Abstract
Objectives
Indication for invasive mechanical ventilation in COVID‐19 pneumonia has been a major challenge. This study aimed to evaluate if lung ultrasound (LUS) can assist identification of requirement of invasive mechanical ventilation in moderate to severe COVID‐19 pneumonia.
Materials and Methods
Between April 23 and November 12, 2020, hospitalized patients with moderate to severe COVID‐19 (oxygen demand ≥4 L/min) were included consecutively. Lung ultrasound was performed daily until invasive mechanical ventilation (IMV‐group) or spontaneous recovery (non‐IMV‐group). Clinical parameters and lung ultrasound findings were compared between groups, at intubation (IMV‐group) and highest oxygen demand (non‐IMV‐group). A reference group with oxygen demand <4 L/min was examined at hospital admission.
Results
Altogether 72 patients were included: 50 study patients (IMV‐group, n = 23; non‐IMV‐group, n = 27) and 22 reference patients. LUS‐score correlated to oxygen demand (SpO2/FiO2‐ratio) (r = 0.728; p < .0001) and was higher in the IMV‐group compared to the non‐IMV‐group (20.0 versus 18.0; p = .026). Based on receiver operating characteristic analysis, a LUS‐score of 19.5 was identified as cut‐off for requirement of invasive mechanical ventilation (area under the curve 0.68; sensitivity 56%, specificity 74%). In 6 patients, LUS identified critical coexisting conditions. Respiratory rate and oxygenation index ((SpO2/FiO2)/respiratory rate) ≥4.88 identified no requirement of invasive mechanical ventilation with a positive predictive value of 87% and negative predictive value of 100%.
Conclusions
LUS‐score had only a moderate diagnostic value for requirement of invasive mechanical ventilation in moderate to severe COVID‐19. However, LUS proved valuable as complement to respiratory parameters in guidance of disease severity and identifying critical coexisting conditions.
Keywords: Coronavirus, COVID‐19, ultrasonography, ventilators, mechanical
Abbreviations
- ARDS
acute respiratory distress syndrome
- COVID‐19
coronavirus disease 2019
- FiO2
inspired fraction of oxygen
- IMV
invasive mechanical ventilation
- LUS
lung ultrasound
- PaO2
arterial partial pressure of oxygen
- ROX index
respiratory rate and oxygenation index
- SpO2
peripheral oxygen saturation
Introduction
Coronavirus disease 2019 (COVID‐19), caused by a previously unknown human pathogen (severe acute respiratory syndrome coronavirus 2; SARS‐Cov‐2), was declared a global pandemic on March 11, 2020. 1 Most patients express mild symptoms, but COVID‐19 can develop into a severe condition with pneumonia, acute respiratory distress syndrome (ARDS) and multi‐organ failure. 2 COVID‐19 has challenged emergency medicine and intensive care worldwide by high numbers of critically ill patients together with deficient evidence for management and treatment of a novel condition. The clinical features of respiratory failure in COVID‐19 have confounded clinicians – severe hypoxia despite lack of respiratory distress (silent hypoxia) and unpredictable clinical deterioration. 3 Identification of requirement of and timing for initiation of invasive mechanical ventilation (IMV) has been a major challenge. 3 , 4 Lung ultrasound (LUS) is an established method in diagnostics of acute respiratory failure. 5 , 6 In COVID‐19 pneumonia, interstitial lung pathologies have been described on LUS. 7 , 8 , 9 , 10 In ARDS, a LUS‐score has previously been developed to classify the severity of respiratory failure by a 36‐point scale based on LUS findings of B‐line artifacts (B‐lines) and consolidations. 11 During the COVID‐19 pandemic, studies exploring the role of LUS in the management of COVID‐19 pneumonia and respiratory failure have been requested. 12 , 13 , 14 So far, only a few studies have evaluated LUS‐score in hospitalized patients with COVID‐19. 15 , 16 , 17 LUS has been found to be a valuable tool in assessment of disease severity and presumably also in guiding patient management, including decisions on mechanical ventilation. 15 , 16 , 17 By being a bedside method, LUS has obvious advantages compared to other radiological entities saving hospital resources and reducing virus transmission. 18 With the aim of determining the significance of LUS examination in identifying patients with requirement of IMV, we assessed a consecutive cohort of patients with moderate to severe (WHO 4–6) respiratory failure due to COVID‐19 infection. 19 The primary objective was to evaluate the association between LUS‐score and indication for IMV treatment. In addition, we assessed the association between LUS‐score and peripheral oxygen saturation/inspired fraction of oxygen (SpO2/FiO2)‐ratio as well as the distribution pattern of LUS findings in moderate to severe COVID‐19 pneumonia.
Materials and Methods
Study design and setting
Between April 23 and November 12, 2020 a prospective single‐center study was conducted at Falun Hospital, Sweden. During the study period, Falun Hospital supplied care for all hospitalized patients with COVID‐19 in the county of Dalarna, with a population of about 270,000. A majority of patients with COVID‐19 were admitted to the hospital due to respiratory failure. In isolation rooms at the care units, oxygen therapy was provided by nasal cannula, face mask or high flow nasal cannula. Patients in need of IMV were admitted to the intensive care unit. Indication for IMV treatment was respiratory failure with impending exhaustion, high oxygen demand with deranged clinical parameters such as respiratory rate, work of breathing and impaired consciousness. Due to aerosol dispersion and risk of virus transmission in open areas at the intensive care unit, high flow nasal cannula and noninvasive ventilation were not utilized for patients with COVID‐19 at the intensive care unit.
All admitted patients (age ≥ 18 years) with the following criteria were eligible for the study: (1) positive for SARS‐CoV‐2 by reverse transcription polymerase chain reaction on nasopharyngeal swab and (2) moderate to severe respiratory failure corresponding to oxygen requirement ≥4 liters/minute (L/min) or high flow nasal cannula with a FiO2 of ≥30% (Airvo 2, Fisher & Paykel) (WHO 4–6). 19 , 20 Exclusion criteria were documented treatment limitation of “not eligible for IMV “and inability to participate in LUS examination or daily follow‐ups. Also patients with the following conditions were excluded to avoid false‐positive findings: interstitial lung diseases (for example pulmonary fibrosis), congestive heart failure, kidney failure on dialysis or age > 80 years. 5 , 21 Patients with pneumothorax or history of pneumonectomy or pleurodesis were excluded due to interference with LUS examination. Between May 21 and June 13 a group of reference patients admitted to the hospital with no or mild respiratory failure (oxygen requirement of <4 L/min, WHO 3–4) were included based on the same criteria. 19 All patients approved participation by written informed consent. Ethical approval was received from the Swedish Ethical Review Authority (Dnr 2020–02184).
