Abstract
Evaluation of the performance of a new set of primers defined from the ORF1ab sequence, and its combination with a previously published set of primers from the N sequence, to detect SARS-CoV-2 RNA by the loop-mediated isothermal amplification technique is presented. The ORF1ab primer set enables visual detection of SARS-CoV-2 RNA in 16 min. In addition, a simultaneous reaction with both ORF1ab and N primers allows for higher sensitivity of detection, particularly when low numbers of copies are present (250 viral RNA copies). Further, the protocol is able to detect viral RNA in saliva samples. The procedure reported could be easily implemented in the generation of a new and sensitive rapid point-of care device for SARS-CoV-2 RNA visual detection.
Keywords: : COVID-19, nucleocapsid primers, ORF1ab primers, SARS-CoV-2 detection, virus LAMP amplification
Graphical abstract
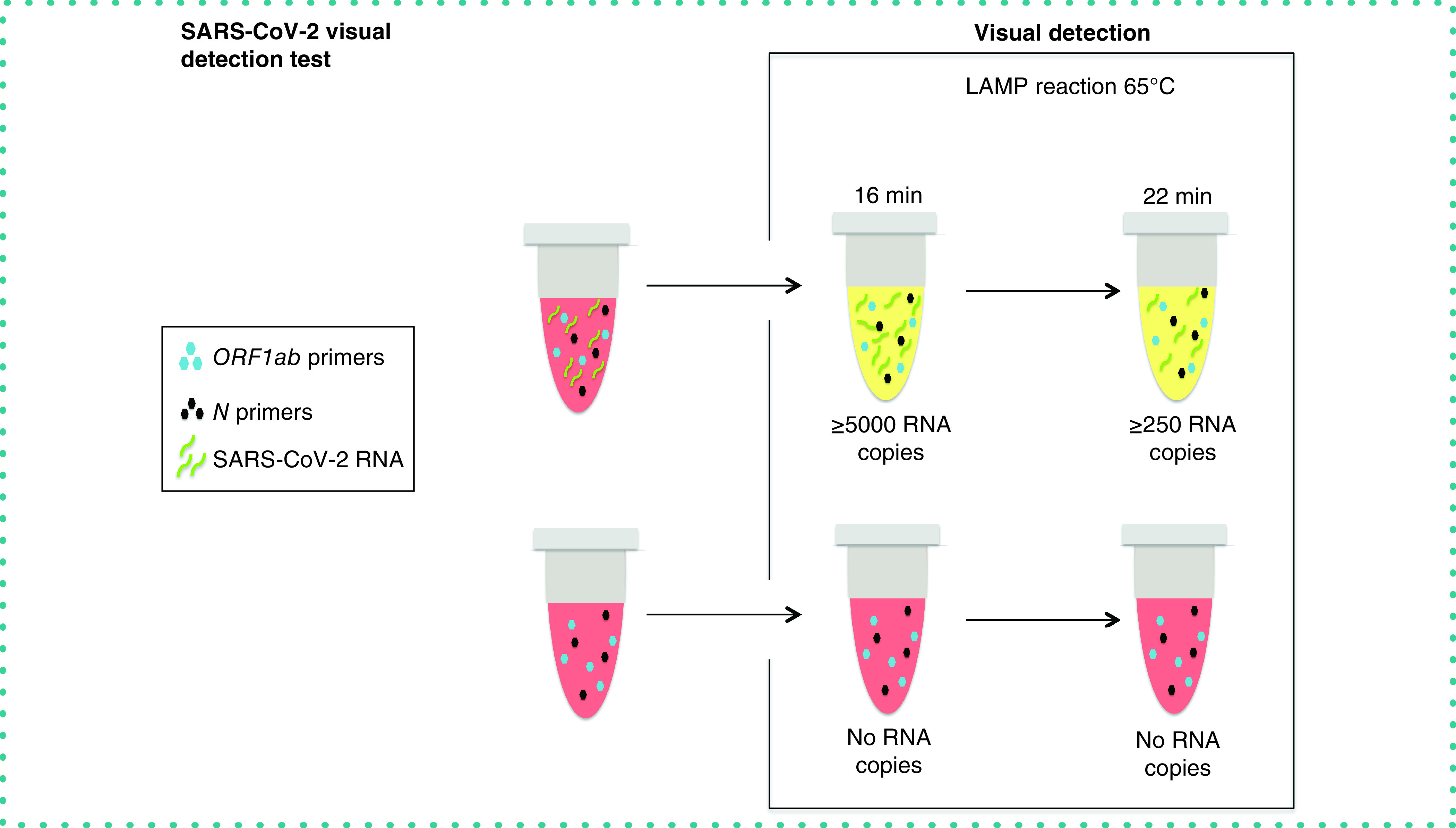
METHOD SUMMARY
Optimized loop-mediated isothermal amplification reactions were carried out using the combination of two sets of primers (designed on ORF1ab and N sequences). Saliva samples were pretreated by heating and proteinase K, before SARS-CoV-2 determination by loop-mediated isothermal amplification.
