Abstract
Stem cell niche is regulated by intrinsic and extrinsic factors. In the Drosophila testis, cyst stem cells (CySCs) support the differentiation of germline stem cells (GSCs). However, the underlying mechanisms remain unclear. In this study, we found that somatic CG6015 is required for CySC maintenance and GSC differentiation in a Drosophila model. Knockdown of CG6015 in CySCs caused aberrant activation of dpERK in undifferentiated germ cells in the Drosophila testis, and disruption of key downstream targets of EGFR signaling (Dsor1 and rl) in CySCs results in a phenotype resembling that of CG6015 knockdown. CG6015, Dsor1, and rl are essential for the survival of Drosophila cell line Schneider 2 (S2) cells. Our data showed that somatic CG6015 regulates CySC maintenance and GSC differentiation via EGFR signaling, and inhibits aberrant activation of germline dpERK signals. These findings indicate regulatory mechanisms of stem cell niche homeostasis in the Drosophila testis.
Subject terms: Cell death, Differentiation
Introduction
Stem cell homeostasis is regulated by its microenvironments or stem cell niches1. In Drosophila, the testes contain a well-structured microenvironment comprising terminally differentiated somatic cells (hub cells), germline stem cells (GSCs), and somatic cyst stem cells (CySCs), which provides functional signals for the homeostasis of self-renewal and differentiation2. Each GSC is tightly enclosed by two CySCs (see refs. 2,3). GSCs and CySCs divide asymmetrically to produce two kinds of daughter cells: one remains in contact with the hub and retains self-renewal characteristics, while the other is displaced from the hub and undergoes initial differentiation4–6. GSCs produce gonialblasts (GBs), and CySCs give rise to early somatic cyst cells. With the encapsulation of early somatic cyst cells, GBs undergo transit amplification (TA) with four rounds of mitosis and then enter the meiotic stage before terminal differentiation. Fusomes facilitate connections and communication among germ cells, with morphological changes from “dot” to “bifurcation”7. During spermatogenesis, autonomous and nonautonomous cell signals are required for germline differentiation. CySCs encapsulate GSCs to establish a tight connection, and exchange signals with GSCs to support germline differentiation8–10. Currently, little is known about the interactions of GSCs in the Drosophila testis stem cell niche.
Several classical pathways contribute to germline differentiation in the Drosophila testis stem cell niche. The Janus kinase-signal transducers and activators of transcription (JAK-STAT) and bone morphogenetic protein (BMP) signaling pathways promote GSC self-renewal by repressing their differentiation11,12. Somatic activation of the Hedgehog (Hh) signaling pathway in CySCs regulates the maintenance of CySC characteristics and GSC fate determination13. The epidermal growth factor receptor (EGFR) signaling pathway, a highly conserved pathway, is involved in proliferation, differentiation, and several tumorigenic processes14–18. The EGFR signaling pathway is specifically activated in somatic cells by EGFR via Spitz (Spi), which is an EGF ligand secreted by germ cells19. Ras, one of the best-known downstream targets of EGFR, activates the mitogen-activated protein kinase (MAPK) cascade, which includes Raf kinase, MEK kinase, and ERK kinase. Phosphorylated ERK (dpERK) is recruited into the nucleus and activates downstream transcription factors through phosphorylation modification20. The EGFR signaling pathway in CySCs promotes TA division in GSCs in Drosophila19,21. Aberrations in the special microenvironment created by somatic cells result from a serious loss of EGFR signaling in somatic cells, causing defective encapsulation of GBs and accumulation of early-stage germ cells14,22. Higher activation levels of EGFR signaling in somatic cyst cells are necessary for the completion of germline terminal differentiation in Drosophila testes23. The EGFR signaling pathway drives germline differentiation indirectly by inhibiting BMP signals in Drosophila ovaries24. Genome-wide RNAi screening of the Drosophila S2 cells revealed a vital role of the splicing process in MAPK expression25, which could indicate that the EGFR signaling pathway plays a role in regulating somatic lineage.
Some intrinsic factors that control germline differentiation in CySCs have recently been identified26. Spliceosome structure and function have been extensively studied; essential spliceosome components like U2A are instrumental in regulating spermatogonial differentiation in Drosophila27. In addition, heterogeneous nuclear ribonucleoproteins (hnRNPs), heterogeneous nuclear ribonucleoprotein L (hnRNPL), and RNA-binding motif protein, X-linked-like-2 (RBMXL2), are essential for normal spermatogenesis in humans28,29. The yeast Prp17 (or CDC40) gene is involved in signaling, cell cycle progression, splicing, and development30–33. CG6015, the homolog of Prp17 in Drosophila34, has been identified as a spliceosome-related gene by a large-scale RNAi screen in Drosophila testes and contributes to GSC maintenance and differentiation35. However, the regulatory mechanism of CG6015 in the Drosophila testis remains unclear.
In this study, the function of CG6015 and the underlying mechanisms were systemically analyzed using in vivo and in vitro approaches in Drosophila. Our data showed that somatic CG6015 and Dsor1 are required for CySC maintenance and GSC differentiation. Moreover, knockdown of rolled (rl), driven by tj-Gal4, causes GSC differentiation defects, but does not affect the maintenance of cyst cells. Surprisingly, somatic silencing of CG6015, Dsor1, and rl results in the ectopic expression of dpERK in undifferentiated germ cells (germline dpERK signals) in Drosophila testes. Our data suggest a novel mechanism involving CG6015 and EGFR signaling, which modulates the differentiation process in the Drosophila testis stem cell niche.
Results
Somatic CG6015 is required for CySC maintenance and GSC differentiation
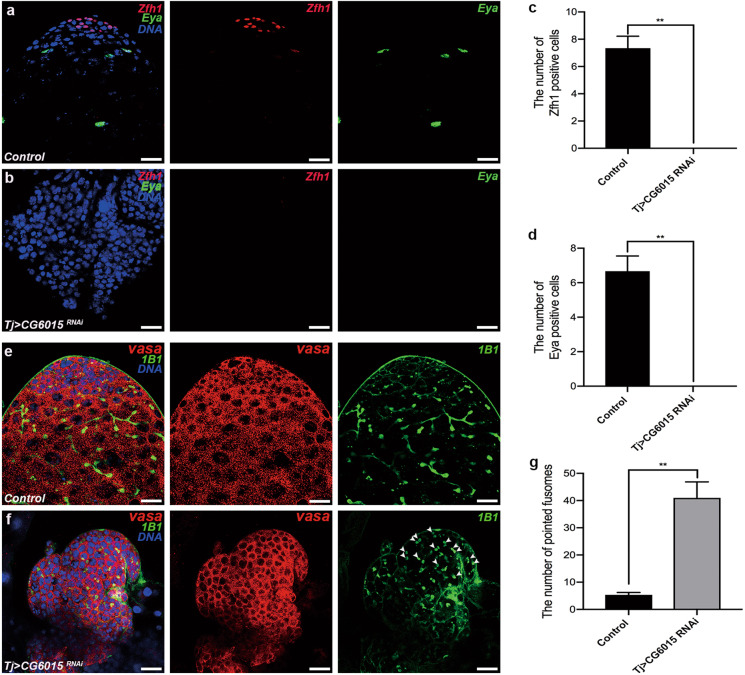
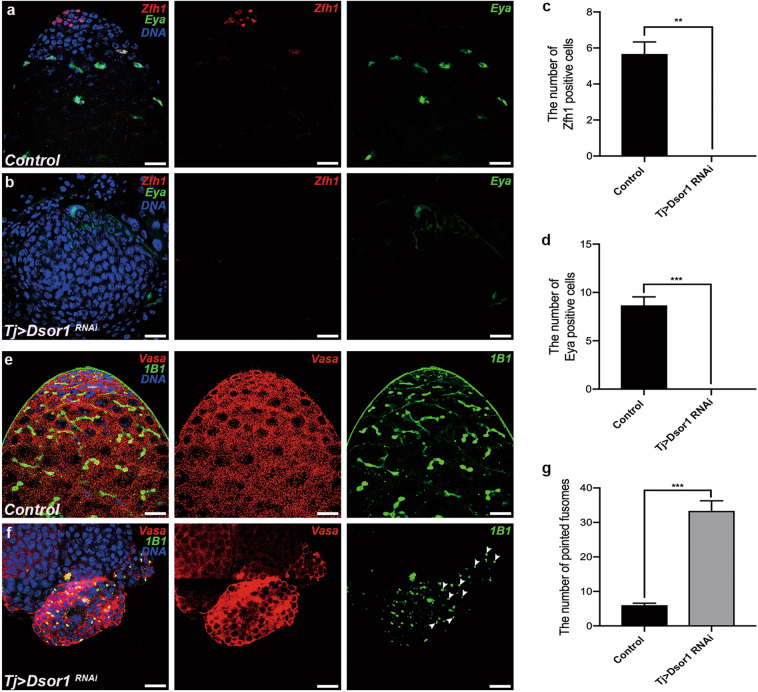
To determine the functions of somatic CG6015 in Drosophila testes, an RNAi-mediated analysis was conducted, driven by tj-GAL4. Zinc-finger homeodomain protein 1 (Zfh1) is a transcription factor that is highly expressed in CySCs and somatic cyst cells surrounding the hub36. High levels of eyes absent (Eya), a mature cyst cell marker, are typically observed in late-stage somatic cyst cells37. Unexpectedly, Zfh1 and Eya have not been detected in tj > CG6015 RNAi testes, indicating total loss of CySCs and mature cyst cells (Fig. 1a–d).