Data collection
Patients eligible for the study were identified through daily contact with all care units. Included patients with oxygen requirement ≥4 L/min were examined daily with LUS until either initiation of IMV treatment or spontaneous improvement to oxygen demand of ≤1–2 L/min. For reference patients, LUS was performed at one occasion within 48 hours after admission to hospital. Clinical respiratory status was noted along with the LUS examination. For patients receiving IMV, data right before initiation of IMV were used for analysis. If LUS data were missing from the time right before intubation, data within 24 hours prior to, or after initiation of IMV were used. For patients who did not receive IMV treatment, data at the day of highest oxygen demand (based on lowest SpO2/FiO2‐ratio) were used for further analysis. To define severity of ARDS (Berlin criteria) for patients on IMV, the lowest arterial partial pressure of oxygen/FiO2‐ratio (PaO2/FiO2‐ratio) within 24 hours after intubation was used. 22 Peripheral oxygen saturation was measured by a pulse oximeter (Nellcor Oximax N‐65; Covidien, Boulder, CO, USA). Respiratory rate, oxygen requirement (L/min) and Glasgow Coma Scale were noted and patients were asked for dyspnea at rest (“yes/no”). Medical history and clinical parameters together with relevant laboratory results were collected from patient charts. A conversion table for estimation of FiO2 provided by oxygen delivery devices was used. 23 , 24 An estimate of PaO2/FiO2‐ratio was obtained from SpO2/FiO2. 20 Severity of respiratory failure was defined by cut‐off values used for ARDS; a SpO2/FiO2‐ratio of 315, 235 and 148 reflecting mild, moderate and severe ARDS, respectively. 20 , 22 In order to evaluate clinical criteria for intubation, we used the respiration rate and oxygenation index (ROX index) based on Roca et al. ((SpO2/FiO2)/respiratory rate). 25 A ROX index of ≥4.88 has been suggested as cut‐off for low risk of requirement of IMV. 25
Lung ultrasound
An ultrasound machine aimed for point‐of‐care examinations with a lung preset adjusted to a depth of 12 cm was used (BK Medical flex focus 500 with a curved probe [BK medical type 8823] of 2‐6 MHz, BK Medical AB, Herley, Denmark). Six lung regions on the left and right hemithorax, respectively, were scanned: upper and basal anterior/lateral/posterior regions. Anterior, lateral and posterior regions were separated by the anterior and posterior axillary lines. 11 For each region, findings of B‐line artifacts (B‐lines) and consolidations were noted. Each region was assessed and added up to a LUS‐score (0–36) based on previously described protocol in ARDS 11 , 26 : score 0 ‐ normal findings with A‐lines or ≤ two B‐lines; score 1 – 3 or more well‐separated B‐lines covering ≤50% of the region; score 2 ‐ coalescent B‐lines covering >50% of the region; score 3 ‐ large consolidation with air bronchogram. Our data were also used to evaluate an alternative LUS‐score developed for COVID‐19 pneumonia by Manivel et al. 12 Therefore, the presence of small (<1 cm) or large (>1 cm) subpleural consolidations was noted for each region. Scanning was performed by carefully sliding sagittal and/or transversal over each region, and the highest score found was considered representative for the region. LUS was performed by 5 consultant anaesthesiologists experienced in LUS examination technique, not blinded for the patients' clinical status. Consensus on interpretation and scoring of LUS‐findings were sought among 2 physicians present at each LUS examination.
Statistical analysis
Demographics, clinical parameters and LUS findings were presented as absolute numbers and percentages for categorical variables and as medians with interquartile range (IQR) for continuous variables. The Chi‐square test or Fisher's exact test was used for comparisons of categorical outcomes and Mann–Whitney U test for unrelated continuous outcomes. The Friedman test compared ordinal data between 3 related samples and Wilcoxon signed rank test was used for post hoc analysis. Pearson correlation was used to estimate association between continuous variables. Receiver‐operating characteristic curve analysis was used to evaluate LUS‐score and ROX index for identification of requirement of IMV treatment. The optimal cut‐off values were identified using the Youden index. P‐value <.05 was considered statistically significant. A sample size of 50 patients was estimated prior to initiation of the present study, based on previous LUS‐data for mild and moderate ARDS (significance level of 0.05, 80% power). 27 IBM SPSS Statistics for Windows, Version 26.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism version 8.4.3 (GraphPad Software, La Jolla, CA, USA) were used for statistical analysis and graphic presentation.
Results
Study population
During the study period, 50 patients with COVID‐19 with an oxygen demand of ≥4 L/min were included. Of these, 23 received IMV treatment (IMV‐group) and 27 did not (non‐IMV‐group) (Figure 1). In addition, a reference group of 22 patients with an oxygen requirement of <4 L/min was recruited. Four of these later developed an oxygen requirement of ≥4 L/min and were included as study patients. Medical history and clinical parameters at admission to hospital are presented in Table 1. The median number of days from onset of symptoms to hospital admission was 9‐10 days for all groups.
Figure 1.
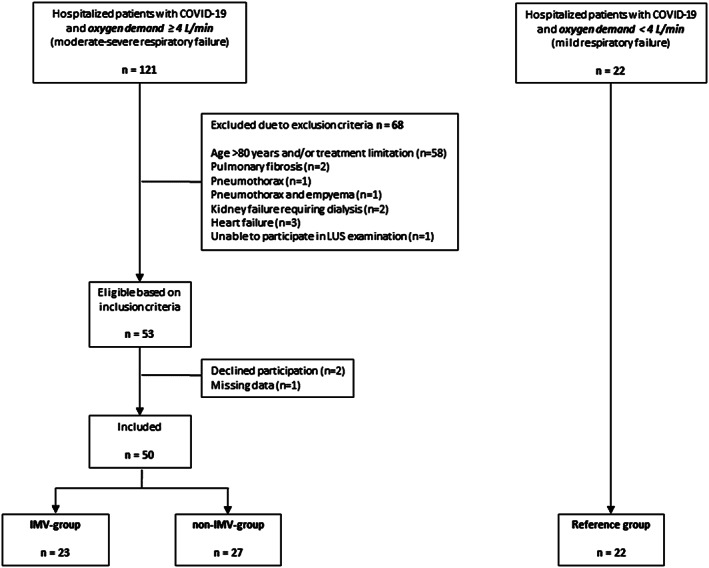
Study flowchart. Patients positive for SARS‐CoV‐2 by reverse transcription polymerase chain reaction and oxygen demand ≥4 L/min were eligible for the study. Patients were followed daily until either initiation of IMV (IMV‐group) or spontaneous improvement without requirement of IMV (non‐IMV‐group). Patients in the reference group were included based on the same inclusion or exclusion criteria but with oxygen demand <4 L/min. IMV = invasive mechanical ventilation.
