Summary
Angiotensin‐converting enzyme 2 (ACE2) has been an increasingly prevalent target for investigation since its discovery 20 years ago. The finding that it serves a counterregulatory function within the traditional renin–angiotensin system, implicating it in cardiometabolic health, has increased its clinical relevance. Focus on ACE2's role in cardiometabolic health has largely centered on its apparent functions in the context of obesity. Interest in ACE2 has become even greater with the discovery that it serves as the cell receptor for severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2), opening up numerous mechanisms for deleterious effects of infection. The proliferation of ACE2 within the literature coupled with its dual role in SARS‐CoV‐2 infection and obesity necessitates review of the current understanding of ACE2's physiological, pathophysiological, and potential therapeutic functions. This review highlights the roles of ACE2 in cardiac dysfunction and obesity, with focus on epicardial adipose tissue, to reconcile the data in the context of SARS‐CoV‐2 infection.
Keywords: ACE2, cardiac injury, epicardial fat, inflammation
Abbreviations
- ACE
angiotensin‐converting enzyme
- ACE2
angiotensin‐converting enzyme 2
- ACE2KO
angiotensin‐converting enzyme 2 knockout
- ACEi
angiotensin‐converting enzyme inhibitor
- Ang I
angiotensin I
- Ang II
angiotensin II
- Ang‐(1‐7)
angiotensin‐(1‐7)
- Ang‐(1‐9)
angiotensin‐(1‐9)
- ARB
angiotensin receptor blockers
- ARDS
Acute respiratory distress syndrome
- AT1R
angiotensin II type 1 receptor
- AT2R
angiotensin II type 2 receptor
- BAT
brown adipose tissue
- BMI
body mass index
- COVID‐19
coronavirus disease 2019
- DIO
diet‐induced obesity
- EAT
epicardial adipose tissue
- HFmrEF
heart failure with mid‐range ejection fraction
- HFpEF
heart failure with preserved ejection fraction
- HFrEF
heart failure with reduced ejection fraction
- MasR
Mas receptor
- PAH
pulmonary arterial hypertension
- RAS
RENIN–angiotensin system
- rhACE2
recombinant human angiotensin‐converting enzyme 2
- SARS‐COV
severe acute respiratory syndrome associated coronavirus
- SARS‐CoV‐2
severe acute respiratory syndrome coronavirus 2
- UCP1
uncoupling protein 1
- WAT
white adipose tissue
1. INTRODUCTION
Angiotensin‐converting enzyme 2 (ACE2), a homolog of angiotensin‐converting enzyme (ACE), was first discovered from a cDNA library in 2000 and was shortly thereafter mapped to a quantitative trait locus on the X chromosome of rats. 1 , 2 , 3 Not much later, it was hypothesized that ACE2 countered ACE's effect on blood pressure as well as cardiovascular function more broadly. 4 , 5 The physiological relevance of ACE2 in comparison to previously discovered ACE is yet to be fully understood. The current coronavirus disease 2019 (COVID‐19) pandemic and the outbreak of severe acute respiratory syndrome coronavirus 2 (SARS‐CoV‐2) shifted the focus back on ACE2 and its role in infectious and cardiometabolic diseases. ACE2 is essential for SARS‐CoV‐2 to enter the host cell. 6 Recent clinical evidence links obesity to elevated risk for COVID‐19 infection and consequent poorer outcomes. 7 , 8 SARS‐CoV‐2 has piqued interest in understanding the role of ACE2 in adipose depots, especially cardiac fat (Figure 1). With the spotlight on ACE2, it is prudent to review the current understanding of its role in adipose tissue as well as possible links to COVID‐19 infection. The major focus of this review is to highlight the roles of ACE2 in (i) cardiometabolic disease, (ii) cardiac fat inflammation, and (iii) in the transdifferentiation of adipose depots.
FIGURE 1.
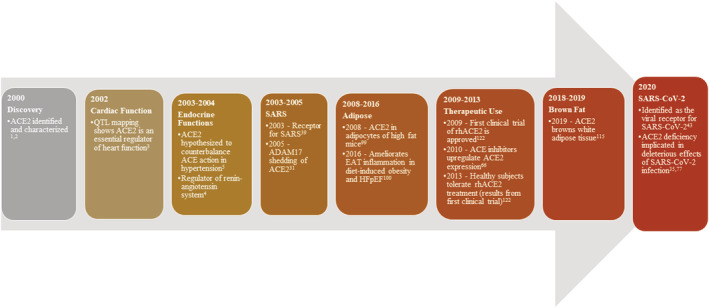
Timeline of important ACE2 discoveries: ACE2 was identified in 2000 as a homologue of ACE. This figure highlights the key discoveries made over the past 20 years that is attributed to the function of ACE2 in health and disease. ACE, angiotensin‐converting enzyme; ACE2, angiotensin‐converting enzyme 2; ADAM17, A disintegrin and metalloproteinase 17; Ang 1–7, angiotensin‐(1‐7); EAT, epicardial adipose tissue; HFpEF, heart failure with preserved ejection fraction; rhACE2, recombinant human angiotensin‐converting enzyme 2; SARS, severe acute respiratory syndrome; SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2; QTL, quantitative trait locus
1.1. ACE and ACE2
The renin–angiotensin system (RAS) plays a key role in the regulation of blood pressure. 9 Renin, produced by the kidneys, cleaves angiotensinogen to angiotensin I (Ang I), which is then converted to angiotensin II (Ang II) by ACE. 10 , 11 Through activation of the angiotensin II type 1 receptor (AT1R), Ang II promotes inflammation, salt and water reabsorption, oxidative stress, and vasoconstriction. 12 , 13 Angiotensin II type 2 receptors (AT2R) are believed to play a role in the protective arm of the RAS by exhibiting antihypertensive effects. 14 , 15 AT2R stimulation has been linked to increased production of vasodilators such as bradykinin, nitric oxide, and cyclic Guanosine monophosphate (GMP) and therefore aids in decreasing blood pressure. 16 , 17 , 18 , 19 , 20 Hence, agents such as angiotensin receptor blockers (ARB) and ACE inhibitors (ACEi) exhibit antihypertensive effects in humans. 21 , 22 , 23 ARBs act by stopping the downstream effects of Ang II by blocking AT1R, and the mechanism of ACEi is inhibition of the conversion of Ang I to Ang II. 24
ACE2, though functionally different from ACE, shares several network protein partners with ACE (Figure 2). 24 , 25 ACE2 acts by converting Ang II to angiotensin‐(1‐7) (Ang‐(1‐7)) as well as Ang I to angiotensin‐(1‐9) (Ang‐(1‐9)). 26 Ang‐(1‐7) acts on the G‐protein coupled Mas receptor (MasR), which signals through the PI3K–Akt pathway, to generate vasodilators such as nitric oxide, bradykinin, and prostaglandins. 27 , 28 Ang II induces the action of tumor necrosis factor alpha converting enzyme (TACE or ADAM17), which causes shedding of catalytically active ACE2 from tissue into the plasma. 29 , 30 , 31 The resulting soluble ACE2 can influence the cardiac remodeling process by altering integrin signaling. 30 , 32 ACE2 and Ang‐(1‐7) have anti‐inflammatory effects that counteract the Ang II and tumor necrosis factor alpha converting enzyme response in the heart and vasculature, effectively protecting the cardiovascular system from damage. 33 Thus, ACE2 plays a vital role in the RAS by exerting a protective effect on the cardiovascular system. 19 ACE2 is expressed ubiquitously and is highest in the small intestine, kidneys, heart, and adipose tissue, moderate in the lungs, liver, and colon, and lowest in the blood, bone marrow, and brain. 34 , 35 Though ACE2 tissue expression does not appear to vary greatly between sexes, races, or ages, 34 recent studies do caution that ACE2 expression in the nasal epithelium was lower in children compared to adults. 36 ACE2 levels are altered in renal, heart, and other disease states. 37 , 38 Thus, Ang II, ACE, ACE2, and Ang‐(1‐7) are all significant components of the RAS and involved in regulating blood pressure, electrolyte balance, and vascular resistance. 19
FIGURE 2.
