Abstract
A chondroitin sulfate-bone marrow (CS-BM) adhesive hydrogel was used to localize rhBMP-2 to enhance articular cartilage tissue formation. Chondrocyte pellet culture informed that 0.1 and 1 μg/ml of rhBMP-2 enhanced sulfated-GAG content. rhBMP-2 localization within the hydrogels was investigated and it was found that BM, CS-NHS, and rhBMP-2 levels and time affected rhBMP-2 retention. Retention was modulated from 82 to 99% over a three-week period for the material formulations investigated. To evaluate carrier efficacy, rhBMP-2 and bovine articular chondrocytes were encapsulated within CS-BM, and biochemical evaluation revealed significant increases in total collagen production with rhBMP-2. Histological analysis revealed more robust tissue formation and greater type-II collagen production with encapsulated rhBMP- 2. Subsequently, a subcutaneous culture of hydrogels revealed increased total collagen, type-II to type-I collagen ratio, and sulfated GAG in samples carrying rhBMP-2. These findings indicate the development of a multifunctional system capable of localizing rhBMP-2 to enhance repair tissue quality.
Keywords: Tissue Adhesive, Intraoperative Biologics, BMP-2, Growth Factor Delivery, Cartilage, Bone Marrow
Graphical Abstract
Introduction
Orthopaedic trauma and disease are largely treated by means of tissue fixation or joint1–3replacement utilizing metals, plastics, and ceramics.1–3 These devices are typically successful at restoring function and eliminating pain, but they are not permanent solutions. Wear-particle induced osteolysis and implant-induced stress shielding of bone result in bone resorption around the implant, limiting the life of these devices to around 10 to 15 years and allowing at most two revisions.4–6 Consequently, prospective patients younger than 55 years old are advised to wait for these therapies, and they must often live with pain for years prior to intervention.6 Injuries as a result of participation in sports and the military have created a burgeoning population of young7,8 adults suffering from early onset osteoarthritis (OA) seeking suitable treatments. There is a need for tissue engineering solutions to move away from these synthetic treatments and establish early interventions to enable tissue repair with long-term homeostasis.
Biologic hydrogel scaffolds offer a promising approach to repairing soft joint tissues such as cartilage. Natural cartilage is composed of a collagenous fiber network within a highly hydrated proteoglycan phase that is very similar to a hydrogel scaffold. Delivering a substrate that is chemically or physically similar to cartilage tissue presents a more natural environment to host cells and may facilitate more efficient tissue repair. Soft hydrogel substrates, both synthetic and naturally derived, have demonstrated the ability to support chondrogenesis by both mature and progenitor cells.9–11 More specifically, the combination of polysaccharide-blood based hydrogels has also previously demonstrated promise for enhancing microfracture repair in humans, and has been approved for human therapies.12 However, successful repair must combine tissue fixation of the scaffold and support for tissue in-growth and regeneration. Additionally, in certain cases involving severe trauma or for the regeneration of specialized tissues, healing will require more specific signaling and instruction.
The delivery of growth factors is an effective approach to stimulating and enhancing the quality of tissue repair. One superfamily of growth factors that has been applied to enhance musculoskeletal tissue engineering is the transforming growth factors (TGF). This group includes the TGF-β factors as well as the bone morphogenetic proteins (BMP). TGF-β is used to induce in vitro differentiation of bone marrow derived mesenchymal stem cells (MSCs) into chondrocytes.13 Additionally, BMPs have been applied in animal models or humans to enhance healing in tendon repair, ligament insertion, bone fusion, and articular cartilage regeneration.14–17 Bolus delivery of these growth factors through injection is typically not a suitable delivery method to stimulate long-term tissue regeneration due to short in vivo half-life.18 One effective way to generate a sustained presence of these growth factors is to load them into a scaffold. Combining BMPs with a multifunctional material specifically designed to enhance regeneration may lead to superior quality of tissue repair and long-term homeostasis.
We have developed a bioactive chondroitin sulfate adhesive to be combined with intraoperative biologics into a hydrogel to enhance quality of tissue repair and integration.19,20 Previously, the bone marrow hydrogel was used to support MSC osteogenesis and meniscus fusion.21,22 Here, the hydrogel was combined with rhBMP-2 to enhance hyaline cartilage tissue production by increasing type-II collagen and sulfated GAG content. Histological, immunohistochemical, and biochemical methods were used to assess the effect of rhBMP-2 on chondrocytes in pellet culture and encapsulated with CS-BM hydrogels in vitro and in vivo in a subcutaneous implant culture model. Additionally, a BMP-2 enzyme-linked immunosorbent assay (ELISA) was used to assess rhBMP-2 retention and release from CS-BM hydrogels with varying material formulation and growth factor loading. In total, we demonstrated the ability of an adhesive bone marrow hydrogel to deliver spatially localized rhBMP-2 to enhance tissue production by articular chondrocytes.
Materials and Methods
Synthesis of chondroitin sulfate succinimidyl succinate (CS-NHS).
The adhesive polymer CS-NHS was synthesized as previously described.19 Briefly, phosphate buffered saline (PBS) containing 7% (w/v) CS (25 KDA, New Zealand Pharmaceuticals Ltd., Palmerston North, New Zealand), 10% (w/v) 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) and 3.8% (w/v) n- hydroxysuccinimide (NHS) was reacted for 10 minutes at 37°C. The solution was frozen and CS-NHS was precipitated and washed with −20°C ethanol. The dried product was stored under argon at −20°C.
Chondrocyte Isolation.
Chondrocytes were isolated from juvenile bovine femoral condyles and patellar groove (Research 87, Inc. Boylston, MA) using a previously described collagenase digest.23
Chondrocyte Pellet Culture.