Table 1.
Background characteristics. Demographics, medical history and laboratory/clinical parameters at hospital admission for IMV‐, non‐IMV‐groups and reference group
| IMV | non‐IMV | reference oxygen demand <4 L/min | ||
|---|---|---|---|---|
| oxygen demand ≥4 L/min | ||||
| n = 23 | n = 27 | p‐value | n = 22 | |
| Age (years) | 65 (17) | 67 (19) | .441 | 61 (22) |
| Sex Male; n (%) | 17 (74) | 17 (63) | .408 | 9 (41) |
| Female; n (%) | 6 (26) | 10 (37) | 13 (59) | |
| Body mass index (kg/m2) | 28 (6) | 28 (5) | .674 | 28 (3) |
| Medical history | ||||
| Hypertension; n (%) | 10 (43) | 13 (48) | .741 | 6 (27) |
| Ischemic heart disease; n (%) | 1 (4) | 3 (11) | .614 | 1 (5) |
| Atrial fibrillation; n (%) | 3 (13) | 3 (11) | 1.000 | 1 (5) |
| Stroke; n (%) | 0 | 0 | NA | 1 (5) |
| COPD; n (%) | 0 | 1 (4) | NA | 1 (5) |
| Astma; n (%) | 3 (1) | 7 (26) | .308 | 4 (18) |
| Diabetes; n (%) | 2 (9) | 8 (30) | .085 | 2 (9) |
| Kidney failure; n (%) | 2 (9) | 0 | NA | 1 (5) |
| Liver disease; n (%) | 0 | 0 | NA | 0 |
| Cancer; n (%) | 1 (4) | 1 (4) | NA | 2 (9) |
| Immunosuppression; n (%) | 0 | 2 (13) | NA | 1 (5) |
| Number of days with symptoms before hospital admission | 9 (7) | 10 (2) | .391 | 9 (6) |
| Laboratory parameters | ||||
| C‐reactive protein (mg/l) | 93 (128) | 109 (93) a | .668 | 46 (75) a |
| Procalcitonin (μg/l) | 0.25 (0.95) b , d | 0.10 (0.20) b , e | .049 | 0.10 (0.02) b , g |
| Leukocyte count (x 103/μl) | 5.8 (5.8) | 7.2 (3.4) | .606 | 5.9 (4.4) |
| Lymphocyte count (x 103/μl) | 0.7 (0.5) e | 0.9 (0.6) f | .472 | 1.0 (0.8) f |
| Trombocyte count (x 103/μl) | 207 (118) | 245 (140) d | .078 | 202 (76) d |
| Troponin I (ng/l) | 23 (27) c , f | 10 (0) c , e | .001 | 10 (0) c , h |
| Kreatinin (μmol/l) | 79 (32) | 70 (23) | .026 | 67 (34) |
| Clinical parameters | ||||
| Mean arterial pressure (mmHg) | 92 (16) | 99 (19) | .072 | 103 (26) |
| Respiratory rate (breath/min) | 24 (17) | 22 (10) | .273 | 21 (12) |
| SpO2/FiO2‐ratio | 339 (340) | 343 (135) | .565 | 446 (141) |
| Body temperature (Celsius) | 37.7 (1.7) | 38.4 (1.2) | .158 | 38.1 (1.0) |
If not stated otherwise, all values are presented as median (interquartile range)
p‐value is presented for comparison of IMV‐ vs non‐IMV‐patients using Mann–Whitney U test for continuous data and Chi‐square/Fisher's exact test for categorical data. NA = not applicable; IMV = invasive mechanical ventilation; COPD = chronic obstructive pulmonary disease; mmHg = millimeter mercury; SpO2 = peripheral oxygen saturation; FiO2 = inspired fraction of oxygen.
1 non‐IMV and 2 reference patients had CRP <4. Calculated as 4.
1 IMV, 3 non‐IMV and 4 reference patients had PCT <0.05. Calculated as 0.05.
6 IMV, 20 non‐IMV and 14 reference patients had Troponin I < 10. Calculated as 10. missing values:
) n = 1;
) n = 2;
) n = 3;
) n = 5;
) n = 6.
Clinical parameters associated with IMV
Time from hospital admission to intubation (IMV‐group) or highest oxygen demand (non‐IMV‐group) showed large individual differences within both groups (0‐10 days) (Table 2). On the day of intubation (IMV‐group) or of highest oxygen demand (non‐IMV‐group), all patients were hemodynamically stable and all but 4 patients were fully oriented. Before intubation, patients in the IMV‐group had lower SpO2/FiO2‐ratio and ROX index, higher respiratory rate and reported dyspnea more often compared to the non‐IMV‐group (Table 2, Figure 2). Based on previously presented cut‐off for ROX index indicating lower risk for requirement of IMV (≥4.88), 4 of 23 (17%) patients in the IMV‐group and all patients in the non‐IMV‐group fulfilled criteria for low risk of IMV treatment (positive predictive value 87%, negative predictive value 100%) (Figure 3C). Median PaO2/FiO2‐ratio in patients on IMV was 140 (IQR 68) after intubation. According to Berlin definition, 20 of these patients (87%), were classified as moderate ARDS and 3 patients (13%) as mild ARDS. 22
Table 2.
