Abstract
Aspergillus flavus is a common saprophytic filamentous fungus that produces the highly toxic natural compound aflatoxin during its growth process. Synthesis of the aflatoxins, which can contaminate food crops causing huge losses to the agricultural economy, is often regulated by epigenetic modification, such as the histone acetyltransferase. In this study, we used Aspergillus flavus as an experimental model to construct the acetyltransferase gene rtt109 knockout strain (△rtt109) and its complementary strain (△rtt109·com) by homologous recombination. The growth of △rtt109 was significantly suppressed compared to the wild type (WT) strain and the △rtt109·com strain. The sclerotium of △rtt109 grew smaller, and the amount of sclerotia generated by △rtt109 was significantly reduced. The number of conidiums of △rtt109 was significantly reduced, especially on the yeast extract sucrose (YES) solid medium. The amount of aflatoxins synthesized by △rtt109 in the PDB liquid medium was significantly decreased We also found that the △rtt109 strain was extremely sensitive to DNA damage stress. Through the maize seed infection experiment, we found that the growth of △rtt109 on the surface of affected corn was largely reduced, and the amount of aerial mycelium decreased significantly, which was consistent with the results on the artificial medium. We further found that H3K9 was the acetylated target of Rtt109 in A. flavus. In conclusion, Rtt109 participated in the growth, conidium formation, sclerotia generation, aflatoxin synthesis, environmental stress response, regulation of infection of A. flavus. The results from this study of rtt109 showed data for acetylation in the regulation of life processes and provided a new thought regarding the prevention and control of A. flavus hazards.
Supplementary Information
The online version contains supplementary material available at 10.1186/s43008-021-00060-4.
Keywords: Aspergillus flavus, Histone, Acetylation, rtt109, H3K9
Introduction
In eukaryotic organisms, chromatin is a dynamic and highly regulated structure that functions in compacting DNA into the nucleus and affects vital functions, such as gene expression, cell differentiation, and cell division. The fundamental repeat unit of chromatin is the nucleosome, which is composed of two copies of histone proteins H2A, H2B, H3, and H4, and wrapped twice around by about 147 base pairs of circumvoluted DNA. Histone modifications are understood to have the capacity of regulating transcription apart from genome expression (Kornberg & Lorch, 1999; Strahl & Allis, 2000). Some nuclear activities are strongly affected by different posttranslational modifications to the residues of histone proteins, including acetylation, methylation, phosphorylation, SUMOylation and ubiquitination (Hamon et al., 2007; Arbibe et al., 2007).
In the case of acetylation, it is a reversible process mediated by both histone acetyltransferases (HATs) and histone deacetylases (HDACs), by which a range of functions operate. HATs interacting with acetyl-coenzyme A, a co-factor that transfers an acetyl-group to a lysine residue, allows chromatin to transform into a flexible structure, and thereby activate transcription (Shahbazian & Grunstein, 2007). In contrast, HDACs deacetylate acetylated-lysine residues, compress DNA sequences, and lead to transcription repression (Kouzarides, 2000). Generally, HATs are grouped into 13 families according to their evolutionary relationships, where each HAT shares similarities with the others and are thus named from KAT1 to KAT13 (Torchia et al., 1998).
There is more evidence implicating that the acetylation on histone residues plays an important role for many cellular physiological processes in eukaryotes. For example, Gcn5, a KAT2-family HAT (Carrozza et al., 2003), was reported to participate in the regulation of nearly 12% of the genome in yeast (Lee et al., 2000). Deletion of KAT5-family HAT Esa1, led to lethality in A. nidulans (Soukup et al., 2012). In spite of the acetylation of histone and the functions of relative HATs have been studied extensively, there are still many functions that have not been fully characterized in fungi.
The regulator of Ty1 transposition 109 (Rtt109) was identified as a fungal-specific KAT11-family histone acetyltransferase, mainly responsible for the acetylation of histone 3 lysine 56 (H3K56) (Carrozza et al., 2003). This regulator was also reported to have a slight relationship with the acetylation of several residues on H3K9 and H3K27 (Ransom et al., 2010; Han et al., 2007a; Driscoll et al., 2007; Tsubota et al., 2007a; Berndsen et al., 2008; Schneider et al., 2006). The H3-H4 histone chaperone Asf1 primarily integrates to H3, and following that, H3K56 undergoes acetylation by Rtt109 (Tsubota et al., 2007b; Selth & Svejstrup, 2007a). A deletion mutant of Asf1 resulted in a large decrease to the level of acetylation on H3K56 (Han et al., 2007b). Another chaperone that has a relationship with Rtt109 is Vps75, a member of the Nucleosome Assembly Protein (NAP-1) family first found in Saccharomyces cerevisiae (Tang et al., 2008). In light of the evidence that Vps75 could combine with Rtt109 to form a Rtt109-Vps75 complex, influencing the level of acetylation on H3K56, Vps75 was found not essential for the acetylation of H3K56 (Selth et al., 2009; Selth & Svejstrup, 2007b; Park et al., 2008; da Rosa et al., 2013). The acetylation of H3K56 is vital for DNA damage repair, implying that the activation of DNA metabolic functions was facilitated by acetylation on H3K56 in Saccharomyces cerevisiae (Kadyk & Hartwell, 1992). Increasing evidence has demonstrated that cells would show a certain degree of inhibition under the pressure of DNA toxin agents without the acetylation on H3K56 in eukaryotic cells (Johnson & Jasin, 2000; Gonzalez-Barrera et al., 2003; Cortes-Ledesma & Aguilera, 2006; Gunjan et al., 2005).
Filamentous fungi are regarded as a class of eukaryote that can produce many kinds of bioactive compounds, known as secondary metabolites, which provide a selective advantage for themselves in the living environment and can be extremely harmful to other species (Yu & Keller, 2005). Many compounds are synthesized and released when fungi invade plants or animals, causing poisoning, diseases, and even death (Yu & Keller, 2005; Amaike & Keller, 2011; Amare & Keller, 2014). Aspergillus flavus is one kind of typical pathogenic filamentous fungus homologous to the well-established model organism A. nidulans (Zhang et al., 2015). It is also known as a common pathogen colonizing crops or food such as maize, peanuts, and rice (Gunjan et al., 2005; Yu & Keller, 2005; Amaike & Keller, 2011; Amare & Keller, 2014). A. flavus can produce aflatoxins resulting in billions of dollars of economic losses and serious food safety worldwide (Yu & Keller, 2005). Certain epigenetic factors could affect the growth, development and aflatoxin synthesis in A. flavus, such as methylation, acetylation, and phosphorylation. Many acetyltransferases including Rtt109 have been reported in fungal species, including S. cerevisiae, A. fumigatus, A. nidulans, and others (Amaike & Keller, 2011). However, the function of acetyltransferase Rtt109 has not been widely reported in A. flavus.
The purpose of this study is to clarify the effects of histone acetylation modification in growth and development, metabolism regulation, and infection of A. flavus. This can be used for which can be used for one of the targets for the control of aflatoxin in the future, and to lay a theoretical foundation for the prevention and control of aflatoxin. In this study, we studied the function of acetyltransferase Rtt109 in A. flavus. We found that Rtt109 in this fungus participated in growth, conidium formation, sclerotia generation, aflatoxin synthesis, environmental stress responses, regulation of infection, and other life processes. We further showed that many of those functions were influenced by the acetylation of H3K9, and revealed that H3K9 might be a candidate target for reducing both the fungal load and aflatoxin quantities in susceptible crops possibly through the application of HIGS (host-induced gene silencing).