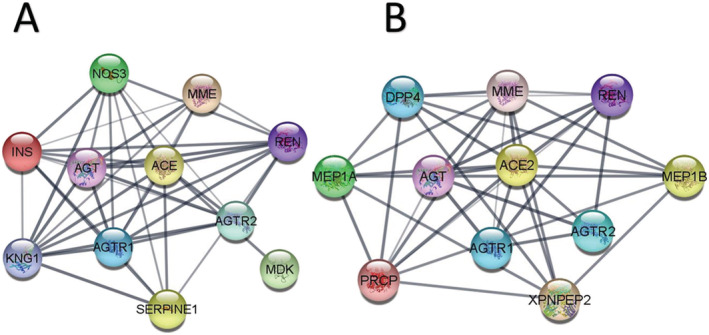
Protein–protein interaction network of ACE and ACE2: ACE and ACE2 have opposing physiological functions. The figure represents some of their protein network partners. Network pathway was developed using STRING database in CytoScape v3.8.0. (a) ACE or (b) ACE2 was used as the seed to identify interacting proteins. The protein interaction network is visualized in the form of a graphical network with the protein molecules forming the nodes and the interactions forming the edges. Some interactions that are connected by multiple lines between two nodes indicate interactions that are derived from more than one source of information. Proteins shared by both ACE and ACE2 are MME, membrane mettaloendopeptidase; REN, renin; AGTR1, angiotensin II receptor type 1; AGTR2, angiotensin II receptor type 2; AGT, angiotensinogen. Network genes unique to ACE are NOS3, nitric oxide synthase 3, INS, insulin, SERPINE1, serpin family E member 1; MDK, midkine; KNG1, kininogen‐1. Network genes unique to ACE2 are DPP4, dipeptidyl peptidase 4; MEP1A, meprin A subunit A; MEP1B, meprin A subunit B; PRCP, lysosomal pro‐X carboxypeptidase precursor; XPNPEP2, X‐prolyl aminopeptidase 2
1.2. SARS‐CoV‐2 and ACE2
The current interest in ACE2 piqued with the knowledge that it serves as the key receptor for SARS‐CoV‐2 infection in humans. 39 , 40 The entry of SARS‐CoV‐2 and SARS‐CoV into cells is facilitated by an interaction between the viral spike protein and the extracellular domains of transmembrane ACE2 in type II alveolar cells. 41 , 42 , 43 ACE2 in type II alveolar epithelial cells of the lungs seem to play a protective role by preventing polarization towards inflammatory status. 40 , 44 Infection with SARS‐CoV‐2, however, can lead to pulmonary edema and development of hyaline membranes in the lungs that lead to acute lung injury and acute respiratory distress syndrome (ARDS). 45 , 46 , 47 These results could be worse in individuals with obesity as a result of reduced lung compliance, increased vascular permeability, and increased inflammatory mediators. 7 , 48 , 49 , 50 The binding affinity of SARS‐CoV‐2 with ACE2 appears to be stronger than SARS‐CoV, which may explain the larger global impact of SARS‐CoV‐2 infection compared to SARS‐CoV. 51 , 52 The binding interaction between SARS‐CoV‐2 and transmembrane ACE2 leads to decreased ACE2 expression. 43 , 53 Earlier rodent studies have shown Ang II infusion downregulated surface ACE2 expression in murine myocardial tissue leading to myocardial injury. 54 Also, a recent small case study (12 patients) reported elevated plasma Ang II levels strongly associated with viral load in SARS‐CoV‐2 patients with pneumonia. Though ACE2 expression or activity was not assessed in this study, the authors suggested the elevation in Ang II was attributed to imbalances in RAS. 55 Thus, it may be hypothesized that tissue ACE2 downregulation and RAS imbalance might contribute to the development of multiorgan damage attributed to SARS‐CoV‐2 infections. 56 A decreased level of ACE2 secondary to SARS‐CoV‐2 infection followed by an increase in Ang II can exacerbate cardiovascular disease symptoms or promote further disease complications. 57 , 58 Nearly all of the most common comorbidities associated with severe outcomes due to SARS‐CoV‐2 infections are conditions in which ACE2 is known to be involved, such as diabetes, hypertension, and obesity. 59 , 60 , 61 Because ACE2 is greatly expressed in the heart and lungs, this infection has deleterious effects in these organs, including heart failure and ARDS. 62 , 63 Despite tissue expression of ACE2 not varying between sex or age, 34 men and the elderly have increased risk of severe disease resulting from SARS‐CoV‐2 infection. 64 Further, patients using ACEi and ARB medications, which have been shown to increase ACE2 expression in the heart, liver, and kidneys, 21 , 65 , 66 do not appear to have increased risk for developing SARS‐CoV‐2 infection, nor do they seem to have an increased risk for severe disease. 67 , 68 The lack of a correlation between initial ACE2 expression and disease severity coupled with the known largest comorbidities associated with SARS‐CoV‐2 suggests that reduced ACE2 activity may play a key role in increasing disease severity rather than the initial high levels of ACE2 expression leading to worsened prognosis. However, these findings do not rule out the possibility that some conditions which produce elevated ACE2 expression could result in increased viral proliferation and thus more severe infection. For instance, with recent studies indicating differing levels of ACE2 expression in nasal epithelium between children and adults, 36 more studies at the level of single cells are needed to further understand the tissue/cell distribution of ACE2 and its influence on SARS‐CoV‐2 infection. There are several emerging therapeutic measures being proposed targeting ACE2 to combat SARS‐CoV‐2. Some examples of the studies include using soluble recombinant human ACE2 (rhACE2) or ARBs targeting cardiac and kidney tissues to counteract the effects of Ang II. 35 , 69 , 70
2. ACE2 AND CARDIOVASCULAR DISEASE
2.1. Heart failure and ACE2
Heart failure affects at least 26 million people worldwide and 5.7 million residents of the United States. 71 This syndrome has been classified into three subtypes: heart failure with altered ejection fractions, abnormal natriuretic peptide levels, and the presence of structural heart disease and diastolic dysfunction. 71 Heart failure with altered ejection fractions can be further classified into heart failure with reduced ejection fraction (HFrEF), heart failure with preserved ejection fraction (HFpEF), and heart failure with mid‐range ejection fraction (HFmrEF). 72 Though not proven in humans, ACE2, Ang II, and Ang‐(1‐7) have been determined to play a significant role in the pathology of both HFpEF and HFrEF in murine models and may also have potential therapeutic effects. 54 , 73 These positive effects correspond to degradation of Ang II leading to reduced cardiac remodeling that is seen in heart failure. 54 , 73
2.2. HFpEF and ACE2/Ang‐(1‐7)
HFpEF accounts for up to 54% of heart failure patients in western populations. 74 HFpEF is usually associated with diastolic filling abnormalities. 75 Several studies have shown loss or downregulation of ACE2 leads to greater diastolic dysfunction as well as cardiac dysfunction, cardiac hypertrophy, and fibrosis due to increased presence of Ang II. 76 A downregulation of ACE2 may occur secondary to a viral infection, such as SARS‐CoV‐2, or from genetic variations. 77 , 78 If an individual is infected with SARS‐CoV‐2, a serious complication could be HFpEF, resulting from the redirection of ACE2 from its normal physiological function to serve as the SARS‐CoV‐2 viral receptor. 41 With lower ACE2 levels secondary to SARS‐CoV‐2 infection, Ang II levels would be expected to rise in the patient. Elevated Ang II leads to hypertension and worsened cardiovascular function that could induce harmful effects to the cardiac health and overall health of the individual. A recent study concluded that ACE2 in the form of recombinant ACE2 affecting murine hearts helps to mediate and resolve several pathologic changes attributed to Ang II; recombinant ACE2 reduced myocardial hypertrophy and fibrosis and corrected diastolic dysfunction. 54 , 79 This finding is significant and demonstrates that ACE2 plays a major role as a cardioprotective enzyme in healthy individuals. Therefore, recombinant ACE2 may be a potential therapeutic treatment for HFpEF when endogenous ACE2 levels are diminished. These results, however, have been obtained using murine models, and further investigations are required to prove the translatability of recombinant ACE2's potential therapeutic benefits in human HFpEF. Additionally, the possibility of rhACE2 to act as a decoy to scavenge viral particles in patients with HFpEF infected with SARS‐CoV‐2 patients also warrants further investigations.
