Abstract
During the second surge of COVID‐19 in France (fall 2020), we assessed the expression of monocyte CD169 (i.e., Siglec‐1, one of the numerous IFN‐stimulated genes) upon admission to intensive care units of 45 patients with RT‐PCR‐confirmed SARS‐CoV2 pulmonary infection. Overall, CD169 expression was strongly induced on circulating monocytes of COVID‐19 patients compared with healthy donors and patients with bacterial sepsis. Beyond its contribution at the emergency department, CD169 testing may be also helpful for patients' triage at the ICU to rapidly reinforce suspicion of COVID‐19 etiology in patients with acute respiratory failure awaiting for PCR results for definitive diagnosis.
Keywords: CD169, COVID‐19, monocyte, sepsis
1. INTRODUCTION
The appearance of severe acute respiratory coronavirus‐2 (SARS‐CoV‐2) has led to a rapidly spreading pandemic. 1 Since the first cases of coronavirus disease‐19 (COVID‐19), more than 73 million cases and 1.6 million deaths have been reported worldwide (by December, 16—Johns Hopkins University). In areas particularly affected by virus spread, the tremendous number of patients coming to hospital threatened to overwhelm emergency departments (ED) and intensive care units (ICU) resources. Thus a crucial issue lied in the local capacity to rapidly test patients in order to confirm or rule out COVID‐19 diagnosis and consequently optimize patients' triage. Diagnosis of COVID‐19 was routinely achieved by detection of SARS‐CoV‐2 RNA in nasopharyngeal swabs via RT‐PCR.
In response to viral infection, host cells immediately produce cytokines as a first alarm bell. 2 , 3 Among these, type‐I interferons (IFN‐I) are crucial components of innate immune system against viruses. IFN‐I have, by themselves, antiviral properties but they also induce the expression of hundreds of IFN‐stimulated genes (ISG) turning cells into antiviral status that limits virus spread. 2 Among various effects, IFN‐I induce sharp rise in CD169 (also known as Siglec‐1) expression on monocytes (mCD169) whereas this molecule is not expressed (or at very low levels) under baseline conditions. 4 Such overexpression is consequently easily detectable by flow cytometry. Therefore, increased mCD169 has been proposed, by itself, as a potential host marker of viral infections (e.g., HIV, influenza). Moreover, in conjunction with concomitant assessment of neutrophil CD64 (nCD64) expression, mCD169 was used to differentiate viral from bacterial infections. 5 Recently, along with increased plasma IFN‐I concentration, mCD169 overexpression was observed in COVID‐19 patients. 6 Within a pandemic context, this highlighted the interest of mCD169 as a rapid marker for the triage of patients with suspected COVID‐19. As this first work in COVID‐19 reported on patients admitted to the ED, we investigated in prospective observational study whether CD169 overexpression would also be present in critically ill COVID‐19 patients upon ICU admission.
2. PATIENTS AND METHODS
2.1. Patients
Critically‐ill patients admitted to ICUs from university hospitals (Hospices Civils de Lyon, Lyon, France) who presented with pulmonary infection with SARS‐CoV2 confirmed by RT‐PCR testing were prospectively included in the study. This study was part of an ongoing prospective observational clinical study (RICO, REA‐IMMUNO‐COVID) 7 and was registered at ClinicalTrials.gov (NCT04392401). This study was approved by ethics committee (Comité de Protection des Personnes Ile de France 1—N°IRB/IORG #: IORG0009918) under agreement number 2020‐A01079‐30. The committee waived the need for written informed consent because the study was observational. Nevertheless, oral information and non‐opposition to inclusion in the study were mandatory and were systematically obtained before any blood sample was drawn and recorded in patients' clinical files. If a patient was unable to consent directly, non‐opposition was obtained from the patient's legally authorized representative and reconfirmed from the patient at the earliest opportunity. Inclusion criteria were: patients aged >18 years, diagnosis of COVID‐19 confirmed by RT‐PCR testing in one respiratory sample. Exclusion criteria were pregnancy, institutionalized patients, inability to obtain informed consent. As controls, patients with septic shock (bacterial infection) from the cohort IMMUNOSEPSIS 8 were included as well as healthy donors from Etablissement Francais du Sang (Lyon, F).