Fig. 1. The necessity of somatic CG6015 for CySC maintenance and GSC differentiation in Drosophila testes.
a, b Immunostaining of control (a) and tj > CG6015 RNAi (b) testes for Zfh1 (red) and Eya (green). c Number of Zfh1-positive cells in control (n = 3) and tj > CG6015 RNAi (n = 3) testes. d Number of Eya-positive cells in control (n = 3) and tj > CG6015 RNAi (n = 3) testes. e, f Immunostaining of control (e) and tj > CG6015 RNAi (f) testes for Vasa (red) and 1B1 (green). g Number of pointed fusomes in control (n = 3) and tj > CG6015 RNAi (n = 3) testes. The W1118 line was used as control. Student’s t test was used for the statistical analysis. **P < 0.01. Scale bar: 20 μm.
Vasa was used as a germ cell marker in the testis38, and fusomes were labeled with 1B1 to observe the differentiation process39. Control testes displayed a strong pattern with the formation of pointed and branched fusomes in Vasa-positive germ cells (Fig. 1e). However, accumulation of undifferentiated germ cells and pointed fusomes was observed in tj > CG6015 RNAi testes (Fig. 1f). The number of pointed fusomes in tj > CG6015 RNAi testes was significantly higher than that in controls (Fig. 1g). These results suggested that CG6015 regulates CySC characteristics, and nonautonomously affects germ cell differentiation.
Inactivation of somatic CG6015 causes differentiation defects without hub signals and modulates germ cell apoptosis and proliferation
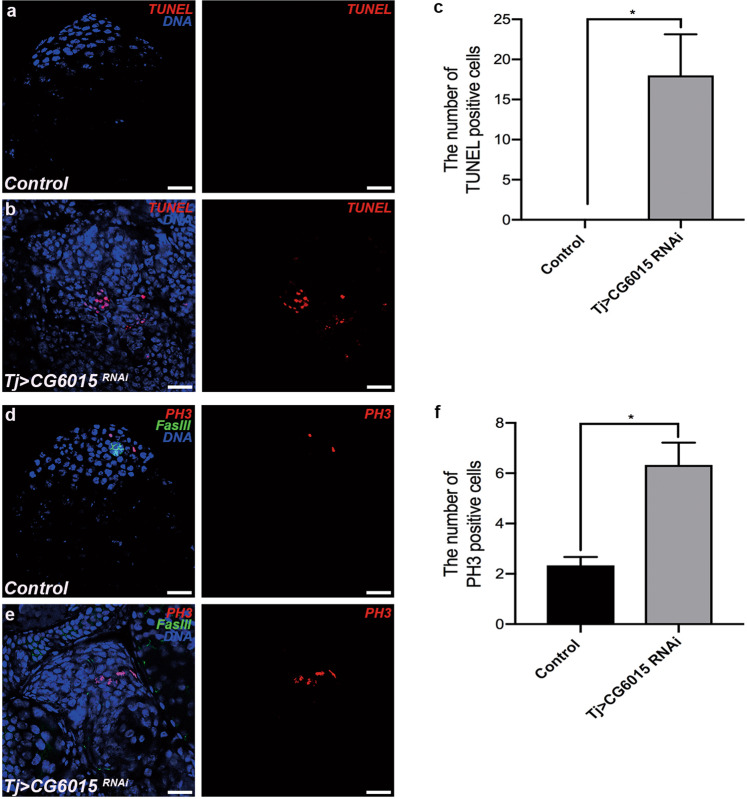
To characterize the accumulated undifferentiated germ cell cysts, we detected apoptosis and proliferation in the Drosophila testis. TUNEL-positive cells dramatically increased in tj > CG6015 RNAi testes (Fig. 2a–c). Fasciclin III (FasIII) protein and phosphohistone-3 (PH3) are markers of hub cells and M-phase cells, respectively. Surprisingly, no hub cells were observed around the accumulated undifferentiated germ cell cysts, while PH3-positive cells were significantly higher in tj > CG6015 RNAi testes than in the controls (Fig. 2d–f). These results suggested that apoptosis and proliferation were maintained without hub signals in undifferentiated germ cells induced by somatic CG6015 inactivation.
Fig. 2. Knockdown of CG6015 in CySCs leads to cell death and proliferation dysfunction.
a, b TUNEL (red) staining of control (a) and tj > CG6015 RNAi (b) testes. c Number of TUNEL-positive cells in control (n = 3) and tj > CG6015 RNAi (n = 3) testes. d, e Immunostaining of control (d) and tj > CG6015 RNAi (e) testes for PH3 (red) and FasIII (green). f Number of PH3-positive cells in control (n = 3) and tj > CG6015 RNAi (n = 3) testes. The W1118 line was used as control. Student’s t test was used for the statistical analysis. *P < 0.05. Scale bar: 20 μm.