2.3. HFrEF and ACE2/Ang‐(1‐7)
HFrEF, sometimes called “systolic dysfunction,” is not as prevalent as HFpEF; however, it remains a significant type of heart failure in the United States. 71 HFrEF is defined as left ventricular ejection fraction ≤40%. 80 Ang II can have detrimental effects on the heart, including cardiomyocyte toxicity, that can lead to development of HFrEF. 81 Serum ACE2 levels increase significantly in patients with hypertension and HFrEF but do not increase in HFpEF. 82 This suggests serum ACE2 is a biomarker for systolic dysfunction and HFrEF. 82 The most widespread approach currently utilized to treat HFrEF is neurohormonal control through a combination of ACEi or ARBs with a beta blocker. 80 It was demonstrated that using pulmonary artery pressure guided management, in conjunction with the recommended ACEi/ARB with beta blocker administration, decreased hospitalizations and mortality in patients with HFrEF. 80 Because Ang II is detrimental in patients experiencing HFrEF, a study may be warranted examining rhACE2 administration for possible benefits. The future study could investigate if rhACE2 possesses therapeutic effects in combination with pulmonary artery pressure guided management to decrease patient usage of ACEi/ARBs with beta blockers or utilize ACE2's previously known cardioprotective function in individuals with HFrEF.
2.4. Protective arm of the RAS and sex differences in obesity‐induced hypertension
ACE2 has also been implicated in sex differences observed in obesity‐induced hypertension which may be attributed to differential expression of either adipose ACE2 or MasR. ACE2 deficiency in adipose tissue augments increased systolic blood pressure in high‐fat diet fed female mice but not male mice. 83 This protection in female mice is postulated to involve the promoter upregulation of ACE2 by 17‐β‐estradiol binding to estrogen‐receptor‐α. 84 Other studies have shown that MasR deficiency augments obesity‐induced hypertension in male mice and abolishes protection from obesity‐induced hypertension in female mice. 85 , 86 This was because male obese mice had increased plasma Ang II levels which could be suppressed by ARBs such as losartan, whereas obese females had higher ACE2 expression in the adipose tissue and a higher plasma Ang‐(1‐7). Hence, the MasR antagonist, D‐Ala‐Ang‐(1–7), was shown to abolish the protection against increased systolic blood pressure in obese female mice. 86 Thus, it seems that the ACE2/Ang‐(1‐7)/MasR axis of the RAS provides protection from obesity‐induced hypertension in a sex‐dependent manner, something that should not be ignored in the context of SARS‐CoV‐2 infection.
3. ACE2 AND CARDIAC FAT
3.1. Epicardial adipose tissue and the heart
In humans, epicardial adipose tissue (EAT) can surround the myocardium and be present in relatively large quantities. This is in contrast to murine EAT which is localized to the atrioventricular groove and is present in relatively small quantities. 87 EAT is contiguous with the myocardium, making it sensible to think that EAT may influence cardiac function, 88 an effect that would be prominent in cases of obesity as a result of increased EAT thickness. 89 , 90 This thought is strengthened by the fact that EAT and the myocardium share the same microcirculation, providing an easy mechanism for EAT secretions to be transferred to the myocardium. 88 Furthermore, it has recently been shown that EAT adipocytes can release exosomes that can enter cardiac cells via endocytosis. 91 , 92 , 93 This opens up numerous mechanisms for EAT‐mediated effects on cardiac function, particularly via the transfer of microRNAs from EAT to the myocardium. 88 , 94 Of interest are the metabolic differences between EAT and other adipose tissue depots. EAT primarily stores saturated fatty acids, whereas other adipose tissues primarily store unsaturated fatty acids. 95 EAT is also more inclined towards lipolysis, resulting in a larger release of free fatty acids in comparison to other adipose tissue depots. 96 It has been hypothesized that this function provides EAT the advantage of supplying free fatty acids to the myocardium during times of high energy demand, though the evidence of this is presently lacking. EAT's proximity to the heart as well as its mechanisms for cardiac interactions indicate that EAT likely plays a critical role in the pathophysiology of cardiometabolic diseases. 97
3.2. ACE2, EAT inflammation, and link to SARS‐CoV‐2
The role of ACE2 in obesity and adipose tissue inflammation and its resulting cardiac effects has become a pressing topic because of the outbreak of SARS‐CoV‐2 and the knowledge that adipocytes can express ACE2 in a diet‐dependent manner. 98 There is a current interest in understanding the critical role for the ACE2/Ang‐(1‐7)/MasR axis in EAT inflammation. It has been seen that ACE2 knockout (ACE2KO) mice have greater EAT inflammation than wild‐type mice with diet‐induced obesity (DIO), indicating that the ACE2/Ang‐(1‐7)/MasR axis is essential in preventing EAT inflammation in response to DIO. 99 There was an increase in macrophage count as well as a polarization of macrophage phenotype towards proinflammatory status in EAT of both ACE2KO‐DIO mice and patients with obesity who suffered HFpEF. 99 The mechanism by which EAT inflammation led to HFpEF was attributed to increased insulin resistance coupled with increased glucose intolerance. 99 Although adipose tissue inflammation was already known to be associated with obesity‐induced insulin resistance, 100 it was found that despite having less severe obesity, the ACE2KO‐DIO mice had greater insulin resistance in comparison to wild‐type DIO mice. 99 , 101 This indicates that the ACE2/Ang‐(1‐7)/MasR axis itself plays a role in the regulation of insulin sensitivity, independent of the severity of obesity. The most critical finding is that the administration of Ang‐(1‐7) prevented the observed EAT inflammation and insulin resistance that was characteristic of the HFpEF phenotype. 99 This points towards Ang‐(1‐7) having therapeutic potential as a cardioprotective treatment in obesity.