2.2. Flow cytometry
EDTA‐whole blood samples were processed by flow cytometry according to a newly described one‐step procedure (no wash, no centrifuge protocol). 9 For each blood sample tested, 500 μl of Versalyse lysing solution (Beckman Coulter, Hialeah, FL), 10 μl of blood and 10 μl IOTest Myeloid Activation antibody cocktail (Beckman Coulter), containing three markers: anti‐CD169‐PE (clone 7‐239), anti‐CD64‐PB (clone 22), anti‐HLA‐DR‐APC (clone Immu357) were incubated for 30 min at room temperature in the dark. Afterwards, samples were directly analyzed on a three‐laser, 10‐color Navios flow cytometer (BeckmanCoulter) according to a compensation‐free protocol. Monocytes were identified on a Side Scatter (SSC)/HLA‐DR dot plot whereas neutrophils and lymphocytes were gated based on SSC and forward scatter characteristics (FSC) characteristics (Figure 1). CD169 results were expressedas mean of fluorescence intensities (MFI) and CD169 ratio (i.e., mCD169 MFI/lymphocyte CD169 MFI). In addition, to fit with results from multi‐parametric paper in COVID‐19, 10 results were also expressed as % of positive cells (positivity threshold was based on lack of CD169 expression on neutrophils). Neutrophil CD64 results were expressed as MFI.
FIGURE 1.
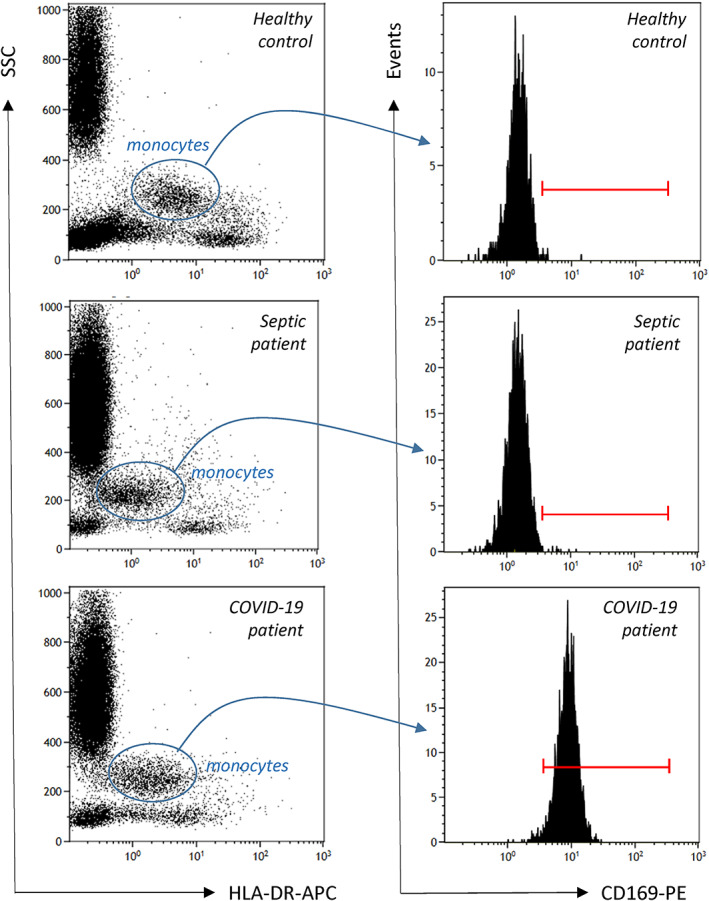
Gating strategy (illustrative examples). Monocytes are first gated out from other cells on the basis of on SSC (side‐scatter) / HLA‐DR histogram (blue circle, left histogram) and CD169 is then measured on their surface (monoparametric histogram, right panel) as mean of fluorescence related to the entire monocyte population or as % of positive cells (positivity threshold, red cursor, was based on lack of CD169 expression on neutrophils). In addition, CD169 MFI was also determined on total lymphocyte population gated on SSC / FSC (forward‐scatter) histogram (not shown) in order to provide results CD169 ratio (i.e., mCD169 MFI/lymphocyte CD169 MFI). One illustrative example of each group is depicted (healthy controls, septic shock, COVID‐19) [Color figure can be viewed at wileyonlinelibrary.com]
3. RESULTS
During the second surge of COVID‐19 pandemic in France, between October 8th and December 8th, 2020, we enrolled 45 critically ill patients with positive SARS‐CoV‐2 RT‐PCR. During the same period, we also included 2 ICU patients with septic shock and 12 healthy controls. Main demographic and characteristics are presented in Table 1. We observed a strong mCD169 overexpression in COVID‐19 ICU patients and clear differences with septic shock or healthy donors with results expressed as MFI, percentages or ratio (Figure 2). Original data for the measurements in the three patients groups are listed in Table S1. When expressed as percentages of CD169+ monocytes, all COVID‐19 patients except two expressed higher mCD169 than healthy donors and septic shock patients. Notably, in more than half of COVID‐19 patients, almost all circulating monocytes expressed CD169. Regarding results as ratio, when applying the threshold recently proposed by Bedin et al. (i.e., 3.5), 6 77% of COVID‐19 ICU patients presented with elevated results whereas all controls and septic patients were <3.5. Of note, although nCD64 values measured in COVID‐19 patients were not as high as observed in septic shock patients, they were nevertheless elevated above healthy controls (data not shown). This might be due to putative co‐infections as reported in COVID‐19 patients arriving at the ICU. 11
TABLE 1.