CG6015 regulates proliferation and apoptosis in S2 cells
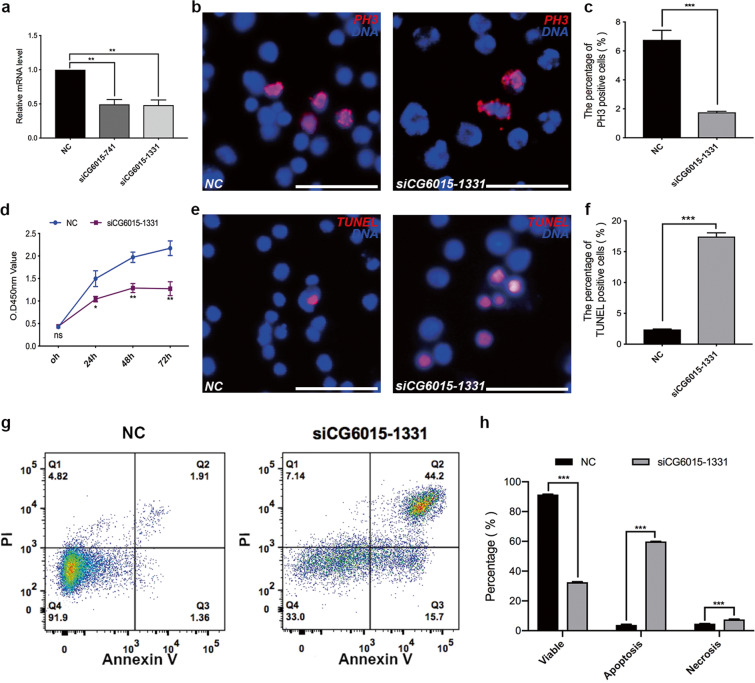
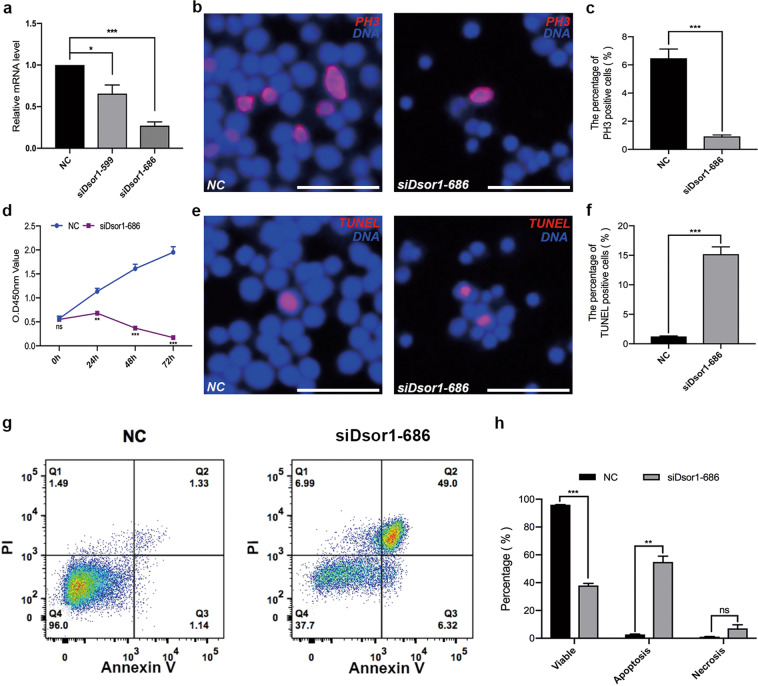
The function CG6015 was further analyzed in vitro. Two small-interfering RNAs (siRNAs: siCG6015-741 and siCG6015-1331) were used to silence the mRNA expression of CG6015, and siCG6015-1331 was selected for further functional analysis in S2 cells (Fig. 3a). First, we found that the relative mRNA expression levels of Prp complex subunits (Prp19 and Prp18) and Sm complex subunits (SmB, SmD1, and SmF) were upregulated after silencing of CG6015 (Supplementary Fig. S1). We also observed that siCG6015 decreased the proportion of PH3-positive S2 cells (Fig. 3b, c). CCK-8 assay was simultaneously conducted to assess cell growth conditions after silencing of CG6015, and the results showed clear suppression of cell growth ability (Fig. 3d). In addition, apoptosis was detected using the TUNEL assay and flow cytometry. Interestingly, the ratio of TUNEL-positive cells increased after CG6015 knockdown (Fig. 3e, f), indicating that siCG6015 treatment promoted apoptosis. Flow cytometric cell component tests confirmed these results and showed that the ratio of apoptosis and necrosis significantly increased in S2 cells after treatment with CG6015 siRNA (Fig. 3g, h). These results indicated that CG6015 is essential for cell proliferation and cell death.
Fig. 3. Functional analysis of CG6015 in S2 cells.
a Relative mRNA levels of CG6015 in the negative control (NC) and siCG6015 (siCG6015-741 and siCG6015-1331)-treated S2 cells to validate knockdown efficiency. b Immunostaining of NC and siCG6015-1331-treated S2 cells by using PH3 (red). c Percentages of PH3-positive cells in NC (n = 3) and siCG6015-1331-treated (n = 3) S2 cells. d CCK-8 test in NC and siCG6015-1331-treated S2 cells. e TUNEL (red) staining in NC and siCG6015-1331-treated S2 cells. f Percentages of TUNEL-positive cells in NC (n = 3) and siCG6015-1331-treated (n = 3) S2 cells. g Cell component analysis in NC and siCG6015-1331-treated S2 cells by flow cytometry. h Percentages of cell components in NC (n = 3) and siCG6015-1331-treated (n = 3) S2 cells. The Student’s t test was used for the statistical analysis. *P < 0.05; **P < 0.01; ***P < 0.001; n.s. not significant. Scale bar: 30 μm.
Somatic CG6015 inactivation disrupts the expression pattern of dpERK
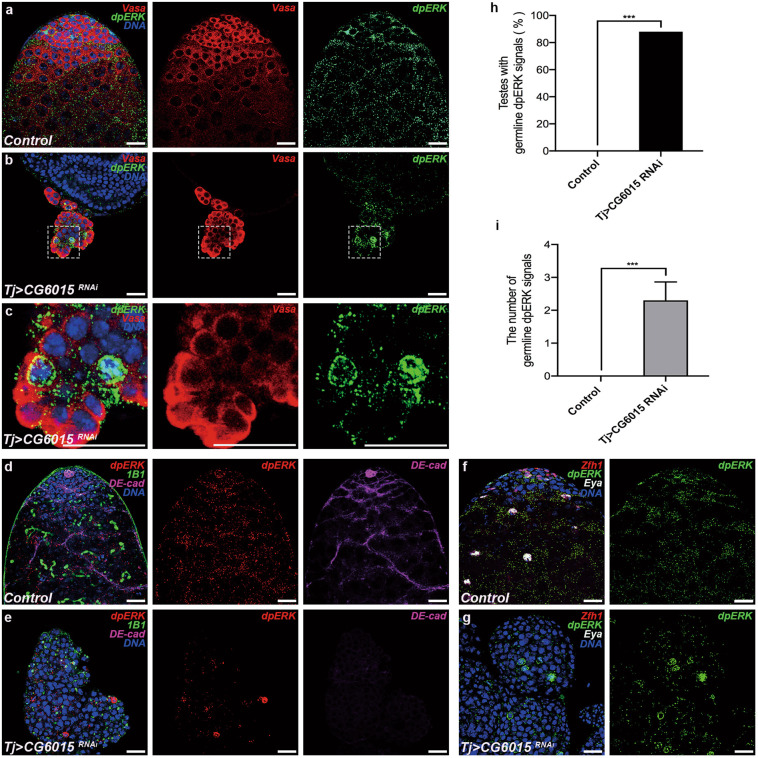
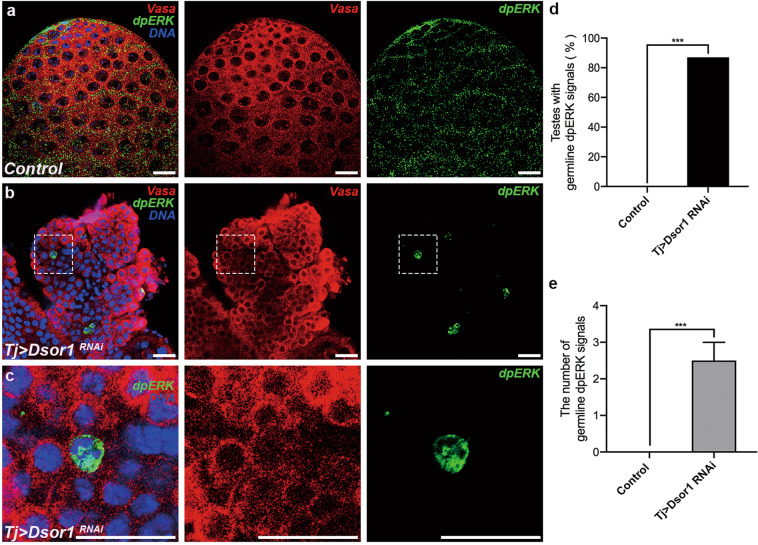
EGFR signaling in somatic cells has critical functions in GSC differentiation22,40. We aimed to determine whether CG6015 mediates GSC differentiation via EGFR signaling by detecting dpERK, a key downstream activator of the EGFR signaling pathway41. In the control testes, dpERK was mainly detected in somatic cyst cells (Fig. 4a), as described previously42. Conversely, we observed ectopic dpERK signals, mostly mislocalized in germ cell nuclei, in accumulated undifferentiated germ cell cysts after knockdown of CG6015 driven by tj-GAL4 (Fig. 4b, c). Moreover, the mislocalized germline dpERK signals were accompanied by pointed fusomes (Fig. 4d, e), and were not co-located with Zfh1 or Eya (Fig. 4f, g). Importantly, the percentage of testes with mislocalized germline dpERK signals increased to 88% (Fig. 4h). Further, the number of mislocalized germline dpERK signals increased by an average of 2.30 ± 0.56 in tj > CG6015 RNAi testes (Fig. 4i). Thus, inactivation of CG6015 in somatic cyst cells led to aberrant activation of ERK kinase in germ cells, along with differentiation defects, suggesting a possible role for CG6015 and EGFR signaling in germline differentiation in the stem cell niche.