In late 2019, Coronavirus disease 2019 (COVID-19) emerged as a new acute infection of the respiratory tract [1]. COVID-19 propagation has grown exponentially worldwide and the WHO declared a global pandemic on 11 March 2020 [2]. Fast, accurate testing is necessary in the fight to slow down and reduce the high spread and impact of the causative agent of COVID-19, SARS-CoV-2. Better control of COVID-19 seems to be dependent on the identification of infected individuals as fast as possible, enabling their isolation and follow-up (and quarantine) of the people with whom they were in contact [3].
To date, different methods for COVID-19 detection are available, often clustered depending on the detected material: nucleic acid, antigen (viral protein), or host antibodies [4]. The most rapid tests are based on the detection of protein antigens or host antibodies, which are highly suitable for point-of-care diagnostics. However, the main problem with these tests is their low sensitivity and, in the case of antibody tests, the fact that they detect advanced or late stages of the disease; so these tests are not useful for early detection [4]. Hence the cornerstone for early diagnosis of COVID-19 involves the detection of viral RNA with sensitive nucleic acid amplification tests (NAAT) [4]. The most widely used NAAT for SARS-CoV-2 amplification is RT-qPCR. However, its implementation requires qualified personnel and technically complex laboratory instruments (such as PCR-related thermocyclers), it takes time (hours or days) and it would be difficult to use in a point-of-care setting [5,6]. For these reasons, the development of new and rapid testing strategies is of the utmost importance to counteract this pandemic.
Loop-mediated isothermal amplification (LAMP) is an isothermal NAAT that can detect the presence of viral RNA in biological samples by amplification of selected regions of its genetic material. The reaction has high specificity and sensitivity, associated with exponential amplification of a set of six different target sequences simultaneously [7]. Furthermore, LAMP is an attractive option to be used as a point-of-care test for SAR-CoV-2 detection [5], as it can be performed in an easily handled, portable thermocycler which is easily manageable by non-expert users. Several studies have demonstrated its usefulness in SARS-CoV-2 diagnosis [6,8–16]. Using fluorescent labeling in the RNA amplification, LAMP may enable the detection of SARS-CoV-2 at a very low concentration (from one SARS-CoV-2 RNA copy) [16] and in a short time [6,9,13,17]. Moreover, this methodology is compatible with simple visual colorimetric detection procedures and with relatively unpurified samples [9,16,18,19]. Published data show that the usual time required for SARS-CoV-2 detection is around 20–30 min, which needs to be added to the viral RNA extraction time [9,16,18]. Only sparse studies have achieved a reduction of the required time, although results can be obtained in around 10 min by using colorimetric LAMP [19] or combining different sets of primers [9].
Here, we tested the use of a newly designed set of six primers of the ORF1ab sequence, as well as their combination with a previously published set of primers of the N sequence, to detect SARS-CoV-2 RNA with an optimized colorimetric LAMP protocol. Data show that the protocol is able to detect a useful range of viral RNA copy numbers (250–10,000) in a short time (16–22 min, depending on the required sensitivity), making it easily applicable in a point-of-care setting for different samples, including saliva.
Materials & methods
Synthetic SARS-CoV-2 RNA
For all determinations Twist Synthetic SARS-CoV-2 RNA Control 2 (MN908947.3) was used (Twist Bioscience, CA, USA).
Saliva treatment
One saliva sample was collected, after at least 1 h without drink and food, in nuclease-free water (VWR, Llinars del Vallès, Spain) and proteinase K (Roche, Barcelona, Spain) solution. The sample was incubated for 15 min at room temperature and then for 5 min at 95°C in order to inactivate the proteinase K.
