Objective
Vaccination of pregnant women can be an important strategy to confer protection to neonates and young infants.1 However, there are limited data on the immunologic response of pregnant women to the messenger RNA (mRNA) coronavirus disease 2019 (COVID-19) vaccinations2 and the kinetics of transplacental antibody transfer.3 , 4 Our objective was to investigate the transfer of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) immunoglobulin G (IgG) to infants following maternal COVID-19 vaccination during pregnancy and the factors associated with an increased efficiency of transfer.
Study Design
This was a prospective case series of pregnant women who delivered at the Prentice Women’s Hospital in Chicago, IL (between January 2021 and March 2021). Women who received a COVID-19 vaccination during pregnancy were identified via review of the electronic health records (EHRs). Demographic and clinical information, including the specific vaccine used and the latency from vaccination to delivery was obtained from the EHRs. At the time of the study, 2 mRNA COVID-19 vaccines, the Pfizer (New York, NY) and Moderna (Cambridge, MA) vaccines, had received emergency use authorization, and healthcare workers were the initial qualifying group. Maternal blood and umbilical cord blood (herein referred to as “infant”) were retrieved from specimens submitted for blood typing. SARS-CoV-2 IgM and IgG antibody levels were measured from the plasma using the Access SARS-CoV-2 IgG and IgM Antibody tests (DXI Platform, Beckman Coulter, Brea, CA) in a Certified Analytics Professional and Clinical Laboratory Improvement Amendments certified clinical laboratory. The chemiluminescent assay quantitatively reports antibodies against the SARS-CoV-2 receptor binding domain in arbitrary units [AU]/mL, with values of ≥1 considered positive. The antibody transfer ratio was calculated as the infant IgG concentration divided by the maternal IgG concentration. Descriptive statistics were calculated. Linear regressions were used to identify factors significantly associated with the antibody transfer ratios and the infant IgG values. This study was institutional review board approved before its initiation.
Results
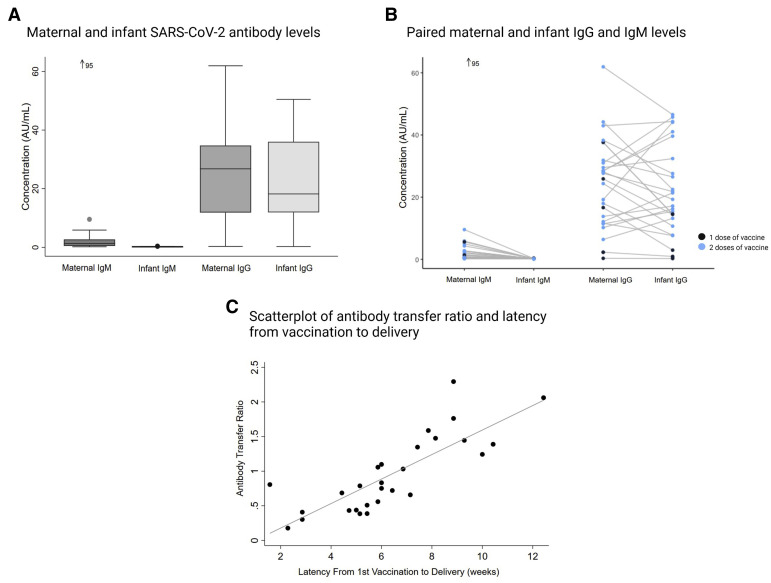
We collected matched maternal plasma and cord blood from 27 women who received a COVID-19 vaccination during pregnancy; they delivered 28 infants (1 twin pair). The average maternal age was 33±3 (mean±standard deviation) years; race and ethnicity was 75% non-Hispanic White and 11% Hispanic. The average gestational age at first vaccine dose was 33±2 weeks. For the vaccine type, 18 (64%) received the Pfizer vaccine, 6 (18%) received the Moderna vaccine, and 4 (14%) received a vaccine with the manufacturer unknown. Of note, 22 women (74%) received both vaccine doses before delivery with a mean latency of 6±3 weeks. About half of the women (15/27) and none of the infants had a positive IgM test (>1) (Figure , A). All but 1 woman (26/27) had a positive SARS-CoV-2 IgG test at the time of delivery. Only 3 infants did not have positive IgG tests (1 set of twins); these 2 women had received their first vaccine dose less than 3 weeks before delivery. The average maternal to infant IgG transfer ratio was 1.0±0.6 (Figure, B), however, an increased latency from vaccination to delivery (weeks) was associated with an increased transfer ratio (β=0.2; 95% confidence interval [CI], 0.1–0.2) (Figure, C). Correspondingly, having received the second vaccine dose before delivery was significantly associated with increased infant IgG levels (β=19.0; 95% CI, 7.1–30.8). Similarly, latency from vaccination to delivery was associated with increased infant IgG levels (β=2.9; 95% CI, 0.7–5.1).