The high prevalence of comorbidities related to EAT inflammation, particularly obesity, in SARS‐CoV‐2 infected cases raises the possibility that cardiac injury is in part a result of the suppression of anti‐inflammatory action by the ACE2/Ang‐(1‐7)/MasR axis. 60 , 61 , 102 , 103 This is partially supported by the observation of cytokine storms in patients with severe cases of SARS‐CoV‐2. 104 , 105 Factors indicative of systemic inflammation such as interleukin 6, tumor necrosis factor alpha, and interleukin 10 are all part of cytokine storm in patients with SARS‐CoV‐2 as well as elevated in EAT during DIO, especially EAT without ACE2. 99 , 106 , 107 Obesity induces EAT inflammation, an effect that is ameliorated by Ang‐(1‐7) action on MasR. 99 The downregulation of ACE2 resulting from SARS‐CoV‐2 infection reduces this action due to reduced conversion of Ang II to Ang‐(1‐7) by ACE2. 56 , 57 This leads to the possibility of EAT‐mediated cardiac injury. 103 Interestingly, it was also shown that DIO leads to greater ACE2 expression in EAT. 99 This opens up the possibility for EAT in patients with obesity to serve as a hub for viral infection that could mediate infection of the heart, although this remains a speculation. Although risk factors for EAT‐mediated cardiac injury are greater in SARS‐CoV‐2 patients with obesity, this result is not limited to patients with obesity; present data show that visceral adiposity, although moderately associated with BMI (body mass index), can vary greatly within a BMI class while also serving as a better predictor of cardiometabolic risk. 108 For instance, Asian American women have been shown to have comparatively larger stores of visceral adipose tissue even after adjustments made for age and total adiposity. 109 Computed tomographic (CT) imaging of the EAT of patients with SARS‐CoV‐2 showed lower CT threshold attenuation values for the EAT in severe cases of SARS‐CoV‐2. 110 , 111 This indicates that EAT inflammation itself is associated with severity of disease. Overall, patients assessed as nonobese by BMI measures may still have increased visceral adiposity which increases the risk of EAT inflammation. Further, EAT's sharing of a microcirculation with the heart and its capability to release exosomes that can enter cardiac cells open up numerous mechanisms by which EAT may contribute to/mediate SARS‐CoV‐2's entry into the heart, causing direct cardiac effects. 88 , 103 Interestingly, the cytokine levels observed in SARS‐CoV‐2 cytokine storm have also been shown to be negatively correlated with T cell counts and positively correlated with T cell exhaustion measures. 106 This raises the possibility that patients with a predisposition to heightened inflammatory cytokine levels, such as individuals with larger EAT depots, could suffer worsened outcomes which result from T cell depletion and exhaustion. Some have suggested that this could mean that vaccines for SARS‐CoV‐2 could be less effective in individuals with obesity due to a weakened immune response. 8 Importantly, this thought may also apply to individuals assessed as nonobese by BMI measures who still have increased visceral adiposity such as Asian American women.
Although there is evidence that the ACE2/Ang‐(1‐7)/MasR axis is involved in the pathophysiology of EAT‐mediated cardiac dysfunction and that its regulation has therapeutic potential, there are still significant questions that necessitate further studies. First, whereas the team responsible for the previously mentioned study acknowledged the necessity of determining whether the inflammatory effects of ACE2 deletion in obesity extend to other adipose tissue depots, 99 others have pointed out that it is possible that ACE2 deletion could result in cardiac dysfunction stemming from myocardial inflammation independent of EAT inflammation. 112 , 113 Determination of the independence of the two is critical for determining the extent to which ACE2/Ang‐(1‐7) action in EAT specifically is responsible for cardioprotection. The second major question is whether Ang‐(1‐7) administration is the best option in the pursuit of developing a therapeutic approach to EAT‐mediated cardiac dysfunction in obesity. A potential candidate to replace Ang‐(1‐7) treatment is rhACE2, an alternative that recent study has shown to reduce markers of adipose tissue inflammation. 114 The added benefit of treatment with rhACE2 would be the reduction of Ang II levels, an effect that has also already been observed. 54 , 114 The limitations of the current data on using rhACE2 as a treatment for EAT inflammation are that it is not specific to EAT, a tissue that has characteristics which generally differentiate it from other adipose tissues, and that it only measured molecular markers of inflammation as opposed to the actual inflammatory status of the tissue. Garnering a more complete understanding of the treatment is especially pertinent for SARS‐CoV‐2 infected individuals. Treatment using soluble rhACE2 has been raised as a potential therapeutic option that has the potential to redirect SARS‐CoV‐2 binding away from membrane‐bound ACE2; binding of SARS‐CoV‐2 to the soluble rhACE2 would allow membrane‐bound ACE2 to continue functioning within tissue, avoiding detrimental effects stemming from reduced ACE2/Ang‐(1‐7)/MasR axis action. 69 Considering that rhACE2 has been suggested for treating both obesity‐induced cardiac dysfunction and SARS‐CoV‐2 infection independent of each other, it may serve as a valuable route towards treatment of the severe cases of SARS‐CoV‐2 infection that are more common in patients with obesity. 7 However, one of rhACE2's chief limitations presently is the lack of knowledge regarding its effects on adipose tissue, particularly its potential to induce the browning of white adipose tissue (WAT) which could negatively affect cardiac function. 114
3.3. rhACE2's role in adipose tissue transdifferentiation
Interest in brown adipose tissue (BAT) stems primarily from its expression of uncoupling protein‐1 (UCP1), which bypasses the electron transport chain in a mechanism that results in less adenosine triphosphate production, more substrate oxidation, and greater thermogenesis. 115 Of growing interest is the presence of UCP1‐expressing adipocytes that are heterogeneously embedded in WAT depots which are referred to as beige (or brite) adipocytes. EAT is one such depot, now being characterized as largely beige in type. 116 , 117 The administration of rhACE2 has been shown to seemingly induce the transdifferentiation of WAT to BAT. 114 There is little quantification of the extent of rhACE2‐induced browning of WAT, leaving the possibility that prolonged treatment in SARS‐CoV‐2 infected individuals could result in heightened fever resulting from increased UCP1‐instigated thermogenesis, though the role of BAT in immune‐induced hyperthermia is now debated in the literature. 118 Hence, although rhACE2 use may potentially be therapeutically beneficial as a treatment for obesity, its use in SARS‐CoV‐2 infected patients may be a cause for concern.
Considering that EAT is inclined towards the release of free fatty acids, 96 and that the myocardium utilizes free fatty acids as its primary source of energy, 119 the proliferation of beige tissue around the myocardium could have adverse effects on myocardial function stemming from hampered metabolic activity. Such a result could be catastrophic in patients with SARS‐CoV‐2 infection because it would add to the observed impairment of cardiac function in these patients. 120 Although rhACE2 has been studied in humans since 2009, 121 studies in the context of SARS‐CoV‐2 infection have not yet been investigated. The currently ongoing clinical trial being conducted in Austria, Germany, and Denmark will hopefully elucidate the cardiac effects of rhACE2 treatment in the context of SARS‐CoV‐2 infection. 122
4. CONCLUSIONS
ACE2 is a critical mediator of both cardiometabolic health and the entry of SARS‐CoV‐2 into human cells. These two functions of ACE2 are not independent of each other; the former serves as an explanation of the observed detrimental effects resulting from the latter. ACE2 has physiological and pathophysiological roles in both cardiac dysfunction and obesity, both of which can be linked to ACE2's functions in EAT (Figure 3). This understanding is crucial as therapeutic approaches to treating SARS‐CoV‐2 infection move forward. Mitigation of the altered RAS balance stemming from infection is a valuable method to reduce the severity of disease, one that may serve the added benefits of diverting viral binding away from membrane‐bound, catalytically active ACE2 as well as elucidating the potential for such treatment in cardiometabolic diseases in the future. In such situations, the role of ACE2 in obesity, particularly in epicardial fat, as well as observed sex differences in the RAS should not be ignored.
FIGURE 3.
