Abstract
Aims/Introduction
Excessive dietary salt or low potassium intakes are strongly correlated with insulin resistance (IR) and type 2 diabetes mellitus. In epidemiological and experimental studies, increased serum retinol‐binding protein 4 (RBP4) contributes to the pathogenesis of type 2 diabetes mellitus. Herein, we hypothesized that RBP4 might be an adipocyte‐derived “signal” that plays the crucial role in salt‐related insulin resistance or type 2 diabetes mellitus. This study aimed to assess whether salt consumption and potassium supplementation influence serum RBP4 levels in healthy individuals.
Materials and Methods
A total of 42 participants (aged 25–50 years) in a rural area of Northern China were successively provided normal (3 days at baseline), low‐salt (7 days; 3 g/day NaCl) and high‐salt (7 days; 18 g/day) diets, and a high‐salt diet with potassium additive (7 days; 18 g/day NaCl and 4.5 g/day KCl). Urinary sodium and potassium were measured to ensure compliance to dietary intervention. Then, RBP4 levels were evaluated by enzyme‐linked immunosorbent assay.
Results
High salt intake significantly raised serum RBP4 levels in healthy participants (17.5 ± 0.68 vs 28.6 ± 1.02 µg/mL). This phenomenon was abrogated by potassium supplementation (28.6 ± 1.02 vs 17.6 ± 0.88 µg/mL). In addition, RBP4 levels presented positive (r = 0.528, P < 0.01) and negative (r = −0.506, P < 0.01) associations with 24‐h urinary sodium‐ and potassium excretion levels.
Conclusions
RBP4 synthesis is motivated by high salt intake and revoked by potassium supplementation. Our pioneer work has contributed to the present understanding of salt‐induced insulin resistance or type 2 diabetes mellitus.
Keywords: Dietary intervention, Insulin resistance, Retinol‐binding protein 4
Excess dietary salt and low potassium intake are strongly correlated with insulin resistance or type 2 diabetes mellitus. Retinol‐binding protein 4, termed “adipokines”, is associated with insulin resistance or type 2 diabetes mellitus. Our study found salt loading can enhance the production of circulating retinol‐binding protein 4; inversely, potassium supplementation can blunt the effects of excessive retinol‐binding protein 4.

Introduction
Individuals consuming high amounts of salt are at elevated risk of cardiovascular events, and the related health outcomes constitute a public health concern worldwide 1 . Multiple reports have established associations of high sodium consumption with insulin resistance (IR) or type 2 diabetes mellitus 2 , 3 . Potassium is considered to exert beneficial effects on cardiovascular diseases and associated mortality. Recently, a population‐based study showed that reduced dietary potassium correlates with new‐onset diabetes 4 . Meanwhile, it was shown that both dietary and serum potassium levels constitute a potential risk factor for thiazide‐induced diabetes 5 , 6 , 7 .
Recent studies have focused on adipocytes as the source of “adipokines,” because they are involved in the pathogenesis of obesity‐associated ailments, including IR, type 2 diabetes mellitus and cardiovascular disease. In addition to adipokines specifically produced in the fat tissue, an increasing number of well‐known molecules are secreted by adipocytes. Animal and human experiments have shown that high‐sodium diets increase fat tissue mass, as well as adipocyte size 3 , 8 . Our previous study also showed that elevated salt consumption stimulates the synthesis of several adipokines, including adiponectin and tumor necrosis factor‐α 9 .
Retinol‐binding protein 4 (RBP4), a 21‐kDa member of the “lipocalin superfamily,” was originally considered to be mostly found in the liver; it constitutes the only retinol (vitamin A) transporter 10 . More recently, Yang et al. 11 found that significant amounts of circulating RBP4 are released from adipocytes. Transgenic overexpression of human RBP4 and administration of recombinant RBP4 in healthy mice reduce insulin sensitivity, which is ameliorated by RBP4 gene deletion or RBP4 level normalization in obese rodents 11 . Indeed, previous reports have shown that high serum RBP4 levels are associated with IR in humans. A study by Sun et al. 12 included 2,091 Chinese adult participants (aged 50–70 years), and found plasma RBP4 to independently correlate with 6‐year odds of developing type 2 diabetes mellitus. Hence, RBP4 is considered a novel adipokine in humans, possibly relating to obesity, IR, type 2 diabetes and metabolic syndrome.