LAMP primers
LAMP primers were designed and prioritized on the ORF1ab sequence, using LAMP Primer Explorer v5, following the manual indications [20]. N primer set, from the nucleocapsid protein sequence, was obtained from the work of Zhang et al. [9]. Primer sets include an outer forward primer (F3), outer backward primer (B3), forward inner primer (FIP), backward inner primer (BIP), loop forward primer (LF) and loop backward primer (LB). LAMP primers for N and Orf1ab coding regions, described in Table 1, were purchased from Sigma Genosys (Haverhill, UK).
Table 1. Primers for SARS-CoV-2 detection.
| Primer | Sequence (5′ to 3′) |
|---|---|
| N | |
| F3 | TGGCTACTACCGAAGAGCT |
| B3 | TGCAGCATTGTTAGCAGGAT |
| FIP (Fic + F2) | TCTGGCCCAGTTCCTAGGTAGTCCAGACGAATTCGTGGTGG |
| BIP (B1c + B2) | AGACGGCATCATATGGGTTGCACGGGTGCCAATGTGATCT |
| LF | GGACTGAGATCTTTCATTTTACCGT |
| LB | ACTGAGGGAGCCTTGAATACA |
| ORF1ab | |
| F3 | CCTCAACTTGAACAGCCCT |
| B3 | CGAAGAAGAACCTTGCGGT |
| FIP (Fic + F2) | GCCTTCGAGTTCTGCTACCAGCTCATCAAACGTTCGGATGCT |
| BIP (B1c + B2) | ACGGTCGTAGTGGTGAGACACTAAGCCACTGGTATTTCGCC |
| LF | ACATGACCATGAGGTGCAGTTCG |
| LB | TGGTGTCCTTGTCCCTCATGTG |
B3: Outer backward primer; BIP: Backward inner primer; F3: Outer forward primer; FIP: Forward inner primer; LB: Loop backward primer; LF: Loop forward primer.
Master mix
LAMP reactions were performed using WarmStart® Colorimetric LAMP 2X Master Mix (New England Biolabs Ltd, Hitchin, UK).
Tested enhancer compounds
Potential enhancer compounds were tested: glycerol ([Gly] 0.87 μM), guanidine hydrochloride ([GHC] 40 mM), bovine serum albumin ([BSA] 0.24 μM; all Sigma, Madrid, Spain), MgCl2 (0.2 and 1mM) and dNTPs (0.5, 1 and 1.5 mM; Thermo Fisher Scientific, Madrid Spain).
Experimental design
In order to determine the sensitivity of the two sets of primers, alone or in combination, with an improved LAMP protocol, different LAMP reactions were performed using the Twist SARS-CoV-2 RNA Control. In a first step, different enhancer compounds of the LAMP reaction were tested (Gly, GHC, BSA, MgCl2 and dNTPs). In a second step, GHC was used as an enhancer compound (having revealed the best improvement), and the sensitivities of N primers, ORF1ab primers and the combination of both primer sets were tested. The sensitivity was calculated considering all detected positive wells versus all true positives plus the false negative wells, multiplied by 100. Finally, in order to test the feasibility of applying the LAMP procedure to saliva samples, LAMP reactions using 1 × 104 copies of SARS-CoV-2 RNA control were executed, adding 5.5 μl of pretreated saliva sample.
LAMP protocol
All LAMP reactions were prepared in an enclosed room using designated pipettes and filter tips in order to prevent contamination.
LAMP reactions were executed in a total volume of 25 μl. Each reaction was performed with 12.5 μl of WarmStart Colorimetric LAMP 2X Master Mix. This Master Mix is an optimized formulation of Bst 2.0 WarmStart DNA Polymerase and WarmStart RTx, in a solution containing a visible pH indicator (phenol red) for rapid and easy LAMP detection [21]. The reaction also contained 1 μl primer mix (1.5 μl was also tested), which was made with: 40μM of FIP and BIP primers, 5 μM of F3 and B3 primers and 10 μM of LB and LF primers; different amounts of fragments of SARS-CoV-2 RNA; 5 μl of GHC (final concentration 40 mM); and the final volume was made up to 25 μl with nuclease-free water. Other enhancer compounds were also tested, but not included in the final protocol. Reactions were set up on ice, then incubated at a constant temperature of 65°C (63°C was also tested) for up to 30 min (14–30 min were visually tested) in an Applied Biosystems 2720 thermal cycler (Applied Biosystems, Madrid, Spain). To end the reaction, a rapid cooling step was performed by immersing the tubes in iced water. When storing in a freezer, the colors remain stable. At the final reaction time point, the absorbance at 440 (yellow) and 560 (red) nm was determined.