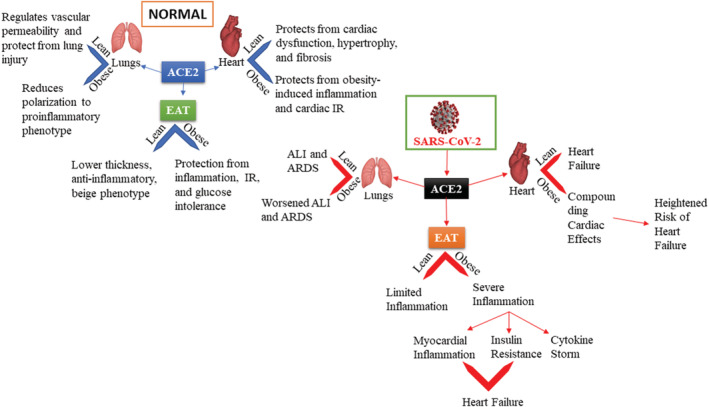
Consequences of SARS‐CoV‐2 infection on ACE2 function in lean and obese individuals. ACE2 plays a key role in the entry of the SARS‐CoV‐2 in humans. This figure represents predicted changes in ACE2 function in normal and infected individuals. ACE2 function is modulated by the patient's obesity status. (a) Normal function of ACE2 in the lungs, heart, and EAT. (b) Deleterious effects of SARS‐CoV‐2 in the lungs, heart, and EAT. ACE2, angiotensin‐converting enzyme 2; ALI, acute lung injury; ARDS, acute respiratory distress syndrome; EAT, epicardial adipose tissue; SARS‐CoV‐2, severe acute respiratory syndrome coronavirus 2
CONFLICT OF INTEREST
No conflict of interest statement.
ACKNOWLEDGEMENTS
NS acknowledges support from National Institute on Aging Grant 1R15AG051062‐01 (NS); BF acknowledges support from the MU Creative Discovery Award and National Aeronautics and Space Administration's West Virginia Space Grant Consortium (NNX15AI01H).
Flinn B, Royce N, Gress T, Chowdhury N, Santanam N. Dual role for angiotensin‐converting enzyme 2 in Severe Acute Respiratory Syndrome Coronavirus 2 infection and cardiac fat. Obesity Reviews. 2021;22:e13225. 10.1111/obr.13225
Brendin Flinn and Nicholas Royce contributed equally to this work.
REFERENCES
- 1. Tipnis SR, Hooper NM, Hyde R, Karran E, Christie G, Turner AJ. A human homolog of angiotensin‐converting enzyme. Cloning and functional expression as a captopril‐insensitive carboxypeptidase. J Biol Chem. 2000;275(43):33238‐33243. [DOI] [PubMed] [Google Scholar]
- 2. Donoghue M, Hsieh F, Baronas E, et al. A novel angiotensin‐converting enzyme‐related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1–9. Circ Res. 2000;87:E1‐E9. [DOI] [PubMed] [Google Scholar]
- 3. Crackower MA, Sarao R, Oudit GY, et al. Angiotensin‐converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417(6891):822‐828. [DOI] [PubMed] [Google Scholar]
- 4. Burrell LM, Johnston CI, Tikellis C, Cooper ME. ACE2, a new regulator of the renin–angiotensin system. Trends Endocrinol Metab. 2004;15(4):166‐169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Yagil Y, Yagil C. Hypothesis: ACE2 modulates blood pressure in the mammalian organism. Hypertension. 2003;41(4):871‐873. [DOI] [PubMed] [Google Scholar]
- 6. Ortiz Bezara ME, Thurman A, Pezzulo AA, et al. Heterogeneous expression of the SARS‐Coronavirus‐2 receptor ACE2 in the human respiratory tract. bioRxiv. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Akoumianakis I, Filippatos T. The renin–angiotensin‐aldosterone system as a link between obesity and coronavirus disease 2019 severity. Obes Rev. 2020;21(9):e13077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Popkin BM, Du S, Green WD, et al. Individuals with obesity and COVID‐19: a global perspective on the epidemiology and biological relationships. Obes Rev. 2020;21:e13128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Hall JE. The renin–angiotensin system: renal actions and blood pressure regulation. Compr Ther. 1991;17(5):8‐17. [PubMed] [Google Scholar]
- 10. Santos RAS, Oudit GY, Verano‐Braga T, Canta G, Steckelings UM, Bader M. The renin–angiotensin system: going beyond the classical paradigms. Am J Physiol Heart Circ Physiol. 2019;316(5):H958‐H970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Ferrario CM. The renin–angiotensin system: importance in physiology and pathology. J Cardiovasc Pharmacol. 1990;15(Suppl 3):S1‐S5. [PubMed] [Google Scholar]
- 12. Chappell MC. Nonclassical renin–angiotensin system and renal function. Compr Physiol. 2012;2(4):2733‐2752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. de Barcelos Ubaldo Martins L, Jabour L, Vieira CC, Nery LCC, Dias RF, Simoes ESAC. Renin–angiotensin system (RAS) and immune system profile in specific subgroups with COVID‐19. Curr Med Chem. 2020. 10.2174/0929867327666200903113117 [DOI] [PubMed] [Google Scholar]
- 14. Steckelings UM, Kloet A, Sumners C. Centrally mediated cardiovascular actions of the angiotensin II type 2 receptor. Trends Endocrinol Metab. 2017;28(9):684‐693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Juillerat‐Jeanneret L. The other angiotensin II receptor: AT2R as a therapeutic target. J Med Chem. 2020;63(5):1978‐1995. [DOI] [PubMed] [Google Scholar]
- 16. Carey RM, Jin XH, Siragy HM. Role of the angiotensin AT2 receptor in blood pressure regulation and therapeutic implications. Am J Hypertens. 2001;14(11):98S‐102S. [DOI] [PubMed] [Google Scholar]
- 17. Siragy HM, Carey RM. The subtype‐2 (AT2) angiotensin receptor regulates renal cyclic guanosine 3′, 5′‐monophosphate and AT1 receptor‐mediated prostaglandin E2 production in conscious rats. J Clin Invest. 1996;97(8):1978‐1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Siragy HM, Carey RM. The subtype 2 (AT2) angiotensin receptor mediates renal production of nitric oxide in conscious rats. J Clin Invest. 1997;100(2):264‐269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Cheng H, Wang Y, Wang GQ. Organ‐protective effect of angiotensin‐converting enzyme 2 and its effect on the prognosis of COVID‐19. J Med Virol. 2020;92(7):726‐730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Ali Q, Wu Y, Hussain T. Chronic AT2 receptor activation increases renal ACE2 activity, attenuates AT1 receptor function and blood pressure in obese Zucker rats. Kidney Int. 2013;84(5):931‐939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Ferrario CM, Jessup J, Chappell MC, et al. Effect of angiotensin‐converting enzyme inhibition and angiotensin II receptor blockers on cardiac angiotensin‐converting enzyme 2. Circulation. 2005;111(20):2605‐2610. [DOI] [PubMed] [Google Scholar]
- 22. ACEIs or ARBs For Adults With Hypertension . Clinician's Guide. Rockville (MD: Comparative Effectiveness Review Summary Guides for Clinicians; 2007. [Google Scholar]
- 23. Batty JA, Tang M, Hall M, Ferrari R, Strauss MH, Hall AS. Blood pressure reduction and clinical outcomes with angiotensin‐converting enzyme inhibitors and angiotensin II receptor blockers: protocol for a systematic review and meta‐regression analysis. Syst Rev. 2018;7(1):131–141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Rice GI, Jones AL, Grant PJ, Carter AM, Turner AJ, Hooper NM. Circulating activities of angiotensin‐converting enzyme, its homolog, angiotensin‐converting enzyme 2, and neprilysin in a family study. Hypertension. 2006;48(5):914‐920. [DOI] [PubMed] [Google Scholar]
- 25. Horiuchi M, Iwanami J, Mogi M. Regulation of angiotensin II receptors beyond the classical pathway. Clin Sci (Lond). 2012;123(4):193‐203. [DOI] [PubMed] [Google Scholar]
- 26. Danilczyk U, Eriksson U, Crackower MA, Penninger JM. A story of two ACEs. J Mol Med (Berl). 2003;81(4):227‐234. [DOI] [PubMed] [Google Scholar]
