Abstract
Multiple ring-enhancing lesions on brain MRI in an immunocompromised patient are frequently associated with opportunistic infections such as toxoplasmosis. In this report, we describe the case of a patient with HIV/AIDS who was found to having multiple ring-enhancing lesions and was diagnosed with a primary central nervous system lymphoma.
Keywords: HIV, Lymphoma, Toxoplasmosis, Antiretroviral therapy, Ring-enhancing lesion
Introduction
Human immunodeficiency virus (HIV) infection results in impaired cellular immunity, which predisposes to development of cancers. Although the overall incidence of NHL in HIV patients has declined with the use of antiretroviral therapy (ART) [2], it is still considerably greater than that of the general population. Primary central nervous system (CNS) lymphoma comprises about 15 % of HIV-related NHL, with the most common subtype being diffuse large B cell lymphoma [5]. Here we present the case of a young female diagnosed with HIV and nonadherent with ART, who presented to the hospital with worsening altered mental status and was found to have an aggressive diffuse large B-cell lymphoma, EBV-related.
Case report
A 29-year-old white female presented to the emergency department with altered mental status, falls, and vomiting that began about 3 days prior. Her past medical history includes intravenous drug use and HIV/AIDS diagnosed in 2014. She is nonadherent with ART. Two months ago, she was admitted to an outside hospital for acute hypoxic respiratory failure and was subsequently diagnosed with Mycobacterium avium complex (MAC) pneumonia and Cytomegalovirus (CMV) pneumonia on bronchoscopy. She was started on azithromycin, ethambutol, and rifabutin at that time and completed a course of intravenous valganciclovir. Since discharge from that hospitalization, she did not follow-up with any doctors and had been having waxing and waning mentation according to her sister. Over the past few days, she had progressively worsening disorientation and focal weakness of the arms, often dropping items from her hands. Additionally, she complained of poor appetite along with multiple episodes of vomiting.
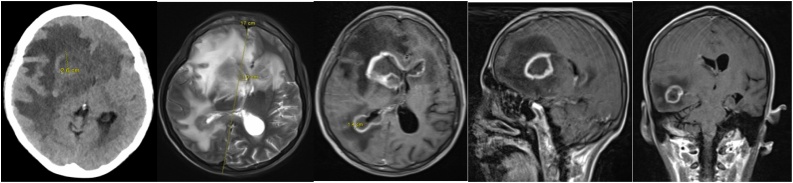
Upon arrival to the emergency department, she was afebrile but tachycardic with a heart rate up to 140 beats per minute. She was noted to have a Glasgow coma scale (GCS) score of 10 and was too lethargic to provide any history. Chest x-ray on admission showed no acute findings. CT head and MRI brain were urgently performed (Fig. 1). CT head showed multiple parenchymal masses clustered in the right cerebral hemisphere, the largest of which was 2.6 cm, as well as extensive vasogenic edema. MRI brain showed multiple peripherally enhancing lesions scattered throughout the cerebral hemispheres, right worse than left, with resultant significant mass effect in the right cerebral hemisphere and associated 16 mm leftward midline shift and suspected right uncal herniation. Given the age of the patient and her HIV status, the differential diagnoses based on the imaging findings were extensive and included abscesses, lymphoma, and opportunistic CNS infections such as Toxoplasma encephalitis. However, given the signal characteristics/findings (i.e. multiple areas of diffusion restriction), there was initially greatest concern for cerebral abscesses with associated right lateral ventriculitis.
Fig. 1.
Left image: CT head showing 2.6 cm parenchymal mass and extensive vasogenic edema. Right 4 images: MRI brain showing multiple peripherally enhancing lesions scattered throughout the cerebral hemispheres, right worse than left, with resultant significant mass effect in the right cerebral hemisphere and associated 16 mm leftward midline shift and suspected right uncal herniation.
Her HIV viral load on admission was 3.4 million copies/mL, with a CD4 count of 3 cells/mm3. She was started on broad-spectrum antibiotics with vancomycin, cefepime, ampicillin, metronidazole, and valganciclovir, in addition to her therapy for MAC. She was also started on dexamethasone given concern for malignancy as well as levetiracetam for seizure prophylaxis. Additionally, she was given TMP-SMX for Toxoplasma gondii and Pneumocystis jiroveci prophylaxis. A craniotomy was performed on hospital day 2. Biopsies of the masses were obtained and a ventricular drain was placed. The pathology resulted as aggressive diffuse large B-cell lymphoma (MUM1+, CD10-, BCL6-), EBV-associated, in the setting of immunosuppression. Immunostaining for MYC was negative.