Chondrocyte pellets (0.33 million cells) were formed in the bottom of 0.5 ml Eppendorf tubes by centrifuging at 160 g for 15 minutes. Pellets were cultured in 300 μl chondrocyte media for three days to allow pellet formation.24 After the third day, chondrocyte media was added containing 0, 0.1, or 1 μg/ml rhBMP-2 (Pfizer, Inc. Cambridge, MA) and this was considered day ‘zero’. Pellets were harvested for biochemistry (n = 3) and histology (n = 1) at days 0, 3, 6, and 10.
Hydrogel Fabrication.
CS-NHS dissolved in PBS was mixed with freeze-thawed bovine bone marrow aspirate (Thomas D. Morris, Inc., Reisterstown, MD) at room temperature to form chondroitin sulfate-bone marrow hydrogels. The bone marrow was freeze-thawed for several cycles to lyse any cells. rhBMP-2 solutions were added to the bone marrow component just prior to mixing. The precursor mixture was allowed ten minutes to cross-link before being placed into collection media for growth factor release study or chondrocyte growth media for chondrogenesis studies. CS-NHS concentrations and bone marrow volume fractions are outlined in the methods sections for each experiment.
rhBMP-2 Retention in CS-BM Hydrogels.
The effect of several factors, including CS-NHS, BM, rhBMP-2, and time, on the retention of rhBMP-2 from CS-BM hydrogels was investigated using a Design of Experiments method. This method allows the user to vary several factors simultaneously while measuring a response. Here, the DOE-Custom Designer option in the statistical software JMP (Version 10, SAS Institute, Inc. Cary, NC) was used to select combinations of CS-NHS concentration (30, 40, 50, 60, 70 mg/ml), BM loading volume fraction (30, 40, 50, 60, 70%), and rhBMP-2 loading concentration (50, 100 μg/ml) for a Response Surface design, which can be used to estimate the coefficients of the factors effecting rhBMP-2 retention. This design generated the following regression model:
where Y = rhBMP-2 retention normalized to initial loading, ai = the factor coefficient, Xi, X2, X3, and X4 = the values of CS-NHS content, BM volume fraction, rhBMP-2 loading concentration, and days. The selected CS-NHS and BM concentrations largely cover the technically feasible range of hydrogel formulations, and the rhBMP-2 values are in the range of doses shown to be effective in animal studies.25,26 The response, rhBMP-2 retention normalized to initial loading, was measured over a 3-week time course (1, 5, 9, 13, 17, and 21 days).
To measure rhBMP-2 retention, 30 μl hydrogels were formed using the methods outlined in the hydrogel fabrication methods section and according to the formulations found in Table 1. The hydrogels were placed into 500 μl of PBS collection medium containing 20% FBS. Hydrogels were incubated at 37°C and shaken at 4 hz. At each time point, medium was collected and replaced with new PBS/FBS. Collection medium was frozen at −80°C until ELISA was performed. rhBMP-2 ELISA (SYD Labs, Inc. Malden, MA) was performed on all samples according to manufacturer protocol, and data was analyzed using JMP software. Retention was calculated by subtracting cumulative mass of rhBMP-2 released at each time point from total mass initially loaded, then normalized as a percentage of initial loading mass.
Table 1:
Summary of hydrogel and loading condition used in retention study
| Run | CS-NHS (mg/ml) |
BM (Vol Fraction) |
rhBMP-2 (μg/ml) |
|---|---|---|---|
| 1 | 30 | 0.3 | 100 |
| 2 | 30 | 0.5 | 50 |
| 3 | 30 | 0.6 | 50 |
| 4 | 40 | 0.4 | 100 |
| 5 | 40 | 0.6 | 50 |
| 6 | 50 | 0.3 | 50 |
| 7 | 50 | 0.4 | 100 |
| 8 | 50 | 0.5 | 50 |
| 9 | 50 | 0.7 | 100 |
| 10 | 60 | 0.3 | 100 |
| 11 | 60 | 0.5 | 50 |
| 12 | 60 | 0.6 | 100 |
| 13 | 60 | 0.7 | 50 |
| 14 | 70 | 0.3 | 50 |
| 15 | 70 | 0.4 | 50 |
| 16 | 70 | 0.5 | 100 |
| 17 | 70 | 0.6 | 100 |
| 18 | 70 | 0.7 | 50 |
In Vitro Chondrogenesis Experiment.
Chondroitin sulfate-bone marrow hydrogels containing bovine chondrocytes and rhBMP-2 were formed to evaluate the efficacy of the encapsulated growth factor. Chondrocytes and rhBMP-2 were suspended in bone marrow prior to mixing with a CS-NHS solution (100 mg/ml) in PBS. The final hydrogel contents contained 30 mg/ml CS- NHS, 70 percent by volume bone marrow, 2×107 chondrocytes/ml and either 0, 0.1, or 1 μg/ml of rhBMP-2. These rhBMP-2 concentrations were chosen because they were shown to stimulate chondrocyte hyaline cartilage production in the chondrocyte pellet study (Fig 1). The hydrogels were transferred into chondrocyte media, and the cell culture media was changed 3 times per week.27 Constructs were harvested at three and 21 days for biochemical (n=4), histological and immunohistochemical (n=1) analysis.