- 27. Jiang T, Gao L, Lu J, Zhang YD. ACE2‐Ang‐(1‐7)‐Mas axis in brain: a potential target for prevention and treatment of ischemic stroke. Curr Neuropharmacol. 2013;11(2):209‐217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Povlsen AL, Grimm D, Wehland M, Infanger M, Kruger M. The vasoactive Mas receptor in essential hypertension. J Clin Med. 2020;9(1):267–278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Patel VB, Clarke N, Wang Z, et al. Angiotensin II induced proteolytic cleavage of myocardial ACE2 is mediated by TACE/ADAM‐17: a positive feedback mechanism in the RAS. J Mol Cell Cardiol. 2014;66:167‐176. [DOI] [PubMed] [Google Scholar]
- 30. Turner AJ. Chapter 25—ACE2 cell biology, regulation, and physiological functions. In: Unger T, Steckelings UM, dos Santos RAS, eds. The Protective Arm of the Renin Angiotensin System (RAS). Boston: Academic Press; 2015:185‐189. [Google Scholar]
- 31. Lambert DW, Yarski M, Warner FJ, et al. Tumor necrosis factor‐alpha convertase (ADAM17) mediates regulated ectodomain shedding of the severe‐acute respiratory syndrome‐coronavirus (SARS‐CoV) receptor, angiotensin‐converting enzyme‐2 (ACE2). J Biol Chem. 2005;280(34):30113‐30119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. English WR, Corvol P, Murphy G. LPS activates ADAM9 dependent shedding of ACE from endothelial cells. Biochem Biophys Res Commun. 2012;421(1):70‐75. [DOI] [PubMed] [Google Scholar]
- 33. Gaddam RR, Chambers S, Bhatia M. ACE and ACE2 in inflammation: a tale of two enzymes. Inflamm Allergy Drug Targets. 2014;13(4):224‐234. [DOI] [PubMed] [Google Scholar]
- 34. Li MY, Li L, Zhang Y, Wang XS. Expression of the SARS‐CoV‐2 cell receptor gene ACE2 in a wide variety of human tissues. Infect Dis Poverty. 2020;9(1):45–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Essig M, Matt M, Massy Z. The COVID‐19 outbreak and the angiotensin‐converting enzyme 2: too little or too much? Nephrol Dial Transplant. 2020;35(6):1073‐1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Patel AB, Verma A. Nasal ACE2 levels and COVID‐19 in children. JAMA. 2020;323(23):2386‐2387. [DOI] [PubMed] [Google Scholar]
- 37. Wang W, Patel VB, Parajuli N, et al. Heterozygote loss of ACE2 is sufficient to increase the susceptibility to heart disease. J Mol Med (Berl). 2014;92(8):847‐858. [DOI] [PubMed] [Google Scholar]
- 38. Soler MJ, Wysocki J, Batlle D. ACE2 alterations in kidney disease. Nephrol Dial Transplant. 2013;28(11):2687‐2697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Li W, Moore MJ, Vasilieva N, et al. Angiotensin‐converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426(6965):450‐454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203(2):631‐637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Walls AC, Park Y‐J, Tortorici MA, Wall A, McGuire AT, Veesler D. Structure, function, and antigenicity of the SARS‐CoV‐2 spike glycoprotein. Cell. 2020;181:281‐92.e6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Zhao Y, Zhao Z, Wang Y, Zhou Y, Ma Y, Zuo W. Single‐cell RNA expression profiling of ACE2, the receptor of SARS‐CoV‐2. Am J Respir Crit Care Med. 2020;202(5):756‐759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Zhou P, Yang X‐L, Wang X‐G, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270‐273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Imai Y, Kuba K, Rao S, et al. Angiotensin‐converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436(7047):112‐116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Ni W, Yang X, Yang D, et al. Role of angiotensin‐converting enzyme 2 (ACE2) in COVID‐19. Critical Care. 2020;24:422–431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Hrenak J, Simko F. Renin–angiotensin system: an important player in the pathogenesis of acute respiratory distress syndrome. Int J Mol Sci. 2020;21:8038–8056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Zhang X, Li S, Niu S. ACE2 and COVID‐19 and the resulting ARDS. Postgrad Med J. 2020;96(1137):403‐407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. de Lucena TMC, da Silva Santos AF, de Lima BR, de Albuquerque Borborema ME, de Azevedo Silva J. Mechanism of inflammatory response in associated comorbidities in COVID‐19. Diabetes Metab Syndr. 2020;14(4):597‐600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Motta‐Santos D, Santos RAS, Santos SHS. Angiotensin‐(1‐7) and obesity: role on cardiorespiratory fitness and COVID‐19 implications. Obesity (Silver Spring). 2020;28(10):1786–1789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Zbinden‐Foncea H, Francaux M, Deldicque L, Hawley JA. Does high cardiorespiratory fitness confer some protection against proinflammatory responses after infection by SARS‐CoV‐2? Obesity (Silver Spring). 2020;28(8):1378‐1381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Shang J, Ye G, Shi K, et al. Structural basis of receptor recognition by SARS‐CoV‐2. Nature. 2020;581(7807):221‐224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Yan R, Zhang Y, Li Y, Xia L, Guo Y, Zhou Q. Structural basis for the recognition of SARS‐CoV‐2 by full‐length human ACE2. Science. 2020;367(6485):1444‐1448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Verdecchia P, Cavallini C, Spanevello A, Angeli F. The pivotal link between ACE2 deficiency and SARS‐CoV‐2 infection. Eur J Intern Med. 2020;76:14‐20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Zhong J, Basu R, Guo D, et al. Angiotensin‐converting enzyme 2 suppresses pathological hypertrophy, myocardial fibrosis, and cardiac dysfunction. Circulation. 2010;122:717‐728.18 p following 28 [DOI] [PubMed] [Google Scholar]
- 55. Liu Y, Yang Y, Zhang C, et al. Clinical and biochemical indexes from 2019‐nCoV infected patients linked to viral loads and lung injury. Sci China Life Sci. 2020;63(3):364‐374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Wang Y, Wang Y, Luo W, et al. A comprehensive investigation of the mRNA and protein level of ACE2, the putative receptor of SARS‐CoV‐2, in human tissues and blood cells. Int J Med Sci. 2020;17(11):1522‐1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Oudit GY, Kassiri Z, Jiang C, et al. SARS‐coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. Eur J Clin Invest. 2009;39(7):618‐625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Aksoy H, Karadag AS, Wollina U. Angiotensin II receptors—impact for COVID‐19 severity. Dermatol Ther. 2020;e13989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Obukhov AG, Stevens BR, Prasad R, et al. SARS‐CoV‐2 infections and ACE2: clinical outcomes linked with increased morbidity and mortality in individuals with diabetes. Diabetes. 2020;69(9):1875‐1886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Richardson S, Hirsch JS, Narasimhan M, et al. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID‐19 in the New York City area. JAMA. 2020;323(20):2052‐2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Stefan N, Birkenfeld AL, Schulze MB, Ludwig DS. Obesity and impaired metabolic health in patients with COVID‐19. Nat Rev Endocrinol. 2020;16(7):341‐342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Imai Y, Kuba K, Penninger JM. Angiotensin‐converting enzyme 2 in acute respiratory distress syndrome. Cell Mol Life Sci. 2007;64(15):2006‐2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Lumbers ER, Delforce SJ, Pringle KG, Smith GR. The lung, the heart, the novel coronavirus, and the renin–angiotensin system; the need for clinical trials. Front Med (Lausanne). 2020;7:248–254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Borges do Nascimento IJ, Cacic N, Abdulazeem HM, et al. Novel coronavirus infection (COVID‐19) in humans: a scoping review and meta‐analysis. J Clin Med. 2020;9:941–954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Ishiyama Y, Gallagher PE, Averill DB, Tallant EA, Brosnihan KB, Ferrario CM. Upregulation of angiotensin‐converting enzyme 2 after myocardial infarction by blockade of angiotensin II receptors. Hypertension. 2004;43(5):970‐976. [DOI] [PubMed] [Google Scholar]