Clinical parameters and lung ultrasound findings. For the IMV‐group, clinical data were collected prior to intubation and LUS examination performed within 24 hours prior to or after intubation. For the non‐IMV group, all data were collected at the day of highest oxygen demand. For reference patients all data were collected within 48 hours of hospital admission
| IMV | non‐IMV | reference oxygen demand <4 L/min | ||
|---|---|---|---|---|
| oxygen demand ≥4 L/min | ||||
| N = 23 | N = 27 | p‐value | N = 22 | |
| Days from admission to IMV/maximum O2‐demand; median (min‐max) | 3 (0–10) | 2 (0–10) | .945 | ‐ |
| Oxygen device | ||||
| Nasal cannula / face mask; n (%) | 11 (48) | 17 (63) | .283 | 9 (41) |
| HFNC; n (%) | 12 (52) | 10 (37) | 0 | |
| Clinical and laboratory parameters | ||||
| Glasgow Coma Scale = 15; n (%) | 20 (87) | 26 (96) | .322 | 22 (100) |
| Respiratory rate (breaths/minute) | 32 (7) | 24 (6) | <.0001 | 20 (9) |
| SpO2/FiO2‐ratio | 106 (51) | 219 (60) | <.0001 | 452 (98) |
| ROX Index ((SpO2/FiO2)/RR) | 3.74 (2.08) | 8.94 (3.76) | <.0001 | 19 (11) |
| Patients with dyspnea; n (%) | 16 (73) a | 9 (33) | .006 | 1 (5) |
| Mean arterial pressure (mmHg) | 88 (28) | 87 (16) | .755 | 91 (10) |
| Body temperature (Celsius) | 38.6 (1.7) | 37.4 (1.2) | .026 | 38.5 (1.4) |
| C‐reactive protein (mg/l) | 146 (80) | 111 (94) a | .113 | 62 (70) b |
| Lung ultrasound | ||||
| LUS‐score (0–36) | 20.0 (5.0) | 18.0 (7.0) | .026 | 8.5 (10.0) |
| LUSS according to Manivel et al. | 23.0 (8.0) | 21.0 (12.0) | .545 | 10.0 (9.3) |
| Number of patients with pathological findings | ||||
| B‐lines (≥3/region); n (%) | 23 (100) | 27 (100) | NA | 19 (86) |
| Subpleural consolidations; n (%) | 21 (91) | 26 (96) | .588 | 16 (73) |
| Large consolidations (score 3); n (%) | 12 (52) | 15 (55) | .811 | 3 (14) |
| Pleural effusion; n (%) | 1 (4) | 3 (11) | NA | 0 (0) |
| Pathological findings in all regions (12/12); n (%) | 10 (43) | 5 (19) | .055 | 1 (5) |
| Number of regions for each patient (median; interquartile range) | ||||
| Separated B‐lines (score 1) | 3.0 (3.0) | 4.0 (2.0) | .435 | 3.0 (2.5) |
| Coalescent B‐lines (score 2) | 6.0 (5.0) | 4.0 (3.0) | .049 | 1.5 (3.3) |
| Large consolidations (score 3) | 1.0 (3.0) | 1.0 (3.0) | .951 | 0.0 (0.0) |
| Subpleural consolidations <1 cm | 2.0 (4.0) | 3.0 (3.0) | .090 | 1.0 (2.0) |
| Subpleural consolidations >1 cm | 1.0 (1.0) | 1.0 (2.0) | .888 | 0.0 (0.0) |
| Pathological findings (score 1/2/3) | 11.0 (1.0) | 11.0 (3.0) | .008 | 5.5 (5.5) |
If not stated otherwise, all values are presented as median (interquartile range).
p‐value is presented for comparison of IMV‐ vs non‐IMV‐patients using Mann–Whitney U test for continuous data and Chi‐square/Fishers exact test for categorical data. NA = not applicable; IMV = invasive mechanical ventilation; HFNC = high flow nasal cannula; SpO2 = peripheral oxygen saturation; FiO2 = inspired fraction of oxygen; ROX index = Respiratory rate and Oxygenation Index; RR = respiratory rate; mmHg = millimeter mercury; LUS = lung ultrasound; LUSS = Lung ultrasound scoring system according to Manivel et al. (0–36). 12 Missing values:
) n = 1;
) n = 2.
Figure 2.
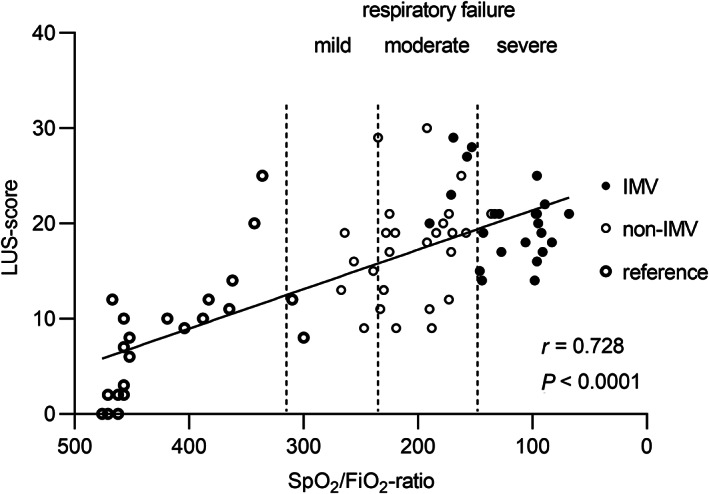
Correlation between LUS‐score and oxygen demand defined as SpO2/FiO2‐ratio. Cut‐off values (dotted lines) for mild, moderate and severe respiratory failure are defined as SpO2/FiO2‐ratio 315, 235 and 148, respectively. Data presented prior to intubation (IMV‐group) and on the day with highest oxygen demand (non‐IMV‐group). For the reference group data were collected within 48 hours after hospital admission. Correlation coefficient and p‐value given for Pearson correlation. See Figure 1 legend for definition of groups. LUS = lung ultrasound; IMV = invasive mechanical ventilation; SpO2 = peripheral oxygen saturation; FiO2 = inspired fraction of oxygen.
Figure 3.
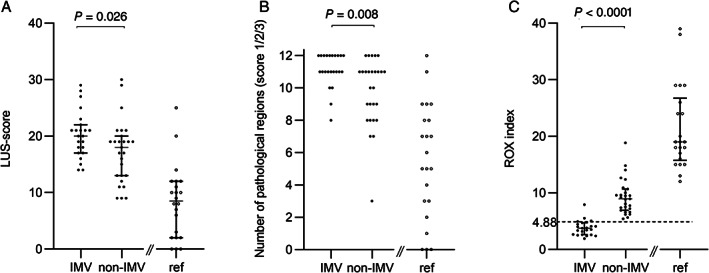
LUS findings and ROX index presented for IMV‐, non‐IMV‐ and reference groups. See Figure 1 legend for definition of groups. (A) LUS‐score (0–36) for each patient based on 12 lung regions examined. (B) Total number of regions with pathological findings (score 1/2/3) for each patient. (C) ROX index ((SpO2/FiO2)/respiratory rate) based on Roca et al. 25 The dotted line marks suggested cut‐off for low risk for intubation (≥4.88). Comparisons between IMV‐ and non‐IMV‐group using the Mann–Whitney U test. LUS = Lung ultrasound; ROX index = Respiratory rate and oxygenation index; IMV = invasive mechanical ventilation; SpO2 = peripheral oxygen saturation; FiO2 = inspired fraction of oxygen.