Little attention has been focused on the link between salt intake and RBP4. To assess the influence of salt or potassium on IR or type 2 diabetes mellitus, prospective trials that minimize bias are required. We tested the hypothesis that RBP4 synthesis contributes to the progression of IR in excessive salt intake and potassium supplementation. Therefore, the effects of salt consumption and added potassium on serum RBP4 levels were assessed in healthy individuals.
Methods
Participants
A total of 42 participants with comparable eating habits were recruited from a rural community of Northern China and briefly queried regarding their medical history. Participants with a history of obesity, hypertension, liver or renal diseases, IR, or type 2 diabetes mellitus were excluded. Hypertension was indicated by systolic blood pressure (BP) ≥140 mmHg and/or diastolic BP ≥90 mmHg. According to the criteria recommended by the Working Group on Obesity in China 13 , obese individuals had body mass index (BMI) values ≥28 kg/m2. The participants had no history of smoking. The study obtained approval from the institutional ethics committee of Xi’an Jiaotong University Medical School, and all participants provided written informed consent.
Protocol
First, the participants were assessed for 3 days in the baseline period, recording their clinical and physical (height, weight and BP) data. During this initial experimental phase, they were provided a regular salt diet. Then, the participants received low‐salt (3 g of NaCl daily; 7 days) and high‐salt (18 g of NaCl daily; 7 days) diets successively, followed by a high‐salt diet and potassium supplementation (4.5 g of KCl daily) for 7 days (Figure 1). The participants received intensive dietary intervention, when the participants arrived at the study kitchen for their breakfast, lunch and dinner during the entire intervention period. All foods were prepared without salt, and prepacked salt was added to meal when it was served by the study staff. The participants were instructed to avoid table or cooking salt, high‐sodium foods and nitrite‐/nitrate‐rich foods throughout the study. The study protocol was consistent with Genetic Epidemiology Network of Salt Sensitivity (GenSalt) 14 .
Figure 1.

Overview of dietary intervention.
Biochemical analysis
Blood glucose levels were assessed by the glucose oxidase method. Serum total cholesterol, triglyceride and high‐density lipoprotein cholesterol levels were also evaluated. Blood specimens for fasting serum RBP4 levels were cooled on ice immediately after collection. The serum was kept at −80°C until analysis. RBP4 levels were assessed by sandwich enzyme‐linked immunosorbent assay with anti‐RBP4 antibodies (Wuhan USCN Sciences Corporation, Wuhan, China). Five serum specimens were used for intra‐ and interassay variations, and coefficients ranging between 3.4% and 4.8% (mean 4.1%), and from 4.7% to 6.2% (mean 5.4%), respectively, were obtained.
Twenty‐four hour urinary sodium and potassium level assessment
Urine specimens (24 h) were obtained at baseline and on the final days of various intervention periods, and frozen at −40°C until use. Sodium and potassium levels in the urine were measured with ion‐selective electrodes (Hitachi Ltd., Tokyo, Japan).
Statistical analysis
Data are shown as the mean ± standard deviation. Repeated measures anova was used to assess biochemical parameters. Differences between biochemical markers obtained during the various dietary intervention periods were calculated by analysis of variance with the repeated measures design. Age, sex and BMI were adjusted in multivariable analysis. SPSS 16.0 (SPSS Inc., Chicago, IL, USA) was used for statistical analyses, with two‐tailed P < 0.05 showing statistical significance.
Results
Characteristics of the participants
All enrolled participants completed the present interventional study. They were aged 50.9 ± 1.29 years, with systolic and diastolic BPs of 110.7 ± 2.2 and 72.6 ± 1.3 mmHg, respectively (Table 1). The mean 24‐h sodium level in urine was 183.2 ± 10.1 mmol/day, corresponding to 8 g of salt consumed per day. The mean potassium level in urine was 45.3 ± 2.8 mmol/day.