- 66. Huang ML, Li X, Meng Y, et al. Upregulation of angiotensin‐converting enzyme (ACE) 2 in hepatic fibrosis by ACE inhibitors. Clin Exp Pharmacol Physiol. 2010;37(1):e1‐e6. [DOI] [PubMed] [Google Scholar]
- 67. Mackey K, King VJ, Gurley S, et al. Risks and impact of angiotensin‐converting enzyme inhibitors or angiotensin‐receptor blockers on SARS‐CoV‐2 infection in adults: a living systematic review. Ann Intern Med. 2020;173(3):195‐203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Bean DM, Kraljevic Z, Searle T, et al. Angiotensin‐converting enzyme inhibitors and angiotensin II receptor blockers are not associated with severe COVID‐19 infection in a multi‐site UK acute hospital trust. Eur J Heart Fail. 2020;22(6):967‐974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69. Batlle D, Wysocki J, Satchell K. Soluble angiotensin‐converting enzyme 2: a potential approach for coronavirus infection therapy? Clin Sci (Lond). 2020;134(5):543‐545. [DOI] [PubMed] [Google Scholar]
- 70. Rico‐Mesa JS, White A, Anderson AS. Outcomes in patients with COVID‐19 infection taking ACEI/ARB. Curr Cardiol Rep. 2020;22(5):31–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Savarese G, Lund LH. Global public health burden of heart failure. Card Fail Rev. 2017;3(01):7‐11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Sama IE, Ravera A, Santema BT, et al. Circulating plasma concentrations of angiotensin‐converting enzyme 2 in men and women with heart failure and effects of renin–angiotensin‐aldosterone inhibitors. Eur Heart J. 2020;41(19):1810‐1817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Patel VB, Zhong JC, Grant MB, Oudit GY. Role of the ACE2/angiotensin 1‐7 axis of the renin–angiotensin system in heart failure. Circ Res. 2016;118(8):1313‐1326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Obokata M, Reddy YNV, Borlaug BA. Diastolic dysfunction and heart failure with preserved ejection fraction: understanding mechanisms by using noninvasive methods. JACC Cardiovasc Imaging. 2020;13(1 Pt 2):245‐257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Gladden JD, Linke WA, Redfield MM. Heart failure with preserved ejection fraction. Pflugers Arch. 2014;466(6):1037‐1053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Garabelli PJ, Modrall JG, Penninger JM, Ferrario CM, Chappell MC. Distinct roles for angiotensin‐converting enzyme 2 and carboxypeptidase A in the processing of angiotensins within the murine heart. Exp Physiol. 2008;93(5):613‐621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Gheblawi M, Wang K, Viveiros A, et al. Angiotensin‐converting enzyme 2: SARS‐CoV‐2 receptor and regulator of the renin–angiotensin system: celebrating the 20th anniversary of the discovery of ACE2. Circ Res. 2020;126:1456‐1474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Yang W, Huang W, Su S, et al. Association study of ACE2 (angiotensin I‐converting enzyme 2) gene polymorphisms with coronary heart disease and myocardial infarction in a Chinese Han population. Clin Sci (Lond). 2006;111(5):333‐340. [DOI] [PubMed] [Google Scholar]
- 79. Zhong J, Guo D, Chen CB, et al. Prevention of angiotensin II‐mediated renal oxidative stress, inflammation, and fibrosis by angiotensin‐converting enzyme 2. Hypertension. 2011;57(2):314‐322. [DOI] [PubMed] [Google Scholar]
- 80. Givertz MM, Stevenson LW, Costanzo MR, et al. Pulmonary artery pressure‐guided management of patients with heart failure and reduced ejection fraction. J am Coll Cardiol. 2017;70(15):1875‐1886. [DOI] [PubMed] [Google Scholar]
- 81. Tan LB, Jalil JE, Pick R, Janicki JS, Weber KT. Cardiac myocyte necrosis induced by angiotensin II. Circ Res. 1991;69(5):1185‐1195. [DOI] [PubMed] [Google Scholar]
- 82. Úri K, Fagyas M, Kertész A, et al. Circulating ACE2 activity correlates with cardiovascular disease development. J Renin Angiotensin Aldosterone Syst. 2016;17(4):1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83. Shoemaker R, Tannock LR, Su W, et al. Adipocyte deficiency of ACE2 increases systolic blood pressures of obese female C57BL/6 mice. Biol Sex Differ. 2019;10(1):45–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Wang Y, Shoemaker R, Thatcher SE, Batifoulier‐Yiannikouris F, English VL, Cassis LA. Administration of 17β‐estradiol to ovariectomized obese female mice reverses obesity‐hypertension through an ACE2‐dependent mechanism. Am J Physiol Endocrinol Metab. 2015;308(12):E1066‐E1075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85. Wang Y, Shoemaker R, Powell D, Su W, Thatcher S, Cassis L. Differential effects of Mas receptor deficiency on cardiac function and blood pressure in obese male and female mice. Am J Physiol Heart Circ Physiol. 2017;312(3):H459‐h68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Gupte M, Thatcher SE, Boustany‐Kari CM, et al. Angiotensin converting enzyme 2 contributes to sex differences in the development of obesity hypertension in C57BL/6 mice. Arterioscler Thromb Vasc Biol. 2012;32(6):1392‐1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Iacobellis G, Corradi D, Sharma AM. Epicardial adipose tissue: anatomic, biomolecular and clinical relationships with the heart. Nat Clin Pract Cardiovasc Med. 2005;2(10):536‐543. [DOI] [PubMed] [Google Scholar]
- 88. Patel VB, Shah S, Verma S, Oudit GY. Epicardial adipose tissue as a metabolic transducer: role in heart failure and coronary artery disease. Heart Fail Rev. 2017;22(6):889‐902. [DOI] [PubMed] [Google Scholar]
- 89. Monfort A, Inamo J, Fagour C, et al. Epicardial fat accumulation is an independent marker of impaired heart rate recovery in obese patients with obstructive sleep apnea. Clin Res Cardiol. 2019;108(11):1226‐1233. [DOI] [PubMed] [Google Scholar]
- 90. Gorter TM, van Woerden G, Rienstra M, et al. Epicardial adipose tissue and invasive hemodynamics in heart failure with preserved ejection fraction. JACC Heart Fail. 2020;8(8):667‐676. [DOI] [PubMed] [Google Scholar]