Materials and methods
Sequence and phylogenetic tree analysis
To acquire an A. flavus Rtt109 protein sequence, a BLASTp search for protein homologs of the S. cerevisiae Rtt109 protein was conducted, and sequences of interest were downloaded from the National Center for Biotechnology Information resources (NCBI). These sequences were aligned with the ClustalW method, using MEGA 5.1 software, and a neighbor-joining phylogenetic tree was constructed according to the results. The visualized domains of Rtt109 or Rtt109-like proteins were constructed with DOG 2.0 software accordingly (Ren et al., 2009).
Mutant strains construction
The fungal strains used in this study are listed in Table S1 and were constructed using the primers listed in Table S2. All strains were constructed via homologous recombination and grown on five plates (Zhang et al., 2015). The strategies for strain construction are shown in Fig. S1. For constructing the rtt109 gene-deletion mutant (Δrtt109), an overlap polymerase chain reaction (PCR) method was used as previously described (Chang et al., 2010; Szewczyk et al., 2006), replacing the original rtt109 gene in A. flavus with the pyrG gene as a selection marker (Fig. S1A). For constructing the rtt109 complementary strain (△rtt109·com), the sequence, which consisted of rtt109 connected with pyrG and the overlaps, were used to replace the pyrG in Δrtt109 (Fig. S1B). For the gene-fluorescent fusion protein strain construction (rtt109-mCherry), the original sequence of rtt109 were mutated into the one lacking a termination codon (TAG), and then joined to the gene sequence of a fluorescent protein (mCherry) (Wong et al., 2008a) followed by the pyrG sequence (Fig. S1C). The point mutant strains were established by an overlap PCR method to construct a sequence, of which the pyrG marker was downstream of the gene coding histone 3. The histone 3 gene sequence was mutated by PCR amplification using different modified PCR primers, changing the lysines into alanine, arginine, and glutamine, and the H3K9R, H3K9A, and H3K9Q strains were constructed, respectively (Fig. S2).
Microscopic examination of the rtt109-mCherry subcellular localization
To assess the rtt109-mCherry localization, the newly formed fresh conidia and mycelia were observed using a Leica Confocal SP8 microscope system (Wong et al., 2008b). To observe the localization of the nuclei in mycelia, the samples were stained with 1 μg/mL 40, 6-diamidino-2-phenylindole (DAPI, Sigma, USA) for 30 min on ice. The samples were stained with 1 μg/mL 7-amino-4-chloroMethlycoumarin (CMAC, Sigma, USA) for 1 h at 37 °C, and then cultured in YGT liquid media at 37 °C for 2 h before observing.
Western blot analysis of histone modification
Similar procedures were conducted in our previous work (Lan et al., 2016). The strains were incubated at 28 °C for 72 h, and the mycelia of each sample were collected, then frozen in liquid nitrogen and ground into powder. Next, 100 mg powder was dissolved in 1 mL radio immuno precipitation assay lysis buffer (RIPA, Beyotime, Shanghai, China), which was used to extract the whole proteins. The concentrations of the protein extracts were measured using a Nanodrop detector and then diluted. We uploaded 40 μg of total protein and separated them by 15% sodium dodecyl sulfate polyacrylamide gel electrophoresis. The proteins were then transferred to a polyvinylidene fluoride membrane (Millipore, USA). We used the specific antibodies anti-actin (1:5000 dilution, Cell Signaling Technology), anti-Histone3 (1:5000 dilution, Abcam), anti-acetyl-histone3 (H3ac, 1:5000 dilution, Abcam), anti-acetyl-H3K56 (H3K56ac, 1:5000 dilution, Active Motif), and anti-acetyl-H3K9 (H3K56ac, 1:5000 dilution, PTM Biolabs) to detect histone modifications.
Analyses of conidia and Sclerotia
Conidia of each strain was removed from the periphery of a 2-day-old colony growing on yeast extract sucrose (YES) solid medium (Yang et al., 2016). The plugs were used to inoculate potato dextrose agar (PDA), YES, YS, and glucose minimal medium (GMM) (Shimizu & Keller, 2001). All strains were incubated at 37 °C without light, and the colony diameters were measured and recorded daily. After 5 days, conidia were collected in triplicate from 1 cm diameter zones of the agar media containing fungal growth. Samples were homogenized and diluted in 3 mL 0.05% Tween-20. Conidia were counted using a hemocytometer and microscope. To analyze the sclerotia, each strain was grown in Wickerham (WKM) agar medium at 37 °C in darkness (Raper & Thom, 1949). After 7 days, the surface of each plate was washed using 75% ethanol. Each strain was analyzed using five plates, and each experiment was repeated three times.
Colonization assay
A colonization assay on maize seed was completed using a modified procedure (Kale et al., 2008). The spore solution, WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A strains were diluted by water (approximately 10,000 spore/mL), and then the embryo-removed maize (Zea mays) seeds were added and cultured for 3 h at 28 °C with shaking (180 r/min). Maize seeds were harvested and transferred to a new plate with filter papers, and then cultured for 7 days. The filter papers were kept wet by adding 1 mL water every day.
Determination of Aflatoxin B1 production
The aflatoxin B1 (AFB1) production was measured by thin-layer chromatography (TLC) and high performance liquid chromatography (HPLC) analysis (Yang et al., 2016). Briefly, strains were inoculated in 50 mL of liquid medium, and cultured at 29 °C for 72 h. The cultures were combined with 50 mL chloroform in 250 mL flasks and shaken for 30 min. The grown mycelia were collected and completely dried and weighed. Next, the organic residue of each sample was collected into a new plate, completely dried and weighed, and then resuspended in chloroform solvent according to the weight of the dried mycelia (1 μL/mg of mycelia) (Lan et al., 2016). Approximately 10 μL of samples were loaded onto TLC plates (Haiyang Chemical, Qingdao, China) and dried. Then, they were separated using the developing solvent (chloroform:acetone = 9:1) and exposed under UV radiation (365 nm). The TLC plates were photographed using a Quantum ST5 imaging system (Vilber Lourmat Deutschland GmbH, Eberhardzell, Germany). For HPLC analysis, the sample of aflatoxin extracts were analyzed using a Mycotox TM column (Waters, Milford, MA, USA) at 42 °C (Lan et al., 2016). AFB1 and AFB2, the main products of A. flavus, were identified using a fluorescent detector (Waters) (Lan et al., 2016).
Stress assay
WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A strains were used to inoculate PDA medium supplemented with the following agents: cell wall stress agent, Congo red (CR, 500 μg/mL); hyperosmotic stress mediator, sodium chloride (NaCl, 1.5 mmol/mL); genotoxic agent, methyl methanesulfonate (MMS, 0.01%, v/v); and oxidative stress agent, hydrogen peroxide (H2O2, 10 mmol/mL). The inoculated plates were incubated at 37 °C in darkness for 5 days. Colony diameters were measured every day. Each strain was cultured on five plates, and each experiment was repeated three times.
Quantitative reverse transcription polymerase chain reaction
Mycelia were grown in YES liquid medium for 24 h, before being harvested and grounded into powder using liquid nitrogen. The total RNA was isolated from approximately 100 mg ground mycelia of each strain using an RNA isolation kit (Promega, USA). For qRT-PCR analysis, we used a Revert Aid First-strand cDNA Synthesis kit (Thermo Fisher Scientific, USA) to synthesize the cDNA from the RNA of each strain. We used the SYBR Green Supermix (Takara, Japan) and PikoReal 96 Real-time PCR system (Thermo Fisher Scientific, USA) to complete qRT-PCR analysis (denaturation at 95 °C for 30 S, renaturation and extension at 60 °C for 30 S, 40 cycles), and the relative quantities of each transcript were calculated using the 2−△△CT method (Livak & Schmittgen, 2001). The transcript levels of qRT-PCR were normalized relative to the transcript level of the β-tubulin housekeeping gene. The qRT-PCR primers are listed in Table S3. The data of qRT-PCR analysis were completed in triplicate, and each experiment was repeated three times. In this study, we analyzed sclerotia production related gene sclR, and the aflatoxin synthesis related genes aflO and aflR to evaluate the acetylation effects on sclerotia production and aflatoxin synthesis respectively.