Table 1.
Baseline demographic and clinical characteristics
| Parameter | Values |
|---|---|
| Mean age (years) | 50.9 ± 1.29 |
| Sex (male/female) | 21/21 |
| BMI (kg/m2) | 23.5 ± 0.42 |
| Systolic BP (mm Hg) | 110.7 ± 2.2 |
| Diastolic BP (mm Hg) | 72.6 ± 1.3 |
| Fasting glucose (mmol/L) | 3.9 ± 0.1 |
| Total cholesterol (mmol/L) | 4.18 ± 0.13 |
| Triglycerides (mmol/L) | 1.31 ± 0.11 |
| LDL cholesterol (mmol/L) | 2.35 ± 0.11 |
| HDL cholesterol (mmol/L) | 1.21 ± 0.04 |
| Serum creatinine (µmol/L) | 57.3 ± 1.35 |
Effects of dietary intervention on BP, and urinary sodium and potassium excretion
As shown in Table 2, BP remained relatively stable after all three interventions. Sodium levels in urine were markedly reduced after a change from baseline (normal) to a low‐salt diet, and showed an increase after low‐salt diet replacement by a high‐salt diet (P < 0.05; Table 2). Potassium supplementation resulted in elevated potassium levels and slightly higher sodium levels in urine.
Table 2.
Blood pressure levels (mm Hg) and 24‐h urinary sodium and potassium excretions (mmol/day) at baseline and during dietary interventions
| SBP | DBP | 24‐h Urinary Na+ (mmol/day) | 24‐h Urinary K+ (mmol/day) | |
|---|---|---|---|---|
| Baseline | 110.7 ± 2.2 | 72.6 ± 1.3 | 183.2 ± 10.1 | 45.3 ± 2.8 |
| Low‐salt diet | 108.7 ± 1.8 | 73.5 ± 1.1 | 98.1 ± 5.3 | 36.9 ± 2.3 |
| High‐salt diet | 117.3 ± 2.6 | 77.7 ± 1.3 | 228.3 ± 10.9 | 42.9 ± 3.6 |
| High‐salt diet with potassium supplement | 107.5 ± 1.9 | 72.2 ± 1.3 | 273.4 ± 9.6 | 79.2 ± 3.1 |
P < 0.05 vs low‐salt diet. DBP, diastolic blood pressure; SBP, systolic blood pressure.
Effects of high salt consumption and potassium supplementation on RBP4 synthesis
The present results showed that serum RBP4 amounts were significantly decreased after low salt intake compared with baseline values (17.5 ± 0.68 vs 26.8 ± 1.22 µg/mL, P < 0.01). A high‐salt diet resulted in elevated serum RBP4 levels compared with a low‐salt diet (28.6 ± 1.02 vs 17.5 ± 0.68 µg/mL; Figure 2). Interestingly, high‐salt diet‐associated elevation of serum RBP4 was prevented by potassium supplementation (28.6 ± 1.02 vs 17.6 ± 0.88 µg/mL). Meanwhile, RBP4 concentration was associated with 24‐h urinary sodium amounts in both low‐ and high‐salt intervention phases (r = 0.528, P < 0.01). Finally, serum RBP4 concentration was negatively correlated with 24 h urinary potassium levels in both high‐salt and high‐salt/potassium supplementation intervention phases (r = −0.506, P < 0.01).
Figure 2.