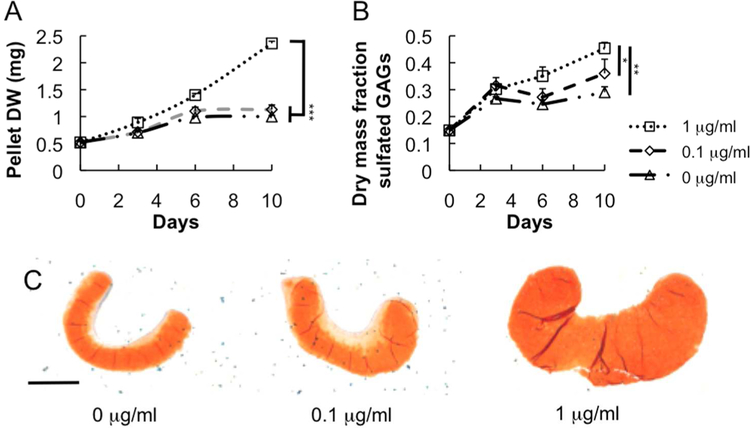
Fig 1.
(A) Dry weight, (B) sulfated GAG mass fraction, and (C) safranin-O staining reveal quantitative and qualitative differences among chondrocyte pellets cultured in various rhBMP-2 conditions. Scale bar = 500 μm. * P < 0.05, ** P < 0.01 *** P < 0.005. For (A) and (B), significant differences refer to time points 6 and 10 days.
In Vivo Chondrogenesis Experiment.
CS-BM hydrogels containing 0 or 1 μg/ml rhBMP-2 were prepared as described in the in vitro methods. Hydrogels were implanted in the dorsal subcutaneous space in nude mice. Animals were anesthetized and maintained with isoflurane. Standard sterile technique was utilized. A 1.5 cm incision was made, and then the fascia was loosened to make a pocket. The hydrogels were placed into the pocket, and the skin was sutured closed. Two hydrogels of each rhBMP-2 condition were implanted into each of four mice, for a total of sixteen implants. Two animals were euthanized and samples harvested at both 3 and 6 weeks post-op. Samples were analyzed for biochemical composition (n=3 for each condition) and histology (n=1 for each condition). Histological samples presented were harvested from a single animal. All procedures were performed with prior approval from the Institute Animal Care and Use Committee.
Biochemical Assays.
Harvested cell pellets and hydrogels designated for biochemical analysis were massed, frozen, and lyophilized. After 48 hours, the samples were massed and then homogenized and solubilized in a papain solution for 16 h at 60°C. DNA content was determined using fluorophotometry with Hoescht 33258 dye (Sigma-Aldrich, St. Louis, MO) and calf- thymus DNA standard. A 1,9-dimethylmethylene blue (DMMB) dye solution was used to quantify sulfated GAG mass per sample. Papain digests and chondroitin sulfate standards (Sigma-Aldrich) were mixed with DMMB and absorbance was read at 525 nm. Total collagen content was measured by performing a hydroxyproline assay. Samples were hydrolyzed in 6N hydrochloric acid at 115°C for 16 hours. Samples were titrated to neutral pH and reacted with p- dimethylamino benzaldehyde and chloramine-T hydrate. Absorbance was measured at 562 nm with a trans-4-hydroxy-L-proline (Sigma-Aldrich) standard.
Histology and Immunohistochemistry.
Cell pellets and hydrogels were fixed in 10% formalin, dehydrated using a series of ethanol solutions, cleared with xylene, paraffin embedded, and sectioned. Samples were stained using Safranin-O, a histological stain that associates with sulfated GAGs indicative of hyaline cartilage production.28 Immunohistochemical staining was performed with a sequence of treatments. The sections were exposed to rabbit polyclonal antibodies against type-I or type-II collagen, biotinylated anti-rabbit secondary antibodies (Invitrogen, Camarillo, CA), and then streptavidin-peroxidase enzyme link. AEC chromagen was applied for 10 minutes to develop the samples before hematoxylin was applied for visualization of cell nuclei.
Matrix Staining Analysis.
ImageJ (National Institutes of Health) was utilized to analyze in vivo type-I and type-II collagen production. Full thickness sections were obtained from each construct. Adjacent sections were stained for type-I or type-II collagen to compare relative production of these matrix components. Four hydrogel regions, each separated by 200 microns, were stained for each matrix marker. Photomicrographs were obtained with constant brightness settings, and regions of interest were selected to encompass the entire scaffold (sectioning artifacts omitted). All images were converted to grayscale, in which pixels have brightness value from 0 (no stain) to 255 (darkest stain). The fraction of maximum intensity is defined as x/255, where x is an arbitrarily chosen intensity threshold. In the threshold-applied images, pixels darker than the threshold value are converted to black pixels, and all others are converted to white. Percent of area stained was calculated as number of black pixels/total pixels for the fractions 0.3, 0.4, 0.5, and 0.6.
Statistical Analysis.
Values are presented as mean and standard deviation. For analysis of biochemistry, statistical analysis was performed using one-way analysis of variance (ANOVA) with SPSS software (version 20; SPSS, Chicago, IL) where appropriate. Post-hoc Tukey tests with an adopted significance level of P = 0.05 was applied to determine statistical significance. For analysis of matrix staining, one-tailed t-tests were used to assess if the presence of rhBMP-2 increased collagen-II levels. P = 0.05 was used to determine statistical significance.
Results
Chondrocyte Pellet Culture.
rhBMP-2 exposure stimulated cartilage ECM production in chondrocyte cell pellets. As demonstrated by weight and gross observation, chondrocyte pellet mass increased upon rhBMP-2 exposure (Fig 1A,C). Additionally, a dose-dependent increase in sulfated GAG mass-fraction (Fig 1B) began at 6 days and was sustained through 10 days. This increase in sulfated GAGs, particularly in the 1 μg/ml treated sample, was confirmed with Safranin-O matrix staining, as evidenced by the more intense red-orange color and positive staining of the entire pellet (Fig 1C).
rhBMP-2 Retention.