Fig. 4. Somatic CG6015 inactivation leads to aberrant dpERK expression.
a–c Immunostaining of control (a) and tj > CG6015 RNAi (b, c) testes for Vasa (red) and dpERK (green). Panel (c) shows enlarged views of sections marked in (b). d, e Immunostaining of control (d) and tj > CG6015 RNAi (e) testes for dpERK (red), 1B1 (green), and DE-cad (magenta). f, g Immunostaining of control (f) and tj > CG6015 RNAi (g) testes for Zfh1 (red), dpERK (green), and Eya (gray). h Percentages of testes with germline dpERK signals in control (n = 20) and tj > CG6015 RNAi (n = 16) testes. Chi-square test was used to evaluate for statistical differences of testes with germline dpERK signals. i Number of germline dpERK signals in control (n = 10) and tj > CG6015 RNAi (n = 10) testes. The Student’s t test was used for statistical differences of the number of germline dpERK signals. ***P < 0.001. The W1118 line was used as control. Scale bar: 20 μm.
Dsor1 is required for CySC maintenance and GSC differentiation in CySC lineages
MEK, also known as downstream of raf (Dsor1), is the direct phosphorylation activator of ERK (also called rl in Drosophila)43. To determine the exact roles of Dsor1 and elucidate the relationship between CG6015 and EGFR signaling in CySC lineages, we knocked down Dsor1 driven by tj-Gal4. Neither Zfh1 nor Eya signal was detected after knocking down of Dsor1 in CySCs (Fig. 5a–d), indicating that Dsor1 is essential for CySC maintenance. Further, undifferentiated germ cells accumulated and pointed fusomes increased in tj > Dsor1 RNAi testes (Fig. 5e–g). Higher TUNEL signals were observed in tj > Dsor1 RNAi testes when compared with control testes (Supplementary Fig. S2a–c). In addition, undifferentiated germ cells in tj > Dsor1 RNAi testes could proliferate without hub signals (Supplementary Fig. S2d–f). These results suggested that Dsor1 could phenocopy CG6015 in Drosophila testes.
Fig. 5. Somatic Dsor1 is indispensable for CySC maintenance and GSC differentiation.
a, b Immunostaining of control (a) and tj > Dsor1 RNAi (b) testes for Zfh1 (red) and Eya (green). c Number of Zfh1-positive cells in control (n = 3) and tj > Dsor1 RNAi (n = 3) testes. d Number of Eya-positive cells in control (n = 3) and tj > Dsor1 RNAi (n = 3) testes. e, f Immunostaining of control (e) and tj > Dsor1 RNAi (f) testes for Vasa (red) and 1B1 (green). g Number of pointed fusomes in control (n = 3) and tj > Dsor1 RNAi (n = 3) testes. The W1118 line was used as control. The Student’s t test was used for the statistical analysis. **P < 0.01; ***P < 0.001. Scale bar: 20 μm.
Somatic downregulation of rl disrupts GSC differentiation
rl is a key downstream target of the EGFR signaling pathway43. We observed Zfh1-positive cells and Eya-positive cells in tj > rl RNAi testes (Supplementary Fig. S3a, b), indicating that rl was not necessary for the maintenance of CySCs. No significant difference was observed in the quantity of Zfh1-positive cells, while the number of Eya-positive cells significantly decreased (Supplementary Fig. S3c, d). Unlike in the control testes, early germ cells dramatically accumulated, with increased pointed fusomes, in the tj > rl RNAi testes (Supplementary Fig. S3e–g). Although hub cells existed in all testes, the number of TUNEL-positive and PH3-positive cells increased in tj > rl RNAi testes (Supplementary Fig. S4). These results emphasized the crucial roles of rl in the somatic cyst lineage, in the promotion of GSC differentiation.
EGFR signaling regulates the survival of Drosophila S2 cells
To elucidate the function of the EGFR signaling pathway, we used multiple small-interfering RNAs (siDsor1-599 and siDsor1-686 and sirl-51 and sirl-785) to downregulate the expression of Dsor1 and rl, respectively, in S2 cells. siDsor1-686 and sirl-785, selected using qRT-PCR, were used for the functional analysis (Fig. 6a and Supplementary Fig. S5a). We found that the percentage of PH3-positive signals was dramatically lower in the siDsor1-686 and sirl-785 groups than in the control groups (Fig. 6b, c and Supplementary Fig. S5b, c). Further, apparent inhibition of cell growth was observed using CCK-8 assay after silencing Dsor1 or rl (Fig. 6d and Supplementary Fig. S5d), suggesting that blockage of EGFR signaling pathway might reduce cell survival. Moreover, to determine whether Dsor1 and rl were involved in apoptosis, TUNEL and flow cytometry assays were performed separately. The percentage of TUNEL-positive cells (Fig. 6e, f and Supplementary Fig. S5e, f), and the ratio of apoptotic cells dramatically increased after silencing Dsor1 or rl, compared with control (Fig. 6g, h and Supplementary Fig. S5g, h). Thus, our results indicated that Dsor1 and rl, as key downstream targets of EGFR signaling, are essential for S2 cell survival.
Fig. 6. Dsor1 regulates proliferation and apoptosis in S2 cells.
a Relative mRNA levels of Dsor1 in the negative control (NC) and siDsor1 (siDsor1-599 and siDsor1-686)-treated S2 cells to validate knockdown efficiency. b Immunostaining of PH3 (red) in NC and siDsor1-686-treated S2 cells. c Proportions of PH3-positive cells in NC (n = 3) and siDsor1-686-treated (n = 3) S2 cells. d CCK-8 test in NC and siDsor1-686-treated S2 cells. e TUNEL (red) staining in NC and siDsor1-686-treated S2 cells. f Percentages of TUNEL-positive cells in NC (n = 4) and siDsor1-686-treated (n = 4) S2 cells. g Flow cytometry test for cell components in NC and siDsor1-686-treated S2 cells. h Percentages of cell components in NC (n = 3) and siDsor1-686-treated (n = 3) S2 cells. The Student’s t test was used for the statistical analysis. *P < 0.05; **P < 0.01; ***P < 0.001; n.s. not significant. Scale bar: 30 μm.