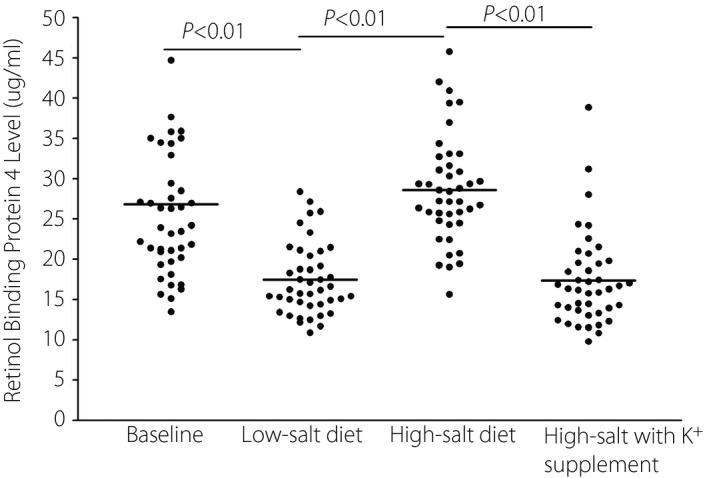
Effect of salt intake and potassium supplementation on serum retinol‐binding protein 4 levels.
Discussion
The present study showed that excessive salt consumption stimulates RBP4 synthesis, which might contribute to IR and diabetes mellitus. Meanwhile, potassium supplementation blunts this phenomenon.
Accumulating evidence suggests that high serum RBP4 levels correlate with IR in humans 12 , 14 , 15 , 16 , 17 , 18 , 19 , as well as cardiovascular disease, including hypertension, stroke and atherosclerosis 20 , 21 , 22 , 23 . RBP4 downregulates the glucose transporter, GLUT4, constituting the rate‐limiting step in insulin‐induced glucose transport across the muscle and adipocyte membrane 11 , 24 . Furthermore, RBP4 can also exacerbate endothelial function and programmed cell death by increasing oxidative stress in the mitochondria through modulation of mitochondrial fusion and fission 19 , 25 , 26 , 27 , which might also promote hypertension and atherosclerosis occurrence.
Human and experimental studies have shown that in addition to promoting hypertension occurrence, high sodium consumption results in IR 2 , 3 , 28 , 29 . Insulin sensitizers (for example, pioglitazone), which ameliorate insulin sensitivity in fat‐fed and obese animals, do not prevent high‐salt‐associated IR 30 , 31 , 32 , showing that high‐salt‐associated IR differs from other IR models. High‐sodium consumption results in IR, mainly because of vascular dysfunction in muscles, in the case of no myocyte‐associated or liver IR 28 . This finding directly contrasts the notion that IR originates from high fat/obesity, in which insulin signaling responsible for glucose uptake is repressed. Interestingly, quinapril prevents the development of high‐sodium‐derived muscle IR by preserving microvascular insulin responsiveness 28 . The present study is the first to show that salt loading significantly enhanced serum RBP4 levels. We previously reported that a high‐salt diet impairs endothelial function in normotensive individuals 33 , especially in salt‐sensitive patients. We believe that sodium‐derived IR might be associated with elevated RBP4, which leads to vascular dysfunction. The current findings provide new insights into the association of sodium intake with IR.
We also found that potassium supplementation reversed the influence of a high‐salt diet on RBP4 levels; however, the underlying mechanisms are unknown. Recent reports have assessed the associations of potassium with obesity and metabolic syndrome, and found that high potassium levels alleviate obesity and metabolic syndrome risk 7 , 33 , 34 , 35 , 36 . We recently reported potassium supplementation in normotensive individuals affects nitrogen oxide production by reducing asymmetric dimethylarginine levels during high salt loading 37 . Furthermore, potassium supplementation counteracts salt‐associated osteoprotegerin elevation through oxidative stress reduction and endothelial function protection 38 . Overall, potassium supplementation might prevent salt‐associated IR by inhibiting the expression of RBP4, which can stimulate the oxygen‐reactive free radical generation and endothelial dysfunction.
The present research had several remarkable advantages. First, the participants were recruited from rural communities where lifestyle and environmental risk factors were homogeneous. In addition, participants with a BMI >28 kg/m2 were excluded; hence, the hybrid effect of BMI might have been minimized. The compliance with the diet intervention was guaranteed through the urinary sodium excretion assessment.