Growth factor retention was a function of CS-NHS, BM, rhBMP-2 loading contents and time (Table 2). rhBMP-2 retention at 21 days ranged from 82 to 99 percent. Generally, as determined using the JMP statistical software, increasing CS-NHS and BM content, or decreasing rhBMP-2 loading content increased retention. Additionally, retention decreased for all samples with time, as a result of release of growth factor from the hydrogel. Using the coefficient data from Table 2 and the equation from the materials and methods, the following relation for rhBMP-2 retention as a function of CS, BM, rhBMP-2, and time was generated; rhBMP-2 retention = 0.92 + 0.099*[BM] - 0.0018*[days] + 0.00078*[CS-NHS] - 0.00045* [rhBMP-2]. This relation allows one to calculate the expected residence of growth factor within the hydrogel over time given initial hydrogel conditions. Additionally, the quantity of rhBMP-2 released to the surrounding tissue over time can be determined, an important consideration when designing a delivery system given that rhBMP-2 can have deleterious effects if delivered unintended to some adjacent tissues.
Table 2:
Summary of coefficients for significant factors affecting rhBMP-2 retention
| Factor | Confidence level |
Coefficient |
|---|---|---|
| Intercept | >99.99% | 9.2* 10^−1 |
| BM | >99.99% | 9.9*10^−2 |
| Time | >99.99% | −1.8*10^−3 |
| CS | >99.99% | 7.8* 10^−4 |
| rhBMP-2 | >99.99% | −4.5* 10^−4 |
Characterization of Chondrocytes Encapsulated with rhBMP-2 in CS-BM Hydrogels.
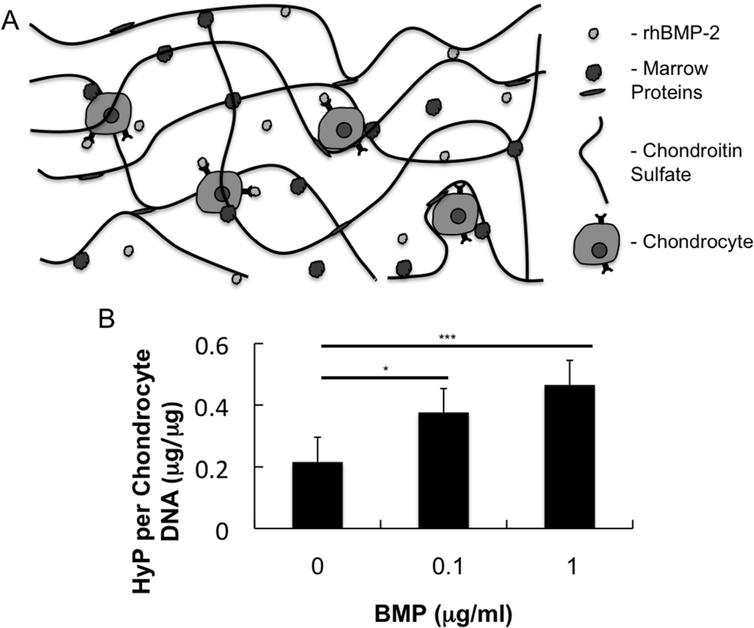
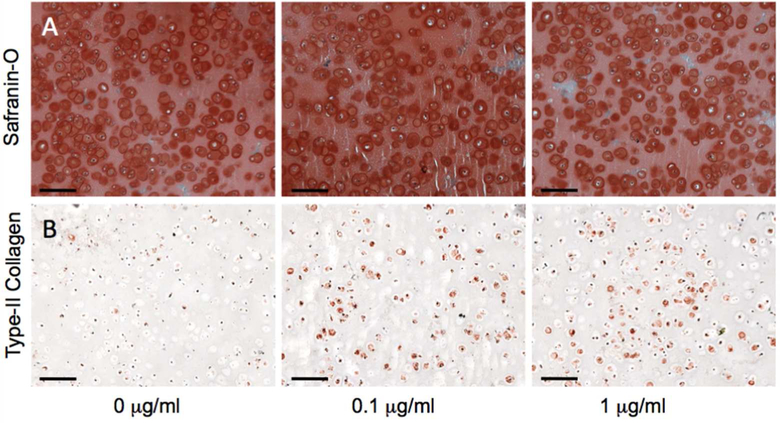
Chondrocytes and rhBMP-2 were encapsulated in biologic adhesive hydrogels composed of CS- NHS and bone marrow. Primary amines located on soluble proteins in the rhBMP-2/bone marrow mixture reacted with activated carboxyl groups on dissolved CS-NHS to cross-link the solution into a hydrogel containing encapsulated chondrocytes (Fig 2A). Over the 3-week duration of culture, the chondrocytes turned over the hydrogel scaffold and produced extracellular cartilage matrix. While there was no measurable accumulation of collagen (as indicated by hydroxyproline content) at the 3-day time point, there was a statistically significant increase in collagen production with respect to increasing rhBMP-2 loading level at three weeks (Fig 2B). Histological analysis of the hydrogels revealed maintained phenotype of encapsulated chondrocytes and rhBMP-2 influence on matrix production (Fig 3). Safranin-O staining of hydrogels of all rhBMP-2 conditions indicated pericellular matrix rich in sulfated GAGs (Fig 3A). Immunohistochemical staining confirmed that type-II collagen was more prevalent in the pericellular matrix surrounding cells in rhBMP-2 treated samples (Fig 3B).
Fig 2.
(A) Schematic of chondrocytes and rhBMP-2 encapsulated within a CS-BM hydrogel. (B) Hydroxyproline normalized to chondrocyte DNA at 21 days of In Vitro hydrogel culture. * P < 0.05, ** P <0.01, *** P < 0.005.
Fig 3.
Histology and immunohistochemistry of chondrocyte encapsulated hydrogels after 21 days of In Vitro culture. (A) Safranin-O and (B) type-II collagen immunohistochemistry were performed to qualitatively assess cell phenotype and hyaline nature of regenerated tissue. Scale bar = 100 μm.