Results & discussion
Rapid diagnosis of active SARS-CoV-2 infection is the most effective method to reduce the spread of COVID-19 [3]. Particularly, NAAT tests have a relevant interest due to their sensitivity, and because they are able to detect the infection at the first stages of its evolution [4]. The LAMP technique is a prominent solution to combine NAAT with fast test detection [5]. In the present study, we evaluated the performance and usefulness of a set of primers for the ORF1ab sequence, as well as their combination with a previously published set of primers of the N sequence [9] to detect SARS-CoV-2 RNA. Our data showed that the ORF1ab primers provided rapid, visual virus detection in only 16 min. In addition, a simultaneous reaction with both ORF1ab and N primer sets increased detection sensitivity, particularly when a low number of copies of SARS-CoV-2 sequences were present. Moreover, the results showed that application of the LAMP technique with these two sets of primers under the conditions tested is able to detect synthetic viral RNA spiked in saliva samples.
Conditions of SARS-COV-2 detection by LAMP using two sets of primers alone or in combination
Firstly, in order to optimize the LAMP protocol for SARS-CoV-2 detection (using 5 × 103 copies of RNA), compounds commonly used to enhance sensitivity of the LAMP technique were tested with the ORF1ab primer set: 0.87 μM Gly [22], 40mM GHC [9], 0.24μM BSA [22] and the addition of different concentrations of MgCl2 (0.2 and 1 mM) and dNTPs (0.5, 1 and 1.5 mM) [23]. A consistent effect of GHC improving the reaction speed was observed (Supplementary Figure 1). A minor impact of Gly, MgCl2 and dNTPs concentration was also observed. BSA alone or in combination with GHC did not produce a significant improvement in detection time (Supplementary Figure 1). In addition, the LAMP reaction temperature (65 and 63°C) and concentration of primer sets (1 and 1.5 μl of primer mix) were tested, and the results indicated that the LAMP reaction at 65°C and with 1 μl of the primer mix constituted the best option (Supplementary Figure 2). Thus 40mM GHC, 1 μl of the primer mix and a LAMP reaction at a constant temperature of 65°C was used subsequently.
The next step was assessing the performance of the primer sets at different viral loads. The results showed that the ORF1ab primer set enabled the detection of SARS-CoV-2 RNA in just 16 min, irrespective of the presence of the N set. Detection sensitivity at 16 min was 100% for 1 × 104 RNA copies of SARS-CoV-2, and 86% for 5 × 103 RNA copies using the ORF1ab or the mix of ORF1ab and N primer sets (Figure 1A). However, sensitivity diminished at lower RNA concentrations, with ORF1ab primers allowing for detection of only 29 or 14% of replicates (seven replicates) containing 1000 and 500 RNA copies, respectively (Figure 1A). A LAMP reaction of 22 min resulted in increased sensitivity using any set of primers, but particularly with the mix and at very low RNA copy number (Figure 1B). In fact, 100% of replicates were detected either using individual sets of primers or their combination (seven replicates) when 5 × 103 RNA copies of synthetic SARS-CoV-2 or higher were present (Figure 1B). Interestingly, the combination of both primer sets in the same well managed to detect 250 RNA copies of SARS-CoV-2, although with a sensitivity of 43% (Figure 1B).
Figure 1. SARS-CoV-2 visual detection.