Main clinical and demographic characteristics of patients and healthy controls
| Healthy donors | COVID‐19 | Septic shock | |
|---|---|---|---|
| Number | 12 | 45 | 2 |
| Age (years) a | 40 (28.7–54.5) | 65 (60.7‐74.5) | 37, 79 |
| Sex (% male) | 25 | 78 | 50 |
| Invasive ventilation, n (%) | 0 | 22 (48.9) | 2 (100) |
Median (interquartile range) except septic shock.
FIGURE 2.
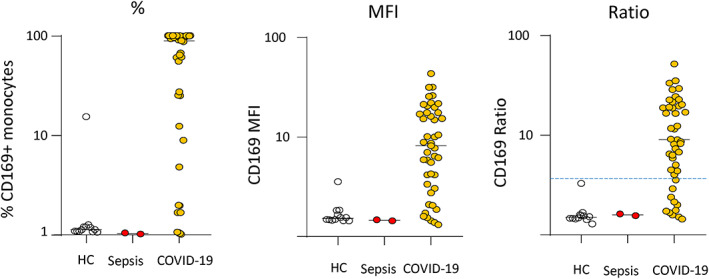
Monocyte CD169 expression in COVID‐19 ICU patients. Results are presented as percentages of positive cells (out of total monocyte population, left graph), median of fluorescence intensity in total monocyte population (MFI, middle graph), ratio monocyte CD169 MFI/lymphocyte CD169 MFI (right graph). The blue dashed line represents threshold proposed in Reference 6. Healthy controls (n = 8) are depicted by empty circles, septic shock patients (n = 2) are depicted by red circles and COVID‐19 ICU patients (n = 45) are depicted by orange circles [Color figure can be viewed at wileyonlinelibrary.com]
4. DISCUSSION
In areas particularly affected by the virus spread, the tremendous number of patients coming to hospital overwhelmed hospital resources. 12 As a consequence, the local capacity to rapidly test patients in order to confirm or rule out COVID‐19 diagnosis and consequently optimize patients' triage has appeared as a major factor in the fight against COVID‐19 pandemic. Due to the surge of patients, resources to address the diagnosis (routinely achieved by detection of SARS‐CoV‐2 RNA in nasopharyngeal swabs via RT‐PCR) in a timely manner have been overwhelmed. Rapid antigen detection tests have been developed to identify active infection but, in their first generation, they lack sensitivity. Consequently, measurement of host marker reflecting viral infection has appeared as a sound approach. Unfortunately, IFN‐I are produced at very low concentrations and most usual ELISA technologies are not able to quantify such low amount of cytokines (concentrations in fg/ml range). Only ultrasensitive digital Elisa (SIMOA) allows for the accurate IFN‐I measurements but this technique is usually not available on a routine basis. The detection of ISG mRNA levels by molecular approach, although possible within the non‐urgent context of auto‐immunity, is not suitable to COVID‐19 emergency due to extended time‐to‐results. 13 , 14 Alternatively, overexpression of mCD169 (siglec‐1, one of ISG) by whole blood flow cytometry has been recently proposed as a sensitive and specific surrogate marker of viral infection within the context of early COVID‐19. 6 This marker was previously described as a tool, in conjunction with nCD64 expression, to discriminate bacterial from viral infections at emergency room. 5 , 15 Specific kinetics of mCD169 expression in response to IFN‐I has been described in vitro. 4
The main result of the present study is to show that, upon ICU admission, critically ill COVID‐19 patients presented with strong mCD169 overexpression. Although altered IFN‐I production is known to undoubtedly concur to the most severe forms of COVID‐19, 16 , 17 , 18 mCD169 overexpression was still observed in this cohort. According to Bedin's threshold, almost 80% of patients were correctly identified as virally infected. Thus, in more severe patients, the present work reinforces the seminal description by Bedin et al. 6 It also extends the interest of mCD169 testing to larger cohorts of patients, especially those directly admitted to ICU for acute respiratory failure after sudden pulmonary worsening and without definitive diagnosis. Thus elevated mCD169 could help for patients triage in the ICU by reinforcing suspicion of COVID‐19 etiology while awaiting for PCR results. mCD169 protocol is a one‐step whole blood method that enables a streamlined flow cytometry sample preparation protocol. The simplified phenotyping procedure reduces training requirements and overcomes logistic constraints inherent to many flow cytometry applications (in particular due to the absence of compensation). Reagents costs are limited (<5 €/test) and results may be provided in less than 45 min. Consequently, mCD169 may contribute to preserving hospital medical capacities by accelerating patients' triage.