Statistical analysis
Our data was presented as the means ± standard deviation of at least three biological replicate samples in figures. Statistical and significance analysis were performed using the data analysis software GraphPad Prism 5.1, and the p-values were lower than 0.05. As for multiple comparisons, we used Tukey’s multiple comparison for significance analysis.
Results
Identification of Rtt109 homologs protein in A. flavus
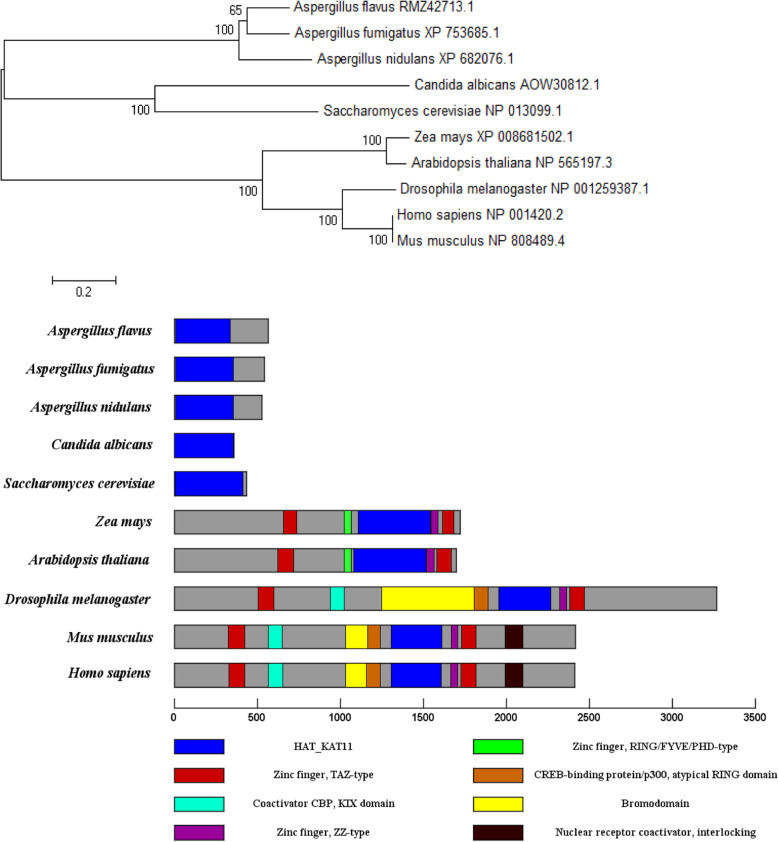
The DNA sequence of the putative DNA damage response protein Rtt109 in A. flavus was identified by searching the NCBI database. The A. flavus rtt109 consisted of 1695 bp with only one intron, encoding a putative histone acetylase with 564 amino acids. A BLAST search and phylogenetic analysis revealed that Rtt109 or its homologous proteins were also found in Saccharomyces cerevisiae, Aspergillus niger, Aspergillus flavus, Aspergillus fumigatus, Aspergillus nidulans, Zea mays, Arabidopsis thaliana, Dorsophilia melanogaster, Mus musculus, and Homo sapiens (Fig. 1a). Despite that the lengths of amino acid sequence above were different, they shared the same protein domain KAT_11 and are highly conserved (Fig. 1b). These data suggested that Rtt109 proteins are conserved from fungi to higher eukaryotes.
Fig. 1.
Our bioinformatics analysis of Rtt109 using the protein sequences of A. flavus (RMZ42713.1), A. nidulans (XP_682076.1), A. fumigatus (XP_753685.1), C. albicans (AOW30812.1), S. cerevisiae (NP_013099.1), Z. mays (XP_008681502.1), A. thaliana (NP_565197.3), D. melanogaster (NP_001259387.1), M. musculus (NP_808489.4), and H. sapiens (NP_001420.2) for (a) phylogenetic trees, and (b) protein domain analysis
Rtt109 is a typical B-type HAT predominantly localized in vacuoles or vesicles
There are two types of intracellular HATs, including A-type HATs for acetyltransferases localized and functioning in the nucleus, and B-type HATs for acetyltransferases predominantly existing in intracytoplasmic regions, which are transported into the nucleus to function. In order to investigate the subcellular localization of the Rtt109 protein, we used a previously described method (Lan et al., 2016) to construct an rtt109-mCherry strain constitutively expressing the mCherry tag at the C-terminal of the rtt109 sequence under the control of the native promoter in A. flavus. The rtt109-mCherry strain exhibited the same phenotype as the WT strains, suggesting that the function of rtt109 was not affected by the mCherry tag. We observed that the rtt109 protein was primarily co-localized with a CMAC stained vacuole or vesicle in fluorescence microscopy (Fig. 2a), but not with a DAPI stained nucleus at the germination stage (Fig. 2b). These data suggest that Rtt109 is predominantly stored in vacuoles or vesicles typical of other B-type HATs.
Fig. 2.
Subcellular localization of acetylase Rtt109 in A. flavus. The newborn mycelium of rtt109-mcherry strain was staining and observed under exciting light by using (a) 1 mg/mL 7-amino-4-chloroMethlycoumarin (CMAC) and (b) 1 mg/mL 6-diamidino-2-phenylindole (DAPI)
Rtt109 is a Bona fide HAT for acetylation of histone 3
In order to confirm our sequence alignment analysis (Fig. 1a), we sought to experimentally confirm that A. flavus rtt109 is a HAT targeting histone 3. As observed, the absence of rtt109 decreased the H3 acetylation levels, confirming that Rtt109 had HAT activity (Fig. 3). In addition, we examined the acetylation level of lysine residues in histone 3 and found that the acetylation level of Rtt109 targeting H3K9 and H3K56 was largely decreased in Δrtt109 compared to WT and △rtt109·com strains (Fig. 3). These results confirmed that the rtt109 was the functional HAT in A. flavus.
Fig. 3.

The effect of rtt109 on the acetylation of histone 3 in A. flavus. Western blot analysis showed the acetylation level targeting on H3K9 and H3K56 compared with actin, H3, and ac-H3. Wild type (WT)
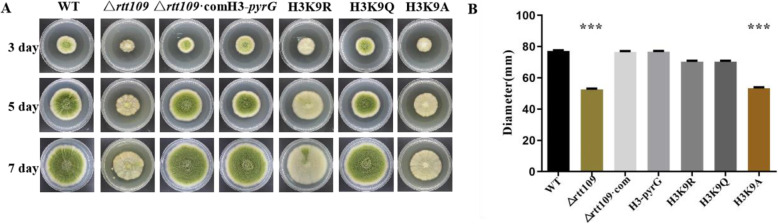
Rtt109 played an important role in A. flavus’s vegetative growth
According to the results of Fig. 4, after knocking out rtt109, the vegetative growth of A. flavus was significantly inhibited. Additionally, the growth rates of the Δrtt109 strains were considerably lower than that of the WT and Δrtt109·com strains on PDA and YES media, as well as in YS medium and GMM (Fig. S3B). Microscopic examinations indicated that the Δrtt109 strain generated less mycelia and more branches at the mycelial tips compared to the WT and Δrtt109·com strains on YES solid medium (Fig. S3C). These results revealed that rtt109 played an important role in the vegetative growth in A. flavus.