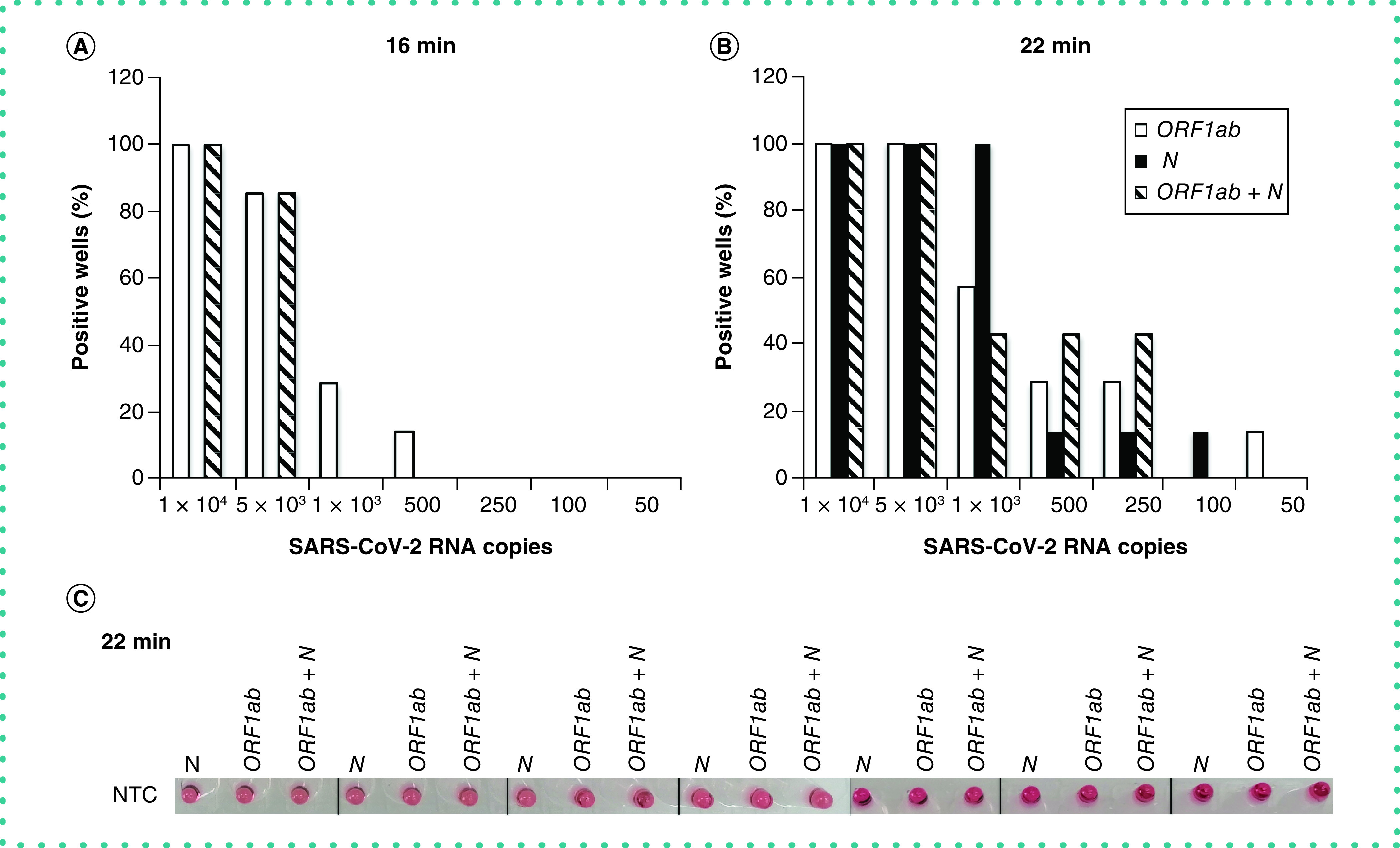
(A & B) Percentage of visually positive replicates (seven replicates) for each primer set (ORF1ab and N) and their combination (ORF1ab + N) after (A) 16 or (B) 22 min of LAMP reaction with different amounts of synthetic SARS-CoV-2 RNA (10,000, 5000, 1000, 500, 250, 100 and 50 copies). (C) Color shown in the test tube of nontemplate control for each primer set (ORF1ab and N) and their combination (ORF1ab + N) after 22 min of LAMP reaction (seven replicates).
It is noteworthy that virus-free samples (nontemplate control [NTC] wells) remained unchanged in color (red) throughout the 22 min of LAMP reaction (Figure 1C). This is of particular interest because a longer LAMP reaction (30 min) turned orange in some wells incubated with ORF1ab primers (either alone or in combination with N primers), which could mask visual detection at this time point (Figure 2D). Thus the final recommended incubation time was up to 22 min, because at 30 min the test could give false positives.
Figure 2. Spectroscopy and visual positive SARS-CoV-2 detection.