In Vivo rhBMP-2 Chondrogenesis.
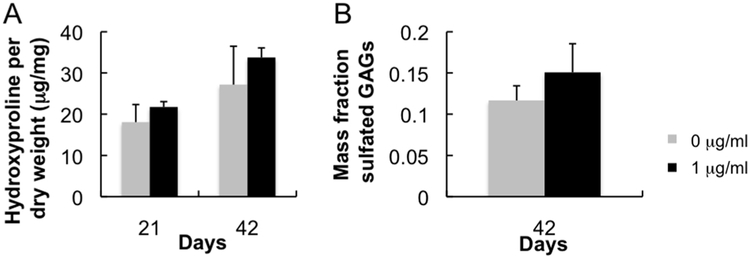
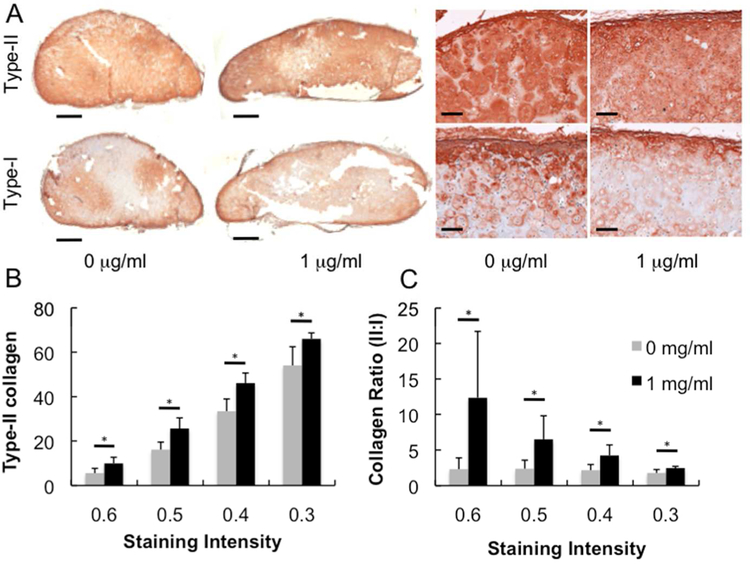
In vivo cartilage production was stimulated by the presence of rhBMP-2. Though not statistically significant, hydroxyproline (Fig 4A) and sulfated GAG (Fig 4B) contents were increased with 1 μg/ml rhBMP-2 compared with control. Immunohistochemical results for type-I and type-II collagen staining indicate strong hyaline cartilage tissue production for constructs with and without rhBMP-2 (Fig 5A). Type-II collagen staining in rhBMP-2 positive sample is more diffuse and evenly distributed with less type-I collagen compared with rhBMP-2 negative control. Closer inspection using brightness threshold analysis indicates (Fig 5B) significant increases in type-II collagen and (Fig 5C) the ratio of type-II to type-I collagen staining density per area at four staining thresholds in the rhBMP-2 loaded sample compared to control. Though total hydroxyproline content did not significantly increase, the presence of rhBMP-2 is stimulating more type-II and less type-I collagen production.
Fig 4.
Biochemistry assays for bovine chondrocytes encapsulated in subcutaneously implanted hydrogels. (A) Hydroxyproline assay was performed at both the three and six-week time points to assess the effect of rhBMP-2 loading on total collagen production. (B) Dimethyl-methylene blue assay was performed at the six- week time point to quantify the effect of rhBMP-2 on sulfated GAG production.
Fig 5.
Immunohistochemistry of subcutaneously implanted CSBM hydrogels containing encapsulated bovine chondrocytes. (A) Low and high magnification images of subcutaneously implanted constructs stained with antibodies against type-I and type-II collagen (scale bars equal 1 mm and 100 μm, respectively). Analysis of the stained neocartilage using a thresholding and fraction of maximum intensity method to assess the effect of rhBMP-2 on (B) the percent of tissue stained positive for type-II collagen and (C) the relative ratio of type-II to type-I collagen. * P < 0.05
Discussion
This study further demonstrates the versatility of the CS-NHS tissue adhesive for application in musculoskeletal tissue repair. Previously, this material was applied to support osteogenic tissue production and fibrocartilage meniscus fusion.21,22 Here, work was performed to assess the chondrogenic capacity and rhBMP-2 carrier efficacy of hydrogel containing 30 mg/ml CS and 70% BM. This material formulation was selected for its superior ability to support cell infiltration and subsequent tissue regeneration compared with other CS-BM formulations.21 The ability to support these cell processes is desired in a material applied to enhance microfracture healing in order to enable chondrocytes at the tissue-hydrogel interface as well as MSCs from the microfracture perforations to infiltrate the material, replace it with regenerated cartilage, and potentially improve integration with the surrounding tissue. In this work, encapsulated articular chondrocytes produced hyaline-like, type-II collagen-rich cartilage within the CS-BM hydrogel. Histological staining with Safranin-O demonstrated that chondrocytes within the material retained their articular phenotype by producing pericellular matrices rich in sulfated GAGs (Fig 3A) and type-II collagen (Fig 3B, Fig 5A). These results indicate that chondroitin sulfate-bone marrow hydrogels are a suitable candidate material to be used as a scaffold in conjunction with cartilage repair techniques.