Figure.
Maternal and infant SARS-CoV-2 antibodies and IgG transfer
A, Anti–SARS-CoV-2 antibody levels in maternal and umbilical cord (infant) blood following maternal COVID-19 vaccination (n=27 mothers, n=28 infants); B, Paired maternal and infant IgM and IgG levels of women with 1 dose (n=5) and 2 doses (n=22) of the vaccine during pregnancy. Paired infant IgM was 0.2 for 1 outlier maternal IgM of 95 AU/mL; C, The association between the latency (weeks) from first vaccine dose and the antibody transfer ratio (infant IgG to maternal IgG).
COVID-19, coronavirus disease 2019; Ig, immunoglobulin; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
Mithal. Antibody transfer after maternal coronavirus disease 2019 vaccination. Am J Obstet Gynecol 2021.
Conclusion
Most pregnant women who received a COVID-19 mRNA vaccine during the third trimester had transplacental transfer of IgG to the infant. The observed mean IgG transfer ratio demonstrated that infant antibody levels are about equal to the maternal levels. This is slightly lower than what has been reported for the pertussis vaccinations (1.19–1.36),5 but greater than the transfer ratio following SARS-CoV-2 infection (0.72±0.57).4 A novel finding is that the transfer ratio seems to increase with latency from vaccination. These data suggest, at least among women in their third trimester, that earlier vaccination may produce a greater infant immunity, the immunobiology of which requires further study. Notably, owing to vaccine eligibility and timing, most women included herein were healthcare workers in their third trimester. Future research, including a more diverse cohort of women and women who received vaccination earlier in their pregnancy, is needed. Nevertheless, these results show promising evidence for passive immunity against SARS-CoV-2 in newborns after maternal receipt of COVID-19 mRNA vaccinations.
Acknowledgments
We acknowledge support from Friends of Prentice (J.A.G.), the Stanley Manne Children’s Research Institute (L.B.M.), and the National Institutes of Health’s National Center for Advancing Translational Sciences under grant number UL1TR001422. Individual authors are supported by the National Institute of Biomedical Imaging and Bioengineering under grant number K08EB030120 (J.A.G.) and the National Institute of Allergy and Infectious Diseases under grant number K23AI139337 (L.B.M.). The Figure was produced with biorender.com. The authors would like to thank the staff of the Prentice Women’s Hospital and Northwestern Memorial Hospital Department of Pathology, specifically the Chemistry Laboratory.
Footnotes
The authors report no conflict of interest.
This study received financial support from the Stanley Manne Children’s Research Institute (L.B.M.) and the Friends of Prentice (J.A.G.) for tracking, biospecimen processing, and antibody assays. The National Institutes of Health (NIH), under grant number UL1TR001422, provided support to the Northwestern University Clinical and Translational Sciences Institute, which provided infrastructure support (REDCap). The individual authors’ research time were supported by the NIH under grant numbers K08EB030120 (J.A.G.) and K23AI139337 (L.B.M).
References
- 1.Gall S.A. Vaccines for pertussis and influenza: recommendations for use in pregnancy. Clin Obstet Gynecol. 2008;51:486–497. doi: 10.1097/GRF.0b013e318181dde1. [DOI] [PubMed] [Google Scholar]
- 2.Gray K.J., Bordt E.A., Atyeo C., et al. COVID-19 vaccine response in pregnant and lactating women: a cohort study. Am J Obstet Gynecol. 2021 doi: 10.1016/j.ajog.2021.03.023. [Epub ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Atyeo C., Pullen K.M., Bordt E.A., et al. Compromised SARS-CoV-2-specific placental antibody transfer. Cell. 2021;184:628–642.e10. doi: 10.1016/j.cell.2020.12.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Edlow A.G., Li J.Z., Collier A.Y., et al. Assessment of maternal and neonatal SARS-CoV-2 viral load, transplacental antibody transfer, and placental pathology in pregnancies During the COVID-19 pandemic. JAMA Netw Open. 2020;3 doi: 10.1001/jamanetworkopen.2020.30455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Munoz F.M., Bond N.H., Maccato M., et al. Safety and immunogenicity of tetanus diphtheria and acellular pertussis (Tdap) immunization during pregnancy in mothers and infants: a randomized clinical trial. JAMA. 2014;311:1760–1769. doi: 10.1001/jama.2014.3633. [DOI] [PMC free article] [PubMed] [Google Scholar]