The limitations of the present study should be mentioned. First, the sample size was relatively small, and these findings should be confirmed in more extensive studies. In addition, the causal relationship between RBP4 and IR was not estimated. Finally, RBP4 amounts were determined by enzyme‐linked immunosorbent assay, whereas immunoblot standardized to RBP4 is the method of choice for such assessment.
The present study might give a tantalizing clue to the mechanisms by which high sodium intake contributes to IR. Potassium supplementation can reverse the effect of elevated RBP4 levels (Figure 3). These findings also shed some new light on a therapeutic target for the treatment of IR.
Figure 3.
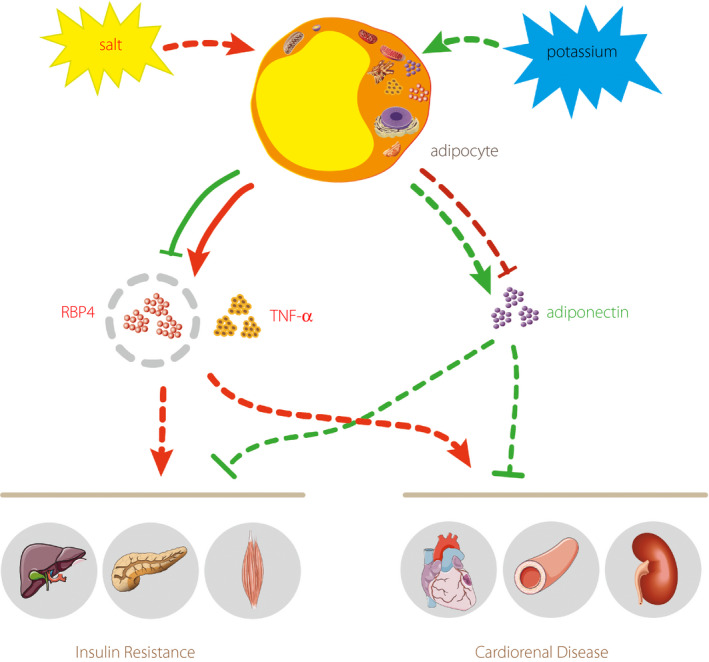
The potential mechanism of diet‐induced insulin resistance and cardiovascular disease. RBP4, retinol‐binding protein 4; TNF‐α, tumor necrosis factor‐α.
Disclosure
The authors declare no conflict of interest.
Acknowledgments
We gratefully acknowledge the study participants for their invaluable contribution. This study was supported by the Natural Science Basic Research Plan of Shaanxi Province (No. 2017KJXX‐70), the Natural Science Foundation of Shaanxi Province (Nos. 2016JM8025 and 2017JM8117) and the National Natural Science Foundation of China (No. 81400328).
J Diabetes Investig. 2021
References
- 1. Bibbins‐Domingo K, Chertow GM, Coxson PG, et al. Projected effect of dietary salt reductions on future cardiovascular disease. N Engl J Med 2010; 362: 590–599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Donovan DS, Solomon CG, Seely EW, et al. Effect of sodium intake on insulin sensitivity. Am J Physiol 1993; 264(5 Pt 1): E730–E734. [DOI] [PubMed] [Google Scholar]
- 3. Oh SW, Han KH, Han SY, et al. Association of sodium excretion with metabolic syndrome, insulin resistance, and body fat. Medicine (Baltimore) 2015; 94: e1650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Chatterjee R, Colangelo LA, Yeh HC, et al. Potassium intake and risk of incident type 2 diabetes mellitus: the Coronary Artery Risk Development in Young Adults (CARDIA) study. Diabetologia 2012; 55: 1295–1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Zillich AJ, Garg J, Basu S, et al. Thiazide diuretics, potassium, and the development of diabetes: a quantitative review. Hypertension 2006; 48: 219–224. [DOI] [PubMed] [Google Scholar]
- 6. Shafi T, Appel LJ, Miller ER, et al. Changes in serum potassium mediate thiazide‐induced diabetes. Hypertension 2008; 52: 1022–1029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lee H, Lee J, Hwang SS, et al. Potassium intake and the prevalence of metabolic syndrome: the Korean National Health and Nutrition Examination Survey 2008–2010. PLoS One 2013; 8: e55106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Zhu H, Pollock NK, Kotak I, et al. Dietary sodium, adiposity, and inflammation in healthy adolescents. Pediatrics 2014; 133: e635–e642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Liu F, Mu J, Yuan Z, et al. High salt intake fails to enhance plasma adiponectin in normotensive salt‐sensitive subjects. Nutrition 2012; 28: 422–425. [DOI] [PubMed] [Google Scholar]
- 10. Kotnik P, Fischer‐Posovszky P, Wabitsch M. RBP4: a controversial adipokine. Eur J Endocrinol 2011; 165: 703–711. [DOI] [PubMed] [Google Scholar]