The combination of avascularity with immobility of chondrocytes due to dense ECM imparts little to no intrinsic healing capacity for cartilage defects, ultimately leading to degenerative joint disease and OA.29 Repairing chondral defects could prevent joint degeneration and eliminate the need for traumatic and invasive total knee replacement surgery. Therefore, there is a market for technologies to enhance articular cartilage repair. Regenerative treatments exist for articular cartilage repair, but are imperfect. In the microfracture procedure, perforations are made in the subchondral bone to form a growth factor and progenitor cell-rich bone marrow clot in the defect space. However, the natural tendency of clots to contract results in an incomplete fill of the defect and poor integration of old and new tissue.30 Additionally, microfracture produces a mechanically inferior fibrocartilage repair that degrades due to wear after a short period.31 It has been shown that combining a polysaccharide biomaterial with the microfracture clot can prevent this clot shrinkage and enhance hyaline quality of microfracture repair in large animals and in humans.12 Our hydrogel system could be combined with the bone marrow clot after microfracture to not only prevent shrinkage and improve mechanics of the blood clot (CS-NHS based hydrogels have moduli greater than 30 kPa compared to reported modulus of 2.2 kPa for a blood clot), but also covalently bind it to the surrounding articular cartilage and subchondral bone.20,32 This strong bond may result in better integration of neocartilage with surrounding tissue.33 And due to the ability of chondroitin sulfate to promote MSC chondrogenesis, the addition of CS-NHS may also support regeneration of hyaline cartilage better than microfracture alone.11 Additionally, the ability to localize therapeutic quantities of rhBMP-2 into the hydrogel implant for an extended duration may enhance the hyaline nature of repair cartilage.
Bone marrow stimulates healing in musculoskeletal tissues in part due to a rich presence of growth factors, but a robust healing response may require growth factor delivery at supra- physiologic doses. Growth factors from the TGF superfamily are an attractive option for enhancing tissue repair due to the role that they play in the development of numerous musculoskeletal tissues.34–37 While TGF-β has been shown to stimulate cartilage repair and is often used in chondrogenic differentiation protocols, it also has been reported to adversely induce fibrosis in treated joints.13,38 These findings lead us to investigate rhBMP-enhanced chondrogenesis. The FDA has approved several of these growth factors, including rhBMP-2 (INFUSE®) and rhBMP-7 (OP-1™ Putty), for bone fusion indications, but rhBMP-2 has the potential to be applied for repair of other musculoskeletal tissues. Consistent with previously reported results, the results of this study showed that rhBMP-2 was capable of stimulating proteoglycan synthesis and ECM production by articular chondrocytes in pellet culture (Fig 1), where fresh rhBMP-2 was supplied within the media at each media change.17 In the hydrogel system where the rhBMP-2 was delivered with a single bolus delivery within the hydrogel, the presence of the growth factor was capable of doubling hydroxyproline production by encapsulated chondrocytes (Fig 2B) and increasing the type-II to type-I collagen ratio (Fig 5C). These results demonstrates that the growth factor was retained within the material and remained active throughout the duration of the experiment, a quality of the system with important implications for the efficacy of CS-BM to be utilized as a carrier for rhBMP-2.
The means of growth factor delivery is critical to success of the intended therapy. Currently, clinical delivery of growth factors is typically accomplished through loading onto a collagen sponge, which is often unsecured or physically entrapped.15,39 These collagen sponges are ineffective at precise control of growth factor retention spatially and temporally, and do little to specifically direct tissue repair. Uncontrolled release of supra-physiological levels of rhBMP-2 have demonstrated adverse effects such as neurological impairment due to ectopic bone formation in the spinal canal when applied for vertebral fusion, suggesting that more sophisticated approaches may be required.40 Carrier technologies such as electrospun fibers and hydrogels are being researched to allow greater control over release/retention and better stimulate repair.41–43 Potential carriers should fulfill certain requirements. First, the carrier should protect the rhBMP-2 and maintain bioactivity while also allowing for spatially and temporally controlled release/retention of the BMP to promote tissue repair.18 Immobilization of the growth factor onto the scaffold can be done through adsorption, entrapment, or covalent binding.44 The characteristic of high retention with limited release over a three-week period from our hydrogel systems suggests that rhBMP-2 is both covalently bound to the CS and physically entrapped within the CS-BM scaffold (Fig 2A). Molecules present in the bone marrow stroma can bind BMP and likely participate in BMP targeting which may aid in the physical retention of the growth factor in this system.45,46 Inclusion of rhBMP-2 enhanced the quantity (Fig 2B, 5C) and quality (Fig 3B, 5) of tissue repair, indicating that the encapsulation conditions did not impair the function of the growth factor. Second, the carrier should be easy to prepare and handle surgically, and should be compatible with established surgical techniques in order to gain widespread acceptance. CS-NHS can be delivered as a powder down a trephine or readily dissolved in saline to form an injectable solution, qualities that impart strong potential compatibility with microfracture procedures. Last, clinically successful carriers should be chemically stable and be able to be produced sterile at commercial scales. Future studies examining stability and sterilization are planned. The novelty of our rhBMP-2 delivery system lies in the multifactorial approach. The adhesive properties of this chondroitin sulfate biomaterial can be leveraged to localize intraoperative biologics and exogenous growth factors to stimulate healing, integration, and enhance quality of musculoskeletal repair tissue contained to the site of interest.