- 91. Pellegrini L, Foglio E, Pontemezzo E, Germani A, Russo MA, Limana F. Cardiac repair: the intricate crosstalk between the epicardium and the myocardium. Curr Stem Cell Res Ther. 2020;15(8):661‐673. [DOI] [PubMed] [Google Scholar]
- 92. Wen Z, Li J, Fu Y, Zheng Y, Ma M, Wang C. Hypertrophic adipocyte‐derived exosomal miR‐802‐5p contributes to insulin resistance in cardiac myocytes through targeting HSP60. Obesity (Silver Spring). 2020;28:1932‐1940. [DOI] [PubMed] [Google Scholar]
- 93. Zhao L, Ma Z, Guo Z, Zheng M, Li K, Yang X. Analysis of long non‐coding RNA and mRNA profiles in epicardial adipose tissue of patients with atrial fibrillation. Biomed Pharmacother. 2020;121:109634–109644. [DOI] [PubMed] [Google Scholar]
- 94. Thomou T, Mori MA, Dreyfuss JM, et al. Adipose‐derived circulating miRNAs regulate gene expression in other tissues. Nature. 2017;542(7642):450‐455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Pezeshkian M, Noori M, Najjarpour‐Jabbari H, et al. Fatty acid composition of epicardial and subcutaneous human adipose tissue. Metab Syndr Relat Disord. 2009;7(2):125‐131. [DOI] [PubMed] [Google Scholar]
- 96. Marchington JM, Mattacks CA, Pond CM. Adipose tissue in the mammalian heart and pericardium: structure, foetal development and biochemical properties. Comp Biochem Physiol B. 1989;94(2):225‐232. [DOI] [PubMed] [Google Scholar]
- 97. Iacobellis G, Barbaro G. Epicardial adipose tissue feeding and overfeeding the heart. Nutrition. 2019;59:1‐6. [DOI] [PubMed] [Google Scholar]
- 98. Gupte M, Boustany‐Kari C, Bharadwaj K, et al. ACE2 is expressed in mouse adipocytes and regulated by a high‐fat diet. American Journal of Physiology Regulatory, Integrative and Comparative Phys Ther. 2008;295:R781‐R788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Patel VB, Mori J, McLean BA, et al. ACE2 deficiency worsens epicardial adipose tissue inflammation and cardiac dysfunction in response to diet‐induced obesity. Diabetes. 2016;65(1):85‐95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Hotamisligil GS, Shargill NS, Spiegelman BM. Adipose expression of tumor necrosis factor‐alpha: direct role in obesity‐linked insulin resistance. Science. 1993;259(5091):87‐91. [DOI] [PubMed] [Google Scholar]
- 101. Van Gaal LF, Mertens IL, De Block CE. Mechanisms linking obesity with cardiovascular disease. Nature. 2006;444(7121):875‐880. [DOI] [PubMed] [Google Scholar]
- 102. Kim IC, Han S. Epicardial adipose tissue: fuel for COVID‐19‐induced cardiac injury? Eur Heart J. 2020;41(24):2334‐2335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Malavazos AE, Goldberger JJ, Iacobellis G. Does epicardial fat contribute to COVID‐19 myocardial inflammation? Eur Heart J. 2020;41(24):2333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Griffin DO, Jensen A, Khan M, et al. Cytokine storm of a different flavor: the different cytokine signature of SARS‐CoV2 the cause of COVID‐19 from the original SARS outbreak. J Glob Antimicrob Resist. 2020;24:90–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105. Vizcaychipi MP, Shovlin CL, McCarthy A, et al. Increase in COVID‐19 inpatient survival following detection of thromboembolic and cytokine storm risk from the point of admission to hospital by a near real time traffic‐light system (TraCe‐Tic). Braz J Infect Dis. 2020;24(5):412‐421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Diao B, Wang C, Tan Y, et al. Reduction and functional exhaustion of T cells in patients with coronavirus disease 2019 (COVID‐19). Front Immunol. 2020;11:827–833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Mazurek T, Zhang L, Zalewski A, et al. Human epicardial adipose tissue is a source of inflammatory mediators. Circulation. 2003;108(20):2460‐2466. [DOI] [PubMed] [Google Scholar]
- 108. Shah RV, Murthy VL, Abbasi SA, et al. Visceral adiposity and the risk of metabolic syndrome across body mass index: the MESA study. JACC Cardiovasc Imaging. 2014;7(12):1221‐1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Park YW, Allison DB, Heymsfield SB, Gallagher D. Larger amounts of visceral adipose tissue in Asian Americans. Obes Res. 2001;9(7):381‐387. [DOI] [PubMed] [Google Scholar]
- 110. Hui H, Zhang Y, Yang X, et al. Clinical and radiographic features of cardiac injury in patients with 2019 novel coronavirus pneumonia. medRxiv. 2020. 2020.02.24.20027052 [Google Scholar]
- 111. Goeller M, Achenbach S, Marwan M, et al. Epicardial adipose tissue density and volume are related to subclinical atherosclerosis, inflammation and major adverse cardiac events in asymptomatic subjects. J Cardiovasc Comput Tomogr. 2018;12(1):67‐73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. Patel VB, Basu R, Oudit GY. ACE2/Ang 1‐7 axis: a critical regulator of epicardial adipose tissue inflammation and cardiac dysfunction in obesity. Adipocyte. 2016;5(3):306‐311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Touyz RM. Protecting the heart in obesity: role of ACE2 and its partners. Diabetes. 2016;65(1):19‐21. [DOI] [PubMed] [Google Scholar]
- 114. Kawabe Y, Mori J, Morimoto H, et al. ACE2 exerts anti‐obesity effect via stimulating brown adipose tissue and induction of browning in white adipose tissue. Am J Physiol Endocrinol Metab. 2019;317(6):E1140‐E1149. [DOI] [PubMed] [Google Scholar]
- 115. Sidossis LS, Porter C, Saraf MK, et al. Browning of subcutaneous white adipose tissue in humans after severe adrenergic stress. Cell Metab. 2015;22(2):219‐227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116. Chechi K, Voisine P, Mathieu P, et al. Functional characterization of the Ucp1‐associated oxidative phenotype of human epicardial adipose tissue. Sci Rep. 2017;7(1):15566–15580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117. Sacks HS, Fain JN, Bahouth SW, et al. Adult epicardial fat exhibits beige features. J Clin Endocrinol Metabol. 2013;98(9):E1448‐E1455. [DOI] [PubMed] [Google Scholar]
- 118. Eskilsson A, Shionoya K, Enerbäck S, Engblom D, Blomqvist A. The generation of immune‐induced fever and emotional stress‐induced hyperthermia in mice does not involve brown adipose tissue thermogenesis. FASEB j. 2020;34(4):5863‐5876. [DOI] [PubMed] [Google Scholar]
- 119. Stanley WC, Recchia FA, Lopaschuk GD. Myocardial substrate metabolism in the normal and failing heart. Physiol Rev. 2005;85(3):1093‐1129. [DOI] [PubMed] [Google Scholar]
- 120. Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497‐506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Haschke M, Schuster M, Poglitsch M, et al. Pharmacokinetics and pharmacodynamics of recombinant human angiotensin‐converting enzyme 2 in healthy human subjects. Clin Pharmacokinet. 2013;52(9):783‐792. [DOI] [PubMed] [Google Scholar]
- 122. Gross S, Jahn C, Cushman S, Bar C, Thum T. SARS‐CoV‐2 receptor ACE2‐dependent implications on the cardiovascular system: from basic science to clinical implications. J Mol Cell Cardiol. 2020;144:47‐53. [DOI] [PMC free article] [PubMed] [Google Scholar]