- 11. Yang Q, Graham TE, Mody N, et al. Serum retinol binding protein 4 contributes to insulin resistance in obesity and type 2 diabetes. Nature 2005; 436: 356–362. [DOI] [PubMed] [Google Scholar]
- 12. Sun L, Qi Q, Zong G, et al. Elevated plasma retinol‐binding protein 4 is associated with increased risk of type 2 diabetes in middle‐aged and elderly Chinese adults. J Nutr 2014; 144: 722–728. [DOI] [PubMed] [Google Scholar]
- 13. Zhou B, Cooperative Meta‐analysis Group of Working Group on Obesity in China . Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults: study on optimal cut‐off points of body mass index and waist circumference in Chinese adults. Asia Pac J Clin Nutr. 2002; 11: S685–S693. [PubMed] [Google Scholar]
- 14. GenSalt Collaborative Research Group . GenSalt: rationale, design, methods and baseline characteristics of study participants. J Hum Hypertens 2007; 21: 639–646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Meisinger C, Ruckert IM, Rathmann W, et al. Retinol‐binding protein 4 is associated with prediabetes in adults from the general population: the Cooperative Health Research in the Region of Augsburg (KORA) F4 Study. Diabetes Care 2011; 34: 1648–1650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Graham TE, Yang Q, Bluher M, et al. Retinol‐binding protein 4 and insulin resistance in lean, obese, and diabetic subjects. N Engl J Med 2006; 354: 2552–2563. [DOI] [PubMed] [Google Scholar]
- 17. Broch M, Vendrell J, Ricart W, et al. Circulating retinol‐binding protein‐4, insulin sensitivity, insulin secretion, and insulin disposition index in obese and nonobese subjects. Diabetes Care 2007; 30: 1802–1806. [DOI] [PubMed] [Google Scholar]
- 18. Takebayashi K, Suetsugu M, Wakabayashi S, et al. Retinol binding protein‐4 levels and clinical features of type 2 diabetes patients. J Clin Endocrinol Metab 2007; 92: 2712–2719. [DOI] [PubMed] [Google Scholar]
- 19. Park SE, Kim DH, Lee JH, et al. Retinol‐binding protein‐4 is associated with endothelial dysfunction in adults with newly diagnosed type 2 diabetes mellitus. Atherosclerosis 2009; 204: 23–25. [DOI] [PubMed] [Google Scholar]
- 20. Ingelsson E, Sundstrom J, Melhus H, et al. Circulating retinol‐binding protein 4, cardiovascular risk factors and prevalent cardiovascular disease in elderly. Atherosclerosis 2009; 206: 239–244. [DOI] [PubMed] [Google Scholar]
- 21. Liu Y, Wang D, Chen H, et al. Circulating retinol binding protein 4 is associated with coronary lesion severity of patients with coronary artery disease. Atherosclerosis 2015; 238: 45–51. [DOI] [PubMed] [Google Scholar]
- 22. Solini A, Santini E, Madec S, et al. Retinol‐binding protein‐4 in women with untreated essential hypertension. Am J Hypertens 2009; 22: 1001–1006. [DOI] [PubMed] [Google Scholar]
- 23. Huang G, Wang D, Khan UI, et al. Associations between retinol‐binding protein 4 and cardiometabolic risk factors and subclinical atherosclerosis in recently postmenopausal women: cross‐sectional analyses from the KEEPS study. Cardiovasc Diabetol 2012; 11: 52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Wolf G. Serum retinol‐binding protein: a link between obesity, insulin resistance, and type 2 diabetes. Nutr Rev 2007; 65: 251–256. [DOI] [PubMed] [Google Scholar]