Fig. 4.
The effect of rtt109 and H3K9 acetylation on growth in A.flavus. a The growth of WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A on potato dextrose agar (PDA) solid medium. b Statistical analysis of WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A in diameter at day 7
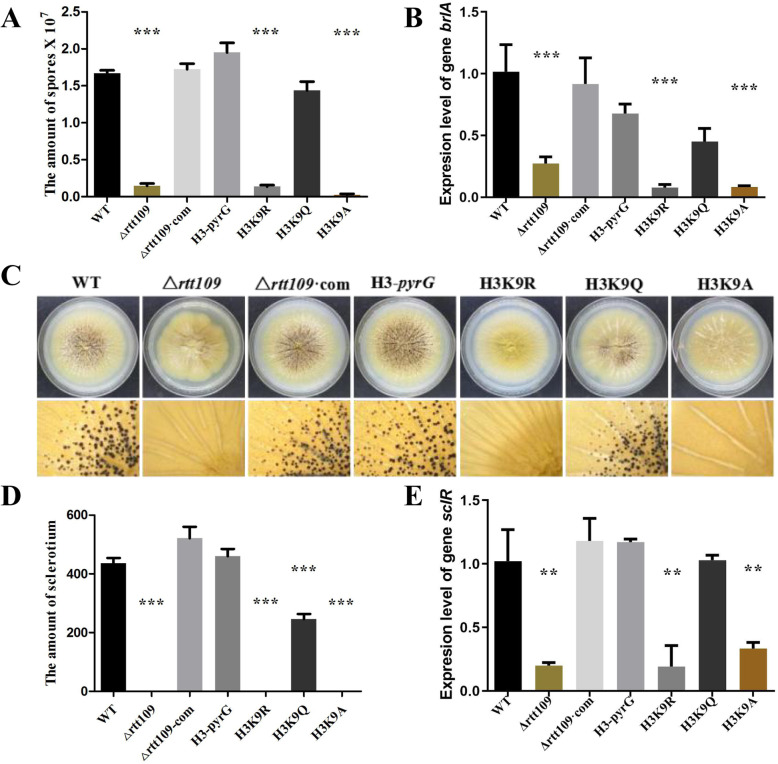
Rtt109 is essential for A. flavus’s conidial formation and Sclerotial generation
The conidiophores and conidial formation were analyzed to characterize the role of Δrtt109 during reproduction (Fig. 5, S4). Microscopic examinations revealed that the conidial heads of the Δrtt109 mutant were smaller than that of the WT and Δrtt109·com strains (Fig. S4A). The quantitative analysis of conidial formation confirmed that the Δrtt109 strains was almost unable to form conidia on YES, in contrast to the WT and Δrtt109·com strains (Fig. 5a). Further, the expression of conidial formation related genes, brlA, was down-regulated in the Δrtt109 strain cultured for 72 h (Fig. 5b). A. flavus generated sclerotia, the resting bodies that enable the fungus to survive in unsuitable environments. The WT and Δrtt109·com strain can normally produce sclerotia, while the Δrtt109 strain produced none in dark conditions, either on the WKM medium (Fig. S5A) or YS medium (Fig. 5c), and the statistical analysis agreed with this (Fig. 5d, S5B). Further, the expression levels of the genes, sclR, related to sclerotial production, were lower in Δrtt109 compared to the WT and Δrtt109·com strains (Fig. 5e). All these results showed that rtt109 played vital roles in the conidial formation and sclerotial generation in A. flavus.
Fig. 5.
The effect of rtt109 and H3K9 acetylation on conidia and sclerotia production. WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A strains were used for (a) the conidia production on PDA solid medium; (b) qRT-PCR analysis of the brlA gene; (c) sclerotia production analysis under dark conditions at 37 °C; (d) statistical analysis; (e) qRT-PCR analysis of the sclR gene. The asterisks *** represent a significant difference level of p < 0.001, and the asterisks ** represent a significant difference level of p < 0.01
Rtt109 played an essential role in pathogenicity towards maize
In order to measure the role that rtt109 played in affecting maize seed, the ability of all the strains to colonize maize seeds was evaluated. As Fig. 6 shows, the WT, △rtt109, and △rtt109·com strains were successfully colonized on the maize seed. Among these three strains, the Δrtt109 mutant strain was less able to colonize on maize seeds compared to the WT and Δrtt109·com strains. The aerial hyphae of Δrtt109 generated on the surface of the colonized maize seeds were much less than those of the WT and Δrtt109·com strains. A lack of conidiation was observed in the Δrtt109 mutant-infected maize seeds in contrast to that in the WT and Δrtt109·com strains. In all, the growth of the hyperacetylated strains, WT, △rtt109·com, H3-pyrG and H3K9Q, are better than the hypoacetylated strains, △rtt109, H3K9R and H3K9A. All these results are consisted with the strains grown on mediums and indicated that Rtt109 played an essential role in pathogenicity towards maize.
Fig. 6.
The effect of rtt109 and H3K9 acetylation on pathogenicity. The maize infection experiment was carried out in WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A strains of A. flavus after 7 days in the dark at 29 °C
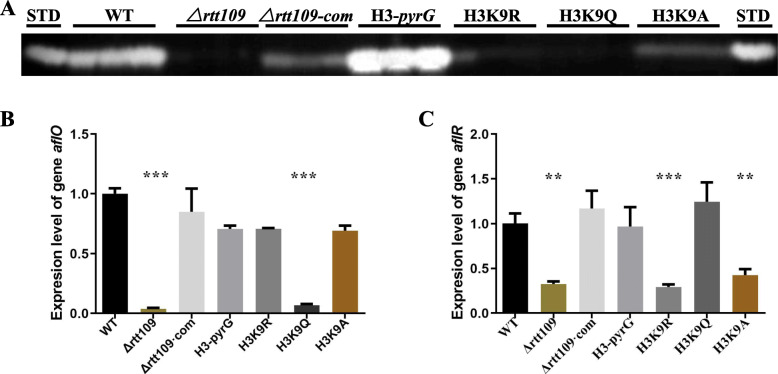
Aflatoxin synthesis in A. flavus conducted by rtt109
In order to investigate the role that rtt109 played in aflatoxin synthesis, we measured the most important and abundant secondary metabolites aflatoxins in A. flavus. The TLC results showed that the aflatoxin productions were largely reduced in Δrtt109 (Fig. 7a), indicating that aflatoxin synthesis was affected by rtt109. To further analyze the impact on aflatoxin synthesis in A. flavus knocking out rtt109, qRT-PCR experiments were conducted. In Fig. 7, the expressions of the aflatoxin-specific regulatory genes aflO and aflR were extremely low in the Δrtt109 strain compared to the WT and Δrtt109·com strains (Fig. 7b, c). All these results showed that rtt109 is important for aflatoxin production.
Fig. 7.