Lung ultrasound findings
LUS data for the IMV‐group was obtained as close as possible in relation to intubation. Twelve patients (52%) were examined right before intubation and 8 (35%) and 3 (13%) patients were scanned within 24 hours prior to, or after intubation respectively. The 3 patients scanned after intubation presented with a LUS‐score of 17, 20 and 25, respectively.
All 72 patients with COVID‐19 infection, except 3 patients in the reference group, had pathological LUS findings including thickening or irregularity of the pleural line, different distributions of B‐lines, subpleural consolidations and/or large consolidations with air bronchogram (Table 2). Based on all examined patients, LUS‐score correlated to SpO2/FiO2‐ratio (r = 0.728, p < .0001) (Figure 2). However, this correlation did not remain when analyzing only patients with oxygen demand ≥4 L/min (r = 0.266, p = .062). Patients in the IMV‐group had an overall higher LUS‐score compared to the non‐IMV‐group (20.0 versus 18.0; p = .026) (Figure 3A). This difference was more pronounced when also including reference patients in the non‐IMV‐group (20.0 versus 12.0; p < .0001). Using the scoring system proposed for COVID‐19 by Manivel et al., no difference was found between patients in the IMV‐ and non‐IMV‐group (Table 2). 12 The total number of regions with pathological LUS‐findings (Score 1, 2 or 3) for each patient showed significant difference between the IMV‐ and non‐IMV group even though the median numbers were the same in both groups (11 regions of 12 examined, p = .008) (Table 2; Figure 3B). Ten patients (43%) presented with pathological findings (LUS‐score 1, 2 or 3) in every region in the IMV‐group, compared to 5 patients (19%) in the non‐IMV group (p = .055) (Figure 3B, Table 2).
Distribution of B‐lines and different categories of consolidations over anterior, lateral and posterior lung fields are presented in Figure 4. The number of large consolidations increased gradually from anterior to posterior fields in both IMV‐ and non‐IMV‐groups (Figure 4). For B‐lines and subpleural consolidations, there was no difference between anterior, lateral and posterior lung fields (Figure 4). Photos of representative LUS findings are presented in Figure 5.
Figure 4.
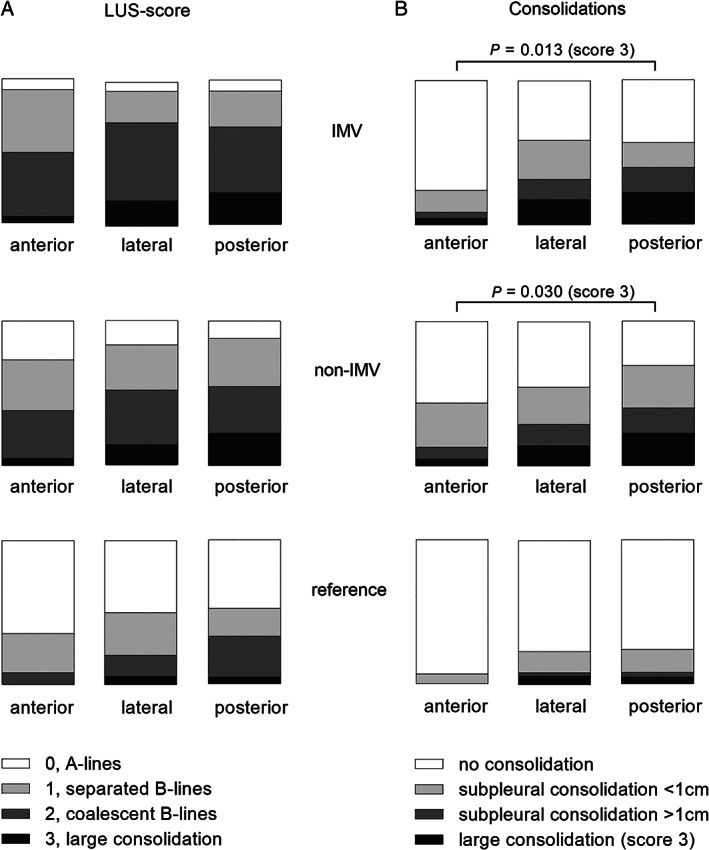
Distribution of LUS‐findings over anterior, lateral and posterior lung fields. Each field presents the sum of 4 regions examined by LUS (2 right and 2 left regions ‐ upper and basal) (A) Different qualities of the LUS‐score and (B) categories of consolidations are presented as percentage for 3 examined fields for each patient (anterior, lateral, posterior). Data are presented separately for each study group: IMV‐, non‐IMV‐ and the reference group; see Figure 1 legend for definition of groups. P‐values given for comparison between fields of large consolidations (score 3) using Friedman's test. Post hoc test for comparison of score 3 between anterior and posterior field using Wilcoxon signed rank test (IMV p = .013; non‐IMV p = .015). LUS = Lung ultrasound; IMV = invasive mechanical ventilation.
Figure 5.
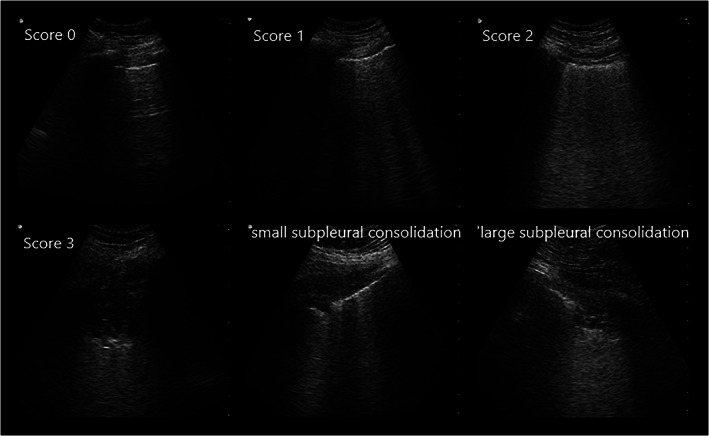
Photos illustrating the different qualities of LUS findings accounting for the LUS‐score and subpleural consolidations found in patients with COVID‐19. Score 0: normal findings with A‐lines or ≤2 B‐lines; Score 1: 3 or more well separated B‐lines; Score 2: coalescent B‐lines; Score 3: large consolidation with air bronchogram; Small subpleural consolidation <1 cm; Large subpleural consolidation >1 cm. LUS = Lung ultrasound.