Ratio of absorbance at 440/560 nm ratio (mean ± SEM, seven replicates), visual detection and positive well ratio (X/7) after 30 min of LAMP reaction with different amounts of synthetic SARS-CoV-2 RNA (10,000, 5000, 1000, 500, 250, 100 and 50 copies) for each set of primers. (A) ORF1ab primers. (B) N primers. (C) Combination of both primer sets (ORF1ab + N). (D) Ratio of absorbance at 440/560 nm ratio (mean ± SEM, seven replicates), visual detection and the maximum detected ratio (440/560 nm) after 30 min of LAMP reaction of nontemplate control. Samples with a ratio <1.5 for the ORF1ab and ORF1ab + N primers sets, and <1.3 for the N primer set, were excluded and considered negative for the positive well ratio. The assay was conducted in 96-well PCR plates and the image has been taken from the bottom.
LAMP: Loop-mediated isothermal amplification; SEM: Standard error of the mean.
To circumvent incorrect readings at 30 min, visual detection could be complemented with absorbance determination by spectroscopy. This would also address the issue of personal variation in the ability to distinguish colors between borderline positive samples, and could be easily implemented in a point-of-care device with a filter or a more complex readout. The ratio at 440/560 nm reflects the relation between the yellow (positive) and orange-pink (negative). Then the sample value could be corrected by the NTC absorbance ratio. In this sense, we have considered as positive the samples that gave a ratio higher than the NTC (≥1.5 for the ORF1ab and ORF1ab + N primer sets, and ≥1.3 for the N primer set). In this case, the limits of detection and sensitivity for ORF1ab and the mix of both primer sets at 30 min, determined by their absorbance ratios, were similar to the results visually detected at 22 min (Figure 2A–C).
Therefore this work shows that SARS-CoV-2 RNA detection is feasible with the application of a LAMP assay and that this method allows for visual identification of positive samples. We propose to perform the assay using the combination of the two sets of primers tested, enabling the determination of a sample, in duplicate in the same test tube, by visual identification. Using the present protocol, the ORF1ab primers would provide rapid detection (in 16 min) followed by the reaction of the N primers (up to 22 min). Additionally, the use of both primer sets together and in the same test tube is able to provide increased sensitivity, particularly at very low RNA concentrations.
LAMP reaction in saliva
Although the WHO has recommended nasopharyngeal specimens for SARS-CoV-2 detection, their obtainment requires trained healthcare workers and the procedure is relatively invasive for subjects [24]. Saliva samples could represent very suitable biological material for COVID-19 diagnosis [25]. Thus we preliminarily tested the feasibility of applying the LAMP procedure, to obtain this first information with these sets of primers, to saliva samples spiked with SARS-Cov-2 RNA. In principle, saliva samples could contain some inhibitors that could make it difficult to detect the virus and this would require RNA isolation before any NAAT. Here we assessed whether the saliva components of a healthy volunteer influenced SARS-CoV-2 RNA detection by LAMP. Our results showed that although visual detection of synthetic SARS-CoV-2 RNA (1 × 104 copies) was delayed in the tubes containing saliva, RNA could be detected, albeit at a longer time than in non-saliva test tubes, regardless of the primer set used. As previously shown, 100% of replicates for ORF1ab acquired the yellow/orange color after 16 min of LAMP reaction, whereas in the replicates with saliva, the change of color appeared after 22 min (Figure 3). In the same way, for the N primer set, 100% of replicates were visually positive at 22 min of LAMP reaction, but only 80% of replicates with saliva were positive after 30 min (Figure 3). Therefore, the results show that SARS-CoV-2 detection is possible from a saliva specimen without performing RNA isolation and increasing the reaction time of the current protocol. The increased time required to detect the presence of SARS-CoV-2 RNA could be associated with the presence of proteins, particularly RNases, present in saliva samples [26] and remaining after the treatment with proteinase K or the thermal protocol required for proteinase K inactivation, which could cause RNA degradation [27].
Figure 3. SARS-CoV-2 visual detection in saliva samples.
Visual detection at 16, 22 and 30 min of LAMP reaction with 1 × 104 copies of synthetic SARS-CoV-2 RNA (five replicates) or nontemplate control (one replicate), for each set of N and ORF1ab primers, with or without a pretreated saliva sample. The assay was conducted in eight-well PCR strips and the image has been taken from the front of the strips.