The effect of rtt109 and H3K9 acetylation on aflatoxin synthesis. a TLC assay of aflatoxins produced by the WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A strains grown on YES liquid medium at 29 °C for 3 days. The qRT-PCR analysis results of WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q, and H3K9A (b) on aflO, and (C)on aflR. The asterisks *** represent a significant difference level of p < 0.001, and the asterisks **represent a significant difference level of p < 0.01
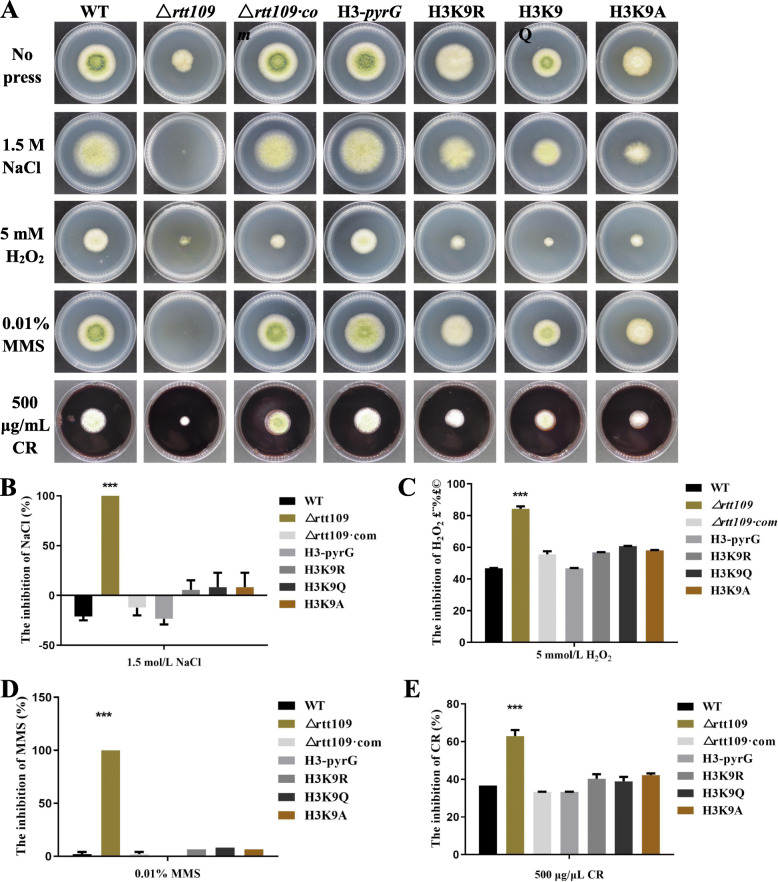
Rtt109 is critical for a stress response in A. flavus
We investigated the sensitivity of the WT, Δrtt109, and Δrtt109·com strains to 500 μg/μL CR, 5 mol/mL H2O2, 0.01% MMS and 1.2 mol/L NaCl, which represent cell wall stress, oxidative stresses, genotoxic stress and osmotic stress, respectively, and find that Δrtt109 strain is sensitive to NaCl, H2O2, MMS and CR. As we can observe in Fig. 8a, the Δrtt109 strain could not grow on the PDA medium containing 1.2 mol/L NaCl or 0.01% MMS, and statistical results supported these (Fig. 8b, d). Although Δrtt109 strain could grow on the PDA medium containing 500 μg/μL CR and 5 mol/mL H2O2 respectively, Δrtt109 strain was largely inhibited in contrast of WT and Δrtt109·com strains (Fig. 8c, e). These results indicated that Rtt109 played important roles in responsing to cell wall stress, oxidative stresses, osmotic stress and especially the genotoxic stress in A. flavus.
Fig. 8.
The effect of rtt109 and H3K9acetylation on stress response. a WT, △rtt109, △rtt109·com, H3-pyrG, H3K9R, H3K9Q and H3K9A strains were cultured on PDA medium containing 0.01% MMS, 5 mol/mL H2O2, 1.2 mol/L NaCl, and 500 μg/μL Congo red (CR) at 37 °C for 3 days. The inhibition of growth of the strains on PDA medium containing (B) 5 mol/mL H2O2, (C) 1.5 mol/L NaCl, (D) 0.01% MMS, (E) 500 μg/μL CR compared with the no press group, respectively. The asterisks *** represent a significant difference level of p < 0.001, and the asterisk * represents a significant difference level of p < 0.05
H3K9 as an important Acetylable target for rtt109 in A. flavus
In Figs. 4, 5, 6, 7, WT, △rtt109·com, H3-pyrG, and the simulative hyperacetylation of H3K9 strain H3K9Q showed similar trends on conidial formation, sclerotial generation, vegetative growth, and pathogenicity towards maize, while △rtt109, the simulative hypoacetylation of H3K9 strains H3K9R, and H3K9A was defective in these functions. These results indicated that the acetylation of H3K9 had a great influence on the functions above. However, there was no obvious differences in responding to stress between WT, Δrtt109·com, H3-pyrg, H3K9R, H3K9Q, and H3K9A, which indicated that the stress of A. flavus might not be influenced by acetylation of H3K9. All these results confirmed that H3K9 was the acetylated target by rtt109 in A. flavus, which played important roles in its full functions.
Discussion
According to the bioinformatics analysis, the Rtt109 homologous proteins did show an evolutionary trails in fungi, plants and animals, however, the fundamental function of Rtt109 is retained due to the conserved KAT_11 domain (Carrozza et al., 2003). In addition, the characteristic structural domain, KAT_11, is extremely conserved among Rtt109, which is orthologous in fungi, plants, and animals (Carrozza et al., 2003). The results in Fig. 1 might indicate that KAT_11 is an ancient domain and has evolved into different branches. Our results also showed that Rtt109 mainly localized to the vacuole or vesica of A. flavus. The previous study of Rtt109 showed that HATs are usually localized in the nucleus and cytoplasm in S. cerevisiae (Shahbazian & Grunstein, 2007; Kouzarides, 2000; Torchia et al., 1998; Carrozza et al., 2003; Lee et al., 2000), but sometimes in vacuole (Soukup et al., 2012). It has been reported that Rtt109 of S. cerevisiae will be transported out from the vacuole by Vps75, and then delivered to the nucleus in order to activate some functions of the histone. However, there are fundamental differences between S. cerevisiae and A. flavus in genomics and cell structure, thus the functions and the localization of the HATs Rtt109 are different. For example, in A. flavus, there are no similar sequences or protein homologous to Vps75 of S. cerevisiae. Therefore, further study on how Rtt109 is delivered from vacuole or vesica of A. flavus is necessary.
The previous study in S. pombe indicated that Rtt109 can acetylate the lysine residues H3K9, H3K27, and H3K56 of histone 3 (Selth & Svejstrup, 2007a; Han et al., 2007b; Tang et al., 2008; Selth et al., 2009), and we found that Rtt109 was a bona fide HAT for acetylation of histone 3 targeting both H3K9 and H3K56. We also found that the rtt109 functions for growth, asexual development, sclerotial formation, secondary metabolites synthesis, and invasion towards hosts of A. flavus were mediated by H3K9, which indicates that rtt109 is involved in many physiological functions of A. flavus due to its acetyltransferase activity. The modification of histones will lead to the expression or silencing of various genes, which is usually conducted by histone modifying enzymes.
When H3K9 was hypoacetylated, growth, sclerotium formation, and infection of A. flavus were significantly lower than that from the simulative hyperacetylated H3K9. Simulative hyperacetylation and hypoacetylation of H3K9 both influenced the synthesis of aflatoxin, but the regulation mechanism might be a dynamic process in A. flavus. Therefore, Rtt109 is a bona fide HAT for the acetylation of histone 3, and the acetylation modification on H3K9 played an important role in the regulation of the sclerotium formation, secondary metabolism, growth, and other physiological process in A. flavus. However, we also found that the hyperacetylation and hypoacetylation were very complex. For example, the expression level of aflO in the putative hypoacetylated strains, H3K9R and H3K9A, were consistent with the WT and Δrtt109·com, but Δrtt109 and H3K9Q were almost the same. Furthermore, there was no difference in the stress responses between the WT, Δrtt109·com, H3K9R, H3K9Q and H3K9A, showing that the hyperacetylation and hypoacetylation were a complex dynamics process. Therefore, further research on the mechanism of acetylation of H3K9 affecting gene expression is needed.