Cerebrospinal fluid (CSF) analysis and cultures were taken at the time of surgical intervention. CSF studies showed a total nucleated cell count of 6 cells/mm3 (42 % lymphocytes, 22 % neutrophils, 22 % monocytes, 14 % mesothelial cells), glucose 53 mg/dL, and protein 52 mg/dL. CSF cultures (i.e. aerobic, anaerobic, fungal, acid-fast bacilli) returned negative as did CSF cryptococcal antigen. Serum cryptococcal antigen, toxoplasma IgM and IgG antibody, histoplasma antigen, galactomannan antigen, Fungitell assay, and QuantiFERON assay all returned negative as well.
She had a prolonged hospital course which was complicated by Mobitz type 1 heart block for which a temporary pacemaker was placed. She also had episodes of supraventricular tachycardia, requiring an esmolol drip. Additionally, she developed a pneumothorax secondary to central line placement for which a chest tube was placed.
Although the patient had no evidence of disseminated disease or lymphadenopathy on physical exam, a bone marrow biopsy was performed on hospital day 8 to complete staging for diffuse large B-cell lymphoma and showed no evidence of disease in the marrow. ART (abacavir/dolutegravir/lamivudine) therapy was started on hospital day 11 after the infectious workup previously mentioned returned negative. For her MAC infection, she was continued on azithromycin, ethambutol, and rifabutin. She was evaluated by oncology for treatment of her CNS lymphoma. However, after further conversations with her sister and decision maker, it was determined that chemotherapy would be too risky given her poor clinical status, and the decision was made to transition to hospice and comfort care.
Discussion
Human immunodeficiency virus (HIV)-related primary central nervous system lymphoma (PCNSL) is an acquired immune deficiency syndrome (AIDS)-defining malignancy that is strongly linked to Epstein-Barr virus (EBV) infection and immunosuppression [7]. HIV-related PCNSL can present with a wide array of focal or generalized signs and symptoms including confusion, lethargy, memory loss, hemiparesis, aphasia, and seizures [1]. Our patient was noncompliant with ART, increasing her risk of HIV-related illnesses, and presented with confusion, focal weakness, and inability to tolerated oral intake.
HIV-related PCNSL is the second leading cause of intracranial lesions in patients with HIV after toxoplasmosis, with a prevalence of 12.3 %. Furthermore, the 5-year survival rate without treatment is 22 % [4]. When compared to immunocompetent counterparts, PCNSL related to AIDS may present with numerous brain lesions rather than solitary lesions, albeit smaller in size [4]. Lesions tend to be supratentorial and favor the cerebral hemispheres and basal ganglia in location, and ependymal seeding and meningeal involvement can be seen with periventricular lesions [4,12]. Additionally, HIV-related lymphoma is particularly more aggressive and rapidly growing than the lesions seen in immunocompetent hosts. The breakdown of the blood-brain barrier leads to ring enhancement, and the rapid growth and ensuing necrosis contributes to irregular and non-homogeneous enhancement [12]. This is in contrast to non-HIV-related PCNSL, which is usually homogeneously enhancing [8]. Other radiographic findings can be variable and may display atypical features in the setting of AIDS. These include hyperintense lesions on diffusion-weighted MRI, and hypointense lesions on apparent diffusion coefficient (ADC) maps due to increased cellularity [4,12].
In patients with a low CD4 cell count (<200 cells/mm3), the main alternative causes of an enhancing mass lesion or lesions with surrounding edema are toxoplasmosis, progressive multifocal leukoencephalopathy (PML), CMV encephalitis, tuberculosis, and other bacterial or fungal abscesses [3]. While often thought that multiple ring enhancing lesions on brain MRI in a HIV patient favor the diagnosis of toxoplasmosis, PCNSL lesions may be solitary or multifocal, in roughly equal proportions [9]. Although solitary lesions are much more likely to favor a diagnosis of PCNSL, the presence of multiple lesions, as seen in this case, should not exclude the diagnosis.
Additionally, toxoplasmosis cannot be easily ruled out with plasma serology, as this can often be inaccurate in immunocompromised individuals [11]. Given its similar presentation and imaging findings, this poses yet another challenge to accurately differentiating between PCNSL and toxoplasmosis. Nonetheless, the diagnosis of PCNSL in a HIV patient should be made by stereotactic biopsy or cerebrospinal fluid (CSF) flow cytometry in the majority of patients. Treatment usually consists of tumor-directed therapy (i.e. methotrexate), in addition to concurrent treatment of HIV with ART [6,10].
In summation, this case demonstrates the importance of accurately diagnosing and managing the extensive intracranial complications of HIV. It highlights why clinicians should not necessarily favor the diagnosis of an opportunistic infection (i.e. toxoplasmosis) even in patients with multiple ring-enhancing brain lesions, as PCNSL lesions are equally likely to be solitary or multifocal. Thus, clinical suspicion for PCNSL should remain high in these situations and early biopsy should be sought to ensure definitive diagnosis.
Ethical approval
None.
Consent
Verbal consent was obtained from the patient’s caregiver/guardian during hospitalization. Written consent was not able to be obtained from the patient or guardian as the patient transitioned to hospice following admission and was lost to follow-up. However, no identifiable information is presented in this case report, and we believe the benefit of publication towards society greatly outweighs any potential harm to the patient.