- 25. Wang J, Chen H, Liu Y, et al. Retinol binding protein 4 induces mitochondrial dysfunction and vascular oxidative damage. Atherosclerosis 2015; 240: 335–344. [DOI] [PubMed] [Google Scholar]
- 26. Du M, Martin A, Hays F, et al. Serum retinol‐binding protein‐induced endothelial inflammation is mediated through the activation of toll‐like receptor 4. Mol Vis 2017; 23: 185–197. [PMC free article] [PubMed] [Google Scholar]
- 27. Farjo KM, Farjo RA, Halsey S, et al. Retinol‐binding protein 4 induces inflammation in human endothelial cells by an NADPH oxidase‐ and nuclear factor kappa B‐dependent and retinol‐independent mechanism. Mol Cell Biol 2012; 32: 5103–5115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Premilovac D, Richards SM, Rattigan S, et al. A vascular mechanism for high‐sodium‐induced insulin resistance in rats. Diabetologia 2014; 57: 2586–2595. [DOI] [PubMed] [Google Scholar]
- 29. Kimura T, Hashimoto Y, Tanaka M, et al. Sodium‐chloride difference and metabolic syndrome: a population‐based large‐scale cohort study. Intern Med 2016; 55: 3085–3090. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Ogihara T, Asano T, Ando K, et al. Insulin resistance with enhanced insulin signaling in high‐salt diet‐fed rats. Diabetes 2001; 50: 573–583. [DOI] [PubMed] [Google Scholar]
- 31. Ye JM, Doyle PJ, Iglesias MA, et al. Peroxisome proliferator‐activated receptor (PPAR)‐alpha activation lowers muscle lipids and improves insulin sensitivity in high fat‐fed rats: comparison with PPAR‐gamma activation. Diabetes 2001; 50: 411–417. [DOI] [PubMed] [Google Scholar]
- 32. Pickavance LC, Brand CL, Wassermann K, et al. The dual PPARalpha/gamma agonist, ragaglitazar, improves insulin sensitivity and metabolic profile equally with pioglitazone in diabetic and dietary obese ZDF rats. Br J Pharmacol 2005; 144: 308–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Liu FQ, Mu JJ, Liu ZQ, et al. Endothelial dysfunction in normotensive salt‐sensitive subjects. J Hum Hypertens 2012; 26: 247–252. [DOI] [PubMed] [Google Scholar]
- 34. Chatterjee R, Colangelo LA, Yeh HC, et al. Potassium intake and risk of incident type 2 diabetes mellitus: the Coronary Artery Risk Development in Young Adults (CARDIA) Study. Diabetologia 2012; 55: 1295–1303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Cai X, Li X, Fan W, et al. Potassium and obesity/metabolic syndrome: a systematic review and meta‐analysis of the epidemiological evidence. Nutrients 2016; 8: 183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Shin D, Joh HK, Kim KH, et al. Benefits of potassium intake on metabolic syndrome: the fourth Korean National Health and Nutrition Examination Survey (KNHANES IV). Atherosclerosis 2013; 230: 80–85. [DOI] [PubMed] [Google Scholar]
- 37. Fang Y, Mu JJ, He LC, et al. Salt loading on plasma asymmetrical dimethylarginine and the protective role of potassium supplement in normotensive salt‐sensitive Asians. Hypertension 2006; 48: 724–729. [DOI] [PubMed] [Google Scholar]
- 38. Liu FQ, Liu SQ, Zhang Y, et al. Effects of salt loading on plasma osteoprotegerin levels and protective role of potassium supplement in normotensive subjects. Circ J 2016; 81: 77–81. [DOI] [PubMed] [Google Scholar]