The importance of Rtt109 for DNA damage repair has been broadly studied in fungal species. Usually, the Rtt109 protein in fungal species is known as a kind of acetyltransferase that can modify H3K56, which is well connected with the expression of rad52 (Alvaro et al., 2007; Malone et al., 1988), the DNA damage repair function gene in many eukaryote species (Tsubota et al., 2007b; Selth & Svejstrup, 2007a; Han et al., 2007b). However, the importance of rtt109 for morphogenesis has not yet been well studied in filamentous fungal species. After knocking out the gene rtt109, the colony of A. flavus was significantly smaller than that of the WT, and branching at the mycelial tips increased significantly. The agent MMS can cause DNA damage of the genome in A. flavus, while acetyltransferase Rtt109 can activate the DNA damage repair function of A. flavus, thus, the deletion of rtt109 may lead to the loss of DNA damage repair function.
By microscopic observation, we found that the conidial heads of △rtt109 were much smaller than those of the WT and △rtt109·com, which indicated that development was regulated by gene rtt109. As for conidial formation, the amounts of spores produced by △rtt109 on PDA, YES, YS, and GMM mediums were greatly decreased, in particular, there was no spore generated on the YES medium. The sclerotia production of △rtt109 on the WKM medium was greatly decreased, in particularly, there was no sclerotia generated on the YS medium. These results indicated that knocking out rtt109 leads to a defect of reproduction ability in A. flavus.
In colonization experiments, we found that △rtt109 can hardly colonize on the surface of maize, which is consistent with the culture experiments, implying that rtt109 also played a role in invasion towards the crops. The synthesis of aflatoxin is a complex process in A. flavus, which is regulated by a cluster of genes. There were 30 genes reported to participate in aflatoxin synthesis, and the total length of this cluster is 70 kb (Yu et al., 2004; Wild & Turner, 2002; Brown et al., 1996; Chang, 2003). The activation of gene expression is connected to histone modification. According to our results, the △rtt109 strain showed an inhibition in aflatoxin synthesis, indicating that rtt109 played an important role in aflatoxin synthesis.
Conclusion
In this study, our results indicated that rtt109 was involved in regulating many physiological processes in A. flavus. After knocking out rtt109, the growth rate, conidium generation, generation of sclerotia, and aflatoxin synthesis of A. flavus were largely reduced, indicating that rtt109 is crucial in A. flavus. Rtt109 is a bona fide HAT for the acetylation of histone 3, and the acetylation modification on H3K9 played important roles in the regulation of many physiological processes in A. flavus.
Supplementary Information
Additional file 1: Table S1. The strains used in this experiment. Table S2. PCR primers used in this study. Table S3. qRT-PCR primers used in this study. Fig. S1. Strategy of construction for △rtt109 and △rtt109·com strains. Diagrammatic representation of the gene replacement strategy for construction of (A) △rtt109, (B) △rtt109·com, and (C) rtt109-mCherry. Sequences A and B referred to the homologous sequences near the target A. flavus rtt109. Fig. S2. Strategy of construction for point-mutant strains. (A) Diagrammatic representation of the gene replacement strategy for construction of H3 point-mutant strains. (B) Sequence alignment results of H3K9A, H3K9Q, H3K9R, H3-pyrG, and WT. Fig. S3. The effect of rtt109 on growth of A. flavus. (A) The growth of WT, △rtt109 and △rtt109·com on YES solid medium, YS solid medium and GMM solid medium. (B) Growth rate of WT, △rtt109 and △rtt109·com as in (A). (C) Mycelial branching observation of WT, △rtt109 and △rtt109·com strains grown on YES solid medium. Fig. S4. The effect of rtt109 on conidia production in A. flavus. (A) The conidophore of WT, △rtt109 and △rtt109·com on YES solid medium, YS solid medium and GMM solid medium. (B) The statistical analysis of conidia production on YES solid medium, YS solid medium and GMM solid medium as in A. The asterisks *** represents a significant different level of p < 0.001. Fig. S5. The effect of rtt109 on sclerotia production in A. flavus. (A) Sclerotia production of the strains cultured on WKM solid medium under light and dark at 37 °C for 10 days. (B) Statistical analysis of sclerotia production in WT, △rtt109 and △rtt109·com grown on WKM solid medium. The asterisks *** represents a significant different level of p < 0.001. Fig. S6. The effect of rtt109 on aflatoxin production in A. flavus. (A) TLC assay of aflatoxin produced by A. flavus WT, △rtt109 and △rtt109·com strains grown on PDB liquid medium at 29 °C from 3 to 6 days. (B) HPLC assay of aflatoxin produced by the WT, △rtt109 and △rtt109·com strains grown on YES liquid medium at 29 °C. Fig. S7. The effect of strains WT, △rtt109, △rtt109·com, H3K56R, H3K56Q, and H3K56A on (A) growth on PDA medium, (B) conidophore on GMM medium, (C) sclerotia production.
Acknowledgements
We especially thank Perng Kuang Chang (Southern Regional Research Center, United States Department of Agriculture, New Orleans, USA), Zhumei He (Sun Yat-senUniversity, Guangzhou, China), Yang Liu (Institute of Food Science and Technology CAAS), and Kong Qing for their kindness to provide the strains. The whole sequence of mCherry was from Bowen Li (Fujian Institute of Research on Structure of Matter, Chinese Academy of Sciences). We thank Yanyun Li (FAFU, Life Science College) for her help in taking the confocal images.
Adherence to national and international regulations
Not applicable.
Authors’ contributions
Conceptualization, R.S. and H.L.; Data curation, R.S., M.W., H.L. and L.W.; Formal analysis, R.S. L.W. and M.W.; Funding acquisition, S.W.; Investigation, R.S. L.W. and H.L.; Methodology, R.S. and H.L.; Project administration S.W. and J.Y.; Resources, S.W. and J.Y.; Software, R.S. and H.L.; Supervision, S.W. and J.Y.; Validation, R.S., M.W., H.L. and L.W.; Visualization, R.S. and M.W.; Roles/Writing - original draft, R.S and H.L.; Writing - review & editing, S.W. and J.Y. The author (s) read and approved the final manuscript.
Funding
This research was funded by the National Natural Science Foundation of China (No. 31772105).
Availability of data and materials
Not applicable.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no conflict of interest.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Ruilin Sun, Meifang Wen and Lianghuan Wu contributed equally to this work.