The release kinetics of rhBMP-2 from the hydrogel is an important consideration, and having the ability to control this release/retention is desirable. It has been shown previously and in this study that enhancement of tissue production can be achieved through sustained residence of the growth factor over an extended duration.18 The results of our rhBMP-2 release study indicate that adjusting CS-NHS, BM, and rhBMP-2 loading concentrations can be used to precisely control the retention kinetics of the growth factor. The ability to control growth factor retention may allow for a variety of applications. In applications where retention is preferred, gels with high CS contents could be delivered. It has been demonstrated that physiologically high concentrations of rhBMP-2 are needed in vivo to induce growth factor-stimulated tissue formation in non-human primates.47,48 The high efficiency for rhBMP-2 retention by certain formulations could localize the rhBMP-2 to the area of interest, thereby reducing ectopic tissue formation associated with diffusion of supra-physiological doses away from the delivery site.40 In applications where growth factor elution is preferred, a combination of low CS-NHS with high rhBMP-2 loading level could be delivered. This could be desired in a joint where several tissues could benefit from rhBMP-2 stimulation and the risks associated with ectopic bone formation are minimal. It is important to note that in vivo release profiles may differ from these measured in vitro, and elution of a greater portion of rhBMP-2 may occur in vivo.49 Humans express more than 50 enzymes capable of hydrolyzing linear amide bonds, which could free scaffold-bound rhBMP-2 and result in greater release.50 The retention of rhBMP-2 should be a target for future in vivo experiments investigating the efficacy of CS-BM as a delivery vehicle for rhBMP-2 enhanced tissue repair.
Conclusion
This work presents a novel carrier technology to deliver and localize rhBMP-2, with the ability to temporally and spatially control growth factor retention as a function of CS-NHS content, bone marrow fraction, and rhBMP-2 loading level. It was further demonstrated that the bone marrow hydrogels were capable of supporting articular chondrocyte viability and phenotype retention, with the ability of cells to produce hyaline-like ECM. rhBMP-2 encapsulation within the bone marrow hydrogel resulted in increased quantity and quality of tissue production by articular chondrocytes, indicating that encapsulated growth factor retained its biological function. In sum, this multifunctional material is a candidate vehicle for rhBMP-2 delivery to enhance outcome in musculoskeletal tissue engineering.
Acknowledgments
The authors would like to thank Matthew Gibson for his assistance in teaching surgical techniques for the in vivo studies, Paul Tershakovic for his work in performing the cell culture and histology of the pellet study, and Eric Vanderploeg for his expertise and advising role in planning the in vitro chondrogenesis and rhBMP-2 release experiments. This research was funded in part by Pfizer, Inc., and by the National Institute of Arthritis and Musculoskeletal and Skin Diseases (5R01AR054005–04).
REFERENCES
- (1).Long M Rack HJ Biomaterials 1998, 19, 1621–1639. [DOI] [PubMed] [Google Scholar]
- (2).Kurtz SM; Muratoglu OK; Evans M Edidin AA Biomaterials 1999, 20, 1659–1688. [DOI] [PubMed] [Google Scholar]
- (3).Jeffers JR Walter WL J Bone Joint Surg Br 2012, 94, 735–745. [DOI] [PubMed] [Google Scholar]
- (4).Amstutz HC; Campbell P; Kossovsky N Clarke IC Clin Orthop Relat Res 1992, 7–18. [PubMed] [Google Scholar]
- (5).Nagels J; Stokdijk M Rozing PM J Shoulder Elbow Surg 2003, 12, 35–39. [DOI] [PubMed] [Google Scholar]
- (6).Forsythe ME; Dunbar MJ; Hennigar AW; Sullivan MJ Gross M Pain ResManag 2008, 13, 335–341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (7).Scher DL; Belmont PJ Jr.; Mountcastle S Owens BD Arthritis Rheum 2009, 61, 468–475. [DOI] [PubMed] [Google Scholar]
- (8).Golightly YM; Marshall SW; Callahan LF Guskiewicz K JPhys Act Health 2009, 6, 638–643. [DOI] [PubMed] [Google Scholar]
- (9).Elisseeff J; Anseth K; Sims D; McIntosh W; Randolph M; Yaremchuk M Langer R Plast Reconstr Surg 1999, 104, 1014–1022. [DOI] [PubMed] [Google Scholar]
- (10).Elisseeff JH; Lee A; Kleinman HK Yamada Y Ann N Y Acad Sci 2002, 961, 118–122. [DOI] [PubMed] [Google Scholar]
- (11).Varghese S; Hwang NS; Canver AC; Theprungsirikul P; Lin DW Elisseeff J Matrix Biol 2008, 27, 12–21. [DOI] [PubMed] [Google Scholar]
- (12).Hoemann CD; Hurtig M; Rossomacha E; Sun J; Chevrier A; Shive MS Buschmann MD J Bone Joint Surg Am 2005, 87, 2671–2686. [DOI] [PubMed] [Google Scholar]
- (13).Lee JW; Kim YH; Kim SH; Han SH Hahn SB Yonsei Med J 2004, 45 Suppl, 41–47. [DOI] [PubMed] [Google Scholar]