At the final point of the LAMP reaction, the ratio of absorbance at 440/560 nm of positive wells was lower in the replicates with saliva than in the replicates without saliva (for ORF1ab primer set, 1.50 ± 0.02 vs 1.74 ± 0.02 and for N primer set, 1.34 ± 0.04 vs 1.59 ± 0.06; saliva and non-saliva samples, respectively). Moreover, saliva did not alter the NTC wells, which showed an absorbance ratio <1.1. Therefore the results show how these primer sets could be used for SARS-CoV-2 detection in saliva samples by LAMP, without a previous RNA isolation or purification step. Nevertheless, more studies with a wider number and variety of saliva samples from COVID-19 patients are needed to confirm the utility of these two primer sets in human rapid detection and its feasibility to be implemented at point-of-care.
Conclusion
Our results demonstrate that the LAMP technique applied to the tested set of ORF1ab primers is able to detect a relatively low number of copies of SARS-CoV-2 RNA within only 16 min of reaction and allows for visual diagnosis of the presence of COVID-19 active infection. Beyond that, the combination of this primer set with a predefined primer set of the SARS-CoV-2 N region [9] in the same reaction tube does not produce interference and increases detection sensitivity, particularly with a lower number of viral RNA copies. Moreover, it is of interest that the combination of both primer sets would reduce the cost and handling of testing samples in duplicate and represent a double check for detection of the disease; even if one of these primer sets does not generate color, the other can generate color and reveal a positive sample. Therefore the use of primer sets designed from two different viral target regions (ORF1ab and N) seems to be a good strategy to generate a new and highly sensitive rapid detection test by the user-friendly LAMP technique. In addition, preliminary results suggest the utility of these primer sets directly with saliva samples, which can contribute to reducing the time taken for diagnosis as well as the use of material, technical equipment and human health resources in the fight against COVID-19. The procedure reported would be easily scalable in a point-of-care setting and could be implemented in the generation of a new and sensitive rapid point-of care device for SARS-CoV-2 RNA visual detection.
Supplementary Material
Supplementary data
To view the supplementary data that accompany this paper please visit the journal website at: www.future-science.com/doi/suppl/10.2144/btn-2020-0159
Author contributions
A Palou and F Serra proposed and supervised the work. B Reynés carried out the experimental procedures. All authors participated in the data analysis and interpretation. All the authors wrote the manuscript, participated in its critical revision and read and approved the final manuscript.
Financial & competing interests disclosure
The authors received funding from Universitat de les Illes Balears and Alimentómica SL. B Reynés is supported by Torres Quevedo Contract Aids (PTQ-17-09044) from the Spanish Government. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations.
Open access
This work is licensed under the Attribution-NonCommercial-NoDerivatives 4.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/4.0/
References
Papers of special note have been highlighted as: • of interest
- 1.Zhu N, Zhang D, Wang W et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 382(8), 727–733 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Huang X, Wei F, Hu L, Wen L, Chen K. Epidemiology and clinical characteristics of COVID-19. Arch. Iran Med. 23(4), 268–271 (2020). [DOI] [PubMed] [Google Scholar]
- 3.Güner R, Hasanoğlu I, Aktaş F. COVID-19: prevention and control measures in community. Turk. J. Med. Sci. 50(SI-1), 571–577 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.La Marca A, Capuzzo M, Paglia T, Roli L, Trenti T, Nelson SM. Testing for SARS-CoV-2 (COVID-19): a systematic review and clinical guide to molecular and serological in vitro diagnostic assays. Reprod. Biomed. Online 41(3), 483–499 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nguyen T, Duong Bang D, Wolff A. 2019 novel coronavirus disease (COVID-19): paving the road for rapid detection and point-of-care diagnostics. Micromachines (Basel) 11(3), 306 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lamb LE, Bartolone SN, Ward E, Chancellor MB. Rapid detection of novel coronavirus/Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) by reverse transcription-loop-mediated isothermal amplification. PLoS ONE 15(6), e0234682 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]; • Development of one of the first rapid (30–45 min) SARS-CoV-2 detection tests using LAMP methodology.
- 7.Kashir J, Yaqinuddin A. Loop mediated isothermal amplification (LAMP) assays as a rapid diagnostic for COVID-19. Med. Hypotheses 141, 109786 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]; • Review of recent advancements in enhanced LAMP protocol assays for COVID-19 diagnosis.
- 8.Park GS, Ku K, Baek SH et al. Development of reverse transcription loop-mediated isothermal amplification assays targeting Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2). J. Mol. Diagn. 22(6), 729–735 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang Y, Ren G, Buss J, Barry AJ, Patton GC, Tanner NA. Enhancing colorimetric loop-mediated isothermal amplification speed and sensitivity with guanidine chloride. BioTechniques 69(3), 178–185 (2020). [DOI] [PubMed] [Google Scholar]; • GHC is presented as an enhancer of the speed and sensitivity of LAMP for SARS-CoV-2 detection.