By nature, this preliminary work, conducted during the second wave of pandemic in France presents with limitations. First, a larger cohort of ICU COVID‐19 patients would be desirable to confirm these results. In addition, before drawing definitive conclusion, the inclusion of a cohort of mixed ICU patients with different etiologies (surgery, trauma, sepsis, burns) needs to be tested for mCD169 to precisely investigate specificity. Finally, the question of a potential interest of a daily mCD169 monitoring should be explored in further investigations. That given, these preliminary results in ICU patients aggregate with previously published data to underline the interest in mCD169 as a complementary tool to PCR results. As such, this could be of major help during the present pandemic. Of note, during the writing of this manuscript, another study, describing concordant mCD169 results at the ED has been released (Bourgoin et al., medRxiv 2020, doi: https://doi.org/10.1101/2020.10.28.20221259). Importantly, by using similar protocol (antibodies, lysis reagent), the authors shortened incubation/staining time at 15 min.
Collectively, these first results from critically ill COVID‐19 patients indicate that increased mCD169 expression, an IFN‐I inducible receptor, may contribute to COVID‐19 patients triage in the ICU while awaiting for definitive PCR results. The development of automated flow cytometric mCD169 measurement should be considered for further viral pandemic.
CONFLICT OF INTEREST
This study was partially supported by Beckman Coulter through donation of the CD169 reagents used in the study. This private company had no role in the study design, or collection and interpretation of the clinical data.
AUTHOR CONTRIBUTIONS
Marine Ortillon: Formal analysis; writing‐original draft. Remy COUDEREAU: Formal analysis; investigation; writing‐review and editing. Martin Cour: Investigation; supervision; validation; writing‐review and editing. Thomas Rimmelé: Investigation; supervision; validation; writing‐review and editing. Marine godignon: Supervision; validation; writing‐review and editing. Morgane Gossez: Validation; writing‐review and editing. hodane yonis: Investigation; supervision; writing‐review and editing. laurent argaud: Investigation; supervision; validation; writing‐review and editing. anne‐claire lukaszewicz: Investigation; supervision; validation; writing‐original draft. Fabienne Venet: Investigation; methodology; supervision; validation; writing‐original draft. Guillame Monneret: Conceptualization; data curation; funding acquisition; investigation; methodology; project administration; resources; supervision; validation; writing‐original draft.
DATA AVAILABILITY STATEMENT
The authors certify that this manuscript reports original clinical research data. Individual data that underlie the results reported in this article are available from the corresponding author following publication, including the study report and study protocol. Additional data are available upon reasonable request.
ETHICS AND PATIENT APPROVAL
This study was part of an ongoing prospective observational clinical study (RICO, REA‐IMMUNO‐COVID) and was registered at ClinicalTrials.gov (NCT04392401). This study was approved by ethics committee (Comité de Protection des Personnes Ile de France 1—N°IRB/IORG #: IORG0009918) under agreement number 2020‐A01079‐30. The committee waived the need for written informed consent because the study was observational. Nevertheless, oral information and non‐opposition to inclusion in the study were mandatory and were systematically obtained before any blood sample was drawn and recorded in patients' clinical files. If a patient was unable to consent directly, non‐opposition was obtained from the patient's legally authorized representative and reconfirmed from the patient at the earliest opportunity.
Supporting information
Table S1: Supporting information.
Data S1: MIFlowCyt item checklist.
Ortillon M, Coudereau R, Cour M, et al. Monocyte CD169 expression in COVID‐19 patients upon intensive care unit admission. Cytometry. 2021;99:466–471. 10.1002/cyto.a.24315
Funding information All authors are supported by Hospices Civils de Lyon and Claude Bernard Lyon‐1 University.