References
- Alvaro D, Lisby M, Rothstein R. Genome-wide analysis of Rad52 foci reveals diverse mechanisms impacting recombination. PLoS Genet. 2007;3(12):e228. doi: 10.1371/journal.pgen.0030228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amaike S, Keller NP. Aspergillus flflavus. Annu Rev Phytopathol. 2011;49(1):107–133. doi: 10.1146/annurev-phyto-072910-095221. [DOI] [PubMed] [Google Scholar]
- Amare MG, Keller NP. Molecular mechanisms of Aspergillus flflavus secondary metabolism and development. Fungal Genet Biol. 2014;66:11–18. doi: 10.1016/j.fgb.2014.02.008. [DOI] [PubMed] [Google Scholar]
- Arbibe L, Kim DW, Batsche E, Pedron T, Mateescu B, Muchardt C, Parsot C, Sansonetti PJ. An injected bacterial effector targets chromatin access for transcription factor NF-kB to alter transcription of host genes involved in immune responses. Na Immunol. 2007;8(1):47–56. doi: 10.1038/ni1423. [DOI] [PubMed] [Google Scholar]
- Berndsen CE, Tsubota T, Lindner SE, Lee S, Holton JM, Kaufman PD, Keck JL, Denu JM. Molecular functions of the histone acetyltransferase chaperone complex Rtt109–Vps75. Nat Struct Mol Biol. 2008;15(9):948–956. doi: 10.1038/nsmb.1459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown DW, Yu JH, Kelkar HS, Fernandes M, Nesbitt TC, Keller NP, Adams TH, Leonard TJ. Twenty-five coregulated transcripts define a sterigmatocystin gene cluster in Aspergillus nidulans[J] Proc Natil Acad Sci. 1996;93(4):1418–1422. doi: 10.1073/pnas.93.4.1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carrozza MJ, Utley RT, Workman JL, Côté J. The diverse functions of histone acetyltransferase complexes[J] Trends Genet. 2003;19(6):321–329. doi: 10.1016/S0168-9525(03)00115-X. [DOI] [PubMed] [Google Scholar]
- Chang PK (2003) The Aspergillus parasiticus protein AFLJ interacts with the aflatoxin pathway-specific regulator AFLR[J]. Mol Genet Genomics 268(6):711–719. 10.1007/s00438-003-0809-3 [DOI] [PubMed]
- Chang PK, Scharfenstein LL, Wei Q, Bhatnagar D. Development and refifinement of a high-effiffifficiency gene-targeting system for Aspergillus flflavus. J Microbiol Methods. 2010;81(3):240–246. doi: 10.1016/j.mimet.2010.03.010. [DOI] [PubMed] [Google Scholar]
- Cortes-Ledesma F, Aguilera A. Double-strand breaks arising by replication through a nick are repaired by cohesin-dependent sister-chromatid exchange. EMBO Rep. 2006;7(9):919–926. doi: 10.1038/sj.embor.7400774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Rosa JL, Bajaj V, Spoonamore J, et al. A small molecule inhibitor of fungal histone acetyltransferase Rtt109[J] Bioorg Med Chem Lett. 2013;23(10):2853–2859. doi: 10.1016/j.bmcl.2013.03.112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Driscoll R, Hudson A, Jackson SP. Yeast Rtt109 Promotes Genome Stability by Acetylating Histone H3 on Lysine 56. Science. 2007;315(5812):649–652. doi: 10.1126/science.1135862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Barrera S, Cortes-Ledesma F, Wellinger RE, Aguilera A. Equal sister chromatid exchange is a major mechanism of double-strand break repair in yeast. Mol Cell. 2003;11(6):1661–1671. doi: 10.1016/S1097-2765(03)00183-7. [DOI] [PubMed] [Google Scholar]
- Gunjan A, Paik J, Verreault A. Regulation of histone synthesis and nucleosome assembly. Biochimie. 2005;87(7):625–635. doi: 10.1016/j.biochi.2005.02.008. [DOI] [PubMed] [Google Scholar]
- Hamon MA, Batsche E, Regnault B, Tham TN, Seveau S, Muchardt C, Cossart P. Histone modifications induced by a family of bacterial toxins. Proc. Natl Acad Sci. U S A. 2007;104(33):13467–13472. doi: 10.1073/pnas.0702729104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Han J, Zhou H, Horazdovsky B, Zhang K, Xu RM, Zhang Z. Rtt109 Acetylates Histone H3 Lysine 56 and Functions in DNA Replication. Science. 2007;315(5812):653–655. doi: 10.1126/science.1133234. [DOI] [PubMed] [Google Scholar]
- Han J, Zhou H, Li Z, Xu RM, Zhang Z. The Rtt109-Vps75 histone acetyltransferase complex acetylates non-nucleosomal histone H3[J] J Biol Chem. 2007;282(19):14158–14164. doi: 10.1074/jbc.M700611200. [DOI] [PubMed] [Google Scholar]
- Johnson RD, Jasin M. Sister chromatid gene conversion is a prominent double-strand break repair pathway in mammalian cells. EMBO J. 2000;19(13):3398–3407. doi: 10.1093/emboj/19.13.3398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kadyk LC, Hartwell LH. Sister chromatids are preferred over homologs as substrates for recombinational repair in Saccharomyces cerevisiae. Genetics. 1992;132(2):387–402. doi: 10.1093/genetics/132.2.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kale SP, Milde L, Trapp MK, Frisvad JC, Keller NP, Bok JW. Requirement of LaeA for secondary metabolism and sclerotial production in Aspergillus flflavus. Fungal Genet Biol. 2008;45(10):1422–1429. doi: 10.1016/j.fgb.2008.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornberg RD, Lorch Y. Twenty-five years of the nucleosome, fundamental particle of the eukaryote chromosome. Cell. 1999;98(3):285–294. doi: 10.1016/S0092-8674(00)81958-3. [DOI] [PubMed] [Google Scholar]
- Kouzarides T. Acetylation: a regulatory modification to rival phosphorylation? EMBO J. 2000;19(6):1176–1179. doi: 10.1093/emboj/19.6.1176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lan H, Sun R, Fan K, et al. The Aspergillus flavus histone acetyltransferase AflGcnE regulates morphogenesis, aflatoxin biosynthesis, and pathogenicity[J] Front Microbiol. 2016;7:1324. doi: 10.3389/fmicb.2016.01324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee TI, Causton HC, Holstege FC, Shen W-C, Hannett N, Jennings EG, et al. Redundant roles for the TFIID and SAGA complexes in global transcription. Nature. 2000;405(6787):701–704. doi: 10.1038/35015104. [DOI] [PubMed] [Google Scholar]
- Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods. 2001;25(4):402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- Malone RE, Montelone BA, Edwards C, Carney K, Hoekstra MF. A reexamination of the role of the RAD52 gene in spontaneous mitotic recombination. Curr Genet. 1988;14(3):211–223. doi: 10.1007/BF00376741. [DOI] [PubMed] [Google Scholar]
- Park YJ, Sudhoff KB, Andrews AJ, Stargell LA, Luger K. Histone chaperone specificity in Rtt109 activation. Nat Struct Mol Biol. 2008;15(9):957–964. doi: 10.1038/nsmb.1480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ransom M, Dennehey BK, Tyler JK. Chaperoning Histones during DNA Replication and Repair. Cell. 2010;140(2):183–195. doi: 10.1016/j.cell.2010.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raper KB, Thom C. A manual of the Penicillia. Baltimore, MD: Williams & Wilkins; 1949. [Google Scholar]
- Ren J, Wen L, Gao X, Jin C, Xue Y, Yao X. DOG 1.0: illustrator of protein domain structures. Cell Res. 2009;19(2):271–273. doi: 10.1038/cr.2009.6. [DOI] [PubMed] [Google Scholar]
- Schneider J, Bajwa P, Johnson FC, Bhaumik SR, Shilatifard A. Rtt109 Is Required for Proper H3K56 Acetylation. J Biol Chem. 2006;281(49):37270–37274. doi: 10.1074/jbc.C600265200. [DOI] [PubMed] [Google Scholar]