LUS identified important differential diagnoses in 6 patients. One IMV patient with high procalcitonin and one non‐IMV patient showed compact basal consolidations and pleural effusions on LUS. They were diagnosed with bacterial pneumonia in addition to COVID‐19 infection. Two IMV‐patients with low LUS‐score (17 and 15, respectively) despite low PaO2/FiO2‐ratio (116 and 129, respectively) raised the concern of pulmonary embolus, which was later confirmed by computed tomography. One non‐IMV patient had a history of dyspnea for several months. In this patient, an elevated hemidiaphragm was seen on LUS, which was confirmed by computed tomography. In one patient, excluded due to pneumothorax, LUS revealed a large pleural effusion later diagnosed as empyema.
Identification of requirement of IMV
The diagnostic value of LUS‐score and ROX index for requirement of IMV were evaluated with receiver operating characteristic curve analysis. Based on patients with oxygen requirement of ≥4 L/min, an optimal cut‐off value of 19.5 for LUS‐score was identified with a sensitivity of 56% and specificity of 74% for IMV treatment (area under the curve 0.68, 95% CI 0.54–0.83; p = .026) (Figure 6A). For the same group of patients, a cut‐off value for ROX index of 5.57 identified patients with requirement for IMV with a sensitivity of 96% and specificity of 96% based on receiver operating characteristic curve analysis (area under the curve, 0.98, 95% CI, 0.94–1.00; p < .0001) (Figure 6B). Receiver operating characteristic curve analysis was also performed including the reference group, that is, patients with an oxygen demand of <4 L/min for comparison (Figures 6A,B).
Figure 6.
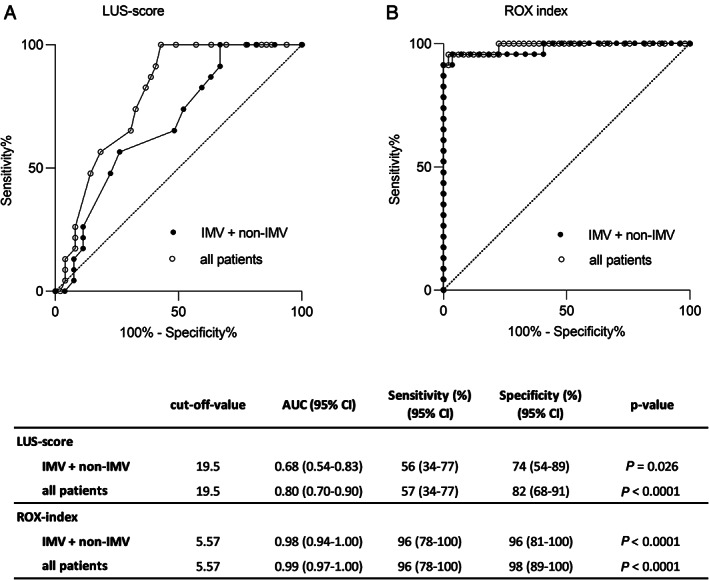
Reciever operating characteristic (ROC) curve analysis evaluating the diagnostic value of LUS‐score and ROX index for prediction of IMV treatment. ROC curves presented for IMV‐ and non‐IMV‐groups and for all patients (IMV, non‐IMV and reference group). See Figure 1 legend for definition of groups. (A) LUS‐score and (B) ROX index as diagnostic tests for determination of requirement of IMV treatment. The table presents suggested cut‐off values (based on Youden index) together with area under the curve (95% CI) and sensitivity and specificity. LUS = Lung ultrasound; ROX index = Respiratory rate and oxygenation index; IMV = invasive mechanical ventilation.
Discussion
In line with previous findings in ARDS and recent reports in COVID‐19, LUS‐score correlated to overall severity of respiratory failure. 15 , 16 , 17 , 27 , 28 However, LUS‐score had a limited diagnostic value for identification of patients with requirement of IMV treatment in a group of patients with moderate to severe COVID‐19 infection (WHO scale 4–6). LUS proved an important bedside instrument for identification of critical differential diagnoses.
In COVID‐19 pneumonia, there are a few studies describing the association between LUS‐findings and severity of disease. 15 , 16 , 17 , 29 , 30 In 3 recent reports, a much higher difference in LUS‐score was presented between patients requiring IMV treatment and patients without IMV compared to our data. 15 , 16 , 17 LUS‐score was also suggested a useful predictor for IMV. 15 , 16 , 17 In all 3 studies, LUS examinations were performed at hospital admission for patients with a wide range of disease severity, including milder conditions. 15 , 16 , 17 When also including reference patients with less severe respiratory failure in our analysis, the diagnostic value of LUS was consistent with previous reports. 15 , 16 , 17 In evaluation of requirement of IMV, selecting patients with an impending risk for mechanical ventilation was relevant for our purpose. Therefore, only patients with a high oxygen demand eligible for IMV treatment were included in our primary study group. Patients with a mild condition were examined foremost to ensure that LUS, in our hands, correlated to disease severity as previously shown in ARDS. 27 , 28 Timing of LUS examination in this study was based on the rationale that clinical assessment for requirement of IMV is routinely performed when patients deteriorate with increasing oxygen demand. It is important to consider that LUS findings in COVID‐19 correlate with disease severity and will change with the clinical course. 15 , 30 , 31 To match the clinical course between groups, all patients were followed daily and LUS findings closest to the time of intubation or at the day of highest oxygen demand were used for analysis. The onset of severe respiratory distress in patients with COVID‐19 has been reported about 5 days into hospitalization, however, with large individual differences. 32 In the presented data, time from admission to start of IMV treatment or highest oxygen demand also showed large variation. Altogether, patient selection and timing of LUS examination will determine the clinical implication of conclusions.
Our data confirmed the typical pattern of COVID‐19 pneumonia on LUS: bilateral B‐lines, thickened pleural lines and consolidations of various sizes mixed with spared areas. 7 , 9 , 26 , 29 Pleural effusions were rare. 29 , 30 As previously reported, large consolidations were predominantly found in posterior lung regions. 8 , 30 Based on this, it is of value to include also posterior regions in the ultrasound examination. In accordance to previous studies, the overall presence of large consolidations was not associated with requirement for IMV treatment in COVID‐19 pneumonia. 15 , 30 Differential diagnoses always need to be considered since the described LUS findings are not specific for COVID‐19. 33 , 34 Unexpected LUS findings assisted in identifying coexisting conditions that required urgent treatment in our study.