REFERENCES
- 1. Zhou F, Yu T, Du R, Fan G, Liu Y, Liu Z, et al. Clinical course and risk factors for mortality of adult inpatients with COVID‐19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Schneider WM, Chevillotte MD, Rice CM. Interferon‐stimulated genes: a complex web of host defenses. Annu Rev Immunol. 2014;32:513–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Sa Ribero M, Jouvenet N, Dreux M, Nisole S. Interplay between SARS‐CoV‐2 and the type I interferon response. PLoS Pathog. 2020;16:e1008737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Bourgoin P, Biechele G, Ait Belkacem I, Morange PE, Malergue F. Role of the interferons in CD64 and CD169 expressions in whole blood: relevance in the balance between viral‐ or bacterial‐oriented immune responses. Immun Inflamm Dis. 2020;8:106–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Bourgoin P, Soliveres T, Ahriz D, Arnoux I, Meisel C, Unterwalder N, et al. Clinical research assessment by flow cytometry of biomarkers for infectious stratification in an Emergency Department. Biomark Med. 2019;13:1373–86. [DOI] [PubMed] [Google Scholar]
- 6. Bedin AS, Makinson A, Picot MC, Mennechet F, Malergue F, Pisoni A, et al. Monocyte CD169 expression as a biomarker in the early diagnosis of COVID‐19. J Infect Dis. 2020. 10.1093/infdis/jiaa724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Monneret G, Cour M, Viel S, Venet F, Argaud L. Coronavirus disease 2019 as a particular sepsis: a 2‐week follow‐up of standard immunological parameters in critically ill patients. Intensive Care Med. 2020. ;46(9):1764–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Waeckel L, Venet F, Gossez M, Monard C, Rimmele T, Monneret G. Delayed persistence of elevated monocytic MDSC associates with deleterious outcomes in septic shock: a retrospective cohort study. Crit Care. 2020;24:132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Bourgoin P, Hayman J, Rimmele T, Venet F, Malergue F, Monneret G. A novel one‐step extracellular staining for flow cytometry: proof‐of‐concept on sepsis‐related biomarkers. J Immunol Methods. 2019;470:59–63. [DOI] [PubMed] [Google Scholar]
- 10. Silvin A, Chapuis N, Dunsmore G, Goubet AG, Dubuisson A, Derosa L, et al. Elevated calprotectin and abnormal myeloid cell subsets discriminate severe from mild COVID‐19. Cell. 2020;182:1401–18.e18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Kreitmann L, Monard C, Dauwalder O, Simon M, Argaud L. Early bacterial co‐infection in ARDS related to COVID‐19. Intensive Care Med. 2020;46:1787–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Rimmele T, Pascal L, Polazzi S, Duclos A. Organizational aspects of care associated with mortality in critically ill COVID‐19 patients. Intensive Care Med. 2021. ;47(1):119–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Crow YJ, Manel N. Aicardi‐Goutieres syndrome and the type I interferonopathies. Nat Rev Immunol. 2015;15:429–40. [DOI] [PubMed] [Google Scholar]
- 14. Pescarmona R, Belot A, Villard M, Besson L, Lopez J, Mosnier I, et al. Comparison of RT‐qPCR and Nanostring in the measurement of blood interferon response for the diagnosis of type I interferonopathies. Cytokine. 2019;113:446–52. [DOI] [PubMed] [Google Scholar]
- 15. Bourgoin P, Lediagon G, Arnoux I, Bernot D, Morange PE, Michelet P, et al. Flow cytometry evaluation of infection‐related biomarkers in febrile subjects in the emergency department. Future Microbiol. 2020;15:189–201. [DOI] [PubMed] [Google Scholar]
- 16. Hadjadj J, Yatim N, Barnabei L, Corneau A, Boussier J, Smith N, et al. Impaired type I interferon activity and inflammatory responses in severe COVID‐19 patients. Science. 2020;369:718–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Bastard P, Rosen LB, Zhang Q, Michailidis E, Hoffmann HH, Zhang Y, et al. Autoantibodies against type I IFNs in patients with life‐threatening COVID‐19. Science. 2020;370:eabd4585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Acharya D, Liu G, Gack MU. Dysregulation of type I interferon responses in COVID‐19. Nat Rev Immunol. 2020;20:397–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Table S1: Supporting information.
Data S1: MIFlowCyt item checklist.
Data Availability Statement
The authors certify that this manuscript reports original clinical research data. Individual data that underlie the results reported in this article are available from the corresponding author following publication, including the study report and study protocol. Additional data are available upon reasonable request.