Inactivation of Dsor1 and rl in CySCs disrupts the expression pattern of dpERK
Since our results revealed a role for CG6015 in GSC differentiation via EGFR signaling, and somatic CG6015 could cause mislocalized expression of dpERK in germ cells, we then determined whether germline dpERK signals reappeared in accumulated undifferentiated germ cells after the downregulation of Dsor1 and rl. Surprisingly, a significant accumulation of aberrantly expressed dpERK signals was detected in the undifferentiated germ cells, compared with control testes (Fig. 7a–c and Supplementary Fig. S6a–c). The percentage of testes with mislocalized germline dpERK signals increased in tj > Dsor1 RNAi testes (87%, Fig. 7d) and tj > rl RNAi testes (78%, Supplementary Fig. S6d). Further, the number of mislocalized germline dpERK signals increased by an average of 2.50 ± 0.50 in tj > Dsor1 RNAi testes (Fig. 7e) and 3.22 ± 0.95 in tj > rl RNAi testes (Supplementary Fig. S6e). The inactivation of the EGFR signaling pathway led to germline differentiation defects and activated ectopic expression of dpERK, providing novel evidence of EGFR-mediated differentiation defects and ERK kinase activation in germ cells.
Fig. 7. Somatic Dsor1 inactivation causes ectopic dpERK expression in undifferentiated germ cells.
a–c Immunostaining of Vasa (red) and dpERK (green) in control (a) and tj > Dsor1 RNAi (b, c) testes. Images in (c) are enlarged views of the areas marked in (b). d Percentages of testes with germline dpERK signals in control (n = 20) and tj > Dsor1 RNAi (n = 15) testes. Chi-square test was used to evaluate for statistical differences of testes with germline dpERK signals. e The number of germline dpERK signals in control (n = 10) and tj > Dsor1 RNAi (n = 10) testes. The Student’s t test was used for statistical differences of the number of germline dpERK signals. ***P < 0.001. The W1118 line was used as control. Scale bar: 20 μm.
Discussion
In this study, we determined the functions of somatic CG6015 in CySC maintenance and GSC differentiation and elucidated novel mechanisms involving CG6015 and EGFR signaling. We discovered that CG6015 and key targets of the EGFR signaling pathway (Dsor1 and rl) were present in CySCs and mediated GSC differentiation by non-cell-autonomous effects. Importantly, germline dpERK signals were activated and observed among the accumulated undifferentiated germ cells in the Drosophila testis after the disruption of CG6015, Dsor1, and rl in CySCs.
Preliminary evidence identified CG6015 as a GSC regulator in the Drosophila testis35. Here, we found that inactivation of CG6015 in CySCs led to GSC differentiation defects, and promoted self-renewal and apoptosis of the accumulated undifferentiated germ cells without normal niche signals. Somatic CG6015, which induced GSC differentiation defects, unexpectedly activated the aberrant expression of germline dpERK signals in undifferentiated germ cells. Importantly, Dsor1 or rl mimicked the phenotype of CG6015 in Drosophila testes and S2 cells, indicating possible correlations between CG6015 and EGFR signaling pathway (Fig. 8).
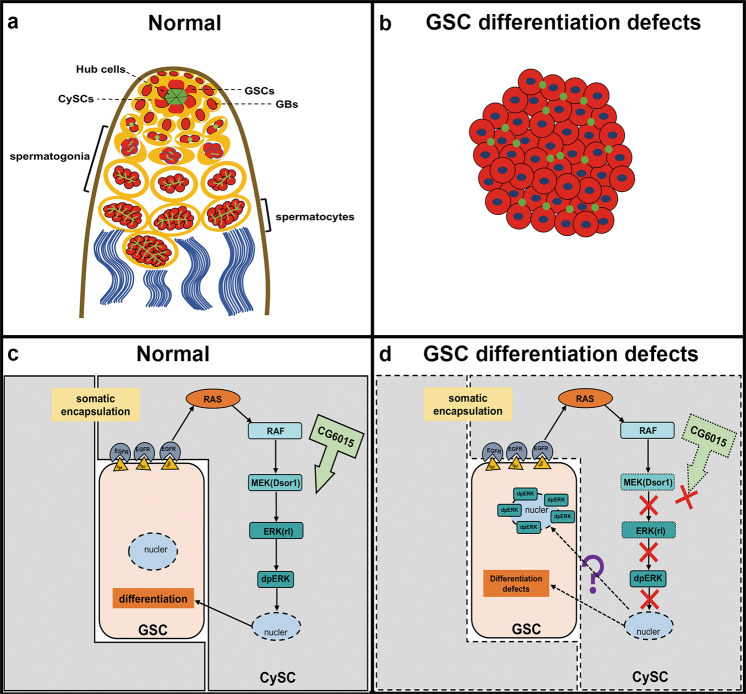
Fig. 8. Schematic representation of CG6015 and EGFR signaling in the Drosophila testis.
a, b Graphical representation of the apical tips of normal testes and testes with GSC differentiation defects. c, d Mechanisms of CG6015 and EGFR signaling. In normal testes, the stem cell niche is composed of GSCs, CySCs, and hub cells, which occupy the apical tips of the testis. For the integrity of germ cell differentiation, somatic cells should envelop germ cells, and fusomes gradually transition from dot to bifurcate. Somatic inactivation of CG6015 and key targets in the EGFR signaling pathway lead to the accumulation of undifferentiated germ cells, and eventually the formation of tumor-like germ cell cysts with pointed fusomes in the Drosophila testis. Our results show the maintenance of stem cell niche homeostasis by CG6015 and EGFR signaling via germline dpERK signals.
Somatic cells are required to encapsulate germ cells for differentiation14. Several studies have demonstrated the roles of EGFR signaling in germline differentiation via EGFR, Ras, and Raf factors23,26,42,44. However, the role of Dsor1 and rl in the homeostasis of the stem cell niche in the Drosophila testis was not extensively studied. Dsor1 is a downstream target that could directly phosphorylate ERK protein45. The mRNA expression of rl could be affected by some splicing factors25. Our results also revealed that one such splicing factor, CG6015, mediated expression changes of several spliceosome components, indicating regulatory roles for CG6015 in spliceosomes. We, therefore, hypothesized that CG6015 likely plays a critical role in mRNA splicing, and regulates EGFR signaling for germ cell differentiation. Inactivation of CG6015 and its targets in EGFR signaling in CySCs disrupted the balance of stem cell niche signals, leading to failure of GSC differentiation.
Considering the functions of Dsor1 and rl in the testis, they likely act via more pathways than only the MEK–ERK cascade to control somatic behavior. EGFR could also activate Vav/Rac1/Rho1 signals to display similar germ cell enclosure functions. Rho1, a negative regulatory factor of rl, could regulate germ cell enclosure antagonistically against EGFR/Vav/Rac1 (see ref. 19). Rho1 is a small GTPase, which regulates the actin cytoskeleton. Several signaling pathways modulate cell motility and cell shape by controlling cytoskeleton dynamics46. Importantly, a somatic permeability barrier has been discovered to regulate germline encapsulation and differentiation47. Hence, Rho1-mediated cytoskeleton changes could damage germline encapsulation by affecting the somatic permeability barrier. Further studies are needed to elucidate the detailed mechanisms by which EGFR signaling modulates GSC encapsulation and differentiation.