- (14).Martinek V; Latterman C; Usas A; Abramowitch S; Woo SL; Fu FH Huard J J Bone Joint Surg Am 2002, 84-A, 1123–1131. [DOI] [PubMed] [Google Scholar]
- (15).Rodeo SA; Suzuki K; Deng XH; Wozney J Warren RF Am J Sports Med 1999, 27, 476–488. [DOI] [PubMed] [Google Scholar]
- (16).Khan SN Lane JM Expert Opin Biol Ther 2004, 4, 741–748. [DOI] [PubMed] [Google Scholar]
- (17).van Beuningen HM; Glansbeek HL; van der Kraan PM van den Berg WB Osteoarthritis Cartilage 1998, 6, 306–317. [DOI] [PubMed] [Google Scholar]
- (18).Bessa PC; Casal M Reis RL J Tissue Eng Regen Med 2008, 2, 81–96. [DOI] [PubMed] [Google Scholar]
- (19).Strehin I; Ambrose WM; Schein O; Salahuddin A Elisseeff J J Cataract Refract Surg 2009, 35, 567–576. [DOI] [PubMed] [Google Scholar]
- (20).Strehin I; Nahas Z; Arora K; Nguyen T Elisseeff J Biomaterials 2010, 31, 2788–2797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (21).Simson J; Strehin I; Allen B Elisseeff JH Submitted,
- (22).Simson J; Crist J; Strehin I; Lu Q Elisseeff JH J Orthop Res 2012, [DOI] [PubMed] [Google Scholar]
- (23).Kim TK; Sharma B; Williams CG; Ruffner MA; Malik A; McFarland EG Elisseeff JH Osteoarthritis Cartilage 2003, 11, 653–664. [DOI] [PubMed] [Google Scholar]
- (24).Musumeci G; Loreto C; Carnazza ML; Strehin I Elisseeff J Histol Histopathol 2011, 26, 1265–1278. [DOI] [PubMed] [Google Scholar]
- (25).M Yamamoto YT, Tabata Y Key Engineering Materials 2005, 288.-, 253–256. [Google Scholar]
- (26).Tamai N; Myoui A; Hirao M; Kaito T; Ochi T; Tanaka J; Takaoka K Yoshikawa H Osteoarthritis Cartilage 2005, 13, 405–417. [DOI] [PubMed] [Google Scholar]
- (27).Gunja NJ Athanasiou KA Arthritis Research and Therapy 2007, 9, [DOI] [PMC free article] [PubMed] [Google Scholar]
- (28).Strehin IA Elisseeff JH Methods Mol Biol 2009, 522, 349–362. [DOI] [PubMed] [Google Scholar]
- (29).Davies-Tuck ML; Wluka AE; Wang Y; Teichtahl AJ; Jones G; Ding C Cicuttini FM Osteoarthritis Cartilage 2008, 16, 337–342. [DOI] [PubMed] [Google Scholar]
- (30).Mithoefer K; McAdams T; Williams RJ; Kreuz PC Mandelbaum BR Am J Sports Med 2009, 37, 2053–2063. [DOI] [PubMed] [Google Scholar]
- (31).Kang SW; Bada LP; Kang CS; Lee JS; Kim CH; Park JH Kim BS BiotechnolLett 2008, 30, 435–439. [DOI] [PubMed] [Google Scholar]
- (32).Roy SC; Paulose M Grimes CA Biomaterials 2007, 28, 4667–4672. [DOI] [PubMed] [Google Scholar]
- (33).Wang DA; Varghese S; Sharma B; Strehin I; Fermanian S; Gorham J; Fairbrother DH; Cascio B Elisseeff JH Nat Mater 2007, 6, 385–392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (34).Ramoshebi LN; Matsaba TN; Teare J; Renton L; Patton J Ripamonti U Expert Rev Mol Med 2002, 4, 1–11. [DOI] [PubMed] [Google Scholar]
- (35).Frenkel SR; Saadeh PB; Mehrara BJ; Chin GS; Steinbrech DS; Brent B; Gittes GK Longaker MT Plast Reconstr Surg 2000, 105, 980–990. [DOI] [PubMed] [Google Scholar]
- (36).McPherron AC; Lawler AM Lee SJ Nature 1997, 387, 83–90. [DOI] [PubMed] [Google Scholar]
- (37).Wolfman NM; Hattersley G; Cox K; Celeste AJ; Nelson R; Yamaji N; Dube JL; DiBlasio-Smith E; Nove J; Song JJ; Wozney JM Rosen V J Clin Invest 1997, 100, 321–330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (38).Allen JB; Manthey CL; Hand AR; Ohura K; Ellingsworth L Wahl SM J Exp Med 1990, 171, 231–247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (39).Geiger M; Li RH Friess W Adv DrugDeliv Rev 2003, 55, 1613–1629. [DOI] [PubMed] [Google Scholar]
- (40).Wong DA; Kumar A; Jatana S; Ghiselli G Wong K Spine J 2008, 8, 1011–1018. [DOI] [PubMed] [Google Scholar]
- (41).Li C; Vepari C; Jin HJ; Kim HJ Kaplan DL Biomaterials 2006, 27, 3115–3124. [DOI] [PubMed] [Google Scholar]
- (42).Bulpitt P Aeschlimann D JBiomedMater Res 1999, 47, 152–169. [DOI] [PubMed] [Google Scholar]
- (43).Park Y; Sugimoto M; Watrin A; Chiquet M Hunziker EB Osteoarthritis Cartilage 2005, 13, 527–536. [DOI] [PubMed] [Google Scholar]
- (44).Luginbuehl V; Meinel L; Merkle HP Gander B Eur J Pharm Biopharm 2004, 58, 197–208. [DOI] [PubMed] [Google Scholar]
- (45).Charbonneau NL; Jordan CD; Keene DR; Lee-Arteaga S; Dietz HC; Rifkin DB; Ramirez F Sakai LY J Biol Chem 2010, 285, 20242–20251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (46).Sengle G; Charbonneau NL; Ono RN; Sasaki T; Alvarez J; Keene DR; Bachinger HP Sakai LY J Biol Chem 2008, 283, 13874–13888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (47).Valentin-Opran A; Wozney J; Csimma C; Lilly L Riedel GE Clin Orthop Relat Res 2002, 110–120. [DOI] [PubMed] [Google Scholar]
- (48).Gittens SA Uludag HJ Drug Target 2001, 9, 407–429. [DOI] [PubMed] [Google Scholar]
- (49).Kempen DH; Lu L; Hefferan TE; Creemers LB; Maran A; Classic KL; Dhert WJ Yaszemski MJ Biomaterials 2008, 29, 3245–3252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (50).Scheer M; Grote A; Chang A; Schomburg I; Munaretto C; Rother M; Sohngen C; Stelzer M; Thiele J Schomburg D Nucleic Acids Res 2011, 39, D670–676. [DOI] [PMC free article] [PubMed] [Google Scholar]