- 10.Yan C, Cui J, Huang L et al. Rapid and visual detection of 2019 novel coronavirus (SARS-CoV-2) by a reverse transcription loop-mediated isothermal amplification assay. Clin. Microbiol. Infect. 26(6), 773–779 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lu R, Wu X, Wan Z, Li Y, Jin X, Zhang C. A novel reverse transcription loop-mediated isothermal amplification method for rapid detection of SARS-CoV-2. Int. J. Mol. Sci. 21(8), 2826 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huang WE, Lim B, Hsu CC et al. RT-LAMP for rapid diagnosis of coronavirus SARS-CoV-2. Microb. Biotechnol. 13(4), 950–961 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kitagawa Y, Orihara Y, Kawamura R et al. Evaluation of rapid diagnosis of novel coronavirus disease (COVID-19) using loop-mediated isothermal amplification. J. Clin. Virol. 129, 104446 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Baek YH, Um J, Antigua KJC et al. Development of a reverse transcription-loop-mediated isothermal amplification as a rapid early-detection method for novel SARS-CoV-2. Emerg. Microbes Infect. 9(1), 998–1007 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ganguli A, Mostafa A, Berger J et al. Rapid isothermal amplification and portable detection system for SARS-CoV-2. Proc. Natl Acad. Sci. USA 117(37), 22727–22735 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lau YL, Ismail I, Mustapa NI et al. Real-time reverse transcription loop-mediated isothermal amplification for rapid detection of SARS-CoV-2. PeerJ 8, e9278 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]; • RT-LAMP may allow the detection of one copy of SARS-CoV-2 RNA in 30 min.
- 17.Jiang M, Pan W, Arasthfer A et al. Development and validation of a rapid, single-step reverse transcriptase loop-mediated isothermal amplification (RT-LAMP) system potentially to be used for reliable and high-throughput screening of COVID-19. Front. Cell Infect. Microbiol. 10, 331 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ben-Assa N, Naddaf R, Gefen T et al. Direct on-the-spot detection of SARS-CoV-2 in patients. Exp. Biol. Med. (Maywood) 245(14), 1187–1193 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]; • Authors developed a LAMP protocol for the detection of SARS-CoV-2 from saliva and clinical swabs without a step of RNA purification.
- 19.Lalli MA, Chen X, Langmade SJ et al. Rapid and extraction-free detection of SARS-CoV-2 from saliva with colorimetric LAMP. Clin. Chem. 67(2), 415–424 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]; • Authors developed a rapid colorimetric assay using optimized RT-LAMP for SARS-CoV-2 detection on saliva samples without an RNA purification step.
- 20.Primer Explorer user manual. https://primerexplorer.jp/e/v5_manual
- 21.New England Biolabs. WarmStart® Colorimetric LAMP 2X Master Mix (DNA & RNA). https://international.neb.com/products/m1800-warmstart-colorimetric-lamp-2x-master-mix-dna-rna#Product%20Information
- 22.Modak SS, Barber CA, Geva E, Abrams WR, Malamud D, Ongagna YS. Rapid point-of-care isothermal amplification assay for the detection of malaria without nucleic acid purification. Infect. Dis. (Auckl.) 9, 1–9 (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zou Y, Mason MG, Botella JR. Evaluation and improvement of isothermal amplification methods for point-of-need plant disease diagnostics. PLoS ONE 15(6), e0235216 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fuji S. How to obtain a nasopharyngeal swab specimen. N. Engl. J. Med. 383(3), e14 (2020). [DOI] [PubMed] [Google Scholar]
- 25.Wong SCY, Tse H, Siu HK et al. Posterior oropharyngeal saliva for the detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Clin. Infect. Dis. 71(11), 2939–2946 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chu AW, Chan W-M, Ip JD et al. Evaluation of simple nucleic acid extraction methods for the detection of SARS-CoV-2 in nasopharyngeal and saliva specimens during global shortage of extraction kits. J. Clin. Virol. 129, 104519 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kuiper JWP, Baade T, Kremer M et al. Detection of SARS-CoV-2 from raw patient samples by coupled high temperature reverse transcription and amplification. PLoS ONE 15(11), e0241740 (2020). [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.