Author contribution
1.) Nishant Patel (corresponding author) – Authored the majority of the manuscript, including abstract, introduction, hospital course, and discussion sections, as wells ascollected the images included in the report.
2) Rishi Charate (secondary author) – Reviewed entirety of case report and offered edits and additions to the manuscript.
Author statement
Nishant Patel: Conceptualization, Data Curation, Writing - original draft, Writing - review & editing, Visualization, Project administration.
Rishi Charate: Conceptualization, Resources, Writing - review & editing.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of Competing Interest
The authors report no declarations of interest.
References
- 1.Baumgartner J.E., Rachlin J.R., Beckstead J.H., Meeker T.C., Levy R.M., Wara W.M. Primary central nervous system lymphomas: natural history and response to radiation therapy in 55 patients with acquired immunodeficiency syndrome. J Neurosurg. 1990;73(August (2)):206–211. doi: 10.3171/jns.1990.73.2.0206. PMID: 2366078. [DOI] [PubMed] [Google Scholar]
- 2.Besson C. Changes in AIDS-related lymphoma since the era of highly active antiretroviral therapy. Blood. 2001;98(October (8)):2339–2344. doi: 10.1182/blood.v98.8.2339. PMID: 11588028. [DOI] [PubMed] [Google Scholar]
- 3.Blumenthal D.T., Raizer J.J., Rosenblum M.K., Bilsky M.H., Hariharan S., Abrey L.E. Primary intracranial neoplasms in patients with HIV. Neurology. 1999;52(May (8)):1648–1651. doi: 10.1212/wnl.52.8.1648. PMID: 10331693. [DOI] [PubMed] [Google Scholar]
- 4.Chirico C., Izzo I., Casari S., Cattaneo C., Doglietto F., Castelli F. AIDS-associated central nervous system lymphoma: the great mime. A case report and literature review. IDTM. 2018;4(June (2)):e472. [Google Scholar]
- 5.Coté T.R., Biggar R.J., Rosenberg P.S., Devesa S.S., Percy C., Yellin F.J. Non-Hodgkin’s lymphoma among people with AIDS: incidence, presentation and public health burden. AIDS/Cancer Study Group. Int J Cancer. 1997;73(November (5)):645–650. doi: 10.1002/(sici)1097-0215(19971127)73:5<645::aid-ijc6>3.0.co;2-x. doi: 10.1002/(sici)1097-0215(19971127)73:5<645::aid-ijc6>3.0.co;2-x. PMID: 9398040. [DOI] [PubMed] [Google Scholar]
- 6.Ferreri A.J., Reni M., Villa E. Therapeutic management of primary central nervous system lymphoma: lessons from prospective trials. Ann Oncol. 2000;11(August (8)):927–937. doi: 10.1023/a:1008376412784. PMID: 11038028. [DOI] [PubMed] [Google Scholar]
- 7.et al Gasser O., Swiss H.I.V. Cohort Study. HIV patients developing primary CNS lymphoma lack EBV-specific CD4+ T cell function irrespective of absolute CD4+ T cell counts. PLoS Med. 2007;4(March (3)):e96. doi: 10.1371/journal.pmed.0040096. PMID: 17388662; PMCID: PMC1831733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jenkins C.N., Colquhoun I.R. Characterization of primary intracranial lymphoma by computed tomography: an analysis of 36 cases and a review of the literature with particular reference to calcification haemorrhage and cyst formation. Clin Radiol. 1998;53(June (6)):428–434. doi: 10.1016/s0009-9260(98)80271-7. PMID: 9651058. [DOI] [PubMed] [Google Scholar]
- 9.Johnson B.A., Fram E.K., Johnson P.C., Jacobowitz R. The variable MR appearance of primary lymphoma of the central nervous system: comparison with histopathologic features. AJNR Am J Neuroradiol. 1997;18(March (3)):563–572. PMID: 9090424. [PMC free article] [PubMed] [Google Scholar]
- 10.Modi M., Mochan A., Modi G. Management of HIV-associated focal brain lesions in developing countries. QJM. 2004;97(July (7)):413–421. doi: 10.1093/qjmed/hch080. PMID: 15208429. [DOI] [PubMed] [Google Scholar]
- 11.Porter S.B., Sande M.A. Toxoplasmosis of the central nervous system in the acquired immunodeficiency syndrome. N Engl J Med. 1992;327(December (23)):1643–1648. doi: 10.1056/NEJM199212033272306. PMID: 1359410. [DOI] [PubMed] [Google Scholar]
- 12.Thurnher M.M., Thurnher S.A., Schindler E. CNS involvement in AIDS: spectrum of CT and MR findings. Eur Radiol. 1997;7(7):1091–1097. doi: 10.1007/s003300050260. PMID: 9265682. [DOI] [PubMed] [Google Scholar]