Patients requiring IMV treatment presented with higher LUS‐score and higher number of pathologic regions, compared to patients not requiring IMV treatment. However, a LUS‐score cut‐off to identify IMV requirement had only a moderate diagnostic value due to high individual variation among patients with moderate to severe respiratory failure. Also in ARDS, attempts to identify a LUS‐score cut‐off to separate mild, moderate and severe ARDS have been conflicting. 26 , 27 , 28 , 35 , 36 IMV requirement partially depends on patient related factors beyond lung pathology identified on LUS. Moreover, individual variations of LUS‐score might be based on the fact that COVID‐19 pneumonia seems to differ from ARDS in general. Compared to ARDS, a discrepancy between degree of lung compliance and hypoxia has been described in COVID‐19. Based on a theoretical model, 2 phenotypes depending on lung compliance have been suggested. 37 , 38 The low compliance type (L‐type) is characterized by a predominant B‐line pattern and the high compliance type (H‐type) presents mainly with large consolidations. 26 , 38 Variation in LUS‐score for individuals with similar oxygen demand might be explained by a comparatively low LUS‐score, for the L‐type, and a high LUS‐score, for the H‐type. Another explanation of the discrepancy of oxygen demand and findings on LUS‐score could be a V/Q‐mismatch due to alveolar capillary microthrombi described in COVID‐19. 39 So far, there is no consensus regarding a LUS‐protocol for COVID‐19 pneumonia. 12 , 13 , 26 , 40 In our cohort, LUS‐score proved valuable by correlation to disease severity. 11 , 26 Manivel et al. have suggested a modified protocol for COVID‐19, adding subpleural consolidations to the score. 12 This scoring system seemed inferior to LUS‐score in our patients. Frequent findings of minor subpleural consolidations in the non‐IMV‐group resulted in a higher score according to Manivel et al., while these findings had no obvious impact on respiratory status. 12
By natural means, respiratory parameters were strong predictors for identification of patients requiring IMV treatment. A majority of patients in the IMV‐group reported dyspnea and had high respiratory rate, low SpO2 despite high oxygen delivery previous to intubation and a PaO2/FiO2‐ratio corresponding to moderate ARDS. 22 ROX index, based on oxygen demand and respiratory rate, has previously proved a high diagnostic value for prediction of lower risk for IMV in pneumonia. 25 ROX index has been suggested to also be useful in COVID‐19 pneumonia but has not been validated so far. 38 Our data confirmed that the previously validated ROX index cut‐off for prediction of IMV treatment in pneumonia can be used also in COVID‐19. 25 In summary, we agree with the panel opinion reported by Liu et al. on using LUS for guidance of severity of disease and differential diagnosis but to “decide on level of care on clinical appearance.” 14
There are some limitations of this study. False low LUS‐score in the IMV‐group might have been recorded due to examination too far prior to intubation or due to improved aeration after initiation of IMV‐treatment. It was not possible to blind the physicians performing LUS examination for the clinical respiratory status of the patients, which could have added bias to the interpretation of LUS‐findings. As a single‐center‐study, with a small number of patients, the findings should be verified in other populations.
Conclusions
LUS‐score had only a moderate diagnostic value for identification of requirement of invasive mechanical ventilation in patients with moderate to severe COVID‐19. Lung ultrasound served as complement to bedside assessment by guidance of disease severity and was an important tool for differential diagnostics. Respiratory rate and oxygenation index (ROX index) proved applicable for guiding need of IMV‐treatment in COVID‐19.
Acknowledgments
We want to thank all patients for their participation and health personnel at the intensive care unit and care units of Falun hospital for help with patient recruitment. A special acknowledgement is given to Jeanette Vestling (MD), Ida Mattsson (MD), Lars Dittlau (MD) and Oskar Ostwald (MD) for LUS examinations. Torbjörn Roos (MD) and Erik Degerman (MD) are acknowledged for supporting and facilitating the study during the hectic time of the pandemic.
C.S. initiated the study and takes responsibility for the content of the manuscript, including data and analysis. C.S. and M.H. performed data analysis and wrote the first draft of the manuscript, with help from C.K. All authors took active part in study design, logistics, data collection and interpretation of the data, reviewed and approved the final manuscript. The authors declare no conflicts of interest. Financial support was provided by Center for Clinical Research (CKF) Dalarna‐Uppsala University. The funding sources did not have influence on study design, data collection or conclusions drawn from the data set.
Data has not been previously published or presented at an official meeting.
Registrator Maria Hårdstedt at https://clinicaltrails.gov; registration number NCT04412551
All of the authors of this article have reported no disclosures.
References
- 1. World Health Organization timeline : WHO's COVID‐19 response. https://www.who.int/emergencies/diseases/novel‐coronavirus‐2019/interactive‐timeline#!
- 2. Guan W, Ni Z, Hu Y, et al. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med 2020; 382:1708–1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Gattinoni L, Chiumello D, Caironi P, et al. COVID‐19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med 2020; 46:1099–1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Brochard L, Slutsky A, Pesenti A. Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am J Respir Crit Care Med 2017; 195:438–442. [DOI] [PubMed] [Google Scholar]
- 5. Volpicelli G, Elbarbary M, Blaivas M, et al. International evidence‐based recommendations for point‐of‐care lung ultrasound. Intensive Care Med 2012; 38:577–591. [DOI] [PubMed] [Google Scholar]
- 6. Lichtenstein DA. Lung ultrasound in the critically ill. Ann Intensive Care 2014; 4:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Peng QY, Wang XT, Zhang LN. Findings of lung ultrasonography of novel corona virus pneumonia during the 2019–2020 epidemic. Intensive Care Med 2020; 46:849–850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Smith MJ, Hayward SA, Innes SM, Miller ASC. Point‐of‐care lung ultrasound in patients with COVID‐19 – a narrative review. Anaesthesia 2020; 75:1096–1104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Lomoro P, Verde F, Zerboni F, et al. COVID‐19 pneumonia manifestations at the admission on chest ultrasound, radiographs, and CT: single‐center study and comprehensive radiologic literature review. Eur J Radiol Open 2020; 7:100231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Yasukawa K, Minami T. Point‐of‐care lung ultrasound findings in patients with COVID‐19 pneumonia. Am J Trop Med Hyg 2020; 102:1198–1202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Bouhemad B, Mongodi S, Via G, Rouquette I. Ultrasound for “lung monitoring” of ventilated patients. Anesthesiology 2015; 122:437–447. [DOI] [PubMed] [Google Scholar]
- 12. Manivel V, Lesnewski A, Shamim S, Carbonatto G, Govindan T. CLUE: COVID‐19 lung ultrasound in emergency department. EMA ‐ Emerg Med Australas 2020; 32:694–696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Soldati G, Smargiassi A, Inchingolo R, et al. Proposal for international standardization of the use of lung ultrasound for patients with COVID‐19: a simple, quantitative, reproducible method. J Ultrasound Med 2020; 39:1413–1419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Liu RB, Tayal VS, Panebianco NL, et al. Ultrasound on the frontlines of COVID‐19: report from an international webinar. Acad Emerg Med 2020; 27:523–526. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Deng Q, Zhang Y, Wang H, et al. Semiquantitative lung ultrasound scores in the evaluation and follow‐up of critically ill patients with COVID‐19: a single‐center study. Acad Radiol 2020; 27:1363–1372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Zieleskiewicz L, Markarian T, Lopez A, et al. Comparative study of lung ultrasound and chest computed tomography scan in the assessment of severity of confirmed COVID‐19 pneumonia. Intensive Care Med 2020; 46:1707–1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Lichter Y, Topilsky Y, Taieb P, et al. Lung ultrasound predicts clinical course and outcomes in COVID‐19 patients. Intensive Care Med 2020; 46:1873–1883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Tung‐Chen Y. Lung ultrasound in the monitoring of COVID‐19 infection. Clin Med 2020; 20:e62–e65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. WHO R&D Blueprint, novel Coronavirus . COVID‐19 therapeutic trial synopsis. https://www.who.int/publications/i/item/covid-19-therapeutic-trial-synopsis. Accessed February 18, 2020.