Normally, dpERK is activated and expressed in CySCs and somatic cyst cells of the testis23. The initial discovery of ectopically expressed dpERK signals in undifferentiated germ cells was unexpected. To confirm the localization of these ectopic dpERK signals, co-staining of Vasa and dpERK was performed. The possibility of co-localization between the ectopic dpERK signals and somatic lineages was assessed and excluded. Germline dpERK signals were reported for the first time in our study. We speculated whether GSC differentiation defects induced the activation of germline dpERK signals, providing us with a specific understanding of the germline EGFR signaling pathway. The effects of MAPK cascades in cell-fate determination, such as proliferation, apoptosis, and differentiation, have been extensively investigated48. The localization of dpERK in the nucleus or cytoplasm was regarded as a switch to determine proliferation and differentiation in mouse muscle progenitors. Increased dpERK nuclear translocation could repress myogenic differentiation in head muscle progenitors49. Prevention of ERK nuclear translocation has been studied as a novel therapeutic strategy for some Ras/ERK-related cancers50. This is a possible explanation for the nuclear accumulation of the germline dpERK signals. However, the detailed mechanism requires further study.
In summary, we discovered that CG6015 was required in CySCs for GSC differentiation through EGFR signaling. Our discovery of activated germline dpERK signals could greatly improve our understanding of stem cell niche regulation, male fertility, and germline tumorigenesis.
Materials and methods
Fly stocks and fly crosses
All flies were raised on standard cornmeal molasses agar medium at 25 °C. The transgenic RNA interference (RNAi) flies used in this study were obtained from TsingHua Fly Center (THFC) and were derived from the same RNAi collection as the Transgenic RNAi Project. UAS-RNAi flies included UAS-CG6015 RNAi (THU1409), UAS-Dsro1 RNAi (THU0677), and UAS-rl RNAi (THU3530). Tj-Gal4 (104055) was acquired from Drosophila Genetic Resource Consortium (DGRC). The W1118 line was used as control.
UAS/Gal4 system was used to mediate tissue-specific expressed knockdown in Drosophila. Two-to-three-day-old flies were used in this study. Males from tj-Gal4 line were randomly selected to cross with transgenic UAS-RNAi virgin females and raised at 25 °C. Then we chose qualified male offsprings with specific genotypes for further functional analysis.
Cell culture and transfection
Drosophila S2 cells were obtained from Drosophila Genomics Resource Center and were cultured in Schneider’s Drosophila medium (21720024, Gibco, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS) (04-001-1ACS, Bioind, Israel) at 28 °C. Every 3–4 days, S2 cells were passaged to another plate in 1:3 or 1:2 ratio as previously described51. Cells have been tested and confirmed to be free of mycoplasma contamination.
Before transfection, S2 cells were seeded into a six-well plate to guarantee that the cell growth area reached 70–80% of the well. For knocking down target genes, Lipofectamine 2000 Transfection Reagent (Lipo2000, 11668019, Invitrogen, USA), Opti-Minimal Essential Medium (MEM) (31985-062, Gibco, USA), and siRNA were used together. The detailed transfected process was as follows: two tubes were prepared to mix reagent, the first one contained 250 μl Opti-MEM and 15 μl Lipo2000 and incubated for 5 min after vortexing for 5 s at room temperature. Then, the other one contained 250 μl Opti-MEM, and 15 μl siRNA was mixed with the first tube and incubated for 20 min after vortexing for 5 s at room temperature. The siRNAs were designed and synthesized by GenePharma (Suzhou, China). The detailed information of siRNAs is listed in Supplementary Table S1.
Quantitative real-time PCR
The total RNA was extracted from S2 cells using RNAiso Plus kit (9108, Takara, Japan). cDNA was synthesized by Prime Script RT Reagent Kit (RR037A, Takara, Japan). TB GreenTM Premix Ex TaqTM (RR420A, Takara, Japan) was used to carry out qRT-PCR and GAPDH was amplified as an internal standard. Fold changes were calculated using the standard curve according to the manufacturer’s protocol. Each experiment was repeated three times independently. Information of all primers used for qRT-PCR is listed in Supplementary Table S2.
Immunostaining and antibodies
Fly testes were dissected in 1× PBS and fixed for 30 min in 4% PFA. They were washed three times in 0.3% PBST and blocked for 30 min in 5% BSA. Testes were incubated with primary antibody overnight at 4 °C. Then the samples were washed three times for 30 min in 0.3% PBST and incubated with secondary antibodies at room temperature for 1 h avoiding light. Testes were then washed three times again by 0.3% PBST. Finally, testes were stained with Hoechst-33342 (1.0 mg/mL, C0031, Solarbio, Beijing, China) for 5 min before finalizing51. The images were captured by LSM800 Zeiss confocal microscope and processed by Adobe Photoshop Software. Drosophila S2 cells were cultured on cover glasses for 48 h, and immunostaining was performed in 24-well plates according to the similar protocols described above.
The antibodies used in this study included rat anti-Zfh1 (a gift from Tong lab, 1:1000), mouse anti-Eya (#AB_528232, DSHB: Developmental Studies Hybridoma Bank, 1:30), rat anti-Vasa (#AB_760351, DSHB, 1:20), mouse anti-1B1 (#AB_528070, DSHB, 1:50), rabbit anti-PH3 (#53348, CST: Cell Signaling Technology, 1:1000), mouse anti-FasIII (#AB_528238, DSHB, 1:50), rat anti-DE-cadherin (#AB_528120, DSHB, 1:15), and rabbit anti-dpERK (#4370, CST, 1:200). Secondary antibodies containing Cy3, A488, and A647 (Molecular Probes and Jackson Immunological) were diluted at 1:400 with 5% BSA.
TUNEL assay
Cell death tests in testes and S2 cells were examined by TUNEL BrightRed Apoptosis Detection Kit (A113, Vazyme, Nanjing, China) according to the manufacturer’s protocols. After blocking, testes or S2 cells were incubated in 50 μl of 1× equilibration buffer that was diluted by ddH2O at 1:5 (room temperature) for 30 min in the dark. Then, a mixture with 34 μl ddH2O, 10 μl 5× equilibration buffer, 5 μl BrightRed Labeling Mix, and 1 μl Recombinant TdT Enzyme was prepared in the dark. After balance, 50 μl mixture was added and incubated with testes or S2 cells for 1 h at 37 °C in the dark. After TUNEL staining, both testes and S2 cells were washed in 1× PBS three times. Before finalizing, testes and S2 cells were stained with Hoechst-33342 (1.0 mg/mL, C0031, Solarbio, Beijing, China) for 5 min.
Flow cytometry assay
After transfection for 48 h in S2 cells, flow cytometry was conducted by Annexin V-Alexa Fluor 647/propidium iodide (PI) Apoptosis Assay Kit (FMSAV647-100, FcMACS, Nanjing, China). According to the manufacturer’s protocols, S2 cells were washed with ice-cold 1× PBS and resuspended by binding buffer that was diluted with DEPC-treated water at the ratio of 1:4 after centrifugation. In all, 5 μl Annexin V-Alexa Fluor 647 and 10 μl PI were added into 100-μl cell suspension. In total, 1 × 106 cells are required in each sample and incubated for 15 min at room temperature in the dark. Before testing on FACScan flow cytometry (BD Biosciences intervals, San Jose, CA, USA), 200 μl 1× PBS was added into each sample to dilute cells. The experiments required more than three replicates, and the final results were analyzed and processed by FlowJo software.
