface scum formation. Water Res. 2019;167:115091. [DOI] [PubMed] [Google Scholar]
- Yoon JH, Lee SY.. Winogradskyella multivorans sp. nov., a polysaccharide-degrading bacterium isolated from seawater of an oyster farm. Antonie Van Leeuwenhoek. 2012;102:231–8. [DOI] [PubMed] [Google Scholar]
- Zäncker B, Engel A, Cunliffe M.. Bacterial communities associated with individual transparent exopolymer particles (TEP). J Plankton Res. 2019;41:561–5. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.