- 20. Rice TW, Wheeler AP, Bernard GR, Hayden DL, Schoenfeld DA, Ware LB. Comparison of the SpO2/FIO2 ratio and the PaO 2/FIO2 ratio in patients with acute lung injury or ARDS. Chest 2007; 132:410–417. [DOI] [PubMed] [Google Scholar]
- 21. Ciccarese F, Chiesa AM, Feletti F, et al. The senile lung as a possible source of pitfalls on chest ultrasonography and computed tomography. Respiration 2015; 90:56–62. [DOI] [PubMed] [Google Scholar]
- 22. Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: The Berlin definition. JAMA 2012; 307:2526–2533. [DOI] [PubMed] [Google Scholar]
- 23. Vincent JL. Convertion tables for estimating FiO2, EPIC II. https://www.intensive.org/epic2/Documents/Estimation%20of%20PO2%20and%20FiO2.pdf.
- 24. Vincent JL, Rello J, Marshall J, et al. International study of the prevalence and outcomes of infection in intensive care units. JAMA 2009; 302:2323–2329. [DOI] [PubMed] [Google Scholar]
- 25. Roca O, Messika J, Caralt B, et al. Predicting success of high‐flow nasal cannula in pneumonia patients with hypoxemic respiratory failure: The utility of the ROX index. J Crit Care 2016; 35:200–205. [DOI] [PubMed] [Google Scholar]
- 26. Gargani L, Soliman‐Aboumarie H, Volpicelli G, Corradi F, Pastore MC, Cameli M. Why, when, and how to use lung ultrasound during the COVID‐19 pandemic: enthusiasm and caution. Eur Heart J Cardiovasc Imaging 2020; 21:941–948. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Pisani L, Vercesi V, van Tongeren PSI, et al. The diagnostic accuracy for ARDS of global versus regional lung ultrasound scores ‐ a post hoc analysis of an observational study in invasively ventilated ICU patients. Intensive Care Med Exp 2019; 7:44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Li L, Yang Q, Li L, et al. The value of lung ultrasound score on evaluating clinical severity and prognosis in patients with acute respiratory distress syndrome. Chinese Crit Care Med 2015; 27:579–584. [DOI] [PubMed] [Google Scholar]
- 29. Zhang Y, Xue H, Wang M, He N, Lv Z, Cui L. Lung ultrasound findings in patients with coronavirus disease (COVID‐19). Am J Roentgenol 2021; 216:80–84. [DOI] [PubMed] [Google Scholar]
- 30. Xing C, Li Q, Du H, Kang W, Lian J, Yuan L. Lung ultrasound findings in patients with COVID‐19 pneumonia. Crit Care 2020; 24:174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Dargent A, Chatelain E, Kreitmann L, et al. Lung ultrasound score to monitor COVID‐19 pneumonia progression in patients with ARDS. PLoS One 2020; 15:e0236312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Xu J, Yang X, Yang L, et al. Clinical course and predictors of 60‐day mortality in 239 critically ill patients with COVID‐19: a multicenter retrospective study from Wuhan, China. Crit Care 2020; 24:219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Vetrugno L, Bove T, Orso D, Bassi F, Boero E, Ferrari G. Lung ultrasound and the COVID‐19 “pattern”: not all that glitters today is gold tomorrow. J Ultrasound Med 2020; 39:2281–2282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Sekiguchi H, Schenck LA, Horie R, et al. Critical care ultrasonography differentiates ARDS, pulmonary edema, and other causes in the early course of acute hypoxemic respiratory failure. Chest 2015; 148:912–918. [DOI] [PubMed] [Google Scholar]
- 35. Zhao Z, Jiang L, Xi X, et al. Prognostic value of extravascular lung water assessed with lung ultrasound score by chest sonography in patients with acute respiratory distress syndrome. BMC Pulm Med 2015; 15:98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Sanjan A, Krishnan SV, Abraham SV, Palatty BU. Utility of point‐of‐care lung ultrasound for initial assessment of acute respiratory distress syndrome patients in the emergency department. J Emerg Trauma Shock 2019; 12:248–253. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Gattinoni L, Coppola S, Cressoni M, Busana M, Rossi S, Chiumello D. COVID‐19 does not lead to a “typical” acute respiratory distress syndrome. Am J Respir Crit Care Med 2020; 201:1299–1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Denault AY, Delisle S, Canty D, et al. A proposed lung ultrasound and phenotypic algorithm for the care of COVID‐19 patients with acute respiratory failure. Can J Anesth 2020; 67:1393–1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Ackermann M, Verleden SE, Kuehnel M, et al. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid‐19. N Engl J Med 2020; 383:120–128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Schultz MJ, Sivakorn C, Dondorp AM. Challenges and opportunities for lung ultrasound in novel Coronavirus disease (COVID‐19). Am J Trop Med Hyg 2020; 102:1162–1163. [DOI] [PMC free article] [PubMed] [Google Scholar]