Cell viability assay
CCK-8 Cell Counting Kit (A311-01-AA, Vazyme, Nanjing, Chania) was used to analyze cell growth situation in S2 cells. Based on the manufacturer’s instructions, after transfection at 0, 24, 48, and 72 h, S2 cells were collected and resuspended after centrifugation using 10% CCK-8 mixture, which was diluted by Schneider’s Drosophila Medium. The cell suspension was seeded into 96-well plates with three accessory wells for each sample and incubated at 37 °C for 2 h. The absorbance in each well was evaluated on a spectrophotometer (Multiskan GO, Thermo Scientific, Waltham, USA) at 450 nm. The results were performed for more than three independent experiments.
Statistical analysis
All the experiments conducted in this study were repeated at least three times. The quantitative results were presented as means ± standard error of the mean (SEM) and evaluated for statistical differences using Student’s t test and one-way ANOVA by Graphpad Software (https://www.graphpad.com/). Chi-square test was used to evaluate for statistical differences of testes with germline dpERK signals. *P < 0.05; **P < 0.01; ***P < 0.001.
Supplementary information
Acknowledgements
The authors wish to thank all study participants, research staff, and students who assisted with this work. We would like to thank Chao Tong from Life Sciences Institute, Zhejiang University, for sharing reagents and stocks. We are grateful to the THFC, DGRC, and DHSB for providing fly strains and antibodies.
Author contributions
J.Y., B.Z., and X.C. initiated the project and designed the study, J.Y., C.Q., Y.W., and Q.Z. coordinated the experiments and wrote the paper. Q.Z., M.W., W.C., X.L. Y.Y., Y.W., B.Z., and C.S. performed the experiments and provided conceptual inputs for the paper; Q.Z., J.Y., J.F., and X.H. analyzed the data. All authors read and approved the final paper.
Funding
This work was supported by the National Natural Science Foundation of China (81701511, 81901532, 81901533, 81803505), Natural Science Foundation of Jiangsu Province (BK20190188), Scientific Research Project of Wuxi Health Committee (Q201926), and Suzhou Key Laboratory of Male Reproduction Research (SZS201718).
Conflict of interest
The authors declare no competing interests.
Footnotes
Edited by Ivano Amelio
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Bo Zheng, Email: mansnoopy@163.com.
Yibo Wu, Email: moliaty@aliyun.com.
Jun Yu, Email: yujun9117@126.com.
Supplementary information
The online version contains supplementary material available at 10.1038/s41420-021-00452-w.
References
- 1.Fairchild MJ, Yang L, Goodwin K, Tanentzapf G. Occluding junctions maintain stem cell niche homeostasis in the fly testes. Curr. Biol. 2016;26:2492–2499. doi: 10.1016/j.cub.2016.07.012. [DOI] [PubMed] [Google Scholar]
- 2.Xu R, et al. Self-restrained regulation of stem cell niche activity by niche components in the Drosophila testis. Dev. Biol. 2018;439:42–51. doi: 10.1016/j.ydbio.2018.04.011. [DOI] [PubMed] [Google Scholar]
- 3.Issigonis M, Matunis E. Snapshot: stem cell niches of the Drosophila testis and ovary. Cell. 2011;145:994–994. e2. doi: 10.1016/j.cell.2011.05.037. [DOI] [PubMed] [Google Scholar]
- 4.de Cuevas M, Matunis EL. The stem cell niche: lessons from the Drosophila testis. Development. 2011;138:2861–2869. doi: 10.1242/dev.056242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Yamashita YM, Jones DL, Fuller MT. Orientation of asymmetric stem cell division by the APC tumor suppressor and centrosome. Science. 2003;301:1547–1550. doi: 10.1126/science.1087795. [DOI] [PubMed] [Google Scholar]
- 6.Zoller R, Schulz C. The Drosophila cyst stem cell lineage: partners behind the scenes? Spermatogenesis. 2012;2:145–157. doi: 10.4161/spmg.21380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eikenes ÅH, Brech A, Stenmark H, Haglund K. Spatiotemporal control of Cindr at ring canals during incomplete cytokinesis in the Drosophila male germline. Dev. Biol. 2013;377:9–20. doi: 10.1016/j.ydbio.2013.02.021. [DOI] [PubMed] [Google Scholar]
- 8.Lim JG, Fuller MT. Somatic cell lineage is required for differentiation and not maintenance of germline stem cells in Drosophila testes. Proc. Natl Acad. Sci. USA. 2012;109:18477–18481. doi: 10.1073/pnas.1215516109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jemc JC. Somatic gonadal cells: the supporting cast for the germline. Genesis. 2011;49:753–775. doi: 10.1002/dvg.20784. [DOI] [PubMed] [Google Scholar]
- 10.Cheng J, Tiyaboonchai A, Yamashita YM, Hunt AJ. Asymmetric division of cyst stem cells in Drosophila testis is ensured by anaphase spindle repositioning. Development. 2011;138:831–837. doi: 10.1242/dev.057901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Amoyel M, Bach EA. Functions of the Drosophila JAK-STAT pathway: lessons from stem cells. JAKSTAT. 2012;1:176–183. doi: 10.4161/jkst.21621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ma X, Xie T. Stem cells: keeping BMP signaling local. Curr. Biol. 2011;21:R809–R811. doi: 10.1016/j.cub.2011.08.051. [DOI] [PubMed] [Google Scholar]
- 13.Zhang Z, Lv X, Jiang J, Zhang L, Zhao Y. Dual roles of Hh signaling in the regulation of somatic stem cell self-renewal and germline stem cell maintenance in Drosophila testis. Cell Res. 2013;23:573–576. doi: 10.1038/cr.2013.29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kiger AA, White-Cooper H, Fuller MT. Somatic support cells restrict germline stem cell self-renewal and promote differentiation. Nature. 2000;407:750–754. doi: 10.1038/35037606. [DOI] [PubMed] [Google Scholar]
- 15.Harden N. New insights from Drosophila into the regulation of EGFR signaling. Methods Mol. Biol. 2017;1652:37–42. doi: 10.1007/978-1-4939-7219-7_2. [DOI] [PubMed] [Google Scholar]
- 16.Fereres S, Hatori Ryo, Hatori M, Kornberg TB. Cytoneme-mediated signaling essential for tumorigenesis. PLoS. Genet. 2019;15:e1008425. doi: 10.1371/journal.pgen.1008415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Shilo BZ. Signaling by the Drosophila epidermal growth factor receptor pathway during development. Exp. Cell Res. 2003;284:140–149. doi: 10.1016/S0014-4827(02)00094-0. [DOI] [PubMed] [Google Scholar]
- 18.Li Z, Liu S, Cai Y. EGFR/MAPK signaling regulates the proliferation of Drosophila renal and nephric stem cells. J. Genet. Genomics. 2015;42:9–20. doi: 10.1016/j.jgg.2014.11.007. [DOI] [PubMed] [Google Scholar]
- 19.Sarkar A, et al. Antagonistic roles of Rac and Rho in organizing the germ cell microenvironment. Curr. Biol. 2007;17:1253–1258. doi: 10.1016/j.cub.2007.06.048. [DOI] [PubMed] [Google Scholar]
- 20.Reddy BV, Irvine KD. Regulation of hippo signaling by EGFR-MAPK signaling through Ajuba family proteins. Dev. Cell. 2013;24:459–471. doi: 10.1016/j.devcel.2013.01.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tran J, Brenner TJ, DiNardo S. Somatic control over the germline stem cell lineage during Drosophila spermatogenesis. Nature. 2000;407:754–757. doi: 10.1038/35037613. [DOI] [PubMed] [Google Scholar]
- 22.Schulz C, Wood CG, Jones DL, Tazuke SI, Fuller MT. Signaling from germ cells mediated by the rhomboid homolog stet organizes encapsulation by somatic support cells. Development. 2002;129:4523–4534. doi: 10.1242/dev.129.19.4523. [DOI] [PubMed] [Google Scholar]