- Selth L, Svejstrup JQ. Vps75, a new yeast member of the NAP histone chaperone family. J Biol Chem. 2007;282(17):12358–12362. doi: 10.1074/jbc.C700012200. [DOI] [PubMed] [Google Scholar]
- Selth L, Svejstrup JQ. Vps75, a new yeast member of the NAP histone chaperone family. J Biol Chem. 2007;282(17):12358–12362. doi: 10.1074/jbc.C700012200. [DOI] [PubMed] [Google Scholar]
- Selth LA, Lorch Y, Ocampo-Hafalla MT, Mitter R, Shales M, Krogan NJ, Kornberg RD, Svejstrup JQ. An rtt109-independent role for vps75 in transcription-associated nucleosome dynamics. Mol Cell Biol. 2009;29(15):4220–4234. doi: 10.1128/MCB.01882-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shahbazian MD, Grunstein M. Functions of site-specific histone acetylation and deacetylation. Annu Rev Biochem. 2007;76(1):75–100. doi: 10.1146/annurev.biochem.76.052705.162114. [DOI] [PubMed] [Google Scholar]
- Shimizu K, Keller NP. Genetic involvement of a cAMP-dependent protein kinase in a G protein signaling pathway regulating morphological and chemical transitions in Aspergillus nidulans. Genetics. 2001;157(2):591–600. doi: 10.1093/genetics/157.2.591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soukup AA, Chiang YM, Bok JW, Reyes-Dominguez Y, Oakley BR, Wang CCC, Strauss J, Keller NP. Overexpression of the a spergillus nidulans histone 4 acetyltransferase EsaA increases activation of secondary metabolite production[J] Mol Microbiol. 2012;86(2):314–330. doi: 10.1111/j.1365-2958.2012.08195.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strahl BD, Allis CD. The language of covalent histone modifications. Nature. 2000;403(6765):41–45. doi: 10.1038/47412. [DOI] [PubMed] [Google Scholar]
- Szewczyk E, Nayak T, Oakley CE, Edgerton H, Xiong Y, Taheri-Talesh N, Osmani SA, Oakley BR. Fusion PCR and gene targeting in Aspergillus nidulans. Nat Protoc. 2006;1(6):3111–3120. doi: 10.1038/nprot.2006.405. [DOI] [PubMed] [Google Scholar]
- Tang Y, Meeth K, Jiang E, Luo C, Marmorstein R. Structure of Vps75 and implications for histone chaperone function. Proc Natl Acad Sci U S A. 2008;105(34):12206–12211. doi: 10.1073/pnas.0802393105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torchia J, Glass C, Rosenfeld MG. Co-activators and co-repressors in the integration of transcriptional responses. Curr Opin Cell Biol. 1998;10(3):373–383. doi: 10.1016/S0955-0674(98)80014-8. [DOI] [PubMed] [Google Scholar]
- Tsubota T, Berndsen CE, Erkmann JA, Smith CL, Yang L, Freitas MA, Denu JM, Kaufman PD. Histone H3-K56 Acetylation Is Catalyzed by Histone Chaperone-Dependent Complexes. Mol Cell. 2007;25(5):703–712. doi: 10.1016/j.molcel.2007.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsubota T, Berndsen CE, Erkmann JA, Smith CL, Yang L, Freitas MA, Denu JM, Kaufman PD. Histone H3-K56 acetylation is catalyzed by histone chaperone-dependent com plexes. Mol Cell. 2007;25(5):703–712. doi: 10.1016/j.molcel.2007.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wild CP, Turner PC. The toxicology of aflatoxins as a basis for public health decisions[J] Mutagenesis. 2002;17(6):471–481. doi: 10.1093/mutage/17.6.471. [DOI] [PubMed] [Google Scholar]
- Wong KH, Todd RB, Oakley BR, Oakley CE, Hynes MJ, Davis MA. Sumoylation in Aspergillus nidulans: sumO inactivation,overexpression and live-cell imaging. Fungal Genet Bio. 2008;45(5):728–737. doi: 10.1016/j.fgb.2007.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong KH, Todd RB, Oakley BR, Oakley CE, Hynes MJ, Davis MA. Sumoylation in Aspergillus nidulans: sumO inactivation, overexpression and live-cell imaging. Fungal Genet Biol. 2008;45(5):728–737. doi: 10.1016/j.fgb.2007.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang K, Liang L, Ran F, Liu Y, Li Z, Lan H, Gao P, Zhuang Z, Zhang F, Nie X, Kalayu Yirga S, Wang S. The DmtA methyltransferase contributes to Aspergillus flflavus conidiation, sclerotial production, aflflatoxin biosynthesis and virulence. Sci Rep. 2016;6(1):23259. doi: 10.1038/srep23259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu J, Chang PK, Ehrlich KC, Cary JW, Bhatnagar D, Cleveland TE, Payne GA, Linz JE, Woloshuk CP, Bennett JW. Clustered pathway genes in aflatoxin biosynthesis[J] Appl Environ Microbiol. 2004;70(3):1253–1262. doi: 10.1128/AEM.70.3.1253-1262.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu JH, Keller N. Regulation of secondary metabolism in filamentous fungi[J] Annu Rev Phytopathol. 2005;43(1):437–458. doi: 10.1146/annurev.phyto.43.040204.140214. [DOI] [PubMed] [Google Scholar]
- Zhang F, Zhong H, Han X, Guo Z, Yang W, Liu Y, Yang K, Zhuang Z, Wang S. Proteomic profifile of Aspergillus flflavus in response to water activity. Fungal Biol. 2015;119(2-3):114–124. doi: 10.1016/j.funbio.2014.11.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Additional file 1: Table S1. The strains used in this experiment. Table S2. PCR primers used in this study. Table S3. qRT-PCR primers used in this study. Fig. S1. Strategy of construction for △rtt109 and △rtt109·com strains. Diagrammatic representation of the gene replacement strategy for construction of (A) △rtt109, (B) △rtt109·com, and (C) rtt109-mCherry. Sequences A and B referred to the homologous sequences near the target A. flavus rtt109. Fig. S2. Strategy of construction for point-mutant strains. (A) Diagrammatic representation of the gene replacement strategy for construction of H3 point-mutant strains. (B) Sequence alignment results of H3K9A, H3K9Q, H3K9R, H3-pyrG, and WT. Fig. S3. The effect of rtt109 on growth of A. flavus. (A) The growth of WT, △rtt109 and △rtt109·com on YES solid medium, YS solid medium and GMM solid medium. (B) Growth rate of WT, △rtt109 and △rtt109·com as in (A). (C) Mycelial branching observation of WT, △rtt109 and △rtt109·com strains grown on YES solid medium. Fig. S4. The effect of rtt109 on conidia production in A. flavus. (A) The conidophore of WT, △rtt109 and △rtt109·com on YES solid medium, YS solid medium and GMM solid medium. (B) The statistical analysis of conidia production on YES solid medium, YS solid medium and GMM solid medium as in A. The asterisks *** represents a significant different level of p < 0.001. Fig. S5. The effect of rtt109 on sclerotia production in A. flavus. (A) Sclerotia production of the strains cultured on WKM solid medium under light and dark at 37 °C for 10 days. (B) Statistical analysis of sclerotia production in WT, △rtt109 and △rtt109·com grown on WKM solid medium. The asterisks *** represents a significant different level of p < 0.001. Fig. S6. The effect of rtt109 on aflatoxin production in A. flavus. (A) TLC assay of aflatoxin produced by A. flavus WT, △rtt109 and △rtt109·com strains grown on PDB liquid medium at 29 °C from 3 to 6 days. (B) HPLC assay of aflatoxin produced by the WT, △rtt109 and △rtt109·com strains grown on YES liquid medium at 29 °C. Fig. S7. The effect of strains WT, △rtt109, △rtt109·com, H3K56R, H3K56Q, and H3K56A on (A) growth on PDA medium, (B) conidophore on GMM medium, (C) sclerotia production.
Data Availability Statement
Not applicable.