- 23.Hudson AG, Parrott BB, Qian Y, Schulz C. A temporal signature of epidermal growth factor signaling regulates the differentiation of germline cells in testes of Drosophila melanogaster. PLoS ONE. 2013;8:e70678. doi: 10.1371/journal.pone.0070678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Liu M, Lim TM, Cai Y. The Drosophila female germline stem cell lineage acts to spatially restrict DPP function within the niche. Sci. Signal. 2010;3:ra57. doi: 10.1126/scisignal.2000740. [DOI] [PubMed] [Google Scholar]
- 25.Ashton-Beaucage D, et al. A functional screen reveals an extensive layer of transcriptional and splicing control underlying RAS/MAPK signaling in Drosophila. PLoS. Biol. 2014;12:e1001809. doi: 10.1371/journal.pbio.1001809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Feng L, Shi Z, Chen X. Enhancer of polycomb coordinates multiple signaling pathways to promote both cyst and germline stem cell differentiation in the Drosophila adult testis. PLoS. Genet. 2017;13:e1006571. doi: 10.1371/journal.pgen.1006571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wu H, et al. Major spliceosome defects cause male infertility and are associated with nonobstructive azoospermia in humans. Proc. Natl Acad. Sci. USA. 2016;113:4134–4139. doi: 10.1073/pnas.1513682113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Westerveld GH, et al. Heterogeneous nuclear ribonucleoprotein G-T (HNRNP G-T) mutations in men with impaired spermatogenesis. Mol. Hum. Reprod. 2004;10:265–269. doi: 10.1093/molehr/gah042. [DOI] [PubMed] [Google Scholar]
- 29.Li J, et al. HnRNPL as a key factor in spermatogenesis: Lesson from functional proteomic studies of azoospermia patients with Sertoli cell only syndrome. J. Proteom. 2012;75:2879–2891. doi: 10.1016/j.jprot.2011.12.040. [DOI] [PubMed] [Google Scholar]
- 30.Dahan, O. & kupiec, M. The Saccharomyces cerevisiae gene CDC40/PRP17 controls cell cycle progression through splicing of the ANC1 gene. Nucleic Acids Res.32, 2529–2540 (2004). [DOI] [PMC free article] [PubMed]
- 31.Kerins JA, Hanazawa M, Dorsett M, Schedl T. PRP-17 and the pre-mRNA splicing pathway are preferentially required for the proliferation versus meiotic development decision and germline sex determination in Caenorhabditis elegans. Dev. Dyn. 2010;239:1555–1572. doi: 10.1002/dvdy.22274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ren L, et al. Systematic two-hybrid and comparative proteomic analyses reveal novel yeast pre-mRNA splicing factors connected to Prp19. PLoS ONE. 2011;6:e16719. doi: 10.1371/journal.pone.0016719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kaplan Y, Kupiec M. A role for the yeast cell cycle/splicing factor Cdc40 in the G1/S transition. Curr. Genet. 2006;51:123–140. doi: 10.1007/s00294-006-0113-y. [DOI] [PubMed] [Google Scholar]
- 34.Andersen DS, Tapon N. Drosophila MFAP1 is required for pre-mRNA processing and G2/M progression. J. Biol. Chem. 2008;283:31256–31267. doi: 10.1074/jbc.M803512200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yu J, et al. Protein synthesis and degradation are essential to regulate germline stem cell homeostasis in Drosophila testes. Development. 2016;143:2930–2945. doi: 10.1242/dev.134247. [DOI] [PubMed] [Google Scholar]
- 36.Leatherman JL, DiNardo S. Zfh-1 controls somatic stem cell self-renewal in the Drosophila testis and nonautonomously influences germline stem cell self-renewal. Cell Stem Cell. 2008;3:44–54. doi: 10.1016/j.stem.2008.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fabrizio JJ, Boyle M, DiNardo S. A somatic role for eyes absent (eya) and sine oculis (so) in Drosophila spermatocyte development. Dev. Biol. 2003;258:117–128. doi: 10.1016/S0012-1606(03)00127-1. [DOI] [PubMed] [Google Scholar]
- 38.Liang L, Diehl-Jones W, Lasko P. Localization of vasa protein to the Drosophila pole plasm is independent of its RNA-binding and helicase activities. Development. 1994;120:1201–1211. doi: 10.1242/dev.120.5.1201. [DOI] [PubMed] [Google Scholar]
- 39.Yu J, et al. Small ribonucleoprotein particle protein SmD3 governs the homeostasis of germline stem cells and the crosstalk between the spliceosome and ribosome signals in Drosophila. Faseb. J. 2019;33:8125–8137. doi: 10.1096/fj.201802536RR. [DOI] [PubMed] [Google Scholar]
- 40.Cheng H, Chen X, Zheng Y. The nuclear lamina regulates germline stem cell niche organization via modulation of EGFR signaling. cell Stem cell. 2013;13:73–86. doi: 10.1016/j.stem.2013.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Paul S, et al. Activation-induced substrate engagement in ERK signaling. Mol. Biol. Cell. 2020;31:235–243. doi: 10.1091/mbc.E19-07-0355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gupta S, Varshney B, Chatterjee S, Ray K. Somatic ERK activation during transit amplification is essential for maintaining the synchrony of germline divisions in Drosophila testis. Open Biol. 2018;8:180033. doi: 10.1098/rsob.180033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Roskoski RJ. ERK1/2 MAP kinases: structure, function, and regulation. Pharm. Res. 2012;66:105–143. doi: 10.1016/j.phrs.2012.04.005. [DOI] [PubMed] [Google Scholar]
- 44.Papagiannouli F, Berry CW, Fuller MT. The Dlg module and clathrin-mediated endocytosis regulate EGFR signaling and cyst cell-germline coordination in the Drosophila testis. Stem Cell Rep. 2019;12:1024–1040. doi: 10.1016/j.stemcr.2019.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Hall ET, Verheyen EM. Ras-activated Dsor1 promotes Wnt signaling in Drosophila development. J. Cell Sci. 2015;128:4499–4511. doi: 10.1242/jcs.175240. [DOI] [PubMed] [Google Scholar]
- 46.Bear JE, Haugh JM. Directed migration of mesenchymal cells: where signaling and the cytoskeleton meet. Curr. Opin. Cell Biol. 2014;30:74–82. doi: 10.1016/j.ceb.2014.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Fairchild MJ, Smendziuk CM, Tanentzapf G. A somatic permeability barrier around the germline is essential for Drosophila spermatogenesis. Development. 2014;142:268–281. doi: 10.1242/dev.114967. [DOI] [PubMed] [Google Scholar]
- 48.Filippi S, et al. Robustness of MEK-ERK dynamics and origins of cell-to-cell variability in MAPK signaling. Cell Rep. 2016;15:2524–2535. doi: 10.1016/j.celrep.2016.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Michailovici I, et al. Nuclear to cytoplasmic shuttling of ERK promotes differentiation of muscle stem/progenitor cells. Development. 2014;141:2611–2620. doi: 10.1242/dev.107078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Flores K, Yadav SS, Katz AA, Seger R. The nuclear translocation of mitogen-activated protein kinases: molecular mechanisms and use as novel therapeutic target. Neuroendocrinology. 2019;108:121–131. doi: 10.1159/000494085. [DOI] [PubMed] [Google Scholar]
- 51.Yu J, et al. Srlp is crucial for the self-renewal and differentiation of germline stem cells via RpL6 signals in Drosophila testes. Cell Death Dis. 2019;10:294. doi: 10.1038/s41419-019-1527-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.